Modulation of the Blood–Brain Barrier for Drug Delivery to Brain
Abstract
1. Introduction
2. Modulation of Tight Junctions
2.1. Osmotic BBB Disruption
2.2. Radiation-Mediated BBB Disruption
2.3. Activating Bradykinin B2 Receptor
2.4. Direct Interference of Tight Junctions
2.5. Other Potential Strategies
3. Modulation of Active Efflux
3.1. Direct Inhibition of Efflux Transporters
3.2. Targeting Regulatory Pathways of Efflux Transporters
4. Modulation of Transcytosis
4.1. Upregulation of LRP1
4.2. Inhibition of Mfsd2a
4.3. Upregulation of GLUT1
5. Multifunctional Strategies by Multiple BBB Modulation
5.1. Focused Ultrasound
5.2. Activating A2A Adenosine Receptor
5.3. Activating Potassium Channels
5.4. Other Potential Multifunctional Strategies
6. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Andreone, B.J.; Chow, B.W.; Tata, A.; Lacoste, B.; Ben-Zvi, A.; Bullock, K.; Deik, A.A.; Ginty, D.D.; Clish, C.B.; Gu, C. Blood-brain barrier permeability is regulated by lipid transport-dependent suppression of caveolae-mediated transcytosis. Neuron 2017, 94, 581–594.e585. [Google Scholar] [CrossRef]
- Wang, J.Z.; Xiao, N.; Zhang, Y.Z.; Zhao, C.X.; Guo, X.H.; Lu, L.M. Mfsd2a-based pharmacological strategies for drug delivery across the blood-brain barrier. Pharmacol. Res. 2016, 104, 124–131. [Google Scholar] [CrossRef]
- Knowland, D.; Arac, A.; Sekiguchi, K.J.; Hsu, M.; Lutz, S.E.; Perrino, J.; Steinberg, G.K.; Barres, B.A.; Nimmerjahn, A.; Agalliu, D. Stepwise recruitment of transcellular and paracellular pathways underlies blood-brain barrier breakdown in stroke. Neuron 2014, 82, 603–617. [Google Scholar] [CrossRef]
- Ben-Zvi, A.; Lacoste, B.; Kur, E.; Andreone, B.J.; Mayshar, Y.; Yan, H.; Gu, C. Mfsd2a is critical for the formation and function of the blood-brain barrier. Nature 2014, 509, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Armulik, A.; Genove, G.; Mae, M.; Nisancioglu, M.H.; Wallgard, E.; Niaudet, C.; He, L.; Norlin, J.; Lindblom, P.; Strittmatter, K.; et al. Pericytes regulate the blood-brain barrier. Nature 2010, 468, 557–561. [Google Scholar] [CrossRef]
- Daneman, R.; Zhou, L.; Kebede, A.A.; Barres, B.A. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature 2010, 468, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Arvanitis, C.D.; Ferraro, G.B.; Jain, R.K. The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 2020, 20, 26–41. [Google Scholar] [CrossRef]
- Han, L.; Jiang, C. Evolution of blood-brain barrier in brain diseases and related systemic nanoscale brain-targeting drug delivery strategies. Acta Pharm. Sin. B 2021, 11, 2306–2325. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Ronnback, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef]
- Gao, X.; Yue, Q.; Liu, Y.; Fan, D.; Fan, K.; Li, S.; Qian, J.; Han, L.; Fang, F.; Xu, F.; et al. Image-guided chemotherapy with specifically tuned blood brain barrier permeability in glioma margins. Theranostics 2018, 8, 3126–3137. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; He, Y.; Zhang, J.; Li, J.; Yu, X.; Cao, Z.; Meng, F.; Zhao, Y.; Wu, X.; Shen, T.; et al. Functionalized nanocarrier combined seizure-specific vector with p-glycoprotein modulation property for antiepileptic drug delivery. Biomaterials 2016, 74, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Soffietti, R.; Ahluwalia, M.; Lin, N.; Ruda, R. Management of brain metastases according to molecular subtypes. Nat. Rev. Neurol. 2020, 16, 557–574. [Google Scholar] [CrossRef]
- Luo, H.; Shusta, E.V. Blood-brain barrier modulation to improve glioma drug delivery. Pharmaceutics 2020, 12, 1085. [Google Scholar] [CrossRef]
- Lochhead, J.J.; Yang, J.; Ronaldson, P.T.; Davis, T.P. Structure, function, and regulation of the blood-brain barrier tight junction in central nervous system disorders. Front. Physiol. 2020, 11, 914. [Google Scholar] [CrossRef] [PubMed]
- Pandit, R.; Chen, L.; Gotz, J. The blood-brain barrier: Physiology and strategies for drug delivery. Adv. Drug Deliv. Rev. 2020, 165–166, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.D.; Winkler, E.A.; Sagare, A.P.; Singh, I.; LaRue, B.; Deane, R.; Zlokovic, B.V. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 2010, 68, 409–427. [Google Scholar] [CrossRef]
- Yanagida, K.; Liu, C.H.; Faraco, G.; Galvani, S.; Smith, H.K.; Burg, N.; Anrather, J.; Sanchez, T.; Iadecola, C.; Hla, T. Size-selective opening of the blood-brain barrier by targeting endothelial sphingosine 1-phosphate receptor 1. Proc. Natl. Acad. Sci. USA 2017, 114, 4531–4536. [Google Scholar] [CrossRef] [PubMed]
- Kodack, D.P.; Askoxylakis, V.; Ferraro, G.B.; Fukumura, D.; Jain, R.K. Emerging strategies for treating brain metastases from breast cancer. Cancer Cell 2015, 27, 163–175. [Google Scholar] [CrossRef]
- Haluska, M.; Anthony, M.L. Osmotic blood-brain barrier modification for the treatment of malignant brain tumors. Clin. J. Oncol. Nurs. 2004, 8, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Kroll, R.A.; Neuwelt, E.A. Outwitting the blood-brain barrier for therapeutic purposes: Osmotic opening and other means. Neurosurgery 1998, 42, 1083–1099; discussion 1099–1100. [Google Scholar] [CrossRef]
- Siegal, T.; Rubinstein, R.; Bokstein, F.; Schwartz, A.; Lossos, A.; Shalom, E.; Chisin, R.; Gomori, J.M. In vivo assessment of the window of barrier opening after osmotic blood-brain barrier disruption in humans. J. Neurosurg. 2000, 92, 599–605. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, E.A.; da Costa Araujo, S.; Redeker, S.; van Schaik, R.; Aronica, E.; Gorter, J.A. Blood-brain barrier leakage may lead to progression of temporal lobe epilepsy. Brain J. Neurol. 2007, 130, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Marchi, N.; Angelov, L.; Masaryk, T.; Fazio, V.; Granata, T.; Hernandez, N.; Hallene, K.; Diglaw, T.; Franic, L.; Najm, I.; et al. Seizure-promoting effect of blood-brain barrier disruption. Epilepsia 2007, 48, 732–742. [Google Scholar] [CrossRef]
- Ikeda, M.; Bhattacharjee, A.K.; Kondoh, T.; Nagashima, T.; Tamaki, N. Synergistic effect of cold mannitol and na(+)/ca(2+) exchange blocker on blood-brain barrier opening. Biochem. Biophys. Res. Commun. 2002, 291, 669–674. [Google Scholar] [CrossRef]
- Stapleton, S.; Jaffray, D.; Milosevic, M. Radiation effects on the tumor microenvironment: Implications for nanomedicine delivery. Adv. Drug Deliv. Rev. 2017, 109, 119–130. [Google Scholar] [CrossRef]
- Brown, W.R.; Thore, C.R.; Moody, D.M.; Robbins, M.E.; Wheeler, K.T. Vascular damage after fractionated whole-brain irradiation in rats. Radiat. Res. 2005, 164, 662–668. [Google Scholar] [CrossRef] [PubMed]
- van Vulpen, M.; Kal, H.B.; Taphoorn, M.J.; El-Sharouni, S.Y. Changes in blood-brain barrier permeability induced by radiotherapy: Implications for timing of chemotherapy? (review). Oncol. Rep. 2002, 9, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Crowe, W.; Wang, L.; Zhang, Z.; Varagic, J.; Bourland, J.D.; Chan, M.D.; Habib, A.A.; Zhao, D. Mri evaluation of the effects of whole brain radiotherapy on breast cancer brain metastasis. Int. J. Radiat. Biol. 2019, 95, 338–346. [Google Scholar] [CrossRef]
- Teng, F.; Tsien, C.I.; Lawrence, T.S.; Cao, Y. Blood-tumor barrier opening changes in brain metastases from pre to one-month post radiation therapy. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2017, 125, 89–93. [Google Scholar] [CrossRef]
- Bouchet, A.; Potez, M.; Coquery, N.; Rome, C.; Lemasson, B.; Brauer-Krisch, E.; Remy, C.; Laissue, J.; Barbier, E.L.; Djonov, V.; et al. Permeability of brain tumor vessels induced by uniform or spatially microfractionated synchrotron radiation therapies. Int. J. Radiat. Oncol. Biol. Phys. 2017, 98, 1174–1182. [Google Scholar] [CrossRef]
- Lemasson, B.; Serduc, R.; Maisin, C.; Bouchet, A.; Coquery, N.; Robert, P.; Le Duc, G.; Tropres, I.; Remy, C.; Barbier, E.L. Monitoring blood-brain barrier status in a rat model of glioma receiving therapy: Dual injection of low-molecular-weight and macromolecular mr contrast media. Radiology 2010, 257, 342–352. [Google Scholar] [CrossRef]
- Fang, L.; Sun, X.; Song, Y.; Zhang, Y.; Li, F.; Xu, Y.; Ma, S.; Lin, N. Whole-brain radiation fails to boost intracerebral gefitinib concentration in patients with brain metastatic non-small cell lung cancer: A self-controlled, pilot study. Cancer Chemother. Pharmacol. 2015, 76, 873–877. [Google Scholar] [CrossRef]
- Zeng, Y.D.; Liao, H.; Qin, T.; Zhang, L.; Wei, W.D.; Liang, J.Z.; Xu, F.; Dinglin, X.X.; Ma, S.X.; Chen, L.K. Blood-brain barrier permeability of gefitinib in patients with brain metastases from non-small-cell lung cancer before and during whole brain radiation therapy. Oncotarget 2015, 6, 8366–8376. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A.; Chandra, R.; Cuccarese, M.F.; Pfirschke, C.; Engblom, C.; Stapleton, S.; Adhikary, U.; Kohler, R.H.; Mohan, J.F.; Pittet, M.J.; et al. Radiation therapy primes tumors for nanotherapeutic delivery via macrophage-mediated vascular bursts. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Le Pechoux, C.; Laplanche, A.; Faivre-Finn, C.; Ciuleanu, T.; Wanders, R.; Lerouge, D.; Keus, R.; Hatton, M.; Videtic, G.M.; Senan, S.; et al. Clinical neurological outcome and quality of life among patients with limited small-cell cancer treated with two different doses of prophylactic cranial irradiation in the intergroup phase iii trial (pci99-01, eortc 22003-08004, rtog 0212 and ifct 99-01). Ann. Oncol. Off. J. Eur. Soc. Med Oncol. 2011, 22, 1154–1163. [Google Scholar] [CrossRef]
- Dietrich, J.; Monje, M.; Wefel, J.; Meyers, C. Clinical patterns and biological correlates of cognitive dysfunction associated with cancer therapy. Oncologist 2008, 13, 1285–1295. [Google Scholar] [CrossRef] [PubMed]
- Emerich, D.F.; Dean, R.L.; Osborn, C.; Bartus, R.T. The development of the bradykinin agonist labradimil as a means to increase the permeability of the blood-brain barrier: From concept to clinical evaluation. Clin. Pharmacokinet. 2001, 40, 105–123. [Google Scholar] [CrossRef]
- de Vries, N.A.; Beijnen, J.H.; Boogerd, W.; van Tellingen, O. Blood-brain barrier and chemotherapeutic treatment of brain tumors. Expert Rev. Neurother. 2006, 6, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Bartus, R.T.; Elliott, P.J.; Dean, R.L.; Hayward, N.J.; Nagle, T.L.; Huff, M.R.; Snodgrass, P.A.; Blunt, D.G. Controlled modulation of bbb permeability using the bradykinin agonist, rmp-7. Exp. Neurol. 1996, 142, 14–28. [Google Scholar] [CrossRef]
- Sanovich, E.; Bartus, R.T.; Friden, P.M.; Dean, R.L.; Le, H.Q.; Brightman, M.W. Pathway across blood-brain barrier opened by the bradykinin agonist, rmp-7. Brain Res. 1995, 705, 125–135. [Google Scholar] [CrossRef]
- Matsukado, K.; Inamura, T.; Nakano, S.; Fukui, M.; Bartus, R.T.; Black, K.L. Enhanced tumor uptake of carboplatin and survival in glioma-bearing rats by intracarotid infusion of bradykinin analog, rmp-7. Neurosurgery 1996, 39, 125–133; discussion 124–133. [Google Scholar] [CrossRef]
- Borlongan, C.V.; Emerich, D.F. Facilitation of drug entry into the cns via transient permeation of blood brain barrier: Laboratory and preliminary clinical evidence from bradykinin receptor agonist, cereport. Brain Res. Bull. 2003, 60, 297–306. [Google Scholar] [CrossRef]
- Ford, J.; Osborn, C.; Barton, T.; Bleehen, N.M. A phase i study of intravenous rmp-7 with carboplatin in patients with progression of malignant glioma. Eur. J. Cancer 1998, 34, 1807–1811. [Google Scholar] [CrossRef]
- Warren, K.; Jakacki, R.; Widemann, B.; Aikin, A.; Libucha, M.; Packer, R.; Vezina, G.; Reaman, G.; Shaw, D.; Krailo, M.; et al. Phase ii trial of intravenous lobradimil and carboplatin in childhood brain tumors: A report from the children’s oncology group. Cancer Chemother. Pharmacol. 2006, 58, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Prados, M.D.; Schold, S.C., Jr.; Fine, H.A.; Jaeckle, K.; Hochberg, F.; Mechtler, L.; Fetell, M.R.; Phuphanich, S.; Feun, L.; Janus, T.J.; et al. A randomized, double-blind, placebo-controlled, phase 2 study of rmp-7 in combination with carboplatin administered intravenously for the treatment of recurrent malignant glioma. Neuro Oncol. 2003, 5, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Deeken, J.F.; Loscher, W. The blood-brain barrier and cancer: Transporters, treatment, and trojan horses. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2007, 13, 1663–1674. [Google Scholar] [CrossRef] [PubMed]
- Riley, M.G.; Kim, N.N.; Watson, V.E.; Gobin, Y.P.; LeBel, C.P.; Black, K.L.; Bartus, R.T. Intra-arterial administration of carboplatin and the blood brain barrier permeabilizing agent, rmp-7: A toxicologic evaluation in swine. J. Neurooncol. 1998, 36, 167–178. [Google Scholar] [CrossRef]
- Hashizume, K.; Black, K.L. Increased endothelial vesicular transport correlates with increased blood-tumor barrier permeability induced by bradykinin and leukotriene c4. J. Neuropathol. Exp. Neurol. 2002, 61, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Rozhkova, E.A. Nanoscale materials for tackling brain cancer: Recent progress and outlook. Adv. Mater. 2011, 23, H136–H150. [Google Scholar] [CrossRef] [PubMed]
- Haseloff, R.F.; Dithmer, S.; Winkler, L.; Wolburg, H.; Blasig, I.E. Transmembrane proteins of the tight junctions at the blood-brain barrier: Structural and functional aspects. Semin. Cell Dev. Biol. 2015, 38, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Zihni, C.; Mills, C.; Matter, K.; Balda, M.S. Tight junctions: From simple barriers to multifunctional molecular gates. Nat. Rev. Mol. Cell Biol. 2016, 17, 564–580. [Google Scholar] [CrossRef] [PubMed]
- Nitta, T.; Hata, M.; Gotoh, S.; Seo, Y.; Sasaki, H.; Hashimoto, N.; Furuse, M.; Tsukita, S. Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J. Cell Biol. 2003, 161, 653–660. [Google Scholar] [CrossRef]
- Campbell, M.; Kiang, A.S.; Kenna, P.F.; Kerskens, C.; Blau, C.; O’Dwyer, L.; Tivnan, A.; Kelly, J.A.; Brankin, B.; Farrar, G.J.; et al. Rnai-mediated reversible opening of the blood-brain barrier. J. Gene Med. 2008, 10, 930–947. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.; Hanrahan, F.; Gobbo, O.L.; Kelly, M.E.; Kiang, A.S.; Humphries, M.M.; Nguyen, A.T.; Ozaki, E.; Keaney, J.; Blau, C.W.; et al. Targeted suppression of claudin-5 decreases cerebral oedema and improves cognitive outcome following traumatic brain injury. Nat. Commun. 2012, 3, 849. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, K.; Hashimoto, Y.; Shirakura, K.; Okada, Y.; Hirayama, R.; Iwashita, Y.; Nishino, I.; Ago, Y.; Takeda, H.; Kuniyasu, H.; et al. Safety and efficacy of an anti-claudin-5 monoclonal antibody to increase blood-brain barrier permeability for drug delivery to the brain in a non-human primate. J. Control. Release Off. J. Control. Release Soc. 2021, 336, 105–111. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Zhou, W.; Hamauchi, K.; Shirakura, K.; Doi, T.; Yagi, K.; Sawasaki, T.; Okada, Y.; Kondoh, M.; Takeda, H. Engineered membrane protein antigens successfully induce antibodies against extracellular regions of claudin-5. Sci. Rep. 2018, 8, 8383. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Shirakura, K.; Okada, Y.; Takeda, H.; Endo, K.; Tamura, M.; Watari, A.; Sadamura, Y.; Sawasaki, T.; Doi, T.; et al. Claudin-5-binders enhance permeation of solutes across the blood-brain barrier in a mammalian model. J. Pharmacol. Exp. Ther. 2017, 363, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Dithmer, S.; Staat, C.; Muller, C.; Ku, M.C.; Pohlmann, A.; Niendorf, T.; Gehne, N.; Fallier-Becker, P.; Kittel, A.; Walter, F.R.; et al. Claudin peptidomimetics modulate tissue barriers for enhanced drug delivery. Ann. N. Y. Acad. Sci. 2017, 1397, 169–184. [Google Scholar] [CrossRef]
- Krug, S.M.; Amasheh, S.; Richter, J.F.; Milatz, S.; Gunzel, D.; Westphal, J.K.; Huber, O.; Schulzke, J.D.; Fromm, M. Tricellulin forms a barrier to macromolecules in tricellular tight junctions without affecting ion permeability. Mol. Biol. Cell 2009, 20, 3713–3724. [Google Scholar] [CrossRef] [PubMed]
- Zeniya, S.; Kuwahara, H.; Daizo, K.; Watari, A.; Kondoh, M.; Yoshida-Tanaka, K.; Kaburagi, H.; Asada, K.; Nagata, T.; Nagahama, M.; et al. Angubindin-1 opens the blood-brain barrier in vivo for delivery of antisense oligonucleotide to the central nervous system. J. Control. Release Off. J. Control. Release Soc. 2018, 283, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Krug, S.M.; Hayaishi, T.; Iguchi, D.; Watari, A.; Takahashi, A.; Fromm, M.; Nagahama, M.; Takeda, H.; Okada, Y.; Sawasaki, T.; et al. Angubindin-1, a novel paracellular absorption enhancer acting at the tricellular tight junction. J. Control. Release Off. J. Control. Release Soc. 2017, 260, 1–11. [Google Scholar] [CrossRef]
- Spampinato, S.F.; Obermeier, B.; Cotleur, A.; Love, A.; Takeshita, Y.; Sano, Y.; Kanda, T.; Ransohoff, R.M. Sphingosine 1 phosphate at the blood brain barrier: Can the modulation of s1p receptor 1 influence the response of endothelial cells and astrocytes to inflammatory stimuli? PLoS ONE 2015, 10, e0133392. [Google Scholar]
- Fan, L.; Yan, H. Fty720 attenuates retinal inflammation and protects blood-retinal barrier in diabetic rats. Investig. Ophthalmol. Vis. Sci. 2016, 57, 1254–1263. [Google Scholar] [CrossRef]
- Gril, B.; Paranjape, A.N.; Woditschka, S.; Hua, E.; Dolan, E.L.; Hanson, J.; Wu, X.; Kloc, W.; Izycka-Swieszewska, E.; Duchnowska, R.; et al. Reactive astrocytic s1p3 signaling modulates the blood-tumor barrier in brain metastases. Nat. Commun. 2018, 9, 2705. [Google Scholar] [CrossRef] [PubMed]
- Dusaban, S.S.; Chun, J.; Rosen, H.; Purcell, N.H.; Brown, J.H. Sphingosine 1-phosphate receptor 3 and rhoa signaling mediate inflammatory gene expression in astrocytes. J. Neuroinflammation 2017, 14, 111. [Google Scholar] [CrossRef]
- Sanna, M.G.; Vincent, K.P.; Repetto, E.; Nguyen, N.; Brown, S.J.; Abgaryan, L.; Riley, S.W.; Leaf, N.B.; Cahalan, S.M.; Kiosses, W.B.; et al. Bitopic sphingosine 1-phosphate receptor 3 (s1p3) antagonist rescue from complete heart block: Pharmacological and genetic evidence for direct s1p3 regulation of mouse cardiac conduction. Mol. Pharmacol. 2016, 89, 176–186. [Google Scholar] [CrossRef]
- Iannitti, T.; Palmieri, B. An update on the therapeutic role of alkylglycerols. Mar. Drugs 2010, 8, 2267–2300. [Google Scholar] [CrossRef]
- Erdlenbruch, B.; Alipour, M.; Fricker, G.; Miller, D.S.; Kugler, W.; Eibl, H.; Lakomek, M. Alkylglycerol opening of the blood-brain barrier to small and large fluorescence markers in normal and c6 glioma-bearing rats and isolated rat brain capillaries. Br. J. Pharmacol. 2003, 140, 1201–1210. [Google Scholar] [CrossRef] [PubMed]
- Erdlenbruch, B.; Jendrossek, V.; Eibl, H.; Lakomek, M. Transient and controllable opening of the blood-brain barrier to cytostatic and antibiotic agents by alkylglycerols in rats. Exp. Brain 2000, 135, 417–422. [Google Scholar]
- Erdlenbruch, B.; Jendrossek, V.; Kugler, W.; Eibl, H.; Lakomek, M. Increased delivery of erucylphosphocholine to c6 gliomas by chemical opening of the blood-brain barrier using intracarotid pentylglycerol in rats. Cancer Chemother. Pharmacol. 2002, 50, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Erdlenbruch, B.; Schinkhof, C.; Kugler, W.; Heinemann, D.E.; Herms, J.; Eibl, H.; Lakomek, M. Intracarotid administration of short-chain alkylglycerols for increased delivery of methotrexate to the rat brain. Br. J. Pharmacol. 2003, 139, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Pyeun, Y.S.; Kim, Y.W.; Cho, B.M.; Lee, T.H.; Moon, T.Y.; Suh, K.T.; Park, B.R. A model for research on the blood-brain barrier disruption induced by unsaturated fatty acid emulsion. Investig. Radiol. 2005, 40, 270–276. [Google Scholar] [CrossRef]
- Sztriha, L.; Betz, A.L. Oleic acid reversibly opens the blood-brain barrier. Brain Res. 1991, 550, 257–262. [Google Scholar] [CrossRef][Green Version]
- Zhou, W.; Chen, C.; Shi, Y.; Wu, Q.; Gimple, R.C.; Fang, X.; Huang, Z.; Zhai, K.; Ke, S.Q.; Ping, Y.F.; et al. Targeting glioma stem cell-derived pericytes disrupts the blood-tumor barrier and improves chemotherapeutic efficacy. Cell Stem Cell 2017, 21, 591–603 e594. [Google Scholar] [CrossRef] [PubMed]
- Guerra, D.A.P.; Paiva, A.E.; Sena, I.F.G.; Azevedo, P.O.; Silva, W.N.; Mintz, A.; Birbrair, A. Targeting glioblastoma-derived pericytes improves chemotherapeutic outcome. Angiogenesis 2018, 21, 667–675. [Google Scholar] [CrossRef]
- Rodriguez, P.L.; Jiang, S.; Fu, Y.; Avraham, S.; Avraham, H.K. The proinflammatory peptide substance p promotes blood-brain barrier breaching by breast cancer cells through changes in microvascular endothelial cell tight junctions. Int. J. Cancer 2014, 134, 1034–1044. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, E.; Zocchi, M.R.; Magni, E.; Panzeri, M.C.; Curnis, F.; Rugarli, C.; Ferrero, M.E.; Corti, A. Roles of tumor necrosis factor p55 and p75 receptors in tnf-alpha-induced vascular permeability. Am. J. Physiol. Cell Physiol. 2001, 281, C1173–C1179. [Google Scholar] [CrossRef] [PubMed]
- Connell, J.J.; Chatain, G.; Cornelissen, B.; Vallis, K.A.; Hamilton, A.; Seymour, L.; Anthony, D.C.; Sibson, N.R. Selective permeabilization of the blood-brain barrier at sites of metastasis. J. Natl. Cancer Inst. 2013, 105, 1634–1643. [Google Scholar] [CrossRef]
- Avraham, H.K.; Jiang, S.; Fu, Y.; Nakshatri, H.; Ovadia, H.; Avraham, S. Angiopoietin-2 mediates blood-brain barrier impairment and colonization of triple-negative breast cancer cells in brain. J. Pathol. 2014, 232, 369–381. [Google Scholar] [CrossRef]
- Gurnik, S.; Devraj, K.; Macas, J.; Yamaji, M.; Starke, J.; Scholz, A.; Sommer, K.; Di Tacchio, M.; Vutukuri, R.; Beck, H.; et al. Angiopoietin-2-induced blood-brain barrier compromise and increased stroke size are rescued by ve-ptp-dependent restoration of tie2 signaling. Acta Neuropathol. 2016, 131, 753–773. [Google Scholar] [CrossRef]
- Loscher, W.; Gericke, B. Novel intrinsic mechanisms of active drug extrusion at the blood-brain barrier: Potential targets for enhancing drug delivery to the brain? Pharmaceutics 2020, 12, 966. [Google Scholar] [CrossRef]
- Potschka, H. Targeting regulation of abc efflux transporters in brain diseases: A novel therapeutic approach. Pharmacol. Ther. 2010, 125, 118–127. [Google Scholar] [CrossRef]
- Bauer, B.; Hartz, A.M.; Fricker, G.; Miller, D.S. Modulation of p-glycoprotein transport function at the blood-brain barrier. Exp. Biol. Med. 2005, 230, 118–127. [Google Scholar] [CrossRef]
- Fox, E.; Bates, S.E. Tariquidar (xr9576): A p-glycoprotein drug efflux pump inhibitor. Expert Rev. Anticancer Ther. 2007, 7, 447–459. [Google Scholar] [CrossRef]
- Bors, L.A.; Erd, F. Overcoming the blood–brain barrier. Challenges and tricks for cns drug delivery. Sci. Pharm. 2019, 87, 6. [Google Scholar] [CrossRef]
- Shan, Y.; Cen, Y.; Zhang, Y.; Tan, R.; Zhao, J.; Nie, Z.; Zhang, J.; Yu, S. Effect of p-glycoprotein inhibition on the penetration of ceftriaxone across the blood-brain barrier. Neurochem. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Erdo, F.; Nagy, I.; Toth, B.; Bui, A.; Molnar, E.; Timar, Z.; Magnan, R.; Krajcsi, P. Abcb1a (p-glycoprotein) limits brain exposure of the anticancer drug candidate seliciclib in vivo in adult mice. Brain Res. Bull. 2017, 132, 232–236. [Google Scholar] [CrossRef]
- Carcaboso, A.M.; Elmeliegy, M.A.; Shen, J.; Juel, S.J.; Zhang, Z.M.; Calabrese, C.; Tracey, L.; Waters, C.M.; Stewart, C.F. Tyrosine kinase inhibitor gefitinib enhances topotecan penetration of gliomas. Cancer Res. 2010, 70, 4499–4508. [Google Scholar] [CrossRef]
- Choo, E.F.; Kurnik, D.; Muszkat, M.; Ohkubo, T.; Shay, S.D.; Higginbotham, J.N.; Glaeser, H.; Kim, R.B.; Wood, A.J.; Wilkinson, G.R. Differential in vivo sensitivity to inhibition of p-glycoprotein located in lymphocytes, testes, and the blood-brain barrier. J. Pharmacol. Exp. Ther. 2006, 317, 1012–1018. [Google Scholar] [CrossRef]
- Ekins, S.; Ecker, G.F.; Chiba, P.; Swaan, P.W. Future directions for drug transporter modelling. Xenobiotica; Fate Foreign Compd. Biol. Syst. 2007, 37, 1152–1170. [Google Scholar] [CrossRef]
- Harmsen, S.; Meijerman, I.; Beijnen, J.H.; Schellens, J.H. The role of nuclear receptors in pharmacokinetic drug-drug interactions in oncology. Cancer Treat Rev. 2007, 33, 369–380. [Google Scholar] [CrossRef]
- Zastre, J.A.; Chan, G.N.; Ronaldson, P.T.; Ramaswamy, M.; Couraud, P.O.; Romero, I.A.; Weksler, B.; Bendayan, M.; Bendayan, R. Up-regulation of p-glycoprotein by hiv protease inhibitors in a human brain microvessel endothelial cell line. J. Neurosci. Res. 2009, 87, 1023–1036. [Google Scholar] [CrossRef]
- Bauer, B.; Hartz, A.M.; Fricker, G.; Miller, D.S. Pregnane x receptor up-regulation of p-glycoprotein expression and transport function at the blood-brain barrier. Mol. Pharmacol. 2004, 66, 413–419. [Google Scholar]
- Huang, H.; Wang, H.; Sinz, M.; Zoeckler, M.; Staudinger, J.; Redinbo, M.R.; Teotico, D.G.; Locker, J.; Kalpana, G.V.; Mani, S. Inhibition of drug metabolism by blocking the activation of nuclear receptors by ketoconazole. Oncogene 2007, 26, 258–268. [Google Scholar] [CrossRef]
- Bankstahl, J.P.; Hoffmann, K.; Bethmann, K.; Loscher, W. Glutamate is critically involved in seizure-induced overexpression of p-glycoprotein in the brain. Neuropharmacology 2008, 54, 1006–1016. [Google Scholar] [CrossRef]
- Loscher, W. Pharmacology of glutamate receptor antagonists in the kindling model of epilepsy. Prog. Neurobiol. 1998, 54, 721–741. [Google Scholar] [CrossRef]
- Bauer, B.; Hartz, A.M.; Pekcec, A.; Toellner, K.; Miller, D.S.; Potschka, H. Seizure-induced up-regulation of p-glycoprotein at the blood-brain barrier through glutamate and cyclooxygenase-2 signaling. Mol. Pharmacol. 2008, 73, 1444–1453. [Google Scholar] [CrossRef]
- Zibell, G.; Unkruer, B.; Pekcec, A.; Hartz, A.M.; Bauer, B.; Miller, D.S.; Potschka, H. Prevention of seizure-induced up-regulation of endothelial p-glycoprotein by cox-2 inhibition. Neuropharmacology 2009, 56, 849–855. [Google Scholar] [CrossRef]
- van Vliet, E.A.; Zibell, G.; Pekcec, A.; Schlichtiger, J.; Edelbroek, P.M.; Holtman, L.; Aronica, E.; Gorter, J.A.; Potschka, H. Cox-2 inhibition controls p-glycoprotein expression and promotes brain delivery of phenytoin in chronic epileptic rats. Neuropharmacology 2010, 58, 404–412. [Google Scholar] [CrossRef]
- Kulkarni, S.K.; Dhir, A. Cyclooxygenase in epilepsy: From perception to application. Drugs Today 2009, 45, 135–154. [Google Scholar] [CrossRef]
- Pekcec, A.; Unkruer, B.; Schlichtiger, J.; Soerensen, J.; Hartz, A.M.; Bauer, B.; van Vliet, E.A.; Gorter, J.A.; Potschka, H. Targeting prostaglandin e2 ep1 receptors prevents seizure-associated p-glycoprotein up-regulation. J. Pharmacol. Exp. Ther. 2009, 330, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.S.; Ahmad, M.; de Brum-Fernandes, A.J.; Dore, S. Prostaglandin ep4 receptor agonist protects against acute neurotoxicity. Brain Res. 2005, 1066, 71–77. [Google Scholar] [CrossRef]
- Bilak, M.; Wu, L.; Wang, Q.; Haughey, N.; Conant, K.; St Hillaire, C.; Andreasson, K. Pge2 receptors rescue motor neurons in a model of amyotrophic lateral sclerosis. Ann. Neurol. 2004, 56, 240–248. [Google Scholar] [CrossRef] [PubMed]
- McCullough, L.; Wu, L.; Haughey, N.; Liang, X.; Hand, T.; Wang, Q.; Breyer, R.M.; Andreasson, K. Neuroprotective function of the pge2 ep2 receptor in cerebral ischemia. J. Neurosci. Off. J. Soc. Neurosci. 2004, 24, 257–268. [Google Scholar] [CrossRef]
- Spudich, A.; Kilic, E.; Xing, H.; Kilic, U.; Rentsch, K.M.; Wunderli-Allenspach, H.; Bassetti, C.L.; Hermann, D.M. Inhibition of multidrug resistance transporter-1 facilitates neuroprotective therapies after focal cerebral ischemia. Nat. Neurosci. 2006, 9, 487–488. [Google Scholar] [CrossRef]
- Cirrito, J.R.; Deane, R.; Fagan, A.M.; Spinner, M.L.; Parsadanian, M.; Finn, M.B.; Jiang, H.; Prior, J.L.; Sagare, A.; Bales, K.R.; et al. P-glycoprotein deficiency at the blood-brain barrier increases amyloid-beta deposition in an alzheimer disease mouse model. J. Clin. Investig. 2005, 115, 3285–3290. [Google Scholar] [CrossRef]
- Vogelgesang, S.; Cascorbi, I.; Schroeder, E.; Pahnke, J.; Kroemer, H.K.; Siegmund, W.; Kunert-Keil, C.; Walker, L.C.; Warzok, R.W. Deposition of alzheimer’s beta-amyloid is inversely correlated with p-glycoprotein expression in the brains of elderly non-demented humans. Pharmacogenetics 2002, 12, 535–541. [Google Scholar] [CrossRef]
- Deane, R.; Zlokovic, B.V. Role of the blood-brain barrier in the pathogenesis of alzheimer’s disease. Curr. Alzheimer Res. 2007, 4, 191–197. [Google Scholar] [CrossRef]
- Lee, G.; Bendayan, R. Functional expression and localization of p-glycoprotein in the central nervous system: Relevance to the pathogenesis and treatment of neurological disorders. Pharm. Res. 2004, 21, 1313–1330. [Google Scholar] [CrossRef]
- Lim, J.C.; Kania, K.D.; Wijesuriya, H.; Chawla, S.; Sethi, J.K.; Pulaski, L.; Romero, I.A.; Couraud, P.O.; Weksler, B.B.; Hladky, S.B.; et al. Activation of beta-catenin signalling by gsk-3 inhibition increases p-glycoprotein expression in brain endothelial cells. J. Neurochem. 2008, 106, 1855–1865. [Google Scholar]
- Ju, X.; Chen, H.; Miao, T.; Ni, J.; Han, L. Prodrug delivery using dual-targeting nanoparticles to treat breast cancer brain metastases. Mol. Pharm. 2021, 18, 2694–2702. [Google Scholar] [CrossRef]
- Khan, N.U.; Ni, J.; Ju, X.; Miao, T.; Chen, H.; Han, L. Escape from abluminal lrp1-mediated clearance for boosted nanoparticle brain delivery and brain metastasis treatment. Acta Pharm. Sin. B 2021, 11, 1341–1354. [Google Scholar] [CrossRef]
- Ni, J.; Miao, T.; Su, M.; Khan, N.U.; Ju, X.; Chen, H.; Liu, F.; Han, L. Psma-targeted nanoparticles for specific penetration of blood-brain tumor barrier and combined therapy of brain metastases. J. Control. Release Off. J. Control. Release Soc. 2021, 329, 934–947. [Google Scholar] [CrossRef]
- Guo, Q.; Zhu, Q.; Miao, T.; Tao, J.; Ju, X.; Sun, Z.; Li, H.; Xu, G.; Chen, H.; Han, L. Lrp1-upregulated nanoparticles for efficiently conquering the blood-brain barrier and targetedly suppressing multifocal and infiltrative brain metastases. J. Control. Release Off. J. Control. Release Soc. 2019, 303, 117–129. [Google Scholar] [CrossRef]
- Guo, Q.; Chang, Z.; Khan, N.U.; Miao, T.; Ju, X.; Feng, H.; Zhang, L.; Sun, Z.; Li, H.; Han, L. Nanosizing noncrystalline and porous silica material-naturally occurring opal shale for systemic tumor targeting drug delivery. ACS Appl. Mater. Interfaces 2018, 10, 25994–26004. [Google Scholar] [CrossRef] [PubMed]
- Dong, A.; Han, L.; Shao, Z.; Fan, P.; Zhou, X.; Yuan, H. Glaucoma drainage device coated with mitomycin c loaded opal shale microparticles to inhibit bleb fibrosis. ACS Appl. Mater. Interfaces 2019, 11, 10244–10253. [Google Scholar] [CrossRef] [PubMed]
- Ju, X.; Miao, T.; Chen, H.; Ni, J.; Han, L. Overcoming mfsd2a-mediated low transcytosis to boost nanoparticle delivery to brain for chemotherapy of brain metastases. Adv. Healthc. Mater. 2021, 10, e2001997. [Google Scholar] [CrossRef] [PubMed]
- Uchida, Y.; Ohtsuki, S.; Katsukura, Y.; Ikeda, C.; Suzuki, T.; Kamiie, J.; Terasaki, T. Quantitative targeted absolute proteomics of human blood-brain barrier transporters and receptors. J. Neurochem. 2011, 117, 333–345. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Ma, D.; Shui, G.; Wong, P.; Cazenave-Gassiot, A.; Zhang, X.; Wenk, M.R.; Goh, E.L.; Silver, D.L. Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid. Nature 2014, 509, 503–506. [Google Scholar] [CrossRef]
- Bengmark, S. Gut microbiota, immune development and function. Pharmacol. Res. 2013, 69, 87–113. [Google Scholar] [CrossRef]
- Alakbarzade, V.; Hameed, A.; Quek, D.Q.; Chioza, B.A.; Baple, E.L.; Cazenave-Gassiot, A.; Nguyen, L.N.; Wenk, M.R.; Ahmad, A.Q.; Sreekantan-Nair, A.; et al. A partially inactivating mutation in the sodium-dependent lysophosphatidylcholine transporter mfsd2a causes a non-lethal microcephaly syndrome. Nat. Genet. 2015, 47, 814–817. [Google Scholar] [CrossRef]
- Zhao, Z.; Sagare, A.P.; Ma, Q.; Halliday, M.R.; Kong, P.; Kisler, K.; Winkler, E.A.; Ramanathan, A.; Kanekiyo, T.; Bu, G.; et al. Central role for picalm in amyloid-beta blood-brain barrier transcytosis and clearance. Nat. Neurosci. 2015, 18, 978–987. [Google Scholar] [CrossRef]
- Andras, I.E.; Eum, S.Y.; Huang, W.; Zhong, Y.; Hennig, B.; Toborek, M. Hiv-1-induced amyloid beta accumulation in brain endothelial cells is attenuated by simvastatin. Mol. Cell. Neurosci. 2010, 43, 232–243. [Google Scholar] [CrossRef]
- Zandl-Lang, M.; Fanaee-Danesh, E.; Sun, Y.; Albrecher, N.M.; Gali, C.C.; Cancar, I.; Kober, A.; Tam-Amersdorfer, C.; Stracke, A.; Storck, S.M.; et al. Regulatory effects of simvastatin and apoj on app processing and amyloid-beta clearance in blood-brain barrier endothelial cells. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 40–60. [Google Scholar] [CrossRef]
- Zlokovic, B.V.; Yamada, S.; Holtzman, D.; Ghiso, J.; Frangione, B. Clearance of amyloid beta-peptide from brain: Transport or metabolism? Nat. Med. 2000, 6, 718–719. [Google Scholar] [CrossRef]
- Tobert, J.A. New developments in lipid-lowering therapy: The role of inhibitors of hydroxymethylglutaryl-coenzyme a reductase. Circulation 1987, 76, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.D.; Zhao, Z.; Montagne, A.; Nelson, A.R.; Zlokovic, B.V. Blood-brain barrier: From physiology to disease and back. Physiol. Rev. 2019, 99, 21–78. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Blood-brain barrier breakdown in alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 2018, 14, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Winkler, E.A.; Nishida, Y.; Sagare, A.P.; Rege, S.V.; Bell, R.D.; Perlmutter, D.; Sengillo, J.D.; Hillman, S.; Kong, P.; Nelson, A.R.; et al. Glut1 reductions exacerbate alzheimer’s disease vasculo-neuronal dysfunction and degeneration. Nat. Neurosci. 2015, 18, 521–530. [Google Scholar] [CrossRef]
- Zhao, C.; Ma, J.; Wang, Z.; Li, H.; Shen, H.; Li, X.; Chen, G. Mfsd2a attenuates blood-brain barrier disruption after sub-arachnoid hemorrhage by inhibiting caveolae-mediated transcellular transport in rats. Transl. Stroke Res. 2020, 11, 1012–1027. [Google Scholar] [CrossRef]
- Montagne, A.; Zhao, Z.; Zlokovic, B.V. Alzheimer’s disease: A matter of blood-brain barrier dysfunction? J. Exp. Med. 2017, 214, 3151–3169. [Google Scholar] [CrossRef]
- Guemez-Gamboa, A.; Nguyen, L.N.; Yang, H.; Zaki, M.S.; Kara, M.; Ben-Omran, T.; Akizu, N.; Rosti, R.O.; Rosti, B.; Scott, E.; et al. Inactivating mutations in mfsd2a, required for omega-3 fatty acid transport in brain, cause a lethal microcephaly syndrome. Nat. Genet. 2015, 47, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Patching, S.G. Glucose transporters at the blood-brain barrier: Function, regulation and gateways for drug delivery. Mol. Neurobiol. 2017, 54, 1046–1077. [Google Scholar] [CrossRef] [PubMed]
- Anraku, Y.; Kuwahara, H.; Fukusato, Y.; Mizoguchi, A.; Ishii, T.; Nitta, K.; Matsumoto, Y.; Toh, K.; Miyata, K.; Uchida, S.; et al. Glycaemic control boosts glucosylated nanocarrier crossing the bbb into the brain. Nat. Commun. 2017, 8, 1001. [Google Scholar] [CrossRef]
- Arsiwala, T.A.; Sprowls, S.A.; Blethen, K.E.; Adkins, C.E.; Saralkar, P.A.; Fladeland, R.A.; Pentz, W.; Gabriele, A.; Kielkowski, B.; Mehta, R.I.; et al. Ultrasound-mediated disruption of the blood tumor barrier for improved therapeutic delivery. Neoplasia 2021, 23, 676–691. [Google Scholar] [CrossRef]
- Meng, Y.; Suppiah, S.; Surendrakumar, S.; Bigioni, L.; Lipsman, N. Low-intensity mr-guided focused ultrasound mediated disruption of the blood-brain barrier for intracranial metastatic diseases. Front. Oncol. 2018, 8, 338. [Google Scholar] [CrossRef]
- Alonso, A. Ultrasound-induced blood-brain barrier opening for drug delivery. Front. Neurol. Neurosci. 2015, 36, 106–115. [Google Scholar] [PubMed]
- Sheikov, N.; McDannold, N.; Sharma, S.; Hynynen, K. Effect of focused ultrasound applied with an ultrasound contrast agent on the tight junctional integrity of the brain microvascular endothelium. Ultrasound Med. Biol. 2008, 34, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Sheikov, N.; McDannold, N.; Vykhodtseva, N.; Jolesz, F.; Hynynen, K. Cellular mechanisms of the blood-brain barrier opening induced by ultrasound in presence of microbubbles. Ultrasound Med. Biol. 2004, 30, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Huang, Q.; Wang, F.; Liu, Y.; Wang, Z.; Wang, Z.; Zhang, Q.; Lei, B.; Cheng, Y. The role of caveolin-1 in blood-brain barrier disruption induced by focused ultrasound combined with microbubbles. J. Mol. Neurosci. MN 2012, 46, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Aryal, M.; Fischer, K.; Gentile, C.; Gitto, S.; Zhang, Y.Z.; McDannold, N. Effects on p-glycoprotein expression after blood-brain barrier disruption using focused ultrasound and microbubbles. PLoS ONE 2017, 12, e0166061. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Gandhi, D.; Melhem, E.R.; Frenkel, V. Mri guided focused ultrasound-mediated delivery of therapeutic cells to the brain: A review of the state-of-the-art methodology and future applications. Front. Neurol. 2021, 12, 669449. [Google Scholar] [CrossRef]
- Chen, K.T.; Lin, Y.J.; Chai, W.Y.; Lin, C.J.; Chen, P.Y.; Huang, C.Y.; Kuo, J.S.; Liu, H.L.; Wei, K.C. Neuronavigation-guided focused ultrasound (navifus) for transcranial blood-brain barrier opening in recurrent glioblastoma patients: Clinical trial protocol. Ann. Transl. Med. 2020, 8, 673. [Google Scholar] [CrossRef]
- Appelboom, G.; Detappe, A.; LoPresti, M.; Kunjachan, S.; Mitrasinovic, S.; Goldman, S.; Chang, S.D.; Tillement, O. Stereotactic modulation of blood-brain barrier permeability to enhance drug delivery. Neuro. Oncol. 2016, 18, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- McDannold, N.; Zhang, Y.; Supko, J.G.; Power, C.; Sun, T.; Vykhodtseva, N.; Golby, A.J.; Reardon, D.A. Blood-brain barrier disruption and delivery of irinotecan in a rat model using a clinical transcranial mri-guided focused ultrasound system. Sci. Rep. 2020, 10, 8766. [Google Scholar] [CrossRef] [PubMed]
- Hersh, D.S.; Wadajkar, A.S.; Roberts, N.; Perez, J.G.; Connolly, N.P.; Frenkel, V.; Winkles, J.A.; Woodworth, G.F.; Kim, A.J. Evolving drug delivery strategies to overcome the blood brain barrier. Curr. Pharm. Des. 2016, 22, 1177–1193. [Google Scholar] [CrossRef]
- Lammers, T.; Koczera, P.; Fokong, S.; Gremse, F.; Ehling, J.; Vogt, M.; Pich, A.; Storm, G.; van Zandvoort, M.; Kiessling, F. Theranostic uspio-loaded microbubbles for mediating and monitoring blood-brain barrier permeation. Adv. Funct. Mater. 2015, 25, 36–43. [Google Scholar] [CrossRef]
- Huang, H.Y.; Liu, H.L.; Hsu, P.H.; Chiang, C.S.; Tsai, C.H.; Chi, H.S.; Chen, S.Y.; Chen, Y.Y. A multitheragnostic nanobubble system to induce blood-brain barrier disruption with magnetically guided focused ultrasound. Adv. Mater. 2015, 27, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Wang, Z.; Shen, J.; Xu, S.; Hu, Z. Nerve growth factor delivery by ultrasound-mediated nanobubble destruction as a treatment for acute spinal cord injury in rats. Int. J. Nanomed. 2017, 12, 1717–1729. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; McDannold, N.; Jolesz, F.A.; Hynynen, K. Noninvasive localized delivery of herceptin to the mouse brain by mri-guided focused ultrasound-induced blood-brain barrier disruption. Proc. Natl. Acad. Sci. USA 2006, 103, 11719–11723. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.J.; Selert, K.; Gao, Z.; Samiotaki, G.; Baseri, B.; Konofagou, E.E. Noninvasive and localized blood-brain barrier disruption using focused ultrasound can be achieved at short pulse lengths and low pulse repetition frequencies. J. Cereb. Blood Flow Metab. 2011, 31, 725–737. [Google Scholar] [CrossRef]
- Baseri, B.; Choi, J.J.; Tung, Y.S.; Konofagou, E.E. Multi-modality safety assessment of blood-brain barrier opening using focused ultrasound and definity microbubbles: A short-term study. Ultrasound Med. Biol. 2010, 36, 1445–1459. [Google Scholar] [CrossRef]
- Hynynen, K.; McDannold, N.; Vykhodtseva, N.; Jolesz, F.A. Noninvasive mr imaging-guided focal opening of the blood-brain barrier in rabbits. Radiology 2001, 220, 640–646. [Google Scholar] [CrossRef]
- Wala, K.; Szlasa, W.; Saczko, J.; Rudno-Rudzinska, J.; Kulbacka, J. Modulation of blood-brain barrier permeability by activating adenosine a2 receptors in oncological treatment. Biomolecules 2021, 11, 633. [Google Scholar] [CrossRef]
- Sassaroli, E.; O’Neill, B.E. Modulation of the interstitial fluid pressure by high intensity focused ultrasound as a way to alter local fluid and solute movement: Insights from a mathematical model. Phys. Med. Biol. 2014, 59, 6775–6795. [Google Scholar] [CrossRef] [PubMed]
- Burgess, A.; Nhan, T.; Moffatt, C.; Klibanov, A.L.; Hynynen, K. Analysis of focused ultrasound-induced blood-brain barrier permeability in a mouse model of alzheimer’s disease using two-photon microscopy. J. Control. Release Off. J. Control. Release Soc. 2014, 192, 243–248. [Google Scholar] [CrossRef] [PubMed]
- McMahon, D.; Poon, C.; Hynynen, K. Evaluating the safety profile of focused ultrasound and microbubble-mediated treatments to increase blood-brain barrier permeability. Expert Opin. Drug Deliv. 2019, 16, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Effendi, W.I.; Nagano, T.; Kobayashi, K.; Nishimura, Y. Focusing on adenosine receptors as a potential targeted therapy in human diseases. Cells 2020, 9, 785. [Google Scholar] [CrossRef]
- Ledent, C.; Vaugeois, J.M.; Schiffmann, S.N.; Pedrazzini, T.; El Yacoubi, M.; Vanderhaeghen, J.J.; Costentin, J.; Heath, J.K.; Vassart, G.; Parmentier, M. Aggressiveness, hypoalgesia and high blood pressure in mice lacking the adenosine a2a receptor. Nature 1997, 388, 674–678. [Google Scholar] [CrossRef]
- Bynoe, M.S.; Viret, C.; Yan, A.; Kim, D.G. Adenosine receptor signaling: A key to opening the blood-brain door. Fluids Barriers CNS 2015, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Bobermin, L.D.; Roppa, R.H.A.; Quincozes-Santos, A. Adenosine receptors as a new target for resveratrol-mediated glioprotection. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 634–647. [Google Scholar] [CrossRef]
- Kim, D.G.; Bynoe, M.S. A2a adenosine receptor modulates drug efflux transporter p-glycoprotein at the blood-brain barrier. J. Clin. Investig. 2016, 126, 1717–1733. [Google Scholar] [CrossRef]
- Jackson, S.; Anders, N.M.; Mangraviti, A.; Wanjiku, T.M.; Sankey, E.W.; Liu, A.; Brem, H.; Tyler, B.; Rudek, M.A.; Grossman, S.A. The effect of regadenoson-induced transient disruption of the blood-brain barrier on temozolomide delivery to normal rat brain. J. Neurooncol. 2016, 126, 433–439. [Google Scholar] [CrossRef]
- Vezina, A.; Manglani, M.; Morris, D.; Foster, B.; McCord, M.; Song, H.; Zhang, M.; Davis, D.; Zhang, W.; Bills, J.; et al. Adenosine a2a receptor activation enhances blood-tumor barrier permeability in a rodent glioma model. Mol. cancer Res. MCR 2021. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.G.; Bynoe, M.S. A2a adenosine receptor regulates the human blood-brain barrier permeability. Mol. Neurobiol. 2015, 52, 664–678. [Google Scholar] [CrossRef] [PubMed]
- Carman, A.J.; Mills, J.H.; Krenz, A.; Kim, D.G.; Bynoe, M.S. Adenosine receptor signaling modulates permeability of the blood-brain barrier. J. Neurosci. Off. J. Soc. Neurosci. 2011, 31, 13272–13280. [Google Scholar] [CrossRef] [PubMed]
- Pak, R.W.; Kang, J.; Valentine, H.; Loew, L.M.; Thorek, D.L.J.; Boctor, E.M.; Wong, D.F.; Kang, J.U. Voltage-sensitive dye delivery through the blood brain barrier using adenosine receptor agonist regadenoson. Biomed. Opt. Express 2018, 9, 3915–3922. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.; Weingart, J.; Nduom, E.K.; Harfi, T.T.; George, R.T.; McAreavey, D.; Ye, X.; Anders, N.M.; Peer, C.; Figg, W.D.; et al. The effect of an adenosine a2a agonist on intra-tumoral concentrations of temozolomide in patients with recurrent glioblastoma. Fluids Barriers CNS 2018, 15, 2. [Google Scholar] [CrossRef]
- Jackson, S.; George, R.T.; Lodge, M.A.; Piotrowski, A.; Wahl, R.L.; Gujar, S.K.; Grossman, S.A. The effect of regadenoson on the integrity of the human blood-brain barrier, a pilot study. J. Neurooncol. 2017, 132, 513–519. [Google Scholar] [CrossRef]
- Meng, L.; Wang, C.; Lu, Y.; Sheng, G.; Yang, L.; Wu, Z.; Xu, H.; Han, C.; Lu, Y.; Han, F. Targeted regulation of blood-brain barrier for enhanced therapeutic efficiency of hypoxia-modifier nanoparticles and immune checkpoint blockade antibodies for glioblastoma. ACS Appl. Mater. Interfaces 2021, 13, 11657–11671. [Google Scholar] [CrossRef]
- Han, L.; Cai, Q.; Tian, D.; Kong, D.K.; Gou, X.; Chen, Z.; Strittmatter, S.M.; Wang, Z.; Sheth, K.N.; Zhou, J. Targeted drug delivery to ischemic stroke via chlorotoxin-anchored, lexiscan-loaded nanoparticles. Nanomedicine 2016, 12, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Kong, D.K.; Zheng, M.Q.; Murikinati, S.; Ma, C.; Yuan, P.; Li, L.; Tian, D.; Cai, Q.; Ye, C.; et al. Increased nanoparticle delivery to brain tumors by autocatalytic priming for improved treatment and imaging. ACS Nano 2016, 10, 4209–4218. [Google Scholar] [CrossRef]
- Zou, Y.; Liu, Y.; Yang, Z.; Zhang, D.; Lu, Y.; Zheng, M.; Xue, X.; Geng, J.; Chung, R.; Shi, B. Effective and targeted human orthotopic glioblastoma xenograft therapy via a multifunctional biomimetic nanomedicine. Adv. Mater. 2018, 30, e1803717. [Google Scholar] [CrossRef]
- Gao, X.; Qian, J.; Zheng, S.; Changyi, Y.; Zhang, J.; Ju, S.; Zhu, J.; Li, C. Overcoming the blood-brain barrier for delivering drugs into the brain by using adenosine receptor nanoagonist. ACS Nano 2014, 8, 3678–3689. [Google Scholar] [CrossRef] [PubMed]
- Khaitan, D.; Reddy, P.L.; Ningaraj, N. Targeting brain tumors with nanomedicines: Overcoming blood brain barrier challenges. Curr. Clin. Pharmacol. 2018, 13, 110–119. [Google Scholar] [CrossRef]
- Ningaraj, N.S.; Rao, M.K.; Black, K.L. Adenosine 5′-triphosphate-sensitive potassium channel-mediated blood-brain tumor barrier permeability increase in a rat brain tumor model. Cancer Res. 2003, 63, 8899–8911. [Google Scholar]
- Brayden, J.E. Functional roles of katp channels in vascular smooth muscle. Clin. Exp. Pharmacol. Physiol. 2002, 29, 312–316. [Google Scholar] [CrossRef]
- Kitazono, T.; Faraci, F.M.; Taguchi, H.; Heistad, D.D. Role of potassium channels in cerebral blood vessels. Stroke 1995, 26, 1713–1723. [Google Scholar] [CrossRef]
- Ruoslahti, E. Specialization of tumour vasculature. Nat. Rev. Cancer 2002, 2, 83–90. [Google Scholar] [CrossRef]
- Ningaraj, N.S.; Sankpal, U.T.; Khaitan, D.; Meister, E.A.; Vats, T.S. Modulation of kca channels increases anticancer drug delivery to brain tumors and prolongs survival in xenograft model. Cancer Biol. Ther. 2009, 8, 1924–1933. [Google Scholar] [CrossRef][Green Version]
- Gu, Y.T.; Xue, Y.X.; Wang, Y.F.; Wang, J.H.; Chen, X.; ShangGuan, Q.R.; Lian, Y.; Zhong, L.; Meng, Y.N. Minoxidil sulfate induced the increase in blood-brain tumor barrier permeability through ros/rhoa/pi3k/pkb signaling pathway. Neuropharmacology 2013, 75, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.T.; Xue, Y.X.; Zhang, H.; Li, Y.; Liang, X.Y. Adenosine 5′-triphosphate-sensitive potassium channel activator induces the up-regulation of caveolin-1 expression in a rat brain tumor model. Cell. Mol. Neurobiol. 2011, 31, 629–634. [Google Scholar] [CrossRef]
- Tinker, A.; Aziz, Q.; Thomas, A. The role of atp-sensitive potassium channels in cellular function and protection in the cardiovascular system. Br. J. Pharmacol. 2014, 171, 12–23. [Google Scholar] [CrossRef]
- Rich, J.N.; Bigner, D.D. Development of novel targeted therapies in the treatment of malignant glioma. Nat. Rev. Drug Discov. 2004, 3, 430–446. [Google Scholar] [CrossRef]
- Lockman, P.R.; Mittapalli, R.K.; Taskar, K.S.; Rudraraju, V.; Gril, B.; Bohn, K.A.; Adkins, C.E.; Roberts, A.; Thorsheim, H.R.; Gaasch, J.A.; et al. Heterogeneous blood-tumor barrier permeability determines drug efficacy in experimental brain metastases of breast cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2010, 16, 5664–5678. [Google Scholar] [CrossRef]
- Gallo, J.M.; Li, S.; Guo, P.; Reed, K.; Ma, J. The effect of p-glycoprotein on paclitaxel brain and brain tumor distribution in mice. Cancer Res 2003, 63, 5114–5117. [Google Scholar]
- Hanton, G.; Sobry, C.; Dagues, N.; Rochefort, G.Y.; Bonnet, P.; Eder, V. Cardiovascular toxicity of minoxidil in the marmoset. Toxicol. Lett. 2008, 180, 157–165. [Google Scholar] [CrossRef]
- Miao, T.T.; Ju, X.F.; Zhu, Q.N.; Wang, Y.M.; Guo, Q.; Sun, T.; Lu, C.Z.; Han, L. Nanoparticles surmounting blood-brain tumor barrier through both transcellular and paracellular pathways to target brain metastases. Adv. Funct. Mater. 2019, 29, 201900259. [Google Scholar] [CrossRef]
- Guo, S.; Som, A.T.; Arai, K.; Lo, E.H. Effects of angiotensin-ii on brain endothelial cell permeability via pparalpha regulation of para- and trans-cellular pathways. Brain Res. 2019, 1722, 146353. [Google Scholar] [CrossRef]
- Patel, B.; Yang, P.H.; Kim, A.H. The effect of thermal therapy on the blood-brain barrier and blood-tumor barrier. Int. J. Hyperth. 2020, 37, 35–43. [Google Scholar] [CrossRef]
- Ashraf, O.; Patel, N.V.; Hanft, S.; Danish, S.F. Laser-induced thermal therapy in neuro-oncology: A review. World Neurosurg. 2018, 112, 166–177. [Google Scholar] [CrossRef] [PubMed]
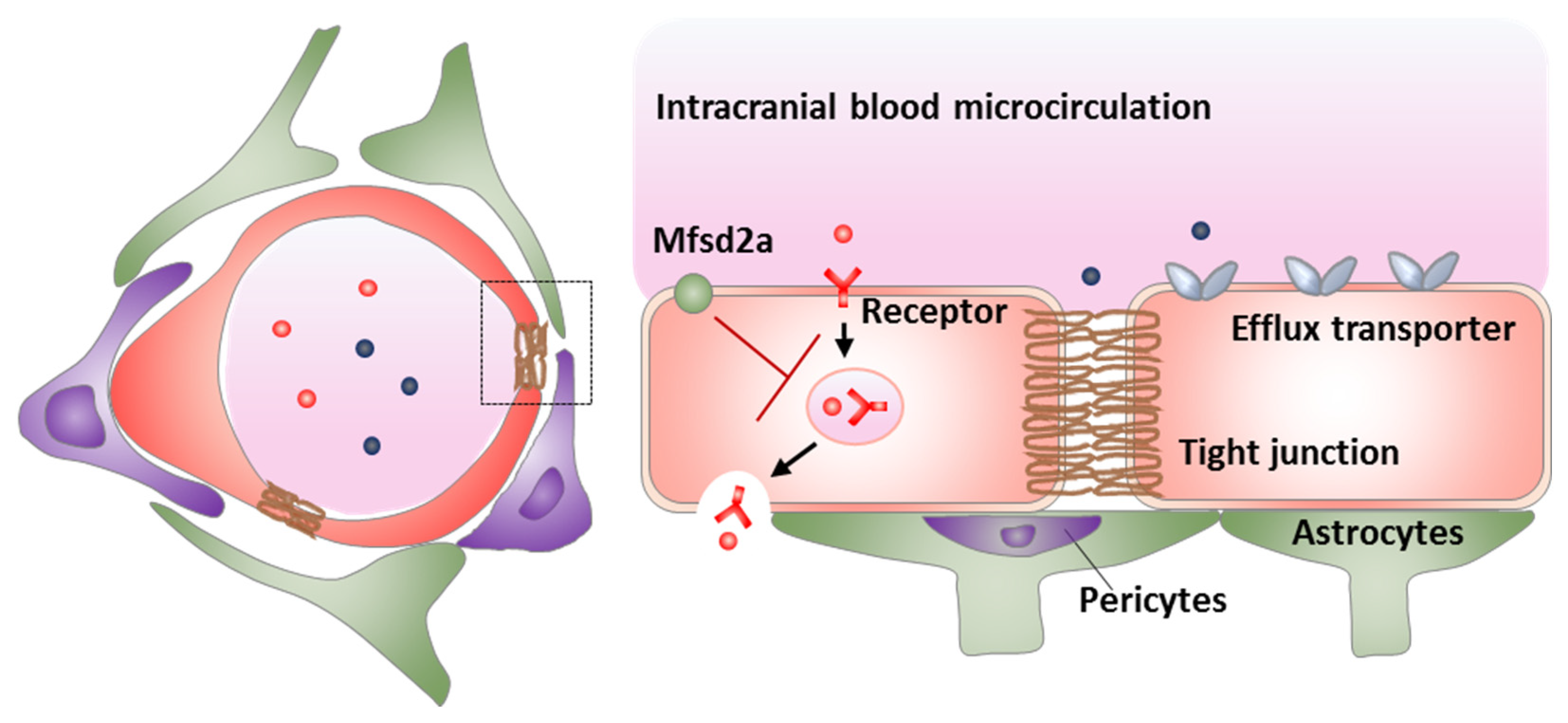

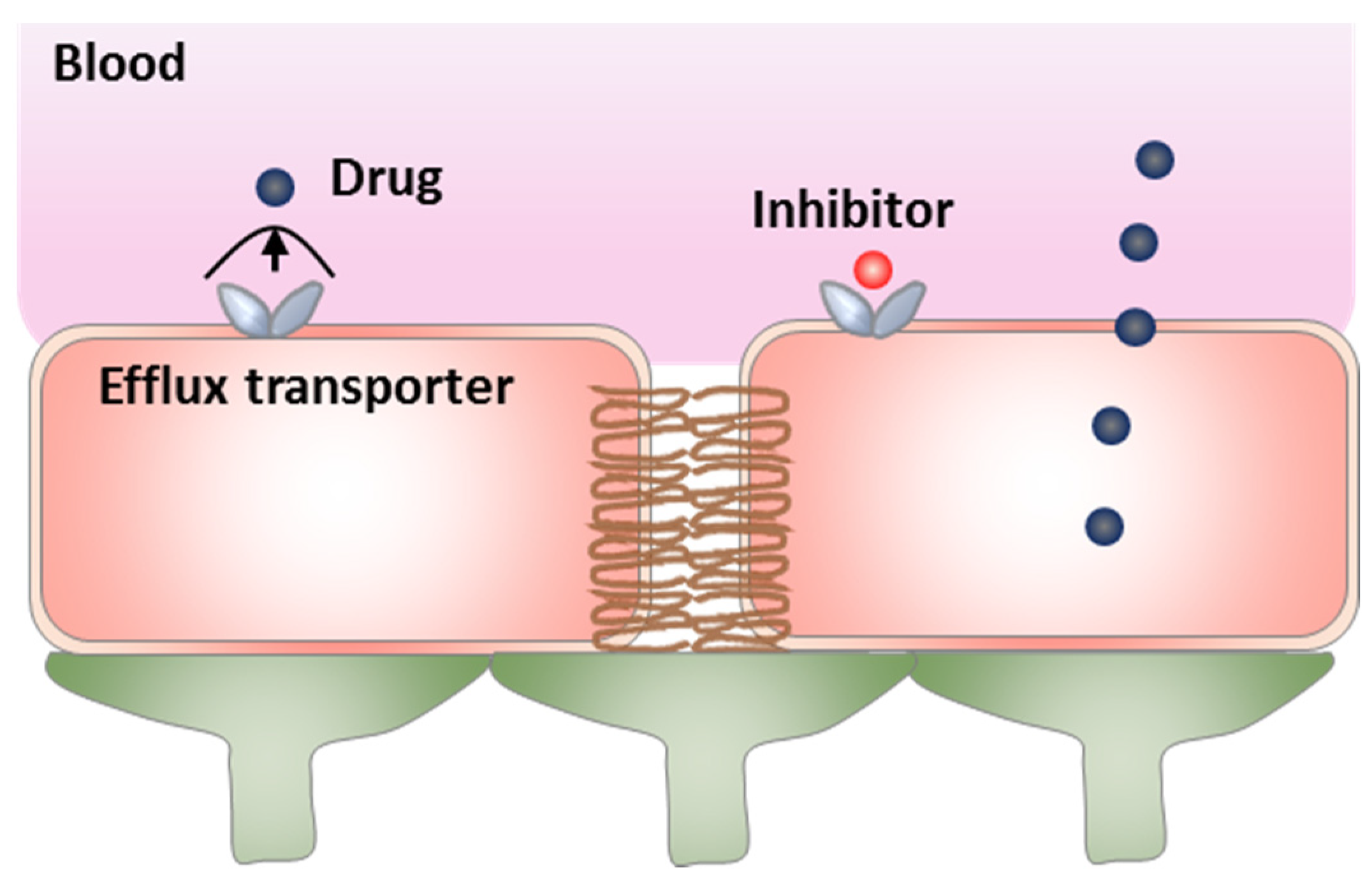

| BBB Modulation Targets | Strategies | Advantages | Disadvantages |
|---|---|---|---|
| Tight junctions | Osmotic disruption | Transient and reversible | Nonselective, uncontrolled flow, invasive, anesthesia, and side effects |
| Radiation-mediated disruption | Disease-specific | Unclear mechanisms and acute, subacute, and chronic dose-dependent toxicity | |
| Activating bradykinin B2 receptor | Disease-specific, rapid and transient | Limited application to only brain tumor and peripheral side effects | |
| Direct interference | Transient and reversible | Peripheral side effects | |
| Active efflux | Direct Inhibition | Transient and reversible… | Tolerability concerns of the inhibitor, and side effects to both brain and peripheral tissues |
| Targeting regulatory pathways | Disease-specific | Slow and side effects | |
| Transcytosis | Upregulation of LRP1 | Drug-specific | Slow and possible LRP1-associated side effects |
| Inhibition of Mfsd2a | Transient and reversible | Possible Mfsd2a-associated side effects | |
| Upregulation of GLUT1 | Efficient | Fasting-associated poor compliance |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, L. Modulation of the Blood–Brain Barrier for Drug Delivery to Brain. Pharmaceutics 2021, 13, 2024. https://doi.org/10.3390/pharmaceutics13122024
Han L. Modulation of the Blood–Brain Barrier for Drug Delivery to Brain. Pharmaceutics. 2021; 13(12):2024. https://doi.org/10.3390/pharmaceutics13122024
Chicago/Turabian StyleHan, Liang. 2021. "Modulation of the Blood–Brain Barrier for Drug Delivery to Brain" Pharmaceutics 13, no. 12: 2024. https://doi.org/10.3390/pharmaceutics13122024
APA StyleHan, L. (2021). Modulation of the Blood–Brain Barrier for Drug Delivery to Brain. Pharmaceutics, 13(12), 2024. https://doi.org/10.3390/pharmaceutics13122024






