Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison
Abstract
:1. Introduction
2. Topical Drug Delivery
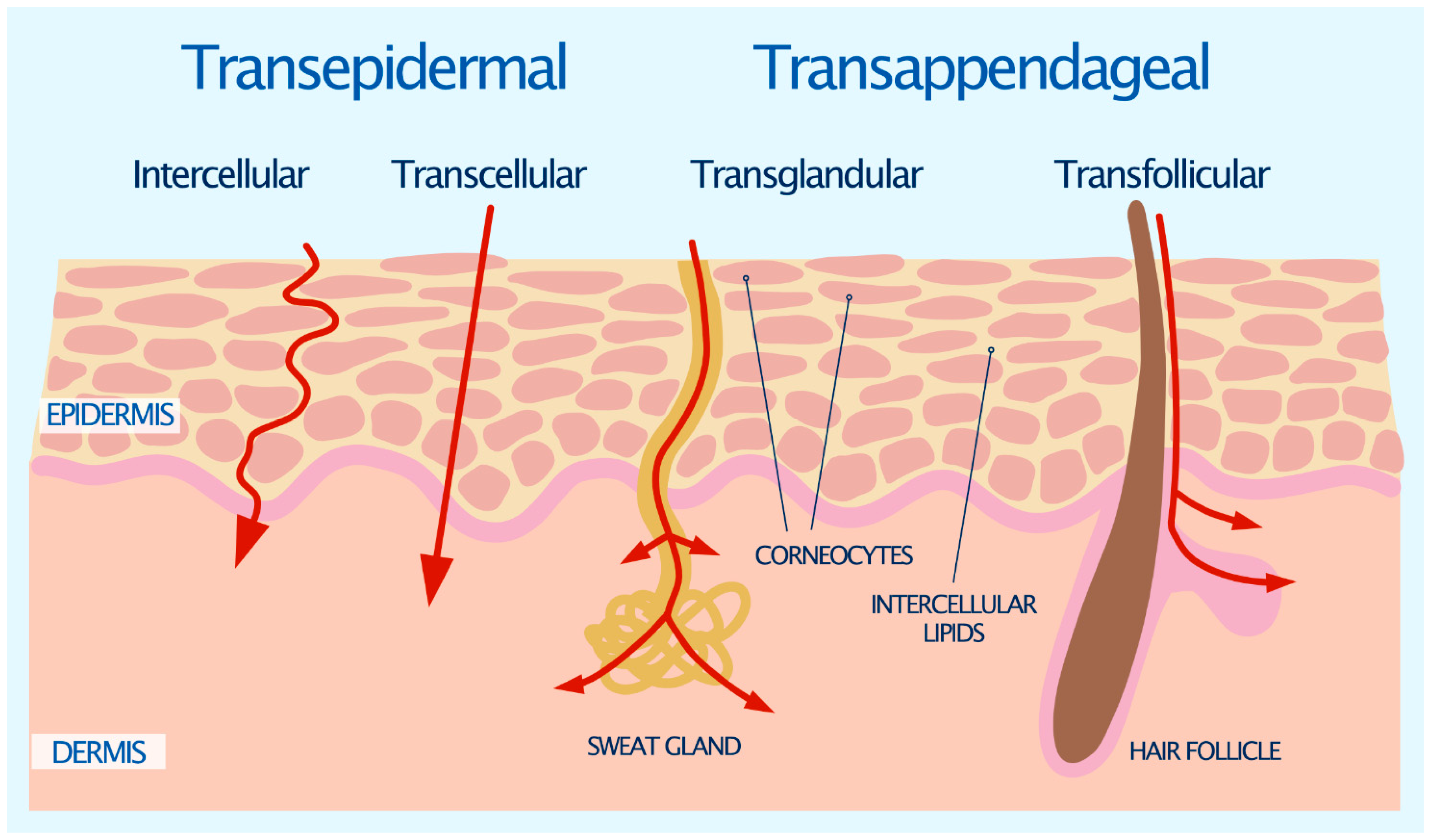
2.1. Transepidermal Route
2.2. Transappendageal Route
3. The Role of the Vehicle
4. Types of Vehicle
4.1. Ointments
4.2. Creams
4.3. Gels
4.4. Lotions
4.5. Solutions
4.6. Foams
4.7. Sprays
5. Choosing a Vehicle
6. The Role of Moisturizers
7. Types of Moisturizers
7.1. Primary Moisturizing Ingredients
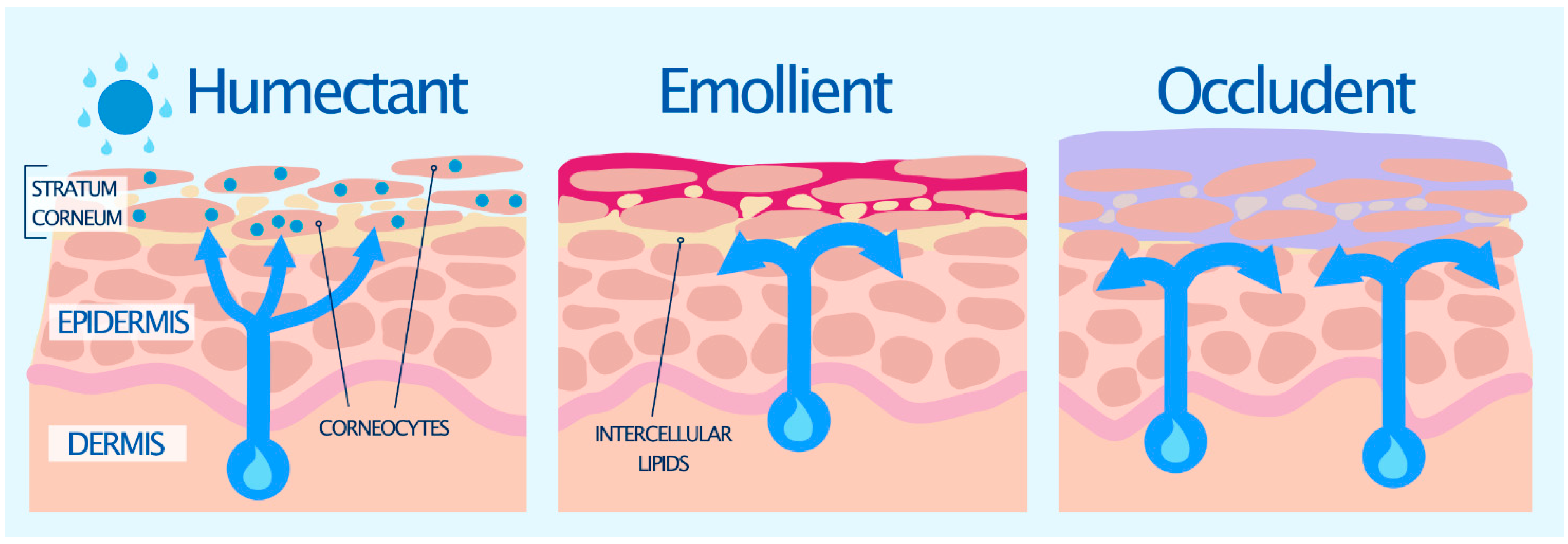
7.1.1. Humectants
7.1.2. Emollients
7.1.3. Occludents
7.2. Secondary Moisturizing Ingredients
8. Choosing a Moisturizer Vehicle
9. The Role of Excipients
9.1. Stiffening Agents
9.2. Thickeners/Gelling Agents
9.3. Silicones
9.4. Humectants
9.5. Emulsifiers/Solubilisers
9.6. Solvents
9.7. Penetration Enhancers
9.8. Chelating Agents
9.9. Acidifying/Alkalising/Buffering Agent
9.10. Antioxidants
9.11. Preservatives
10. Regulatory Considerations
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Elias, P.M. Stratum corneum defensive functions: An integrated view. J. Investig. Dermatol. 2005, 125, 183–200. [Google Scholar] [CrossRef]
- Haftek, M.; Roy, D.C.; Liao, I.-C. Evolution of skin barrier science for healthy and compromised skin. J. Drugs. Dermatol. 2021, 20, s3–s9. [Google Scholar] [CrossRef]
- Roure, R.; Lanctin, M.; Nollent, V.; Bertin, C. Methods to assess the protective efficacy of emollients against climatic and chemical aggressors. Dermatol. Res. Pract. 2012, 2012, 864734. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lim, K.M. Skin barrier dysfunction and filaggrin. Arch. Pharm. Res. 2021, 44, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Chandan, N.; Rajkumar, J.R.; Shi, V.Y.; Lio, P.A. A new era of moisturizers. J. Cosmet. Dermatol. 2021, 20, 2425–2430. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, B.E.; Leung, D.Y.M. Pathophysiology of atopic dermatitis: Clinical implications. Allergy Asthma Proc. 2019, 40, 84–92. [Google Scholar] [CrossRef]
- Harrison, I.P.; Spada, F. Breaking the itch-scratch cycle: Topical options for the management of chronic cutaneous itch in atopic dermatitis. Medicines 2019, 6, 76. [Google Scholar] [CrossRef] [Green Version]
- Kahremany, S.; Hofmann, L.; Harari, M.; Gruzman, A.; Cohen, G. Pruritus in psoriasis and atopic dermatitis: Current treatments and new perspectives. Pharmacol. Rep. 2021, 73, 443–453. [Google Scholar] [CrossRef]
- Morris-Jones, R.; Robertson, S.J.; Ross, J.S. Dermatitis caused by physical irritants. Br. J. Dermatol. 2002, 147, 270–275. [Google Scholar] [CrossRef]
- Cork, M.J. The role of Staphylococcus aureus in atopic eczema: Treatment strategies. J. Eur. Acad. Dermatol. Venereol. 1996, 7, 31–37. [Google Scholar] [CrossRef]
- Greive, K. Cleansers and moisturisers: The basics. Wound. Pract. Res. 2015, 23, 76–81. [Google Scholar]
- Wang, Z.; Man, M.-Q.; Li, T.; Elias, P.M.; Mauro, T.M. Aging-associated alterations in epidermal function and their clinical significance. Aging 2020, 12, 5551–5565. [Google Scholar] [CrossRef] [PubMed]
- Surber, C. Vehicles and Topical Therapy. Chapter 47. In Percutaneous Absorption. Drugs, Cosmetics, Mechanisms, Methods, 5th ed.; Dragićević, N., Maibach, H., Eds.; CRC Press: Boca Raton, FL, USA, 2021; pp. 687–691. [Google Scholar]
- Barry, B.W. Novel mechanisms and devices to enable successful transdermal drug delivery. Eur. J. Pharm. Sci. 2001, 14, 101–114. [Google Scholar] [CrossRef]
- Ghaffarian, R.; Muro, S. Models and methods to evaluate transport of drug delivery systems across cellular barriers. J. Vis. Exp. 2013, 80, e50638. [Google Scholar] [CrossRef] [PubMed]
- Elias, P.M.; Grayson, S.; Lampe, M.A.; Williams, M.L.; Brown, B.E. The Intercomeocyte Space. In Stratum Corneum, 1st ed.; Marks, R., Plewig, G., Eds.; Springer: Berlin/Heidelberg, Germany, 1983; pp. 53–67. [Google Scholar]
- Hadgraft, J. Skin deep. Eur. J. Pharm. Biopharm. 2004, 58, 291–299. [Google Scholar] [CrossRef]
- Potts, R.O.; Francoeur, M.L. Lipid biophysics of water loss through the skin. Proc. Natl. Acad. Sci. USA 1990, 87, 3871–3873. [Google Scholar] [CrossRef] [Green Version]
- Ng, K.W.; Lau, W.M. Skin Deep: The basics of Human Skin Structure and Drug Penetration. In Percutaneous Penetration Enhancers Chemical Methods in Penetration Enhancement, 1st ed.; Dragicevic, N., Maibach, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 3–11. [Google Scholar]
- Otberg, N.; Richter, H.; Schaefer, H.; Blume-Peytavi, U.; Sterry, W.; Lademann, J. Variations of hair follicle size and distribution in different body sites. J. Investig. Dermatol. 2004, 122, 14–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morrow, D.I.J.; McCarron, P.A.; Woolfson, A.D.; Donnelly, R.F. Innovative strategies for enhancing topical and transdermal drug delivery. Open Drug. Deliv. J. 2007, 1, 36–59. [Google Scholar] [CrossRef]
- Benson, H.A.E. Transdermal drug delivery: Penetration enhancement techniques. Curr. Drug. Deliv. 2005, 2, 23–33. [Google Scholar] [CrossRef]
- Kircik, L.; Bikowski, J.; Cohen, D.E.; Draelos, Z.D.; Hebert, A.; West, D.P. Formulation development, testing, and approval: Part 1of 2. Pract. Dermatol. 2010, 1–16. [Google Scholar]
- Brown, M.B.; Turner, M.; Lim, S.T. Topical Product Formulation Development. In Transdermal and Topical Drug Delivery: Principles and Practice, 1st ed.; Benson, H.A.E., Watkinson, A., Eds.; John Wiley and Sons: Hoboken, NJ, USA, 2012; pp. 255–286. [Google Scholar]
- Danby, S.G.; Draelos, Z.D.; Stein Gold, L.F.; Cha, A.; Vlahos, B.; Aikman, L.; Sanders, P.; Wu-Linhares, D.; Cork, M.J. Vehicles for atopic dermatitis therapies: More than just a placebo. J. Dermatolog. Treat. 2020, 1–14. [Google Scholar] [CrossRef]
- Mayba, J.N.; Gooderham, M.J. A Guide to topical vehicle formulations. J. Cutan. Med. Surg. 2018, 22, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.; Landriscina, A.; Friedman, A.J. Principles and approaches for optimizing therapy with unique topical vehicles. J. Drugs. Dermatol. 2014, 13, 1431–1435. [Google Scholar]
- Buhse, L.; Kolinski, R.; Westenberger, B.; Wokovich, A.; Spencer, J.; Chen, C.W.; Turujman, S.; Gautam-Basak, M.; Kang, G.J.; Kibbe, A.; et al. Topical drug classification. Int. J. Pharm. 2005, 295, 101–112. [Google Scholar] [CrossRef] [PubMed]
- De Souza, A.; Strober, B.E. Principles of Topical Therapy. Chapter 214. In Fitzpatrick’s Dermatology in General Medicine, 8th ed.; Goldsmith, L.A., Katz, S.I., Gilchrest, B.A., Paller, A.S., Leffell, D.J., Wolff, K., Eds.; McGraw-Hill: New York, NY, USA, 2012; pp. 2643–2651. [Google Scholar]
- Hughes, J.; Rustin, M. Corticosteroids. Clin. Dermatol. 1997, 15, 715–721. [Google Scholar] [CrossRef]
- Tan, X.; Feldman, S.R.; Chang, J.; Balkrishnan, R. Topical drug delivery systems in dermatology: A review of patient adherence issues. Expert Opin. Drug. Deliv. 2012, 9, 1263–1271. [Google Scholar] [CrossRef]
- Del Rosso, J.; Friedlander, S.F. Corticosteroids: Options in the era of steroid-sparing therapy. J. Am. Acad. Dermatol. 2005, 53, 50–58. [Google Scholar] [CrossRef]
- Weiss, S.C. Conventional topical delivery systems. Dermatol. Ther. 2011, 24, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Tanojo, H.; Lenn, J.; Deng, C.H.; Krochmal, L. A novel foam vehicle for delivery of topical corticosteroids. J. Am. Acad. Dermatol. 2005, 53, 26–38. [Google Scholar] [CrossRef]
- Ricciatti-Sibbald, D.; Sibbald, R.G. Dermatologic vehicles. Clin. Dermatol. 1989, 7, 11–24. [Google Scholar] [CrossRef]
- Van De Kerkhof, P.C.M.; Kragballe, K.; Segaert, S.; Lebwohl, M. Factors impacting the combination of topical corticosteroid therapies for psoriasis: Perspectives from the International Psoriasis Council. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 1130–1139. [Google Scholar] [CrossRef] [PubMed]
- Draelos, Z. Differences in cutaneous irritation of five commonly used topical products. J. Drugs. Dermatol. 2016, 15, 870–873. [Google Scholar]
- Surber, C.; Smith, E.W. The mystical effects of dermatological vehicles. Dermatology 2005, 210, 157–168. [Google Scholar] [CrossRef]
- Berth-Jones, J. Principles of Topical Therapy. Chapter 18. In Rook’s Textbook of Dermatology, 9th ed.; Griffiths, C., Barker, J., Bleiker, T.O., Chalmers, R., Creamer, D.J., Eds.; Wiley & Sons: London, UK, 2016; Volume 4, pp. 1–51. [Google Scholar]
- Rougier, A.; Lotte, C.; Corcuff, T.P. Relationship between skin permeability and cornecyte size according to anatomic site, age and sex in man. J. Soc. Cosmet. Chem. 1988, 39, 15–21. [Google Scholar]
- Kovács, A.; Péter-Héderi, D.; Perei, K.; Budai-Szucs, M.; Léber, A.; Gácsi, A.; Csányi, E.; Berkó, S. Effects of formulation excipients on skin barrier function in creams used in pediatric care. Pharmaceutics 2020, 12, 729. [Google Scholar] [CrossRef] [PubMed]
- Kuehl, B.; Shear, N.H. The evolution of topical formulations in psoriasis. Skin. Ther. Lett. 2018, 23, 5–9. [Google Scholar]
- Reich, K.; Daudén, E. Treatment adherence: A hurdle for real-life effectiveness in psoriasis? J. Eur. Acad. Dermatol. Venereol. 2014, 28, 1–3. [Google Scholar] [CrossRef]
- Daudén, E.; Bewley, A.; Lambert, J.; Girolomoni, G.; Cambazard, F.; Reich, K. Expert recommendations: The use of the fixed combination calcipotriol and betamethasone dipropionate gel for the topical treatment of psoriasis. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.K.; Rehmus, W.E.; Kimball, A.B. Determining the relative importance of patient motivations for nonadherence to topical corticosteroid therapy in psoriasis. J. Am. Acad. Dermatol. 2006, 55, 607–613. [Google Scholar] [CrossRef]
- Bewley, A.; Page, B. Maximizing patient adherence for optimal outcomes in psoriasis. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 9–14. [Google Scholar] [CrossRef]
- Stacey, S.K.; McEleney, M. Topical corticosteroids: Choice and application. Am. Fam. Physician. 2021, 103, 337–343. [Google Scholar]
- Brown, S.J. Atopic eczema. Clin. Med. 2016, 16, 66–69. [Google Scholar] [CrossRef] [Green Version]
- Purnamawati, S.; Indrastuti, N.; Danarti, R.; Saefudin, T. The role of moisturizers in addressing various kinds of dermatitis: A Review. Clin. Med. Res. 2017, 15, 75–87. [Google Scholar] [CrossRef] [Green Version]
- Jemec, G.B.; Na, R. Hydration and plasticity following long-term use of a moisturizer: A single-blind study. Acta Derm. Venereol. 2002, 82, 322–324. [Google Scholar] [CrossRef] [PubMed]
- Lodén, M. Role of topical emollients and moisturizers in the treatment of dry skin barrier disorders. Am. J. Clin. Dermatol. 2003, 4, 771–788. [Google Scholar] [CrossRef]
- Draelos, Z.D. Therapeutic moisturizers. Dermatol. Clin. 2000, 18, 597–607. [Google Scholar] [CrossRef]
- Lodén, M. Effect of moisturizers on epidermal barrier function. Clin. Dermatol. 2012, 30, 286–296. [Google Scholar] [CrossRef]
- Nolan, K.; Marmur, E. Moisturizers: Reality and the skin benefits. Dermatol. Ther. 2012, 25, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Sethi, A.; Kaur, T.; Malhotra, S.K.; Gambhir, M.L. Moisturizers: The slippery road. Indian J. Dermatol. 2016, 61, 279–287. [Google Scholar] [CrossRef]
- Rawlings, A.V.; Canestrari, D.A.; Dobkowski, B. Moisturizer technology versus clinical performance. Dermatol. Ther. 2004, 17, 49–56. [Google Scholar] [CrossRef]
- Spada, F.; Barnes, T.M.; Greive, K.A. Skin hydration is significantly increased by a cream formulated to mimic the skin’s own natural moisturizing systems. Clin. Cosmet. Investig. Dermatol. 2018, 11, 491–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caussin, J.; Rozema, E.; Gooris, G.S.; Wiechers, J.W.; Pavel, S.; Bouwstra, J.A. Hydrophilic and lipophilic moisturisers have similar penetration profiles but different effects on SC water distribution in vivo. Exp. Dermatol. 2009, 18, 954–961. [Google Scholar] [CrossRef]
- Chang, R.-K.; Raw, A.; Lionberger, R.; Yu, L. Generic development of topical dermatologic products: Formulation development, process development, and testing of topical dermatologic products. AAPS J. 2013, 15, 41–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kraft, J.N.; Lynde, C.W. Moisturizers: What they are and a practical approach to product selection. Skin Ther. Lett. 2005, 10, 1–8. [Google Scholar]
- Lodén, M. The clinical benefit of moisturizers. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 672–688. [Google Scholar] [CrossRef]
- Downie, J.B. Understanding moisturizers and their clinical benefits. Pract. Dermatol. Pediatr. 2010, 19–22. [Google Scholar]
- Levin, J.; Miller, R. A guide to the ingredients and potential benefits of over-the-counter cleansers and moisturizers for rosacea patients. J. Clin. Aesthet. Dermatol. 2011, 4, 31–49. [Google Scholar] [PubMed]
- Wiechers, J.M.; Barlow, T. Skin moisturisation and elasticity originate from at least two different mechanisms. Int. J. Cosmet. Sci. 1999, 21, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Lynde, C.W. Moisturizers: What they are and how they work. Skin Ther. Lett. 2001, 6, 3–5. [Google Scholar]
- Spada, F.; Harrison, I.P.; Barnes, T.M.; Greive, K.A.; Daniels, D.; Townley, J.P.; Mostafa, N.; Fong, A.T.; Tong, P.L.; Shumack, S. A daily regimen of a ceramide-dominant moisturizing cream and cleanser restores the skin permeability barrier in adults with moderate eczema: A randomized trial. Dermatol. Ther. 2021, 34, e14970. [Google Scholar] [CrossRef]
- Zeichner, J.A.; del Rosso, J.Q. Multivesicular emulsion ceramide-containing moisturizers: An evaluation of their role in the management of common skin disorders. J. Clin. Aesthet. Dermatol. 2016, 9, 26–32. [Google Scholar] [PubMed]
- Escudero-Góngora, M.M.; Fernández-Peñas, P. Nicotinamide: New indications in dermatology. Actas Dermosifiliogr. 2016, 107, 777–778. [Google Scholar] [CrossRef] [PubMed]
- Al-Niaimi, F.; Chiang, N.Y.Z. Topical vitamin C and the skin: Mechanisms of action and clinical applications. J. Clin. Aesthet. Dermatol. 2017, 10, 14–17. [Google Scholar]
- Perrelli, A.; Goitre, L.; Salzano, A.M.; Moglia, A.; Scaloni, A.; Retta, S.F. Biological activities, health benefits, and therapeutic properties of avenanthramides: From skin protection to prevention and treatment of cerebrovascular diseases. Oxid. Med. Cell. Longev. 2018, 2018, 6015351. [Google Scholar] [CrossRef]
- Hon, K.L.; Leung, A.K.C.; Barankin, B. Barrier repair therapy in atopic dermatitis: An overview. Am. J. Clin. Dermatol. 2013, 14, 289–399. [Google Scholar] [CrossRef]
- Bikowski, J. The use of therapeutic moisturizers in various dermatologic disorders. Cutis 2001, 68, 3–11. [Google Scholar]
- Draelos, Z.D. The science behind skin care: Moisturizers. J. Cosmet. Dermatol. 2018, 17, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Idson, B. Moisturizers, Emollients, And Bath Oils. In Principles of Cosmetics for the Dermatologist, 1st ed.; Frost, P., Horwitz, S.N., Eds.; Mosby Company: St. Louis, MO, USA, 1982; pp. 37–44. [Google Scholar]
- Schmitt, W.H. Skin-Care Products. In Chemistry and Technology of the Cosmetics and Toiletries Industry, 2nd ed.; Williams, D.F., Schmitt, W.H., Eds.; Blackie Academic & Professional: London, UK, 1992; pp. 104–148. [Google Scholar]
- De Villiers, M.M. Surfactants and Emulsifying Agents. Chapter 20. In A Practical Guide to Contemporary Pharmacy Practice, 3rd ed.; Thompson, J.E., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2009; pp. 251–256. [Google Scholar]
- Moser, K.; Kriwet, K.; Kalia, Y.N.; Guy, R.H. Enhanced skin permeation of a lipophilic drug using supersaturated formulations. J. Control. Release 2001, 73, 245–253. [Google Scholar] [CrossRef]
- Haque, T.; Talukder, M.M.U. Chemical enhancer: A simplistic way to modulate barrier function of the stratum corneum. Adv. Pharm. Bull. 2018, 8, 169–179. [Google Scholar] [CrossRef] [Green Version]
- Williams, A.C.; Barry, B.W. Penetration enhancers. Adv. Drug. Deliv. Rev. 2012, 64, 128–137. [Google Scholar] [CrossRef]
- Lessmann, H.; Schnuch, A.; Geier, J.; Uter, W. Skin-sensitizing and irritant properties of propylene glycol. Contact Dermat. 2005, 53, 247–259. [Google Scholar] [CrossRef]
- Jain, A.; Panchagnula, R. Transdermal drug delivery of tricyclic antidepressants: Effect of fatty acids. Methods. Find. Exp. Clin. Pharmacol. 2003, 25, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Jakasa, I.; Verberk, M.M.; Bunge, A.L.; Kruse, J.; Kezic, S. Increased permeability for polyethylene glycols through skin compromised by sodium lauryl sulphate. Exp. Dermatol. 2006, 15, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Raj, R.K.; Chandrul, K.K. Regulatory requirements for cosmetics in relation with regulatory authorities in India against US, Europe, Australia and Asean countries. Int. J. Pharm. Res. Health. Sci. 2016, 4, 1332–1341. [Google Scholar] [CrossRef]
- Draelos, Z.D. Cosmetic moisturizers, topical drug vehicles. Dermatol. Times 2021, 42, 22. [Google Scholar]
- Qualification of Excipients for Use in Pharmaceuticals. Available online: https://ipecamericas.org (accessed on 13 October 2021).
| Vehicle | Definition | Areas of Application | Advantages | Disadvantages |
|---|---|---|---|---|
| Ointment | A suspension or emulsion semisolid formulation that contains <20% water and volatiles and >50% of hydrocarbons, waxes, or polyethylene glycols | Glabrous areas such as palms and soles Skin with short or sparse hair Drier areas of the body, such as the trunk and extremities Thickened and lichenified skin | Occlusive effect increases skin hydration, penetration and efficacy of active drug Emollient properties soothe and soften the skin Water resistance increases contact time Fewer preservatives reduce the risk of sensitization | Greasy which is less cosmetically appealing Difficult to wash off More difficult to spread than creams, lotions and solutions |
| Cream | An emulsion semisolid formulation that contains >20% water and volatiles and/or <50% of hydrocarbons, waxes, or polyethylene glycols | Dry and weepy/exudative skin conditions, especially if significant exudate Infected and exudative plaques Flexural and genital areas Can be used on all areas of the body including face | Moistening properties Emollient properties soothe and soften the skin More spreadable and less greasy than ointments resulting in increased patient compliance | Less occlusive and hydrating than ointments which decreases percutaneous drug absorption |
| Gel | A semisolid formulation that contains a gelling agent to provide stiffness to a solution or colloidal dispersion | Hairy areas, such as the scalp Oily areas, such as the face | Dries as a greaseless non-occlusive film Cosmetically elegant Easy to apply and wash off, especially on hairy areas Non-greasy Provides cooling sensation | No occlusive effects and minimal skin hydration Not an emollient Perspiration removes the gel Alcohol-based gels may cause stinging |
| Lotion | An emulsion liquid formulation with >50% water and volatiles | Exudative dermatoses Hairy areas, such as the scalp | Provide cooling effect as the aqueous phase evaporates Easy to apply to hairy areas Spreads rapidly | Less occlusive and hydrating than ointments or creams |
| Solution | A clear, homogeneous liquid formulation | Hairy areas, including the scalp Non-hairy areas | Easy to spread over various areas of the body, including the scalp Very simple to produce | Messy application No occlusive effects and minimal skin hydration Not an emollient Alcohol-based products can cause stinging, dryness, or skin irritation |
| Foam | Multiphase suspension containing a propellant stored under pressure and forming a foam upon release that quickly breaks down on the skin | Hairy areas of the body, such as the scalp Oily areas, such as the face Non-hairy areas Inflamed or sensitive areas, as application requires minimal mechanical sheering force to disperse the medication | Do not contain any fragrances, formaldehyde, or non-formaldehyde preservatives Low density and easy to apply and spread on skin surface Increased skin absorption Low-residue cosmetic elegance | No occlusive effects and minimal skin hydration Depending on the vehicle base, may result in stinging or burning to excoriated skin |
| Spray | A solution formulation with pressurized propellant | Can be used on all skin areas, but caution in skin folds where absorption is higher | Treats large areas of affected skin (up to 15–20% body surface area) Easier to apply to difficult to reach areas Easily applied in a thin layer with little waste and good absorption Cooling sensation upon application No risk of contamination of the unused portion | May be associated with stinging and burning upon application |
| Moisturizer Type | Examples | Effects on the Skin |
|---|---|---|
| Humectants | Glycerin, gelatin, propylene glycol, butylene glycol, panthenol, sorbitol, urea, hyaluronic acid, glycolic acid, lactic acid, sodium pyrrolidine carboxylic acid | Mostly low molecular weight substances which attract and hold water in the skin May cause water to evaporate into the environment and therefore need to be used with an occludent |
| Emollients | Cholesterol, squalene, linoleic acid, stearic acid, oleic acid, fatty alcohols | Saturated and unsaturated variable length hydrocarbons which improve the ‘feel’ of the skin by filling the spaces in between corneocytes Provide what has been termed ‘skin slip’ or lubricity, imparting a sense of softness and plasticity to the skin Improve the overall appearance and texture of the skin Often used in combination with emulsifiers |
| Occludents | White soft paraffin/petrolatum, beeswax, mineral oil, dimethicone, lanolin, carnauba wax, cetyl alcohol, caprylic/capric triglyceride | Oils and waxes which form an inert layer on the skin and physically block transepidermal water loss (TEWL) Some occludents enter the intercellular space and interact with the stratum corneum lipids, reinforcing the skin barrier |
| Excipient | Examples | Role in Topical Vehicle Formulation | Effects on the Skin |
|---|---|---|---|
| Stiffening agents (lipids and hydrocarbons) | White soft paraffin/petrolatum, liquid paraffin, lanolin, beeswax, carnauba wax, cetyl alcohol, isohexadecane, | Main structure forming materials for semisolid formulations | Occlusive and skin protecting Hydrating Soothing and softening |
| Thickening/ gelling agents | Carbomer, cetyl alcohol, stearic acid, carnauba wax, hydroxyethyl cellulose, guar gum, xanthan gum, gelatin, magnesium aluminium silicate, silica, bentonite, cetyl palmitate, ammonium acryloyldimethyltaurate | Main structure-forming ingredients for gels and viscosity-enhancing ingredients for creams and lotions | - |
| Silicones | Dimethicone, cyclomethicone | Lubricant and film-forming ingredient | Occlusive and skin protecting |
| Humectants (polyols) | Glycerol, sorbitol, propylene glycol, polyethylene glycol, 1,2,6-hexanetriol, triacetin | Promotes the retention of water in the vehicle and the skin | Moisturizing and skin protecting Skin barrier stabilizing |
| Emulsifiers/ solubilizers | Glycerol monostearate, cetostearyl alcohol, cetyl palmitate, sorbitan monostearate, polysorbate 20, polysorbate 80, polysorbate 60, poloxamer, emulsifying wax, sorbitan monooleate, sodium lauryl sulfate, propylene glycol monostearate, diethylene glycol monoethyl ether | Used to reduce the interfacial tension to stabilize emulsions and to improve the wetting and solubility of hydrophobic ingredients | Skin conditioning Harsh surfactants may have deleterious effects by dissolving lipids and irritating the skin |
| Solvents | Purified water, propylene glycol, hexylene glycol, oleyl alcohol, mineral oil/liquid paraffin, propylene carbonate | Used to dissolve or disperse the active drug | Various effects, as described for other excipients |
| Penetration enhancers | Propylene glycol, oleic acid, isopropyl myristate, ethanol, polyethylene glycol | Increases permeation by promoting the diffusion, partitioning, or the solubility of an active drug through the stratum corneum | Disrupts the skin barrier to enhance drug delivery May be irritating at high concentrations |
| Chelating agents | Ethylene diamine tetraacetate |
Binds metal ions to minimize metal-catalyzed degradation and to enhance the preservative effect | - |
|
Acidifying/alkalizing/ buffering agents | Citric acid, lactic acid, phosphoric acid, sodium hydroxide | Maintains optimum pH for drug delivery | Products with high buffering capacity can alter skin surface pH |
| Antioxidants | Butylated hydroxyanisole, butylated hydroxytoluene, tocopherol, ascorbyl palmitate, sodium ascorbyl phosphate | To minimize oxidative deterioration and help stabilize the active drug | May reduce barrier disruption and inflammation due to oxidative stress within the stratum corneum |
| Preservatives |
Benzoic acid, propyl paraben, methyl paraben, imidurea, sorbic acid, potassium sorbate, benzalkonium chloride, phenyl mercuric acetate, chlorobutanol, phenoxyethanol | Prevents microbial growth and contamination of the formulation | Added to protect the formulation rather than to exert an antiseptic effect on the skin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barnes, T.M.; Mijaljica, D.; Townley, J.P.; Spada, F.; Harrison, I.P. Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison. Pharmaceutics 2021, 13, 2012. https://doi.org/10.3390/pharmaceutics13122012
Barnes TM, Mijaljica D, Townley JP, Spada F, Harrison IP. Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison. Pharmaceutics. 2021; 13(12):2012. https://doi.org/10.3390/pharmaceutics13122012
Chicago/Turabian StyleBarnes, Tanya M., Dalibor Mijaljica, Joshua P. Townley, Fabrizio Spada, and Ian P. Harrison. 2021. "Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison" Pharmaceutics 13, no. 12: 2012. https://doi.org/10.3390/pharmaceutics13122012
APA StyleBarnes, T. M., Mijaljica, D., Townley, J. P., Spada, F., & Harrison, I. P. (2021). Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison. Pharmaceutics, 13(12), 2012. https://doi.org/10.3390/pharmaceutics13122012