Antidepressants and Circadian Rhythm: Exploring Their Bidirectional Interaction for the Treatment of Depression
Abstract
1. Introduction
2. Pharmacokinetics of Antidepressants
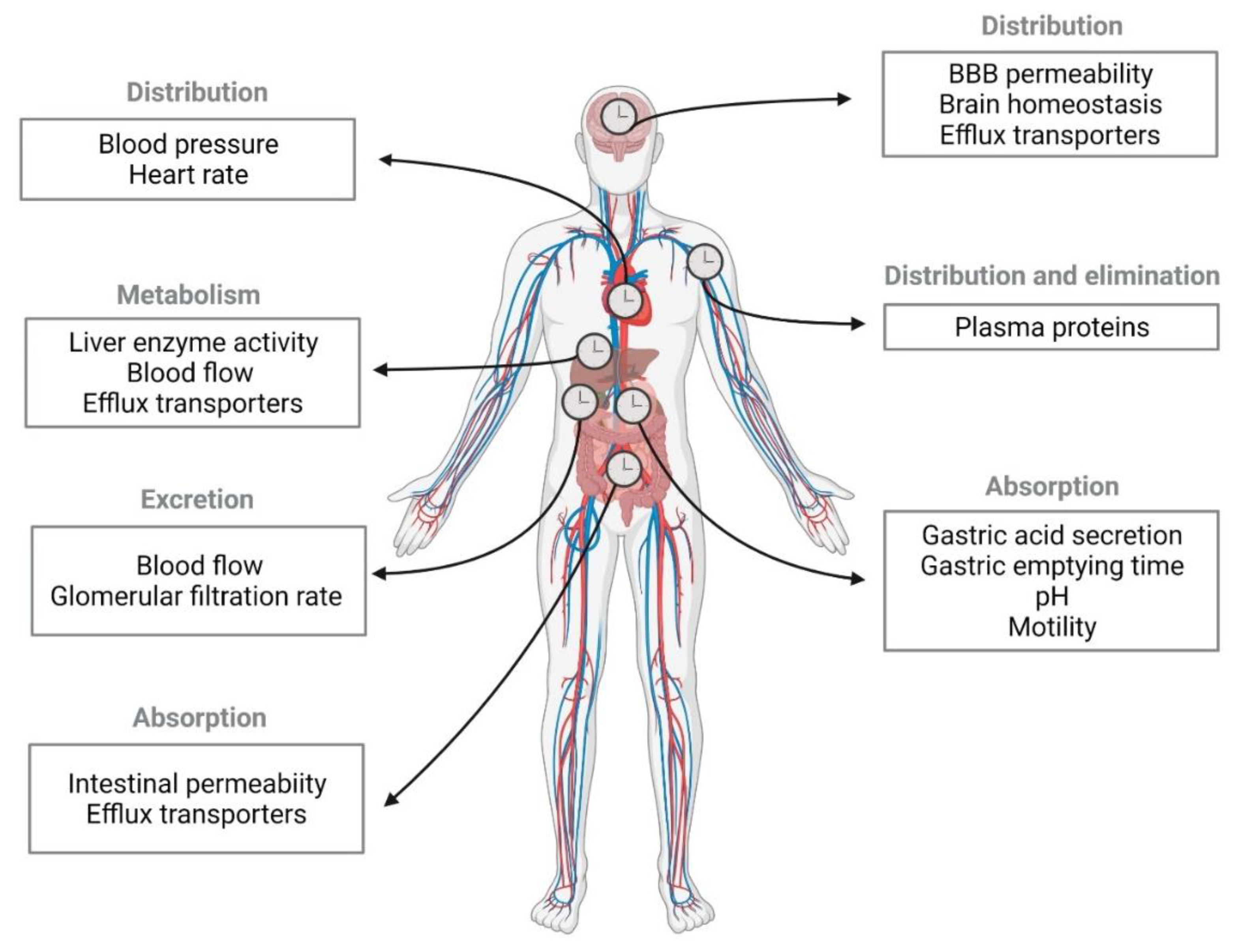
2.1. Circadian Rhythm Effect on Pharmacokinetic Stages
2.1.1. Absorption
2.1.2. Distribution
2.1.3. Metabolism and Excretion
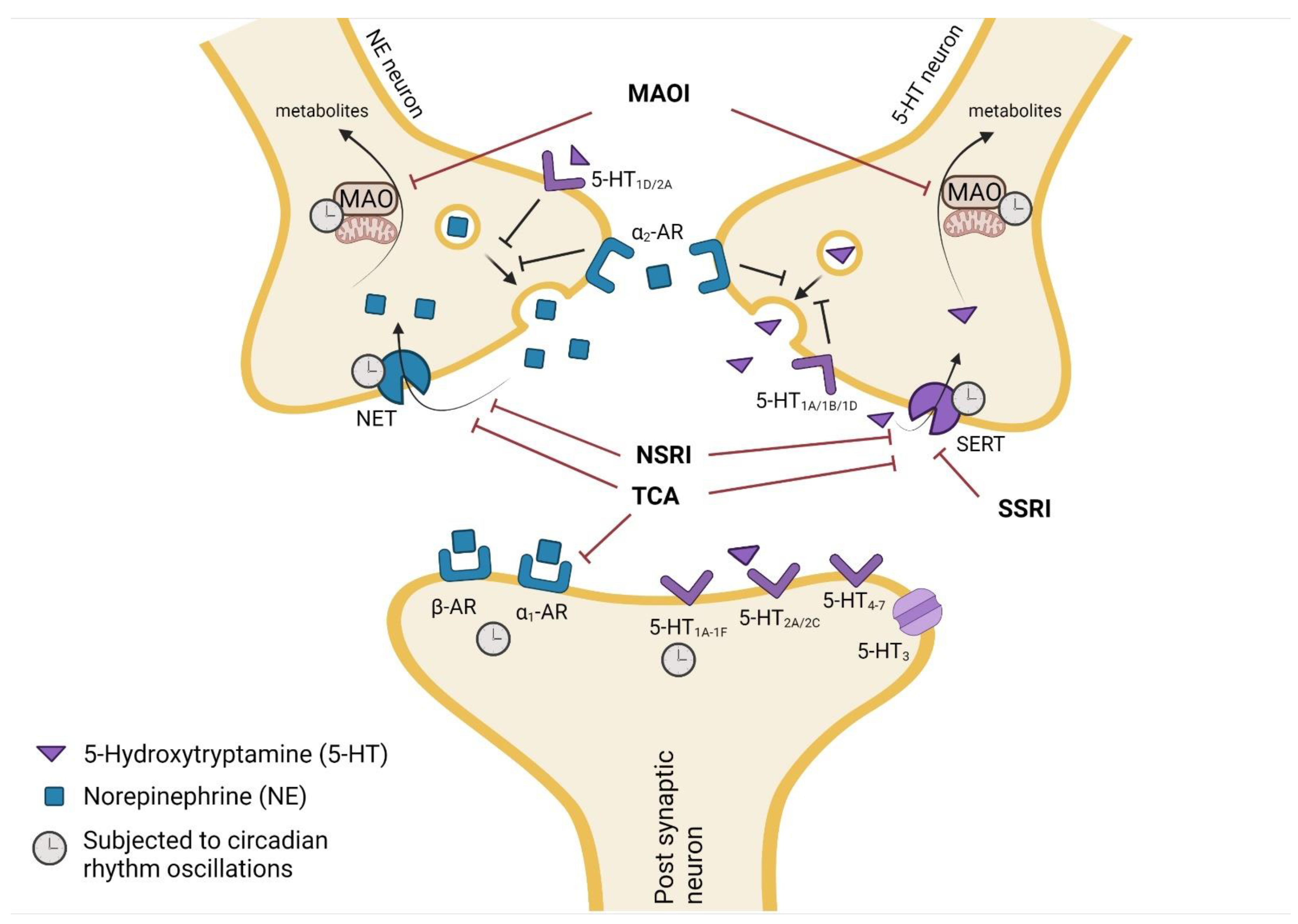
3. Pharmacodynamics of Antidepressants
3.1. Circadian Rhythm Effect on Antidepressant Drug Targets
3.1.1. Animal Studies
3.1.2. Human Data
3.2. Antidepressant Effects on Circadian Rhythms
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brown, G.M. Light, melatonin and the sleep-wake cycle. J. Psychiatr. Neurosci. 1994, 19, 345–353. [Google Scholar]
- Tordjman, S.; Chokron, S.; Delorme, R.; Charrier, A.; Bellissant, E.; Jaafari, N.; Fougerou, C. Melatonin: Pharmacology, functions and therapeutic benefits. Curr. Neuropharmacol. 2017, 15, 434–443. [Google Scholar] [CrossRef]
- Brodsky, V.Y. Circahoralian (ultradian) metabolic rhythms. Biochemistry 2014, 79, 483–495. [Google Scholar] [CrossRef]
- Prendergast, B.J.; Nelson, R.J.; Zucker, I. 19—Mammalian seasonal rhythms: Behavior and neuroendocrine substrates. In Hormones, Brain and Behavior; Pfaff, D.W., Arnold, A.P., Fahrbach, S.E., Etgen, A.M., Rubin, R.T., Eds.; Academic Press: Cambridge, MA, USA, 2002; pp. 93–156. [Google Scholar]
- Pilorz, V.; Helfrich-Forster, C.; Oster, H. The role of the circadian clock system in physiology. Pflüg. Arch. 2018, 470, 227–239. [Google Scholar] [CrossRef]
- Robinson, I.; Reddy, A.B. Molecular mechanisms of the circadian clockwork in mammals. FEBS Lett. 2014, 588, 2477–2483. [Google Scholar] [CrossRef]
- Preitner, N.; Damiola, F.; Molina, L.-L.; Zakany, J.; Duboule, D.; Albrecht, U.; Schibler, U. the orphan nuclear receptor REV-ERBα controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell 2002, 110, 251–260. [Google Scholar] [CrossRef]
- Honma, S. The mammalian circadian system: A hierarchical multi-oscillator structure for generating circadian rhythm. J. Physiol. Sci. 2018, 68, 207–219. [Google Scholar] [CrossRef]
- Buijs, R.M.; Kalsbeek, A. Hypothalamic integration of central and peripheral clocks. Nat. Rev. Neurosci. 2001, 2, 521–526. [Google Scholar] [CrossRef]
- Masri, S.; Sassone-Corsi, P. The emerging link between cancer, metabolism, and circadian rhythms. Nat. Med. 2018, 24, 1795–1803. [Google Scholar] [CrossRef]
- Reddy, A.B.; O’Neill, J.S. Healthy clocks, healthy body, healthy mind. Trends Cell. Biol. 2010, 20, 36–44. [Google Scholar] [CrossRef]
- WHO. Depression and Other Common Mental Disorders: Global Health Estimates; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Santomauro, D.F.; Mantilla Herrera, A.M.; Shadid, J.; Zheng, P.; Ashbaugh, C.; Pigott, D.M.; Abbafati, C.; Adolph, C.; Amlag, J.O.; Aravkin, A.Y.; et al. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet 2021, 398, 1700–1712. [Google Scholar] [CrossRef]
- Charrier, A.; Olliac, B.; Roubertoux, P.; Tordjman, S. Clock genes and altered sleep-wake rhythms: Their role in the development of psychiatric disorders. Int. J. Mol. Sci. 2017, 18, 938. [Google Scholar] [CrossRef]
- Oglodek, E.A.; Just, M.J.; Szromek, A.R.; Araszkiewicz, A. Melatonin and neurotrophins NT-3, BDNF, NGF in patients with varying levels of depression severity. Pharm. Rep. 2016, 68, 945–951. [Google Scholar] [CrossRef]
- Sundberg, I.; Ramklint, M.; Stridsberg, M.; Papadopoulos, F.C.; Ekselius, L.; Cunningham, J.L. Salivary Melatonin in relation to depressive symptom severity in young adults. PLoS ONE 2016, 11, e0152814. [Google Scholar] [CrossRef]
- Hidalgo, M.P.; Caumo, W.; Dantas, G.; Franco, D.G.; Torres, I.L.; Pezzi, J.; Elisabetsky, E.; Detanico, B.C.; Piato, A.; Markus, R.P. 6-Sulfatoxymelatonin as a predictor of clinical outcome in depressive patients. Hum. Psychopharmacol 2011, 26, 252–257. [Google Scholar] [CrossRef]
- Jury Freitas, J.; Bertuol Xavier, N.; Comiran Tonon, A.; Carissimi, A.; Timm Pizutti, L.; Vieira Ilgenfritz, C.A.; Pekelmann Markus, R.; Paz Hidalgo, M. 6-Sulfatoxymelatonin predicts treatment response to fluoxetine in major depressive disorder. Adv. Psychopharmacol. 2019, 9, 1–6. [Google Scholar] [CrossRef]
- Bhagwagar, Z.; Hafizi, S.; Cowen, P.J. Increased salivary cortisol after waking in depression. Psychopharmacology 2005, 182, 54–57. [Google Scholar] [CrossRef]
- Keller, J.; Flores, B.; Gomez, R.G.; Solvason, H.B.; Kenna, H.; Williams, G.H.; Schatzberg, A.F. Cortisol circadian rhythm alterations in psychotic major depression. Biol. Psychiatry 2006, 60, 275–281. [Google Scholar] [CrossRef]
- Moffoot, A.P.R.; O’Carroll, R.E.; Bennie, J.; Carroll, S.; Dick, H.; Ebmeier, K.P.; Goodwin, G.M. Diurnal variation of mood and neuropsychological function in major depression with melancholia. J. Affect. Disord. 1994, 32, 257–269. [Google Scholar] [CrossRef]
- Adan, A.; Archer, S.N.; Hidalgo, M.P.; Di Milia, L.; Natale, V.; Randler, C. Circadian typology: A comprehensive review. Chronobiol. Int. 2012, 29, 1153–1175. [Google Scholar] [CrossRef]
- Au, J.; Reece, J. The relationship between chronotype and depressive symptoms: A meta-analysis. J. Affect. Disord. 2017, 218, 93–104. [Google Scholar] [CrossRef]
- McGlashan, E.M.; Drummond, S.P.A.; Cain, S.W. Evening types demonstrate reduced SSRI treatment efficacy. Chronobiol. Int. 2018, 35, 1175–1178. [Google Scholar] [CrossRef]
- Hasler, G. Pathophysiology of depression: Do we have any solid evidence of interest to clinicians? World Psychiatry 2010, 9, 155–161. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, J.M.; Bies, R.R.; Shelton, R.C. Chapter 15: Drug therapy of depression and anxiety disorders. In Goodman & Gilman’s: The Pharmacological Basis of Therapeutics, 13th ed.; Brunton, L.L., Hilal-Dandan, R., Knollmann, B.C., Eds.; McGraw Hill: New York, NY, USA, 2017. [Google Scholar]
- Bauer, M.; Severus, E.; Moller, H.J.; Young, A.H.; Disorders, W.T.F.o.U.D. Pharmacological treatment of unipolar depressive disorders: Summary of WFSBP guidelines. Int. J. Psychiatry Clin. Pract. 2017, 21, 166–176. [Google Scholar] [CrossRef]
- Alvano, S.A.; Zieher, L.M. An updated classification of antidepressants: A proposal to simplify treatment. Pers. Med. Psychiatry 2020, 19-20, 100042. [Google Scholar] [CrossRef]
- Uchida, H.; Fleischhacker, W.; Juckel, G.; Grunder, G.; Bauer, M. Naming for psychotropic drugs: Dilemma and challenge. Pharmacopsychiatry 2017, 50, 1–2. [Google Scholar] [CrossRef]
- Montgomery, S.A. Why do we need new and better antidepressants? Int. Clin. Psychopharmacol. 2006, 21, S1–S10. [Google Scholar] [CrossRef] [PubMed]
- Uher, R.; Farmer, A.; Henigsberg, N.; Rietschel, M.; Mors, O.; Maier, W.; Kozel, D.; Hauser, J.; Souery, D.; Placentino, A.; et al. Adverse reactions to antidepressants. Br. J. Psychiatry 2009, 195, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.; Posternak, M.; Alpert, J.E. Toward achieving optimal response: Understanding and managing antidepressant side effects. Dialogues Clin. Neurosci. 2008, 10, 409–418. [Google Scholar] [PubMed]
- Martiny, K.; Refsgaard, E.; Lund, V.; Lunde, M.; Thougaard, B.; Lindberg, L.; Bech, P. Maintained superiority of chronotherapeutics vs. exercise in a 20-week randomized follow-up trial in major depression. Acta. Psychiatr. Scand. 2015, 131, 446–457. [Google Scholar] [CrossRef]
- Bicker, J.; Alves, G.; Falcao, A.; Fortuna, A. Timing in drug absorption and disposition: The past, present, and future of chronopharmacokinetics. Br. J. Pharm. 2020, 177, 2215–2239. [Google Scholar] [CrossRef]
- Keller, F.; Hann, A. Clinical Pharmacodynamics: Principles of drug response and alterations in kidney disease. Clin. J. Am. Soc. Nephrol. 2018, 13, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Kawai, H.; Kodaira, N.; Tanaka, C.; Ishibashi, T.; Kudo, N.; Kawashima, Y.; Mitsumoto, A. Time of administration of acute or chronic doses of imipramine affects its antidepressant action in rats. J. Circadian Rhythm. 2018, 16, 5. [Google Scholar] [CrossRef]
- Nakano, S.; Hollister, L.E. Chronopharmacology of amitriptyline. Clin. Pharm. 1983, 33, 453–459. [Google Scholar]
- Erkekoglu, P.; Baydar, T. Chronopharmacodynamics of drugs in toxicological aspects: A short review for clinical pharmacists and pharmacy practitioners. J. Res. Pharm. Pract. 2012, 1, 41–47. [Google Scholar] [CrossRef]
- Liu, J.; Li, H.; Xu, S.; Xu, Y.; Liu, C. Circadian Clock Gene Expression and Drug/Toxicant Interactions as Novel Targets of Chronopharmacology and Chronotoxicology; InTechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Ruben, M.D.; Smith, D.F.; FitzGerald, G.A.; Hogenesch, J.B. Dosing time matters. Science 2019, 365, 547–549. [Google Scholar] [CrossRef]
- Gaspar, L.S.; Alvaro, A.R.; Carmo-Silva, S.; Mendes, A.F.; Relogio, A.; Cavadas, C. The importance of determining circadian parameters in pharmacological studies. Br. J. Pharm. 2019, 176, 2827–2847. [Google Scholar] [CrossRef]
- Dallmann, R.; Brown, S.A.; Gachon, F. Chronopharmacology: New insights and therapeutic implications. Annu. Rev. Pharm. Toxicol. 2014, 54, 339–361. [Google Scholar] [CrossRef]
- Stephan, F.K. The “other” Circadian system: Food as a Zeitgeber. J. Biol. Rhythm. 2002, 17, 284–292. [Google Scholar] [CrossRef]
- Koziolek, M.; Alcaro, S.; Augustijns, P.; Basit, A.W.; Grimm, M.; Hens, B.; Hoad, C.L.; Jedamzik, P.; Madla, C.M.; Maliepaard, M.; et al. The mechanisms of pharmacokinetic food-drug interactions—A perspective from the UNGAP group. Eur. J. Pharm. Sci. 2019, 134, 31–59. [Google Scholar] [CrossRef]
- Haenisch, B.; Hiemke, C.; Bonisch, H. Inhibitory potencies of trimipramine and its main metabolites at human monoamine and organic cation transporters. Psychopharmacology 2011, 217, 289–295. [Google Scholar] [CrossRef]
- Bougerolle, A.M.; Chabard, J.L.; Jbilou, M.; Dordain, G.; Eschalier, A.; Aumaitre, O.; Gaillol, J.; Piron, J.J.; Petit, J.; Berger, J.A. Chronopharmacokinetic and bioequivalence studies of two formulations of trimipramine after oral administration in man. Eur. J. Drug. Metab. Pharm. 1989, 4, 139–144. [Google Scholar] [CrossRef]
- Ronfeld, R.A.; Wilner, K.D.; Baris, B.A. Sertraline: Chronopharmacokinetics and the effect of coadministration with food. Clin. Pharm. 1997, 32, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Rutkowska, A.; Piekoszewski, W.; Brandys, J. Chronopharmacokinetics of Amitriptyline in Rats. Biopharm. Drug Dispos. 1999, 20, 117–124. [Google Scholar] [CrossRef]
- Lemmer, B.; Holle, L. Chronopharmacokinetics of imipramine and desipramine in rat forebrain and plasma after single and chronic treatment with imipramine. Chronobiol. Int. 1991, 8, 176–185. [Google Scholar] [CrossRef]
- Nakano, S.; Hollister, L.E. No circadian effect on nortriptyline kinetics in man. Clin. Pharm. 1978, 23, 199–203. [Google Scholar] [CrossRef]
- Oh-oka, K.; Kono, H.; Ishimaru, K.; Miyake, K.; Kubota, T.; Ogawa, H.; Okumura, K.; Shibata, S.; Nakao, A. Expressions of tight junction proteins Occludin and Claudin-1 are under the circadian control in the mouse large intestine: Implications in intestinal permeability and susceptibility to colitis. PLoS ONE 2014, 9, e98016. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, K.; Kitagawa, E.; Wada, M.; Haraguchi, A.; Orihara, K.; Tahara, Y.; Nakao, A.; Shibata, S. Antigen exposure in the late light period induces severe symptoms of food allergy in an OVA-allergic mouse model. Sci. Rep. 2015, 5, 14424. [Google Scholar] [CrossRef]
- Tran, L.; Jochum, S.B.; Shaikh, M.; Wilber, S.; Zhang, L.; Hayden, D.M.; Forsyth, C.B.; Voigt, R.M.; Bishehsari, F.; Keshavarzian, A.; et al. Circadian misalignment by environmental light/dark shifting causes circadian disruption in colon. PLoS ONE 2021, 16, e0251604. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Kubera, M.; Leunis, J.C. The gut-brain barrier in major depression: Intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuroendocr. Lett. 2008, 29, 117–124. [Google Scholar]
- Uhr, M.; Grauer, M.T. abcb1ab P-glycoprotein is involved in the uptake of citalopram and trimipramine into the brain of mice. J. Psychiatr. Res. 2003, 37, 179–185. [Google Scholar] [CrossRef]
- Bundgaard, C.; Eneberg, E.; Sanchez, C. P-glycoprotein differentially affects escitalopram, levomilnacipran, vilazodone and vortioxetine transport at the mouse blood-brain barrier in vivo. Neuropharmacology 2016, 103, 104–111. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.E.; O’Connor, R.M.; Clarke, G.; Dinan, T.G.; Griffin, B.T.; Cryan, J.F. P-glycoprotein inhibition increases the brain distribution and antidepressant-like activity of escitalopram in rodents. Neuropsychopharmacology 2013, 38, 2209–2219. [Google Scholar] [CrossRef]
- Uhr, M.; Steckler, T.; Yassouridis, A.; Holsboer, F. Penetration of amitriptyline, but not of fluoxetine, into brain is enhanced in mice with blood-brain barrier deficiency due to Mdr1a P-glycoprotein gene disruption. Neuropsychopharmacology 2000, 22, 380–387. [Google Scholar] [CrossRef]
- Spieler, D.; Namendorf, C.; Namendorf, T.; Uhr, M. abcb1ab p-glycoprotein is involved in the uptake of the novel antidepressant vortioxetine into the brain of mice. J. Psychiatr. Res. 2019, 109, 48–51. [Google Scholar] [CrossRef]
- Feng, S.; Zheng, L.; Tang, S.; Gu, J.; Jiang, X.; Wang, L. In-vitro and in situ assessment of the efflux of five antidepressants by breast cancer resistance protein. J. Pharm. Pharm. 2019, 71, 1133–1141. [Google Scholar] [CrossRef]
- O’Leary, O.F.; Dinan, T.G.; Cryan, J.F. Faster, better, stronger: Towards new antidepressant therapeutic strategies. Eur. J. Pharm. 2015, 753, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Gachon, F.; Olela, F.F.; Schaad, O.; Descombes, P.; Schibler, U. The circadian PAR-domain basic leucine zipper transcription factors DBP, TEF, and HLF modulate basal and inducible xenobiotic detoxification. Cell. Metab. 2006, 4, 25–36. [Google Scholar] [CrossRef]
- Murakami, Y.; Higashi, Y.; Matsunaga, N.; Koyanagi, S.; Ohdo, S. Circadian clock-controlled intestinal expression of the multidrug-resistance gene mdr1a in mice. Gastroenterology 2008, 135, 1636–1644. [Google Scholar] [CrossRef]
- Ando, H.; Yanagihara, H.; Sugimoto, K.; Hayashi, Y.; Tsuruoka, S.; Takamura, T.; Kaneko, S.; Fujimura, A. Daily rhythms of P-glycoprotein expression in mice. Chronobiol. Int. 2005, 22, 655–665. [Google Scholar] [CrossRef]
- Stearns, A.T.; Balakrishnan, A.; Rhoads, D.B.; Ashley, S.W.; Tavakkolizadeh, A. Diurnal rhythmicity in the transcription of jejunal drug transporters. J. Pharm. Sci 2008, 108, 144–148. [Google Scholar] [CrossRef]
- Okyar, A.; Kumar, S.A.; Filipski, E.; Piccolo, E.; Ozturk, N.; Xandri-Monje, H.; Pala, Z.; Abraham, K.; Gomes, A.; Orman, M.N.; et al. Sex-, feeding-, and circadian time-dependency of P-glycoprotein expression and activity—implications for mechanistic pharmacokinetics modeling. Sci. Rep. 2019, 9, 10505. [Google Scholar] [CrossRef]
- Iwasaki, M.; Koyanagi, S.; Suzuki, N.; Katamune, C.; Matsunaga, N.; Watanabe, N.; Takahashi, M.; Izumi, T.; Ohdo, S. Circadian modulation in the intestinal absorption of P-glycoprotein substrates in monkeys. Mol. Pharm. 2015, 88, 29–37. [Google Scholar] [CrossRef]
- Abaut, A.Y.; Chevanne, F.; Le Corre, P. Oral bioavailability and intestinal secretion of amitriptyline: Role of P-glycoprotein? Int. J. Pharm. 2007, 330, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Koyanagi, S.; Hamdan, A.M.; Horiguchi, M.; Kusunose, N.; Okamoto, A.; Matsunaga, N.; Ohdo, S. cAMP-response element (CRE)-mediated transcription by activating transcription factor-4 (ATF4) is essential for circadian expression of the Period2 gene. J. Biol. Chem. 2011, 286, 32416–32423. [Google Scholar] [CrossRef] [PubMed]
- Hamdan, A.M.; Koyanagi, S.; Wada, E.; Kusunose, N.; Murakami, Y.; Matsunaga, N.; Ohdo, S. Intestinal expression of mouse Abcg2/breast cancer resistance protein (BCRP) gene is under control of circadian clock-activating transcription factor-4 pathway. J. Biol.Chem. 2012, 287, 17224–17231. [Google Scholar] [CrossRef]
- Chen, L.; Yang, G. Recent advances in circadian rhythms in cardiovascular system. Front. Pharm. 2015, 6, 71. [Google Scholar] [CrossRef] [PubMed]
- Lemmer, B.; Soloviev, M. Chronobiology and the implications for safety pharmacology. In Drug Discovery and Evaluation: Safety and Pharmacokinetic Assays; Vogel, H.G., Maas, J., Hock, F.J., Mayer, D., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; Volume 21. [Google Scholar]
- Douma, L.G.; Gumz, M.L. Circadian clock-mediated regulation of blood pressure. Free Radic. Biol. Med. 2018, 119, 108–114. [Google Scholar] [CrossRef]
- Scheving, L.E.; Pauly, J.E.; Tsai, T.H. Circadian fluctuation of plasma proteins of the rat. Am. J. Physiol. 1968, 215, 1096–1101. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Harten, J.v. Clinical pharmacokinetics of selective serotonin reuptake inhibitors. Clin. Pharm. 1993, 24, 203–220. [Google Scholar] [CrossRef] [PubMed]
- Borga, O.; Azarnoff, D.L.; Forshell, G.P.; Sjoqvist, F. Plasma protein binding of tricyclic anti-depressants in man. Biochem. Pharm. 1969, 18, 2135–2143. [Google Scholar] [CrossRef]
- Yost, R.L.; DeVane, C.L. Diurnal variation of α1-acid glycoprotein concentration in normal volunteers. J. Pharm. Sci. 1985, 74, 777–779. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Available online: https://go.drugbank.com/drugs/ (accessed on 25 January 2021).
- Nakazato, R.; Kawabe, K.; Yamada, D.; Ikeno, S.; Mieda, M.; Shimba, S.; Hinoi, E.; Yoneda, Y.; Takarada, T. Disruption of Bmal1 impairs blood-brain barrier integrity via pericyte dysfunction. J. Neurosci. 2017, 37, 10052–10062. [Google Scholar] [CrossRef]
- Myung, J.; Schmal, C.; Hong, S.; Tsukizawa, Y.; Rose, P.; Zhang, Y.; Holtzman, M.J.; De Schutter, E.; Herzel, H.; Bordyugov, G.; et al. The choroid plexus is an important circadian clock component. Nat. Commun. 2018, 9, 1062. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Kang, H.; Xu, Q.; Chen, M.J.; Liao, Y.; Thiyagarajan, M.; O’Donnell, J.; Christensen, D.J.; Nicholson, C.; Iliff, J.J.; et al. Sleep drives metabolite clearance from the adult brain. Science 2013, 342, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.L.; Lahens, N.F.; Yue, Z.; Arnold, D.M.; Pakstis, P.P.; Schwarz, J.E.; Sehgal, A. A circadian clock regulates efflux by the blood-brain barrier in mice and human cells. Nat. Commun. 2021, 12, 617. [Google Scholar] [CrossRef] [PubMed]
- Pulido, R.S.; Munji, R.N.; Chan, T.C.; Quirk, C.R.; Weiner, G.A.; Weger, B.D.; Rossi, M.J.; Elmsaouri, S.; Malfavon, M.; Deng, A.; et al. Neuronal activity regulates blood-brain barrier efflux transport through endothelial circadian genes. Neuron 2020, 108, 937–952. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.L.; Yue, Z.; Arnold, D.M.; Artiushin, G.; Sehgal, A. A Circadian clock in the blood-brain barrier regulates xenobiotic efflux. Cell 2018, 173, 130–139. [Google Scholar] [CrossRef]
- Kervezee, L.; Hartman, R.; Berg, D.J.v.d.; Shimizu, S.; Emoto-Yamamoto, Y.; Meijer, J.H.; Lange, E.C.M.d. Diurnal variation in p-glycoprotein-mediated transport and cerebrospinal fluid turnover in the brain. AAPS J. 2014, 16, 1029–1037. [Google Scholar] [CrossRef]
- Savolainen, H.; Meerlo, P.; Elsinga, P.H.; Windhorst, A.D.; Dierckx, R.A.; Colabufo, N.A.; van Waarde, A.; Luurtsema, G. P-glycoprotein function in the rodent brain displays a daily rhythm, a quantitative in vivo PET study. AAPS J. 2016, 18, 1524–1531. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.E.; Clarke, G.; Dinan, T.G.; Cryan, J.F.; Griffin, B.T. Human P-glycoprotein differentially affects antidepressant drug transport: Relevance to blood-brain barrier permeability. Int. J. Neuropsychopharmacol. 2013, 16, 2259–2272. [Google Scholar] [CrossRef]
- Uhr, M.; Grauer, M.T.; Yassouridis, A.; Ebinger, M. Blood-brain barrier penetration and pharmacokinetics of amitriptyline and its metabolites in p-glycoprotein (abcb1ab) knock-out mice and controls. J. Psychiatr. Res. 2007, 41, 179–188. [Google Scholar] [CrossRef]
- O’Brien, F.E.; Clarke, G.; Fitzgerald, P.; Dinan, T.G.; Griffin, B.T.; Cryan, J.F. Inhibition of P-glycoprotein enhances transport of imipramine across the blood-brain barrier: Microdialysis studies in conscious freely moving rats. Br. J. Pharm. 2012, 166, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Lemmer, B.; Nold, G. Circadian changes in estimated hepatic blood flow in healthy subjects. Br. J. Clin. Pharm. 1991, 32, 627–629. [Google Scholar] [CrossRef]
- Hodgson, K.; Tansey, K.E.; Uher, R.; Dernovšek, M.Z.; Mors, O.; Hauser, J.; Souery, D.; Maier, W.; Henigsberg, N.; Rietschel, M.; et al. Exploring the role of drug-metabolising enzymes in antidepressant side effects. Psychopharmacology 2015, 232, 2609–2617. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Yu, F.; Guo, L.; Chen, M.; Yuan, X.; Wu, B. Small heterodimer partner regulates circadian cytochromes p450 and drug-induced hepatotoxicity. Theranostics 2018, 8, 5246–5258. [Google Scholar] [CrossRef]
- Zhang, Y.K.; Yeager, R.L.; Klaassen, C.D. Circadian expression profiles of drug-processing genes and transcription factors in mouse liver. Drug Metab. Dispos. 2009, 37, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, T.; Tomita, M.; Matsunaga, N.; Nakagawa, H.; Koyanagi, S.; Ohdo, S. Molecular basis for rhythmic expression of CYP3A4 in serum-shocked HepG2 cells. Pharm. Genom. 2007, 17, 1047–1056. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, N.; Inoue, M.; Kusunose, N.; Kakimoto, K.; Hamamura, K.; Hanada, Y.; Toi, A.; Yoshiyama, Y.; Sato, F.; Fujimoto, K.; et al. Time-dependent interaction between differentiated embryo chondrocyte-2 and CCAAT/enhancer-binding protein alpha underlies the circadian expression of CYP2D6 in serum-shocked HepG2 cells. Mol. Pharm. 2012, 81, 739–747. [Google Scholar] [CrossRef]
- Mauvoisin, D.; Wang, J.; Jouffe, C.; Martin, E.; Atger, F.; Waridel, P.; Quadroni, M.; Gachon, F.; Naef, F. Circadian clock-dependent and -independent rhythmic proteomes implement distinct diurnal functions in mouse liver. Proc. Natl. Acad. Sci. USA 2014, 111, 167–172. [Google Scholar] [CrossRef]
- Oh, J.H.; Lee, J.H.; Han, D.H.; Cho, S.; Lee, Y.J. Circadian clock is involved in regulation of hepatobiliary transport mediated by multidrug resistance-associated protein 2. J. Pharm. Sci. 2017, 106, 2491–2498. [Google Scholar] [CrossRef]
- Carmona-Antonanzas, G.; Santi, M.; Migaud, H.; Vera, L.M. Light- and clock-control of genes involved in detoxification. Chronobiol. Int. 2017, 34, 1026–1041. [Google Scholar] [CrossRef]
- Wyska, E. Pharmacokinetic considerations for current state-of-the-art antidepressants. Expert Opin. Drug Metab. Toxicol. 2019, 15, 831–847. [Google Scholar] [CrossRef]
- White, C.A.; Pardue, R.; Huang, C.; Warren, J. Chronobiological evaluation of the active biliary and renal secretion of ampicillin. Chronobiol. Int. 1995, 12, 410–418. [Google Scholar] [CrossRef]
- Oda, M.; Koyanagi, S.; Tsurudome, Y.; Kanemitsu, T.; Matsunaga, N.; Ohdo, S. Renal circadian clock regulates the dosing-time dependency of cisplatin-induced nephrotoxicity in mice. Mol. Pharm. 2014, 85, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Minami, Y.; Ohashi, M.; Tsuchiya, Y.; Kusaba, T.; Tamagaki, K.; Koike, N.; Umemura, Y.; Inokawa, H.; Yagita, K. Robust circadian clock oscillation and osmotic rhythms in inner medulla reflecting cortico-medullary osmotic gradient rhythm in rodent kidney. Sci. Rep. 2017, 7, 7306. [Google Scholar] [CrossRef]
- Prins, J.M.; Weverling, G.J.; Ketel, R.J.v.; Speelman, P. Circadian variations in serum levels and the renal toxicity of aminoglycosides in patients. Clin. Pharm. 1997, 62, 106–111. [Google Scholar] [CrossRef]
- Kawai, H.; Machida, M.; Ishibashi, T.; Kudo, N.; Kawashima, Y.; Mitsumoto, A. Chronopharmacological analysis of antidepressant activity of a dual-action serotonin noradrenaline reuptake inhibitor (SNRI), milnacipran, in rats. Biol. Pharm. Bull. 2018, 41, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Ushijima, K.; Sakaguchi, H.; Sato, Y.; To, H.; Koyanagi, S.; Higuchi, S.; Ohdo, S. Chronopharmacological study of antidepressants in forced swimming test of mice. J. Pharm. Exp. Ther. 2005, 315, 764–770. [Google Scholar] [CrossRef]
- Matheson, G.J.; Schain, M.; Almeida, R.; Lundberg, J.; Cselenyi, Z.; Borg, J.; Varrone, A.; Farde, L.; Cervenka, S. Diurnal and seasonal variation of the brain serotonin system in healthy male subjects. Neuroimage 2015, 112, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Pangerl, B.; Pangerl, A.; Reiter, R.J. Circadian variations of adrenergic receptors in the mammalian pineal gland: A review. J. Neural Transm. 1990, 81, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Hampp, G.; Ripperger, J.A.; Houben, T.; Schmutz, I.; Blex, C.; Perreau-Lenz, S.; Brunk, I.; Spanagel, R.; Ahnert-Hilger, G.; Meijer, J.H.; et al. Regulation of monoamine oxidase A by circadian-clock components implies clock influence on mood. Curr. Biol. 2008, 18, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Perez-Caballero, L.; Torres-Sanchez, S.; Bravo, L.; Mico, J.A.; Berrocoso, E. Fluoxetine: A case history of its discovery and preclinical development. Expert Opin. Drug Discov. 2014, 9, 567–578. [Google Scholar] [CrossRef]
- Tatsumi, M.; Groshan, K.; Blakely, R.D.; Richelson, E. Pharmacological profile of antidepressants and related compounds at human monoamine transporters. Eur. J. Pharm. 1997, 340, 249–258. [Google Scholar] [CrossRef]
- Praschak-Rieder, N.; Willeit, M.; Wilson, A.A.; Houle, S.; Meyer, J.H. Seasonal variation in human brain serotonin transporter binding. Arch. Gen. Psychiatry 2008, 65, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Gillman, P.K. Tricyclic antidepressant pharmacology and therapeutic drug interactions updated. Br. J. Pharm. 2007, 151, 737–748. [Google Scholar] [CrossRef]
- Marquez, E.; Pavia, J.; Laukonnen, S.; Martos, F.; Gomez, A.; Rius, F.; Cuesta, F.S.d.I. Circadian rhythm in muscarinic receptor subtypes in rat forebrain. Chronobiol. Int. 1990, 7, 277–282. [Google Scholar] [CrossRef]
- Maletic, V.; Eramo, A.; Gwin, K.; Offord, S.J.; Duffy, R.A. The role of norepinephrine and its alpha-adrenergic receptors in the pathophysiology and treatment of major depressive disorder and schizophrenia: A systematic review. Front. Psychiatry 2017, 8, 42. [Google Scholar] [CrossRef]
- Doze, V.A.; Handel, E.M.; Jensen, K.A.; Darsie, B.; Luger, E.J.; Haselton, J.R.; Talbot, J.N.; Rorabaugh, B.R. alpha(1A)-And alpha(1B)-adrenergic receptors differentially modulate antidepressant-like behavior in the mouse. Brain Res. 2009, 1285, 148–157. [Google Scholar] [CrossRef]
- Nestler, E.J.; Barrot, M.; DiLeone, R.J.; Eisch, A.J.; Gold, S.J.; Monteggia, L.M. Neurobiology of depression. Neuron 2002, 34, 13–25. [Google Scholar] [CrossRef]
- Castañeda, T.R.; Prado, B.M.; Prieto, D.; Mora, F. Circadian rhythms of dopamine, glutamate and GABA in the striatum and nucleus accumbens of the awake rat: Modulation by light. J. Pineal Res. 2004, 36, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Eckel-Mahan, K.; Sassone-Corsi, P. Phenotyping circadian rhythms in mice. Curr. Protoc. Mouse Biol. 2015, 5, 271–281. [Google Scholar] [CrossRef]
- Ripperger, J.A.; Jud, C.; Albrecht, U. The daily rhythm of mice. FEBS Lett. 2011, 585, 1384–1392. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.P.; Godinho, S.I.; Pothecary, C.A.; Hankins, M.W.; Foster, R.G.; Peirson, S.N. Rapid assessment of sleep-wake behavior in mice. J. Biol. Rhythms 2012, 27, 48–58. [Google Scholar] [CrossRef]
- Krishnan, V.; Nestler, E.J. Animal models of depression: Molecular perspectives. Curr. Top. Behav. Neurosci. 2011, 7, 121–147. [Google Scholar] [CrossRef]
- Landgraf, D.; Long, J.E.; Proulx, C.D.; Barandas, R.; Malinow, R.; Welsh, D.K. Genetic disruption of circadian rhythms in the suprachiasmatic nucleus causes helplessness, behavioral despair, and anxiety-like behavior in mice. Biol. Psychiatry 2016, 80, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Slattery, D.A.; Cryan, J.F. Using the rat forced swim test to assess antidepressant-like activity in rodents. Nat. Protoc. 2012, 7, 1009–1014. [Google Scholar] [CrossRef]
- Cryan, J.F.; Mombereau, C.; Vassout, A. The tail suspension test as a model for assessing antidepressant activity: Review of pharmacological and genetic studies in mice. Neurosci. Biobehav. Rev. 2005, 29, 571–625. [Google Scholar] [CrossRef]
- Jin, Z.L.; Chen, X.F.; Ran, Y.H.; Li, X.R.; Xiong, J.; Zheng, Y.Y.; Gao, N.N.; Li, Y.F. Mouse strain differences in SSRI sensitivity correlate with serotonin transporter binding and function. Sci. Rep. 2017, 7, 8631. [Google Scholar] [CrossRef]
- Lucki, I.; Dalvi, A.; Mayorga, A.J. Sensitivity to the effects of pharmacologically selective antidepressants in different strains of mice. Psychopharmacology 2001, 155, 315–322. [Google Scholar] [CrossRef]
- Mombereau, C.; Kaupmann, K.; Froestl, W.; Sansig, G.; van der Putten, H.; Cryan, J.F. Genetic and pharmacological evidence of a role for GABA(B) receptors in the modulation of anxiety- and antidepressant-like behavior. Neuropsychopharmacology 2004, 29, 1050–1062. [Google Scholar] [CrossRef]
- Cryan, J.F.; Markou, A.; Lucki, I. Assessing antidepressant activity in rodents: Recent developments and future needs. Trends Pharm. Sci. 2002, 23, 238–245. [Google Scholar] [CrossRef]
- Detke, M.J.; Rickels, M.; Lucki, I. Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmacology 1995, 121, 66–72. [Google Scholar] [CrossRef]
- Richetto, J.; Polesel, M.; Weber-Stadlbauer, U. Effects of light and dark phase testing on the investigation of behavioural paradigms in mice: Relevance for behavioural neuroscience. Pharm. Biochem. Behav. 2019, 178, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Kelliher, P.; Connor, T.J.; Harkin, A.; Sanchez, C.; Kelly, J.P.; Leonard, B.E. Varying responses to the rat forced-swim test under diurnal and nocturnal conditions. Physiol. Behav. 2000, 69, 531–539. [Google Scholar] [CrossRef]
- Borsini, F.; Lecci, A.; Stasi, M.A.; Pessia, M.; Meli, A. Seasonal and circadian variations of behavioural response to antidepressants in the forced swimming test in rats. Behav. Pharmacol. 1990, 1, 395–401. [Google Scholar] [CrossRef]
- Weiner, N.; Clement, H.-W.; Gemsa, D.; Wesemann, W. Circadian and seasonal rhythms of 5-HT receptor subtypes, membrane anisotropy and 5-HT release in hippocampus and cortex of the rat. Neurochem. Int. 1992, 21, 7–14. [Google Scholar] [CrossRef]
- Kawai, H.; Iwadate, R.; Ishibashi, T.; Kudo, N.; Kawashima, Y.; Mitsumoto, A. Antidepressants with different mechanisms of action show different chronopharmacological profiles in the tail suspension test in mice. Chronobiol. Int. 2019, 36, 1194–1207. [Google Scholar] [CrossRef]
- Tsang, A.H.; Astiz, M.; Leinweber, B.; Oster, H. Rodent models for the analysis of tissue clock function in metabolic rhythms research. Front. Endocrinol. 2017, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- Subhash, M.N.; Nagaraja, M.R.; Sharada, S.; Vinod, K.Y. Cortical alpha-adrenoceptor downregulation by tricyclic antidepressants in the rat brain. Neurochem. Int. 2003, 43, 603–609. [Google Scholar] [CrossRef]
- Redolat, R.; Vidal, J.; Gómez, M.C.; Carrasco, M.C. Effects of acute bupropion administration on locomotor activity in adolescent and adult mice. Behav. Pharmacol. 2005, 16, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Belovicova, K.; Bogi, E.; Csatlosova, K.; Dubovicky, M. Animal tests for anxiety-like and depression-like behavior in rats. Interdiscip. Toxicol. 2017, 10, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Kavakli, I.H.; Sancar, A. Circadian photoreception in humans and mice. Mol. Interv. 2002, 2, 484–492. [Google Scholar] [CrossRef]
- Nagayama, H.; Nagano, K.; Ikezaki, A.; Tashiro, T. Double-blind study of the chronopharmacotherapy of depression. Chronobiol. Int. 1991, 8, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Philipp, M.; Marneros, A. Chronobiology and its implications for pharmacotherapy of endogenous depression. Pharmacopsychiatry 1978, 11, 235–240. [Google Scholar] [CrossRef]
- Emens, J.; Lewy, A.; Kinzie, J.M.; Arntz, D.; Rough, J. Circadian misalignment in major depressive disorder. Psychiatry Res 2009, 168, 259–261. [Google Scholar] [CrossRef]
- Kripke, D.F.; Nievergelt, C.M.; Joo, E.; Shekhtman, T.; Kelsoe, J.R. Circadian polymorphisms associated with affective disorders. J. Circadian Rhythm. 2009, 7, 2. [Google Scholar] [CrossRef]
- Takahashi, T.; Sasabayashi, D.; Yucel, M.; Whittle, S.; Lorenzetti, V.; Walterfang, M.; Suzuki, M.; Pantelis, C.; Malhi, G.S.; Allen, N.B. Pineal gland volume in major depressive and bipolar disorders. Front. Psychiatry 2020, 11, 450. [Google Scholar] [CrossRef]
- Gorwood, P. Restoring circadian rhythms: A new way to successfully manage depression. J. Psychopharmacol. 2010, 24, 15–19. [Google Scholar] [CrossRef]
- Nomura, K.; Castanon-Cervantes, O.; Davidson, A.; Fukuhara, C. Selective serotonin reuptake inhibitors and raft inhibitors shorten the period of Period1-driven circadian bioluminescence rhythms in rat-1 fibroblasts. Life Sci. 2008, 82, 1169–1174. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, S.X.; Liu, L.J.; Xu, L.Z.; Gao, L.; Wang, X.F.; Zhang, J.T.; Lu, L. Diurnal alterations in circadian genes and peptides in major depressive disorder before and after escitalopram treatment. Psychoneuroendocrinology 2013, 38, 2789–2799. [Google Scholar] [CrossRef]
- McGlashan, E.M.; Nandam, L.S.; Vidafar, P.; Mansfield, D.R.; Rajaratnam, S.M.W.; Cain, S.W. The SSRI citalopram increases the sensitivity of the human circadian system to light in an acute dose. Psychopharmacology 2018, 235, 3201–3209. [Google Scholar] [CrossRef]
- Carvalho, L.A.; Gorenstein, C.; Moreno, R.; Pariante, C.; Markus, R.P. Effect of antidepressants on melatonin metabolite in depressed patients. J. Psychopharmacol. 2008, 23, 315–321. [Google Scholar] [CrossRef]
- Cuesta, M.; Clesse, D.; Pevet, P.; Challet, E. New light on the serotonergic paradox in the rat circadian system. J. Neurochem. 2009, 110, 231–243. [Google Scholar] [CrossRef]
- Sprouse, J.; Braselton, J.; Reynolds, L. Fluoxetine modulates the circadian biological clock via phase advances of suprachiasmatic nucleus neuronal firing. Biol. Psychiatry 2006, 60, 896–899. [Google Scholar] [CrossRef]
- Schaufler, J.; Ronovsky, M.; Savalli, G.; Cabatic, M.; Sartori, S.B.; Singewald, N.; Pollak, D.D. Fluoxetine normalizes disrupted light-induced entrainment, fragmented ultradian rhythms and altered hippocampal clock gene expression in an animal model of high trait anxiety- and depression-related behavior. Ann. Med. 2015, 48, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Demisch, K.; Demisch, L.; Bochnik, H.J.; Nickelsen, T.; Althoff, P.H.; Schöffling, K.; Rieth, R. Melatonin and cortisol increase after fluvoxamine. Br. J. Clin. Pharmacol. 1986, 22, 620–622. [Google Scholar] [CrossRef] [PubMed]
- Demisch, K.; Demisch, L.; Nickelsen, T.; Rieth, R. The influence of acute and subchronic administration of various antidepressants on early morning melatonin plasma levels in healthy subjects: Increases following fluvoxamine. J. Neural Transm. 1987, 68, 257–270. [Google Scholar] [CrossRef]
- Hao, Y.; Hu, Y.; Wang, H.; Paudel, D.; Xu, Y.; Zhang, B. The effect of fluvoxamine on sleep architecture of depressed patients with insomnia: An 8-week, open-label, baseline-controlled study. Nat. Sci. Sleep 2019, 11, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Murata, Y.; Kamishioiri, Y.; Tanaka, K.; Sugimoto, H.; Sakamoto, S.; Kobayashi, D.; Mine, K. Severe sleepiness and excess sleep duration induced by paroxetine treatment is a beneficial pharmacological effect, not an adverse reaction. J. Affect. Disord. 2013, 150, 1209–1212. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.; Hojer, A.M.; Buchberg, J.; Areberg, J.; Nutt, D.J. Differentiated effects of the multimodal antidepressant vortioxetine on sleep architecture: Part 1, a pharmacokinetic/pharmacodynamic comparison with paroxetine in healthy men. J. Psychopharmacol. 2015, 29, 1085–1091. [Google Scholar] [CrossRef]
- Hariharasubramanian, N.; Nair, N.P.V.; Pilapil, C.; Isaac, I.; Quirion, R. Effect of imipramine on the circadian rhythm of plasma melatonin in unipolar depression. Chronobiol. Int. 1986, 3, 65–69. [Google Scholar] [CrossRef]
- Thompson, C.; Mezey, G.; Corn, T.; Franey, C.; English, J.; Arendt, J.; Checkley, S.A. The effect of desipramine upon melatonin and cortisol secretion in depressed and normal subjects. Br. J. Psychiatry 1985, 147, 389–393. [Google Scholar] [CrossRef]
- Spulber, S.; Conti, M.; Elberling, F.; Raciti, M.; Borroto-Escuela, D.O.; Fuxe, K.; Ceccatelli, S. Desipramine restores the alterations in circadian entrainment induced by prenatal exposure to glucocorticoids. Transl. Psychiatry 2019, 9, 263. [Google Scholar] [CrossRef]
- Refinetti, R.; Menaker, M. Effects of imipramine on circadian rhythms in the golden hamster. Pharm. Biochem. Behav. 1993, 45, 27–33. [Google Scholar] [CrossRef]
- Castanho, A.; Bothorel, B.; Seguin, L.; Mocaer, E.; Pevet, P. Like melatonin, agomelatine (S20098) increases the amplitude of oscillations of two clock outputs: Melatonin and temperature rhythms. Chronobiol. Int. 2014, 31, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Redman, J.R.; Francis, A.J.P. Entrainment of rat circadian rhythms by the melatonin agonist s-20098 requires intact suprachiasmatic nuclei but not the pineal. J. Biol. Rhythms 1998, 13, 39–51. [Google Scholar] [CrossRef]
- Redman, J.R.; Guardiola-Lemaitre, B.; Brown, M.; Delagrange, P.; Armstrong, S.M. Dose dependent effects of S.20098, a melatonin agonist, on direction of re-entrainment of rat circadian activity rhythms. Psychopharmacology 1995, 118, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Kräuchi, K.; Cajochen, C.; Möri, D.; Graw, P.; Wirz-Justice, A. Early evening melatonin and S-20098 advance circadian phase and nocturnal regulation of core body temperature. Am. J. Physiol. 1997, 272, R1178–R1188. [Google Scholar] [CrossRef]
- Leproult, R.; Van Onderbergen, A.; L’Hermite-Baleriaux, M.; Van Cauter, E.; Copinschi, G. Phase-shifts of 24-h rhythms of hormonal release and body temperature following early evening administration of the melatonin agonist agomelatine in healthy older men. Clin. Endocrinol. 2005, 63, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Cajochen, C.; Krauchi, K.; Mori, D.; Graw, P.; Wirz-Justice, A. Melatonin and S-20098 increase REM sleep and wake-up propensity without modifying NREM sleep homeostasis. Am. J. Physiol. 1997, 272, R1189–R1196. [Google Scholar] [CrossRef] [PubMed]
- Descamps, A.; Rousset, C.; Millan, M.J.; Spedding, M.; Delagrange, P.; Cespuglio, R. Influence of the novel antidepressant and melatonin agonist/serotonin2C receptor antagonist, agomelatine, on the rat sleep-wake cycle architecture. Psychopharmacology 2009, 205, 93–106. [Google Scholar] [CrossRef]
- Mairesse, J.; Silletti, V.; Laloux, C.; Zuena, A.R.; Giovine, A.; Consolazione, M.; van Camp, G.; Malagodi, M.; Gaetani, S.; Cianci, S.; et al. Chronic agomelatine treatment corrects the abnormalities in the circadian rhythm of motor activity and sleep/wake cycle induced by prenatal restraint stress in adult rats. Int. J. Neuropsychopharmacol. 2013, 16, 323–338. [Google Scholar] [CrossRef]
- Schmelting, B.; Corbach-Sohle, S.; Kohlhause, S.; Schlumbohm, C.; Flugge, G.; Fuchs, E. Agomelatine in the tree shrew model of depression: Effects on stress-induced nocturnal hyperthermia and hormonal status. Eur. Neuropsychopharmacol. 2014, 24, 437–447. [Google Scholar] [CrossRef]
- Rainer, Q.; Xia, L.; Guilloux, J.-P.; Gabriel, C.; Mocaër, E.; Hen, R.; Enhamre, E.; Gardier, A.M.; David, D.J. Beneficial behavioural and neurogenic effects of agomelatine in a model of depression/anxiety. Int. J. Neuropsychopharmacol. 2011, 15, 321–335. [Google Scholar] [CrossRef]
- Barden, N.; Shink, E.; Labbe, M.; Vacher, R.; Rochford, J.; Mocaer, E. Antidepressant action of agomelatine (S 20098) in a transgenic mouse model. Prog. Neuropsychopharmacol. Biol. Psychiatry 2005, 29, 908–916. [Google Scholar] [CrossRef]
- Kasper, S.; Hajak, G.; Wulff, K.; Hoogendijk, W.J.; Montejo, A.L.; Smeraldi, E.; Rybakowski, J.K.; Quera-Salva, M.A.; Wirz-Justice, A.M.; Picarel-Blanchot, F.; et al. Efficacy of the novel antidepressant agomelatine on the circadian rest-activity cycle and depressive and anxiety symptoms in patients with major depressive disorder: A randomized, double-blind comparison with sertraline. J. Clin. Psychiatry 2010, 71, 109–120. [Google Scholar] [CrossRef]
- Quera Salva, M.A.; Vanier, B.; Laredo, J.; Hartley, S.; Chapotot, F.; Moulin, C.; Lofaso, F.; Guilleminault, C. Major depressive disorder, sleep EEG and agomelatine: An open-label study. Int. J. Neuropsychopharmacol. 2007, 10, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Bellet, M.M.; Vawter, M.P.; Bunney, B.G.; Bunney, W.E.; Sassone-Corsi, P. Ketamine influences CLOCK:BMAL1 function leading to altered circadian gene expression. PLoS ONE 2011, 6, e23982. [Google Scholar] [CrossRef] [PubMed]
- Duncan, W.C., Jr.; Slonena, E.; Hejazi, N.S.; Brutsche, N.; Yu, K.C.; Park, L.; Ballard, E.D.; Zarate, C.A., Jr. Motor-activity markers of circadian timekeeping are related to ketamine’s rapid antidepressant properties. Biol. Psychiatry 2017, 82, 361–369. [Google Scholar] [CrossRef]
- Schmid, D.A.; Wichniak, A.; Uhr, M.; Ising, M.; Brunner, H.; Held, K.; Weikel, J.C.; Sonntag, A.; Steiger, A. Changes of sleep architecture, spectral composition of sleep EEG, the nocturnal secretion of cortisol, ACTH, GH, prolactin, melatonin, ghrelin, and leptin, and the DEX-CRH test in depressed patients during treatment with mirtazapine. Neuropsychopharmacology 2006, 31, 832–844. [Google Scholar] [CrossRef]
- Osredkar, D.; Kržan, M. Expression of serotonin receptor subtypes in rat brain and astrocyte cell cultures: An age- and tissue-dependent process. Period Biol. 2009, 111, 129–135. [Google Scholar]
- Challet, E.; Turek, F.W.; Laute, M.-A.; Reeth, O.V. Sleep deprivation decreases phase-shift responses of circadian rhythms to light in the mouse: Role of serotonergic and metabolic signals. Brain Res. 2001, 909, 81–91. [Google Scholar] [CrossRef]
- Bang-Andersen, B.; Ruhland, T.; Jorgensen, M.; Smith, G.; Frederiksen, K.; Jensen, K.G.; Zhong, H.; Nielsen, S.M.; Hogg, S.; Mork, A.; et al. Discovery of 1-[2-(2,4-dimethylphenylsulfanyl)phenyl]piperazine (Lu AA21004): A novel multimodal compound for the treatment of major depressive disorder. J. Med. Chem. 2011, 54, 3206–3221. [Google Scholar] [CrossRef] [PubMed]
- Swanson, L.M.; Burgess, H.J.; Huntley, E.D.; Bertram, H.; Mooney, A.; Zollars, J.; Dopp, R.; Hoffmann, R.; Armitage, R.; Todd Arnedt, J. Relationships between circadian measures, depression, and response to antidepressant treatment: A preliminary investigation. Psychiatry Res. 2017, 252, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Monteggia, L.M. A neurotrophic model for stress-related mood disorders. Biol. Psychiatry 2006, 59, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Malberg, J.; Nakagawa, S.; D’Sa, C. Neuronal plasticity and survival in mood disorders. Biol. Psychiatry 2000, 48, 732–739. [Google Scholar] [CrossRef]
- Faraguna, U.; Vyazovskiy, V.V.; Nelson, A.B.; Tononi, G.; Cirelli, C. A causal role for brain-derived neurotrophic factor in the homeostatic regulation of sleep. J. Neurosci. 2008, 28, 4088–4095. [Google Scholar] [CrossRef] [PubMed]
- Schaaf, M.J.M.; Duurland, R.; Kloet, E.R.; Vreugdenhil, E. Circadian variation in BDNF mRNA expression in the rat hippocampus. Brain Res.Mol. 2000, 75, 342–344. [Google Scholar] [CrossRef]
- Begliuomini, S.; Lenzi, E.; Ninni, F.; Casarosa, E.; Merlini, S.; Pluchino, N.; Valentino, V.; Luisi, S.; Luisi, M.; Genazzani, A.R. Plasma brain-derived neurotrophic factor daily variations in men: Correlation with cortisol circadian rhythm. J. Endocrinol. 2008, 197, 429–435. [Google Scholar] [CrossRef]
- Cain, S.W.; Chang, A.M.; Vlasac, I.; Tare, A.; Anderson, C.; Czeisler, C.A.; Saxena, R. Circadian rhythms in plasma brain-derived neurotrophic factor differ in men and women. J. Biol. Rhythms 2017, 32, 75–82. [Google Scholar] [CrossRef]
- Giese, M.; Beck, J.; Brand, S.; Muheim, F.; Hemmeter, U.; Hatzinger, M.; Holsboer-Trachsler, E.; Eckert, A. Fast BDNF serum level increase and diurnal BDNF oscillations are associated with therapeutic response after partial sleep deprivation. J. Psychiatr. Res. 2014, 59, 1–7. [Google Scholar] [CrossRef]
- Lu, Y.; Ho, C.S.; McIntyre, R.S.; Wang, W.; Ho, R.C. Effects of vortioxetine and fluoxetine on the level of Brain Derived Neurotrophic Factors (BDNF) in the hippocampus of chronic unpredictable mild stress-induced depressive rats. Brain Res. Bull. 2018, 142, 1–7. [Google Scholar] [CrossRef]
- Lu, Y.; Ho, C.S.; McIntyre, R.S.; Wang, W.; Ho, R.C. Agomelatine-induced modulation of brain-derived neurotrophic factor (BDNF) in the rat hippocampus. Life Sci. 2018, 210, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Malberg, J.E.; Eisch, A.J.; Nestler, E.J.; Duman, R.S. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J. Neurosci. 2000, 20, 9104–9110. [Google Scholar] [CrossRef]
- Kasper, S.; Hamon, M. Beyond the monoaminergic hypothesis: Agomelatine, a new antidepressant with an innovative mechanism of action. World J. Biol. Psychiatry 2009, 10, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Martinet, L.; Guardiola-Lemaitre, B.; Mocaer, E. Entrainment of circadian rhythms by S-20098, a melatonin agonist, is dose and plasma concentration dependent. Pharmacol. Biochem. Behav. 1996, 54, 713–718. [Google Scholar] [CrossRef]
- Pitrosky, B.; Kirsch, R.; Malan, A.; Mocaer, E.; Pevet, P. Organization of rat circadian rhythms during daily infusion of melatonin or S20098, a melatonin agonist. Am. J. Physiol. 1999, 277, R812–R828. [Google Scholar] [CrossRef] [PubMed]
- Papp, M.; Gruca, P.; Boyer, P.A.; Mocaer, E. Effect of agomelatine in the chronic mild stress model of depression in the rat. Neuropsychopharmacology 2003, 28, 694–703. [Google Scholar] [CrossRef]
- Liu, M.Y.; Yin, C.Y.; Zhu, L.J.; Zhu, X.H.; Xu, C.; Luo, C.X.; Chen, H.; Zhu, D.Y.; Zhou, Q.G. Sucrose preference test for measurement of stress-induced anhedonia in mice. Nat. Protoc. 2018, 13, 1686–1698. [Google Scholar] [CrossRef]
- Moriguchi, S.; Takamiya, A.; Noda, Y.; Horita, N.; Wada, M.; Tsugawa, S.; Plitman, E.; Sano, Y.; Tarumi, R.; ElSalhy, M.; et al. Glutamatergic neurometabolite levels in major depressive disorder: A systematic review and meta-analysis of proton magnetic resonance spectroscopy studies. Mol. Psychiatry 2019, 24, 952–964. [Google Scholar] [CrossRef]
- Paul, K.N.; Fukuhara, C.; Karom, M.; Tosini, G.; Albers, H.E. AMPA/kainate receptor antagonist DNQX blocks the acute increase of Per2 mRNA levels in most but not all areas of the SCN. Brain Res. Mol. Brain Res. 2005, 139, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Paul, K.N.; Fukuhara, C.; Tosini, G.; Albers, H.E. Transduction of light in the suprachiasmatic nucleus: Evidence for two different neurochemical cascades regulating the levels of Per1 mRNA and pineal melatonin. Neuroscience 2003, 119, 137–144. [Google Scholar] [CrossRef]
- Berman, R.M.; Cappiello, A.; Anand, A.; Oren, D.A.; Heninger, G.R.; Charney, D.S.; Krystal, J.H. Antidepressant effects of ketamine in depressed patients. Biol. Psychiatry 2000, 47, 351–354. [Google Scholar] [CrossRef]
- Ma, Z.; Zang, T.; Birnbaum, S.G.; Wang, Z.; Johnson, J.E.; Zhang, C.L.; Parada, L.F. TrkB dependent adult hippocampal progenitor differentiation mediates sustained ketamine antidepressant response. Nat. Commun. 2017, 8, 1668. [Google Scholar] [CrossRef]
- Goldsmith, C.S.; Bell-Pedersen, D. Diverse roles for MAPK signaling in circadian clocks. Adv. Genet. 2013, 84, 1–39. [Google Scholar] [CrossRef] [PubMed]
- Reus, G.Z.; Vieira, F.G.; Abelaira, H.M.; Michels, M.; Tomaz, D.B.; dos Santos, M.A.; Carlessi, A.S.; Neotti, M.V.; Matias, B.I.; Luz, J.R.; et al. MAPK signaling correlates with the antidepressant effects of ketamine. J. Psychiatr. Res. 2014, 55, 15–21. [Google Scholar] [CrossRef]
- Orozco-Solis, R.; Montellier, E.; Aguilar-Arnal, L.; Sato, S.; Vawter, M.P.; Bunney, B.G.; Bunney, W.E.; Sassone-Corsi, P. A Circadian genomic signature common to ketamine and sleep deprivation in the anterior cingulate cortex. Biol. Psychiatry 2017, 82, 351–360. [Google Scholar] [CrossRef] [PubMed]

| Antidepressant or Active Metabolite | Animal Species (Gender) | Daily Dose (mg/kg) | Duration (Days) | Route | Biological Matrix | ZT | Pharmacokinetic Parameters | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| tmax (h) | Cmax (mg/L) | AUC (mg.h/L) | t1/2α (h) | t1/2β (h) | ka (h−1) | ||||||||
| Amitriptyline | Wistar rat (male) | 64 | 10 | Intragastric | Plasma | ZT4 | - | - | 5.43 | - | 3.88 | 0.96 | [48] |
| ZT16 | - | - | 4.70 | - | 2.90 | 1.40 | |||||||
| Desipramine | Wistar rat (male) | 10 | 1 | Intravenous | Plasma | ZT0.5 | 0.78 | 54.5 | 272 | 0.18 | 2.76 | - | [49] |
| ZT12.5 | 1.71 | 47.7 | 259 | 0.71 | 1.90 | - | |||||||
| Brain | ZT0.5 | 1.86 | 1.01 1 | 6.08 2 | 0.75 | 1.08 | - | ||||||
| ZT12.5 | 2.25 | 1.02 1 | 6.50 2 | 1.20 | 0.91 | - | |||||||
| Intraperitoneal | Brain | ZT0.5 | 2.32 | 0.85 1 | 6.90 2 | 0.83 | 3.72 | - | |||||
| ZT12.5 | 1.59 | 1.80 1 | 10.69 2 | 0.53 | 2.91 | - | |||||||
| Imipramine | Wistar rat (male) | 10 | 1 | Intravenous | Plasma | ZT0.5 | - | - | 482 | - | 1.08 | - | [49] |
| ZT12.5 | - | - | 474 | - | 0.91 | - | |||||||
| Brain | ZT0.5 | - | - | 40.43 2 | - | 1.40 | - | ||||||
| ZT12.5 | - | - | 53.21 2 | - | 2.83 | - | |||||||
| Intraperitoneal | Brain | ZT0.5 | 0.43 | 2.85 1 | 6.44 2 | 0.11 | 1.23 | - | |||||
| ZT12.5 | 0.40 | 4.80 1 | 9.67 2 | 0.11 | 1.08 | - | |||||||
| Antidepressant or Active Metabolite | Subjects | Study Design | Daily Dose (mg) | Duration (Days) | Formulation | Time of Administration | Plasma Pharmacokinetic Parameters | Ref. | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| tmax (h) | Cmax (mg/L) | AUC (mg.h/L) | t1/2β (h) | kel (h−1) | ka (h−1) | MRT (h) | ||||||||
| Amitriptyline | 10 healthy subjects (♂), 22–31 years old. | Crossover | 50 | 21 | Injectable solution | 9h00 | 3.2 * | 96.1 | 1270 | 15.7 | - | 0.36 * | - | [37] |
| 21h00 | 4.4 * | 72.8 | 1224 | 17.2 | - | 0.25 * | - | |||||||
| Nortriptyline | 10 healthy subjects (♂), 22–30 years old. | Crossover | 100 | 14 | Oral formulation: 25 mg capsules | 9h00 | 6.2 | 32 | 730 | 15.0 | - | - | - | [50] |
| 21h00 | 8.8 | 31 | 730 | 16.0 | - | - | - | |||||||
| Trimipramine | 12 healthy subjects (6 ♀, 6 ♂), 22–37 years old. | Crossover | 100 | 15 | Oral formulation: 100 mg tablet | 8h00 | 2.5 | 37.8 | 362 | 10.9 | - | - | 10.8 | [46] |
| 20h00 | 2.8 | 39.2 | 376 | 9.9 | - | - | 11.5 | |||||||
| Oral formulation: solution | 8h00 | 1.5 * | 48.2 * | 372 | 9.9 | - | - | 9.8 * | ||||||
| 20h00 | 2.5 * | 28.8 * | 322 | 11.1 | - | - | 11.8 * | |||||||
| Sertraline | 10 healthy subjects (♂), 18–45 years old. | Crossover | 100 | 1 | Oral formulation: 100 mg tablet | Morning | 7.0 | 24.5 | 0.664 | 20.0 | 0.0347 | - | - | [47] |
| Evening | 7.3 | 24.4 | 0.705 | 20.8 | 0.0333 | - | - | |||||||
| Antidepressant | Species (Gender) | Dose (mg/Kg) | Initial of Experiment after Administration (h) | Route | Zeitgeber Time (ZT) Administrations | Pharmacodynamic | Drug Concentration | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Test | 24 h Rhythm Variation | Observations | ||||||||
| Amitriptyline | ICR mice (male) | 15 | 0.5 | Intraperitoneal | ZT2, ZT6, ZT10, ZT14, ZT18, ZT22 | FST | Yes | Lowest immobility at ZT14. | - | [105] |
| Bupropion | C57BL/6 mice (male) | 20 | 1 | Intraperitoneal | ZT1, ZT7, ZT13, ZT19 | TST | No, but significantly different between ZT | Lowest immobility at ZT1. | No significant differences between dosing times in plasma and brain. | [134] |
| Locomotor activity | No | Increased | ||||||||
| Desipramine | CD-COBS rats (male) | 20 | 24, 5 and 1 | Intraperitoneal | ZT3, ZT7, ZT11, ZT15, ZT19, ZT23 | FST | No | - | - | [132] |
| Fluoxetine | C57BL/6 mice (male) | 30 | 1 | Intraperitoneal | ZT1, ZT7, ZT13, ZT19 | TST | Yes | Lowest immobility at ZT1. | No significant differences between dosing times in plasma and brain. | [134] |
| Locomotor activity | Yes | Lowest locomotion activity at ZT1 | ||||||||
| Fluvoxamine | ICR mice (male) | 30 | 0.5 | Intraperitoneal | ZT2, ZT6, ZT10, ZT14, ZT18, ZT22 | FST | Yes | Lowest immobility at ZT14. | ZT2 > ZT14 in plasma, significantly different after 1h of drug injection. No differences in brain. | [105] |
| ZT2, ZT14 | Locomotor activity | No | No effect | |||||||
| Imipramine | C57BL/6 mice (male) | 30 | 1 | Intraperitoneal | ZT1, ZT7, ZT13, ZT19 | TST | Yes | Lowest immobility at ZT13. | No significant differences between dosing times in plasma and brain | [134] |
| Locomotor activity | No | Reduced | ||||||||
| Wistar Hannover rats (male) | 30 | 1 | Intraperitoneal | ZT1, ZT13 | FST | Yes | Lowest immobility and highest climbing at ZT1. | ZT1 > ZT13 for imipramine and desipramine in plasma but not significantly different | [36] | |
| 10 for 2 weeks | 1 | Intraperitoneal | ZT1, ZT13 | FST | Yes | Lowest immobility and highest climbing at ZT1. | - | |||
| 30 for 2 weeks | 1 | Intraperitoneal | ZT1, ZT13 | FST | No | - | - | |||
| Mianserin | CD-COBS rats (male) | 15 | 24, 5 and 1 | Intraperitoneal | ZT3, ZT7, ZT11, ZT15, ZT19, ZT23 | FST | No | - | - | [132] |
| Milnacipran | Wistar Hannover rats (male) | 60 | 1 | Oral | ZT1, ZT13 | FST | Yes | Lowest immobility and highest swimming at ZT1. | No significant differences between dosing times in plasma and brain | [104] |
| Nomifensine | CD-COBS rats (male) | 5 | 24, 5 and 1 | Intraperitoneal | ZT3, ZT7, ZT11, ZT15, ZT19, ZT23 | FST | Yes | Lowest immobility at ZT7 | - | [132] |
| Venlafaxine | C57BL/6 mice (male) | 30 | 1 | Intraperitoneal | ZT1, ZT7, ZT13, ZT19 | TST | Yes | Lowest immobility at ZT7. | No significant differences between dosing times in plasma and brain | [134] |
| Locomotor activity | Yes | Lowest locomotion activity at ZT7. | ||||||||
| Antidepressant | Subjects | Study Design | Daily Dose (mg) | Duration (Days) | Time Administrations | Pharmacodynamic | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|
| Test | 24 h Rhythm Variation | Observations | |||||||
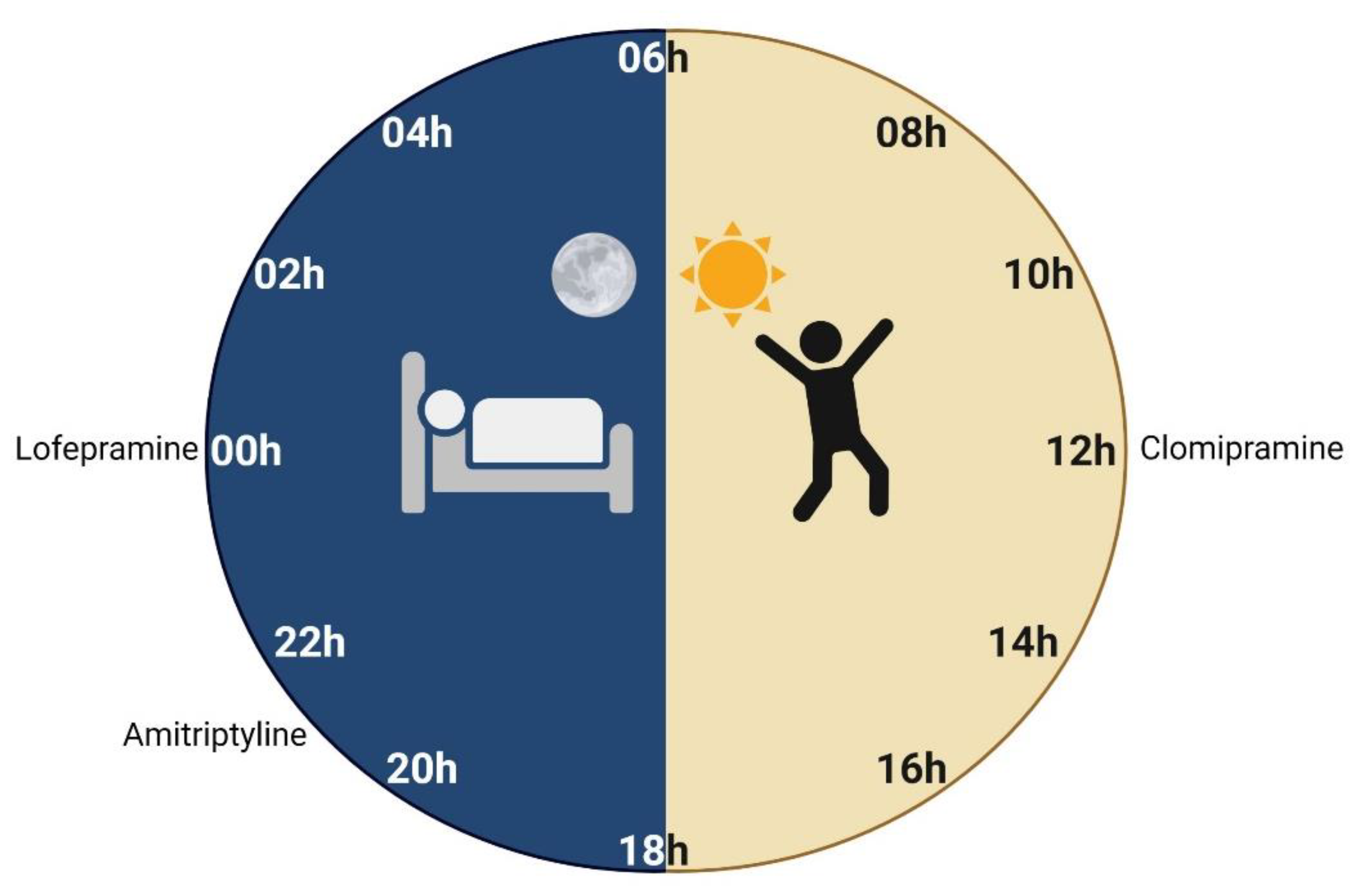
| Amitriptyline | 10 healthy (♂) subjects. Range age: 22–31 years old. | Crossover | 50 | 21 | 9h00 21h00 | Antimuscarinic action (saliva flow) and sedation effect by self-rating scales | Yes | Highest salivary flow and lowest sedative effect at 21h00 | [37] |
| Clomipramine | 40 patients with MDD (15 ♀, 25 ♂). Range age: 18–65 years old. | Crossover | 150 | 28 | 8h20 12h20 20h30 | HRSD and BDRS | Yes | Lowest depressive symptoms at 12h20 | [140] |
| Lofepramine | 30 patients with MDD (22 ♀, 8 ♂). Range age: 25–60 years old. | Parallel | 210 | 21 | 8h00 16h00 24h00 | HRSD and CSRS | Yes | Lowest depressive symptoms at 24h00 | [141] |
| Antidepressant | Pre-Clinical Studies | Clinical Studies | References |
|---|---|---|---|
| SSRI | |||
| Citalopram/escitalopram |
|
| [146,147,148] |
| Fluoxetine |
|
| [146,149,150,151,152] |
| Fluvoxamine |
|
| [146,153,154,155] |
| Paroxetine |
|
| [146,156,157] |
| Sertraline |
| [146] | |
| SNRI | |||
| Duloxetine |
| [149] | |
| TCA | |||
| Desipramine |
|
| [158,159,160] |
| Imipramine |
|
| [158,159,161] |
| Atypical | |||
| Agomelatine |
|
| [162,163,164,165,166,167,168,169,170,171,172,173,174] |
| Ketamine |
|
| [175,176] |
| Mirtazapine |
| [177] | |
| Vortioxetine |
| [157] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, S.; Bicker, J.; Falcão, A.; Fortuna, A. Antidepressants and Circadian Rhythm: Exploring Their Bidirectional Interaction for the Treatment of Depression. Pharmaceutics 2021, 13, 1975. https://doi.org/10.3390/pharmaceutics13111975
Silva S, Bicker J, Falcão A, Fortuna A. Antidepressants and Circadian Rhythm: Exploring Their Bidirectional Interaction for the Treatment of Depression. Pharmaceutics. 2021; 13(11):1975. https://doi.org/10.3390/pharmaceutics13111975
Chicago/Turabian StyleSilva, Soraia, Joana Bicker, Amílcar Falcão, and Ana Fortuna. 2021. "Antidepressants and Circadian Rhythm: Exploring Their Bidirectional Interaction for the Treatment of Depression" Pharmaceutics 13, no. 11: 1975. https://doi.org/10.3390/pharmaceutics13111975
APA StyleSilva, S., Bicker, J., Falcão, A., & Fortuna, A. (2021). Antidepressants and Circadian Rhythm: Exploring Their Bidirectional Interaction for the Treatment of Depression. Pharmaceutics, 13(11), 1975. https://doi.org/10.3390/pharmaceutics13111975








