Green Metallic Nanoparticles for Cancer Therapy: Evaluation Models and Cancer Applications
Abstract
1. Introduction
2. Overview: Green Biosynthesis of Metallic Nanoparticles
2.1. Nanomaterials Classification: From Geometrical Aspects to Functionality
2.2. Green Biosynthesis of MNPs
2.2.1. Phytochemical Synthesis
2.2.2. Microorganism-Assisted Synthesis
2.3. Development of Stimuli-Responsive MNPs
2.3.1. Thermo-Responsive Polymers
2.3.2. pH-Responsive Mechanisms
2.3.3. Light-Responsive Groups
2.3.4. Magnetic Targeting and Theragnostic
2.4. The Influence of the Physicochemical Properties of MNPs
3. Cancer Evaluation Models
3.1. In Silico Analysis
3.2. Conventional 2D In Vitro Assays
3.3. Mimetic 3D Cell Culture
3.4. Ex Vivo Models
3.4.1. Isolated Tissue System
3.4.2. Isolated and Perfused Organ System
3.4.3. Integrative Evidence about the Physiological Profile of AgNPs and AuNPs
3.5. In Vivo Evaluation Models
3.5.1. C. elegans and D. rerio (Zebrafish) Models
3.5.2. Murine Model
4. Application of Biosynthetic MNPs in Cancer Therapy
4.1. Skin Cancer
4.2. Breast Cancer
4.3. Lung Cancer
4.4. Prostate Cancer
4.5. Colorectal Cancer
4.6. Cervical Cancer
4.7. Leukemia
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Kashkooli, F.M.; Soltani, M.; Momeni, M.M.; Rahmim, A. Enhanced Drug Delivery to Solid Tumors via Drug-Loaded Nanocarriers: An Image-Based Computational Framework. Front. Oncol. 2021, 11, 655781. [Google Scholar] [CrossRef]
- Kashkooli, F.M.; Soltani, M.; Souri, M. Controlled anti-cancer drug release through advanced nano-drug delivery systems: Static and dynamic targeting strategies. J. Control. Release 2020, 327, 316–349. [Google Scholar] [CrossRef]
- Lane, L.A. Physics in nanomedicine: Phenomena governing the in vivo performance of nanoparticles. Appl. Phys. Rev. 2020, 7, 011316. [Google Scholar] [CrossRef]
- Andleeb, A.; Andleeb, A.; Asghar, S.; Zaman, G.; Tariq, M.; Mehmood, A.; Nadeem, M.; Hano, C.; Lorenzo, J.; Abbasi, B. A Systematic Review of Biosynthesized Metallic Nanoparticles as a Promising Anti-Cancer-Strategy. Cancers 2021, 13, 2818. [Google Scholar] [CrossRef]
- Neouze, M.-A.; Schubert, U. Surface Modification and Functionalization of Metal and Metal Oxide Nanoparticles by Organic Ligands. Mon. Chem. Chem. Mon. 2008, 139, 183–195. [Google Scholar] [CrossRef]
- Yang, G.; Chen, C.; Zhu, Y.; Liu, Z.; Xue, Y.; Zhong, S.; Wang, C.; Gao, Y.; Zhang, W. GSH-Activatable NIR Nanoplatform with Mitochondria Targeting for Enhancing Tumor-Specific Therapy. ACS Appl. Mater. Interfaces 2019, 11, 44961–44969. [Google Scholar] [CrossRef] [PubMed]
- Stephen, B.J.; Suchanti, S.; Mishra, R.; Singh, A. Cancer Nanotechnology in Medicine: A Promising Approach for Cancer Detection and Diagnosis. Crit. Rev. Ther. Drug Carr. Syst. 2020, 37, 375–405. [Google Scholar] [CrossRef] [PubMed]
- Stylianopoulos, T.; Jain, R.K. Design considerations for nanotherapeutics in oncology. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1893–1907. [Google Scholar] [CrossRef] [PubMed]
- Phan, H.; Haes, A.J. What Does Nanoparticle Stability Mean? J. Phys. Chem. C 2019, 123, 16495–16507. [Google Scholar] [CrossRef]
- Buerki-Thurnherr, T.; Schaepper, K.; Aengenheister, L.; Wick, P. Developmental Toxicity of Nanomaterials: Need for a Better Understanding of Indirect Effects. Chem. Res. Toxicol. 2018, 31, 641–642. [Google Scholar] [CrossRef]
- Abreu, T.; Biscaia, M.; Gonçalves, N.; Fonseca, N.A.; Moreira, J.N. In Vitro and In Vivo Tumor Models for the Evaluation of Anticancer Nanoparticles. In Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2021; Volume 1295, pp. 271–299. [Google Scholar]
- Ramos-Gomes, F.; Ferreira, N.; Kraupner, A.; Alves, F.; Markus, M.A. Ex vivo Live Cell Imaging of Nanoparticle-Cell Interactions in the Mouse Lung. Front. Bioeng. Biotechnol. 2020, 8, 588922. [Google Scholar] [CrossRef]
- Nethi, S.K.; Mukherjee, A.; Mukherjee, S. Biosynthesized Gold and Silver Nanoparticles in Cancer Theranostics. In Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications; Springer, Cham: Cham, Switzerland, 2020; Volume 2020, pp. 1–15. [Google Scholar] [CrossRef]
- Gao, W.; Zhang, Y.; Zhang, Q.; Zhang, L. Nanoparticle-Hydrogel: A Hybrid Biomaterial System for Localized Drug Delivery. Ann. Biomed. Eng. 2016, 44, 2049–2061. [Google Scholar] [CrossRef]
- Peng, J.; Liang, X. Progress in research on gold nanoparticles in cancer management. Medicine 2019, 98, e15311. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.; Sazgarnia, A. Gold nanoparticles as cancer theranostic agent. Nanomed. J. 2019, 6, 147–160. [Google Scholar] [CrossRef]
- McNeilly, O.; Mann, R.; Hamidian, M.; Gunawan, C. Emerging Concern for Silver Nanoparticle Resistance in Acinetobacter baumannii and Other Bacteria. Front. Microbiol. 2021, 12, 12. [Google Scholar] [CrossRef]
- Gomes, H.; Martins, C.; Prior, J. Silver Nanoparticles as Carriers of Anticancer Drugs for Efficient Target Treatment of Cancer Cells. Nanomaterials 2021, 11, 964. [Google Scholar] [CrossRef] [PubMed]
- ISO-ISO/TR 13014:2012-Nanotechnologies—Guidance on Physico-Chemical Characterization of Engineered Nanoscale Materials for Toxicologic Assessment. Available online: https://www.iso.org/standard/52334.html (accessed on 24 July 2021).
- Pokropivny, V.; Skorokhod, V. Classification of nanostructures by dimensionality and concept of surface forms engineering in nanomaterial science. Mater. Sci. Eng. C 2007, 27, 990–993. [Google Scholar] [CrossRef]
- Goodilin, E.A.; Weiss, P.S.; Gogotsi, Y. Nanotechnology Facets of the Periodic Table of Elements. ACS Nano 2019, 13, 10879–10886. [Google Scholar] [CrossRef]
- Fang, X.; Wang, Y.; Wang, Z.; Jiang, Z.; Dong, M. Microorganism Assisted Synthesized Nanoparticles for Catalytic Applications. Energies 2019, 12, 190. [Google Scholar] [CrossRef]
- Kraegeloh, A.; Suarez-Merino, B.; Sluijters, T.; Micheletti, C. Implementation of Safe-by-Design for Nanomaterial Development and Safe Innovation: Why We Need a Comprehensive Approach. Nanomaterials 2018, 8, 239. [Google Scholar] [CrossRef] [PubMed]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice, 12 Principles of Green Chemistry; Oxford University Press: Oxford, UK, 1998; p. 30. ISBN 0198506988. [Google Scholar]
- Leza, D.S.; Porcel, E.; Yang, X.; Stefancikova, L.; Bolsa-Ferruz, M.; Savina, F.; Dragoe, D.; Guerquin-Kern, J.-L.; Wu, T.-D.; Hirayama, R.; et al. Green One-Step Synthesis of Medical Nanoagents for Advanced Radiation Therapy. Nanotechnol. Sci. Appl. 2020, 13, 61–76. [Google Scholar] [CrossRef]
- Huang, Y.; Xiao, D.; Burton-Freeman, B.M.; Edirisinghe, I. Chemical Changes of Bioactive Phytochemicals during Thermal Processing. Food Sci. 2016, 1–9. [Google Scholar] [CrossRef]
- Ahmad, F.; Ashraf, N.; Ashraf, T.; Zhou, R.-B.; Yin, D.-C. Biological synthesis of metallic nanoparticles (MNPs) by plants and microbes: Their cellular uptake, biocompatibility, and biomedical applications. Appl. Microbiol. Biotechnol. 2019, 103, 2913–2935. [Google Scholar] [CrossRef]
- Soliwoda, K.; Tomaszewska, E.; Małek, K.; Celichowski, G.; Orlowski, P.; Krzyzowska, M.; Grobelny, J. The synthesis of monodisperse silver nanoparticles with plant extracts. Colloids Surfaces B Biointerfaces 2019, 177, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Boomi, P.; Poorani, G.P.; Selvam, S.; Palanisamy, S.; Jegatheeswaran, S.; Anand, K.; Balakumar, C.; Premkumar, K.; Prabu, H.G. Green biosynthesis of gold nanoparticles using Croton sparsiflorus leaves extract and evaluation of UV protection, antibacterial and anticancer applications. Appl. Organomet. Chem. 2020, 34, e5574. [Google Scholar] [CrossRef]
- Patil, M.; Kang, M.-J.; Niyonizigiye, I.; Singh, A.; Kim, J.-O.; Seo, Y.B.; Kim, G.-D. Extracellular synthesis of gold nanoparticles using the marine bacterium Paracoccus haeundaensis BC74171T and evaluation of their antioxidant activity and antiproliferative effect on normal and cancer cell lines. Colloids Surfaces B Biointerfaces 2019, 183, 110455. [Google Scholar] [CrossRef]
- Krishnan, S.; Bhardwaj, S.K.; Liu, S.; Xing, R.; Chavali, M. Virus-assisted Biological Methods for Greener Synthesis of Nano-Materials. In Handbook of Greener Synthesis of Nanomaterials and Compounds; Elsevier: Amsterdam, The Netherlands, 2021; pp. 785–806. [Google Scholar]
- Hu, P.; Chen, L.; Kang, X.; Chen, S. Surface Functionalization of Metal Nanoparticles by Conjugated Metal–Ligand Interfacial Bonds: Impacts on Intraparticle Charge Transfer. Acc. Chem. Res. 2016, 49, 2251–2260. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.I.; Qari, H.A.; Umar, K.; Ibrahim, M.N.M. Recent Advances in Metal Decorated Nanomaterials and Their Various Biological Applications: A Review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Nieh, M.-P.; Li, Y. Decorating Nanoparticle Surface for Targeted Drug Delivery: Opportunities and Challenges. Polymers 2016, 8, 83. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Diwan, A.; Singh, P.; Gulati, S.; Choudhary, D.; Mongia, A.; Shukla, S.; Gupta, A. Functionalized gold nanostructures: Promising gene delivery vehicles in cancer treatment. RSC Adv. 2019, 9, 23894–23907. [Google Scholar] [CrossRef]
- Li, X.; Jian, M.; Sun, Y.; Zhu, Q.; Wang, Z. The Peptide Functionalized Inorganic Nanoparticles for Cancer-Related Bioanalytical and Biomedical Applications. Molecules 2021, 26, 3228. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.Z.; Ali, I.; Khan, W.S.; Kong, X.; Dempsey, E. Reversible self-assembly of gold nanoparticles in response to external stimuli. Mater. Des. 2021, 205, 109694. [Google Scholar] [CrossRef]
- Sponchioni, M.; Palmiero, U.C.; Moscatelli, D. Thermo-responsive polymers: Applications of smart materials in drug delivery and tissue engineering. Mater. Sci. Eng. C 2019, 102, 589–605. [Google Scholar] [CrossRef] [PubMed]
- Iida, R.; Mitomo, H.; Matsuo, Y.; Niikura, K.; Ijiro, K. Thermoresponsive Assembly of Gold Nanoparticles Coated with Oligo (Ethylene Glycol) Ligands with an Alkyl Head. J. Phys. Chem. C 2016, 120, 15846–15854. [Google Scholar] [CrossRef]
- De La Rosa, V.R. Poly(2-oxazoline)s as materials for biomedical applications. J. Mater. Sci. Mater. Med. 2013, 25, 1211–1225. [Google Scholar] [CrossRef]
- De la Rosa, V.R.; Zhang, Z.; De Geest, B.G.; Hoogenboom, R. Colorimetric Logic Gates Based on Poly(2-alkyl-2-oxazoline)-Coated Gold Nanoparticles. Adv. Funct. Mater. 2015, 25, 2511–2519. [Google Scholar] [CrossRef]
- Li, C.; Wang, C.; Ji, Z.; Jiang, N.; Lin, W.; Li, D. Synthesis of thiol-terminated thermoresponsive polymers and their enhancement effect on optical limiting property of gold nanoparticles. Eur. Polym. J. 2019, 113, 404–410. [Google Scholar] [CrossRef]
- Cortez-Lemus, N.A.; Licea-Claverie, A. Poly(N-vinylcaprolactam), a comprehensive review on a thermoresponsive polymer becoming popular. Prog. Polym. Sci. 2016, 53, 1–51. [Google Scholar] [CrossRef]
- Beija, M.; Marty, J.-D.; Destarac, M. Thermoresponsive poly(N-vinyl caprolactam)-coated gold nanoparticles: Sharp reversible response and easy tunability. Chem. Commun. 2011, 47, 2826–2828. [Google Scholar] [CrossRef]
- Liu, J.; Detrembleur, C.; Hurtgen, M.; Debuigne, A.; De Pauw-Gillet, M.-C.; Mornet, S.; Duguet, E.; Jérôme, C. Thermo-responsive gold/poly(vinyl alcohol)-b-poly(N-vinylcaprolactam) core–corona nanoparticles as a drug delivery system. Polym. Chem. 2014, 5, 5289–5299. [Google Scholar] [CrossRef]
- Hao, G.; Xu, Z.P.; Li, L. Manipulating extracellular tumour pH: An effective target for cancer therapy. RSC Adv. 2018, 8, 22182–22192. [Google Scholar] [CrossRef]
- Liu, J.; Huang, Y.; Kumar, A.; Tan, A.; Jin, S.; Mozhi, A.; Liang, X.-J. pH-Sensitive nano-systems for drug delivery in cancer therapy. Biotechnol. Adv. 2014, 32, 693–710. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, G.; Demir, B.; Timur, S.; Becer, C.R. Poly(methacrylic acid)-Coated Gold Nanoparticles: Functional Platforms for Theranostic Applications. Biomacromolecules 2016, 17, 2901–2911. [Google Scholar] [CrossRef]
- Blazhynska, M.M.; Kyrychenko, A.; Kalugin, O.N. pH-Responsive Coating of Silver Nanoparticles with Poly(2-(N,N-dimethylamino)ethyl methacrylate): The Role of Polymer Size and Degree of Protonation. J. Phys. Chem. C 2021, 125, 12118–12130. [Google Scholar] [CrossRef]
- Ma, K.; Cheng, Y.; Wei, X.; Chen, D.; Zhao, X.; Jia, P. Gold embedded chitosan nanoparticles with cell membrane mimetic polymer coating for pH-sensitive controlled drug release and cellular fluorescence imaging. J. Biomater. Appl. 2021, 35, 857–868. [Google Scholar] [CrossRef]
- Li, D.; He, Q.; Cui, A.Y.; Li, J. Fabrication of pH-Responsive Nanocomposites of Gold Nanoparticles/Poly(4-vinylpyridine). Chem. Mater. 2007, 19, 412–417. [Google Scholar] [CrossRef]
- Fan, C.; Bian, T.; Shang, L.; Shi, R.; Wu, L.-Z.; Tung, C.-H.; Zhang, T. pH-Responsive reversible self-assembly of gold nanoparticles into nanovesicles. Nanoscale 2016, 8, 3923–3925. [Google Scholar] [CrossRef]
- Hu, M.; Ai, X.; Wang, Z.; Zhang, Z.; Cheong, H.; Zhang, W.; Lin, J.; Li, J.; Yang, H.; Xing, B. Nanoformulation of metal complexes: Intelligent stimuli-responsive platforms for precision therapeutics. Nano Res. 2018, 11, 5474–5498. [Google Scholar] [CrossRef]
- Yang, K.-N.; Zhang, C.-Q.; Wang, W.; Wang, P.C.; Zhou, J.-P.; Liang, X.-J. pH-responsive mesoporous silica nanoparticles employed in controlled drug delivery systems for cancer treatment. Cancer Biol. Med. 2014, 11, 34–43. [Google Scholar] [CrossRef]
- Zhuo, S.; Zhang, F.; Yu, J.; Zhang, X.; Yang, G.; Liu, X. pH-sensitive biomaterials for drug delivery. Molecules 2020, 25, 5649. [Google Scholar] [CrossRef] [PubMed]
- Zayed, G.M.; Kamal, I.; Abdelhafez, W.A.; Alsharif, F.M.; Amin, M.A.; Shaykoon, M.S.A.; Sarhan, H.A.; Abdelsalam, A.M. Effect of chemical binding of doxorubicin hydrochloride to gold nanoparticles, versus electrostatic adsorption, on the in vitro drug release and cytotoxicity to breast cancer cells. Pharm. Res. 2018, 35, 112. [Google Scholar] [CrossRef] [PubMed]
- Manna, D.; Udayabhaskararao, T.; Zhao, H.; Klajn, R. Orthogonal light-induced self-assembly of nanoparticles using differently substituted azobenzenes. Angew. Chem. Int. Ed. 2015, 54, 12394–12397. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Han, Y.; Bian, T.; De, S.; Král, P.; Klajn, R. Supramolecular control of azobenzene switching on nanoparticles. J. Am. Chem. Soc. 2018, 141, 1949–1960. [Google Scholar] [CrossRef]
- Shiraishi, Y.; Shirakawa, E.; Tanaka, K.; Sakamoto, H.; Ichikawa, S.; Hirai, T. Spiropyran-modified gold nanoparticles: Reversible size control of aggregates by UV and visible light irradiations. ACS Appl. Mater. Interfaces 2014, 6, 7554–7562. [Google Scholar] [CrossRef]
- He, H.; Feng, M.; Chen, Q.; Zhang, X.; Zhan, H. Light-induced reversible self-assembly of gold nanoparticles surface-immobilized with coumarin ligands. Angew. Chem. Int. Ed. 2016, 55, 936–940. [Google Scholar] [CrossRef]
- Bian, T.; Chu, Z.; Klajn, R. The many ways to assemble nanoparticles using light. Adv. Mater. 2019, 32, e1905866. [Google Scholar] [CrossRef] [PubMed]
- Rwei, A.Y.; Wang, W.; Kohane, D.S. Photoresponsive nanoparticles for drug delivery. Nano Today 2015, 10, 451–467. [Google Scholar] [CrossRef]
- Mayer, G.; Heckel, A. Biologically active molecules with a “light switch”. Angew. Chem. Int. Ed. 2006, 45, 4900–4921. [Google Scholar] [CrossRef]
- Zhao, H.; Sterner, E.S.; Coughlin, E.B.; Theato, P. o-Nitrobenzyl alcohol derivatives: Opportunities in polymer and materials science. Macromolecules 2012, 45, 1723–1736. [Google Scholar] [CrossRef]
- Agasti, S.S.; Chompoosor, A.; You, C.-C.; Ghosh, P.; Kim, C.K.; Rotello, V.M. photoregulated release of caged anticancer drugs from gold nanoparticles. J. Am. Chem. Soc. 2009, 131, 5728–5729. [Google Scholar] [CrossRef]
- Cheng, X.; Sun, R.; Yin, L.; Chai, Z.; Shi, H.; Gao, M. Light-triggered assembly of gold nanoparticles for photothermal therapy and photoacoustic imaging of tumors in vivo. Adv. Mater. 2017, 29, 1–6. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Simchi, A.; Imani, M.; Milani, A.S.; Stroeve, P. Anin vitrostudy of bare and poly(ethylene glycol)-co-fumarate-coated superparamagnetic iron oxide nanoparticles: A new toxicity identification procedure. Nanotechnology 2009, 20, 225104. [Google Scholar] [CrossRef]
- Arias, L.S.; Pessan, J.P.; Vieira, A.P.M.; Lima, T.M.T.d.; Delbem, A.C.B.; Monteiro, D.R. Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity. Antibiotics 2018, 7, 46. [Google Scholar] [CrossRef]
- Anderson, S.; Gwenin, V.V.; Gwenin, C.D. Magnetic Functionalized Nanoparticles for Biomedical, Drug Delivery and Imaging Applications. Nanoscale Res. Lett. 2019, 14, 1–16. [Google Scholar] [CrossRef]
- Surface Modification of Nanoparticles for Targeted Drug Delivery. Surf. Modif. Nanoparticles Target. Drug Deliv. 2019, 391–403. [CrossRef]
- Zhu, N.; Ji, H.; Yu, P.; Niu, J.; Farooq, M.U.; Akram, M.W.; Udego, I.O.; Li, H.; Niu, X. Surface Modification of Magnetic Iron Oxide Nanoparticles. Nanomaterials 2018, 8, 810. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.M.; Tavallaie, R.; Sandiford, L.; Tilley, R.; Gooding, J.J. Gold coated magnetic nanoparticles: From preparation to surface modification for analytical and biomedical applications. Chem. Commun. 2016, 52, 7528–7540. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wu, Z.; Yu, T.; Jiang, C.; Kim, W.-S. Recent progress on magnetic iron oxide nanoparticles: Synthesis, surface functional strategies and biomedical applications. Sci. Technol. Adv. Mater. 2015, 16, 023501. [Google Scholar] [CrossRef] [PubMed]
- Tarkistani, M.; Komalla, V.; Kayser, V. Recent Advances in the Use of Iron–Gold Hybrid Nanoparticles for Biomedical Applications. Nanomaterials 2021, 11, 1227. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Hou, Y.; Sun, S. Magnetic Core/Shell Fe3O4/Au and Fe3O4/Au/Ag Nanoparticles with Tunable Plasmonic Properties. J. Am. Chem. Soc. 2007, 129, 8698–8699. [Google Scholar] [CrossRef] [PubMed]
- Eyvazzadeh, N.; Shakeri-Zadeh, A.; Fekrazad, R.; Amini, E.; Ghaznavi, H.; Kamrava, S.K. Gold-coated magnetic nanoparticle as a nanotheranostic agent for magnetic resonance imaging and photothermal therapy of cancer. Lasers Med. Sci. 2017, 32, 1469–1477. [Google Scholar] [CrossRef]
- Yao, Y.; Zhou, Y.; Liu, L.; Xu, Y.; Chen, Q.; Wang, Y.; Wu, S.; Deng, Y.; Zhang, J.; Shao, A. Nanoparticle-Based Drug Delivery in Cancer Therapy and Its Role in Overcoming Drug Resistance. Front. Mol. Biosci. 2020, 7, 193. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Tang, H.; Liu, Z.; Chen, B. Effects of major parameters of nanoparticles on their physical and chemical properties and recent application of nanodrug delivery system in targeted chemotherapy. Int. J. Nanomed. 2017, 12, 8483–8493. [Google Scholar] [CrossRef]
- Elahi, N.; Kamali, M.; Baghersad, M.H. Recent biomedical applications of gold nanoparticles: A review. Talanta 2018, 184, 537–556. [Google Scholar] [CrossRef]
- Drummer, S.; Madzimbamuto, T.; Chowdhury, M. Green Synthesis of Transition-Metal Nanoparticles and Their Oxides: A Review. Materials 2021, 14, 2700. [Google Scholar] [CrossRef] [PubMed]
- Grassian, V.H. When Size Really Matters: Size-Dependent Properties and Surface Chemistry of Metal and Metal Oxide Nanoparticles in Gas and Liquid Phase Environments. J. Phys. Chem. C 2008, 112, 18303–18313. [Google Scholar] [CrossRef]
- Javed, R.; Zia, M.; Naz, S.; Aisida, S.O.; Ain, N.U.; Ao, Q. Role of capping agents in the application of nanoparticles in bio-medicine and environmental remediation: Recent trends and future prospects. J. Nanobiotechnology 2020, 18, 172. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ma, X.-L.; Gu, Y.; Huang, H.; Zhang, G.-W. Green Synthesis of Metallic Nanoparticles and Their Potential Applications to Treat Cancer. Front. Chem. 2020, 8, 799. [Google Scholar] [CrossRef]
- Vanlalveni, C.; Lallianrawna, S.; Biswas, A.; Selvaraj, M.; Changmai, B.; Rokhum, S.L. Green synthesis of silver nanoparticles using plant extracts and their antimicrobial activities: A review of recent literature. RSC Adv. 2021, 11, 2804–2837. [Google Scholar] [CrossRef]
- Xie, X.; Liao, J.; Shao, X.; Li, Q.; Lin, Y. The Effect of shape on Cellular Uptake of Gold Nanoparticles in the forms of Stars, Rods, and Triangles. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Lee, S.H.; Jun, B.-H. Silver Nanoparticles: Synthesis and Application for Nanomedicine. Int. J. Mol. Sci. 2019, 20, 865. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; El-Sayed, M.A. Gold nanoparticles: Optical properties and implementations in cancer diagnosis and photothermal therapy. J. Adv. Res. 2010, 1, 13–28. [Google Scholar] [CrossRef]
- De Freitas, L.F.; Varca, G.H.C.; Batista, J.G.D.S.; Lugão, A.B. An Overview of the Synthesis of Gold Nanoparticles Using Radiation Technologies. Nanomaterials 2018, 8, 939. [Google Scholar] [CrossRef] [PubMed]
- De Leersnyder, I.; De Gelder, L.; Van Driessche, I.; Vermeir, P. Revealing the Importance of Aging, Environment, Size and Stabilization Mechanisms on the Stability of Metal Nanoparticles: A Case Study for Silver Nanoparticles in a Minimally Defined and Complex Undefined Bacterial Growth Medium. Nanomaterials 2019, 9, 1684. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.U.; Saleh, T.A.; Wahab, A.; Khan, M.H.U.; Khan, D.; Khan, W.U.; Rahim, A.; Kamal, S.; Khan, F.U.; Fahad, S. Nanosilver: New ageless and versatile biomedical therapeutic scaffold. Int. J. Nanomed. 2018, 13, 733–762. [Google Scholar] [CrossRef] [PubMed]
- Botteon, C.E.A.; Silva, L.B.; Ccana-Ccapatinta, G.V.; Silva, T.S.; Ambrosio, S.R.; Veneziani, R.C.S.; Bastos, J.K.; Marcato, P.D. Biosynthesis and characterization of gold nanoparticles using Brazilian red propolis and evaluation of its antimicrobial and anticancer activities. Sci. Rep. 2021, 11, 1–16. [Google Scholar] [CrossRef]
- Oves, M.; Khan, M.S.; Zaidi, A.; Ahmed, A.S.; Ahmed, F.; Ahmad, E.; Sherwani, A.; Owais, M.; Azam, A. Antibacterial and Cytotoxic Efficacy of Extracellular Silver Nanoparticles Biofabricated from Chromium Reducing Novel OS4 Strain of Stenotrophomonas maltophilia. PLoS ONE 2013, 8, e59140. [Google Scholar] [CrossRef]
- Wei, Q.-Y.; Xu, Y.-M.; Lau, A.T.Y. Recent Progress of Nanocarrier-Based Therapy for Solid Malignancies. Cancers 2020, 12, 2783. [Google Scholar] [CrossRef]
- Minami, H.; Kiyota, N.; Kimbara, S.; Ando, Y.; Shimokata, T.; Ohtsu, A.; Fuse, N.; Kuboki, Y.; Shimizu, T.; Yamamoto, N.; et al. Guidelines for clinical evaluation of anti-cancer drugs. Cancer Sci. 2021, 112, 2563–2577. [Google Scholar] [CrossRef]
- De Dios-Figueroa, G.T.; Aguilera-Marquez, J.d.R.; Camacho-Villegas, T.A.; Lugo-Fabres, P.H. 3d cell culture models in covid-19 times: A review of 3D technologies to understand and accelerate therapeutic drug discovery. Biomedicines 2021, 9, 602. [Google Scholar] [CrossRef]
- Fan, H.; Demirci, U.; Chen, P. Emerging organoid models: Leaping forward in cancer research. J. Hematol. Oncol. 2019, 12, 1–10. [Google Scholar] [CrossRef]
- Lesko, J.; Triebl, A.; Stacher-Priehse, E.; Fink-Neuböck, N.; Lindenmann, J.; Smolle-Jüttner, F.-M.; Köfeler, H.C.; Hrzenjak, A.; Olschewski, H.; Leithner, K. Phospholipid dynamics in ex vivo lung cancer and normal lung explants. Exp. Mol. Med. 2021, 53, 81–90. [Google Scholar] [CrossRef]
- Soltani, M.; Kashkooli, F.M.; Souri, M.; Harofte, S.Z.; Harati, T.; Khadem, A.; Pour, M.H.; Raahemifar, K. Enhancing Clinical Translation of Cancer Using Nanoinformatics. Cancers 2021, 13, 2481. [Google Scholar] [CrossRef] [PubMed]
- Kashkooli, F.M.; Soltani, M.; Souri, M.; Meaney, C.; Kohandel, M. Nexus between in silico and in vivo models to enhance clinical translation of nanomedicine. Nano Today 2021, 36, 101057. [Google Scholar] [CrossRef]
- Jeyarani, S.; Vinita, N.M.; Puja, P.; Senthamilselvi, S.; Devan, U.; Velangani, A.J.; Biruntha, M.; Pugazhendhi, A.; Kumar, P. Biomimetic gold nanoparticles for its cytotoxicity and biocompatibility evidenced by fluorescence-based assays in cancer (MDA-MB-231) and non-cancerous (HEK-293) cells. J. Photochem. Photobiol. B Biol. 2020, 202, 111715. [Google Scholar] [CrossRef] [PubMed]
- Manivasagan, P.; Venkatesan, J.; Senthilkumar, K.; Sivakumar, K.; Kim, S.-K. Biosynthesis, Antimicrobial and Cytotoxic Effect of Silver Nanoparticles Using a NovelNocardiopsissp. BioMed Res. Int. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Han, J.W.; Eppakayala, V.; Jeyaraj, M.; Kim, J.-H. Cytotoxicity of Biologically Synthesized Silver Nanoparticles in MDA-MB-231 Human Breast Cancer Cells. BioMed Res. Int. 2013, 2013, 535796. [Google Scholar] [CrossRef] [PubMed]
- Kumar, C.G.; Poornachandra, Y.; Mamidyala, S.K. Green synthesis of bacterial gold nanoparticles conjugated to resveratrol as delivery vehicles. Colloids Surf. B Biointerfaces 2014, 123, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Tolosa, L.; Donato, M.T.; Gómez-Lechón, M.J. General Cytotoxicity Assessment by Means of the MTT Assay. In Microglia; Humana Press: New York, NY, USA, 2015; Volume 1250, pp. 333–348. [Google Scholar]
- Ahn, E.-Y.; Jin, H.; Park, Y. Green synthesis and biological activities of silver nanoparticles prepared by Carpesium cernuum extract. Arch. Pharmacal Res. 2019, 42, 926–934. [Google Scholar] [CrossRef]
- Ahn, E.-Y.; Park, Y. Anticancer prospects of silver nanoparticles green-synthesized by plant extracts. Mater. Sci. Eng. C 2020, 116, 111253. [Google Scholar] [CrossRef]
- Liu, J.; Liang, H.; Li, M.; Luo, Z.; Zhang, J.; Guo, X.; Cai, K. Tumor acidity activating multifunctional nanoplatform for NIR-mediated multiple enhanced photodynamic and photothermal tumor therapy. Biomaterials 2018, 157, 107–124. [Google Scholar] [CrossRef] [PubMed]
- Louis, K.S.; Siegel, A.C. Cell Viability Analysis Using Trypan Blue: Manual and Automated Methods. Microglia 2011, 740, 7–12. [Google Scholar] [CrossRef]
- Strober, W. Trypan Blue Exclusion Test of Cell Viability. Curr. Protoc. Immunol. 2015, 111, A3.B.1–A3.B.3. [Google Scholar] [CrossRef]
- Mendes, R.; Pedrosa, P.; Lima, J.C.; Fernandes, A.R.; Baptista, P.V. Photothermal enhancement of chemotherapy in breast cancer by visible irradiation of Gold Nanoparticles. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Karuppaiya, P.; Satheeshkumar, E.; Chao, W.-T.; Kao, L.-Y.; Chen, E.C.-F.; Tsay, H.-S. Anti-metastatic activity of biologically synthesized gold nanoparticles on human fibrosarcoma cell line HT-1080. Colloids Surf. B Biointerfaces 2013, 110, 163–170. [Google Scholar] [CrossRef]
- Repetto, G.; Del Peso, A.; Zurita, J.L. Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat. Protoc. 2008, 3, 1125–1131. [Google Scholar] [CrossRef]
- El-Hawary, S.; El-Hefnawy, H.; Mokhtar, F.A.; Sobeh, M.; Mostafa, E.; Osman, S.; El-Raey, M. Green Synthesis of Silver Nanoparticles Using Extract of Jasminum officinal L. Leaves and Evaluation of Cytotoxic Activity Towards Bladder (5637) and Breast Cancer (MCF-7) Cell Lines. Int. J. Nanomed. 2020, 15, 9771–9781. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Nagarajan, A.; Uchil, P. Analysis of Cell Viability by the Lactate Dehydrogenase Assay. Cold Spring Harb. Protoc. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Arantes-Rodrigues, R.; Colaço, A.; Pinto-Leite, R.; Oliveira, P.A. In vitro and in vivo experimental models as tools to investigate the efficacy of antineoplastic drugs on urinary bladder cancer. Anticancer Res. 2013, 33, 1273–1296. [Google Scholar]
- Ahmed, T.; Shahid, M.; Noman, M.; Niazi, M.B.K.; Zubair, M.; Almatroudi, A.; Khurshid, M.; Tariq, F.; Mumtaz, R.; Li, B. Bioprospecting a native silver-resistant Bacillus safensis strain for green synthesis and subsequent antibacterial and anticancer activities of silver nanoparticles. J. Adv. Res. 2020, 24, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Ben Tahar, I.; Fickers, P.; Dziedzic, A.; Płoch, D.; Skóra, B.; Kus-Liśkiewicz, M. Green pyomelanin-mediated synthesis of gold nanoparticles: Modelling and design, physico-chemical and biological characteristics. Microb. Cell Factories 2019, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, R.; Manikandan, B.; Raman, T.; Arunagirinathan, K.; Prabhu, N.M.; Basu, J.; Perumal, M.; Palanisamy, S.; Munusamy, A. Biosynthesis of silver nanoparticles using ethanolic petals extract of Rosa indica and characterization of its antibacterial, anticancer and anti-inflammatory activities. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 138, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Pijuan, J.; Barceló, C.; Moreno, D.F.; Maiques, O.; Sisó, P.; Marti, R.M.; Macià, A.; Panosa, A. In vitro Cell Migration, Invasion, and Adhesion Assays: From Cell Imaging to Data Analysis. Front. Cell Dev. Biol. 2019, 7, 107. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Lei, P.-J.; Padera, T. Progression of Metastasis through Lymphatic System. Cells 2021, 10, 627. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ma, D.; Chen, W.; Chen, X.; Qian, Y.; Zhao, Y.; Hu, T.; Yin, R.; Zhu, Y.; Zhang, Y.; et al. Gold Nanoparticles Suppressed Proliferation, Migration, and Invasion in Papillary Thyroid Carcinoma Cells via Downregulation of CCT. J. Nanomater. 2019, 2019, 1–12. [Google Scholar] [CrossRef]
- Nie, C.; Du, P.; Zhao, H.; Xie, H.; Li, Y.; Yao, L.; Shi, Y.; Hu, L.; Si, S.; Zhang, M.; et al. Ag@TiO2 Nanoprisms with Highly Efficient Near-Infrared Photothermal Conversion for Melanoma Therapy. Chem. Asian J. 2019, 15, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Uggeri, J.; Gatti, R.; Belletti, S.; Scandroglio, R.; Corradini, R.; Rotoli, B.M.; Orlandini, G. Calcein-AM is a detector of intracellular oxidative activity. Histochem. Cell Biol. 2000, 122, 499–505. [Google Scholar] [CrossRef]
- Intracellular hydrogen peroxide and superoxide anion detection in endothelial cells. J. Leukoc. Biol. 1994, 55, 253–258. [CrossRef]
- Żamojć, K.; Zdrowowicz, M.; Rudnicki-Velasquez, P.B.; Krzymiński, K.; Zaborowski, B.; Niedziałkowski, P.; Jacewicz, D.; Chmurzyński, L. The development of 1,3-diphenylisobenzofuran as a highly selective probe for the detection and quantitative determination of hydrogen peroxide. Free Radic. Res. 2017, 51, 38–46. [Google Scholar] [CrossRef]
- Bi, S.; Zhou, H.; Wu, J.; Sun, X. Micronomicin/tobramycin binding with DNA: Fluorescence studies using of ethidium bromide as a probe and molecular docking analysis. J. Biomol. Struct. Dyn. 2018, 37, 1464–1476. [Google Scholar] [CrossRef]
- Vidhya, M.S.; Ameen, F.; Dawoud, T.; Yuvakkumar, R.; Ravi, G.; Kumar, P.; Velauthapillai, D. Anti-cancer applications of Zr, Co, Ni-doped ZnO thin nanoplates. Mater. Lett. 2020, 283, 128760. [Google Scholar] [CrossRef]
- Liegler, T.J.; Hyun, W.; Yen, T.S.; Stites, D.P. Detection and quantification of live, apoptotic, and necrotic human peripheral lymphocytes by single-laser flow cytometry. Clin. Diagn. Lab. Immunol. 1995, 2, 369–376. [Google Scholar] [CrossRef]
- Kumar, R.; Saneja, A.; Panda, A.K. An Aannexin V-FITC—Propidium iodide-based method for detecting apoptosis in a non-small cell lung cancer cell line. Methods Mol. Biol. 2021, 2279, 213–223. [Google Scholar]
- Vermes, I.; Haanen, C.; Steffens-Nakken, H.; Reutellingsperger, C. A novel assay for apoptosis Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J. Immunol. Methods 1995, 184, 39–51. [Google Scholar] [CrossRef]
- Jawaid, P.; Rehman, M.U.; Zhao, Q.-L.; Misawa, M.; Ishikawa, K.; Hori, M.; Shimizu, T.; Saitoh, J.; Noguchi, K.; Kondo, T. Small size gold nanoparticles enhance apoptosis-induced by cold atmospheric plasma via depletion of intracellular GSH and modification of oxidative stress. Cell Death Discov. 2020, 6, 83. [Google Scholar] [CrossRef]
- Boucher, D.; Duclos, C.; Denault, J.-B. General In Vitro Caspase Assay Procedures. Adv. Struct. Saf. Stud. 2014, 1133, 3–39. [Google Scholar] [CrossRef]
- Darzynkiewicz, Z.; Juan, G.; Bedner, E. Determining Cell Cycle Stages by Flow Cytometry. Curr. Protoc. Cell Biol. 1999, 1, 8.4.1.–8.4.18. [Google Scholar] [CrossRef] [PubMed]
- Nunes, A.S.; Barros, A.S.; Costa, E.C.; Moreira, A.F.; Correia, I.J. 3D tumor spheroids as in vitro models to mimic in vivo human solid tumors resistance to therapeutic drugs. Biotechnol. Bioeng. 2019, 116, 206–226. [Google Scholar] [CrossRef] [PubMed]
- Mikhail, A.S.; Eetezadi, S.; Allen, C. Multicellular Tumor Spheroids for Evaluation of Cytotoxicity and Tumor Growth Inhibitory Effects of Nanomedicines In Vitro: A Comparison of Docetaxel-Loaded Block Copolymer Micelles and Taxotere®. PLoS ONE 2013, 8, e62630. [Google Scholar] [CrossRef] [PubMed]
- Langhans, S.A. Three-Dimensional in Vitro Cell Culture Models in Drug Discovery and Drug Repositioning. Front. Pharmacol. 2018, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.; Teng, Y. Is It time to start transitioning from 2D to 3D cell culture? Front. Mol. Biosci. 2020, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Vemuri, S.K.; Banala, R.R.; Mukherjee, S.; Uppula, P.; GPV, S.; Gurava Reddy, A.V.; Malarvilli, T. Novel biosynthesized gold nanoparticles as anti-cancer agents against breast cancer: Synthesis, biological evaluation, molecular modelling studies. Mater. Sci. Eng. C 2019, 99, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, K.; Das, S.; Choudhury, P.; Ghosh, S.; Baral, R.; Choudhuri, S.K. A Novel Approach of Synthesizing and Evaluating the Anticancer Potential of Silver Oxide Nanoparticles in vitro. Chemotherapy 2017, 62, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Souza, W.; Piperni, S.G.; Laviola, P.; Rossi, A.; Rossi, M.I.D.; Archanjo, B.; Leite, P.E.; Fernandes, M.H.; Rocha, L.A.; Granjeiro, J.M.; et al. The two faces of titanium dioxide nanoparticles bio-camouflage in 3D bone spheroids. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Roma-Rodrigues, C.; Pombo, I.; Fernandes, A.; Baptista, P. Hyperthermia Induced by Gold Nanoparticles and Visible Light Photothermy Combined with Chemotherapy to Tackle Doxorubicin Sensitive and Resistant Colorectal Tumor 3D Spheroids. Int. J. Mol. Sci. 2020, 21, 8017. [Google Scholar] [CrossRef]
- Wang, X.; Wei, F.; Liu, A.; Wang, L.; Wang, J.-C.; Ren, L.; Liu, W.; Tu, Q.; Li, L.; Wang, J. Cancer stem cell labeling using poly(l-lysine)-modified iron oxide nanoparticles. Biomaterials 2012, 33, 3719–3732. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.; Huynh, E.; Tang, S.; Uskoković, V. Brain and bone cancer targeting by a ferrofluid composed of superparamagnetic iron-oxide/silica/carbon nanoparticles (earthicles). Acta Biomater. 2019, 88, 422–447. [Google Scholar] [CrossRef]
- Arora, N.; Shome, R.; Ghosh, S.S. Deciphering therapeutic potential of PEGylated recombinant PTEN-silver nanoclusters ensemble on 3D spheroids. Mol. Biol. Rep. 2019, 46, 5103–5112. [Google Scholar] [CrossRef]
- Leite, P.E.C.; Pereira, M.; Harris, G.; Pamies, D.; Dos Santos, L.M.G.; Granjeiro, J.; Hogberg, H.T.; Hartung, T.; Smirnova, L. Suitability of 3D human brain spheroid models to distinguish toxic effects of gold and poly-lactic acid nanoparticles to assess biocompatibility for brain drug delivery. Part. Fibre Toxicol. 2019, 16, 1–20. [Google Scholar] [CrossRef]
- Chen, L.; Wu, M.; Jiang, S.; Zhang, Y.; Li, R.; Lu, Y.; Liu, L.; Wu, G.; Liu, Y.; Xie, L.; et al. Skin Toxicity Assessment of Silver Nanoparticles in a 3D Epidermal Model Compared to 2D Keratinocytes. Int. J. Nanomed. 2019, 14, 9707–9719. [Google Scholar] [CrossRef]
- Kokkinos, J.; Sharbeen, G.; Haghighi, K.S.; Ignacio, R.M.C.; Kopecky, C.; Gonzales-Aloy, E.; Youkhana, J.; Timpson, P.; Pereira, B.A.; Ritchie, S.; et al. Ex vivo culture of intact human patient derived pancreatic tumour tissue. Sci. Rep. 2021, 11, 1–15. [Google Scholar] [CrossRef]
- Wick, P.; Chortarea, S.; Guenat, O.T.; Roesslein, M.; Stucki, J.D.; Hirn, S.; Fink, A.; Rothen-Rutishauser, B. In vitro-ex vivo model systems for nanosafety assessment. Eur. J. Nanomed. 2015, 7, 7. [Google Scholar] [CrossRef][Green Version]
- Gokulan, K.; Williams, K.; Orr, S.; Khare, S. Human Intestinal Tissue Explant Exposure to Silver Nanoparticles Reveals Sex Dependent Alterations in Inflammatory Responses and Epithelial Cell Permeability. Int. J. Mol. Sci. 2020, 22, 9. [Google Scholar] [CrossRef] [PubMed]
- Maldonado-Ortega, D.A.; Navarro-Tovar, G.; Martínez-Castañón, G.; Gonzalez, C. Effect of gold nanoparticles (AuNPs) on isolated rat tracheal segments. Toxicol. Rep. 2021, 8, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Von Goetz, N.; Bachler, G.; Hungerbühler, K. A physiologically based pharmacokinetic model for ionic silver and silver nanoparticles. Int. J. Nanomed. 2013, 8, 3365–3382. [Google Scholar] [CrossRef] [PubMed]
- Christensen, F.M.; Johnston, H.J.; Stone, V.; Aitken, R.J.; Hankin, S.; Peters, S.; Aschberger, K. Nano-silver–feasibility and challenges for human health risk assessment based on open literature. Nanotoxicology 2010, 4, 284–295. [Google Scholar] [CrossRef] [PubMed]
- Ko, E.A.; Song, M.Y.; Donthamsetty, R.; Makino, A.; Yuan, J.X.-J. Tension measurement in isolated rat and mouse pulmonary artery. Drug Discov. Today: Dis. Model. 2010, 7, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Cameron, J.N. Principles of Physiological Measurement; Elsevier: Amsterdam, The Netherlands, 1988; ISBN 9780121569556. [Google Scholar]
- Mohamed, S.A.; Radtke, A.; Saraei, R.; Bullerdiek, J.; Sorani, H.; Nimzyk, R.; Karluss, A.; Sievers, H.H.; Belge, G. Locally Different Endothelial Nitric Oxide Synthase Protein Levels in Ascending Aortic Aneurysms of Bicuspid and Tricuspid Aortic Valve. Cardiol. Res. Pract. 2012, 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.R.; Lunardi, C.N.; Araki, K.; Biazzotto, J.C.; Da Silva, R.S.; Bendhack, L.M. Gold nanoparticle modifies nitric oxide release and vasodilation in rat aorta. J. Chem. Biol. 2014, 7, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Meiring, J.J.; Borm, P.J.; Bagaté, K.; Semmler, M.; Seitz, J.; Takenaka, S.; Kreyling, W.G. The influence of hydrogen peroxide and histamine on lung permeability and translocation of iridium nanoparticles in the isolated perfused rat lung. Part. Fibre Toxicol. 2005, 2, 3. [Google Scholar] [CrossRef]
- Ramirez-Lee, M.A.; Espinosa-Tanguma, R.; Mejía-Elizondo, R.; Medina-Hernández, A.; Martinez-Castañon, G.A.; Gonzalez, C. Effect of silver nanoparticles upon the myocardial and coronary vascular function in isolated and perfused diabetic rat hearts. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2587–2596. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.M.; Mocanu, M.M.; Yellon, D.M. Retrograde heart perfusion: The Langendorff technique of isolated heart perfusion. J. Mol. Cell. Cardiol. 2011, 50, 940–950. [Google Scholar] [CrossRef] [PubMed]
- Dhein, S. The Langendorff Heart. In Practical Methods in Cardiovascular Research; Dhein, S., Mohr, F.W., Delmar, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 155–172. ISBN 978-3-540-26574-0. [Google Scholar]
- Skrzypiec-Spring, M.; Grotthus, B.; Szeląg, A.; Schulz, R. Isolated heart perfusion according to Langendorff—Still viable in the new millennium. J. Pharmacol. Toxicol. Methods 2007, 55, 113–126. [Google Scholar] [CrossRef] [PubMed]
- González, C.; Salazar-García, S.; Palestino, G.; Martínez-Cuevas, P.P.; Lee, M.A.R.; Jurado-Manzano, B.B.; Rosas-Hernandez, H.; Gaytán-Pacheco, N.; Martel, G.; Espinosa-Tanguma, R.; et al. Effect of 45nm silver nanoparticles (AgNPs) upon the smooth muscle of rat trachea: Role of nitric oxide. Toxicol. Lett. 2011, 207, 306–313. [Google Scholar] [CrossRef]
- Gonzalez, C.; Rosas-Hernandez, H.; Lee, M.A.R.; Salazar-García, S.; Ali, S.F. Role of silver nanoparticles (AgNPs) on the cardiovascular system. Arch. Toxicol. 2014, 90, 493–511. [Google Scholar] [CrossRef]
- Ramirez-Lee, M.A.; Aguirre-Bañuelos, P.; Martinez-Cuevas, P.P.; Espinosa-Tanguma, R.; Chi-Ahumada, E.; Martinez-Castañon, G.A.; Gonzalez, C. Evaluation of cardiovascular responses to silver nanoparticles (AgNPs) in spontaneously hypertensive rats. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 385–395. [Google Scholar] [CrossRef]
- Rosas-Hernandez, H.; Jiménez-Badillo, S.; Martínez-Cuevas, P.P.; Gracia-Espino, E.; Terrones, H.; Terrones, M.; Hussain, S.M.; Ali, S.F.; González, C. Effects of 45-nm silver nanoparticles on coronary endothelial cells and isolated rat aortic rings. Toxicol. Lett. 2009, 191, 305–313. [Google Scholar] [CrossRef]
- Holen, I.; Speirs, V.; Morrissey, B.; Blyth, K. In vivo models in breast cancer research: Progress, challenges and future directions. Dis. Model. Mech. 2017, 10, 359–371. [Google Scholar] [CrossRef]
- Guerin, M.V.; Finisguerra, V.; Eynde, B.J.V.D.; Bercovici, N.; Trautmann, A. Preclinical murine tumor models: A structural and functional perspective. eLife 2020, 9, 1–24. [Google Scholar] [CrossRef]
- Kyriakakis, E.; Markaki, M.; Tavernarakis, N. Caenorhabditis elegans as a model for cancer research. Mol. Cell. Oncol. 2014, 2, 2. [Google Scholar] [CrossRef]
- Harris, T.W. WormBase: A multi-species resource for nematode biology and genomics. Nucleic Acids Res. 2004, 32, 411D–417. [Google Scholar] [CrossRef]
- Hason, M.; Bartůnĕk, P. Zebrafish models of cancer-new insights on modeling human cancer in a non-mammalian vertebrate. Genes 2019, 10, 935. [Google Scholar] [CrossRef] [PubMed]
- Tsyusko, O.V.; Unrine, J.M.; Spurgeon, D.; Blalock, E.; Starnes, D.; Tseng, M.; Joice, G.; Bertsch, P.M. Toxicogenomic Responses of the Model Organism Caenorhabditis elegans to Gold Nanoparticles. Environ. Sci. Technol. 2012, 46, 4115–4124. [Google Scholar] [CrossRef]
- Hu, C.-C.; Wu, G.-H.; Lai, S.-F.; Shanmugam, M.M.; Hwu, Y.; Wagner, O.I.; Yen, T.-J. Toxic Effects of Size-tunable Gold Nanoparticles on Caenorhabditis elegans Development and Gene Regulation. Sci. Rep. 2018, 8, 15245. [Google Scholar] [CrossRef] [PubMed]
- Botha, T.L.; Brand, S.J.; Ikenaka, Y.; Nakayama, S.M.; Ishizuka, M.; Wepener, V. How toxic is a non-toxic nanomaterial: Behaviour as an indicator of effect in Danio rerio exposed to nanogold. Aquat. Toxicol. 2019, 215, 105287. [Google Scholar] [CrossRef] [PubMed]
- Viau, C.; Haçariz, O.; Karimian, F.; Xia, J. Comprehensive phenotyping and transcriptome profiling to study nanotoxicity inC. elegans. PeerJ 2020, 8, e8684. [Google Scholar] [CrossRef] [PubMed]
- Griffitt, R.J.; Lavelle, C.M.; Kane, A.S.; Denslow, N.D.; Barber, D.S. Chronic nanoparticulate silver exposure results in tissue accumulation and transcriptomic changes in zebrafish. Aquat. Toxicol. 2013, 130-131, 192–200. [Google Scholar] [CrossRef]
- Cambier, S.; Røgeberg, M.; Georgantzopoulou, A.; Serchi, T.; Karlsson, C.; Verhaegen, S.; Iversen, T.-G.; Guignard, C.; Kruszewski, M.; Hoffmann, L.; et al. Fate and effects of silver nanoparticles on early life-stage development of zebrafish (Danio rerio) in comparison to silver nitrate. Sci. Total. Environ. 2018, 610, 972–982. [Google Scholar] [CrossRef]
- Zhao, G.; Wang, Z.; Xu, L.; Xia, C.-X.; Liu, J.-X. Silver nanoparticles induce abnormal touch responses by damaging neural circuits in zebrafish embryos. Chemosphere 2019, 229, 169–180. [Google Scholar] [CrossRef]
- Ghobashy, M.M.; Elkodous, M.A.; Shabaka, S.H.; Younis, S.A.; Alshangiti, D.M.; Madani, M.; Al-Gahtany, S.A.; Elkhatib, W.F.; Noreddin, A.M.; Nady, N.; et al. An overview of methods for production and detection of silver nanoparticles, with emphasis on their fate and toxicological effects on human, soil, and aquatic environment. Nanotechnol. Rev. 2021, 10, 954–977. [Google Scholar] [CrossRef]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; Van De Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Mural, R.J.; Adams, M.D.; Myers, E.W.; Smith, H.O.; Miklos, G.L.G.; Wides, R.; Halpern, A.; Li, P.W.; Sutton, G.G.; Nadeau, J.; et al. A Comparison of Whole-Genome Shotgun-Derived Mouse Chromosome 16 and the Human Genome. Science 2002, 296, 1661–1671. [Google Scholar] [CrossRef]
- Li, Z.; Zheng, W.; Wang, H.; Cheng, Y.; Fang, Y.; Wu, F.; Sun, G.; Sun, G.; Lv, C.; Hui, B. Application of Animal Models in Cancer Research: Recent Progress and Future Prospects. Cancer Manag. Res. 2021, 13, 2455–2475. [Google Scholar] [CrossRef]
- Blaas, L.; Pucci, F.; Messal, H.A.; Andersson, A.B.; Ruiz, E.J.; Gerling, M.; Douagi, I.; Spencer-Dene, B.; Musch, A.; Mitter, R.; et al. Lgr6 labels a rare population of mammary gland progenitor cells that are able to originate luminal mammary tumours. Nat. Cell Biol. 2016, 18, 1346–1356. [Google Scholar] [CrossRef] [PubMed]
- Arun, G.; Diermeier, S.; Akerman, M.; Chang, K.-C.; Wilkinson, J.E.; A Hearn, S.; Kim, Y.; MacLeod, A.R.; Krainer, A.; Norton, L.; et al. Differentiation of mammary tumors and reduction in metastasis uponMalat1lncRNA loss. Genes Dev. 2016, 30, 34–51. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.M. GFP imaging of tumor growth, metastasis, and angiogenesis in mouse. Lancet 2002, 3, 546–556. [Google Scholar] [CrossRef]
- Hendi, A.A.; El-Nagar, D.M.; Awad, M.A.; Ortashi, K.M.; Alnamlah, R.A.; Merghani, N.M. Green nanogold activity in experimental breast carcinoma in vivo. Biosci. Rep. 2020, 40, 1–12. [Google Scholar] [CrossRef]
- Khoobchandani, M.; Katti, K.K.; Karikachery, A.R.; Thipe, V.C.; Srisrimal, D.; Mohandoss, D.K.D.; Darshakumar, R.D.; Joshi, C.M.; Katti, K.V. New Approaches in Breast Cancer Therapy Through Green Nanotechnology and Nano-Ayurvedic Medicine–Pre-Clinical and Pilot Human Clinical Investigations. Int. J. Nanomed. 2020, 15, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.Q.; Shao, G.; Gan, F.; Ye, G. One-step, Rapid and Green Synthesis of Multifunctional Gold Nanoparticles for Tumor-Targeted Imaging and Therapy. Nanoscale Res. Lett. 2020, 15, 15–29. [Google Scholar] [CrossRef]
- He, Y.; Du, Z.; Ma, S.; Liu, Y.; Li, D.; Huang, H.; Jiang, S.; Cheng, S.; Wu, W.; Zhang, K.; et al. Effects of green-synthesized silver nanoparticles on lung cancer cells in vitro and grown as xenograft tumors in vivo. Int. J. Nanomed. 2016, 11, 1879–1887. [Google Scholar] [CrossRef]
- Murugesan, K.; Koroth, J.; Srinivasan, P.P.; Singh, A.; Mukundan, S.; Karki, S.S.; Choudhary, B.; Gupta, C.M. Effects of green synthesised silver nanoparticles (ST06-AgNPs) using curcumin derivative (ST06) on human cervical cancer cells (HeLa) in vitro and EAC tumor bearing mice models. Int. J. Nanomed. 2019, 14, 5257–5270. [Google Scholar] [CrossRef]
- El-Naggar, N.E.-A.; Hussein, M.H.; El-Sawah, A.A. Phycobiliprotein-mediated synthesis of biogenic silver nanoparticles, characterization, in vitro and in vivo assessment of anticancer activities. Sci. Rep. 2018, 8, 8925. [Google Scholar] [CrossRef]
- Rahimi-Moghaddam, F.; Azarpira, N.; Sattarahmady, N. Evaluation of a nanocomposite of PEG-curcumin-gold nanoparticles as a near-infrared photothermal agent: An in vitro and animal model investigation. Lasers Med Sci. 2018, 33, 1769–1779. [Google Scholar] [CrossRef]
- Ahmeda, A.; Zangeneh, A.; Zangeneh, M.M. Green synthesis and chemical characterization of gold nanoparticle synthesized using Camellia sinensis leaf aqueous extract for the treatment of acute myeloid leukemia in comparison to daunorubicin in a leukemic mouse model. Appl. Organomet. Chem. 2020, 34, 1–13. [Google Scholar] [CrossRef]
- Jones, O.T.; Ranmuthu, C.K.I.; Hall, P.N.; Funston, G.; Walter, F. Recognising Skin Cancer in Primary Care. Adv. Ther. 2019, 37, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Linos, E.; Katz, K.A.; Colditz, G.A. Skin cancer-The importance of prevention. JAMA Intern. Med. 2016, 176, 1435–1436. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Dorrell, D.N.; Strowd, L.C. Skin Cancer Detection Technology. Dermatol. Clin. 2019, 37, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.; Holman, D.M.; Maguire-Eisen, M. Ultraviolet Radiation Exposure and Its Impact on Skin Cancer Risk. Semin. Oncol. Nurs. 2016, 32, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Brandt, M.G.; Moore, C.C. Nonmelanoma Skin Cancer. Facial Plast. Surg. Clin. N. Am. 2019, 27, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.; Berman, R. The Landmark Series: Non-melanoma Skin Cancers. Ann. Surg. Oncol. 2019, 27, 22–27. [Google Scholar] [CrossRef]
- Tanese, K. Diagnosis and Management of Basal Cell Carcinoma. Curr. Treat. Options Oncol. 2019, 20, 13. [Google Scholar] [CrossRef] [PubMed]
- Bagde, A.; Mondal, A.; Singh, M. Drug delivery strategies for chemoprevention of UVB-induced skin cancer: A review. Photodermatol. Photoimmunol. Photomed. 2018, 34, 60–68. [Google Scholar] [CrossRef]
- Wu, F.; Zhu, J.; Li, G.; Wang, J.; Veeraraghavan, V.P.; Mohan, S.K.; Zhang, Q. Biologically synthesized green gold nanoparticles from Siberian ginseng induce growth-inhibitory effect on melanoma cells (B16). Artif. Cells Nanomed. Biotechnol. 2019, 47, 3297–3305. [Google Scholar] [CrossRef]
- González-Pedroza, M.; Argueta-Figueroa, L.; García-Contreras, R.; Jiménez-Martínez, Y.; Martínez-Martínez, E.; Navarro-Marchal, S.; Marchal, J.; Morales-Luckie, R.; Boulaiz, H. Silver Nanoparticles from Annona muricata Peel and Leaf Extracts as a Potential Potent, Biocompatible and Low Cost Antitumor Tool. Nanomaterials 2021, 11, 1273. [Google Scholar] [CrossRef]
- Safwat, M.A.; Soliman, G.M.; Sayed, D.; Attia, M.A. Fluorouracil-Loaded Gold Nanoparticles for the Treatment of Skin Cancer: Development, in Vitro Characterization, and in Vivo Evaluation in a Mouse Skin Cancer Xenograft Model. Mol. Pharm. 2018, 15, 2194–2205. [Google Scholar] [CrossRef]
- Kim, D.; Amatya, R.; Hwang, S.; Lee, S.; Min, K.; Shin, M. BSA-Silver Nanoparticles: A Potential Multimodal Therapeutics for Conventional and Photothermal Treatment of Skin Cancer. Pharmaceutics 2021, 13, 575. [Google Scholar] [CrossRef]
- Liu, Y.; Bailey, J.T.; Abu-Laban, M.; Li, S.; Chen, C.; Glick, A.B.; Hayes, D.J. Photocontrolled miR-148b nanoparticles cause apoptosis, inflammation and regression of Ras induced epidermal squamous cell carcinomas in mice. Biomaterials 2020, 256, 120212. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Tang, Y.; Liu, L.; Lv, X.; Wang, Q.; Ke, H.; Deng, Y.; Yang, H.; Yang, X.; Liu, G.; et al. Size-Dependent Ag2S Nanodots for Second Near-Infrared Fluorescence/Photoacoustics Imaging and Simultaneous Photothermal Therapy. ACS Nano 2017, 11, 1848–1857. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Chang, Y.; Feng, Y.; Liu, N.; Sun, X.; Feng, Y.; Li, X.; Zhang, H. Simulated Sunlight-Mediated Photodynamic Therapy for Melanoma Skin Cancer by Titanium-Dioxide-Nanoparticle-Gold-Nanocluster-Graphene Heterogeneous Nanocomposites. Small 2017, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- Shivashankarappa, A.; Sanjay, K.R. Photodynamic therapy on skin melanoma and epidermoid carcinoma cells using conjugated 5-aminolevulinic acid with microbial synthesised silver nanoparticles. J. Drug Target. 2019, 27, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global Cancer Statistics. CA Cancer J. Clin. 2005, 55, 74–108. [Google Scholar] [CrossRef]
- American Cancer Society Cancer Treatment and Survivorship Facts and Figures 2019; American Cancer Society: Atlanta, GA, USA, 2019; pp. 1–48.
- Moo, T.-A.; Sanford, R.; Dang, C.; Morrow, M. Overview of Breast Cancer Therapy. PET Clin. 2018, 13, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Wang, L. Early Diagnosis of Breast Cancer. Sensors 2017, 17, 1572. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment: A Review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef]
- Fernández-Grijalva, A.; Aguilar-Lemarroy, A.; Jave-Suarez, L.; Gutiérrez-Ortega, A.; Godinez-Melgoza, P.; Herrera-Rodríguez, S.; Mariscal-Ramírez, I.; Martínez-Velázquez, M.; Gawinowicz, M.; Martínez-Silva, M.; et al. Alpha 2HS-glycoprotein, a tumor-associated antigen (TAA) detected in Mexican patients with early-stage breast cancer. J. Proteom. 2015, 112, 301–312. [Google Scholar] [CrossRef]
- Oaxaca-Camacho, A.; Ochoa-Mojica, O.; Aguilar-Lemarroy, A.; Jave-Suárez, L.; Muñoz-Valle, J.; Padilla-Camberos, E.; Núñez-Hernández, J.; Herrera-Rodríguez, S.; Martínez-Velázquez, M.; Carranza-Aranda, A.; et al. Serum Analysis of Women with Early-Stage Breast Cancer Using a Mini-Array of Tumor-Associated Antigens. Biosensors 2020, 10, 149. [Google Scholar] [CrossRef]
- Saravanan, M.; Vahidi, H.; Cruz, D.M.; Vernet-Crua, A.; Mostafavi, E.; Stelmach, R.; Webster, T.J.; Mahjoub, M.A.; Rashedi, M.; Barabadi, H. Emerging Antineoplastic Biogenic Gold Nanomaterials for Breast Cancer Therapeutics: A Systematic Review. Int. J. Nanomed. 2020, 15, 3577–3595. [Google Scholar] [CrossRef]
- Wypij, M.; Jędrzejewski, T.; Trzcińska-Wencel, J.; Ostrowski, M.; Rai, M.; Golińska, P. Green Synthesized Silver Nanoparticles: Antibacterial and Anticancer Activities, Biocompatibility, and Analyses of Surface-Attached Proteins. Front. Microbiol. 2021, 12, 632505. [Google Scholar] [CrossRef]
- Khateef, R.; Khadri, H.; Almatroudi, A.; Alsuhaibani, S.A.; Mobeen, S.A.; Khan, R.A. Potential in-vitro anti-breast cancer activity of green-synthesized silver nanoparticles preparation against human MCF-7 cell-lines. Adv. Nat. Sci. Nanosci. Nanotechnol. 2019, 10, 045012. [Google Scholar] [CrossRef]
- Rajawat, S.; Kurchania, R.; Rajukumar, K.; Pitale, S.; Saha, S.; Qureshi, M.S. Study of anti-cancer properties of green silver nanoparticles against MCF-7 breast cancer cell lines. Green Process. Synth. 2016, 5, 173–181. [Google Scholar] [CrossRef]
- Hamouda, R.A.; Hussein, M.H.; Abo-Elmagd, R.A.; Bawazir, S.S. Synthesis and biological characterization of silver nanoparticles derived from the cyanobacterium Oscillatoria limnetica. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Singh, A.K.; Tiwari, R.; Singh, V.K.; Singh, P.; Khadim, S.R.; Singh, U.; Laxmi; Srivastava, V.; Hasan, S.; Asthana, R. Green synthesis of gold nanoparticles from Dunaliella salina, its characterization and in vitro anticancer activity on breast cancer cell line. J. Drug Deliv. Sci. Technol. 2019, 51, 164–176. [Google Scholar] [CrossRef]
- Ullah, I.; Khalil, A.T.; Ali, M.; Iqbal, J.; Ali, W.; Alarifi, S.; Shinwari, Z.K. Green-Synthesized Silver Nanoparticles Induced Apoptotic Cell Death in MCF-7 Breast Cancer Cells by Generating Reactive Oxygen Species and Activating Caspase 3 and 9 Enzyme Activities. Oxidative Med. Cell. Longev. 2020, 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Al-Radadi, N.S. Green Biosynthesis of Flaxseed Gold Nanoparticles (Au-NPs) as Potent Anti-cancer Agent Against Breast Cancer Cells. J. Saudi Chem. Soc. 2021, 25, 101243. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Roy, B.; Shaw, P.; Mondal, P.; Mondal, M.K.; Chowdhury, P.; Bhattacharya, S.; Chattopadhyay, A. Cytotoxic effect of green synthesized silver nanoparticles in MCF7 and MDA-MB-231 human breast cancer cells in vitro. Nucleus 2020, 63, 191–202. [Google Scholar] [CrossRef]
- Moshfegh, A.; Jalali, A.; Salehzadeh, A.; Jozani, A.S. Biological synthesis of silver nanoparticles by cell-free extract of Polysiphonia algae and their anticancer activity against breast cancer MCF-7 cell lines. Micro Nano Lett. 2019, 14, 581–584. [Google Scholar] [CrossRef]
- Erdogan, O.; Abbak, M.; Demirbolat, G.M.; Birtekocak, F.; Aksel, M.; Pasa, S.; Cevik, O. Green synthesis of silver nanoparticles via Cynara scolymus leaf extracts: The characterization, anticancer potential with photodynamic therapy in MCF7 cells. PLoS ONE 2019, 14, e0216496. [Google Scholar] [CrossRef] [PubMed]
- Madakka, M.; Jayaraju, N.; Rajesh, N. Evaluating the antimicrobial activity and antitumor screening of green synthesized silver nanoparticles compounds, using Syzygium jambolanum, towards MCF7 cell line (Breast cancer cell line). J. Photochem. Photobiol. 2021, 6, 100028. [Google Scholar] [CrossRef]
- Gomathi, A.; Rajarathinam, S.X.; Sadiq, A.M.; Rajeshkumar, S. Anticancer activity of silver nanoparticles synthesized using aqueous fruit shell extract of Tamarindus indica on MCF-7 human breast cancer cell line. J. Drug Deliv. Sci. Technol. 2020, 55, 101376. [Google Scholar] [CrossRef]
- Uzmaa, M.; Sunayanab, N.; Raghavendra, V.B.; Madhu, C.S.; Shanmuganathand, R.; Brindhadevie, K. Biogenic synthesis of gold nanoparticles using Commiphora wightii and their cytotoxic effects on breast cancer cell line (MCF-7). Process. Biochem. 2020, 92, 269–276. [Google Scholar] [CrossRef]
- Xavier, H.F.M.; Nadar, V.M.; Patel, P.; Umapathy, D.; Joseph, A.V.; Manivannan, S.; Santhiyagu, P.; Pandi, B.; Muthusamy, G.; Rathinam, Y.; et al. Selective antibacterial and apoptosis-inducing effects of hybrid gold nanoparticles—A green approach. J. Drug Deliv. Sci. Technol. 2020, 59, 101890. [Google Scholar] [CrossRef]
- Li, S.; Al-Misned, F.A.; El-Serehy, H.A.; Yang, L. Green synthesis of gold nanoparticles using aqueous extract of Mentha Longifolia leaf and investigation of its anti-human breast carcinoma properties in the in vitro condition. Arab. J. Chem. 2021, 14, 102931. [Google Scholar] [CrossRef]
- Sargazi, A.; Barani, A.; Majd, M.H. Synthesis and Apoptotic Efficacy of Biosynthesized Silver Nanoparticles Using Acacia luciana Flower Extract in MCF-7 Breast Cancer Cells: Activation of Bak1 and Bclx for Cancer Therapy. BioNanoScience 2020, 10, 683–689. [Google Scholar] [CrossRef]
- Zulkifli, N.I.; Muhamad, M.; Zain, N.N.M.; Tan, W.-N.; Yahaya, N.; Bustami, Y.; Aziz, A.A.; Kamal, N.N.S.N.M. A Bottom-Up Synthesis Approach to Silver Nanoparticles Induces Anti-Proliferative and Apoptotic Activities Against MCF-7, MCF-7/TAMR-1 and MCF-10A Human Breast Cell Lines. Molecules 2020, 25, 4332. [Google Scholar] [CrossRef] [PubMed]
- Velsankar, K.; Preethi, R.; Ram, P.J.; Ramesh, M.; Sudhahar, S. Evaluations of biosynthesized Ag nanoparticles via Allium Sativum flower extract in biological applications. Appl. Nanosci. 2020, 10, 1–17. [Google Scholar] [CrossRef]
- Munawer, U.; Raghavendra, V.B.; Ningaraju, S.; Krishna, K.L.; Ghosh, A.R.; Melappa, G.; Pugazhendhi, A. Biofabrication of gold nanoparticles mediated by the endophytic Cladosporium species: Photodegradation, in vitro anticancer activity and in vivo antitumor studies. Int. J. Pharm. 2020, 588, 119729. [Google Scholar] [CrossRef] [PubMed]
- Elbialy, N.S.; Abdelfatah, E.A.; Khalil, W.A. Antitumor Activity of Curcumin-Green Synthesized Gold Nanoparticles: In Vitro Study. BioNanoScience 2019, 9, 813–820. [Google Scholar] [CrossRef]
- Divakaran, D.; Lakkakula, J.; Thakur, M.; Kumawat, M.K.; Srivastava, R. Dragon fruit extract capped gold nanoparticles: Synthesis and their differential cytotoxicity effect on breast cancer cells. Mater. Lett. 2019, 236, 498–502. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Casillas, C.E.H.; Fernández, J.M.F.; Camberos, E.P.; López, E.J.H.; Pacheco, G.L.; Velázquez, M.M. Current status of circulating protein biomarkers to aid the early detection of lung cancer. Futur. Oncol. 2014, 10, 1501–1513. [Google Scholar] [CrossRef]
- Mph, K.D.M.; Nogueira, L.; Mariotto, A.B.; Rowland, J.H.; Yabroff, K.R.; Alfano, C.M.; Jemal, A.; Kramer, J.L.; Siegel, R.L. Cancer treatment and survivorship statistics. CA Cancer J. Clin. 2019, 69, 363–385. [Google Scholar] [CrossRef]
- Zhang, X.; Tan, Z.; Jia, K.; Zhang, W.; Dang, M. Rabdosia rubescens Linn: Green synthesis of gold nanoparticles and their anticancer effects against human lung cancer cells. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2171–2178. [Google Scholar] [CrossRef]
- Kumari, R.; Saini, A.; Kumar, A.; Saini, R.V. Apoptosis induction in lung and prostate cancer cells through silver nanoparticles synthesized from Pinus roxburghii bioactive fraction. JBIC J. Biol. Inorg. Chem. 2020, 25, 23–37. [Google Scholar] [CrossRef]
- Castro-Aceituno, V.; Abbai, R.; Moon, S.S.; Ahn, S.; Mathiyalagan, R.; Kim, Y.-J.; Kim, Y.-J.; Yang, D.C. Pleuropterus multiflorus (Hasuo) mediated straightforward eco-friendly synthesis of silver, gold nanoparticles and evaluation of their anti-cancer activity on A549 lung cancer cell line. Biomed. Pharmacother. 2017, 93, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Ghosh, M.; Pandey, D.M. Method development for optimised green synthesis of gold nanoparticles from Millettia pinnata and their activity in non-small cell lung cancer cell lines. IET Nanobiotechnol. 2019, 13, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Dadashpour, M.; Firouzi-Amandi, A.; Pourhassan-Moghaddam, M.; Maleki, M.J.; Soozangar, N.; Jeddi, F.; Nouri, M.; Zarghami, N.; Pilehvar-Soltanahmadi, Y. Biomimetic synthesis of silver nanoparticles using Matricaria chamomilla extract and their potential anticancer activity against human lung cancer cells. Mater. Sci. Eng. C 2018, 92, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Hu, N.; Han, L.; Pi, Y.; Gao, Y.; Chen, K. Anticancer activity of green synthesised gold nanoparticles from Marsdenia tenacissima inhibits A549 cell proliferation through the apoptotic pathway. Artif. Cells Nanomed. Biotechnol. 2019, 47, 4012–4019. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, K.; Ahmad, H.; Manikandan, E.; Arul, K.T.; Kavitha, K.; Moodley, M.K.; Rajagopal, K.; Balabhaskar, R.; Bhaskar, M. The impact of anticancer activity upon Beta vulgaris extract mediated biosynthesized silver nanoparticles (ag-NPs) against human breast (MCF-7), lung (A549) and pharynx (Hep-2) cancer cell lines. J. Photochem. Photobiol. B Biol. 2017, 173, 99–107. [Google Scholar] [CrossRef]
- Park, J.S.; Ahn, E.-Y.; Park, Y. Asymmetric dumbbell-shaped silver nanoparticles and spherical gold nanoparticles green-synthesized by mangosteen (Garcinia mangostana) pericarp waste extracts. Int. J. Nanomed. 2017, 12, 6895–6908. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Vaseeharan, B.; Malaikozhundan, B.; Gopi, N.; Ekambaram, P.; Pachaiappan, R.; Velusamy, P.; Murugan, K.; Benelli, G.; Kumar, R.S.; et al. Therapeutic effects of gold nanoparticles synthesized using Musa paradisiaca peel extract against multiple antibiotic resistant Enterococcus faecalis biofilms and human lung cancer cells (A549). Microb. Pathog. 2017, 102, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Wang, C.; Mathiyalagan, R.; Aceituno, V.C.; Singh, P.; Ahn, S.; Wang, D.; Yang, D.C. Rapid green synthesis of silver and gold nanoparticles using Dendropanax morbifera leaf extract and their anticancer activities. Int. J. Nanomed. 2016, 11, 3691–3701. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ye, X.; Qingkui, G.; Wenliang, Q.; Wen, Z.; Ning, W.; Min, Z. Anticancer activity of green synthesised AgNPs from Cymbopogon citratus (LG) against lung carcinoma cell line. AIET Nanobiotechnol. 2019, 13, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, R.; Joseph, S.; Mathew, B. Indigofera tinctoria leaf extract mediated green synthesis of silver and gold nanoparticles and assessment of their anticancer, antimicrobial, antioxidant and catalytic properties. Artif. Cells Nanomed. Biotechnol. 2017, 46, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Fu, B.; Jiang, L.; Sun, T. Biosynthesis, characterization, and anticancer effect of plant-mediated silver nanoparticles using Coptis chinensis. Int. J. Nanomed. 2019, 14, 1969–1978. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Zhang, J.; Zhang, R.; Luo, Z.; Wang, C.; Shi, S. Gold nano particles synthesized from Magnolia officinalis and anticancer activity in A549 lung cancer cells. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3101–3109. [Google Scholar] [CrossRef]
- Selvan, D.A.; Mahendiran, D.; Kumar, R.S.; Rahiman, A.K. Garlic, green tea and turmeric extracts-mediated green synthesis of silver nanoparticles: Phytochemical, antioxidant and in vitro cytotoxicity studies. J. Photochem. Photobiol. B Biol. 2018, 180, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Kyzioła, A.; Łukasiewicz, S.; Sebastiancd, V.; Kuśtrowskia, P.; Kozieła, M.; Majdaa, D.; Cierniake, A. Towards plant-mediated chemistry–Au nanoparticles obtained using aqueous extract of Rosa damascena and their biological activity in vitro. J. Inorg. Biochem. 2021, 214, 111300. [Google Scholar] [CrossRef]
- Gnanakani, P.E.; Santhanam, P.; Premkumar, K.; Kumar, K.E.; Dhanaraju, M.D. Nannochloropsis Extract–Mediated Synthesis of Biogenic Silver Nanoparticles, Characterization and In Vitro Assessment of Antimicrobial, Antioxidant and Cytotoxic Activities. Asian Pac. J. Cancer Prev. 2019, 20, 2353–2364. [Google Scholar] [CrossRef]
- Singh, H.; Du, J.; Yi, T.-H. Green and rapid synthesis of silver nanoparticles using Borago officinalis leaf extract: Anticancer and antibacterial activities. Artif. Cells Nanomed. Biotechnol. 2016, 45, 1310–1316. [Google Scholar] [CrossRef]
- Tiloke, C.; Phulukdaree, A.; Anand, K.; Gengan, R.M.; Chuturgoon, A.A. Moringa oleifera Gold Nanoparticles Modulate Oncogenes, Tumor Suppressor Genes, and Caspase-9 Splice Variants in A549 Cells. J. Cell. Biochem. 2016, 117, 2302–2314. [Google Scholar] [CrossRef]
- Singh, H.; Du, J.; Singh, P.; Yi, T.H. Ecofriendly synthesis of silver and gold nanoparticles by Euphrasia officinalis leaf extract and its biomedical applications. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1163–1170. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, Z.; Du, X.; Liu, Y.; Alarfaj, A.; Hirad, A.; Ansari, S.; Zhang, Z. Novel green synthesis of silver nanoparticles mediated by Curcumae kwangsiensis for anti-lung cancer activities: A preclinical trial study. Arch. Med. Sci. 2021, 1–9. [Google Scholar] [CrossRef]
- Maharani, V.; Sundaramanickam, A.; Balasubramanian, T. In vitro anticancer activity of silver nanoparticle synthesized by Escherichia coli VM1 isolated from marine sediments of Ennore southeast coast of India. Enzym. Microb. Technol. 2016, 95, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Leone, G.; Buttigliero, C.; Pisano, C.; Di Stefano, R.F.; Tabbò, F.; Turco, F.; Vignani, F.; Scagliotti, G.V.; Di Maio, M.; Tucci, M. Bipolar androgen therapy in prostate cancer: Current evidences and future perspectives. Crit. Rev. Oncol. 2020, 152, 102994. [Google Scholar] [CrossRef] [PubMed]
- Kluger, J.; Roy, A.; Chao, H.H. Androgen Deprivation Therapy and Cognitive Function in Prostate Cancer. Curr. Oncol. Rep. 2020, 22, 24. [Google Scholar] [CrossRef] [PubMed]
- Clarke, N.W. Landmarks in non-hormonal pharmacological therapies for castration-resistant prostate cancer. BJU Int. 2012, 110, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Berthold, D.R.; Pond, G.R.; Soban, F.; De Wit, R.; Eisenberger, M.; Tannock, I.F. Docetaxel Plus Prednisone or Mitoxantrone Plus Prednisone for Advanced Prostate Cancer: Updated Survival in the TAX 327 Study. J. Clin. Oncol. 2008, 26, 242–245. [Google Scholar] [CrossRef]
- Bansal, D.; Reimers, M.; Knoche, E.; Pachynski, R. Immunotherapy and Immunotherapy Combinations in Metastatic Castration-Resistant Prostate Cancer. Cancers 2021, 13, 334. [Google Scholar] [CrossRef]
- Sutherland, S.I.M.; Ju, X.; Horvath, L.G.; Clark, G.J. Moving on From Sipuleucel-T: New Dendritic Cell Vaccine Strategies for Prostate Cancer. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Pugazhendhi, A.; Edison, T.N.J.I.; Karuppusamy, I.; Kathirvel, B. Inorganic nanoparticles: A potential cancer therapy for human welfare. Int. J. Pharm. 2018, 539, 104–111. [Google Scholar] [CrossRef]
- El Raey, M.A.; El-Hagrassi, A.M.; Osman, A.F.; Darwish, K.M.; Emam, M. Acalypha wilkesiana flowers: Phenolic profiling, cytotoxic activity of their biosynthesized silver nanoparticles and molecular docking study for its constituents as Topoisomerase-I inhibitors. Biocatal. Agric. Biotechnol. 2019, 20, 101243. [Google Scholar] [CrossRef]
- Aboyewa, J.A.; Sibuyi, N.R.S.; Meyer, M.; Oguntibeju, O.O. Gold Nanoparticles Synthesized Using Extracts of Cyclopia intermedia, Commonly Known as Honeybush, Amplify the Cytotoxic Effects of Doxorubicin. Nanomaterials 2021, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Sibuyi, N.R.S.; Thipe, V.C.; Panjtan-Amiri, K.; Meyer, M.; Katti, K.V. Green synthesis of gold nanoparticles using Acai berry and Elderberry extracts and investigation of their effect on prostate and pancreatic cancer cells. Nanobiomedicine 2021, 8, 184954352199531. [Google Scholar] [CrossRef] [PubMed]
- Ghodake, G.; Kim, D.-Y.; Jo, J.H.; Jang, J.; Lee, D.S. One-step green synthesis of gold nanoparticles using casein hydrolytic peptides and their anti-cancer assessment using the DU145 cell line. J. Ind. Eng. Chem. 2016, 33, 185–189. [Google Scholar] [CrossRef]
- Dhamecha, D.; Jalalpure, S.; Jadhav, K. Doxorubicin functionalized gold nanoparticles: Characterization and activity against human cancer cell lines. Process. Biochem. 2015, 50, 2298–2306. [Google Scholar] [CrossRef]
- Kanniah, P.; Chelliah, P.; Thangapandi, J.R.; Gnanadhas, G.; Mahendran, V.; Robert, M. Green synthesis of antibacterial and cytotoxic silver nanoparticles by Piper nigrum seed extract and development of antibacterial silver based chitosan nanocomposite. Int. J. Biol. Macromol. 2021, 189, 18–33. [Google Scholar] [CrossRef]
- Zhang, K.; Liu, X.; Ravi, S.O.A.S.; Ramachandran, A.; Ibrahim, I.A.A.; Nassir, A.M.; Yao, J. Synthesis of silver nanoparticles (AgNPs) from leaf extract of Salvia miltiorrhiza and its anticancer potential in human prostate cancer LNCaP cell lines. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2846–2854. [Google Scholar] [CrossRef]
- İlhan, S.; Çamli, Ç. Biogenic silver nanoparticles synthesized from Piper longum fruit extract inhibit HIF-1α/VEGF mediated angiogenesis in prostate cancer cells. Cumhur. Sci. J. 2021, 42, 236–244. [Google Scholar] [CrossRef]
- Jadhav, K.; Deore, S.; Dhamecha, D.; Hr, R.; Jagwani, S.; Jalalpure, S.; Bohara, R. Phytosynthesis of Silver Nanoparticles: Characterization, Biocompatibility Studies, and Anticancer Activity. ACS Biomater. Sci. Eng. 2018, 4, 892–899. [Google Scholar] [CrossRef]
- Patra, N.; Kar, D.; Pal, A.; Behera, A. Antibacterial, anticancer, anti-diabetic and catalytic activity of bio-conjugated metal nanoparticles. Adv. Nat. Sci. Nanosci. Nanotechnol. 2018, 9, 035001. [Google Scholar] [CrossRef]
- He, Y.; Li, X.; Wang, J.; Yang, Q.; Yao, B.; Zhao, Y.; Zhao, A.; Sun, W.; Zhang, Q. Synthesis, characterization and evaluation cytotoxic activity of silver nanoparticles synthesized by Chinese herbal Cornus officinalis via environment friendly approach. Environ. Toxicol. Pharmacol. 2017, 56, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Prasannaraj, G.; Sahi, S.V.; Benelli, G.; Venkatachalam, P. Coating with Active Phytomolecules Enhances Anticancer Activity of Bio-Engineered Ag Nanocomplex. J. Clust. Sci. 2017, 28, 2349–2367. [Google Scholar] [CrossRef]
- Bello, B.A.; Khan, S.A.; Khan, J.A.; Syed, F.Q.; Anwar, Y.; Khan, S.B. Antiproliferation and antibacterial effect of biosynthesized AgNps from leaves extract of Guiera senegalensis and its catalytic reduction on some persistent organic pollutants. J. Photochem. Photobiol. B Biol. 2017, 175, 99–108. [Google Scholar] [CrossRef]
- Netala, V.R.; Bethu, M.S.; Pushpalatha, B.; Baki, V.B.; Aishwarya, S.; Rao, J.V.; Tartte, V. Biogenesis of silver nanoparticles using endophytic fungus Pestalotiopsis microspora and evaluation of their antioxidant and anticancer activities. Int. J. Nanomed. 2016, 11, 5683–5696. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Du, Z.; Ma, S.; Cheng, S.; Jiang, S.; Liu, Y.; Li, D.; Huang, H.; Zhang, K.; Zheng, X. Biosynthesis, Antibacterial Activity and Anticancer Effects Against Prostate Cancer (PC-3) Cells of Silver Nanoparticles Using Dimocarpus Longan Lour. Peel Extract. Nanoscale Res. Lett. 2016, 11, 1–10. [Google Scholar] [CrossRef]
- Prasannaraj, G.; Sahi, S.V.; Ravikumar, S.; Venkatachalam, P. Enhanced Cytotoxicity of Biomolecules Loaded Metallic Silver Nanoparticles Against Human Liver (HepG2) and Prostate (PC3) Cancer Cell Lines. J. Nanosci. Nanotechnol. 2016, 16, 4948–4959. [Google Scholar] [CrossRef]
- Priyadharshini, R.I.; Prasannaraj, G.; Geetha, N.; Venkatachalam, P. Microwave-Mediated Extracellular Synthesis of Metallic Silver and Zinc Oxide Nanoparticles Using Macro-Algae (Gracilaria edulis) Extracts and Its Anticancer Activity Against Human PC3 Cell Lines. Appl. Biochem. Biotechnol. 2014, 174, 2777–2790. [Google Scholar] [CrossRef]
- Firdhouse, M.J.; Lalitha, P. Biosynthesis of silver nanoparticles using the extract of Alternanthera sessilis—antiproliferative effect against prostate cancer cells. Cancer Nanotechnol. 2013, 4, 137–143. [Google Scholar] [CrossRef]
- Rajeswaran, S.; Thirugnanasambandan, S.; Dewangan, N.K.; Moorthy, R.K.; Kandasamy, S.; Vilwanathan, R. Multifarious Pharmacological Applications of Green Routed Eco-Friendly Iron Nanoparticles Synthesized by Streptomyces Sp. (SRT12). Biol. Trace Elem. Res. 2019, 194, 273–283. [Google Scholar] [CrossRef]
- Naz, S.; Islam, M.; Tabassum, S.; Fernandes, N.F.; De Blanco, E.J.C.; Zia, M. Green synthesis of hematite (α-Fe2O3) nanoparticles using Rhus punjabensis extract and their biomedical prospect in pathogenic diseases and cancer. J. Mol. Struct. 2019, 1185, 1–7. [Google Scholar] [CrossRef]
- Kanagamani, K.; Muthukrishnan, P.; Saravanakumar, K.; Shankar, K.; Kathiresan, A. Photocatalytic degradation of environmental perilous gentian violet dye using leucaena-mediated zinc oxide nanoparticle and its anticancer activity. Rare Met. 2019, 38, 277–286. [Google Scholar] [CrossRef]
- He, F.; Yu, W.; Fan, X.; Jin, B. In vitro cytotoxicity of biosynthesized titanium dioxide nanoparticles in human prostate cancer cell lines. Trop. J. Pharm. Res. 2018, 16, 2793. [Google Scholar] [CrossRef][Green Version]
- Xie, Y.-H.; Chen, Y.-X.; Fang, J.-Y. Comprehensive review of targeted therapy for colorectal cancer. Signal Transduct. Target. Ther. 2020, 5, 1–30. [Google Scholar] [CrossRef]
- Takahashi, H.; Berber, E. Role of thermal ablation in the management of colorectal liver metastasis. HepatoBiliary Surg. Nutr. 2020, 9, 49–58. [Google Scholar] [CrossRef]
- Venkatadri, B.; Shanparvish, E.; Rameshkumar, M.; Arasu, M.V.; Al-Dhabi, N.A.; Ponnusamy, V.K.; Agastian, P. Green synthesis of silver nanoparticles using aqueous rhizome extract of Zingiber officinale and Curcuma longa: In-vitro anti-cancer potential on human colon carcinoma HT-29 cells. Saudi J. Biol. Sci. 2020, 27, 2980–2986. [Google Scholar] [CrossRef]
- Shejawal, K.P.; Randive, D.S.; Bhinge, S.D.; Bhutkar, M.A.; Todkar, S.S.; Mulla, A.S.; Jadhav, N.R. Green synthesis of silver, iron and gold nanoparticles of lycopene extracted from tomato: Their characterization and cytotoxicity against COLO320DM, HT29 and Hella cell. J. Mater. Sci. Mater. Med. 2021, 32, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Malaikolundhan, H.; Mookkan, G.; Krishnamoorthi, G.; Matheswaran, N.; Alsawalha, M.; Veeraraghavan, V.P.; Mohan, S.K.; Di, A. Anticarcinogenic effect of gold nanoparticles synthesized from Albizia lebbeck on HCT-116 colon cancer cell lines. Artif. Cells Nanomed. Biotechnol. 2020, 48, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, V.K.; Yadav, N.; Rai, N.K.; Ellah, N.H.A.; Bohara, R.A.; Rehan, I.F.; Marraiki, N.; Batiha, G.E.-S.; Hetta, H.F.; Singh, P. Pleurotus sajor-caju-Mediated Synthesis of Silver and Gold Nanoparticles Active against Colon Cancer Cell Lines: A New Era of Herbonanoceutics. Molecules 2020, 25, 3091. [Google Scholar] [CrossRef]
- Elumalai, D.; Suman, T.Y.; Hemavathi, M.; Swetha, C.; Kavitha, R.; Arulvasu, C.; Kaleena, P.K. Biofabrication of gold nanoparticles using Ganoderma lucidum and their cytotoxicity against human colon cancer cell line (HT-29). Bull. Mater. Sci. 2021, 44, 1–6. [Google Scholar] [CrossRef]
- Javed, B.; Mashwani, Z.-U.-R. Synergistic Effects of Physicochemical Parameters on Bio-Fabrication of Mint Silver Nanoparticles: Structural Evaluation and Action Against HCT116 Colon Cancer Cells. Int. J. Nanomed. 2020, 15, 3621–3637. [Google Scholar] [CrossRef]
- Han, X.; Jiang, X.; Guo, L.; Wang, Y.; Veeraraghavan, V.P.; Mohan, S.K.; Wang, Z.; Cao, D. Anticarcinogenic potential of gold nanoparticles synthesized from Trichosanthes kirilowii in colon cancer cells through the induction of apoptotic pathway. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3577–3584. [Google Scholar] [CrossRef]
- Chengzheng, W.; Jiazhi, W.; Shuangjiang, C.; Swamy, M.K.; Sinniah, U.R.; Akhtar, M.S.; Umar, A. Biogenic Synthesis, Characterization and Evaluation of Silver Nanoparticles from Aspergillus niger JX556221 Against Human Colon Cancer Cell Line HT-J. Nanosci. Nanotechnol. 2018, 18, 3673–3681. [Google Scholar] [CrossRef]
- Dehghanizade, S.; Arasteh, J.; Mirzaie, A. Green synthesis of silver nanoparticles using Anthemis atropatana extract: Characterization and in vitro biological activities. Artif. Cells Nanomed. Biotechnol. 2018, 46, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Acharya, D.; Satapathy, S.; Somu, P.; Parida, U.K.; Mishra, G. Apoptotic Effect and Anticancer Activity of Biosynthesized Silver Nanoparticles from Marine Algae Chaetomorpha linum Extract Against Human Colon Cancer Cell HCT-Biol. Trace Elem. Res. 2021, 199, 1812–1822. [Google Scholar] [CrossRef] [PubMed]
- Shejawal, K.P.; Randive, D.S.; Bhinge, S.D.; Bhutkar, M.A.; Wadkar, G.H.; Jadhav, N.R. Green synthesis of silver and iron nanoparticles of isolated proanthocyanidin: Its characterization, antioxidant, antimicrobial, and cytotoxic activities against COLO320DM and HT. J. Genet. Eng. Biotechnol. 2020, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mata, R.; Nakkala, J.R.; Sadras, S.R. Polyphenol stabilized colloidal gold nanoparticles from Abutilon indicum leaf extract induce apoptosis in HT-29 colon cancer cells. Colloids Surf. B Biointerfaces 2016, 143, 499–510. [Google Scholar] [CrossRef]
- Bin-Meferij, M.; Hamida, R.S. Biofabrication and Antitumor Activity Of Silver Nanoparticles Utilizing Novel Nostoc sp. Bahar M. Int. J. Nanomed. 2019, 14, 9019–9029. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.; Li, H.; Hou, T.; Bethu, M.; Ren, Z.; Zhang, Z. Phytosynthesis of Silver Nanoparticles Using Perilla frutescens Leaf Extract: Characterization and Evaluation of Antibacterial, Antioxidant, and Anticancer Activities. Int. J. Nanomed. 2021, 16, 15–29. [Google Scholar] [CrossRef]
- Movagharnia, R.; Sa, S.S. Cytotoxicity effects of green synthesized silver nanoparticles on human colon cancer (HT29) cells Abstract: Background: Nowadays, investigations on the role of nanoparticles for diagnosis, and treatment of cancers are being increased. Feyz J. Kashan Univ. Med. Sci. 2018, 22, 31–38. [Google Scholar]
- Dey, A.; Yogamoorthy, A.; Sundarapandian, S.M. Green Synthesis of Gold Nanoparticles and Evaluation of its Cytotoxic Property against Colon Cancer Cell Line. Res. J. Life Sci. Bioinform. Pharm. Chem. Sci. 2018, 4, 1–17. [Google Scholar] [CrossRef]
- Venkatas, J.; Singh, M. Cervical cancer: A meta-analysis, therapy and future of nanomedicine. Ecancermedicalscience 2020, 14, 1111. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Bai, Q.; Zhang, X.; Yang, H. Folate-mediated chemotherapy and diagnostics: An updated review and outlook. J. Control. Release 2017, 252, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Combination Treatment of Cervical Cancer Using Folate-Decorated, pH-Sensitive, Carboplatin and Paclitaxel Co-Loaded Lipid-Polymer Hybrid Nanoparticles. Drug Des. Dev. Ther. 2020, 14, 823–832. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, Z.; Zhao, L. Folate-decorated poly(3-hydroxybutyrate-co-3-hydroxyoctanoate) nanoparticles for targeting delivery: Optimization andin vivoantitumor activity. Drug Deliv. 2016, 23, 1830–1837. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Zhao, C.; Wang, L. Molecular-targeted agents combination therapy for cancer: Developments and potentials. Int. J. Cancer 2013, 134, 1257–1269. [Google Scholar] [CrossRef]
- Pandurangan, M.; Enkhtaivan, G.; Venkitasamy, B.; Mistry, B.; Noorzai, R.; Jin, B.Y.; Kim, D.H. Time and Concentration-Dependent Therapeutic Potential of Silver Nanoparticles in Cervical Carcinoma Cells. Biol. Trace Elem. Res. 2016, 170, 309–319. [Google Scholar] [CrossRef]
- Lam, P.-L.; Wong, W.-Y.; Bian, Z.; Chui, C.-H.; Gambari, R. Recent advances in green nanoparticulate systems for drug delivery: Efficient delivery and safety concern. Nanomedicine 2017, 12, 357–385. [Google Scholar] [CrossRef]
- Zangeneh, M.M.; Bovandi, S.; Gharehyakheh, S.; Zangeneh, A.; Irani, P. Green synthesis and chemical characterization of silver nanoparticles obtained using Allium saralicum aqueous extract and survey of in vitro antioxidant, cytotoxic, antibacterial and antifungal properties. Appl. Organomet. Chem. 2019, 33, 1–11. [Google Scholar] [CrossRef]
- Donga, S.; Chanda, S. Facile green synthesis of silver nanoparticles using Mangifera indica seed aqueous extract and its antimicrobial, antioxidant and cytotoxic potential (3-in-1 system). Artif. Cells Nanomed. Biotechnol. 2021, 49, 292–302. [Google Scholar] [CrossRef]
- Al-Sheddi, E.S.; Farshori, N.N.; Al-Oqail, M.M.; Al-Massarani, S.M.; Saquib, Q.; Wahab, R.; Musarrat, J.; Al-Khedhairy, A.; Siddiqui, M. Anticancer Potential of Green Synthesized Silver Nanoparticles Using Extract of Nepeta deflersiana against Human Cervical Cancer Cells (HeLA). Bioinorg. Chem. Appl. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Sarkar, S.; Kotteeswaran, V. Green synthesis of silver nanoparticles from aqueous leaf extract of Pomegranate (Punica granatum) and their anticancer activity on human cervical cancer cells. Adv. Nat. Sci. Nanosci. Nanotechnol. 2018, 9, 025014. [Google Scholar] [CrossRef]
- Du, J.; Singh, H.; Yi, T.-H. Antibacterial, anti-biofilm and anticancer potentials of green synthesized silver nanoparticles using benzoin gum (Styrax benzoin) extract. Bioprocess Biosyst. Eng. 2016, 39, 1923–1931. [Google Scholar] [CrossRef]
- Sreekanth, T.V.M.; Pandurangan, M.; Jung, M.-J.; Lee, Y.R.; Eom, I.-Y. Eco-friendly decoration of graphene oxide with green synthesized silver nanoparticles: Cytotoxic activity. Res. Chem. Intermed. 2016, 42, 5665–5676. [Google Scholar] [CrossRef]
- Xu, Z.; Feng, Q.; Wang, M.; Zhao, H.; Lin, Y.; Zhou, S. Green Biosynthesized Silver Nanoparticles with Aqueous Extracts of Ginkgo Biloba Induce Apoptosis via Mitochondrial Pathway in Cervical Cancer Cells. Front. Oncol. 2020, 10, 575415. [Google Scholar] [CrossRef]
- Venkatachalam, P.; Bhuvaneswari, T.; Geetha, N. Nanotechnology in the Life Sciences. In Nanomaterial Bioin-teractions at the Cellular, Organismal and System Levels; Springer International Publishing: Cham, Swiitzerland, 2021; ISBN 9783030657918. [Google Scholar]
- Kajani, A.A.; Esfahani, S.H.Z.; Bordbar, A.-K.; Khosropour, A.R.; Razmjou, A.; Kardi, M. Anticancer effects of silver nanoparticles encapsulated by Taxus baccata extracts. J. Mol. Liq. 2016, 223, 549–556. [Google Scholar] [CrossRef]
- Qian, L.; Su, W.; Wang, Y.; Dang, M.; Zhang, W.; Wang, C. Synthesis and characterization of gold nanoparticles from aqueous leaf extract of Alternanthera sessilis and its anticancer activity on cervical cancer cells (HeLa). Artif. Cells Nanomed. Biotechnol. 2019, 47, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Al Saqr, A.; Khafagy, E.-S.; Alalaiwe, A.; Aldawsari, M.; Alshahrani, S.; Anwer, K.; Khan, S.; Lila, A.; Arab, H.; Hegazy, W. Synthesis of Gold Nanoparticles by Using Green Machinery: Characterization and In Vitro Toxicity. Nanomaterials 2021, 11, 808. [Google Scholar] [CrossRef]
- Ke, Y.; Al Aboody, M.S.; Alturaiki, W.; Alsagaby, S.; Alfaiz, F.A.; Veeraraghavan, V.P.; Mickymaray, S. Photosynthesized gold nanoparticles from Catharanthus roseus induces caspase-mediated apoptosis in cervical cancer cells (HeLa). Artif. Cells Nanomed. Biotechnol. 2019, 47, 1938–1946. [Google Scholar] [CrossRef]
- Mai, X.-T.; Tran, M.-C.; Hoang, A.-Q.; Nguyen, P.D.-N.; Nguyen, T.-H.; Tran, H.N.; Nguyen, P.-T. Gold nanoparticles from Celastrus hindsii and HAuCl4: Green synthesis, characteristics, and their cytotoxic effects on HeLa cells. Green Process. Synth. 2021, 10, 73–84. [Google Scholar] [CrossRef]
- Baharara, J.; Ramezani, T.; Divsalar, A.; Mousavi, M.; Seyedarabi, A. Induction of apoptosis by green synthesized gold na-noparticles through activation of caspase-3 and 9 in human cervical cancer cells. Avicenna J. Med. Biotechnol. 2016, 8, 75–83. [Google Scholar]
- Tebbi, C. Etiology of Acute Leukemia: A Review. Cancers 2021, 13, 2256. [Google Scholar] [CrossRef] [PubMed]
- Bispo, J.A.B.; Pinheiro, P.S.; Kobetz, E.K. Epidemiology and Etiology of Leukemia and Lymphoma. Cold Spring Harb. Perspect. Med. 2019, 10, a034819. [Google Scholar] [CrossRef]
- Deng, R.; Ji, B.; Yu, H.; Bao, W.; Yang, Z.; Yu, Y.; Cui, Y.; Du, Y.; Song, M.; Liu, S.; et al. Multifunctional Gold Nanoparticles Overcome MicroRNA Regulatory Network Mediated-Multidrug Resistant Leukemia. Sci. Rep. 2019, 9, 5348. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, S.; Pourbagheri-Sigaroodi, A.; Yousefi, A.-M.; Fakhroueian, Z.; Momeny, M.; Bashash, D. ZnO Q-Dots-Induced Apoptosis Was Coupled with the Induction of PPARγ in Acute Promyelocytic Leukemia Cells; Proposing a Novel Application of Nanoparticles in Combination with Pioglitazone. J. Clust. Sci. 2021, 0123456789, 1–13. [Google Scholar] [CrossRef]
- Karmous, I.; Pandey, A.; Ben Haj, K.; Chaoui, A. Efficiency of the Green Synthesized Nanoparticles as New Tools in Cancer Therapy: Insights on Plant-Based Bioengineered Nanoparticles, Biophysical Properties, and Anticancer Roles. Biol. Trace Elem. Res. 2020, 196, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Hossen, S.; Hossain, M.K.; Basher, M.K.; Mia, M.N.H.; Rahman, M.T.; Uddin, J. Smart nanocarrier-based drug delivery systems for cancer therapy and toxicity studies: A review. J. Adv. Res. 2019, 15, 1–18. [Google Scholar] [CrossRef]
- Pallarès, V.; Unzueta, U.; Falgàs, A.; Sánchez-García, L.; Serna, N.; Gallardo, A.; Morris, G.; Alba-Castellón, L.; Álamo, P.; Sierra, J.; et al. An Auristatin nanoconjugate targeting CXCR4+ leukemic cells blocks acute myeloid leukemia dissemination. J. Hematol. Oncol. 2020, 13, 1–19. [Google Scholar] [CrossRef]
- Cruz, D.M.; Mostafavi, E.; Vernet-Crua, A.; Barabadi, H.; Shah, V.; Cholula-Díaz, J.L.; Guisbiers, G.; Webster, T.J. Green nanotechnology-based zinc oxide (ZnO) nanomaterials for biomedical applications: A review. J. Phys. Mater. 2020, 3, 034005. [Google Scholar] [CrossRef]
- Jadoun, S.; Arif, R.; Jangid, N.K.; Meena, R.K. Green synthesis of nanoparticles using plant extracts: A review. Environ. Chem. Lett. 2021, 19, 355–374. [Google Scholar] [CrossRef]
- Bhatia, S. Nanotechnology and Its Drug Delivery Applications. In Natural Polymer Drug Delivery Systems; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–32. [Google Scholar] [CrossRef]
- Ahmeda, A.; Zangeneh, A.; Zangeneh, M.M. Characterization and anti-acute T cell leukemia properties of silver nanoparticles synthesized by a green approach for bioremediation applications: Introducing a new chemotherapeutic drug for clinical trial studies. Appl. Organomet. Chem. 2020, 34, 1–11. [Google Scholar] [CrossRef]
- Namvar, F.; Rahman, H.S.; Mohamad, R.; Rasedee, A.; Yeap, S.K.; Chartrand, M.S.; Azizi, S.; Tahir, P.M. Apoptosis Induction in Human Leukemia Cell Lines by Gold Nanoparticles Synthesized Using the Green Biosynthetic Approach. J. Nanomater. 2015, 1–10. [Google Scholar] [CrossRef]
- Zangeneh, M.M.; Zangeneh, A. Novel green synthesis of Hibiscus sabdariffa flower extract conjugated gold nanoparticles with excellent anti-acute myeloid leukemia effect in comparison to daunorubicin in a leukemic rodent model. Appl. Organomet. Chem. 2019, 34, 1–13. [Google Scholar] [CrossRef]
- Hemmati, S.; Joshani, Z.; Zangeneh, A.; Zangeneh, M.M. Green synthesis and chemical characterization of Thymus vulgaris leaf aqueous extract conjugated gold nanoparticles for the treatment of acute myeloid leukemia in comparison to doxorubicin in a leukemic mouse model. Appl. Organomet. Chem. 2020, 34, 1–14. [Google Scholar] [CrossRef]
- Azizi, S.; Shahri, M.M.; Rahman, H.S.; Rahim, R.A.; Rasedee, A.; Mohamad, R. Green synthesis palladium nanoparticles mediated by white tea (Camellia sinensis) extract with antioxidant, antibacterial, and antiproliferative activities toward the human leukemia (MOLT-4) cell line. Int. J. Nanomed. 2017, 12, 8841–8853. [Google Scholar] [CrossRef]
- Karimi, S.; Shahri, M.M. Medical and cytotoxicity effects of green synthesized silver nanoparticles using Achillea millefolium extract on MOLT-4 lymphoblastic leukemia cell line. J. Med Virol. 2021, 93, 3899–3906. [Google Scholar] [CrossRef] [PubMed]
- Palombarini, F.; Masciarelli, S.; Incocciati, A.; Liccardo, F.; Di Fabio, E.; Iazzetti, A.; Fabrizi, G.; Fazi, F.; Macone, A.; Bonamore, A.; et al. Self-assembling ferritin-dendrimer nanoparticles for targeted delivery of nucleic acids to myeloid leukemia cells. J. Nanobiotechnol. 2021, 19, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Dobrucka, R.; Romaniuk-Drapała, A.; Kaczmarek, M. Anti-Leukemia Activity of Au/CuO/ZnO Nanoparticles Synthesized used Verbena officinalis Extract. J. Inorg. Organomet. Polym. Mater. 2021, 31, 191–202. [Google Scholar] [CrossRef]
- Dobrucka, R.; Romaniuk-Drapała, A.; Kaczmarek, M. Facile synthesis of Au/ZnO/Ag nanoparticles using Glechoma hederacea L. extract, and their activity against leukemia. Biomed. Microdevices 2021, 23, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Parial, D.; Gopal, P.K.; Paul, S.; Pal, R. Gold (III) bioreduction by cyanobacteria with special reference to in vitro biosafety assay of gold nanoparticles. Environ. Biol. Fishes 2016, 28, 3395–3406. [Google Scholar] [CrossRef]
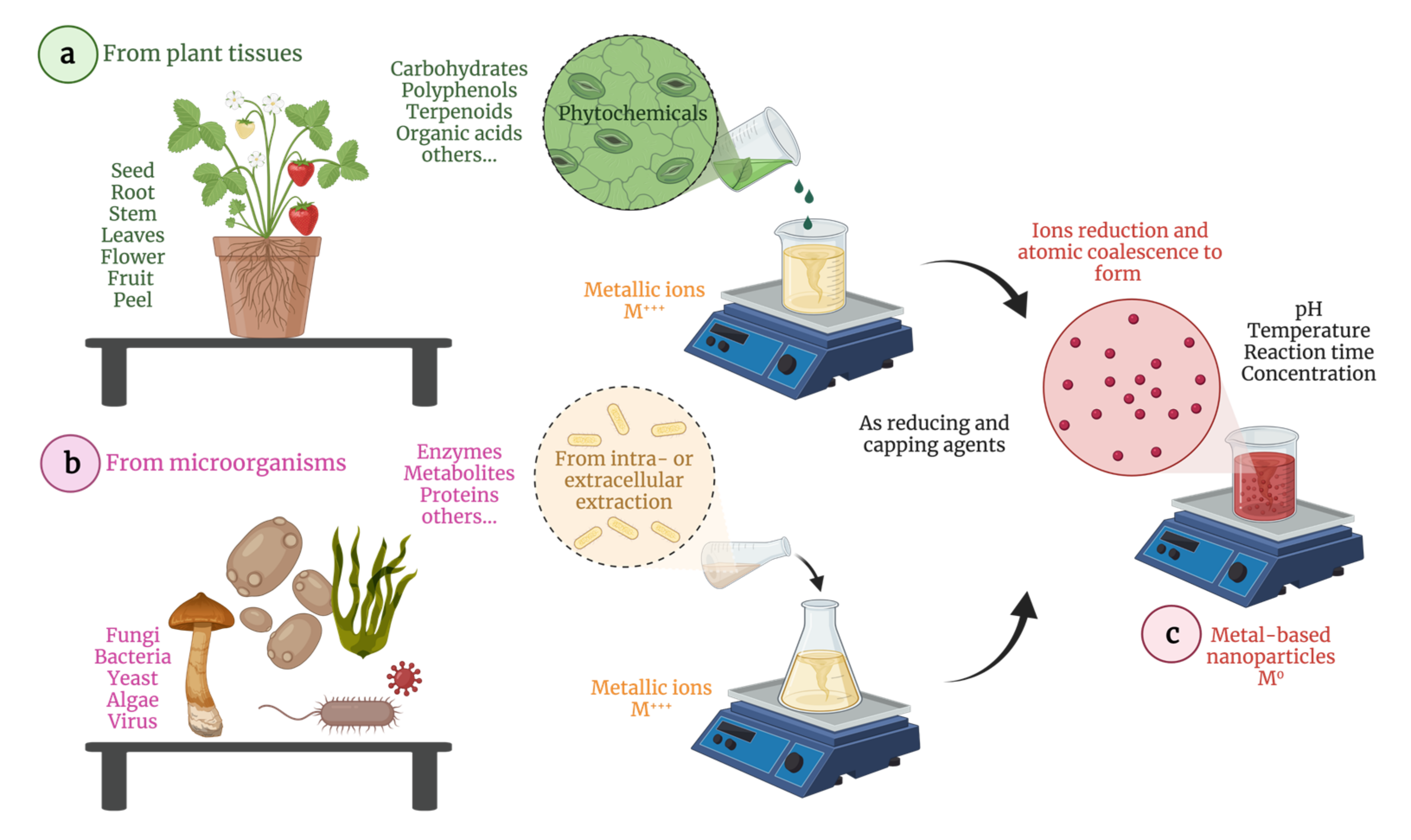

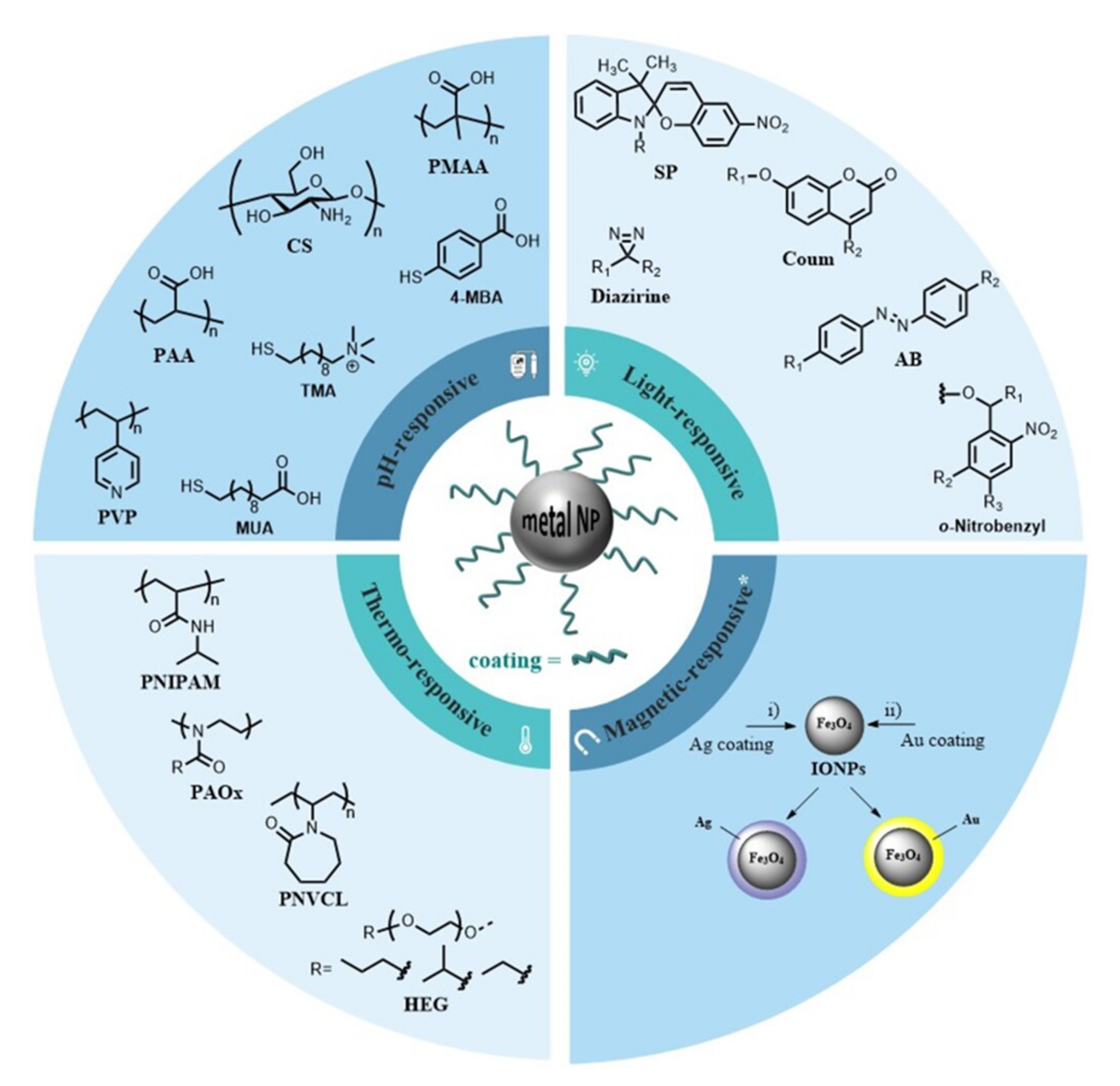
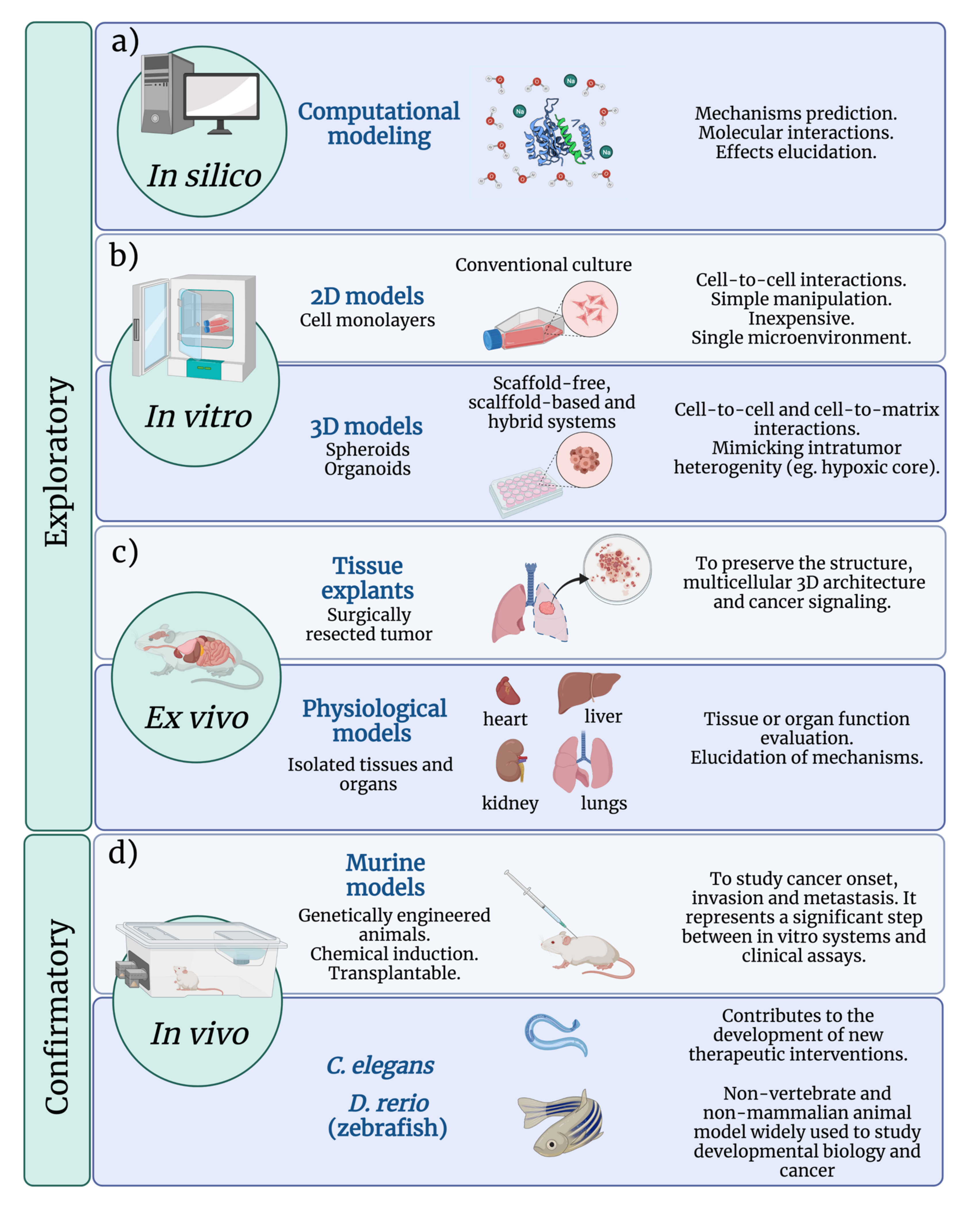

| pH-Sensitive Bonds | Chemical Mechanisms |
|---|---|
| Imine |  |
| Hydrazone |  |
| Oxime |  |
| Amide |  |
| Acetals |  |
| Orthoester |  |
| Type of NP, Concentration and Specimen | Stage of Development at RNA Extraction and Method of Transcriptome Profiling | Up- or Down-Regulation | Gene Ontology | Reference |
|---|---|---|---|---|
| Citrate-coated AuNPs 5.9 mg/L C. elegans L3 larval stage exposed for 12 h. | L3 larval stage Affymetrix C. elegans gene chip | Up-regulation | Amyloid Processing Citrate cycle Clathrin-mediated endocytosis Apoptosis Unfolded protein response G-protein signalling | [171] |
| Bare AuNPs 0.1 and 0.2 mg/L MUA/Au = 0.5 MUA/Au = 3 C. elegans embryos exposed for 72 h. | Adults Affymetrix C. elegans gene chip | Up-regulation Down-regulation | Defense response Lipid catabolic processes Lipid storage Body morphogenesis Body shape and size regulation Metal detoxification(homeostasis) and stress Lifespan | [172] |
| Citrate-capped AuNPs 20 mg/L Liver of adult Zebrafish fish exposed for 96 h. | Adults Affymetrix GeneChip Zebrafish Genome Array | Up-regulation Down-regulation | Stress response Development The establishment of localization Biogenesis Metabolic process Locomotion Biological adhesion Response to a stimulus | [173] |
| AgNPs 10 μg/mL C. elegans L1 larvae trough adulthood exposed for 72 h. | Adults RNA seq/C. elegans | Up-regulation Down-regulation | Cellular process: cell cycle, meiosis, apoptosis, etc. Growth Metabolic process Reproduction Cellular component organization and biogenesis Behavior Cellular process: cell communication, cell surface receptor signaling pathway Developmental process. Morphogenesis, generation of neurons Metabolic process Response to stimulus Growth Locomotion Reproduction | [174] |
| AgNPs 50 μg/L Gills from adult zebrafish exposed for 28 days | Adults Agilent Technologies 4x44 K zebrafish microarray | Over-represented ND | Extracellular components (Matrix) Mithocondria Ribonucleoprotein complexes DNA damage DNA/RNA processing Heat shock proteins Cell growth and migration Anatomical, organ and cell morphogenesis Embryonic, skeletal and organ development | [175] |
| Bare AgNPs 0.01 mg/L 6 dpf larvae exposed for 15 days | Larvae Agilent’s zebrafish (V3) oligonucleotide microarrays | Up-regulation Down-regulation | Photoreception Circadian clock regulation Cardiovascular disease Genetic disorder Hematological disease Hypersensitivity response Ophthalmic disease | [176] |
| Citrate-coated AgNPs 0.4 mg/L 24 hpf embryos exposed for 24 h | Embryos Agilent Technologies 4x44 K zebrafish microarray | Down-regulation | Ligans-gated ion channel activity Dopaminergic receptor signaling and neuro differentiation Neuron recognition, Regulation of neurogenesis | [177] |
| Model | Way of Generation | Advantages | Disadvantages |
|---|---|---|---|
| Ectopic CDX Cell line-Derived Xenograft | Human tumor cells (fluorescents or not) Implanted subcutaneously | Easy, fast, and cheap Commercial cell lines or primary cell cultures Eeasily measurable | Immunodeficient host Some cancer types fail to grow Not specific tissue growing |
| Orthotopic CDX Cell line-Derived Xenograft | Implantation on specific tissue | Microenvironment similar to the origin of the tumor Eeasily measurable | They are more technically complex than ectopic Immunodeficient host Not all cancer types grow |
| Metastatic CDX | By injection of tumor cells by vein or intra-cardiac | Tumors can grow in a variety of tissues or organs | The model does not mimic the original tumor Technically demanding to detect the location of tumor |
| PDXPatient-Derived Xenografts | By implantation of tumor cells or fragment derived from human tumors. (ectopically or orthotopically) | Tumors generated maintain the phenotypic and genotypic characteristics of the original tumor derived from the patient | Requires fresh patient tumor tissue Immunodeficient host Relatively expensive Slow implementation Technically demanding |
| Syngeneic | Mouse tumor tissue or cells implanted on same strain mouse | Good growing tumors Microenvironment adequate Immunocompetent host | Mouse microenvironment Not useful as human model |
| Conventional GEMM Genetically Modified Mouse Models | Oncogenic-driven transgenic mice to develop specific cancer. | Natural microenvironment Intact immune system Modelling of early/late stages of tumor progression | Mouse microenvironment Not truly of human disease Not valuable for certain test therapies |
| In Vivo Model | Biological Source | Type of NP | Size (nm) and Shape | Cell Line | Administration Route | Reference |
|---|---|---|---|---|---|---|
| Subcutaneous Breast cancer Mice | Curcuma longa | AuNPs | 278 Spherical | DMBA | Oral | [185] |
| Breast cancer Mice | Mangifera indica | AuNPs | 55.5–65.5 Spherical | MDA-MB-231 | Oral | [186] |
| Subcutaneous Cacu Mice | Peptides | AuNPs | 100–150 Spherical Conjugate | HeLa | Tail vein | [187] |
| Subcutaneous Lung cancer Mice | Dimocarpus longan | AgNPs | 13 Spherical | H1299 | Intraperitoneal | [188] |
| Intravenous Mice | Curcumin | AgNPs | 50–100 | HeLa | Intraperitoneal | [189] |
| Mice | Nostoc carneum | AgNPs | 16 Spherical | EAC | Intraperitoneal | [190] |
| Subcutaneous C57 Mice | Curcumin | AuNPs | 16 Spherical | C540 (B16/F10) | Intratumoral | [191] |
| Intravenous Mice | Camellia sinensis | AuNPs | 20–30 Spherical | HL-60 | Intravenous | [192] |
| Biological Source or Hybridization | Type of NP | Size (nm) | Shape | IC50 (µg/mL) | Cell Line/In Vivo Model | Reference |
|---|---|---|---|---|---|---|
| Siberian ginseng | AuNPs | 200 | Spherical | 10 µg/mL | B16 | [202] |
| Fluorouracil | AuNPs | 16–150 | Spherical | ND | A431 | [204] |
| TiO2 | AuNPs | 148–333 | Spherical | ND | B16F1 | [208] |
| Bacillus licheniformis | AgNPs | 20–80 | Triangular | 2457.5 | B16F10, A431 | [209] |
| TiO2 | AgNPs | 50 | Nanoprism | 100 | B16F10, C57BL/6J mice | [122] |
| A. muricata | AgNPs | 50 | Spherical | ND | A375 | [203] |
| BSA | AgNPs | 58 | Spherical | 200 | B16F10 | [206] |
| Biological Source | Type of NP | Size (nm) | Shape | IC50 (µg/mL) | Cell Line | Reference |
|---|---|---|---|---|---|---|
| Actinobacterial-SF23 | AgNPs | 3–36 | Spherical | 16.30 | MCF7 | [218] |
| Buchanania axillaris | AgNPs | 17–80 | Spherical | 31.20 | MCF7 | [219] |
| Black Tea mistry | AgNPs | 9–15 | Spherical | 30 | MCF7 | [220] |
| Oscillatoria limnetica | AgNPs | 3–17 | Spherical | 6.14 | MCF7 | [221] |
| Dunaliella salina | AuNPs | ~22 | Spherical | 98 | MCF7 | [222] |
| Fagonia indica | AgNPs | 10–60 | Spherical | 12.35 | MCFT | [223] |
| Linum usitatissimum | AuNPs | ~31 | Triangular | 5 | MCF7 | [224] |
| Chitosan-functionalized | AgNPs | 13–22 | Spherical | 6.40 6.56 | MCF7 MDA-MB-231 | [225] |
| Polysiphonia algae | AgNPs | 5–25 | Spherical | 4.19 | MCF7 | [226] |
| Cynara scolymus | AgNPs | 98 | Spherical | 10 | MDA-MB-231 | [227] |
| Syzygium jambolanum | AgNPs | 20–25 | Spherical | ND | MCF7 | [228] |
| Tamarindus indica | AgNPs | 20–52 | Spherical | 20 | MCF7 | [229] |
| Commiphora wightii | AuNPs | ~28 | Spherical | 66.11 | MCF7 | [230] |
| Sargassum ilicifolium | AuNPs | 20–25 | Spherical | 24 | MDA-MB-231 | [231] |
| Mentha longifolia | AuNPs | ~36 | Spherical | 274 | MCF7 | [232] |
| Acacia luciana | AgNPs | 50 | Spherical | 4.37 | MCF7 | [233] |
| Garcinia atroviridis | AgNPs | 5–30 | Spherical | 2.00 | MCF7 | [234] |
| Allium saticum | AgNPs | 20–35 | Spherical | 89.86 | MCF7 | [235] |
| Cladosporium sp | AuNPs | 5–10 | Spherical | 38.23 | MCF7 | [236] |
| Curcuma mangga | AuNPs | ~28 | Spherical | 0.41 | MCF7 | [237] |
| Dragon fruit | AuNPs | 10–20 | Spherical | ND | MCF7 MDA-MB-231 | [238] |
| Biological Source | Type of NP | Size (nm) | Shape | IC50 (µg/mL) | Cell Line | Reference |
|---|---|---|---|---|---|---|
| Rabdosia rubescens | AuNPs | 130 | Spherical | 25 | A549 | [242] |
| Millettia pinnata | AuNPs | 37 | Spherical | 14.76 | A549 | [245] |
| Pinus roxburghii | AgNPs | 80 | Spherical | 11.28 | A549 | [243] |
| Pleuropterus multiflorus | AgNPs AuNPs | ~275 ~105 | Spherical | 35.16 | A549 | [244] |
| Matricaria chamomilla | AgNPs | ~45 | Spherical | 62.82 | A549 | [246] |
| Carpesium cernuum | AgNPs | 13 | Spherical | ND | A549 | [105] |
| Marsdenia tenacissima | AuNPs | 50 | Spherical | 15 | A549 | [247] |
| Beta vulgaris | AgNPs | 5–20 | Spherical | 48.20 | A549 | [248] |
| Garcinia mangostana | AgNPs AuNPs | 13–31 15–44 | Asymmetric dumbbell Spherical | ND | A549 | [249] |
| Cratoxylum formosum Mucuna birdwoodiana | AgNPs | ~9 ~35 | Spherical | ND | A549 | [249] |
| Musa paradisiaca | AuNPs | 50 | Spherical to triangular | 58 | A549 | [250] |
| Dendropanax morbifera | AgNPs AuNPs | 100–150 10–20 | Polygonal Hexagonal | ND | A549 | [251] |
| Cymbopogon citratus | AgNPs | 17–26 | Spherical | 25 | A549 | [252] |
| Indigofera tinctoria | AgNPs AuNPs | ~19 | Spherical | 56.62 59.33 | A549 | [253] |
| Coptis chinensis | AgNPs | 6–45 | Spherical | 15 | A549 | [254] |
| Magnolia officinalis | AuNPs | 128 | Spherical | ~18 | A549 | [255] |
| Garlic, Green tea, Turmeric | AgNPs | 8 | Spherical | 13.26 17.25 11.11 | A549 | [256] |
| Rosa damascena | AuNPs | 8–45 | Spherical to triangular | ND | A549 | [257] |
| Nannochloropsis sp. | AgNPs | ~57 | Spherical | 15 | A549 | [258] |
| Borago officinalis | AgNPs | 30–80 | Spherical Hexagonal | ~7 | A549 | [259] |
| Moringa oleifera | AuNPs | 10–20 | Spherical | 98.46 | A549 | [260] |
| Euphrasia officinalis | AgNPs AuNPs | ~40 ~50 | Quasi-spherical | ~2 ND | A549 | [261] |
| Curcumae kwangsiensis | AgNPs | 15–21 | Spherical | 249 187 152 | HLC-1 LC-2/ad PC-14 | [262] |
| Escherichia coli VM1 | AgNPs | 10–15 | Spherical | 40 | A549 | [263] |
| Biological Source | Type of NP | Size (nm) | Shape | IC50 µg/mL | Cell Line | Reference |
|---|---|---|---|---|---|---|
| Cyclopia intermedia | AuNPs | 20 | Spherical and triangular | ND | PC-3 | [272] |
| Acai berry | AuNPs | 172 | Spherical and triangular | 1000 | PC-3 | [273] |
| Casein hydrolytic peptide | AuNPs | ~20 | Hexagonal | ND | DU-145 | [274] |
| Doxorubicin | AuNPs | ~75 | Spherical | ND | LN-CaP | [275] |
| Piper Nigrum | AgNPs | 15-38 | Spherical | ND | PC3 | [276] |
| Salvia miltiorrhiza | AgNPs | 100 | Spherical, oval, hexagonal, and triangular | ND | LNCaP | [277] |
| Piperlongum | AgNPs | 5–35 | Quasi-spherical | 73.41 38.53 | PC-3 DU-145 | [278] |
| Salacia chinensis | AgNPs | 40–80 | Rods, triangular and hexagonal | 7.46 | PC-3 | [279] |
| Saraca asoca | AgNPs | 36 | Spherical | 50.00 | DU-145 | [280] |
| Cornus officinal | AgNPs | ~12 | Quasi-spherical | 25.54 | PC-3 | [281] |
| Eclipta prostrata | AgNPs | 50–75 | Spherical | 9.84 | PC-3 | [282] |
| Moringa oleifera | AgNPs | 44–60 | Spherical | 25.21 | PC-3 | [282] |
| Thespesia populnea | AgNPs | 47–97 | Spherical | 31.21 | PC-3 | [282] |
| Guiera senegalensis | AgNPs | 50 | Spherical | 23.48 | PC-3 | [283] |
| Pestalotiopsis microspora | AgNPs | 2–10 | Spherical | 27.71 | PC-3 | [284] |
| Dimocarpus Longan Lour | AgNPs | 9–32 | Spherical | <10.00 | PC-3 | [285] |
| Plumbago zeylanica | AgNPs | 80–98 | Spherical and cuboid | 58.61 | PC-3 | [286] |
| Semecarpus anacardium | AgNPs | 60–95 | Spherical and cuboid | 42.77 | PC-3 | [286] |
| Terminalia arjuna | AgNPs | 34–70 | Spherical and cuboid | 41.78 | PC-3 | [286] |
| Gracilaria edulis | AgNPs | 55–99 | Spherical | 39.60 | PC-3 | [287] |
| Alternanthera sessilis | AgNPs | 30–50 | Spherical | 6.85 | PC-3 | [288] |
| Cell-free supernatant of actinobacteria | FeNPs | 65–87 | Spherical | 65 | PC-3 | [289] |
| Rhus punjabensis | FeNPs | ~48 | Spherical | 12.79 | DU-145 | [290] |
| Leucaena leucocephala | ZnNPs | 50–200 | spherical | 103.72 | PC-3 | [291] |
| Cinnamomum tamala | TiNPs | 23 | Irregular | ND | DU-145 | [292] |
| Biological Source | Type of NP | Size (nm) | Shape | IC50 (µg/mL) | Cell Line | Reference |
|---|---|---|---|---|---|---|
| Zingiber officinale | AgNPs | 42–61 | Spherical | 150.80 | HT29 | [295] |
| Albizia lebbeck | AuNPs | 20–30 | Spherical | 48 | HCT-116 | [297] |
| Pleurotus sajor-caju | AgNPs AuNPs | 16–18 4–22 | Spherical | 50 80 | HCT-116 | [298] |
| Trichosanthes kirilowii | AuNPs | 50 | Spherical | ND | HCT-116 | [301] |
| Aspergillus niger | AgNPs | 20–25 | Spherical | 160 | HT-29 | [302] |
| Anthemis atropatana | AgNPs | 38.89 | Spherical | 4.88 | HT29 | [303] |
| Chaetomorpha linum | AgNPs | 35 | Spherical | 48.84 | HCT-116 | [304] |
| Bergenia ciliata | AgNPs | 50–100 | Spherical | ND | HT29 | [305] |
| Albizia lebbeck | AuNPs | 20–30 | Spherical | 48 | HCT116 | [297] |
| Abutilon indicum | AuNPs | 1–20 | Spherical | 210 | HT-29 | [306] |
| Nostoc sp. | AgNPs | 14.9 | Spherical | 150 | Caco2 | [307] |
| Annona muricata | AgNPs | 16–20 | Spherical | ND | HCT116 | [203] |
| Mentha arvensis | AgNPs | 12–40 | Spherical triangular | 1.7 | HCT116 | [300] |
| Perilla frutescens | AgNPs | ~26 | Spherical rhombic triangle | 39.28 | Colo205 | [308] |
| Ulva lactuca L. | AuNPs | 6–20 | Spherical | 98.46 | HT29 Caco-2 | [260] |
| Curcuma longa Zingiber officinale | AgNPs | 20–51 | Quasi-spherical | 150.80 | HT29 | [295] |
| Artemisa tournefortiana | AgNPs | 22 | Spherical | 40.71 | HT29 | [309] |
| Wedelia trilobata | AuNPs | 10–50 | Spherical | ND | HCT15 | [310] |
| Source of Nanoparticles | Type of NP | Size [nm] | Shape | IC50 (µg/mL) | Cell Lines | Authors |
|---|---|---|---|---|---|---|
| Allium saralicum | AgNPs | 20–40 | Spherical | >1000 | HeLa | [318] |
| Lycopene | AgNPs | 50–100 | Spherical | ND | HeLa | [296] |
| Mangifera indica | AgNPs | 9–61 | ~400 | HeLa | [319] | |
| Nepeta deflersiana | AgNPs | 33 | Spherical | ~3.9 | HeLa | [320] |
| Punica granatum | AgNPs | 41–69 | Spherical | ND | HeLa | [321] |
| Styrax benzoin | AgNPs | 12–38 | Spherical | ND | HeLa | [322] |
| Ziziphus jujube + graphene oxide | AgNPs | 26 7 | Spherical | ND | HeLa | [323] |
| Curcumine derivative (ST06) | AgNPs | 50–100 | Spherical | 1 µM | HeLa | [189] |
| Ginkgo biloba | AgNPs | 40 | Spherical | 4 (HeLa) 6 (SiHa) | HeLa SiHa | [324] |
| Leucas aspera | AgNPs | 35–54 | Spherical | 36 | HeLa | [325] |
| Taxus baccata | AgNPs | 75–91 | Spherical | 10 µg/mL | HeLa | [326] |
| Alternanthera sessilis | AuNPs | 30–50 | Spherical | >5 µg/mL | HeLa | [327] |
| Benincasa hispida | AuNPs | 22 | Spherical | 2.3 µg/mL | HeLa | [328] |
| Catharanthus roseus | AuNPs | 25–35 | Spherical | 5 µg/mL | HeLa | [329] |
| Celastrus hindsii | AuNPs | 13–53 | Spherical | 12.5 | HeLa | [330] |
| Zataria multiflora | AuNPs | 42 | Spherical | ND | HeLa | [331] |
| Biological Source | Type of NP | Size (nm) | Shape | IC50 (µg/mL) | Cell Line | Reference |
|---|---|---|---|---|---|---|
| Glycyrrhiza glabra L. Extract | AgNPs | 20 | Spherical | 604 467 445 438 | J45.01, J.Clone E6–1, J.CaM1.6, J.RT3-T3.5 | [342] |
| Sargassum muticum water Extract | AuNPs | 10 | Spherical | 4.22 5.71 6.55 7.29 | K562, HL-60, Jurkat, CEM | [343] |
| Hibiscus sabdariffa Extract | AuNPs | 15–45 | Spherical | 761 803 882 | C1498, Human HL, 32D-FLT3-ITD | [344] |
| Thymus vulgaris | AuNPs | 10–30 | Spherical | 397 | C1498, Human HL, 32D-FLT3-ITD | [345] |
| Camellia sinensis Tea | PdNPs | 6–18 | Spherical | ND | MOLT-4 | [346] |
| Achillea millefolium | AgNPs | ~22 | Spherical | 0.011 | MOLT-4 | [347] |
| Archaeoglobus fulgidus chimeric ferritin | AgNPs | 4.5 | Spherical | ND | NB4 | [348] |
| Verbena officinalis Extract | Au/CuO/ZnONPs | 35 | Spherical | 0.64 µmol | Jurkat | [349] |
| Glechoma hederacea L. Extract | Au/CuO/ZnONPs | 10 | Spherical | ND | Jurkat | [350] |
| Cyanobacterial strains Leptolyngbya tenuis, Coleofasciculus chthonoplastes, and Nostoc ellipsosporum | AuNPs | 8–42 | Spherical | 150 | MOLT-4 T-ALL | [351] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tinajero-Díaz, E.; Salado-Leza, D.; Gonzalez, C.; Martínez Velázquez, M.; López, Z.; Bravo-Madrigal, J.; Knauth, P.; Flores-Hernández, F.Y.; Herrera-Rodríguez, S.E.; Navarro, R.E.; et al. Green Metallic Nanoparticles for Cancer Therapy: Evaluation Models and Cancer Applications. Pharmaceutics 2021, 13, 1719. https://doi.org/10.3390/pharmaceutics13101719
Tinajero-Díaz E, Salado-Leza D, Gonzalez C, Martínez Velázquez M, López Z, Bravo-Madrigal J, Knauth P, Flores-Hernández FY, Herrera-Rodríguez SE, Navarro RE, et al. Green Metallic Nanoparticles for Cancer Therapy: Evaluation Models and Cancer Applications. Pharmaceutics. 2021; 13(10):1719. https://doi.org/10.3390/pharmaceutics13101719
Chicago/Turabian StyleTinajero-Díaz, Ernesto, Daniela Salado-Leza, Carmen Gonzalez, Moisés Martínez Velázquez, Zaira López, Jorge Bravo-Madrigal, Peter Knauth, Flor Y. Flores-Hernández, Sara Elisa Herrera-Rodríguez, Rosa E. Navarro, and et al. 2021. "Green Metallic Nanoparticles for Cancer Therapy: Evaluation Models and Cancer Applications" Pharmaceutics 13, no. 10: 1719. https://doi.org/10.3390/pharmaceutics13101719
APA StyleTinajero-Díaz, E., Salado-Leza, D., Gonzalez, C., Martínez Velázquez, M., López, Z., Bravo-Madrigal, J., Knauth, P., Flores-Hernández, F. Y., Herrera-Rodríguez, S. E., Navarro, R. E., Cabrera-Wrooman, A., Krötzsch, E., Carvajal, Z. Y. G., & Hernández-Gutiérrez, R. (2021). Green Metallic Nanoparticles for Cancer Therapy: Evaluation Models and Cancer Applications. Pharmaceutics, 13(10), 1719. https://doi.org/10.3390/pharmaceutics13101719








