Application of Nanomaterials as an Advanced Strategy for the Diagnosis, Prevention, and Treatment of Viral Diseases
Abstract
:1. Introduction
2. Diagnosis
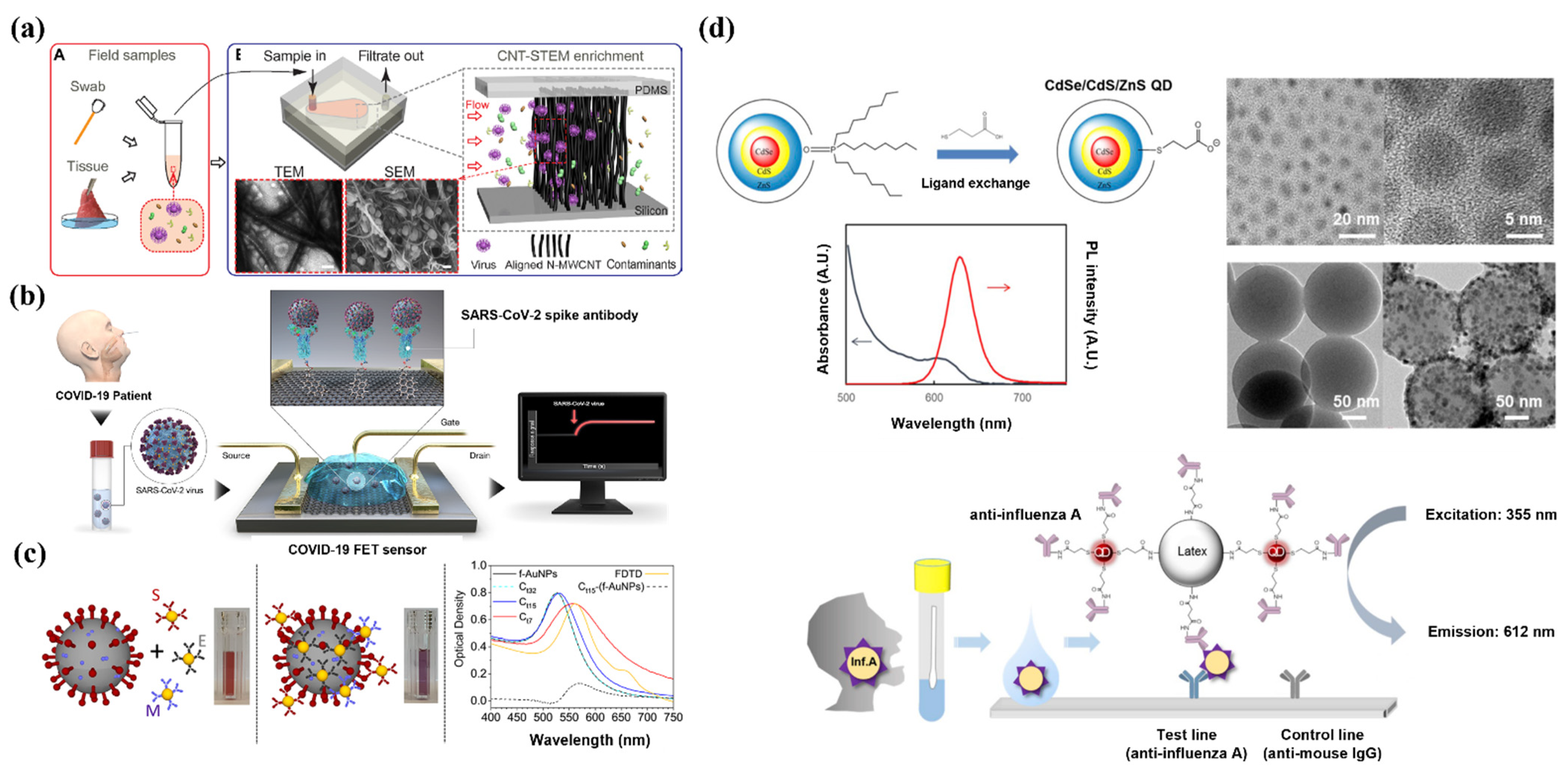
2.1. Carbon Nanotubes
2.2. Graphene
2.3. Gold NPs (AuNPs)
2.4. Quantum Dots (QDs)
2.5. Synthetic Polymers
3. NP Vaccines for Emerging Viruses
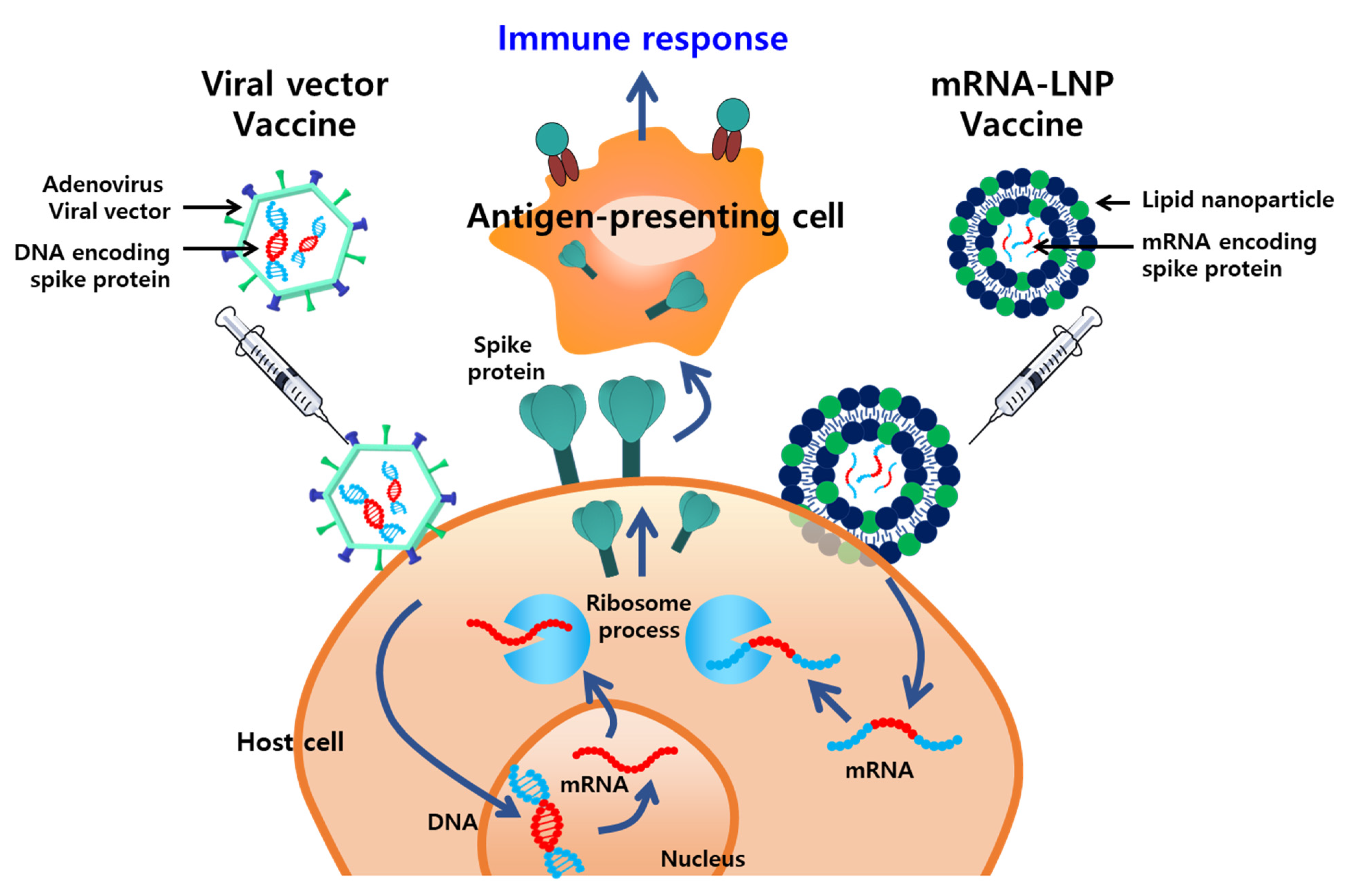
3.1. LNPs
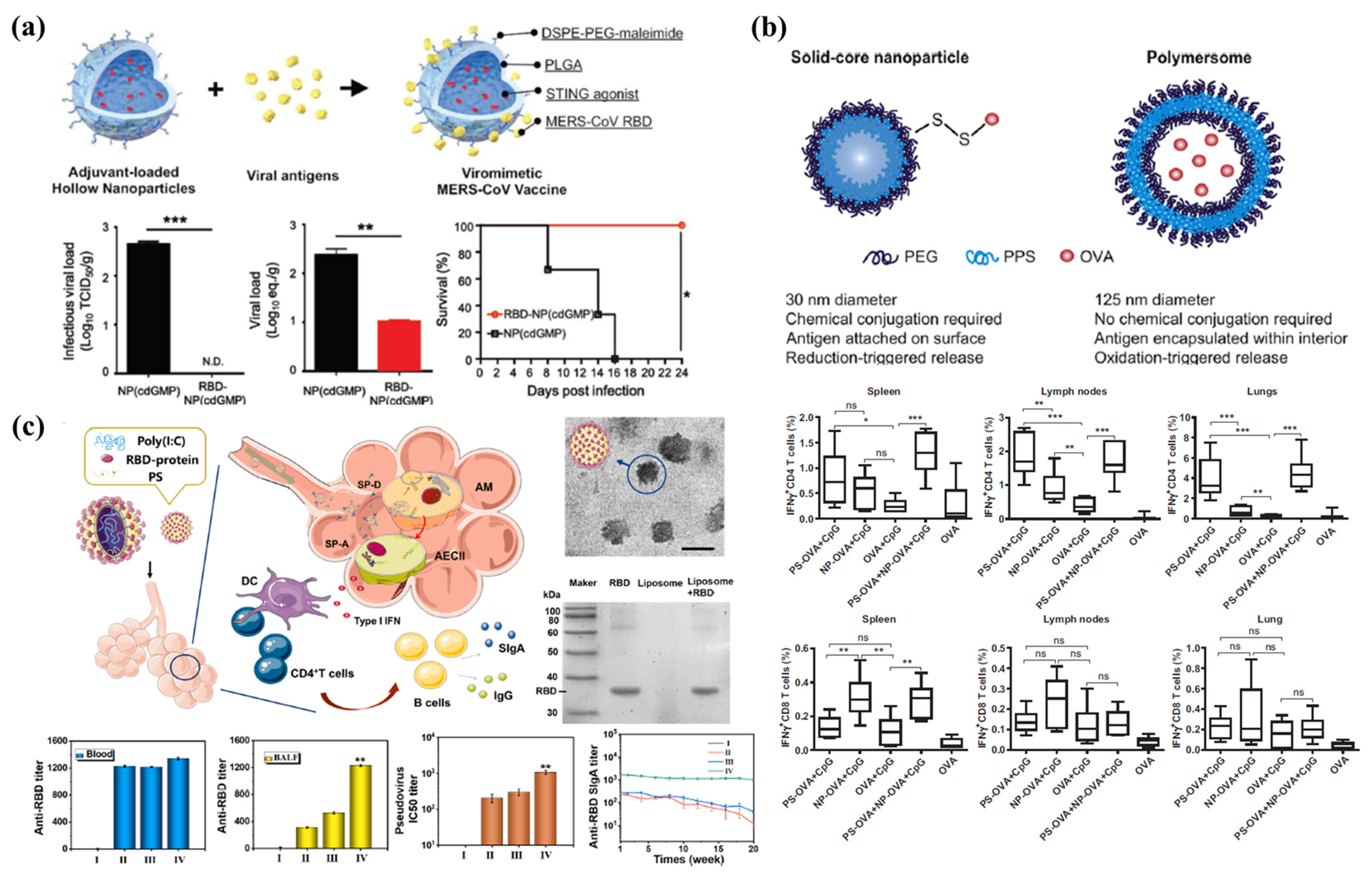
3.2. Polymeric NPs
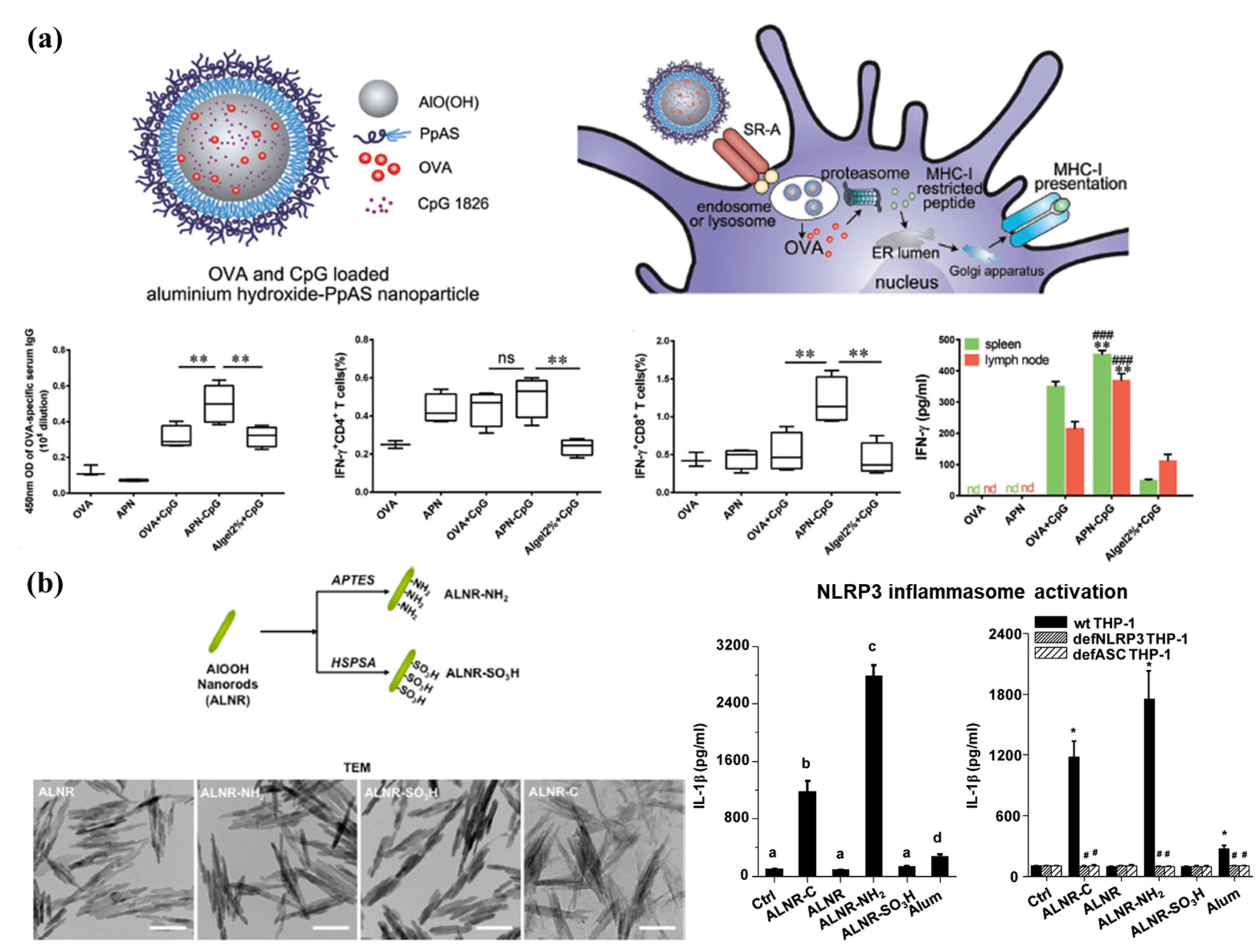
3.3. Nanoparticulate Adjuvants
3.4. Future Directions of Vaccine Development Using Nanoplatforms
4. Treatment
4.1. Delivery
4.2. Viral Inhibition
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Park, S.E. Epidemiology, virology, and clinical features of severe acute respiratory syndrome -coronavirus-2 (SARS-CoV-2; coronavirus disease-19). Clin. Exp. Pediatr. 2020, 63, 119–124. [Google Scholar] [CrossRef] [Green Version]
- Chintagunta, A.D.; Sai Krishna, M.; Nalluru, S.; Sampath Kumar, N.S. Nanotechnology: An emerging approach to combat COVID-19. Emergent Mater. 2021, 1–12. [Google Scholar]
- Díez-Pascual, A.M. Recent progress in antimicrobial nanomaterials. Nanomaterials 2020, 10, 2315. [Google Scholar] [CrossRef]
- Huang, H.; Lovell, J.F. Advanced functional nanomaterials for theranostics. Adv. Funct. Mater. 2017, 27, 1603524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Look, M.; Bandyopadhyay, A.; Blum, J.S.; Fahmy, T.M. Application of nanotechnologies for improved immune response against infectious diseases in the developing world. Adv. Drug. Deliv. Rev. 2010, 62, 378–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wasik, D.; Mulchandani, A.; Yates, M.V. A heparin-functionalized carbon nanotube-based affinity biosensor for dengue virus. Biosens. Bioelectron. 2017, 91, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Shao, W.; Shurin, M.R.; Wheeler, S.E.; He, X.; Star, A. Rapid detection of SARS-CoV-2 antigens using high-purity semiconducting single-walled carbon nanotube-based field-effect transistors. ACS Appl. Mater. Interfaces 2021, 13, 10321–10327. [Google Scholar] [CrossRef]
- Singh, R.; Sharma, A.; Hong, S.; Jang, J. Electrical immunosensor based on dielectrophoretically-deposited carbon nanotubes for detection of influenza virus h1n1. Analyst 2014, 139, 5415–5421. [Google Scholar] [CrossRef]
- Roberts, A.; Chauhan, N.; Islam, S.; Mahari, S.; Ghawri, B.; Gandham, R.K.; Majumdar, S.; Ghosh, A.; Gandhi, S. Graphene functionalized field-effect transistors for ultrasensitive detection of japanese encephalitis and avian influenza virus. Sci. Rep. 2020, 10, 1–12. [Google Scholar]
- Huang, J.; Xie, Z.; Xie, Z.; Luo, S.; Xie, L.; Huang, L.; Fan, Q.; Zhang, Y.; Wang, S.; Zeng, T. Silver nanoparticles coated graphene electrochemical sensor for the ultrasensitive analysis of avian influenza virus h7. Anal. Chim. Acta 2016, 913, 121–127. [Google Scholar] [CrossRef]
- Gong, Q.; Wang, Y.; Yang, H. A sensitive impedimetric DNA biosensor for the determination of the hiv gene based on graphene-nafion composite film. Biosens. Bioelectron. 2017, 89, 565–569. [Google Scholar] [CrossRef]
- Li, C.; Zou, Z.; Liu, H.; Jin, Y.; Li, G.; Yuan, C.; Xiao, Z.; Jin, M. Synthesis of polystyrene-based fluorescent quantum dots nanolabel and its performance in h5n1 virus and SARS-CoV-2 antibody sensing. Talanta 2021, 225, 122064. [Google Scholar] [CrossRef]
- Afsahi, S.; Lerner, M.B.; Goldstein, J.M.; Lee, J.; Tang, X.; Bagarozzi, D.A., Jr.; Pan, D.; Locascio, L.; Walker, A.; Barron, F.; et al. Novel graphene-based biosensor for early detection of zika virus infection. Biosens. Bioelectron. 2018, 100, 85–88. [Google Scholar] [CrossRef]
- Moitra, P.; Alafeef, M.; Dighe, K.; Frieman, M.B.; Pan, D. Selective naked-eye detection of SARS-CoV-2 mediated by n gene targeted antisense oligonucleotide capped plasmonic nanoparticles. ACS Nano 2020, 14, 7617–7627. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Hu, M.; Liu, A.A.; Lin, Y.; Liu, L.; Yu, B.; Zhou, X.; Pang, D.W. Detection of SARS-CoV-2 by crispr/cas12a-enhanced colorimetry. ACS Sens. 2021, 6, 1086–1093. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, M.; Liu, C.; Chen, J.; Luo, X.; Xue, Y.; Liang, Q.; Zhou, L.; Tao, Y.; Li, M.; et al. Sensitive and rapid on-site detection of SARS-CoV-2 using a gold nanoparticle-based high-throughput platform coupled with crispr/cas12-assisted rt-lamp. Sens. Actuators B Chem. 2021, 345, 130411. [Google Scholar] [CrossRef]
- Kim, J.; Oh, S.Y.; Shukla, S.; Hong, S.B.; Heo, N.S.; Bajpai, V.K.; Chun, H.S.; Jo, C.H.; Choi, B.G.; Huh, Y.S.; et al. Heteroassembled gold nanoparticles with sandwich-immunoassay lspr chip format for rapid and sensitive detection of hepatitis b virus surface antigen (hbsag). Biosens. Bioelectron. 2018, 107, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Ding, L.; Zhou, J.; Chen, S.; Chen, F.; Zhao, C.; Xu, J.; Hu, W.; Ji, J.; Xu, H.; et al. One-step rapid quantification of SARS-CoV-2 virus particles via low-cost nanoplasmonic sensors in generic microplate reader and point-of-care device. Biosens. Bioelectron. 2021, 171, 112685. [Google Scholar] [CrossRef] [PubMed]
- Funari, R.; Chu, K.Y.; Shen, A.Q. Detection of antibodies against SARS-CoV-2 spike protein by gold nanospikes in an opto-microfluidic chip. Biosens. Bioelectron. 2020, 169, 112578. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Zhang, L.Q.; Wei, W.; Zhao, H.Y.; Zhou, Z.X.; Zhang, Y.J.; Liu, S.Q. Colorimetric detection of influenza a virus using antibody-functionalized gold nanoparticles. Analyst 2015, 140, 3989–3995. [Google Scholar] [CrossRef] [PubMed]
- Steinmetz, M.; Lima, D.; Viana, A.G.; Fujiwara, S.T.; Pessoa, C.A.; Etto, R.M.; Wohnrath, K. A sensitive label-free impedimetric DNA biosensor based on silsesquioxane-functionalized gold nanoparticles for zika virus detection. Biosens. Bioelectron. 2019, 141, 111351. [Google Scholar] [CrossRef]
- Bai, Z.K.; Wei, H.J.; Yang, X.S.; Zhu, Y.H.; Peng, Y.J.; Yang, J.; Wang, C.W.; Rong, Z.; Wang, S.Q. Rapid enrichment and ultrasensitive detection of influenza a virus in human specimen using magnetic quantum dot nanobeads based test strips. Sens. Actuators B Chem. 2020, 325, 128780. [Google Scholar] [CrossRef]
- Wu, F.; Yuan, H.; Zhou, C.H.; Mao, M.; Liu, Q.; Shen, H.B.; Cen, Y.; Qin, Z.F.; Ma, L.; Li, L.S. Multiplexed detection of influenza a virus subtype h5 and h9 via quantum dot-based immunoassay. Biosens. Bioelectron. 2016, 77, 464–470. [Google Scholar] [CrossRef]
- Wang, C.; Yang, X.; Zheng, S.; Cheng, X.; Xiao, R.; Li, Q.; Wang, W.; Liu, X.; Wang, S. Development of an ultrasensitive fluorescent immunochromatographic assay based on multilayer quantum dot nanobead for simultaneous detection of SARS-CoV-2 antigen and influenza a virus. Sens. Actuators B Chem. 2021, 345, 130372. [Google Scholar] [CrossRef]
- Ngo, D.B.; Chaibun, T.; Yin, L.S.; Lertanantawong, B.; Surareungchai, W. Electrochemical DNA detection of hepatitis e virus genotype 3 using pbs quantum dot labelling. Anal. Bioanal. Chem. 2021, 413, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Roh, C.; Jo, S.K. Quantitative and sensitive detection of sars coronavirus nucleocapsid protein using quantum dots-conjugated rna aptamer on chip. J. Chem. Technol. Biot. 2011, 86, 1475–1479. [Google Scholar] [CrossRef] [PubMed]
- Son, S.U.; Seo, S.B.; Jane, S.; Choi, J.; Lim, J.W.; Lee, D.K.; Kim, H.; Seo, S.; Kang, T.; Jung, J.; et al. Naked-eye detection of pandemic influenza a (ph1n1) virus by polydiacetylene (pda)-based paper sensor as a point-of-care diagnostic platform. Sens. Actuators B Chem. 2019, 291, 257–265. [Google Scholar] [CrossRef]
- Eissa, S.; Zourob, M. Development of a low-cost cotton-tipped electrochemical immunosensor for the detection of SARS-CoV-2. Anal. Chem. 2021, 93, 1826–1833. [Google Scholar] [CrossRef] [PubMed]
- Lebre, F.; Hearnden, C.H.; Lavelle, E.C. Modulation of immune responses by particulate materials. Adv. Mater. 2016, 28, 5525–5541. [Google Scholar] [CrossRef]
- Yeh, Y.T.; Tang, Y.; Sebastian, A.; Dasgupta, A.; Perea-Lopez, N.; Albert, I.; Lu, H.G.; Terrones, M.; Zheng, S.Y. Tunable and label-free virus enrichment for ultrasensitive virus detection using carbon nanotube arrays. Sci. Adv. 2016, 2, e1601026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, G.; Lee, G.; Kim, M.J.; Baek, S.H.; Choi, M.; Ku, K.B.; Lee, C.S.; Jun, S.; Park, D.; Kim, H.G.; et al. Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor (vol 14, pg 5135, 2020). ACS Nano 2020, 14, 12257–12258. [Google Scholar] [CrossRef]
- Della Ventura, B.; Cennamo, M.; Minopoli, A.; Campanile, R.; Censi, S.B.; Terracciano, D.; Portella, G.; Velotta, R. Colorimetric test for fast detection of SARS-CoV-2 in nasal and throat swabs. ACS Sens. 2020, 5, 3043–3048. [Google Scholar] [CrossRef]
- Yezhelyev, M.V.; Gao, X.; Xing, Y.; Al-Hajj, A.; Nie, S.M.; O’Regan, R.M. Emerging use of nanoparticles in diagnosis and treatment of breast cancer. Lancet Oncol. 2006, 7, 657–667. [Google Scholar] [CrossRef]
- Kevadiya, B.D.; Machhi, J.; Herskovitz, J.; Oleynikov, M.D.; Blomberg, W.R.; Bajwa, N.; Soni, D.; Das, S.; Hasan, M.; Patel, M.; et al. Diagnostics for SARS-CoV-2 infections. Nat. Mater. 2021, 20, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.Y.; Wang, T.; Zhang, B.; Luo, Y.; Mao, L.; Wang, F.; Wu, S.J.; Sun, Z.Y. Detection of igm and igg antibodies in patients with coronavirus disease 2019. Clin. Transl. Immunol. 2020, 9, e1136. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.X.; Liu, B.Z.; Deng, H.J.; Wu, G.C.; Deng, K.; Chen, Y.K.; Liao, P.; Qiu, J.F.; Lin, Y.; Cai, X.F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845. [Google Scholar] [CrossRef] [PubMed]
- Padoan, A.; Sciacovelli, L.; Basso, D.; Negrini, D.; Zuin, S.; Cosma, C.; Faggian, D.; Matricardi, P.; Plebani, M. Iga-ab response to spike glycoprotein of SARS-CoV-2 in patients with COVID-19: A longitudinal study. Clin. Chim. Acta. 2020, 507, 164–166. [Google Scholar] [CrossRef] [PubMed]
- Petherick, A. Developing antibody tests for SARS-CoV-2. Lancet 2020, 395, 1101–1102. [Google Scholar] [CrossRef]
- Hildebrandt, N.; Spillmann, C.M.; Algar, W.R.; Pons, T.; Stewart, M.H.; Oh, E.; Susumu, K.; Diaz, S.A.; Delehanty, J.B.; Medintz, I.L. Energy transfer with semiconductor quantum dot bioconjugates: A versatile platform for biosensing, energy harvesting, and other developing applications. Chem. Rev. 2017, 117, 536–711. [Google Scholar] [CrossRef]
- Kim, H.O.; Na, W.; Yeom, M.; Choi, J.; Kim, J.; Lim, J.W.; Yun, D.; Chun, H.; Park, G.; Park, C.; et al. Host cell mimic polymersomes for rapid detection of highly pathogenic influenza virus via a viral fusion and cell entry mechanism. Adv. Funct. Mater. 2018, 28, 1800960. [Google Scholar] [CrossRef]
- Farzin, L.; Shamsipur, M.; Samandari, L.; Sheibani, S. Hiv biosensors for early diagnosis of infection: The intertwine of nanotechnology with sensing strategies. Talanta 2020, 206, 120201. [Google Scholar] [CrossRef]
- Tymm, C.; Zhou, J.H.; Tadimety, A.; Burklund, A.; Zhang, J.X.J. Scalable COVID-19 detection enabled by lab-on-chip biosensors. Cell Mol. Bioeng. 2020, 13, 313–329. [Google Scholar] [CrossRef] [PubMed]
- Mauter, M.S.; Elimelech, M. Environmental applications of carbon-based nanomaterials. Environ. Sci. Technol. 2008, 42, 5843–5859. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.W.; Zhan, H.; Wang, J.N. A direct foaming approach for carbon nanotube aerogels with ultra-low thermal conductivity and high mechanical stability. Nanoscale 2021, 13, 11878–11886. [Google Scholar] [CrossRef] [PubMed]
- Pasinszki, T.; Krebsz, M.; Tung, T.T.; Losic, D. Carbon nanomaterial based biosensors for non-invasive detection of cancer and disease biomarkers for clinical diagnosis. Sensors 2017, 17, 1919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ehtesabi, H.; Amirfazli, M.; Massah, F.; Bagheri, Z. Application of functionalized carbon dots in detection, diagnostic, disease treatment, and desalination: A review. Adv. Nat. Sci. Nanosci. 2020, 11, 025017. [Google Scholar] [CrossRef]
- Tahriri, M.; Del Monico, M.; Moghanian, A.; Yaraki, M.T.; Torres, R.; Yadegari, A.; Tayebi, L. Graphene and its derivatives: Opportunities and challenges in dentistry. Mater. Sci. Eng. C Mater. 2019, 102, 171–185. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Dai, Z.H. Carbon nanomaterial-based electrochemical biosensors: An overview. Nanoscale 2015, 7, 6420–6431. [Google Scholar] [CrossRef]
- Ehtesabi, H. Carbon nanomaterials for salivary-based biosensors: A review. Mater. Today Chem. 2020, 17, 100342. [Google Scholar] [CrossRef]
- Zhu, Z.Z. An overview of carbon nanotubes and graphene for biosensing applications. Nano-Micro Lett. 2017, 9, 25. [Google Scholar] [CrossRef] [Green Version]
- Tran, T.L.; Nguyen, T.T.; Tran, T.T.H.; Chu, V.T.; Tran, Q.T.; Mai, A.T. Detection of influenza a virus using carbon nanotubes field effect transistor based DNA sensor. Phys. E 2017, 93, 83–86. [Google Scholar] [CrossRef]
- Yu, M.F.; Lourie, O.; Dyer, M.J.; Moloni, K.; Kelly, T.F.; Ruoff, R.S. Strength and breaking mechanism of multiwalled carbon nanotubes under tensile load. Science 2000, 287, 637–640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, R.J.; Zhao, X.; Li, J.; Niu, P.H.; Yang, B.; Wu, H.L.; Wang, W.L.; Song, H.; Huang, B.Y.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef] [Green Version]
- Elias, D.C.; Nair, R.R.; Mohiuddin, T.M.G.; Morozov, S.V.; Blake, P.; Halsall, M.P.; Ferrari, A.C.; Boukhvalov, D.W.; Katsnelson, M.I.; Geim, A.K.; et al. Control of graphene’s properties by reversible hydrogenation: Evidence for graphane. Science 2009, 323, 610–613. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Zhang, H.L.; Aldalbahi, A.; Zuo, X.L.; Fan, C.H.; Mi, X.Q. Fluorescent biosensors enabled by graphene and graphene oxide. Biosens. Bioelectron. 2017, 89, 96–106. [Google Scholar] [CrossRef]
- Pumera, M. Graphene in biosensing. Mater. Today 2011, 14, 308–315. [Google Scholar] [CrossRef]
- Janegitz, B.C.; Silva, T.A.; Wong, A.; Ribovski, L.; Vicentini, F.C.; Sotomayor, M.D.T.; Fatibello, O. The application of graphene for in vitro and in vivo electrochemical biosensing. Biosens. Bioelectron. 2017, 89, 224–233. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.; Hong, S.; Jang, J. Label-free detection of influenza viruses using a reduced graphene oxide-based electrochemical immunosensor integrated with a microfluidic platform. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Dahl, J.A.; Maddux, B.L.S.; Hutchison, J.E. Toward greener nanosynthesis. Chem. Rev. 2007, 107, 2228–2269. [Google Scholar] [CrossRef] [Green Version]
- Harper, B.; Sinche, F.; Wu, R.H.; Gowrishankar, M.; Marquart, G.; Mackiewicz, M.; Harper, S.L. The impact of surface ligands and synthesis method on the toxicity of glutathione-coated gold nanoparticles. Nanomaterials 2014, 4, 355–371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahato, K.; Nagpal, S.; Shah, M.A.; Srivastava, A.; Maurya, P.K.; Roy, S.; Jaiswal, A.; Singh, R.; Chandra, P. Gold nanoparticle surface engineering strategies and their applications in biomedicine and diagnostics. 3 Biotech 2019, 9, 57. [Google Scholar] [CrossRef]
- Chang, C.C.; Chen, C.P.; Wu, T.H.; Yang, C.H.; Lin, C.W.; Chen, C.Y. Gold nanoparticle-based colorimetric strategies for chemical and biological sensing applications. Nanomaterials 2019, 9, 861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glynou, K.; Ioannou, P.C.; Christopoulos, T.K.; Syriopoulou, V. Oligonucleotide-functionalized gold nanoparticles as probes in a dry-reagent strip biosensor for DNA analysis by hybridization. Anal. Chem. 2003, 75, 4155–4160. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xu, T.L.; Wang, W.Q.; Wen, Y.Q.; Li, K.; Qian, L.S.; Zhang, X.J.; Liu, G.D. Lateral flow biosensors based on the use of micro- and nanomaterials: A review on recent developments. Microchim. Acta 2020, 187, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wen, T.; Shi, F.J.; Zeng, X.Y.; Jiao, Y.J. Rapid detection of igm antibodies against the SARS-CoV-2 virus via colloidal gold nanoparticle-based lateral-flow assay. ACS Omega 2020, 5, 12550–12556. [Google Scholar] [CrossRef] [PubMed]
- Yrad, F.M.; Castanares, J.M.; Alocilja, E.C. Visual detection of dengue-1 rna using gold nanoparticle-based lateral flow biosensor. Diagnostics 2019, 9, 74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, F.; Xiao, Y.X.; Chen, B.; Wang, L.Y.; Liu, F.M.; Yao, H.P.; Wu, N.P.; Wu, H.B. Development of a colloidal gold-based immunochromatographic strip test using two monoclonal antibodies to detect h7n9 avian influenza virus. Virus Genes 2020, 56, 396–400. [Google Scholar] [CrossRef]
- Cutler, J.I.; Auyeung, E.; Mirkin, C.A. Spherical nucleic acids. J. Am. Chem. Soc. 2012, 134, 1376–1391. [Google Scholar] [CrossRef]
- Karami, A.; Hasani, M.; Jalilian, F.A.; Ezati, R. Conventional pcr assisted single-component assembly of spherical nucleic acids for simple colorimetric detection of SARS-CoV-2. Sens. Actuators B Chem. 2021, 328, 128971. [Google Scholar] [CrossRef]
- Aldewachi, H.; Chalati, T.; Woodroofe, M.N.; Bricklebank, N.; Sharrack, B.; Gardiner, P. Gold nanoparticle-based colorimetric biosensors. Nanoscale 2018, 10, 18–33. [Google Scholar] [CrossRef] [Green Version]
- Salata, O. Applications of nanoparticles in biology and medicine. J. Nanobiotechnol. 2004, 2, 3. [Google Scholar] [CrossRef] [Green Version]
- Fortina, P.; Kricka, L.J.; Surrey, S.; Grodzinski, P. Nanobiotechnology: The promise and reality of new approaches to molecular recognition. Trends Biotechnol. 2005, 23, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Talapin, D.V.; Lee, J.S.; Kovalenko, M.V.; Shevchenko, E.V. Prospects of colloidal nanocrystals for electronic and optoelectronic applications. Chem. Rev. 2010, 110, 389–458. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Shen, Y.; Adogla, E.A.; Viswanath, A.; Tan, R.; Benicewicz, B.C.; Greytak, A.B.; Lin, Y.; Wang, Q. Surface labeling of enveloped virus with polymeric imidazole ligand-capped quantum dots via the metabolic incorporation of phospholipids into host cells. J. Mater. Chem. B 2016, 4, 2421–2427. [Google Scholar] [CrossRef]
- Azzazy, H.M.; Mansour, M.M.; Kazmierczak, S.C. From diagnostics to therapy: Prospects of quantum dots. Clin. Biochem. 2007, 40, 917–927. [Google Scholar] [CrossRef]
- Wang, R.; Wu, X.; Xu, K.; Zhou, W.; Shang, Y.; Tang, H.; Chen, H.; Ning, Z. Highly efficient inverted structural quantum dot solar cells. Adv. Mater. 2018, 30, 1704882. [Google Scholar] [CrossRef]
- Bruchez, M.P. Turning all the lights on: Quantum dots in cellular assays. Curr. Opin. Chem. Biol. 2005, 9, 533–537. [Google Scholar] [CrossRef]
- Nguyen, A.V.T.; Dao, T.D.; Trinh, T.T.T.; Choi, D.Y.; Yu, S.T.; Park, H.; Yeo, S.J. Sensitive detection of influenza a virus based on a cdse/cds/zns quantum dot-linked rapid fluorescent immunochromatographic test. Biosens. Bioelectron. 2020, 155, 112090. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Chen, Y.; Liang, X.; Zhang, G.; Ma, H.; Nie, L.; Wang, Y. Detection of hepatitis b virus m204i mutation by quantum dot-labeled DNA probe. Sensors 2017, 17, 961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, Y.P.; Li, W.; Zhang, F.; Liu, Z.H.; Ezazi, N.Z.; Liu, D.F.; Santos, H.A. Electrospun fibrous architectures for drug delivery, tissue engineering and cancer therapy. Adv. Funct. Mater. 2019, 29, 1802852. [Google Scholar] [CrossRef]
- Kim, H.; Park, Y.; Stevens, M.M.; Kwon, W.; Hahn, S.K. Multifunctional hyaluronate—Nanoparticle hybrid systems for diagnostic, therapeutic and theranostic applications. J. Control. Release 2019, 303, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.H.; Chen, X.Y.; Fu, L.Q.; Du, W.L.; Yang, X.; Mou, X.Z.; Hu, P.Y. Design and development of hybrid hydrogels for biomedical applications: Recent trends in anticancer drug delivery and tissue engineering. Front. Bioeng. Biotechnol. 2021, 9, 630943. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.O.; Kim, E.; An, Y.; Choi, J.; Jang, E.; Choi, E.B.; Kukreja, A.; Kim, M.H.; Kang, B.; Kim, D.J.; et al. A biodegradable polymersome containing bcl-xl sirna and doxorubicin as a dual delivery vehicle for a synergistic anticancer effect. Macromol. Biosci. 2013, 13, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Y.; Jiang, H.B.; Kim, J.E.; Zhang, S.X.; Kim, K.M.; Kwon, J.S. Bioresorbable magnesium-reinforced pla membrane for guided bone/tissue regeneration. J. Mech. Behav. Biomed. 2020, 112, 104061. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.H.; Xu, A.P.; Zhang, Z.Q.; Chen, Z.H.; Wan, L.; Shi, X.; Lin, S.X.; Yuan, Z.Y.; Deng, L.B. Bioactive silver doped hydroxyapatite composite coatings on metal substrates: Synthesis and characterization. Mater. Chem. Phys. 2018, 218, 130–139. [Google Scholar] [CrossRef]
- Wei, X.; Cai, J.; Lin, S.; Li, F.; Tian, F. Controlled release of monodisperse silver nanoparticles via in situ cross-linked polyvinyl alcohol as benign and antibacterial electrospun nanofibers. Colloids Surf B Biointerfaces 2021, 197, 111370. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Ha, K.; Guk, K.; Hwang, S.G.; Choi, J.M.; Kang, T.; Bae, P.; Jung, J.; Lim, E.K. Colorimetric detection of influenza a (h1n1) virus by a peptide-functionalized polydiacetylene (pep-pda) nanosensor. RSC Adv. 2016, 6, 48566–48570. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.O.; Yeom, M.; Kim, J.; Kukreja, A.; Na, W.; Choi, J.; Kang, A.; Yun, D.; Lim, J.W.; Song, D.; et al. Reactive oxygen species-regulating polymersome as an antiviral agent against influenza virus. Small 2017, 13, 1700818. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Chung, S.J.; Park, H.G.; Cho, J.-H.; Kim, M.; Chung, B.H. Mixed self-assembly of polydiacetylenes for highly specific and sensitive strip biosensors. Biosens. Bioelectron. 2008, 24, 480–484. [Google Scholar] [CrossRef]
- Jiang, L.; Luo, J.; Dong, W.; Wang, C.; Jin, W.; Xia, Y.; Wang, H.; Ding, H.; Jiang, L.; He, H. Development and evaluation of a polydiacetylene based biosensor for the detection of h5 influenza virus. J. Virol. Methods 2015, 219, 38–45. [Google Scholar] [CrossRef]
- Dhand, C.; Das, M.; Datta, M.; Malhotra, B.D. Recent advances in polyaniline based biosensors. Biosens. Bioelectron. 2011, 26, 2811–2821. [Google Scholar] [CrossRef]
- Dutta, R.; Thangapandi, K.; Mondal, S.; Nanda, A.; Bose, S.; Sanyal, S.; Jana, S.K.; Ghorai, S. Polyaniline based electrochemical sensor for the detection of dengue virus infection. Avicenna J. Med. Biotechnol. 2020, 12, 77–84. [Google Scholar]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef]
- Smith, D.M.; Simon, J.K.; Baker Jr, J.R. Applications of nanotechnology for immunology. Nat. Rev. Immunol. 2013, 13, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-G.; Park, J.Y.; Shon, Y.; Kim, G.; Shim, G.; Oh, Y.-K. Nanotechnology and vaccine development. Asian J. Pharm. Sci. 2014, 9, 227–235. [Google Scholar] [CrossRef] [Green Version]
- van Riet, E.; Ainai, A.; Suzuki, T.; Kersten, G.; Hasegawa, H. Combatting infectious diseases; nanotechnology as a platform for rational vaccine design. Adv. Drug Deliv. 2014, 74, 28–34. [Google Scholar] [CrossRef]
- Kim, E.; Lim, E.K.; Park, G.; Park, C.; Lim, J.W.; Lee, H.; Na, W.; Yeom, M.; Kim, J.; Song, D. Advanced nanomaterials for preparedness against (re-) emerging viral diseases. Adv. Mater. 2021, 2005927. [Google Scholar] [CrossRef] [PubMed]
- Elberry, M.H.; Darwish, N.H.; Mousa, S.A. Hepatitis c virus management: Potential impact of nanotechnology. Virol. J. 2017, 14, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Yan, Y.; Wang, X.; Lou, P.; Hu, Z.; Qu, P.; Li, D.; Li, Q.; Xu, Y.; Niu, J.; He, Y. A nanoparticle-based hepatitis c virus vaccine with enhanced potency. J. Infect. Dis. 2020, 221, 1304–1314. [Google Scholar]
- Tian, Y.; Wang, H.; Liu, Y.; Mao, L.; Chen, W.; Zhu, Z.; Liu, W.; Zheng, W.; Zhao, Y.; Kong, D.; et al. A peptide-based nanofibrous hydrogel as a promising DNA nanovector for optimizing the efficacy of hiv vaccine. Nano Lett. 2014, 14, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, C. Role of nanotechnology in hiv/aids vaccine development. Adv. Drug Deliv. Rev. 2016, 103, 76–89. [Google Scholar] [CrossRef]
- Shin, H.N.; Iwasaki, A. A vaccine strategy that protects against genital herpes by establishing local memory t cells. Nature 2012, 491, 463. [Google Scholar] [CrossRef] [Green Version]
- Luo, M.; Wang, H.; Wang, Z.; Cai, H.; Lu, Z.; Li, Y.; Du, M.; Huang, G.; Wang, C.; Chen, X. A sting-activating nanovaccine for cancer immunotherapy. Nat. Nanotechnol. 2017, 12, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Zhang, F.; Ni, Q.; Niu, G.; Chen, X. Efficient nanovaccine delivery in cancer immunotherapy. ACS Nano 2017, 11, 2387–2392. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Angelova, A. Coronavirus-induced host cubic membranes and lipid-related antiviral therapies: A focus on bioactive plasmalogens. Front. Cell Dev. Biol. 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Malone, R.W.; Felgner, P.L.; Verma, I.M. Cationic liposome-mediated rna transfection. Proc. Nat. Acad. Sci. USA 1989, 86, 6077–6081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Felgner, J.H.; Kumar, R.; Sridhar, C.; Wheeler, C.J.; Tsai, Y.J.; Border, R.; Ramsey, P.; Martin, M.; Felgner, P.L. Enhanced gene delivery and mechanism studies with a novel series of cationic lipid formulations. J. Biol. Chem. 1994, 269, 2550–2561. [Google Scholar] [CrossRef]
- Brunel, F.; Darbouret, A.; Ronco, J. Cationic lipid dc-chol induces an improved and balanced immunity able to overcome the unresponsiveness to the hepatitis b vaccine. Vaccine 1999, 17, 2192–2203. [Google Scholar] [CrossRef]
- Henriksen-Lacey, M.; Christensen, D.; Bramwell, V.W.; Lindenstrøm, T.; Agger, E.M.; Andersen, P.; Perrie, Y. Liposomal cationic charge and antigen adsorption are important properties for the efficient deposition of antigen at the injection site and ability of the vaccine to induce a cmi response. J. Control. Release 2010, 145, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Henriksen-Lacey, M.; Christensen, D.; Bramwell, V.W.; Lindenstrøm, T.; Agger, E.M.; Andersen, P.; Perrie, Y. Comparison of the depot effect and immunogenicity of liposomes based on dimethyldioctadecylammonium (dda), 3β-[n-(n′, n′-dimethylaminoethane) carbomyl] cholesterol (dc-chol), and 1, 2-dioleoyl-3-trimethylammonium propane (dotap): Prolonged liposome retention mediates stronger th1 responses. Mol. Pharm. 2011, 8, 153–161. [Google Scholar]
- Pardi, N.; Hogan, M.J.; Pelc, R.S.; Muramatsu, H.; Andersen, H.; DeMaso, C.R.; Dowd, K.A.; Sutherland, L.L.; Scearce, R.M.; Parks, R. Zika virus protection by a single low-dose nucleoside-modified mrna vaccination. Nature 2017, 543, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Lou, G.; Anderluzzi, G.; Schmidt, S.T.; Woods, S.; Gallorini, S.; Brazzoli, M.; Giusti, F.; Ferlenghi, I.; Johnson, R.N.; Roberts, C.W. Delivery of self-amplifying mrna vaccines by cationic lipid nanoparticles: The impact of cationic lipid selection. J. Control. Release 2020, 325, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Lappalainen, K.; Jääskeläinen, I.; Syrjänen, K.; Urtti, A.; Syrjänen, S. Comparison of cell proliferation and toxicity assays using two cationic liposomes. Pharm. Res. 1994, 11, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Lizarzaburu, M.E.; Kurth, M.J.; Liu, L.; Wege, H.; Zern, M.A.; Nantz, M.H. Cationic lipid polymerization as a novel approach for constructing new DNA delivery agents. Bioconj. Chem. 2001, 12, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Webster, P.; Davis, M.E. Pegylation significantly affects cellular uptake and intracellular trafficking of non-viral gene delivery particles. Eur. J. Cell Biol. 2004, 83, 97–111. [Google Scholar] [CrossRef]
- Zhang, J.-S.; Liu, F.; Huang, L. Implications of pharmacokinetic behavior of lipoplex for its inflammatory toxicity. Adv. Drug Deliv. Rev. 2005, 57, 689–698. [Google Scholar] [CrossRef]
- Yi Xue, H.; Guo, P.; Wen, W.-C.; Lun Wong, H. Lipid-based nanocarriers for rna delivery. Curr. Pharm. Des. 2015, 21, 3140–3147. [Google Scholar] [CrossRef]
- Siefert, A.L.; Caplan, M.J.; Fahmy, T.M. Artificial bacterial biomimetic nanoparticles synergize pathogen-associated molecular patterns for vaccine efficacy. Biomaterials 2016, 97, 85–96. [Google Scholar] [CrossRef]
- Wei, X.; Zhang, G.; Ran, D.; Krishnan, N.; Fang, R.H.; Gao, W.; Spector, S.A.; Zhang, L. T-cell-mimicking nanoparticles can neutralize hiv infectivity. Adv. Mater. 2018, 30, 1802233. [Google Scholar] [CrossRef]
- Lin, L.C.W.; Huang, C.Y.; Yao, B.Y.; Lin, J.C.; Agrawal, A.; Algaissi, A.; Peng, B.H.; Liu, Y.H.; Huang, P.H.; Juang, R.H. Viromimetic sting agonist-loaded hollow polymeric nanoparticles for safe and effective vaccination against middle east respiratory syndrome coronavirus. Adv. Funct. Mater. 2019, 29, 1807616. [Google Scholar] [CrossRef] [Green Version]
- Stano, A.; Scott, E.A.; Dane, K.Y.; Swartz, M.A.; Hubbell, J.A. Tunable t cell immunity towards a protein antigen using polymersomes vs. Solid-core nanoparticles. Biomaterials 2013, 34, 4339–4346. [Google Scholar] [CrossRef]
- Zheng, B.; Peng, W.; Guo, M.; Huang, M.; Gu, Y.; Wang, T.; Ni, G.; Ming, D. Inhalable nanovaccine with biomimetic coronavirus structure to trigger mucosal immunity of respiratory tract against COVID-19. Chem. Eng. J. 2021, 418, 129392. [Google Scholar] [CrossRef]
- Lim, J.-W.; Na, W.; Kim, H.-O.; Yeom, M.; Kang, A.; Park, G.; Park, C.; Ki, J.; Lee, S.; Jung, B. Co-delivery of antigens and immunostimulants via a polymersome for improvement of antigen-specific immune response. J. Mater. Chem. B 2020, 8, 5620–5626. [Google Scholar] [CrossRef]
- Wegmann, F.; Gartlan, K.H.; Harandi, A.M.; Brinckmann, S.A.; Coccia, M.; Hillson, W.R.; Kok, W.L.; Cole, S.; Ho, L.-P.; Lambe, T. Polyethyleneimine is a potent mucosal adjuvant for viral glycoprotein antigens. Nat. Biotechnol. 2012, 30, 883–888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noh, Y.W.; Hong, J.H.; Shim, S.M.; Park, H.S.; Bae, H.H.; Ryu, E.K.; Hwang, J.H.; Lee, C.H.; Cho, S.H.; Sung, M.H. Polymer nanomicelles for efficient mucus delivery and antigen-specific high mucosal immunity. Angew. Chemie 2013, 125, 7838–7843. [Google Scholar] [CrossRef]
- Bernocchi, B.; Carpentier, R.; Lantier, I.; Ducournau, C.; Dimier-Poisson, I.; Betbeder, D. Mechanisms allowing protein delivery in nasal mucosa using npl nanoparticles. J. Control. Release 2016, 232, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Qiao, D.; Liu, L.; Chen, Y.; Xue, C.; Gao, Q.; Mao, H.-Q.; Leong, K.W.; Chen, Y. Potency of a scalable nanoparticulate subunit vaccine. Nano Lett. 2018, 18, 3007–3016. [Google Scholar] [CrossRef]
- Kim, E.H.; Woodruff, M.C.; Grigoryan, L.; Maier, B.; Lee, S.H.; Mandal, P.; Cortese, M.; Natrajan, M.S.; Ravindran, R.; Ma, H. Squalene emulsion-based vaccine adjuvants stimulate cd8 t cell, but not antibody responses, through a ripk3-dependent pathway. Elife 2020, 9, e52687. [Google Scholar] [CrossRef] [PubMed]
- Garçon, N.; Vaughn, D.W.; Didierlaurent, A.M. Development and evaluation of as03, an adjuvant system containing α-tocopherol and squalene in an oil-in-water emulsion. Exp. Rev. Vaccines 2012, 11, 349–366. [Google Scholar] [CrossRef] [PubMed]
- Klucker, M.F.; Dalençon, F.; Probeck, P.; Haensler, J. Af03, an alternative squalene emulsion-based vaccine adjuvant prepared by a phase inversion temperature method. J. Pharm. Sci. 2012, 101, 4490–4500. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Ji, Z.; Liao, Y.-P.; Chang, C.H.; Wang, X.; Ku, J.; Xue, C.; Mirshafiee, V.; Xia, T. Enhanced immune adjuvant activity of aluminum oxyhydroxide nanorods through cationic surface functionalization. ACS Appl. Mater. Interfaces 2017, 9, 21697–21705. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Wang, Q.; Li, L.; Zeng, Q.; Li, H.; Gong, T.; Zhang, Z.; Sun, X. Turning the old adjuvant from gel to nanoparticles to amplify cd8+ t cell responses. Adv. Sci. 2018, 5, 1700426. [Google Scholar] [CrossRef]
- Joffre, O.P.; Segura, E.; Savina, A.; Amigorena, S. Cross-presentation by dendritic cells. Nat. Rev. Immunol. 2012, 12, 557–569. [Google Scholar] [CrossRef]
- Sun, B.; Ji, Z.; Liao, Y.-P.; Wang, M.; Wang, X.; Dong, J.; Chang, C.H.; Li, R.; Zhang, H.; Nel, A.E. Engineering an effective immune adjuvant by designed control of shape and crystallinity of aluminum oxyhydroxide nanoparticles. ACS Nano 2013, 7, 10834–10849. [Google Scholar] [CrossRef] [Green Version]
- Reimer, J.M.; Karlsson, K.H.; Lövgren-Bengtsson, K.; Magnusson, S.E.; Fuentes, A.; Stertman, L. Matrix-m™ adjuvant induces local recruitment, activation and maturation of central immune cells in absence of antigen. PLoS ONE 2012, 7, e41451. [Google Scholar] [CrossRef] [Green Version]
- Magnusson, S.E.; Reimer, J.M.; Karlsson, K.H.; Lilja, L.; Bengtsson, K.L.; Stertman, L. Immune enhancing properties of the novel matrix-m™ adjuvant leads to potentiated immune responses to an influenza vaccine in mice. Vaccine 2013, 31, 1725–1733. [Google Scholar] [CrossRef]
- Venkatraman, N.; Anagnostou, N.; Bliss, C.; Bowyer, G.; Wright, D.; Lövgren-Bengtsson, K.; Roberts, R.; Poulton, I.; Lawrie, A.; Ewer, K.; et al. Safety and immunogenicity of heterologous prime-boost immunization with viral-vectored malaria vaccines adjuvanted with matrix-m™. Vaccine 2017, 35, 6208–6217. [Google Scholar] [CrossRef]
- Magnusson, S.E.; Altenburg, A.F.; Bengtsson, K.L.; Bosman, F.; de Vries, R.D.; Rimmelzwaan, G.F.; Stertman, L. Matrix-m™ adjuvant enhances immunogenicity of both protein- and modified vaccinia virus ankara-based influenza vaccines in mice. Immunol. Res. 2018, 66, 224–233. [Google Scholar] [CrossRef] [Green Version]
- Lou, Z.Y.; Sun, Y.N.; Rao, Z.H. Current progress in antiviral strategies. Trends Pharmacol. Sci. 2014, 35, 86–102. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E. Antiviral agents active against influenza a viruses. Nat. Rev. Drug Discov. 2006, 5, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Riva, L.; Yuan, S.F.; Yin, X.; Martin-Sancho, L.; Matsunaga, N.; Pache, L.; Burgstaller-Muehlbacher, S.; De Jesus, P.D.; Teriete, P.; Hull, M.V.; et al. Discovery of SARS-CoV-2 antiviral drugs through large-scale compound repurposing. Nature 2020, 586, 113. [Google Scholar] [CrossRef]
- Medhi, R.; Srinoi, P.; Ngo, N.; Tran, H.V.; Lee, T.R. Nanoparticle-based strategies to combat COVID-19. ACS Appl. Nano Mater. 2020, 3, 8557–8580. [Google Scholar] [CrossRef]
- Derudas, M.; McGuigan, C.; Brancale, A.; Bugert, J.; Andrei, G.; Snoeck, R.; Balzarini, J. Design, synthesis and biological evaluation of novel acyclovir protides. Antivir. Res. 2008, 78, A55–A56. [Google Scholar] [CrossRef]
- Javan, F.; Vatanara, A.; Azadmanesh, K.; Nabi-Meibodi, M.; Shakouri, M. Encapsulation of ritonavir in solid lipid nanoparticles: In-vitro anti-hiv-1 activity using lentiviral particles. J. Pharm. Pharmacol. 2017, 69, 1002–1009. [Google Scholar] [CrossRef]
- Mehranfar, A.; Izadyar, M. Theoretical design of functionalized gold nanoparticles as antiviral agents against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). J. Phys. Chem. Lett. 2020, 11, 10284–10289. [Google Scholar] [CrossRef]
- Chun, H.; Yeom, M.; Kim, H.O.; Lim, J.W.; Na, W.; Park, G.; Park, C.; Kang, A.; Yun, D.; Kim, J.; et al. Efficient antiviral co-delivery using polymersomes by controlling the surface density of cell-targeting groups for influenza a virus treatment. Polym. Chem. 2018, 9, 2116–2123. [Google Scholar] [CrossRef]
- Figueira, T.N.; Domingues, M.M.; Illien, F.; Cadima-Couto, I.; Todorovski, T.; Andreu, D.; Sagan, S.; Castanho, M.A.R.B.; Walrant, A.; Veiga, A.S. Enfuvirtide-protoporphyrin ix dual-loaded liposomes: In vitro evidence of synergy against hiv-1 entry into cells. ACS Infect. Dis. 2020, 6, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Joshy, K.S.; Snigdha, S.; George, A.; Kalarikkal, N.; Pothen, L.A.; Thomas, S. Core-shell nanoparticles of carboxy methyl cellulose and compritol-peg for antiretroviral drug delivery. Cellulose 2017, 24, 4759–4771. [Google Scholar] [CrossRef]
- Cao, S.J.; Slack, S.D.; Levy, C.N.; Hughes, S.M.; Jiang, Y.H.; Yogodzinski, C.; Roychoudhury, P.; Jerome, K.R.; Schiffer, J.T.; Hladik, F.; et al. Hybrid nanocarriers incorporating mechanistically distinct drugs for lymphatic cd4(+) t cell activation and hiv-1 latency reversal. Sci. Adv. 2019, 5, eaav6322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joshy, K.S.; Snigdha, S.; George, A.; Kalarikkal, N.; Pothen, L.A.; Thomas, S. Poly (vinyl pyrrolidone)-lipid based hybrid nanoparticles for anti viral drug delivery. Chem. Phys. Lipids 2018, 210, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Lima, T.L.C.; Feitosa, R.D.; dos Santos-Silva, E.; dos Santos-Silva, A.M.; Siqueira, E.M.D.; Machado, P.R.L.; Cornelio, A.M.; do Egito, E.S.T.; Fernandes-Pedrosa, M.D.; Farias, K.J.S.; et al. Improving encapsulation of hydrophilic chloroquine diphosphate into biodegradable nanoparticles: A promising approach against herpes virus simplex-1 infection. Pharmaceutics 2018, 10, 255. [Google Scholar] [CrossRef] [Green Version]
- Kong, B.; Moon, S.; Kim, Y.; Heo, P.; Jung, Y.; Yu, S.H.; Chung, J.; Ban, C.; Kim, Y.H.; Kim, P.; et al. Virucidal nano-perforator of viral membrane trapping viral rnas in the endosome. Nat. Commun. 2019, 10, 185. [Google Scholar] [CrossRef] [Green Version]
- Papp, I.; Sieben, C.; Ludwig, K.; Roskamp, M.; Bottcher, C.; Schlecht, S.; Herrmann, A.; Haag, R. Inhibition of influenza virus infection by multivalent sialic-acid-functionalized gold nanoparticles. Small 2010, 6, 2900–2906. [Google Scholar] [CrossRef] [PubMed]
- Cagno, V.; Andreozzi, P.; D’Alicarnasso, M.; Jacob Silva, P.; Mueller, M.; Galloux, M.; Le Goffic, R.; Jones, S.T.; Vallino, M.; Hodek, J.; et al. Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nat. Mater. 2018, 17, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.N.; Hsueh, Y.H.; Hsieh, C.T.; Tzou, D.Y.; Chang, P.L. Antiviral activity of graphene-silver nanocomposites against non-enveloped and enveloped viruses. Int. J. Environ. Res. Public Health 2016, 13, 430. [Google Scholar] [CrossRef] [Green Version]
- Lv, X.N.; Wang, P.; Bai, R.; Cong, Y.Y.; Suo, S.Q.G.W.; Ren, X.F.; Chen, C.Y. Inhibitory effect of silver nanomaterials on transmissible virus-induced host cell infections. Biomaterials 2014, 35, 4195–4203. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.; Ansar, M.; Speshock, J.; Ivanciuc, T.; Qu, Y.; Casola, A.; Garofalo, R.P. Antiviral and immunomodulatory activity of silver nanoparticles in experimental rsv infection. Viruses 2019, 11, 732. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.F.; Li, Y.H.; Guo, M.; Xiao, M.S.; Wang, C.B.; Zhao, M.Q.; Xu, T.T.; Xia, Y.; Zhu, B. Inhibition of h1n1 influenza virus by selenium nanoparticles loaded with zanamivir through p38 and jnk signaling pathways. RSC Adv. 2017, 7, 35290–35296. [Google Scholar] [CrossRef] [Green Version]
- Ghaffari, H.; Tavakoli, A.; Moradi, A.; Tabarraei, A.; Bokharaei-Salim, F.; Zahmatkeshan, M.; Farahmand, M.; Javanmard, D.; Kiani, S.J.; Esghaei, M.; et al. Inhibition of h1n1 influenza virus infection by zinc oxide nanoparticles: Another emerging application of nanomedicine. J. Biomed. Sci. 2019, 26, 70. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.M.D.E.; Hanchuk, T.D.M.; Santos, M.I.; Kobarg, J.; Bajgelman, M.C.; Cardoso, M.B. Viral inhibition mechanism mediated by surface-modified silica nanoparticles. ACS Appl. Mater. Interfaces 2016, 8, 16564–16572. [Google Scholar] [CrossRef]
- Bromberg, L.; Bromberg, D.J.; Hatton, T.A.; Bandin, I.; Concheiro, A.; Alvarez-Lorenzo, C. Antiviral properties of polymeric aziridine- and biguanide-modified core-shell magnetic nanoparticles. Langmuir 2012, 28, 4548–4558. [Google Scholar] [CrossRef]
- Du, T.; Liang, J.G.; Dong, N.; Lu, J.; Fu, Y.Y.; Fang, L.R.; Xiao, S.B.; Han, H.Y. Glutathione-capped ag2s nanoclusters inhibit coronavirus proliferation through blockage of viral rna synthesis and budding. ACS Appl. Mater. Interfaces 2018, 10, 4369–4378. [Google Scholar] [CrossRef] [PubMed]
- Loczechin, A.; Seron, K.; Barras, A.; Giovanelli, E.; Belouzard, S.; Chen, Y.T.; Metzler-Nolte, N.; Boukherroub, R.; Dubuisson, J.; Szunerits, S. Functional carbon quantum dots as medical countermeasures to human coronavirus. Acs Appl. Mater. Interfaces 2019, 11, 42964–42974. [Google Scholar] [CrossRef] [PubMed]
- Ting, D.; Dong, N.; Fang, L.; Lu, J.; Bi, J.; Xiao, S.; Han, H. Multisite inhibitors for enteric coronavirus: Antiviral cationic carbon dots based on curcumin. ACS Appl. Nano Mater. 2018, 1, 5451–5459. [Google Scholar] [CrossRef]
- Tong, T.; Hu, H.W.; Zhou, J.W.; Deng, S.F.; Zhang, X.T.; Tang, W.T.; Fang, L.R.; Xiao, S.B.; Liang, J.G. Glycyrrhizic-acid-based carbon dots with high antiviral activity by multisite inhibition mechanisms. Small 2020, 16, 1906206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, S.M.; Gu, J.J.; Ye, J.; Fang, B.; Wan, S.F.; Wang, C.Y.; Ashraf, U.; Li, Q.; Wang, X.G.; Shao, L.; et al. Benzoxazine monomer derived carbon dots as a broad-spectrum agent to block viral infectivity. J. Colloid Interface Sci. 2019, 542, 198–206. [Google Scholar] [CrossRef]
- Zoppe, J.O.; Ruottinen, V.; Ruotsalainen, J.; Ronkko, S.; Johansson, L.S.; Hinkkanen, A.; Jarvinen, K.; Seppala, J. Synthesis of cellulose nanocrystals carrying tyrosine sulfate mimetic ligands and inhibition of alphavirus infection. Biomacromolecules 2014, 15, 1534–1542. [Google Scholar] [CrossRef]
- Ye, S.Y.; Shao, K.; Li, Z.H.; Guo, N.; Zuo, Y.P.; Li, Q.; Lu, Z.C.; Chen, L.; He, Q.G.; Han, H.Y. Antiviral activity of graphene oxide: How sharp edged structure and charge matter. ACS Appl. Mater. Interfaces 2015, 7, 21571–21579. [Google Scholar] [CrossRef]
- Chakravarthy, K.V.; Bonoiu, A.C.; Davis, W.G.; Ranjan, P.; Ding, H.; Hu, R.; Bowzard, J.B.; Bergey, E.J.; Katz, J.M.; Knight, P.R.; et al. Gold nanorod delivery of an ssrna immune activator inhibits pandemic h1n1 influenza viral replication. Proc. Natl. Acad. Sci. USA 2010, 107, 10172–10177. [Google Scholar] [CrossRef] [Green Version]
- Surnar, B.; Kamran, M.Z.; Shah, A.S.; Dhar, S. Clinically approved antiviral drug in an orally administrable nanoparticle for COVID-19. ACS Pharmacol. Transl. 2020, 3, 1371–1380. [Google Scholar] [CrossRef]
- Delshadi, R.; Bahrami, A.; McClements, D.J.; Moore, M.D.; Williams, L. Development of nanoparticle-delivery systems for antiviral agents: A review. J. Control. Release 2021, 331, 30–44. [Google Scholar] [CrossRef]
- Chakravarty, M.; Vora, A. Nanotechnology-based antiviral therapeutics. Drug Deliv. Transl. Res. 2021, 11, 748–787. [Google Scholar] [CrossRef]
- Kwon, S.J.; Na, D.H.; Kwak, J.H.; Douaisi, M.; Zhang, F.; Park, E.J.; Park, J.H.; Youn, H.; Song, C.S.; Kane, R.S.; et al. Nanostructured glycan architecture is important in the inhibition of influenza a virus infection. Nat. Nanotechnol. 2017, 12, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Nie, C.; Stadtmüller, M.; Yang, H.; Xia, Y.; Wolff, T.; Cheng, C.; Haag, R. Spiky nanostructures with geometry-matching topography for virus inhibition. Nano Lett. 2020, 20, 5367–5375. [Google Scholar] [CrossRef] [PubMed]
- Lauster, D.; Glanz, M.; Bardua, M.; Ludwig, K.; Hellmund, M.; Hoffmann, U.; Hamann, A.; Bottcher, C.; Haag, R.; Hackenberger, C.P.R.; et al. Multivalent peptide-nanoparticle conjugates for influenza-virus inhibition. Angew. Chem. Int. Ed. 2017, 56, 5931–5936. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.X.; Li, C.M.; Huang, C.Z. Curcumin modified silver nanoparticles for highly efficient inhibition of respiratory syncytial virus infection. Nanoscale 2016, 8, 3040–3048. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Xia, S.; Xu, W.; Tian, R.; Yu, G.; Gu, C.; Pan, P.; Meng, Q.-F.; Cai, X.; Qu, D.; et al. Decoy nanoparticles protect against COVID-19 by concurrently adsorbing viruses and inflammatory cytokines. Proc. Natl. Acad. Sci. USA 2020, 117, 27141. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, Z.; Dinh, P.-U.C.; Zhu, D.; Popowski, K.D.; Lutz, H.; Hu, S.; Lewis, M.G.; Cook, A.; Andersen, H.; et al. Cell-mimicking nanodecoys neutralize SARS-CoV-2 and mitigate lung injury in a non-human primate model of COVID-19. Nat. Nanotechnol. 2021, 16, 942–951. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.J.; Chang, L.; Chu, H.W.; Lin, H.J.; Chang, P.C.; Wang, R.Y.L.; Unnikrishnan, B.; Mao, J.Y.; Chen, S.Y.; Huang, C.C. High amplification of the antiviral activity of curcumin through transformation into carbon quantum dots. Small 2019, 15, 1902641. [Google Scholar] [CrossRef]
- Li, Y.H.; Lin, Z.F.; Guo, M.; Zhao, M.Q.; Xia, Y.; Wang, C.B.; Xu, T.T.; Zhu, B. Inhibition of h1n1 influenza virus-induced apoptosis by functionalized selenium nanoparticles with amantadine through ros-mediated akt signaling pathways. Int. J. Nanomed. 2018, 13, 2005–2016. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.H.; Lin, Z.F.; Zhao, M.Q.; Xu, T.T.; Wang, C.B.; Hua, L.; Wang, H.Z.; Xia, H.M.; Zhu, B. Silver nanoparticle based codelivery of oseltamivir to inhibit the activity of the h1n1 influenza virus through ros-mediated signaling pathways. ACS Appl. Mater. Interfaces 2016, 8, 24385–24393. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.F.; Li, Y.H.; Guo, M.; Xu, T.T.; Wang, C.B.; Zhao, M.Q.; Wang, H.Z.; Chen, T.F.; Zhu, B. The inhibition of h1n1 influenza virus-induced apoptosis by silver nanoparticles functionalized with zanamivir. RSC Adv. 2017, 7, 742–750. [Google Scholar] [CrossRef] [Green Version]
- Rakowska, P.D.; Tiddia, M.; Faruqui, N.; Bankier, C.; Pei, Y.; Pollard, A.J.; Zhang, J.; Gilmore, I.S. Antiviral surfaces and coatings and their mechanisms of action. Commun. Mater. 2021, 2, 53. [Google Scholar] [CrossRef]
- Borkow, G.; Zhou, S.S.; Page, T.; Gabbay, J. A novel anti-influenza copper oxide containing respiratory face mask. PLoS ONE 2010, 5, e11295. [Google Scholar] [CrossRef] [Green Version]
- Peddinti, B.S.T.; Scholle, F.; Ghiladi, R.A.; Spontak, R.J. Photodynamic polymers as comprehensive anti-infective materials: Staying ahead of a growing global threat. ACS Appl. Mater. Interfaces 2018, 10, 25955–25959. [Google Scholar] [CrossRef]
- Mori, Y.; Ono, T.; Miyahira, Y.; Nguyen, V.Q.; Matsui, T.; Ishihara, M. Antiviral activity of silver nanoparticle/chitosan composites against h1n1 influenza a virus. Nanoscale Res. Lett. 2013, 8, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Nanomaterials | Diagnostic Techniques | Target | LOD | Time | Ref. |
|---|---|---|---|---|---|
| Carbon nanotubes | RDT | DENV | 8.4 × 102 TCID50/mL | >10 min | [6] |
| SARS-CoV-2 | 0.55 fg/mL | >5 min | [7] | ||
| Immunological | Influenza A Virus (H1N1) | 1 PFU/ml | 30 min | [8] | |
| Graphene | Immunological | JEV/AIV | 1 fM/10 fM | 1 h | [9] |
| AIV | 1.6 pg/mL | 30 min | [10] | ||
| HIV-1 | 2.3 × 10−14 M | 1 h | [11] | ||
| Influenza A Virus (H5N1) | 25 PFU/mL | 15 min | [12] | ||
| Zika Virus | 450 pmol/L | 5 min | [13] | ||
| AuNPs | Optical | SARS-CoV-2 | 0.18 ng/μL | >10 min | [14] |
| 50 RNA copies per reaction | 30 min | [15] | |||
| 4 copies/μL | 40 min | [16] | |||
| Hepatitis B virus | 100 fg/mL | 10–15 min | [17] | ||
| Immunological | SARS-CoV-2 | 370 vp/mL | 15 min | [18] | |
| 0.08 ng/mL | 30 min | [19] | |||
| Influenza A Virus | 7.8 HAU | 30 min | [20] | ||
| Zika Virus | 0.82 pmol/L | 50 min | [21] | ||
| Quantum dots | ELISA | Influenza A Virus | 22 pfu/mL | >35 min | [22] |
| Influenza A Virus (H5N1) | 0.016 HAU | >15 min | [23] | ||
| SARS-CoV-2 | 5 pg/mL | >15 min | [24] | ||
| Immunological | HEV3 | 1.23 fM | 20 min | [25] | |
| SARS-CoV | 0.1 pg/mL | 1 h | [26] | ||
| Synthetic polymer | Immunological | Influenza A Virus (H1N1) | 5 × 103~104 TCID50 | 9 min | [27] |
| Nanofibers | Immunological | SARS-CoV-2 | 0.8 pg/mL | 20 min | [28] |
| Nanoparticles | Drugs | Target Virus | Strategy | Level of Study | Advantages in Antiviral Therapy | Ref. |
|---|---|---|---|---|---|---|
| Cellulose PEG NPs | Zidovudine | HIV | Inhibition of viral replication | In vitro | Improved encapsulation efficiency Targeted delivery with sustained release | [148] |
| Lipid PLGA NPs | Latency -reversing agents | HIV | Inhibition of viral replication | In vitro | Synergistic latency-reversal and low toxicity | [149] |
| PVP/SA PEG NPs | Zidovudine | HIV | Inhibition of viral replication | In vitro | Improved cellular internalization | [150] |
| PLA NPs | Chloroquine | HSV-1 | Blocking viral entry | In vitro | Targeted delivery with sustained release | [151] |
| Lipid nanodisc | IAV | Viral inactivation | In vitro In vivo | 60% reduction of viral infection 40% reduction of death rate in mice | [152] | |
| AuNPs | IAV | Inhibition of viral infection | In vitro | 40% reduction of viral infection | [153] | |
| RSV, VSV HPV, dengue | Viral inactivation | In vivo Ex vivo | 87% inactivation of virus | [154] | ||
| GO-AgNPs | FCoV IBDV | Blocking viral entry | In vitro | 25% inhibition of FCoV infection 23% inhibition of IBDV infection | [155] | |
| AgNPs | TGEV-CoV | Inhibition of viral replication | In vitro | 67.35% reduction of viral replication | [156] | |
| RSV | Inhibition of viral replication | In vitro In vivo | 75% reduction of viral replication | [157] | ||
| SeNPs | Zanamivir | IAV | Inhibition of viral infection | In vitro | Improved infected cell viability up to 73% | [158] |
| ZnO NPs | Oseltamivir | IAV | Viral inactivation | In vitro | Improved infected cell viability up to 90% | [159] |
| SiO2NPs | HIV VSV | Inhibition of viral infection | In vitro | 50% reduction of viral infection | [160] | |
| FE3O4@SiO2 | HSV | Viral inactivation | In vitro | Improved antiviral activity | [161] | |
| Ag2S | Glutathione | PEDV-CoV model | Inhibition of viral replication | In vitro | Reduction of viral titer | [162] |
| CDs | HCoV-229E | Blocking viral entry | In vitro | CD-mediated inhibition of viral entry | [163] | |
| PEDV-CoV Model | Inhibition of viral replication | In vitro | 80% reduction of viral replication | [164] | ||
| PRRSV-CoV Model | Viral Inactivation | In vitro | Reduction of viral replication | [165] | ||
| Zika, dengue | Inhibition of viral infection | In vitro | Improved infected cell viability up to 90% | [166] | ||
| Cellulose nanocrystals | Alphavirus | Inhibition of viral infection | In vitro | 100% inhibition of viral infection | [167,168] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, J.-W.; Ahn, Y.-R.; Park, G.; Kim, H.-O.; Haam, S. Application of Nanomaterials as an Advanced Strategy for the Diagnosis, Prevention, and Treatment of Viral Diseases. Pharmaceutics 2021, 13, 1570. https://doi.org/10.3390/pharmaceutics13101570
Lim J-W, Ahn Y-R, Park G, Kim H-O, Haam S. Application of Nanomaterials as an Advanced Strategy for the Diagnosis, Prevention, and Treatment of Viral Diseases. Pharmaceutics. 2021; 13(10):1570. https://doi.org/10.3390/pharmaceutics13101570
Chicago/Turabian StyleLim, Jong-Woo, Yu-Rim Ahn, Geunseon Park, Hyun-Ouk Kim, and Seungjoo Haam. 2021. "Application of Nanomaterials as an Advanced Strategy for the Diagnosis, Prevention, and Treatment of Viral Diseases" Pharmaceutics 13, no. 10: 1570. https://doi.org/10.3390/pharmaceutics13101570
APA StyleLim, J.-W., Ahn, Y.-R., Park, G., Kim, H.-O., & Haam, S. (2021). Application of Nanomaterials as an Advanced Strategy for the Diagnosis, Prevention, and Treatment of Viral Diseases. Pharmaceutics, 13(10), 1570. https://doi.org/10.3390/pharmaceutics13101570







