Hyaluronan/Poly-L-lysine/Berberine Nanogels for Impaired Wound Healing
Abstract
1. Introduction
2. Materials and Methods
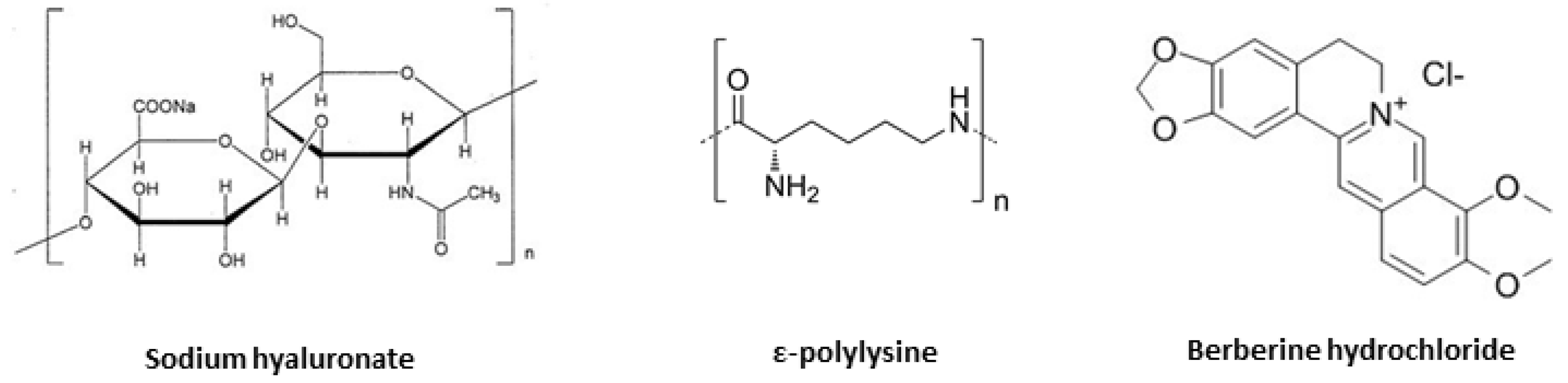
2.1. Materials
2.2. Preliminary Design of Nanogels
2.3. Light Scattering
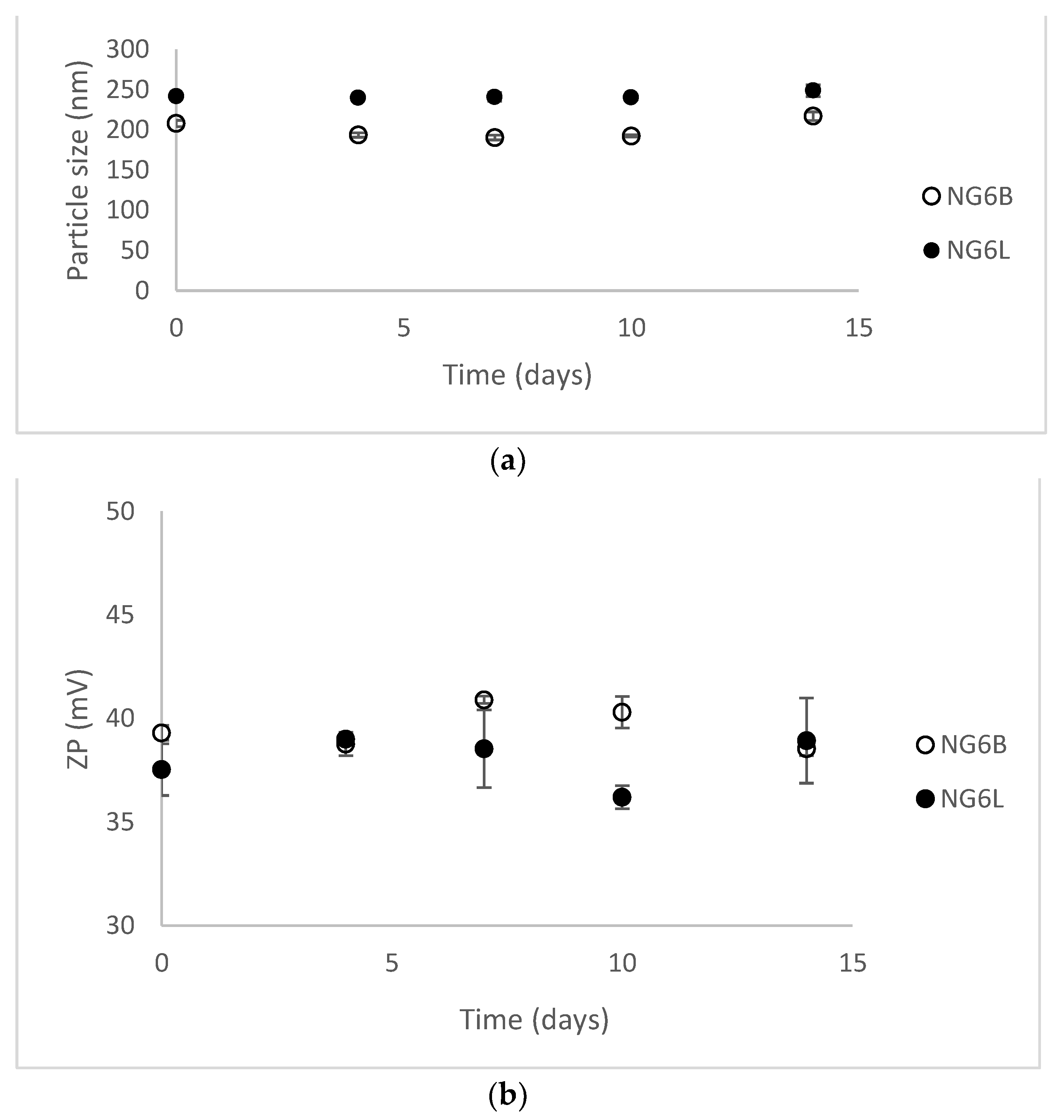
2.4. Stability after Freeze-Drying
2.5. Selected Nanogels Preparation and Berberine Loading
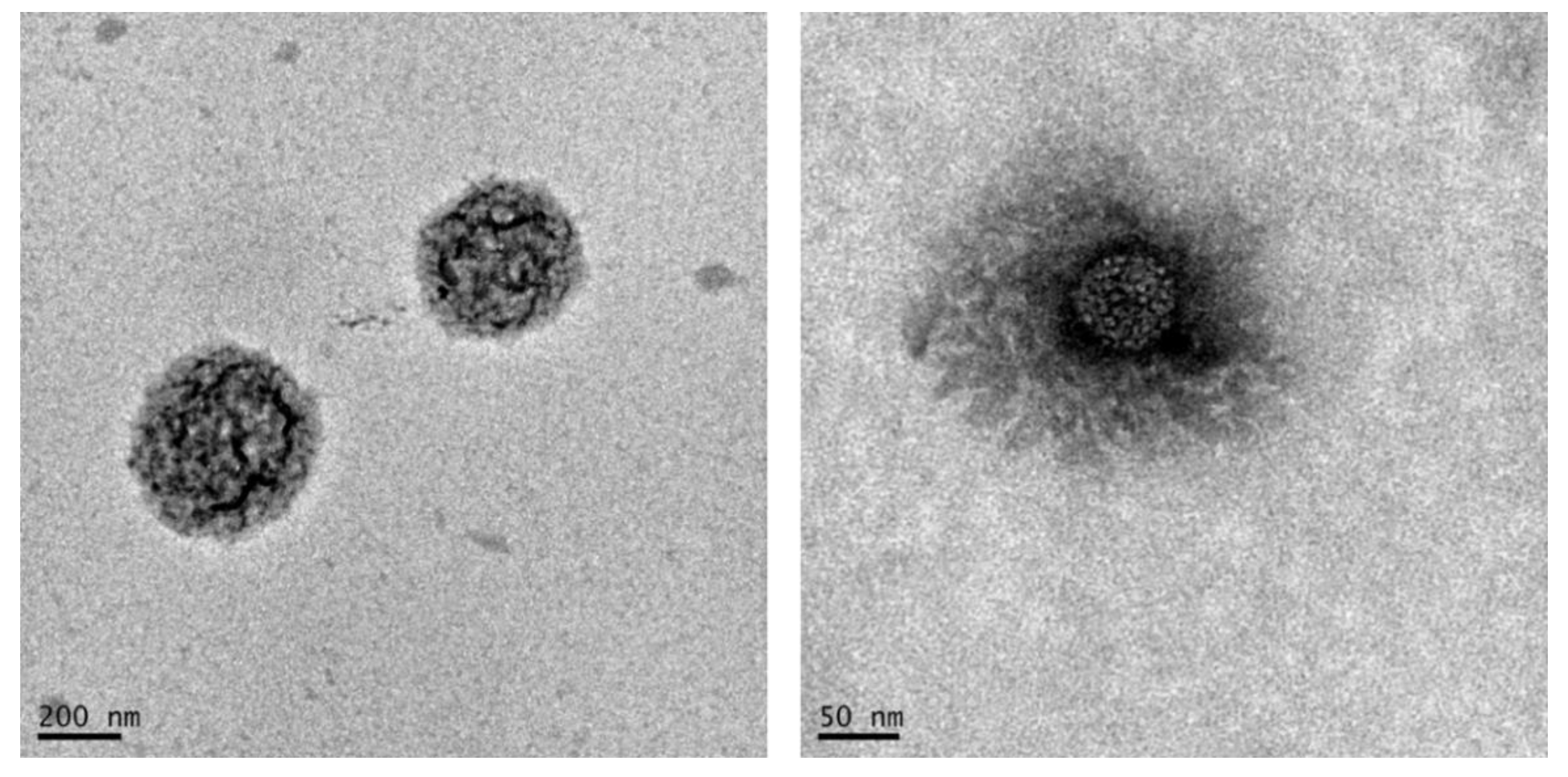
2.6. Nanogels Morphology
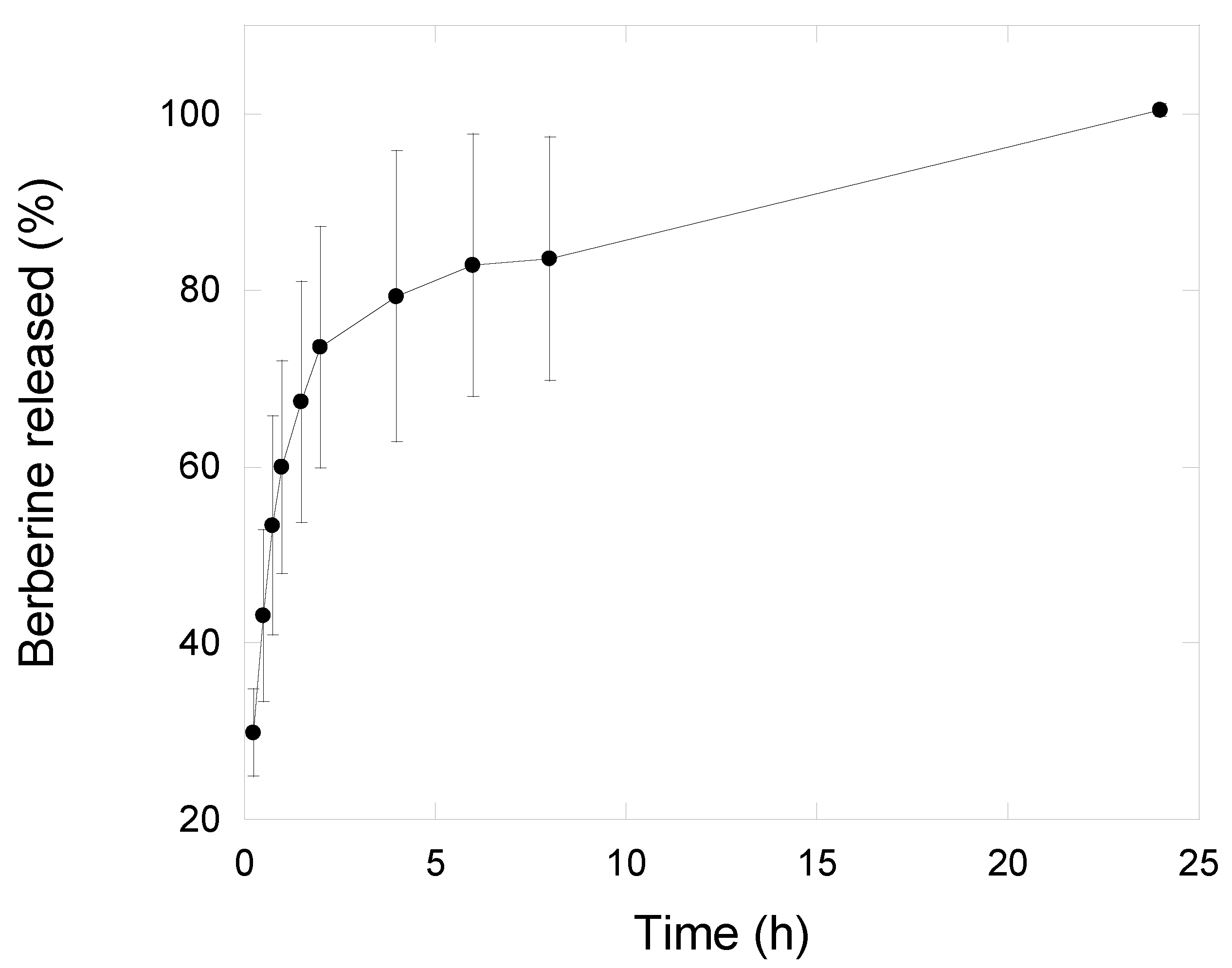
2.7. Berberine Release Studies
2.8. In Vitro Fibroblasts Migration Assay
2.9. Statistical Analysis
3. Results and Discussion
3.1. Nanogels Formulative Studies
3.2. Nanogels Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Greaves, N.S.; Ashcroft, K.J.; Baguneid, M.; Bayat, A. Current understanding of molecular and cellular mechanisms in fibroplasia and angiogenesis during acute wound healing. J. Dermatol. Sci. 2013, 72, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Mihai, M.M.; Preda, M.; Lungu, I.; Gestal, M.C.; Popa, M.I.; Holban, A.M. Nanocoatings for Chronic Wound Repair-Modulation of Microbial Colonization and Biofilm Formation. Int. J. Mol. Sci. 2018, 19, 1179. [Google Scholar] [CrossRef] [PubMed]
- Stephens, P. 1—Dysfunctional wound healing in chronic wounds. In Advanced Wound Repair Therapies; Farrar, D., Ed.; Woodhead Publishing: Sawston, UK, 2011; pp. 3–38. [Google Scholar] [CrossRef]
- Edwards, R.; Harding, K.G. Bacteria and wound healing. Curr. Opin. Infect. Dis. 2004, 17, 91–96. [Google Scholar] [CrossRef]
- Bowler, P.G. Wound pathophysiology, infection and therapeutic options. Ann. Med. 2002, 34, 419–427. [Google Scholar] [CrossRef]
- Elviri, L.; Bianchera, A.; Bergonzi, C.; Bettini, R. Controlled local drug delivery strategies from chitosan hydrogels for wound healing. Expert Opin. Drug Deliv. 2017, 14, 897–908. [Google Scholar] [CrossRef]
- Naseri-Nosar, M.; Ziora, Z.M. Wound dressings from naturally-occurring polymers: A review on homopolysaccharide-based composites. Carbohydr. Polym. 2018, 189, 379–398. [Google Scholar] [CrossRef]
- Sasaki, Y.; Akiyoshi, K. Nanogel engineering for new nanobiomaterials: From chaperoning engineering to biomedical applications. Chem. Rec. 2010, 10, 366–376. [Google Scholar] [CrossRef]
- Zarekar, N.S.; Lingayat, V.J.; Pande, V.V.J.N.; Research, N. Nanogel as a Novel Platform for Smart Drug Delivery System. Nanosci. Nanotechnol. 2017, 4, 25–31. [Google Scholar]
- Raemdonck, K.; Demeester, J.; De Smedt, S. Advanced nanogel engineering for drug delivery. Soft Matter 2009, 5, 707–715. [Google Scholar] [CrossRef]
- Rancan, F.; Asadian-Birjand, M.; Dogan, S.; Graf, C.; Cuellar, L.; Lommatzsch, S.; Blume-Peytavi, U.; Calderón, M.; Vogt, A. Effects of thermoresponsivity and softness on skin penetration and cellular uptake of polyglycerol-based nanogels. J. Control. Release 2016, 228, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Moya-Ortega, M.D.; Alvarez-Lorenzo, C.; Concheiro, A.; Loftsson, T. Cyclodextrin-based nanogels for pharmaceutical and biomedical applications. Int. J. Pharm. 2012, 428, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.-H.; Jones, S.; Forbes, B.; Martin, G.; Brown, M. Hyaluronan: Pharmaceutical Characterization and Drug Delivery. Drug Deliv. 2008, 12, 327–342. [Google Scholar] [CrossRef] [PubMed]
- Voigt, J.; Driver, V.R. Hyaluronic acid derivatives and their healing effect on burns, epithelial surgical wounds, and chronic wounds: A systematic review and meta-analysis of randomized controlled trials. Wound Repair Regen. 2012, 20, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Gariboldi, S.; Palazzo, M.; Zanobbio, L.; Selleri, S.; Sommariva, M.; Sfondrini, L.; Cavicchini, S.; Balsari, A.; Rumio, C. Low molecular weight hyaluronic acid increases the self-defense of skin epithelium by induction of beta-defensin 2 via TLR2 and TLR4. J. Immunol. 2008, 181, 2103–2110. [Google Scholar] [CrossRef] [PubMed]
- Kaya, G.; Tran, C.; Sorg, O.; Hotz, R.; Grand, D.; Carraux, P.; Didierjean, L.; Stamenkovic, I.; Saurat, J.H. Hyaluronate fragments reverse skin atrophy by a CD44-dependent mechanism. PLoS Med. 2006, 3, e493. [Google Scholar] [CrossRef]
- Epand, R.M.; Vogel, H.J. Diversity of antimicrobial peptides and their mechanisms of action. Biochim. Biophys. Acta (BBA)-Biomembr. 1999, 1462, 11–28. [Google Scholar] [CrossRef]
- Zhilei, T.; Shi, Y.; Xing, B.; Hou, Y.; Jia, S. The antimicrobial effects and mechanism of ε-poly-lysine against Staphylococcus aureus. Bioresour. Bioprocess. 2019, 6. [Google Scholar] [CrossRef]
- Yoshida, T.; Nagasawa, T. ε-Poly-l-lysine: Microbial production, biodegradation and application potential. Appl. Microbiol. Biotechnol. 2003, 62, 21–26. [Google Scholar] [CrossRef]
- Shima, S.; Matsuoka, H.; Iwamoto, T.; Sakai, H. Antimicrobial action of epsilon-poly-L-lysine. J. Antibiot. 1984, 37, 1449–1455. [Google Scholar] [CrossRef]
- Shi, C.; He, Y.; Feng, X.; Fu, D. ε-Polylysine and next-generation dendrigraft poly-L-lysine: Chemistry, activity, and applications in biopharmaceuticals. J. Biomater. Sci. Polym. Ed. 2015, 26, 1343–1356. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Pei, H.; Han, Z.; Feng, G.; Li, D. The antimicrobial effects and synergistic antibacterial mechanism of the combination of ε-Polylysine and nisin against Bacillus subtilis. Food Control. 2015, 47, 444–450. [Google Scholar] [CrossRef]
- Ye, R.; Xu, H.; Wan, C.; Peng, S.; Wang, L.; Xu, H.; Aguilar, Z.P.; Xiong, Y.; Zeng, Z.; Wei, H. Antibacterial activity and mechanism of action of ε-poly-l-lysine. Biochem. Biophys. Res. Commun. 2013, 439, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Shih, I.-L.; Shen, M.-H.; Van, Y.-T. Microbial synthesis of poly(ε-lysine) and its various applications. Bioresour. Technol. 2006, 97, 1148–1159. [Google Scholar] [CrossRef]
- Prokopovic, V.Z.; Vikulina, A.S.; Sustr, D.; Shchukina, E.M.; Shchukin, D.G.; Volodkin, D.V. Binding Mechanism of the Model Charged Dye Carboxyfluorescein to Hyaluronan/Polylysine Multilayers. ACS Appl. Mater. Interfaces 2017, 9, 38908–38918. [Google Scholar] [CrossRef]
- Lee, I.C.; Wu, Y.-C.; Cheng, E.-M.; Yang, W.-T. Biomimetic niche for neural stem cell differentiation using poly-L-lysine/hyaluronic acid multilayer films. J. Biomater. Appl. 2014, 29, 1418–1427. [Google Scholar] [CrossRef]
- Niepel, M.S.; Ekambaram, B.K.; Schmelzer, C.E.H.; Groth, T. Polyelectrolyte multilayers of poly (l-lysine) and hyaluronic acid on nanostructured surfaces affect stem cell response. Nanoscale 2019, 11, 2878–2891. [Google Scholar] [CrossRef]
- Dreaden, E.C.; Morton, S.W.; Shopsowitz, K.E.; Choi, J.-H.; Deng, Z.J.; Cho, N.-J.; Hammond, P.T. Bimodal Tumor-Targeting from Microenvironment Responsive Hyaluronan Layer-by-Layer (LbL) Nanoparticles. ACS Nano 2014, 8, 8374–8382. [Google Scholar] [CrossRef]
- Liu, S.; Liu, X.; Ren, Y.; Wang, P.; Pu, Y.; Yang, R.; Wang, X.; Tan, X.; Ye, Z.; Maurizot, V.; et al. Mussel-Inspired Dual-Cross-linking Hyaluronic Acid/ε-Polylysine Hydrogel with Self-Healing and Antibacterial Properties for Wound Healing. ACS Appl. Mater. Interfaces 2020, 12, 27876–27888. [Google Scholar] [CrossRef]
- Simonson, A.W.; Lawanprasert, A.; Goralski, T.D.P.; Keiler, K.C.; Medina, S.H. Bioresponsive peptide-polysaccharide nanogels —A versatile delivery system to augment the utility of bioactive cargo. Nanomed. Nanotechnol. Biol. Med. 2019, 17, 391–400. [Google Scholar] [CrossRef]
- Blass, S.C.; Goost, H.; Tolba, R.H.; Stoffel-Wagner, B.; Kabir, K.; Burger, C.; Stehle, P.; Ellinger, S. Time to wound closure in trauma patients with disorders in wound healing is shortened by supplements containing antioxidant micronutrients and glutamine: A PRCT. Clin. Nutr. 2012, 31, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Roy, S. Redox signals in wound healing. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2008, 1780, 1348–1361. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Gilani, A.-U.-H.; Abdollahi, M.; Daglia, M.; Nabavi, S.F.; Nabavi, S.M. Berberine and neurodegeneration: A review of literature. Pharmacol. Rep. 2015, 67, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Geng, Y.N.; Jiang, J.D.; Kong, W.J. Antioxidant and anti-inflammatory activities of berberine in the treatment of diabetes mellitus. Evid. Based Complementary Altern. Med. 2014, 2014, 289264. [Google Scholar] [CrossRef]
- Neamtu, I.; Rusu, A.G.; Diaconu, A.; Nita, L.E.; Chiriac, A.P. Basic concepts and recent advances in nanogels as carriers for medical applications. Drug Deliv. 2017, 24, 539–557. [Google Scholar] [CrossRef]
- Wen, Y.; Oh, J.K. Recent Strategies to Develop Polysaccharide-Based Nanomaterials for Biomedical Applications. Macromol. Rapid Commun. 2014, 35, 1819–1832. [Google Scholar] [CrossRef]
- Ahmad, N.; Ahmad, F. Green Chemistry: Principle and its Application. In Proceedings of the 2nd International Conference on Advancement in Engineering, Applied Science and Management, ICAEASM 2017, New Delhi, India, 23–24 September 2017. [Google Scholar]
- Pistone, S.; Qoragllu, D.; Smistad, G.; Hiorth, M. Multivariate analysis for the optimization of polysaccharide-based nanoparticles prepared by self-assembly. Colloids Surf. BBiointerfaces 2016, 146, 136–143. [Google Scholar] [CrossRef]
- Kaisersberger-Vincek, M.; Štrancar, J.; Kokol, V. Antibacterial activity of chemically versus enzymatic functionalized wool with ɛ-poly-L-lysine. Text. Res. J. 2017, 87, 1604–1619. [Google Scholar] [CrossRef]
- Zahouani, S.; Chaumont, A.; Senger, B.; Boulmedais, F.; Schaaf, P.; Jierry, L.; Lavalle, P. Stretch-Induced Helical Conformations in Poly(l-lysine)/Hyaluronic Acid Multilayers. ACS Appl. Mater. Interfaces 2016, 8, 14958–14965. [Google Scholar] [CrossRef]
- Stephany Cares, H.; Priscyla Daniely, M.; Nelson, D.; Andrea Monte Alto, C.; Joyce Maria, A.-B. Preparation and Application of Mucoadhesive Nanoparticles Containing Enoxaparin in a Wound Healing Animal Model. Curr. Nanosci. 2014, 10, 779–785. [Google Scholar] [CrossRef]
- Soni, K.S.; Desale, S.S.; Bronich, T.K. Nanogels: An overview of properties, biomedical applications and obstacles to clinical translation. J. Control. Release 2016, 240, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Franken, L.E.; Boekema, E.J.; Stuart, M.C.A. Transmission Electron Microscopy as a Tool for the Characterization of Soft Materials: Application and Interpretation. Adv. Sci. 2017, 4, 1600476. [Google Scholar] [CrossRef] [PubMed]
- Modi, S.; Anderson, B.D. Determination of Drug Release Kinetics from Nanoparticles: Overcoming Pitfalls of the Dynamic Dialysis Method. Mol. Pharm. 2013, 10, 3076–3089. [Google Scholar] [CrossRef] [PubMed]
- Mavuso, S.; Marimuthu, T.; Choonara, Y.E.; Kumar, P.; du Toit, L.C.; Pillay, V. A review of polymeric colloidal nanogels in transdermal drug delivery. Curr. Pharm. Des. 2015, 21, 2801–2813. [Google Scholar] [CrossRef]
- El-Feky, G.; El-Banna, S.; El-Bahy, G.S.; Abdelrazek, E.; Kamal, M.K. Alginate coated chitosan nanogel for the controlled topical delivery of Silver sulfadiazine. Carbohydr. Polym. 2017, 177. [Google Scholar] [CrossRef]
- Luo, A.; Fan, Y. Antioxidant activities of berberine hydrochloride. J. Med. Plants Res. 2011, 5, 3702–3707. [Google Scholar]
- Zhao, L.; Zhang, C. Berberine Inhibits MDA-MB-231 Cells by Attenuating Their Inflammatory Responses. Biomed Res. Int. 2020, 2020, 3617514. [Google Scholar] [CrossRef]

| Code | ε-PL Solution | HA Solution | Molar Ratio nε-PL/nHA | Post-Centrifugation | |||||
|---|---|---|---|---|---|---|---|---|---|
| V (mL) | Conc (mg/mL) | V (mL) | Conc (mg/mL) | MW (kDa) | Size (nm) | PDI | ZP (mV) | ||
| A | 1 | 0.5 | 1 | 2 | 200 | 1.5 | 200.4 ± 4.4 | 0.161 ± 0.017 | −38.8 ± 0.1 |
| B | 2 | 0.5 | 1 | 2 | 200 | 3 | 505.7 ± 14.7 | 0.673 ± 0.056 | −0.05 ± 0.14 |
| C | 2 | 0.5 | 1 | 5 | 200 | 1.2 | 244.7 ± 4.0 | 0.089 ± 0.024 | −38.8 ± 0.5 |
| D | 2.5 | 0.5 | 1 | 5 | 200 | 1.5 | 254.7 ± 27.6 | 0.447 ± 0.060 | −51.1 ± 2.6 |
| E | 1 | 0.5 | 1 | 2 | 700 | 1.5 | 206.7 ± 8.5 | 0.207 ± 0.022 | −40.9 ± 2.5 |
| F | 2 | 0.5 | 1 | 5 | 700 | 1.2 | 279.7 ± 9.4 | 0.256 ± 0.005 | −55.4 ± 2.77 |
| G | 2.5 | 0.5 | 1 | 5 | 700 | 1.5 | 243.6 ± 16.1 | 0.426 ± 0.046 | 1.18 ± 0.06 |
| H | 1 | 0.5 | 1 | 2 | 1200 | 1.5 | 277.5 ± 5.7 | 0.273 ± 0.18 | −44.2 ± 2.21 |
| I | 2 | 0.5 | 1 | 5 | 1200 | 1.2 | Macrogelation | ||
| L | 2.5 | 0.5 | 1 | 5 | 1200 | 1.5 | Macrogelation | ||
| Sample | Particle Size (nm) | PDI | ZP (mV) | Yield (%) | EE (%) |
|---|---|---|---|---|---|
| NG6B‡ | 207.9 ± 7.3 | 0.232 ± 0.019 | +40.9 ± 4.6 | ≈40 | - |
| NG6L§ | 245.2 ± 11.2 | 0.208 ± 0.050 | +38.2 ± 1.6 | ≈50 | ≈10 |
| NG7B# | 195.5 ± 9.9 | 0.169 ± 0.076 | +36.0 ± 5.1 | ≈40 | - |
| NG7L * | 222.8 ± 19.4 | 0.136 ± 0.060 | +37.7 ± 0.8 | ≈30 | ≈10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amato, G.; Grimaudo, M.A.; Alvarez-Lorenzo, C.; Concheiro, A.; Carbone, C.; Bonaccorso, A.; Puglisi, G.; Musumeci, T. Hyaluronan/Poly-L-lysine/Berberine Nanogels for Impaired Wound Healing. Pharmaceutics 2021, 13, 34. https://doi.org/10.3390/pharmaceutics13010034
Amato G, Grimaudo MA, Alvarez-Lorenzo C, Concheiro A, Carbone C, Bonaccorso A, Puglisi G, Musumeci T. Hyaluronan/Poly-L-lysine/Berberine Nanogels for Impaired Wound Healing. Pharmaceutics. 2021; 13(1):34. https://doi.org/10.3390/pharmaceutics13010034
Chicago/Turabian StyleAmato, Giovanni, Maria Aurora Grimaudo, Carmen Alvarez-Lorenzo, Angel Concheiro, Claudia Carbone, Angela Bonaccorso, Giovanni Puglisi, and Teresa Musumeci. 2021. "Hyaluronan/Poly-L-lysine/Berberine Nanogels for Impaired Wound Healing" Pharmaceutics 13, no. 1: 34. https://doi.org/10.3390/pharmaceutics13010034
APA StyleAmato, G., Grimaudo, M. A., Alvarez-Lorenzo, C., Concheiro, A., Carbone, C., Bonaccorso, A., Puglisi, G., & Musumeci, T. (2021). Hyaluronan/Poly-L-lysine/Berberine Nanogels for Impaired Wound Healing. Pharmaceutics, 13(1), 34. https://doi.org/10.3390/pharmaceutics13010034









