Stability and Safety Traits of Novel Cyclosporine A and Tacrolimus Ophthalmic Galenic Formulations Involved in Vernal Keratoconjunctivitis Treatment by a High-Resolution Mass Spectrometry Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Standard Solutions
2.3. Cyclosporine A and Tacrolimus Ophthalmic Formulations
2.3.1. Cyclosporine A-based Ophthalmic Formulations (1%)
- Classical formulation (SAND): Sandimmun® (concentrated injectable solution, 50 mg/mL) was diluted 2:8 vol/vol in commercial artificial drops Lacrimart® and homogenized under agitation to obtain a clear lipid solution as the most common galenic protocol.
- Novel ethanol-free formulation (CSA): cyclosporine A 0.1 g, polyvinylpyrrolidone 0.2 g Cremophor RH-40 1 g, 10 mL injectable-grade water were used as ingredients.
2.3.2. Tacrolimus-based Ophthalmic Formulations (0.1%)
- Classical formulation (PROG): Prograf® (5 mg/mL) was diluted 2:8 vol/vol in in commercial artificial drops Lacrimart® and homogenized under agitation to obtain a clear limpid solution as the most common galenic protocol.
- Novel ethanol-free formulation (TAC): Tacrolimus 0.01 g, polyvinylpyrrolidone 0.2 g Cremophor RH-40 1 g, 10 mL injectable-grade water were used as ingredients.
2.4. Stability of Cyclosporine A and Tacrolimus Ophthalmic Formulations
- -
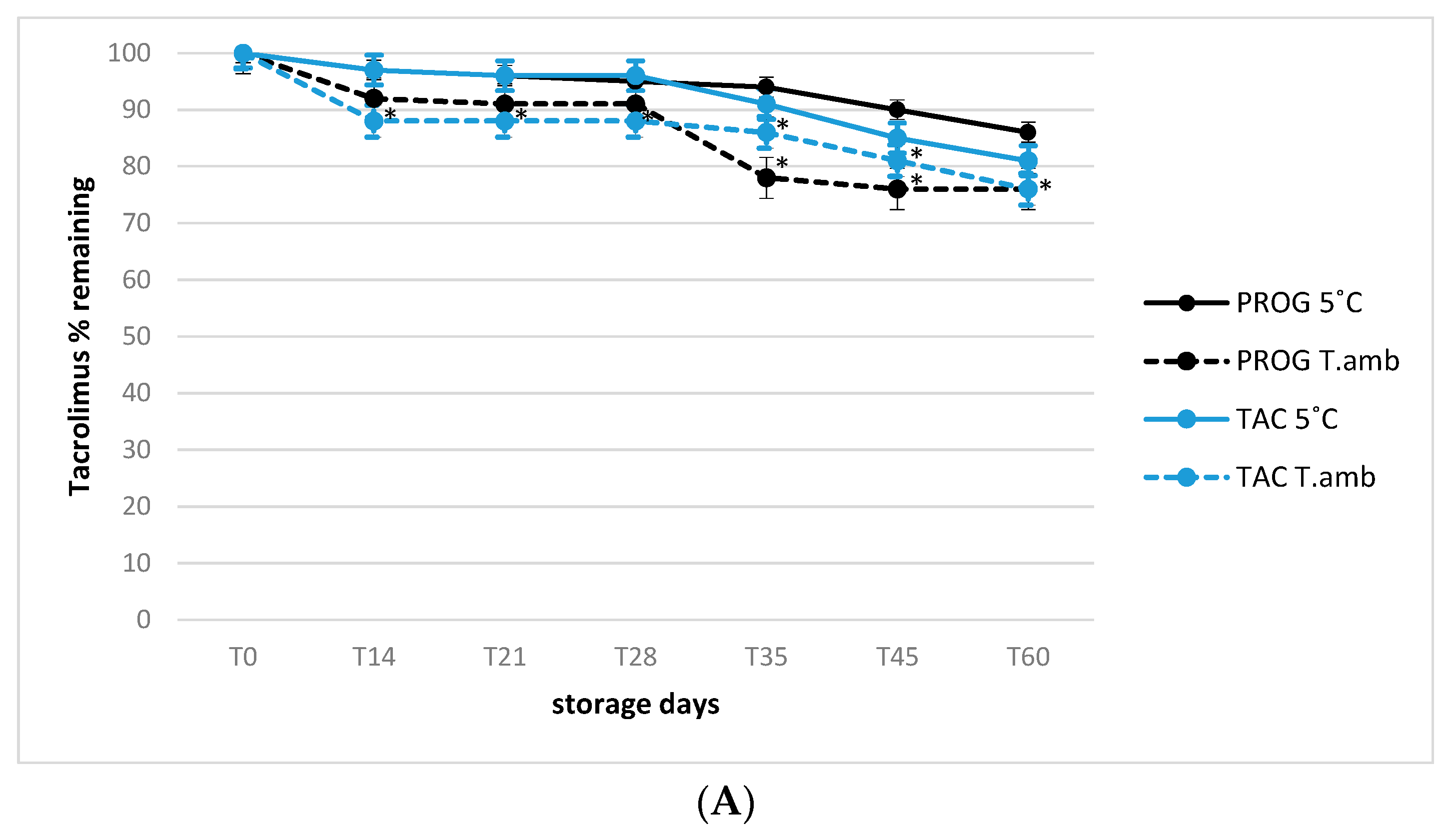
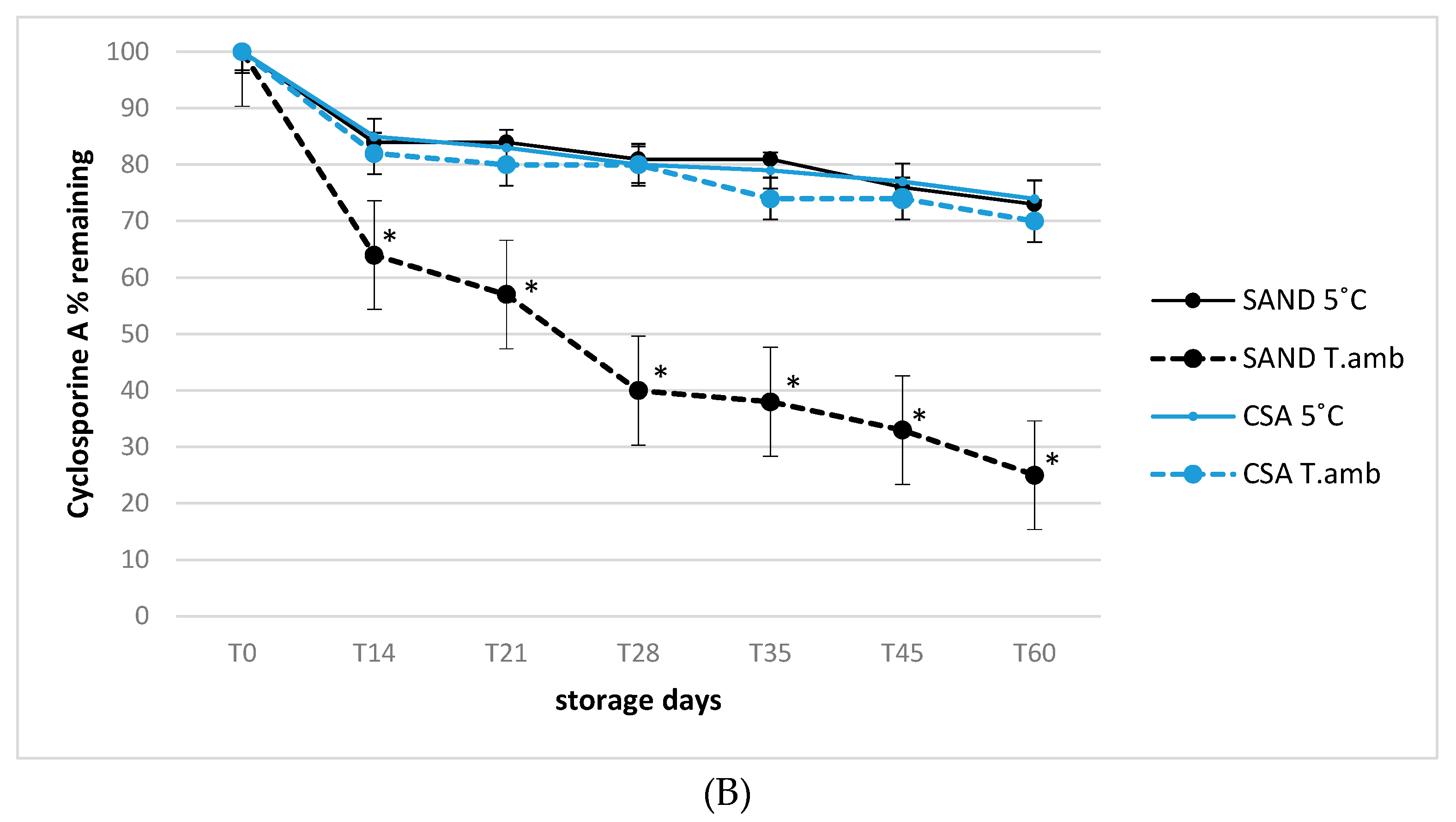
- Real use of simulated conditions in opened products: all formulations underwent analysis, testing two different temperatures (25 °C as room temperature and 5 °C ± 2 °C as refrigerated) during 90 days of storage in which all bottles were regularly gently shaken and opened (4 s each time, three time per day) in order to simulate the real use conditions. As partially investigated, the role of temperature in maintaining active substances stability can play a central role for quality of preparations.
- -
- Shelf-life investigation on unopened products: all formulations underwent active compound analysis during the 90-day testing. The unopened bottles were assessed to verify the decay of cyclosporine A and tacrolimus to assess the possibility to prepare in advance the formulations leading to several advantages for galenic pharmacies in term of stock feasibility.
- -
- Stressed temperature conditions during delivery: all formulations underwent active compounds analysis, simulating an unfavorable temperature condition during transport (simulating courier time delivery), since most of the therapies are also administrated during summer period. 40 °C was selected as the stress parameter compared with the refrigerated one during 4 days by using unopened bottles.
2.5. Sample Processing
2.6. HPLC-HRMS Analysis
2.7. Method Validation
2.8. Microbiological Assay
2.9. Data Analysis
3. Results
3.1. Tacrolimus and Cyclosporine A Quantification in Different Galenic Preparations
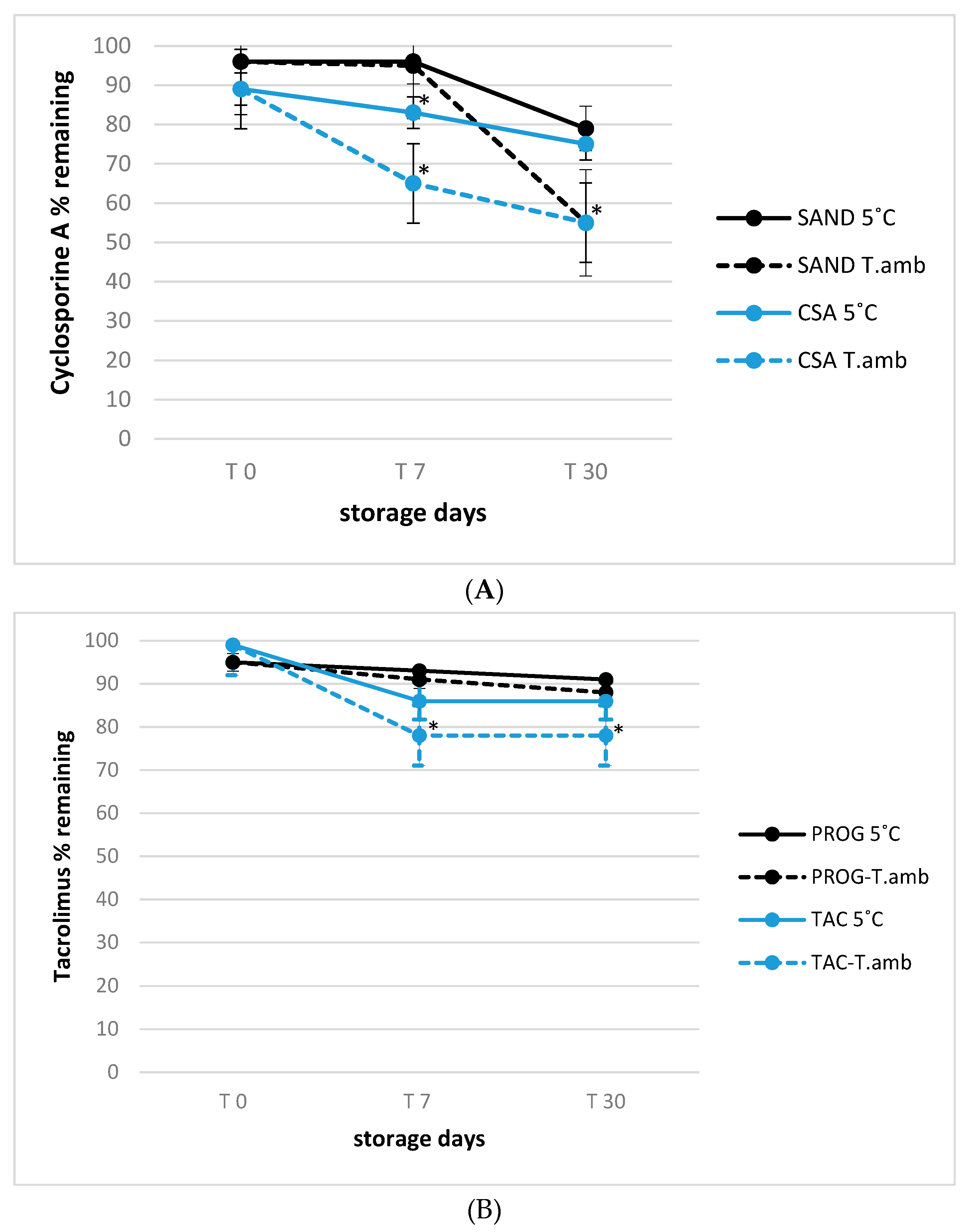
3.2. Stability of Tacrolimus and Cyclosporine A in Eye Drop Formulations under Usage-simulated Conditions
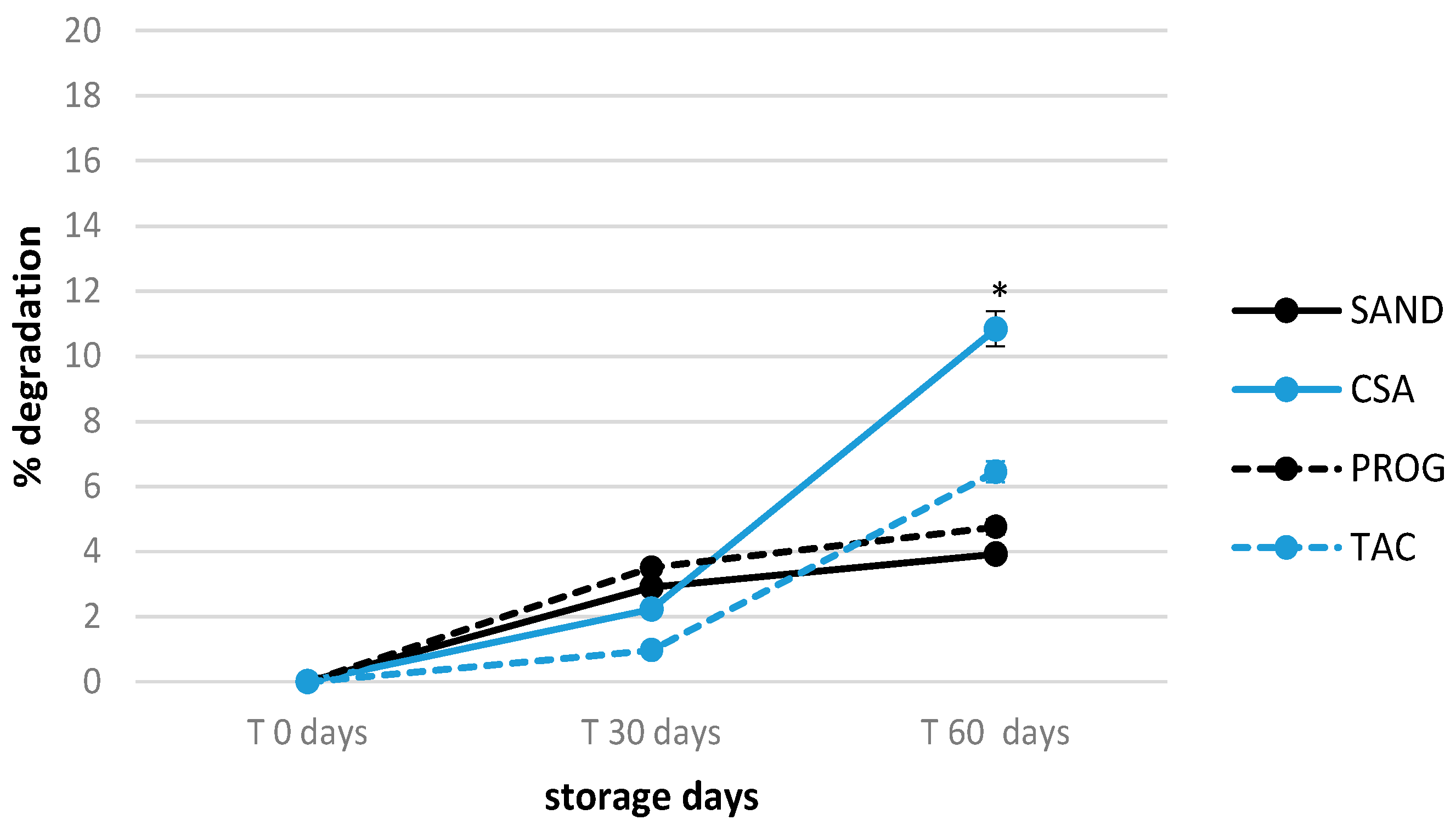
3.3. Shelf-life of Tacrolimus and Cyclosporine A Formulations in Unopened Products
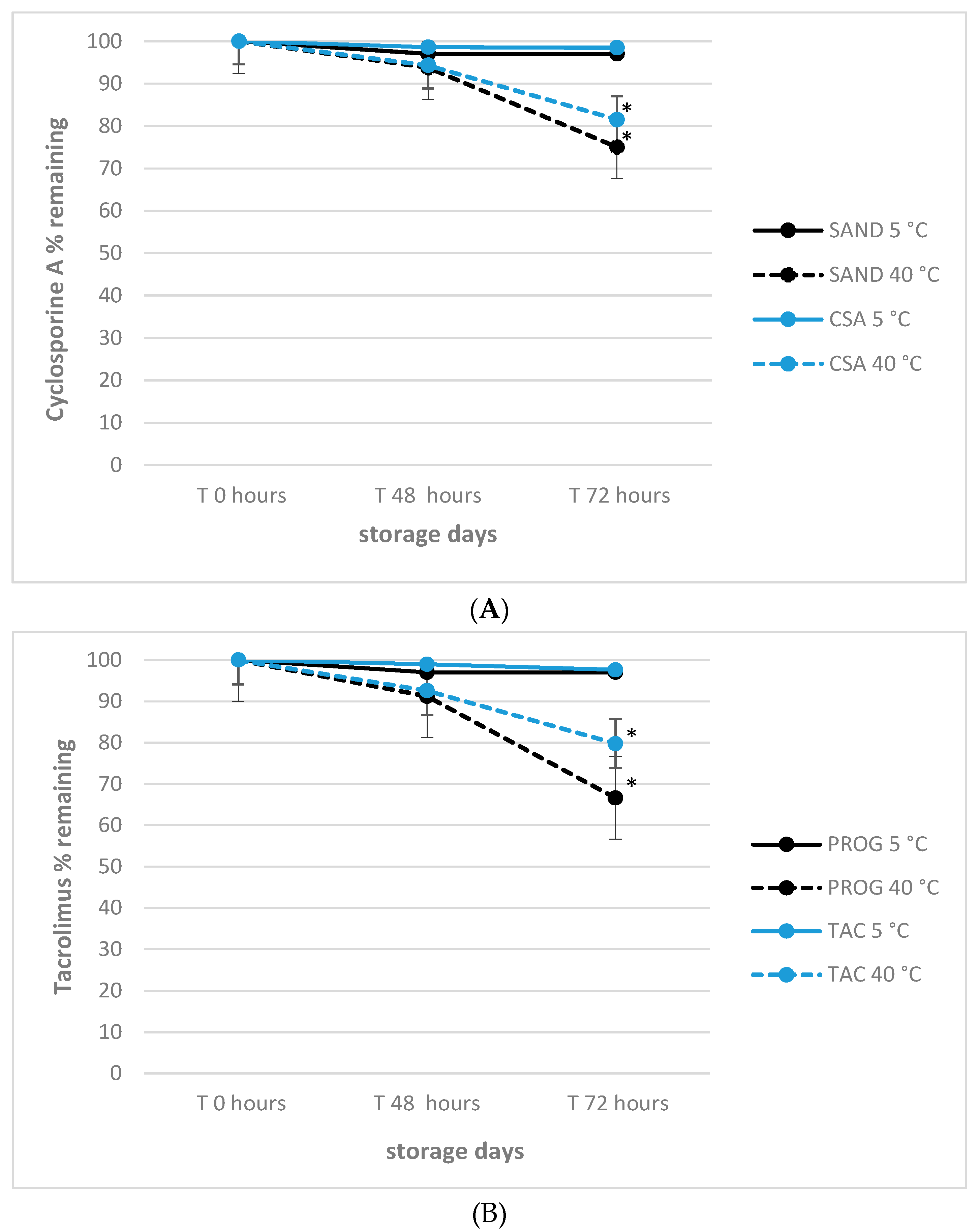
3.4. Effect of Delivery-simulated Conditions on Tacrolimus and Cyclosporine A
3.5. Microbiological Stability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Leonardi, A.; Silva, D.; Perez Formigo, D.; Bozkurt, B.; Sharma, V.; Allegri, P.; Rondon, C.; Calder, V.; Ryan, D.; Kowalski, M.L.; et al. Management of ocular allergy. Allergy 2019, 74, 1611–1630. [Google Scholar] [CrossRef] [PubMed]
- Singhal, D.; Sahay, P.; Maharana, P.K.; Raj, N.; Sharma, N.; Titiyal, J.S. Vernal Keratoconjunctivitis. Surv. Ophthalmol. 2019, 64, 289–311. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, P.; Prokopich, C.L.; Hynes, A.; Kim, H. A contemporary look at allergic conjunctivitis. Allergy Asthma Clin. Immunol. 2020, 16, 5. [Google Scholar] [CrossRef] [PubMed]
- Zicari, A.M.; Capata, G.; Nebbioso, M.; De Castro, G.; Midulla, F.; Leonardi, L.; Loffredo, L.; Spalice, A.; Perri, L.; Duse, M. Vernal Keratoconjunctivitis: An update focused on clinical grading system. Ital. J. Pediatr. 2019, 45, 64. [Google Scholar] [CrossRef] [PubMed]
- Pucci, N.; Novembre, E.; Cianferoni, A.; Lombardi, E.; Bernardini, R.; Caputo, R.; Campa, L.; Vierucci, A. Efficacy and safety of cyclosporine eyedrops in vernal keratoconjunctivitis. Ann. Allergy Asthma Immunol. 2002, 89, 298–303. [Google Scholar] [CrossRef]
- Spadavecchia, L.; Fanelli, P.; Tesse, R.; Brunetti, L.; Cardinale, F.; Bellizzi, M.; Rizzo, G.; Procoli, U.; Bellizzi, G.; Armenio, L. Efficacy of 1.25 and 1% topical cyclosporine in the treatment of severe vernal keratoconjunctivitis in childhood. Pediatr. Allergy Immunol. 2006, 17, 527–532. [Google Scholar] [CrossRef]
- Bonini, S.; Sacchetti, M.; Mantelli, F.; Lambiase, A. Clinical grading of vernal keratoconjunctivitis. Curr. Opin. Allergy Clin. Immunol. 2007, 7, 436–441. [Google Scholar] [CrossRef]
- Keklikci, U.; Dursun, B.; Cingu, A.K. Topical cyclosporine a 0.05% eyedrops in the treatment of vernal keratoconjunctivitis—Randomized placebo-controlled trial. Adv. Clin. Exp. Med. 2011, 23, 455–461. [Google Scholar] [CrossRef]
- Gokhale, N.S. Systematic approach to managing vernal keratoconjunctivitis in clinical practice: Severity grading system and a treatment algorithm. Indian J. Ophthalmol. 2016, 64, 145–148. [Google Scholar] [CrossRef]
- Leonardi, A.; Lazzarini, D.; La Gloria, V.A.; Scalora, T.; Fregona, I. Corneal staining patterns in vernal keratoconjunctivitis: The new VKC-CLEK scoring scale. Br. J. Ophthalmol. 2018, 102, 1448–1453. [Google Scholar] [CrossRef]
- Esposito, S.; Fior, G.; Mori, A.; Osnaghi, S.; Ghiglioni, D. An Update on the Therapeutic Approach to Vernal Keratoconjunctivitis. Paediatr. Drugs 2016, 18, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Piliego, F.; Castegnaro, A. Allergic conjunctivitis: A cross sectional study. Clin. Exp. Allergy 2015, 45, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Castegnaro, A.; Valerio, A.L.; Lazzarini, D. Epidemiology of allergic conjunctivitis: Clinical appearance and treatment patterns in a population-based study. Curr. Opin. Allergy Clin. Immunol. 2015, 15, 482–488. [Google Scholar] [CrossRef]
- Meyer, E.; Kraus, E.; Zonis, S. Efficacy of antiprostaglandin therapy in vernal conjunctivitis. Br. J. Ophthalmol. 1987, 71, 497–499. [Google Scholar] [CrossRef]
- Senthil, S.; Thakur, M.; Rao, H.L.; Mohamed, A.; Jonnadula, G.B.; Sangwan, V.; Garudadri, C.S. Steroid-induced glaucoma and blindness in vernal keratoconjunctivitis. Br. J. Ophthalmol. 2020, 104, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Lallemand, F.; Schmitt, M.; Bourges, J.L.; Gurny, R.; Benita, S.; Garrigue, J.S. Cyclosporine A delivery to the eye: A comprehensive review of academic and industrial efforts. Eur. J. Pharm. Biopharm. 2017, 117, 14–28. [Google Scholar] [CrossRef]
- Vichyanond, P.; Kosrirukvongs, P. Use of cyclosporine A and tacrolimus in treatment of vernal keratoconjunctivitis. Curr. Allergy Asthma Rep. 2013, 13, 308–314. [Google Scholar] [CrossRef]
- Yücel, O.E.; Ulus, N.D. Efficacy and safety of topical cyclosporine A 0.05% in vernal keratoconjunctivitis. Singap. Med. J. 2016, 57, 507–510. [Google Scholar]
- Leonardi, A.; Doan, S.; Amrane, M.; Ismail, D.; Montero, J.; Németh, J.; Aragona, P.; Bremond-Gignac, D.; VEKTIS Study Group. A Randomized, Controlled Trial of Cyclosporine A Cationic Emulsion in Pediatric Vernal Keratoconjunctivitis: The VEKTIS Study. Ophthalmology 2019, 126, 671–681. [Google Scholar] [CrossRef]
- Bremond-Gignac, D.; Doan, S.; Amrane, M.; Ismail, D.; Montero, J.; Németh, J.; Aragona, P.; Leonardi, A.; VEKTIS Study Group. 12-Month Results of Cyclosporine A Cationic Emulsion in a Randomized, Study in Patients With Pediatric Vernal Keratoconjunctivitis. Am. J. Ophthalmol. 2019, 212, 116–126. [Google Scholar] [CrossRef]
- Fiscella, R.G.; Le, H.; Lam, T.T.; Labib, S. Stability of cyclosporine 1% in artificial tears. J. Ocul. Pharm. Ther. 1996, 12, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Chennell, P.; Delaborde, L.; Wasiak, M.; Jouannet, M.; Feschet-Chassot, E.; Chiambaretta, F.; Sautou, V. Stability of an ophthalmic micellar formulation of cyclosporine A in unopened multidose eyedroppers and in simulated use conditions. Eur. J. Pharm. Sci. 2017, 100, 230–237. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. ICH Topic Q2(R1) Validation of Analytical Procedures: Text and Methodology; European Medicines Agency: London, UK, 2006; pp. 1–15. [Google Scholar]
- European Compliance Academy. International Conference on Harmonization of Technical Requirements for the Registration of Pharmaceuticals for Human Use (ICH), Guideline Q2(R1)-Validation of Analytical Procedures: Text and Methodology; European Compliance Academy: Heidelberg, Germany, 2005; p. 1. [Google Scholar]
- Ezquer-Garin, C.; Ferriols-Lisart, R.; Alós-Almiñana, M. Stability of tacrolimus ophthalmic solution. Am. J. Health Syst. Pharm. 2017, 74, 1002–1006. [Google Scholar] [CrossRef] [PubMed]
- Tamrat, L.; Gelaw, Y.; Beyene, G.; Gize, A. Microbial Contamination and Antimicrobial Resistance in Use of Ophthalmic Solutions at the Department of Ophthalmology, Jimma University Specialized Hospital, Southwest Ethiopia. Can. J. Infect. Dis. Med. Microbiol. 2019, 2019, 5372530. [Google Scholar] [CrossRef] [PubMed]
- Tsegaw, A.; Tsegaw, A.; Tefera Abula, Y.A. Bacterial contamination of multi-dose eye drops at ophthalmology department, university of Gondar, northwest Ethiopia. Middle East Afr. J. Ophthalmol. 2017, 24, 81. [Google Scholar] [CrossRef]
- Danny, H.; Jokl, K.; Wormser, G.P.; Nichols, N.S.; Montecalvo, M.A.; Karmen, C.L. Bacterial contamination of ophthalmic solutions used in an extended care facility. Br. J. Ophthalmol. 2007, 91, 1308–1310. [Google Scholar]
- Rushood, A.A.; Azmat, S.; Shariq, M.; Khamis, A.; Lakho, K.A.; Jadoon, M.Z.; Sial, N.; Rushood, A.A.; Kamil, E.A. Ocular disorders among schoolchildren in Khartoum State, Sudan. East Mediterr. Health J. 2013, 19, 282–288. [Google Scholar] [CrossRef]
- Leonardi, A.; Busca, F.; Motterle, L.; Cavarzeran, F.; Fregona, I.A. Case series of 406 vernal keratoconjunctivitis patients: A demographic and epidemiological study. Acta Ophthalmol. Scand. 2006, 84, 406–410. [Google Scholar] [CrossRef]
- Tesse, R.; Spadavecchia, L.; Fanelli, P.; Paglialunga, C.; Capozza, M. New insights into childhood Vernal keratoconjunctivitis-associated factors. Pediatric Allergy Immunol. 2012, 23, 682–685. [Google Scholar] [CrossRef]
- Cameron, J.A. Shield ulcers and plaques of the cornea in vernal keratoconjunctivitis. Ophthalmology 1995, 102, 985–993. [Google Scholar] [CrossRef]
- Labcharoenwongs, P.; Jirapongsanananuruk, O.; Visitsunthorn, N.; Kosrirukvongs, P.; Saengin, P. A double-masked comparison of 0.1% tacrolimus ointment and 2% cyclosporine eye drops in the treatment of vernal keratocojunctivitis in children. Asian Pac. J. Allergy Immunol. 2012, 30, 177–184. [Google Scholar] [PubMed]
- Ebihara, N.; Ohashi, Y.; Uchio, E.; Okamoto, S.; Kumagai, N.; Shoji, J.; Takamura, E.; Nakagawa, Y.; Nanba, K.; Fukushima, A.; et al. A large prospective observational study of novel cyclosporine 0.1% aqueous ophthalmic solution in the treatment of severe allergic conjunctivitis. J. Ocul. Pharm. Ther. 2009, 25, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Bandyopadhyay, S.; Kumar Bandyopadhyay, S. Efficacy, Safety and Steroid-sparing Effect of Topical Cyclosporine A 0.05% for Vernal Keratoconjunctivitis in Indian Children. J. Ophthalmic Vis. Res. 2019, 14, 412–418. [Google Scholar] [CrossRef] [PubMed]

| Formulation | Composition/Quantity | Description/Function | |
|---|---|---|---|
| Cyclosporine A (1%) | |||
| Cyclosporine A ethanol free formulation (CSA) |
| 100 mg | Active substance |
| 1000 mg | Emulsifier | |
| 200 mg | Polymer/Co-Emulsifier | |
| to reach final volume | ||
| Cyclosporine A —classical galenic (SAND) |
| 50 mg/mL | Active substance— injectable solution |
| 100 mg | ||
| 556 mg | Solvent | |
| 1444 mg | Emulsifier | |
| 8 mL: | Artificial tear | |
| Preservative | ||
| Thickener | ||
| Osmotic agent | ||
| Chelating agent | ||
| to reach final volume | ||
| Tacrolimus (0.1%) | |||
| Tacrolimus ethanol free formulation (TAC) |
| 10 mg | Active substance |
| 1000 mg | Emulsifier | |
| 200 mg | Polymer/Co-Emulsifier | |
| to reach final volume | ||
| Tacrolimus— classical galenic (PROG) |
| 5 mg/mL | Active substance— injectable solution |
| 10 mg | ||
| 1620 mg | Solvent | |
| 380 mg | Emulsifier | |
| 8 mL: | Artificial tear | |
| Preservative | ||
| Thickener | ||
| Osmotic agent | ||
| Chelating agent | ||
| to reach final volume | ||
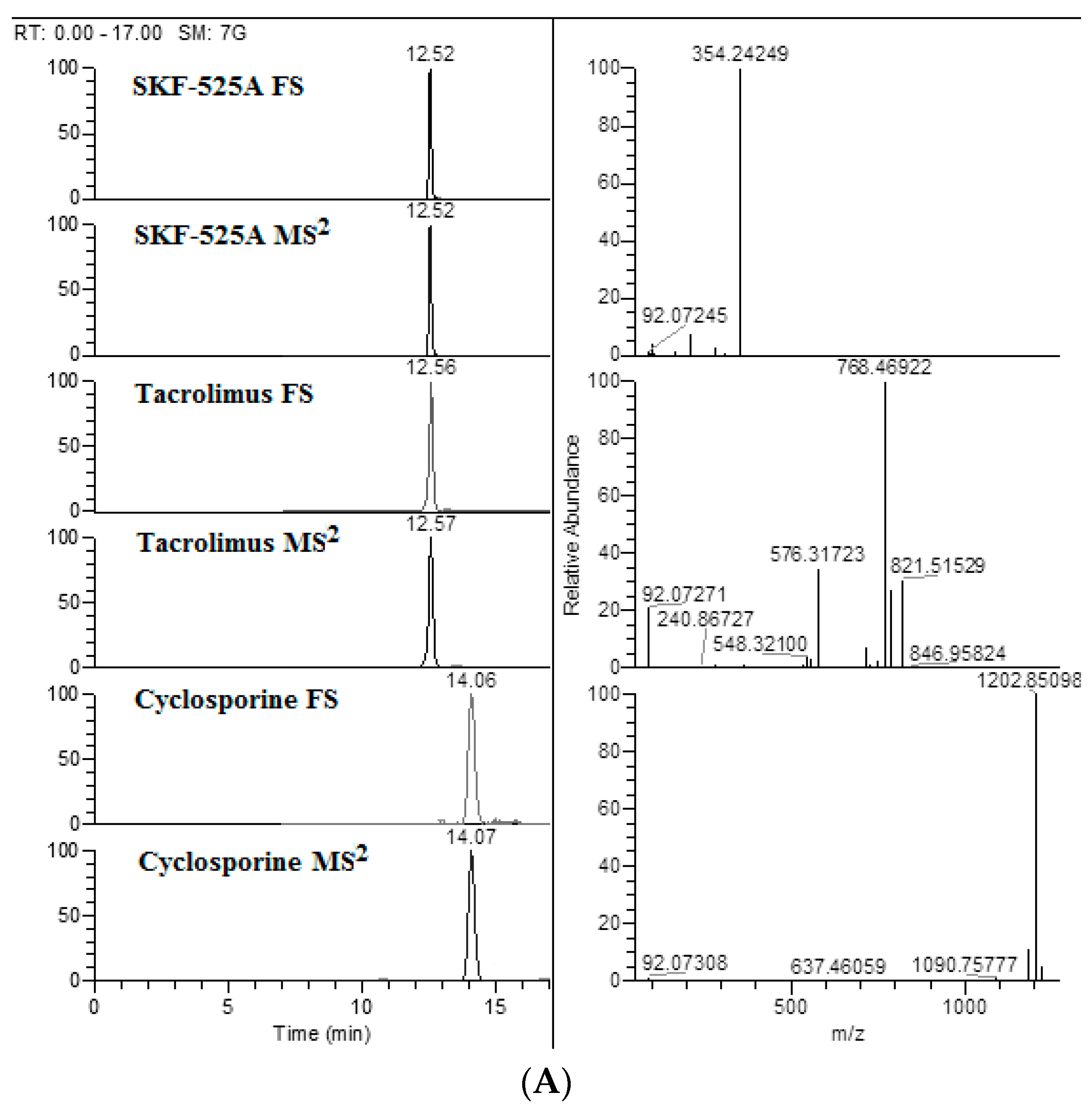
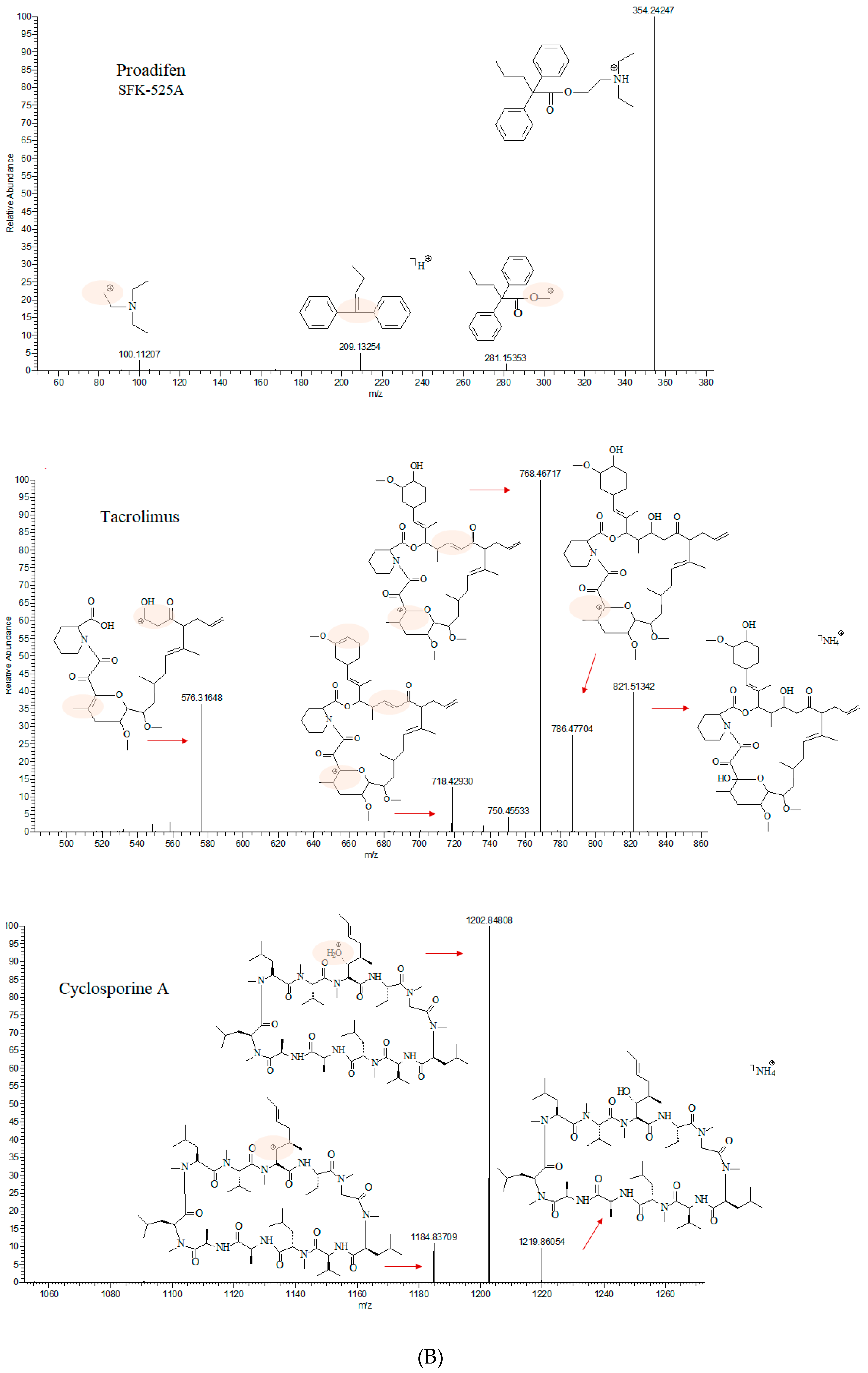
| Compound | Formula | Parent Exact Mass (m/z) | Main Fragment Mass (m/z) | Ionization Polarity |
|---|---|---|---|---|
| Tacrolimus | C44H69NO12 | 821.51580 * | 768.46922 | (+) |
| Cyclosporine A | C62H111N11O12 | 1219.87519 * | 1202.85098 | (+) |
| Proadifen (SFK-525A) | C23H31NO2 | 354.24276 | 209.13289 | (+) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghiglioni, D.G.; Martino, P.A.; Bruschi, G.; Vitali, D.; Osnaghi, S.; Corti, M.G.; Beretta, G. Stability and Safety Traits of Novel Cyclosporine A and Tacrolimus Ophthalmic Galenic Formulations Involved in Vernal Keratoconjunctivitis Treatment by a High-Resolution Mass Spectrometry Approach. Pharmaceutics 2020, 12, 378. https://doi.org/10.3390/pharmaceutics12040378
Ghiglioni DG, Martino PA, Bruschi G, Vitali D, Osnaghi S, Corti MG, Beretta G. Stability and Safety Traits of Novel Cyclosporine A and Tacrolimus Ophthalmic Galenic Formulations Involved in Vernal Keratoconjunctivitis Treatment by a High-Resolution Mass Spectrometry Approach. Pharmaceutics. 2020; 12(4):378. https://doi.org/10.3390/pharmaceutics12040378
Chicago/Turabian StyleGhiglioni, Daniele Giovanni, Piera Anna Martino, Gaia Bruschi, Davide Vitali, Silvia Osnaghi, Maria Grazia Corti, and Giangiacomo Beretta. 2020. "Stability and Safety Traits of Novel Cyclosporine A and Tacrolimus Ophthalmic Galenic Formulations Involved in Vernal Keratoconjunctivitis Treatment by a High-Resolution Mass Spectrometry Approach" Pharmaceutics 12, no. 4: 378. https://doi.org/10.3390/pharmaceutics12040378
APA StyleGhiglioni, D. G., Martino, P. A., Bruschi, G., Vitali, D., Osnaghi, S., Corti, M. G., & Beretta, G. (2020). Stability and Safety Traits of Novel Cyclosporine A and Tacrolimus Ophthalmic Galenic Formulations Involved in Vernal Keratoconjunctivitis Treatment by a High-Resolution Mass Spectrometry Approach. Pharmaceutics, 12(4), 378. https://doi.org/10.3390/pharmaceutics12040378





