Kinetic Cytokine Secretion Profile of LPS-Induced Inflammation in the Human Skin Organ Culture
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Skin Organ Cultures
2.2. Determination of Epidermal and Organ Culture Viability
2.3. Cytokine Quantification
2.4. Statistical Analysis
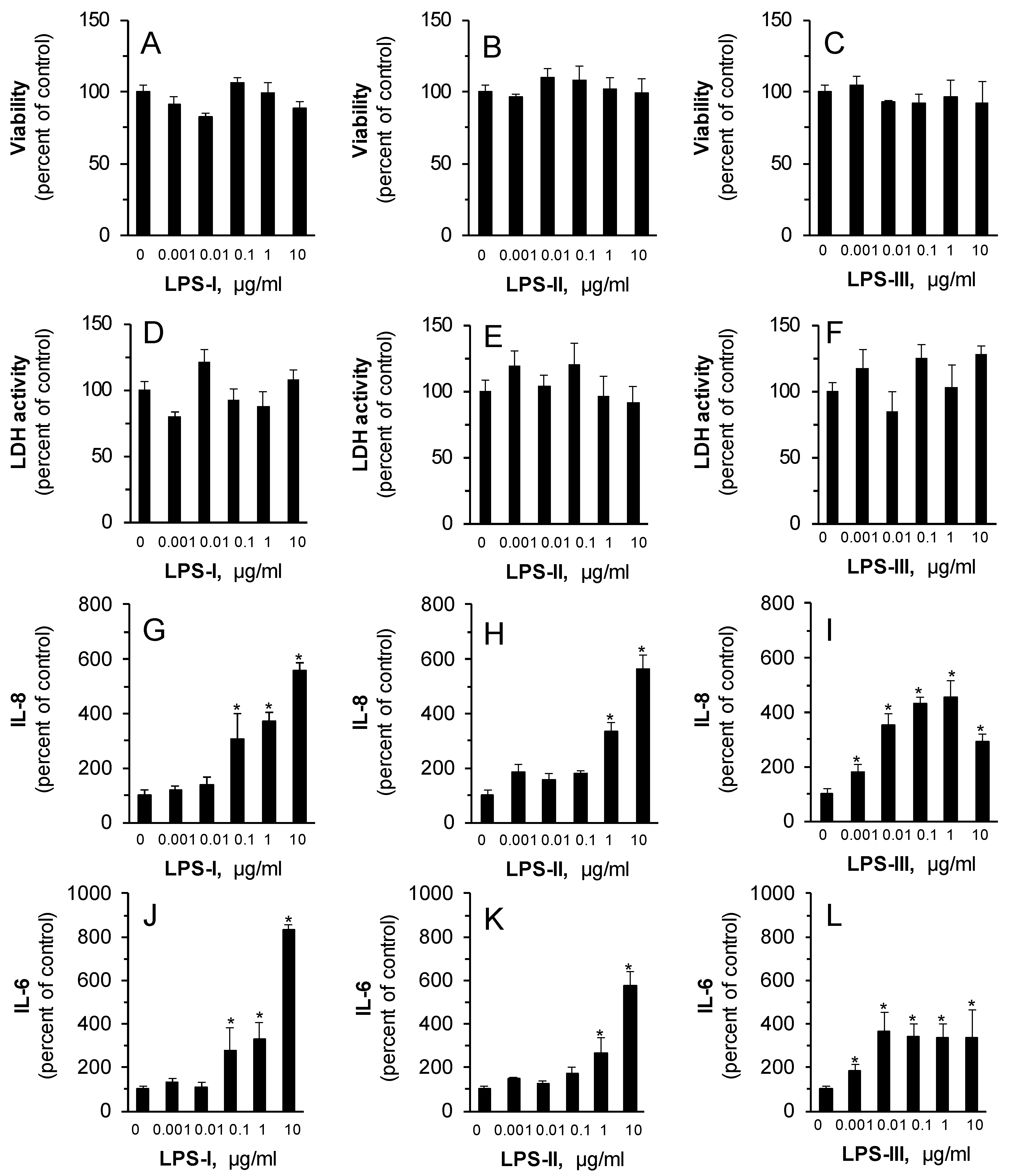
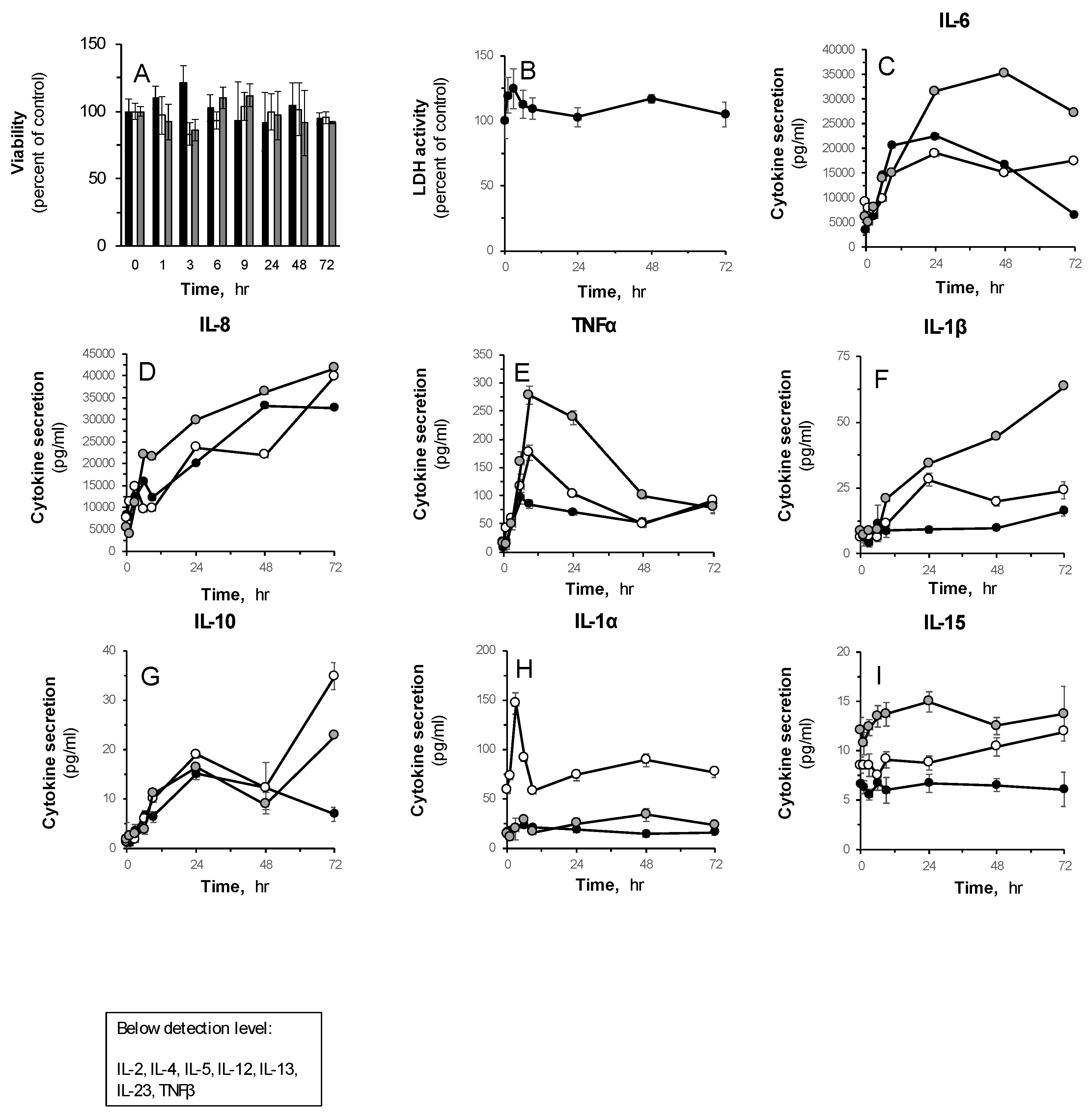
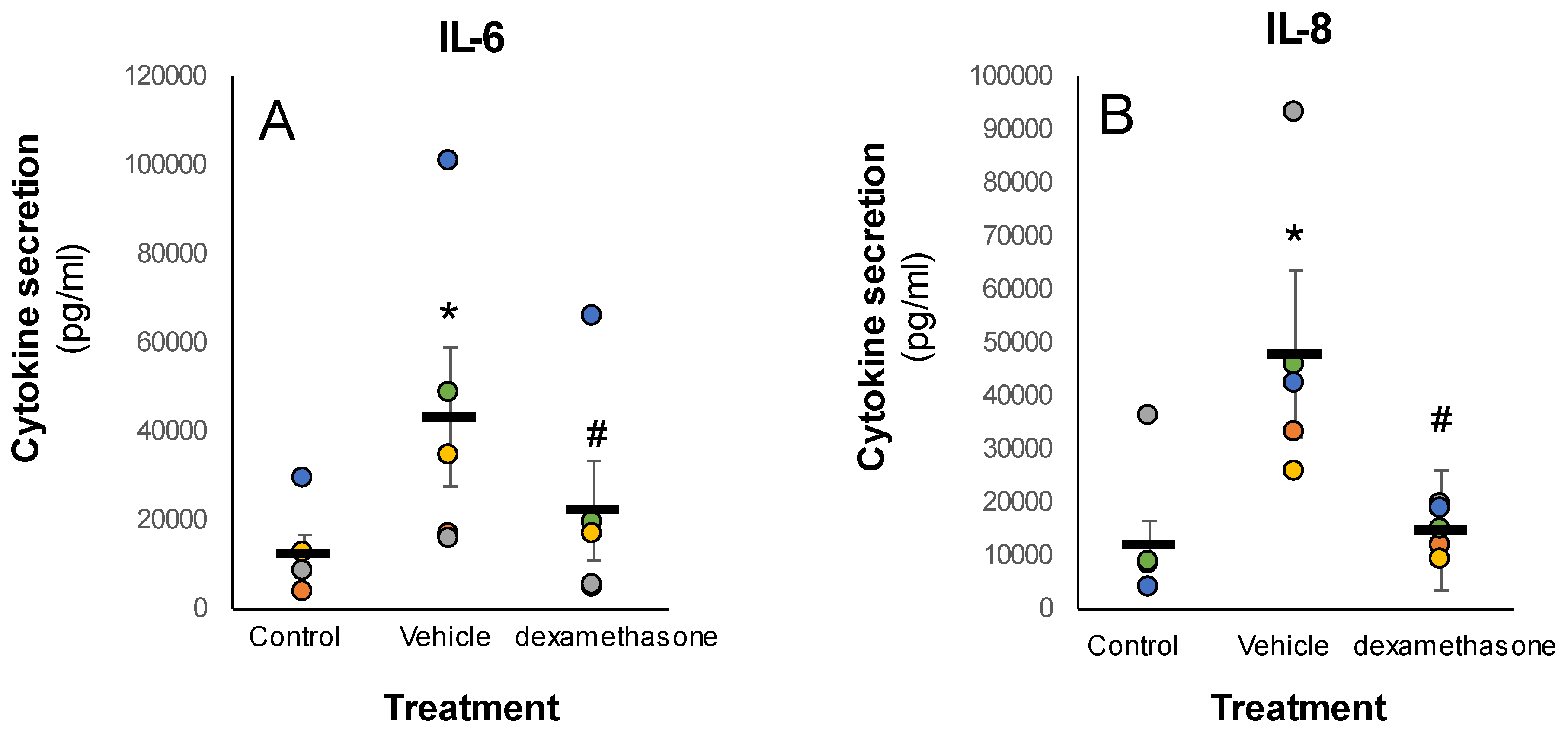
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Pasparakis, M.; Haase, I.; Nestle, F.O. Mechanisms regulating skin immunity and inflammation. Nat. Rev. Immunol. 2014, 14, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Nedoszytko, B.; Sokołowska-Wojdyło, M.; Ruckemann-Dziurdzińska, K.; Roszkiewicz, J.; Nowicki, R.J. Chemokines and cytokines network in the pathogenesis of the inflammatory skin diseases: Atopic dermatitis, psoriasis and skin mastocytosis. Postep. Dermatologii i Alergol. 2014, 31, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Proksch, E.; Brandner, J.M.; Jensen, J.M. The skin: An indispensable barrier. Exp. Dermatol. 2008, 17, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Coondoo, A. Cytokines in dermatology—A basic overview. Indian J. Dermatol. 2011, 56, 368. [Google Scholar] [CrossRef]
- Andrade, T.A.; Aguiar, A.F.; Guedes, F.A.; Leite, M.N.; Caetano, G.F.; Coelho, E.B.; Das, P.K.; Frade, M.A. Ex vivo model of human skin (hOSEC) as alternative to animal use for cosmetic tests. Procedia Eng. 2015, 110, 67–73. [Google Scholar] [CrossRef]
- Semlin, L.; Schäfer-Korting, M.; Borelli, C.; Korting, H.C. In vitro models for human skin disease. Drug Discov. Today 2011, 16, 132–139. [Google Scholar] [CrossRef]
- Mathes, S.H.; Ruffner, H.; Graf-Hausner, U. The use of skin models in drug development. Adv. Drug Deliv. Rev. 2014, 69–70, 81–102. [Google Scholar] [CrossRef]
- Corzo-León, D.E.; Munro, C.A.; MacCallum, D.M. An ex vivo human skin model to study superficial fungal infections. Front. Microbiol. 2019, 10, 1172. [Google Scholar] [CrossRef]
- Maboni, G.; Davenport, R.; Sessford, K.; Baiker, K.; Jensen, T.K.; Blanchard, A.M.; Wattegedera, S.; Entrican, G.; Tötemeyer, S. A novel 3D skin explant model to study anaerobic bacterial infection. Front. Cell. Infect. Microbiol. 2017, 7, 404. [Google Scholar] [CrossRef]
- Park, K.; Lee, J.-H.; Cho, H.-C.; Cho, S.-Y.; Cho, J.-W. Down-regulation of IL-6, IL-8, TNF-α and IL-1β by glucosamine in HaCaT cells, but not in the presence of TNF-α. Oncol. Lett. 2010, 1, 289–292. [Google Scholar] [CrossRef]
- Balato, A.; Lembo, S.; Mattii, M.; Schiattarella, M.; Marino, R.; De Paulis, A.; Balato, N.; Ayala, F. IL-33 is secreted by psoriatic keratinocytes and induces pro-inflammatory cytokines via keratinocyte and mast cell activation. Exp. Dermatol. 2012, 21, 892–894. [Google Scholar] [CrossRef] [PubMed]
- Netzlaff, F.; Lehr, C.M.; Wertz, P.W.; Schaefer, U.F. The human epidermis models EpiSkin®, SkinEthic® and EpiDerm®: An evaluation of morphology and their suitability for testing phototoxicity, irritancy, corrosivity, and substance transport. Eur. J. Pharm. Biopharm. 2005, 60, 167–178. [Google Scholar] [CrossRef]
- Portugal-Cohen, M.; Soroka, Y.; Frušić-Zlotkin, M.; Verkhovsky, L.; Brégégère, F.M.; Neuman, R.; Kohen, R.; Milner, Y. Skin organ culture as a model to study oxidative stress, inflammation and structural alterations associated with UVB-induced photodamage. Exp. Dermatol. 2011, 20, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Yehuda, H.; Soroka, Y.; Zlotkin-Frušić, M.; Gilhar, A.; Milner, Y.; Tamir, S. Isothiocyanates inhibit psoriasis-related proinflammatory factors in human skin. Inflamm. Res. 2012, 61, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Wineman, E.; Douglas, I.; Wineman, V.; Sharova, K.; Jaspars, M.; Meshner, S.; Bentwich, Z.; Cohen, G.; Shtevi, A. Commiphora gileadensis sap extract induces cell cycle-dependent death in immortalized keratinocytes and human dermoid carcinoma cells. J. Herb. Med. 2015, 5, 199–206. [Google Scholar] [CrossRef]
- Kahremany, S.; Babaev, I.; Gvirtz, R.; Ogen-Stern, N.; Azoulay-Ginsburg, S.; Senderowitz, H.; Cohen, G.; Gruzman, A. Nrf2 Activation by SK-119 Attenuates Oxidative Stress, UVB, and LPS-Induced Damage. Skin Pharmacol. Physiol. 2019, 32, 1–9. [Google Scholar] [CrossRef]
- Ogen-Shtern, N.; Chumin, K.; Cohen, G.; Borkow, G. Increased pro-collagen 1, elastin, and TGF-β1 expression by copper ions in an ex-vivo human skin model. J. Cosmet. Dermatol. 2019. [Google Scholar] [CrossRef]
- Hodgkinson, T.; Bayat, A. In vitro and ex vivo analysis of hyaluronan supplementation of integra® dermal template on human dermal fibroblasts and keratinocytes. J. Appl. Biomater. Funct. Mater. 2016, 14, 9–18. [Google Scholar] [CrossRef]
- Messager, S.; Hann, A.C.; Goddard, P.A.; Dettmar, P.W.; Maillard, J.Y. Assessment of skin viability: Is it necessary to use different methodologies? Ski. Res. Technol. 2003, 9, 321–330. [Google Scholar] [CrossRef]
- Portugal-Cohen, M.; Ish-Shalom, E.; Mallon, R.; Corral, P.; Michoux, F.; Ma’or, Z. Apple of Sodom (Calatropis procera) Callus Extract, a Novel Skincare Active and Its Biological Activity in Skin Models When Combined with Dead Sea Water. J. Cosmet. Dermatol. Sci. Appl. 2018, 8, 73–91. [Google Scholar] [CrossRef]
- Ng, K.W.; Pearton, M.; Coulman, S.; Anstey, A.; Gateley, C.; Morrissey, A.; Allender, C.; Birchall, J. Development of an ex vivo human skin model for intradermal vaccination: Tissue viability and Langerhans cell behaviour. Vaccine 2009, 27, 5948–5955. [Google Scholar] [CrossRef] [PubMed]
- Hanson, K.M.; Clegg, R.M. Observation and quantification of ultraviolet-induced reactive oxygen species in Ex Vivo human skin. Photochem. Photobiol. 2002, 76, 57–63. [Google Scholar] [CrossRef]
- Guilloteau, K.; Paris, I.; Pedretti, N.; Boniface, K.; Juchaux, F.; Huguier, V.; Guillet, G.; Bernard, F.X.; Lecron, J.C.; Morel, F. Skin inflammation induced by the synergistic action of IL-17A, IL-22, Oncostatin M, IL-1, and TNF-Recapitulates some features of psoriasis. J. Immunol. 2010, 184, 5263–5270. [Google Scholar] [CrossRef] [PubMed]
- Coolen, N.A.; Vlig, M.; Van Den Bogaerdt, A.J.; Middelkoop, E.; Ulrich, M.M.W. Development of an in vitro burn wound model. Wound Repair Regen. 2008, 16, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Bak, R.O.; Mikkelsen, J.G. Regulation of cytokines by small RNAs during skin inflammation. J. Biomed. Sci. 2010, 17, 53. [Google Scholar] [CrossRef]
- La Vecchia, C.; Garattini, S. An ideal minister of health. J. Epidemiol. Community Health 2002, 56, 890. [Google Scholar] [CrossRef] [PubMed]
- Helen, F.; Kanchana, G. Investigation on the properties of L-serine doped zinc tris (thiourea) sulphate crystal for NLO application. Indian J. Pure Appl. Phys. 2014, 52, 821–828. [Google Scholar]
- Companjen, A.R.; Van Der Wel, L.I.; Wei, L.; Laman, J.D.; Prens, E.P. A modifiedex vivo skin organ culture system for functional studies. Arch. Dermatol. Res. 2001, 293, 184–190. [Google Scholar] [CrossRef]
- Newton, K.; Dixit, V.M. Signaling in innate immunity and inflammation. Cold Spring Harb. Perspect. Biol. 2012, 4, a006049. [Google Scholar] [CrossRef]
- Portou, M.J.; Baker, D.; Abraham, D.; Tsui, J. The innate immune system, toll-like receptors and dermal wound healing: A review. Vascul. Pharmacol. 2015, 71, 31–36. [Google Scholar] [CrossRef]
- Miller, L.S. Toll-like receptors in skin. Adv. Dermatol. 2008, 24, 71–87. [Google Scholar] [CrossRef] [PubMed]
- Good, D.W.; George, T.; Watts, B.A. Toll-like receptor 2 is required for LPS-induced toll-like receptor 4 signaling and inhibition of ion transport in renal thick ascending limb. J. Biol. Chem. 2012, 287, 20208–20220. [Google Scholar] [CrossRef] [PubMed]
- Couper, K.N.; Blount, D.G.; Riley, E.M. IL-10: The Master Regulator of Immunity to Infection. J. Immunol. 2008, 180, 5771–5777. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, A.P.; Diez, D.; Miranda-Saavedra, D. The IL-10/STAT3-mediated anti-inflammatory response: Recent developments and future challenges. Brief. Funct. Genomics 2013, 12, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Banno, T.; Gazel, A.; Blumenberg, M. Effects of tumor necrosis factor-α (TNFα) in epidermal keratinocytes revealed using global transcriptional profiling. J. Biol. Chem. 2004, 279, 32633–32642. [Google Scholar] [CrossRef]
- Wills-Karp, M.; Finkelman, F.D. Untangling the complex web of IL-4-and IL-13-mediated signaling pathways. Sci. Signal. 2008, 1, pe55. [Google Scholar] [CrossRef]
- Carneiro, J.R.M.; Fuzii, H.T.; Kayser, C.; Alberto, F.L.; Soares, F.A.; Sato, E.I.; Andrade, L.E.C. IL-2, IL-5, TNF-α and IFN-γ mRNA expression in epidermal keratinocytes of systemic lupus erythematosus skin lesions. Clinics 2011, 66, 77–82. [Google Scholar] [CrossRef]
- Teng, M.W.L.; Bowman, E.P.; McElwee, J.J.; Smyth, M.J.; Casanova, J.L.; Cooper, A.M.; Cua, D.J. IL-12 and IL-23 cytokines: From discovery to targeted therapies for immune-mediated inflammatory diseases. Nat. Med. 2015, 21, 719. [Google Scholar] [CrossRef]
- Kang, J.; Kim, S.C.; Han, S.C.; Hong, H.J.; Jeon, Y.J.; Kim, B.; Koh, Y.S.; Yoo, E.S.; Kang, H.K. Hair-loss preventing effect of Grateloupia elliptica. Biomol. Ther. 2012, 20, 118. [Google Scholar] [CrossRef]
- Peiser, M.; Wanner, R.; Kolde, G. Human epidermal Langerhans cells differ from monocyte-derived Langerhans cells in CD80 expression and in secretion of IL-12 after CD40 cross-linking. J. Leukoc. Biol. 2004, 76, 616–622. [Google Scholar] [CrossRef]
- Schirmer, C.; Klein, C.; Von Bergen, M.; Simon, J.C.; Saalbach, A. Human fibroblasts support the expansion of IL-17-producing T cells via up-regulation of IL-23 production by dendritic cells. Blood 2010, 116, 1715–1725. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.M.; Bonefeld, C.M.; Poulsen, S.S.; Geisler, C.; Skov, L. IL-23 and TH17-mediated inflammation in human allergic contact dermatitis. J. Allergy Clin. Immunol. 2009, 123, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Piskin, G.; Sylva-Steenland, R.M.R.; Bos, J.D.; Teunissen, M.B.M. In Vitro and In Situ Expression of IL-23 by Keratinocytes in Healthy Skin and Psoriasis Lesions: Enhanced Expression in Psoriatic Skin. J. Immunol. 2006, 176, 1908–1915. [Google Scholar] [CrossRef]
- Shiraki, Y.; Ishibashi, Y.; Hiruma, M.; Nishikawa, A.; Ikeda, S. Cytokine secretion profiles of human keratinocytes during Trichophyton tonsurans and Arthroderma benhamiae infections. J. Med. Microbiol. 2006, 55, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gvirtz, R.; Ogen-Shtern, N.; Cohen, G. Kinetic Cytokine Secretion Profile of LPS-Induced Inflammation in the Human Skin Organ Culture. Pharmaceutics 2020, 12, 299. https://doi.org/10.3390/pharmaceutics12040299
Gvirtz R, Ogen-Shtern N, Cohen G. Kinetic Cytokine Secretion Profile of LPS-Induced Inflammation in the Human Skin Organ Culture. Pharmaceutics. 2020; 12(4):299. https://doi.org/10.3390/pharmaceutics12040299
Chicago/Turabian StyleGvirtz, Raanan, Navit Ogen-Shtern, and Guy Cohen. 2020. "Kinetic Cytokine Secretion Profile of LPS-Induced Inflammation in the Human Skin Organ Culture" Pharmaceutics 12, no. 4: 299. https://doi.org/10.3390/pharmaceutics12040299
APA StyleGvirtz, R., Ogen-Shtern, N., & Cohen, G. (2020). Kinetic Cytokine Secretion Profile of LPS-Induced Inflammation in the Human Skin Organ Culture. Pharmaceutics, 12(4), 299. https://doi.org/10.3390/pharmaceutics12040299






