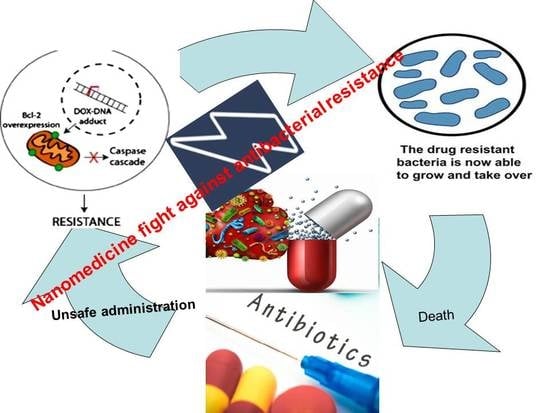
Nanomedicine Fight against Antibacterial Resistance: An Overview of the Recent Pharmaceutical Innovations
Abstract
1. Introduction
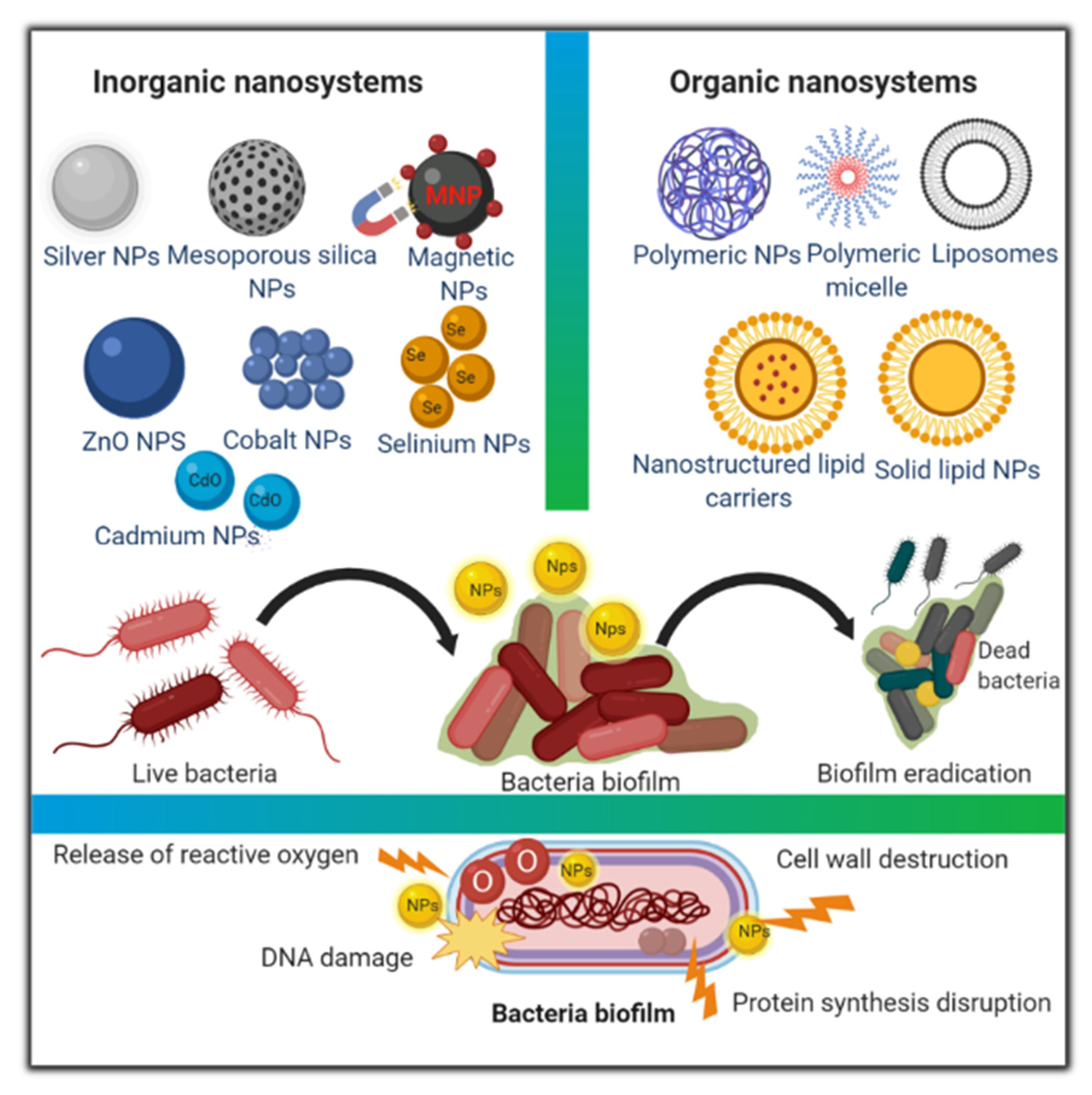
2. Nanosystems’ Role in Overcoming Antibiotic Resistance
2.1. Mechanism of Nanosystems as Antibacterial Drug Delivery Agents
2.2. Classification of Nanosystems
2.2.1. Organic Nanosystems
Liposomes
- 1-
- Physical and chemical instability problems, that can be minimized by addition of antioxidants and/or freeze-drying [28].
- 2-
- The possibility of antibiotic leakage from liposomes under physiological conditions, that can be controlled by adding cholesterol which lead to stabilization of liposomal membrane [48].
- 3-
- 4-
- Special sterilization techniques are needed due to the sensitivity of lipids to high temperatures [51].
- 5-
- Fabrication techniques are very complex, expensive, and difficult to be scaled up [52].
Lipid-Based Nanoparticles
Polymeric Micelles
Polymeric Nanoparticles
2.2.2. Inorganic Nanosystems
Silver NPs (Ag-NPs)
Silica Nanoparticles
Magnetic Nanoparticles, MNPs
Zinc Oxide (ZnO) Nanoparticles
Cobalt NPs
Selenium Nanoparticles (Se-NPs)
Cadmium Nanoparticles (CdO-Nps)
Advantages and Drawbacks of Inorganic Nanoparticles
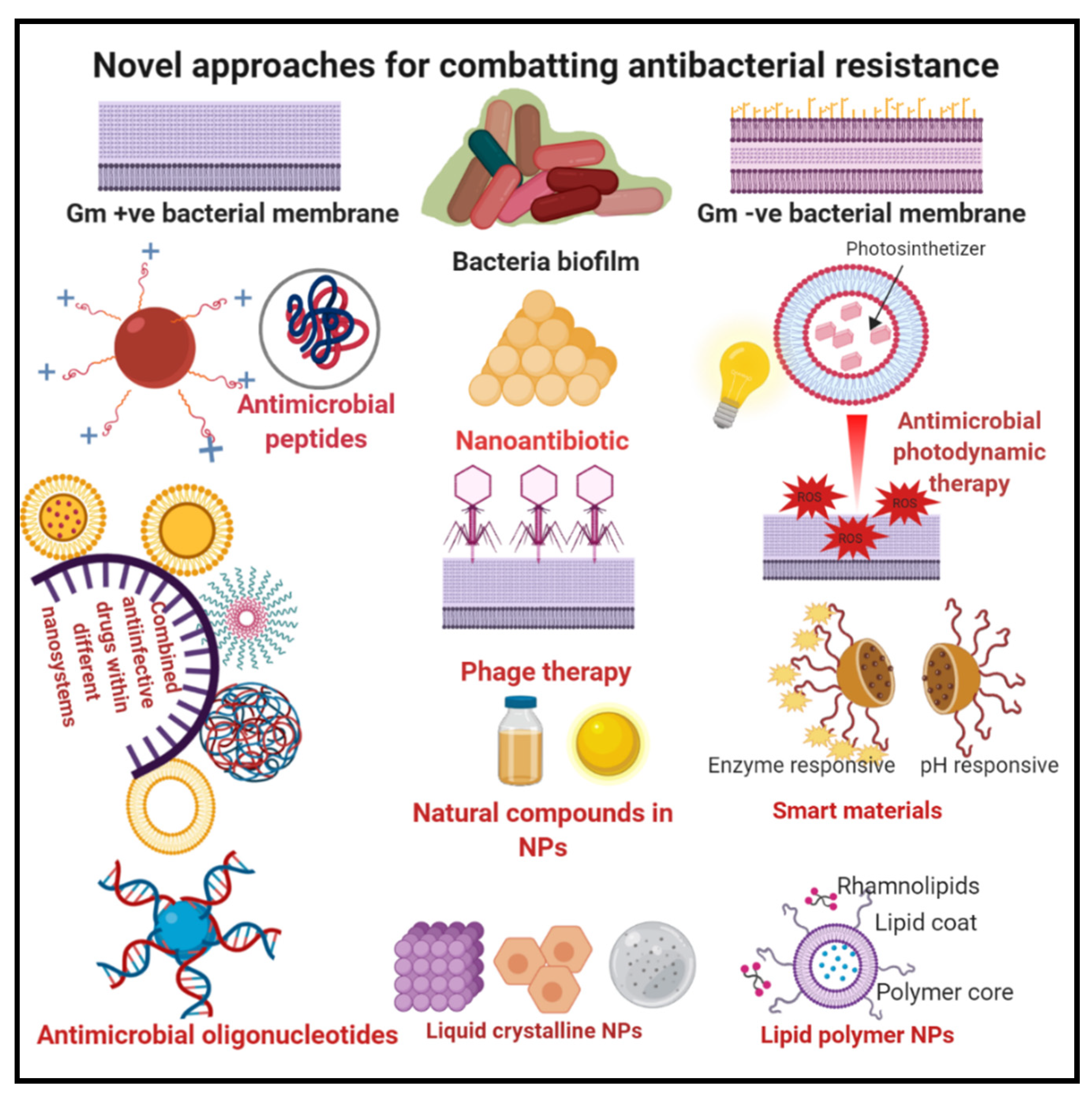
3. Novel Approaches for Combatting Antibacterial Resistance
3.1. Lipid Polymer Nanoparticles (LPNs)
3.2. Nonlamellar Lyotropic Liquid Crystalline Nanoparticles
3.3. Anti-Microbial Oligonucleotides
3.4. Combination of Nanotechnology and Natural Compounds
3.5. Smart Materials
3.6. Cationic Peptides
3.7. Antimicrobial Photodynamic Therapy (aPDT)
3.8. Nano Systems with Combination Drug Therapy
3.9. Nano-Antibiotic
3.10. Phage Therapy
4. Clinical Trials
4.1. Current and Future Market of Nanosystem Antibiotics
4.1.1. Antibiotic Agents
4.1.2. Anti-Toxin Agents
4.1.3. Antimicrobial Peptides
5. Challenges in the Clinical Translation of Nanomedicine
6. Recommendations and Perspectives
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Willyard, C. The drug-resistant bacteria that pose the greatest health threats. Nature 2017, 543, 15. [Google Scholar] [CrossRef]
- Kourtis, A.P.; Hatfield, K.; Baggs, J.; Mu, Y.; See, I.; Epson, E.; Nadle, J.; Kainer, M.A.; Dumyati, G.; Petit, S. Vital Signs: Epidemiology and Recent Trends in Methicillin-Resistant and in Methicillin-Susceptible Staphylococcus aureus Bloodstream Infections—United States. Morb. Mortal. Wkly. Rep. 2019, 68, 214. [Google Scholar] [CrossRef]
- Livorsi, D.J.; Chorazy, M.L.; Schweizer, M.L.; Balkenende, E.C.; Blevins, A.E.; Nair, R.; Samore, M.H.; Nelson, R.E.; Khader, K.; Perencevich, E.N. A systematic review of the epidemiology of carbapenem-resistant Enterobacteriaceae in the United States. Antimicrob. Resist. Infect. Control 2018, 7, 55. [Google Scholar] [CrossRef]
- Li, B.; Webster, T.J. Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections. J. Orthop. Res. 2018, 36, 22–32. [Google Scholar] [CrossRef]
- Blair, J.M.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015, 13, 42–51. [Google Scholar] [CrossRef]
- Aslam, B.; Wang, W.; Arshad, M.I.; Khurshid, M.; Muzammil, S.; Rasool, M.H.; Nisar, M.A.; Alvi, R.F.; Aslam, M.A.; Usman, Q.M. Antibiotic resistance: A rundown of a global crisis. Infect. Drug Resist. 2018, 11, 1645. [Google Scholar] [CrossRef]
- Parisi, O.I.; Scrivano, L.; Sinicropi, M.S.; Puoci, F. Polymeric nanoparticle constructs as devices for antibacterial therapy. Curr. Opin. Pharmacol. 2017, 36, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Berlanga, M.; Guerrero, R. Living together in biofilms: The microbial cell factory and its biotechnological implications. Microb. Cell Factories 2016, 15, 165. [Google Scholar] [CrossRef] [PubMed]
- Rizzato, C.; Torres, J.; Kasamatsu, E.; Camorlinga, M.; Bravo, M.M.; Canzian, F.; Kato, I. Potential role of biofilm formation in the development of digestive tract cancer with special reference to Helicobacter pylori infection. Front. Microbiol. 2019, 10, 846. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, S.; Pal, S. Bacterial intelligence: Imitation games, time-sharing, and long-range quantum coherence. J. Cell Commun. Signal. 2017, 11, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Blair, K.M.; Turner, L.; Winkelman, J.T.; Berg, H.C.; Kearns, D.B. A molecular clutch disables flagella in the Bacillus subtilis biofilm. Science 2008, 320, 1636–1638. [Google Scholar] [CrossRef] [PubMed]
- Rossi-Fedele, G.; Roberts, A. A preliminary study investigating the survival of tetracycline resistant Enterococcus faecalis after root canal irrigation with high concentrations of tetracycline. Int. Endod. J. 2007, 40, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Valappil, S.P. Nanosystems and antibacterial applications. In Drug Delivery Nanosystems for Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 75–90. [Google Scholar]
- Marsich, E.; Travan, A.; Donati, I.; Turco, G.; Bellomo, F.; Paoletti, S. Tissue-implant antimicrobial interfaces. In Antimicrobial Polymers; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 379–428. [Google Scholar]
- Zhang, L.; Mah, T.-F. Involvement of a novel efflux system in biofilm-specific resistance to antibiotics. J. Bacteriol. 2008, 190, 4447–4452. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K. Persister cells and the riddle of biofilm survival. Biochemistry 2005, 70, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.L.; Schneider, T.; Peoples, A.J.; Spoering, A.L.; Engels, I.; Conlon, B.P.; Mueller, A.; Schäberle, T.F.; Hughes, D.E.; Epstein, S. A new antibiotic kills pathogens without detectable resistance. Nature 2015, 517, 455. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, D.; Kiselev, M.A.; Caccamo, M.T. Smart nanoparticles for drug delivery application: Development of versatile nanocarrier platforms in biotechnology and nanomedicine. J. Nanomater. 2019, 2019, 3702518. [Google Scholar] [CrossRef]
- Abed, N.; Couvreur, P. Nanocarriers for antibiotics: A promising solution to treat intracellular bacterial infections. Int. J. Antimicrob. Agents 2014, 43, 485–496. [Google Scholar] [CrossRef]
- Dacoba, T.G.; Olivera, A.; Torres, D.; Crecente-Campo, J.; Alonso, M.J. Modulating the immune system through nanotechnology. In Seminars in Immunology; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Gustafson, H.H.; Holt-Casper, D.; Grainger, D.W.; Ghandehari, H. Nanoparticle uptake: The phagocyte problem. Nano Today 2015, 10, 487–510. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; González, B.; Izquierdo-Barba, I. Nanomaterials as Promising Alternative in the Infection Treatment. Int. J. Mol. Sci. 2019, 20, 3806. [Google Scholar] [CrossRef]
- Baptista, P.V.; McCusker, M.P.; Carvalho, A.; Ferreira, D.A.; Mohan, N.M.; Martins, M.; Fernandes, A.R. Nano-strategies to fight multidrug resistant bacteria—“A Battle of the Titans”. Front. Microbiol. 2018, 9, 1441. [Google Scholar] [CrossRef]
- Baranwal, A.; Srivastava, A.; Kumar, P.; Bajpai, V.K.; Maurya, P.K.; Chandra, P. Prospects of nanostructure materials and their composites as antimicrobial agents. Front. Microbiol. 2018, 9, 422. [Google Scholar] [CrossRef]
- Ashik, U.P.M.; Kudo, S.; Hayashi, J.-I. Chapter 2—An Overview of Metal Oxide Nanostructures. In Synthesis of Inorganic Nanomaterials; Mohan Bhagyaraj, S., Oluwafemi, O.S., Kalarikkal, N., Thomas, S., Eds.; Woodhead Publishing: Sawston, UK, 2018; pp. 19–57. [Google Scholar]
- Martin-Serrano, Á.; Gómez, R.; Ortega, P.; de la Mata, F.J. Nanosystems as vehicles for the delivery of antimicrobial peptides (AMPs). Pharmaceutics 2019, 11, 448. [Google Scholar] [CrossRef]
- Drulis-Kawa, Z.; Dorotkiewicz-Jach, A. Liposomes as delivery systems for antibiotics. Int. J. Pharm. 2010, 387, 187–198. [Google Scholar] [CrossRef]
- Omar, M.M.; Hasan, O.A.; El Sisi, A.M. Preparation and optimization of lidocaine transferosomal gel containing permeation enhancers: A promising approach for enhancement of skin permeation. Int. J. Nanomed. 2019, 14, 1551–1562. [Google Scholar] [CrossRef]
- Omar, M.M.; Eleraky, N.E.; El Sisi, A.M.; Hasan, O.A. Development and Evaluation of in-situ Nasal Gel Formulations of Nanosized Transferosomal Sumatriptan: Design, Optimization, in vitro and in vivo Evaluation. Drug Des. Dev. Ther. 2019, 13, 4413–4430. [Google Scholar] [CrossRef]
- Rukavina, Z.; Vanić, Ž. Current trends in development of liposomes for targeting bacterial biofilms. Pharmaceutics 2016, 8, 18. [Google Scholar] [CrossRef]
- Sharma, A.; Sharma, U.S. Liposomes in drug delivery: Progress and limitations. Int. J. Pharm. 1997, 154, 123–140. [Google Scholar] [CrossRef]
- Alavi, M.; Karimi, N.; Safaei, M. Application of various types of liposomes in drug delivery systems. Adv. Pharm. Bull. 2017, 7, 3. [Google Scholar] [CrossRef]
- Atbiaw, N.; Aman, E.; Dessalegn, B. Review on targeted drug delivery against intracellular pathogen. Pharm. Pharmacol. Int. J. 2018, 6, 183–189. [Google Scholar] [CrossRef]
- Rukavina, Z.; Klarić, M.Š.; Filipović-Grčić, J.; Lovrić, J.; Vanić, Ž. Azithromycin-loaded liposomes for enhanced topical treatment of methicillin-resistant staphyloccocus aureus (mrsa) infections. Int. J. Pharm. 2018, 553, 109–119. [Google Scholar] [CrossRef]
- Vanić, Ž.; Rukavina, Z.; Manner, S.; Fallarero, A.; Uzelac, L.; Kralj, M.; Klarić, D.A.; Bogdanov, A.; Raffai, T.; Virok, D.P. Azithromycin-liposomes as a novel approach for localized therapy of cervicovaginal bacterial infections. Int. J. Nanomed. 2019, 14, 5957. [Google Scholar] [CrossRef]
- Castoldi, A.; Herr, C.; Niederstraßer, J.; Labouta, H.I.; Melero, A.; Gordon, S.; Schneider-Daum, N.; Bals, R.; Lehr, C.M. Calcifediol-loaded liposomes for local treatment of pulmonary bacterial infections. Eur. J. Pharm. Biopharm. 2017, 118, 62–67. [Google Scholar] [CrossRef]
- Lai, S.; Wei, Y.; Wu, Q.; Zhou, K.; Liu, T.; Zhang, Y.; Jiang, N.; Xiao, W.; Chen, J.; Liu, Q. Liposomes for effective drug delivery to the ocular posterior chamber. J. Nanobiotechnol. 2019, 17, 64. [Google Scholar] [CrossRef]
- Yadav, D.; Sandeep, K.; Pandey, D.; Dutta, R. Liposomes for drug delivery. J. Biotechnol. Biomater. 2017, 7, 276. [Google Scholar] [CrossRef]
- Abed, N.; Saïd-Hassane, F.; Zouhiri, F.; Mougin, J.; Nicolas, V.; Desmaële, D.; Gref, R.; Couvreur, P. An efficient system for intracellular delivery of beta-lactam antibiotics to overcome bacterial resistance. Sci. Rep. 2015, 5, 13500. [Google Scholar] [CrossRef]
- Uhl, P.; Pantze, S.; Storck, P.; Parmentier, J.; Witzigmann, D.; Hofhaus, G.; Huwyler, J.; Mier, W.; Fricker, G. Oral delivery of vancomycin by tetraether lipid liposomes. Eur. J. Pharm. Sci. 2017, 108, 111–118. [Google Scholar] [CrossRef]
- Alshamsan, A.; Aleanizy, F.S.; Badran, M.; Alqahtani, F.Y.; Alfassam, H.; Almalik, A.; Alosaimy, S. Exploring anti-MRSA activity of chitosan-coated liposomal dicloxacillin. J. Microbiol. Methods 2019, 156, 23–28. [Google Scholar] [CrossRef]
- Wang, W.; Shao, A.; Feng, S.; Ding, M.; Luo, G. Physicochemical characterization and gastrointestinal adhesion of S-layer proteins-coating liposomes. Int. J. Pharm. 2017, 529, 227–237. [Google Scholar] [CrossRef]
- Wijetunge, S.S.; Wen, J.; Yeh, C.-K.; Sun, Y. Wheat germ agglutinin liposomes with surface grafted cyclodextrins as bioadhesive dual-drug delivery nanocarriers to treat oral cells. Colloids Surf. B Biointerfaces 2020, 185, 110572. [Google Scholar] [CrossRef]
- Alhariri, M.; Majrashi, M.A.; Bahkali, A.H.; Almajed, F.S.; Azghani, A.O.; Khiyami, M.A.; Alyamani, E.J.; Aljohani, S.M.; Halwani, M.A. Efficacy of neutral and negatively charged liposome-loaded gentamicin on planktonic bacteria and biofilm communities. Int. J. Nanomed. 2017, 12, 6949–6961. [Google Scholar] [CrossRef] [PubMed]
- Niu, N.-K.; Yin, J.-J.; Yang, Y.-X.; Wang, Z.-L.; Zhou, Z.-W.; He, Z.-X.; Chen, X.W.; Zhang, X.; Wei, D.; Yang, T. Novel targeting of PEGylated liposomes for codelivery of TGF-β1 siRNA and four antitubercular drugs to human macrophages for the treatment of mycobacterial infection: A quantitative proteomic study. Drug Des. Dev. Ther. 2015, 9, 4441. [Google Scholar]
- Bilton, D.; Pressler, T.; Fajac, I.; Clancy, J.P.; Sands, D.; Minic, P.; Cipolli, M.; Galeva, I.; Sole, A.; Quittner, A.L. Amikacin liposome inhalation suspension for chronic Pseudomonas aeruginosa infection in cystic fibrosis. J. Cyst. Fibros. 2019. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, A.S. Biophysical aspects of using liposomes as delivery vehicles. Biosci. Rep. 2002, 22, 129–150. [Google Scholar] [CrossRef]
- Alhajlan, M.; Alhariri, M.; Omri, A. Efficacy and safety of liposomal clarithromycin and its effect on Pseudomonas aeruginosa virulence factors. Antimicrob. Agents Chemother. 2013, 57, 2694–2704. [Google Scholar] [CrossRef]
- Moyá, M.L.; López-López, M.; Lebrón, J.A.; Ostos, F.J.; Pérez, D.; Camacho, V.; Beck, I.; Merino-Bohórquez, V.; Camean, M.; Madinabeitia, N. Preparation and Characterization of New Liposomes. Bactericidal Activity of Cefepime Encapsulated into Cationic Liposomes. Pharmaceutics 2019, 11, 69. [Google Scholar] [CrossRef]
- Toh, M.-R.; Chiu, G.N. Liposomes as sterile preparations and limitations of sterilisation techniques in liposomal manufacturing. Asian J. Pharm. Sci. 2013, 8, 88–95. [Google Scholar] [CrossRef]
- Nkanga, C.I.; Bapolisi, A.M.; Okafor, N.I.; Krause, R.W.M. General Perception of Liposomes: Formation, Manufacturing and Applications. In Liposomes-Advances and Perspectives; IntechOpen: London, UK, 2019. [Google Scholar]
- Clancy, J.; Dupont, L.; Konstan, M.; Billings, J.; Fustik, S.; Goss, C.; Lymp, J.; Minic, P.; Quittner, A.L.; Rubenstein, R.C. Phase II studies of nebulised Arikace in CF patients with Pseudomonas aeruginosa infection. Thorax 2013, 68, 818–825. [Google Scholar] [CrossRef]
- Bruinenberg, P.; Blanchard, J.D.; Cipolla, D.C.; Dayton, F.; Mudumba, S.; Gonda, I. Inhaled liposomal ciprofloxacin: Once a day management of respiratory infections. In Respiratory Drug Delivery; Davis Healthcare International Publishing River Grove: Orlando, FL, USA, 2010. [Google Scholar]
- Pignatello, R.; Nicolosi, D.; Nicolosi, V.M. Fusogenic liposomes as new carriers to enlarge the spectrum of action of antibiotic drugs against Gram-negative bacteria. In Science against Microbial Pathogens: Communicating Current Research and Technological Advances; Formatex Research Center: Badajoz, Spain, 2011; pp. 52–56. [Google Scholar]
- Beaulac, C.; Sachetelli, S.; Lagace, J. In vitro bactericidal efficacy of sub-MIC concentrations of liposome-encapsulated antibiotic against gram-negative and gram-positive bacteria. J. Antimicrob. Chemother. 1998, 41, 35–41. [Google Scholar] [CrossRef]
- Beaulac, C.; Sachetelli, S.; Lagacé, J. Aerosolization of low phase transition temperature liposomal tobramycin as a dry powder in an animal model of chronic pulmonary infection caused by Pseudomonas aeruginosa. J. Drug Target. 1999, 7, 33–41. [Google Scholar] [CrossRef]
- Zhao Wang, Y.M.; Khalil, H.; Wang, R.; Lu, T.; Zhao, W.; Zhang, Y.; Chen, J.; Chen, T. Fusion between fluid liposomes and intact bacteria: Study of driving parameters and in vitro bactericidal efficacy. Int. J. Nanomed. 2016, 11, 4025. [Google Scholar]
- Ishida, T.; Ichikawa, T.; Ichihara, M.; Sadzuka, Y.; Kiwada, H. Effect of the physicochemical properties of initially injected liposomes on the clearance of subsequently injected PEGylated liposomes in mice. J. Control. Release 2004, 95, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.; Muiruri, P.W.; Jones, G.H.; Scott, M.J.; Jones, M.N. The effect of grafted poly (ethylene glycol) on the electrophoretic properties of phospholipid liposomes and their adsorption to bacterial biofilms. Colloids Surf. A Physicochem. Eng. Asp. 2001, 194, 287–296. [Google Scholar] [CrossRef]
- Moghadas-Sharif, N.; Fazly Bazzaz, B.S.; Khameneh, B.; Malaekeh-Nikouei, B. The effect of nanoliposomal formulations on Staphylococcus epidermidis biofilm. Drug Dev. Ind. Pharm. 2015, 41, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Kaszuba, M.; Jones, M.N. Hydrogen peroxide production from reactive liposomes encapsulating enzymes. Biochimica Et Biophysica Acta (BBA) Biomembr. 1999, 1419, 221–228. [Google Scholar] [CrossRef]
- Bai, J.; Yang, E.; Chang, P.-S.; Ryu, S. Preparation and characterization of endolysin-containing liposomes and evaluation of their antimicrobial activities against gram-negative bacteria. Enzym. Microb. Technol. 2019, 128, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.J.; Kaszuba, M.; Creeth, J.E.; Jones, M.N. Reactive liposomes encapsulating a glucose oxidase-peroxidase system with antibacterial activity. Biochimica Et Biophysica Acta (BBA) Biomembr. 1997, 1326, 37–46. [Google Scholar] [CrossRef]
- Jones, M.N.; Hill, K.J.; Kaszuba, M.; Creeth, J.E. Antibacterial reactive liposomes encapsulating coupled enzyme systems. Int. J. Pharm. 1998, 162, 107–117. [Google Scholar] [CrossRef]
- Goss, C.H.; Kaneko, Y.; Khuu, L.; Anderson, G.D.; Ravishankar, S.; Aitken, M.L.; Lechtzin, N.; Zhou, G.; Czyz, D.M.; McLean, K. Gallium disrupts bacterial iron metabolism and has therapeutic effects in mice and humans with lung infections. Sci. Transl. Med. 2018, 10, eaat7520. [Google Scholar] [CrossRef]
- Alipour, M.; Suntres, Z.E.; Lafrenie, R.M.; Omri, A. Attenuation of Pseudomonas aeruginosa virulence factors and biofilms by co-encapsulation of bismuth–ethanedithiol with tobramycin in liposomes. J. Antimicrob. Chemother. 2010, 65, 684–693. [Google Scholar] [CrossRef]
- Halwani, M.; Yebio, B.; Suntres, Z.; Alipour, M.; Azghani, A.; Omri, A. Co-encapsulation of gallium with gentamicin in liposomes enhances antimicrobial activity of gentamicin against Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2008, 62, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Alhariri, M.; Omri, A. Efficacy of liposomal bismuth-ethanedithiol-loaded tobramycin after intratracheal administration in rats with pulmonary Pseudomonas aeruginosa infection. Antimicrob. Agents Chemother. 2013, 57, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Mokhtar, M.; Nair, A.; Al-Dhubiab, B.; Shehata, T. Hydrogels and Their Combination with Liposomes, Niosomes, or Transfersomes for Dermal and Transdermal Drug Delivery. Liposomes 2017, 155, 155–186. [Google Scholar]
- Eroğlu, İ.; Aslan, M.; Yaman, Ü.; Gultekinoglu, M.; Çalamak, S.; Kart, D.; Ulubayram, K. Liposome Based Combination Therapy for Acne Treatment. J. Liposome Res. 2019. [Google Scholar] [CrossRef]
- Thapa, R.K.; Kiick, K.L.; Sullivan, M.O. Encapsulation of collagen mimetic peptide-tethered vancomycin liposomes in collagen-based scaffolds for infection control in wounds. Acta Biomater. 2020, 103, 115–128. [Google Scholar] [CrossRef]
- Liu, L.; Xiang, Y.; Wang, Z.; Yang, X.; Yu, X.; Lu, Y.; Deng, L.; Cui, W. Adhesive liposomes loaded onto an injectable, self-healing and antibacterial hydrogel for promoting bone reconstruction. NPG Asia Mater. 2019, 11, 81. [Google Scholar] [CrossRef]
- Hui, T.; Yongqing, X.; Tiane, Z.; Gang, L.; Yonggang, Y.; Muyao, J.; Jun, L.; Jing, D. Treatment of osteomyelitis by liposomal gentamicin-impregnated calcium sulfate. Arch. Orthop. Trauma Surg. 2009, 129, 1301–1308. [Google Scholar] [CrossRef]
- Zhu, C.-T.; Xu, Y.-Q.; Shi, J.; Li, J.; Ding, J. Liposome combined porous β-TCP scaffold: Preparation, characterization, and anti-biofilm activity. Drug Deliv. 2010, 17, 391–398. [Google Scholar] [CrossRef]
- Liu, X.-M.; Ren, K.; Wu, G.; Wang, D. Preparation and Evaluation of Biomineral-Binding Antibiotic Liposomes. Liposome-Based Drug Deliv. Syst. 2018. [Google Scholar] [CrossRef]
- Mourtas, S.; Diamanti, G.; Foka, A.; Dracopoulos, V.; Klepetsanis, P.; Stamouli, V.; Spiliopoulou, I.; Antimisiaris, S. Inhibition of Bacterial Attachment on Surfaces by Immobilization of Tobramycin-Loaded Liposomes. J. Biomed. Nanotechnol. 2015. [Google Scholar] [CrossRef]
- Campardelli, R.; Trucillo, P.; Reverchon, E. Supercritical assisted process for the efficient production of liposomes containing antibiotics for ocular delivery. J. CO2 Util. 2018, 25, 235–241. [Google Scholar] [CrossRef]
- Jadhav, M.; Kalhapure, R.S.; Rambharose, S.; Mocktar, C.; Singh, S.; Kodama, T.; Govender, T. Novel lipids with three C18-fatty acid chains and an amino acid head group for pH-responsive and sustained antibiotic delivery. Chem. Phys. Lipids 2018, 212, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Khatib, I.; Khanal, D.; Ruan, J.; Cipolla, D.; Dayton, F.; Blanchard, J.D.; Chan, H.K. Ciprofloxacin nanocrystals liposomal powders for controlled drug release via inhalation. Int. J. Pharm. 2019. [Google Scholar] [CrossRef]
- Risaliti, L.; Kehagia, A.; Daoultzi, E.; Lazari, D.; Bergonzi, M.C.; Vergkizi-Nikolakaki, S.; Hadjipavlou-Litina, D.; Bilia, A.R. Liposomes loaded with Salvia triloba and Rosmarinus officinalis essential oils: In vitro assessment of antioxidant, antiinflammatory and antibacterial activities. J. Drug Deliv. Sci. Technol. 2019, 51, 493–498. [Google Scholar] [CrossRef]
- Giordani, B.; Costantini, P.E.; Fedi, S.; Cappelletti, M.; Abruzzo, A.; Parolin, C.; Foschi, C.; Frisco, G.; Calonghi, N.; Cerchiara, T. Liposomes containing biosurfactants isolated from Lactobacillus gasseri exert antibiofilm activity against methicillin resistant Staphylococcus aureus strains. Eur. J. Pharm. Biopharm. 2019, 139, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Cheng, F.; Swing, C.J.; Xia, S.; Zhang, X. Modulation effect of core-wall ratio on the stability and antibacterial activity of cinnamaldehyde liposomes. Chem. Phys. Lipids 2019, 223, 104790. [Google Scholar] [CrossRef]
- Nicolosi, D.; Cupri, S.; Genovese, C.; Tempera, G.; Mattina, R.; Pignatello, R. Nanotechnology approaches for antibacterial drug delivery: Preparation and microbiological evaluation of fusogenic liposomes carrying fusidic acid. Int. J. Antimicrob. Agents 2015, 45, 622–626. [Google Scholar] [CrossRef]
- Cavalcanti, I.; Pontes-Neto, J.; Kocerginsky, P.; Bezerra-Neto, A.; Lima, J.; Lira-Nogueira, M.; Maciel, M.A.V.; Neves, R.P.; Pimentel, M.F.; Santos-Magalhães, N.S. Antimicrobial activity of β-lapachone encapsulated into liposomes against meticillin-resistant Staphylococcus aureus and Cryptococcus neoformans clinical strains. J. Glob. Antimicrob. Resist. 2015, 3, 103–108. [Google Scholar] [CrossRef]
- Monteiro, N.; Martins, M.; Martins, A.; Fonseca, N.A.; Moreira, J.N.; Reis, R.L.; Neves, N.M. Antibacterial activity of chitosan nanofiber meshes with liposomes immobilized releasing gentamicin. Acta Biomater. 2015, 18, 196–205. [Google Scholar] [CrossRef]
- Zhang, Y.; Pu, C.; Tang, W.; Wang, S.; Sun, Q. Gallic acid liposomes decorated with lactoferrin: Characterization, in vitro digestion and antibacterial activity. Food Chem. 2019, 293, 315–322. [Google Scholar] [CrossRef]
- Cheng, C.; Wu, Z.; McClements, D.J.; Zou, L.; Peng, S.; Zhou, W.; Liu, W. Improvement on stability, loading capacity and sustained release of rhamnolipids modified curcumin liposomes. Colloids Surf. B Biointerfaces 2019, 183, 110460. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Guo, B.; Wang, S.; Ding, J.; Zhou, W. A thermo-responsive and self-healing liposome-in-hydrogel system as an antitubercular drug carrier for localized bone tuberculosis therapy. Int. J. Pharm. 2019, 558, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, A.A.; Aygül, A.; Şenel, B. Influence of glyceryl behenate, tripalmitin and stearic acid on the properties of clarithromycin incorporated solid lipid nanoparticles (SLNs): Formulation, characterization, antibacterial activity and cytotoxicity. J. Drug Deliv. Sci. Technol. 2019, 54, 101240. [Google Scholar] [CrossRef]
- Öztürk, A.A.; Gündüz, A.B.; Ozisik, O. Supervised Machine Learning Algorithms for Evaluation of Solid Lipid Nanoparticles and Particle Size. Comb. Chem. High Throughput Screen. 2018, 21, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Mishra, V.; Bansal, K.K.; Verma, A.; Yadav, N.; Thakur, S.; Sudhakar, K.; Rosenholm, J.M. Solid lipid nanoparticles: Emerging colloidal nano drug delivery systems. Pharmaceutics 2018, 10, 191. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Gupta, N.; Gupta, S. Implications of designing clarithromycin loaded solid lipid nanoparticles on their pharmacokinetics, antibacterial activity and safety. RSC Adv. 2016, 6, 76621–76631. [Google Scholar] [CrossRef]
- Mahajan, P.S.; Mahajan, K.B.; Darekar, A. A review on solid lipid nanoparticle (SLN): An advanced treatment modality. Int. J. Pharmaceut. Sci. Res. 2015. [Google Scholar] [CrossRef]
- Battaglia, L.; Ugazio, E. Lipid Nano-and Microparticles: An Overview of Patent-Related Research. J. Nanomater. 2019, 2019, 2834941. [Google Scholar] [CrossRef]
- Patravale, V.; Patel, P. Lipidic Nanoparticles Based Composition and Method of Formulation and Use Thereof. Patent IN3329/MUM/2010, 7 December 2010. [Google Scholar]
- Salvi, V.R.; Pawar, P. Nanostructured lipid carriers (NLC) system: A novel drug targeting carrier. J. Drug Deliv. Sci. Technol. 2019, 51, 255–267. [Google Scholar] [CrossRef]
- Tran, T.H.; Ramasamy, T.; Truong, D.H.; Choi, H.-G.; Yong, C.S.; Kim, J.O. Preparation and characterization of fenofibrate-loaded nanostructured lipid carriers for oral bioavailability enhancement. AAPS Pharmscitech 2014, 15, 1509–1515. [Google Scholar] [CrossRef]
- Yu, Y.; Feng, R.; Yu, S.; Li, J.; Wang, Y.; Song, Y.; Yang, X.; Pan, W.; Li, S. Nanostructured lipid carrier-based pH and temperature dual-responsive hydrogel composed of carboxymethyl chitosan and poloxamer for drug delivery. Int. J. Biol. Macromol. 2018, 114, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, Y.; Xie, X.; Cai, X.; Mei, X. Preparation and characterization of photo-responsive cell-penetrating peptide-mediated nanostructured lipid carrier. J. Drug Target. 2014, 22, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Kalhapure, R.S.; Mocktar, C.; Sikwal, D.R.; Sonawane, S.J.; Kathiravan, M.K.; Skelton, A.; Govender, T. Ion pairing with linoleic acid simultaneously enhances encapsulation efficiency and antibacterial activity of vancomycin in solid lipid nanoparticles. Colloids Surf. B Biointerfaces 2014, 117, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Kalhapure, R.S.; Sonawane, S.J.; Sikwal, D.R.; Jadhav, M.; Rambharose, S.; Mocktar, C.; Govender, T. Solid lipid nanoparticles of clotrimazole silver complex: An efficient nano antibacterial against Staphylococcus aureus and MRSA. Colloids Surf. B Biointerfaces 2015, 136, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Bazzaz, B.S.F.; Khameneh, B.; Zarei, H.; Golmohammadzadeh, S. Antibacterial efficacy of rifampin loaded solid lipid nanoparticles against Staphylococcus epidermidis biofilm. Microb. Pathog. 2016, 93, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Baig, M.S.; Ahad, A.; Aslam, M.; Imam, S.S.; Aqil, M.; Ali, A. Application of Box–Behnken design for preparation of levofloxacin-loaded stearic acid solid lipid nanoparticles for ocular delivery: Optimization, in vitro release, ocular tolerance, and antibacterial activity. Int. J. Biol. Macromol. 2016, 85, 258–270. [Google Scholar] [CrossRef]
- Bolla, P.K.; Kalhapure, R.S.; Rodriguez, V.A.; Ramos, D.V.; Dahl, A.; Renukuntla, J. Preparation of solid lipid nanoparticles of furosemide-silver complex and evaluation of antibacterial activity. J. Drug Deliv. Sci. Technol. 2019, 49, 6–13. [Google Scholar] [CrossRef]
- Osman, N.; Omolo, C.A.; Gannimani, R.; Waddad, A.Y.; Rambharose, S.; Mocktar, C.; Singh, S.; Parboosing, R.; Govender, T. Novel fatty acid-based pH-responsive nanostructured lipid carriers for enhancing antibacterial delivery. J. Drug Deliv. Sci. Technol. 2019, 53, 101125. [Google Scholar] [CrossRef]
- Alcantara, K.P.; Zulfakar, M.H.; Castillo, A.L. Development, characterization and pharmacokinetics of mupirocin-loaded nanostructured lipid carriers (NLCs) for intravascular administration. Int. J. Pharm. 2019, 571, 118705. [Google Scholar] [CrossRef]
- Carneiro, S.P.; Carvalho, K.V.; Soares, R.D.d.O.A.; Carneiro, C.M.; de Andrade, M.H.G.; Duarte, R.S.; David, O. Functionalized rifampicin-loaded nanostructured lipid carriers enhance macrophages uptake and antimycobacterial activity. Colloids Surf. B Biointerfaces 2019, 175, 306–313. [Google Scholar] [CrossRef]
- Ding, X.; Wang, A.; Tong, W.; Xu, F.J. Biodegradable Antibacterial Polymeric Nanosystems: A New Hope to Cope with Multidrug-Resistant Bacteria. Small 2019, 15, 1900999. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Zhang, E.; Yang, J.; Cao, Z. Strategies to improve micelle stability for drug delivery. Nano Res. 2018, 11, 4985–4998. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Ling, P.; Zhang, T. Polymeric micelles, a promising drug delivery system to enhance bioavailability of poorly water-soluble drugs. J. Drug Deliv. 2013, 2013, 340315. [Google Scholar] [CrossRef] [PubMed]
- Hill, L.E.; Gomes, C.L. Characterization of temperature and pH-responsive poly-N-isopropylacrylamide-co-polymer nanoparticles for the release of antimicrobials. Mater. Res. Express 2014, 1, 035405. [Google Scholar] [CrossRef]
- Kalhapure, R.S.; Renukuntla, J. Thermo-and pH dual responsive polymeric micelles and nanoparticles. Chem.-Biol. Interact. 2018, 295, 20–37. [Google Scholar] [CrossRef]
- Liu, L.; Venkatraman, S.S.; Yang, Y.Y.; Guo, K.; Lu, J.; He, B.; Moochhala, S.; Kan, L. Polymeric micelles anchored with TAT for delivery of antibiotics across the blood–brain barrier. Pept. Sci. 2008, 90, 617–623. [Google Scholar] [CrossRef]
- Cong, Y.; Quan, C.; Liu, M.; Liu, J.; Huang, G.; Tong, G.; Yin, Y.; Zhang, C.; Jiang, Q. Alendronate-decorated biodegradable polymeric micelles for potential bone-targeted delivery of vancomycin. J. Biomater. Sci. Polym. Ed. 2015, 26, 629–643. [Google Scholar] [CrossRef]
- Liu, Y.; Busscher, H.J.; Zhao, B.; Li, Y.; Zhang, Z.; van der Mei, H.C.; Ren, Y.; Shi, L. Surface-adaptive, antimicrobially loaded, micellar nanocarriers with enhanced penetration and killing efficiency in staphylococcal biofilms. ACS Nano 2016, 10, 4779–4789. [Google Scholar] [CrossRef]
- Huang, F.; Gao, Y.; Zhang, Y.; Cheng, T.; Ou, H.; Yang, L.; Liu, J.; Shi, L.; Liu, J. Silver-decorated polymeric micelles combined with curcumin for enhanced antibacterial activity. ACS Appl. Mater. Interfaces 2017, 9, 16880–16889. [Google Scholar] [CrossRef]
- Sheth, U.; Tiwari, S.; Bahadur, A. Preparation and characterization of anti-tubercular drugs encapsulated in polymer micelles. J. Drug Deliv. Sci. Technol. 2018, 48, 422–428. [Google Scholar] [CrossRef]
- Chen, M.; Xie, S.; Wei, J.; Song, X.; Ding, Z.; Li, X. Antibacterial Micelles with Vancomycin-Mediated Targeting and pH/Lipase-Triggered Release of Antibiotics. ACS Appl. Mater. Interfaces 2018, 10, 36814–36823. [Google Scholar] [CrossRef] [PubMed]
- Farhangi, M.; Kobarfard, F.; Mahboubi, A.; Vatanara, A.; Mortazavi, S.A. Preparation of an optimized ciprofloxacin-loaded chitosan nanomicelle with enhanced antibacterial activity. Drug Dev. Ind. Pharm. 2018, 44, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Cong, Y.; Geng, J.; Wang, H.; Su, J.; Arif, M.; Dong, Q.; Chi, Z.; Liu, C. Ureido-modified carboxymethyl chitosan-graft-stearic acid polymeric nano-micelles as a targeted delivering carrier of clarithromycin for Helicobacter pylori: Preparation and in vitro evaluation. Int. J. Biol. Macromol. 2019, 129, 686–692. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Chen, X.; Su, J.; Huang, C. Enhanced efficacy of baicalin-loaded TPGS polymeric micelles against periodontitis. Mater. Sci. Eng. C 2019, 101, 387–395. [Google Scholar] [CrossRef]
- Sandreschi, S.; Piras, A.M.; Batoni, G.; Chiellini, F. Perspectives on polymeric nanostructures for the therapeutic application of antimicrobial peptides. Nanomedicine 2016, 11, 1729–1744. [Google Scholar] [CrossRef] [PubMed]
- Kravanja, G.; Primožič, M.; Knez, Ž.; Leitgeb, M. Chitosan-based (Nano) materials for novel biomedical applications. Molecules 2019, 24, 1960. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wu, Y.; Zhao, L. Antibacterial activity and mechanism of chitosan with ultra high molecular weight. Carbohydr. Polym. 2016, 148, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Kiruthika, V.; Maya, S.; Suresh, M.K.; Kumar, V.A.; Jayakumar, R.; Biswas, R. Comparative efficacy of chloramphenicol loaded chondroitin sulfate and dextran sulfate nanoparticles to treat intracellular Salmonella infections. Colloids Surf. B Biointerfaces 2015, 127, 33–40. [Google Scholar] [CrossRef]
- Gaspar, L.M.d.A.C.; Dórea, A.C.S.; Droppa-Almeida, D.; de Mélo Silva, I.S.; Montoro, F.E.; Alves, L.L.; Macedo, M.L.H.; Padilha, F.F. Development and characterization of PLGA nanoparticles containing antibiotics. J. Nanopart. Res. 2018, 20, 289. [Google Scholar] [CrossRef]
- Lotfipour, F.; Valizadeh, H.; Milani, M.; Bahrami, N.; Ghotaslou, R. Study of antimicrobial effects of clarithromycin loaded PLGA nanoparticles against clinical strains of Helicobacter pylori. Drug Res. 2016, 66, 41–45. [Google Scholar] [CrossRef]
- Malikmammadov, E.; Tanir, T.E.; Kiziltay, A.; Hasirci, V.; Hasirci, N. PCL and PCL-based materials in biomedical applications. J. Biomater. Sci. Polym. Ed. 2018, 29, 863–893. [Google Scholar] [CrossRef] [PubMed]
- Trousil, J.; Filippov, S.K.; Hrubý, M.; Mazel, T.; Syrová, Z.; Cmarko, D.; Svidenská, S.; Matějková, J.; Kováčik, L.; Porsch, B. System with embedded drug release and nanoparticle degradation sensor showing efficient rifampicin delivery into macrophages. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.M.; El-Bisi, M.K.; Taha, G.M.; El-Alfy, E.A. Chitosan nanoparticles loaded antibiotics as drug delivery biomaterial. J. Appl. Pharm. Sci. 2015, 5, 85–90. [Google Scholar] [CrossRef]
- Kariminia, S.; Shamsipur, A.; Shamsipur, M. Analytical characteristics and application of novel chitosan coated magnetic nanoparticles as an efficient drug delivery system for ciprofloxacin. Enhanced drug release kinetics by low-frequency ultrasounds. J. Pharm. Biomed. Anal. 2016, 129, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.V.; Su, C.-H.; Velusamy, P. Ciprofloxacin loaded genipin cross-linked chitosan/heparin nanoparticles for drug delivery application. Mater. Lett. 2016, 180, 119–122. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Li, R.-Y.; Chen, J.-Y.; Chen, J.-K. Biphasic release of gentamicin from chitosan/fucoidan nanoparticles for pulmonary delivery. Carbohydr. Polym. 2016, 138, 114–122. [Google Scholar] [CrossRef]
- Kalhapure, R.S.; Jadhav, M.; Rambharose, S.; Mocktar, C.; Singh, S.; Renukuntla, J.; Govender, T. pH-responsive chitosan nanoparticles from a novel twin-chain anionic amphiphile for controlled and targeted delivery of vancomycin. Colloids Surf. B Biointerfaces 2017, 158, 650–657. [Google Scholar] [CrossRef]
- Elbi, S.; Nimal, T.; Rajan, V.; Baranwal, G.; Biswas, R.; Jayakumar, R.; Sathianarayanan, S. Fucoidan coated ciprofloxacin loaded chitosan nanoparticles for the treatment of intracellular and biofilm infections of Salmonella. Colloids Surf. B Biointerfaces 2017, 160, 40–47. [Google Scholar]
- Chavan, C.; Bala, P.; Pal, K.; Kale, S. Cross-linked chitosan-dextran sulphate vehicle system for controlled release of ciprofloxaxin drug: An ophthalmic application. OpenNano 2017, 2, 28–36. [Google Scholar] [CrossRef]
- Patel, K.K.; Tripathi, M.; Pandey, N.; Agrawal, A.K.; Gade, S.; Anjum, M.M.; Tilak, R.; Singh, S. Alginate lyase immobilized chitosan nanoparticles of ciprofloxacin for the improved antimicrobial activity against the biofilm associated mucoid P. aeruginosa infection in cystic fibrosis. Int. J. Pharm. 2019, 563, 30–42. [Google Scholar] [CrossRef]
- Baelo, A.; Levato, R.; Julián, E.; Crespo, A.; Astola, J.; Gavaldà, J.; Engel, E.; Mateos-Timoneda, M.A.; Torrents, E. Disassembling bacterial extracellular matrix with DNase-coated nanoparticles to enhance antibiotic delivery in biofilm infections. J. Control. Release 2015, 209, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Sabaeifard, P.; Abdi-Ali, A.; Soudi, M.R.; Gamazo, C.; Irache, J.M. Amikacin loaded PLGA nanoparticles against Pseudomonas aeruginosa. Eur. J. Pharm. Sci. 2016, 93, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Türeli, N.G.; Torge, A.; Juntke, J.; Schwarz, B.C.; Schneider-Daum, N.; Türeli, A.E.; Lehr, C.M.; Schneider, M. Ciprofloxacin-loaded PLGA nanoparticles against cystic fibrosis P. aeruginosa lung infections. Eur. J. Pharm. Biopharm. 2017, 117, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Greene, M.K.; Insua, J.L.; Pessoa, J.S.; Small, D.M.; Smyth, P.; McCann, A.P.; Cogo, F.; Bengoechea, J.A.; Taggart, C.C. Clearance of intracellular Klebsiella pneumoniae infection using gentamicin-loaded nanoparticles. J. Control. Release 2018, 279, 316–325. [Google Scholar] [CrossRef]
- Ernst, J.; Klinger-Strobel, M.; Arnold, K.; Thamm, J.; Hartung, A.; Pletz, M.W.; Makarewicz, O.; Fischer, D. Polyester-based particles to overcome the obstacles of mucus and biofilms in the lung for tobramycin application under static and dynamic fluidic conditions. Eur. J. Pharm. Biopharm. 2018, 131, 120–129. [Google Scholar] [CrossRef]
- Hasan, N.; Cao, J.; Lee, J.; Naeem, M.; Hlaing, S.P.; Kim, J.; Jung, Y.; Lee, B.-L.; Yoo, J.-W. PEI/NONOates-doped PLGA nanoparticles for eradicating methicillin-resistant Staphylococcus aureus biofilm in diabetic wounds via binding to the biofilm matrix. Mater. Sci. Eng. C 2019, 103, 109741. [Google Scholar] [CrossRef]
- Abdelghany, S.; Parumasivam, T.; Pang, A.; Roediger, B.; Tang, P.; Jahn, K.; Britton, W.J.; Chan, H.K. Alginate modified-PLGA nanoparticles entrapping amikacin and moxifloxacin as a novel host-directed therapy for multidrug-resistant tuberculosis. J. Drug Deliv. Sci. Technol. 2019, 52, 642–651. [Google Scholar] [CrossRef]
- Mukherjee, S.; Chowdhury, D.; Kotcherlakota, R.; Patra, S. Potential theranostics application of bio-synthesized silver nanoparticles (4-in-1 system). Theranostics 2014, 4, 316–335. [Google Scholar] [CrossRef]
- Kasithevar, M.; Periakaruppan, P.; Muthupandian, S.; Mohan, M. Antibacterial efficacy of silver nanoparticles against multi-drug resistant clinical isolates from post-surgical wound infections. Microb. Pathog. 2017, 107, 327–334. [Google Scholar] [CrossRef]
- Sathishkumar, R.; Sundaramanickam, A.; Srinath, R.; Ramesh, T.; Saranya, K.; Meena, M.; Surya, P. Green synthesis of silver nanoparticles by bloom forming marine microalgae Trichodesmium erythraeum and its applications in antioxidant, drug-resistant bacteria, and cytotoxicity activity. J. Saudi Chem. Soc. 2019, 23, 1180–1191. [Google Scholar] [CrossRef]
- Swaroop, K.; Francis, S.; Somashekarappa, H. Gamma irradiation synthesis of Ag/PVA hydrogels and its antibacterial activity. Mater. Today Proc. 2016, 3, 1792–1798. [Google Scholar] [CrossRef]
- Neihaya, H.; Zaman, H. Investigating the effect of biosynthesized silver nanoparticles as antibiofilm on bacterial clinical isolates. Microb. Pathog. 2018, 116, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, M.; Pandey, S.K.; Nain, T.; Brar, S.K.; Singh, P.; Singh, S.; Wangoo, N.; Sharma, R.K. Stabilized cationic dipeptide capped gold/silver nanohybrids: Towards enhanced antibacterial and antifungal efficacy. Colloids Surf. B Biointerfaces 2017, 158, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Carmona, M.; Gun’ko, Y.K.; Vallet-Regí, M. Mesoporous silica materials as drug delivery: “The Nightmare” of bacterial infection. Pharmaceutics 2018, 10, 279. [Google Scholar] [CrossRef]
- Jang, J.; Kim, Y. Fabrication of monodisperse silica–polymer core–shell nanoparticles with excellent antimicrobial efficacy. Chem. Commun. 2008, 4016–4018. [Google Scholar] [CrossRef]
- Dong, A.; Huang, J.; Lan, S.; Wang, T.; Xiao, L.; Wang, W.; Zhao, T.; Zheng, X.; Liu, F.; Gao, G. Synthesis of N-halamine-functionalized silica–polymer core–shell nanoparticles and their enhanced antibacterial activity. Nanotechnology 2011, 22, 295602. [Google Scholar] [CrossRef]
- Mohammed, L.; Gomaa, H.G.; Ragab, D.; Zhu, J. Magnetic nanoparticles for environmental and biomedical applications: A review. Particuology 2017, 30, 1–14. [Google Scholar] [CrossRef]
- Flores-González, M.; Talavera-Rojas, M.; Soriano-Vargas, E.; Rodríguez-González, V. Practical mediated-assembly synthesis of silver nanowires using commercial Camellia sinensis extracts and their antibacterial properties. New J. Chem. 2018, 42, 2133–2139. [Google Scholar] [CrossRef]
- Madubuonu, N.; Aisida, S.O.; Ali, A.; Ahmad, I.; Zhao, T.-K.; Botha, S.; Maaza, M.; Ezema, F.I. Biosynthesis of iron oxide nanoparticles via a composite of Psidium guavaja-Moringa oleifera and their antibacterial and photocatalytic study. J. Photochem. Photobiol. B Biol. 2019, 199, 111601. [Google Scholar] [CrossRef]
- Davarpanah, A.; Rahdar, A.; Dastnae, M.A.; Zeybek, O.; Beyzaei, H. (1−x) BaFe12O19/xCoFe2O4 hard/soft magnetic nanocomposites: Synthesis, physical characterization, and antibacterial activities study. J. Mol. Struct. 2019, 1175, 445–449. [Google Scholar] [CrossRef]
- Petros, R.A.; DeSimone, J.M. Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov. 2010, 9, 615. [Google Scholar] [CrossRef] [PubMed]
- Pati, R.; Mehta, R.K.; Mohanty, S.; Padhi, A.; Sengupta, M.; Vaseeharan, B.; Goswami, C.; Sonawane, A. Topical application of zinc oxide nanoparticles reduces bacterial skin infection in mice and exhibits antibacterial activity by inducing oxidative stress response and cell membrane disintegration in macrophages. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Hoseyni, S.J.; Manoochehri, M.; Asli, M.D. Synthesis of cobalt nanoparticles by complex demolition method using the reaction between organic ligand Schiff base and cobalt chloride by ultrasonication. Bulletin de la Société Royale des Sciences de Liège 2017, 86, 325–331. [Google Scholar]
- Gargibala satpathy, E.M. Cobalt Nanoparticle as The Antibacterial Tool: In Vitro. Int. J. Eng. Adv. Technol. 2019, 8, 3684–3687. [Google Scholar]
- Khan, S.; Ansari, A.A.; Khan, A.A.; Ahmad, R.; Al-Obaid, O.; Al-Kattan, W. In vitro evaluation of anticancer and antibacterial activities of cobalt oxide nanoparticles. JBIC J. Biol. Inorg. Chem. 2015, 20, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.T.; Ovais, M.; Ullah, I.; Ali, M.; Shinwari, Z.K.; Maaza, M. Physical properties, biological applications and biocompatibility studies on biosynthesized single phase cobalt oxide (Co3O4) nanoparticles via Sageretia thea (Osbeck.). Arab. J. Chem. 2020, 13, 606–619. [Google Scholar] [CrossRef]
- Dogra, V.; Kaur, G.; Jindal, S.; Kumar, R.; Kumar, S.; Singhal, N.K. Bactericidal effects of metallosurfactants based cobalt oxide/hydroxide nanoparticles against Staphylococcus aureus. Sci. Total Environ. 2019, 681, 350–364. [Google Scholar] [CrossRef]
- Cui, D.; Yan, C.; Miao, J.; Zhang, X.; Chen, J.; Sun, L.; Meng, L.; Liang, T.; Li, Q. Synthesis, characterization and antitumor properties of selenium nanoparticles coupling with ferulic acid. Mater. Sci. Eng. C 2018, 90, 104–112. [Google Scholar] [CrossRef]
- Xu, C.; Qiao, L.; Guo, Y.; Ma, L.; Cheng, Y. Preparation, characteristics and antioxidant activity of polysaccharides and proteins-capped selenium nanoparticles synthesized by Lactobacillus casei ATCC 393. Carbohydr. Polym. 2018, 195, 576–585. [Google Scholar] [CrossRef]
- Yazhiniprabha, M.; Vaseeharan, B. In vitro and in vivo toxicity assessment of selenium nanoparticles with significant larvicidal and bacteriostatic properties. Mater. Sci. Eng. C 2019, 103, 109763. [Google Scholar] [CrossRef] [PubMed]
- Ghotekar, S. A review on plant extract mediated biogenic synthesis of CdO nanoparticles and their recent applications. Asian J. Green Chem. 2019, 3, 187–200. [Google Scholar]
- Zahera, M.; Khan, S.A.; Khan, I.A.; Elgorban, A.M.; Bahkali, A.H.; Alghamdi, S.M.; Khan, M.S. Enhancing using glucose encapsulation, the efficacy of CdO NPs against multi-drug resistant Escherichia coli. Microb. Pathog. 2018, 119, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Simões, M.; Simões, L.C.; Vieira, M.J. A review of current and emergent biofilm control strategies. LWT Food Sci. Technol. 2010, 43, 573–583. [Google Scholar] [CrossRef]
- Sood, U.; Singh, D.N.; Hira, P.; Lee, J.-K.; Kalia, V.C.; Lal, R.; Shakarad, M. Rapid and solitary production of mono-rhamnolipid biosurfactant and biofilm inhibiting pyocyanin by a taxonomic outlier Pseudomonas aeruginosa strain CR1. J. Biotechnol. 2020, 307, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Chen, X.; Shen, Y.; Li, H.; Zou, Y.; Yuan, G.; Hu, P.; Hu, H. Mucus penetration enhanced lipid polymer nanoparticles improve the eradication rate of Helicobacter pylori biofilm. J. Control. Release 2019, 300, 52–63. [Google Scholar] [CrossRef]
- Khalid, H.F.; Tehseen, B.; Sarwar, Y.; Hussain, S.Z.; Khan, W.S.; Raza, Z.A.; Bajwa, S.Z.; Kanaras, A.G.; Hussain, I.; Rehman, A. Biosurfactant coated silver and iron oxide nanoparticles with enhanced anti-biofilm and anti-adhesive properties. J. Hazard. Mater. 2019, 364, 441–448. [Google Scholar] [CrossRef]
- Zhai, J.; Fong, C.; Tran, N.; Drummond, C.J. Nonlamellar lyotropic liquid crystalline lipid nanoparticles for the next generation of nanomedicine. ACS Nano 2019, 13, 6178–6206. [Google Scholar] [CrossRef]
- Boge, L.; Hallstensson, K.; Ringstad, L.; Johansson, J.; Andersson, T.; Davoudi, M.; Larsson, P.T.; Mahlapuu, M.; Håkansson, J.; Andersson, M. Cubosomes for topical delivery of the antimicrobial peptide LL-37. Eur. J. Pharm. Biopharm. 2019, 134, 60–67. [Google Scholar] [CrossRef]
- Mendes, C.; Meirelles, G.C.; Barp, C.G.; Assreuy, J.; Silva, M.A.; Ponchel, G. Cyclodextrin based nanosponge of norfloxacin: Intestinal permeation enhancement and improved antibacterial activity. Carbohydr. Polym. 2018, 195, 586–592. [Google Scholar] [CrossRef]
- Tran, N.; Hocquet, M.; Eon, B.; Sangwan, P.; Ratcliffe, J.; Hinton, T.M.; White, J.; Ozcelik, B.; Reynolds, N.P.; Muir, B.W. Nonlamellar lyotropic liquid crystalline nanoparticles enhance the antibacterial effects of rifampicin against Staphylococcus aureus. J. Colloid Interface Sci. 2018, 519, 107–118. [Google Scholar] [CrossRef] [PubMed]
- McArthur, M. Transcription factor decoys for the treatment and prevention of infections caused by bacteria including clostridium difficile. U.S. Patents US20140274800A1, 13 March 2013. [Google Scholar]
- González-Paredes, A.; Sitia, L.; Ruyra, A.; Morris, C.J.; Wheeler, G.N.; McArthur, M.; Gasco, P. Solid lipid nanoparticles for the delivery of anti-microbial oligonucleotides. Eur. J. Pharm. Biopharm. 2019, 134, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Mamusa, M.; Barbero, F.; Montis, C.; Cutillo, L.; Gonzalez-Paredes, A.; Berti, D. Inclusion of oligonucleotide antimicrobials in biocompatible cationic liposomes: A structural study. J. Colloid Interface Sci. 2017, 508, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Xue, X.; Hou, Z.; Zhou, Y.; Meng, J.; Luo, X. Antisense antibiotics: A brief review of novel target discovery and delivery. Curr. Drug Discov. Technol. 2010, 7, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.-Y.; Mao, X.-G.; Zhou, Y.; Chen, Z.; Hu, Y.; Hou, Z.; Li, M.-K.; Meng, J.-R.; Luo, X.X. Advances in the delivery of antisense oligonucleotides for combating bacterial infectious diseases. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 745–758. [Google Scholar] [CrossRef]
- Hegarty, J.P.; Stewart, D.B. Advances in therapeutic bacterial antisense biotechnology. Appl. Microbiol. Biotechnol. 2018, 102, 1055–1065. [Google Scholar] [CrossRef]
- Rai, M.; Paralikar, P.; Jogee, P.; Agarkar, G.; Ingle, A.P.; Derita, M.; Zacchino, S. Synergistic antimicrobial potential of essential oils in combination with nanoparticles: Emerging trends and future perspectives. Int. J. Pharm. 2017, 519, 67–78. [Google Scholar] [CrossRef]
- Rodenak-Kladniew, B.; Montoto, S.S.; Sbaraglini, M.; Di Ianni, M.; Ruiz, M.; Talevi, A.; Alvarez, V.A.; Durán, N.; Castro, G.R.; Islan, G.A. Hybrid Ofloxacin/eugenol co-loaded solid lipid nanoparticles with enhanced and targetable antimicrobial properties. Int. J. Pharm. 2019, 569, 118575. [Google Scholar] [CrossRef]
- Gao, H.; Cheng, T.; Zhang, Y.; Liu, J.; Huang, F.; Yang, C.; Shi, L.; Liu, J. A charge-adaptive nanosystem for prolonged and enhanced in vivo antibiotic delivery. Chem. Coummun. 2016, 52, 6265–6268. [Google Scholar]
- Li, Y.; Liu, G.; Wang, X.; Hu, J.; Liu, S. Enzyme-Responsive Polymeric Vesicles for Bacterial-Strain-Selective Delivery of Antimicrobial Agents. Angew. Chem. Int. Ed. 2016, 55, 1760–1764. [Google Scholar] [CrossRef]
- Augustine, R.; Kalva, N.; Kim, H.A.; Zhang, Y.; Kim, I. pH-Responsive Polypeptide-Based Smart Nano-Carriers for Theranostic Applications. Molecules 2019, 24, 2961. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Algburi, A.; Wang, N.; Kholodovych, V.; Oh, D.O.; Chikindas, M.; Uhrich, K.E. Self-assembled cationic amphiphiles as antimicrobial peptides mimics: Role of hydrophobicity, linkage type, and assembly state. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Fillion, M.; Valois-Paillard, G.; Lorin, A.; Noël, M.; Voyer, N.; Auger, M. Membrane interactions of synthetic peptides with antimicrobial potential: Effect of electrostatic interactions and amphiphilicity. Probiotics Antimicrob. Proteins 2015, 7, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, H.; Bahador, A.; Pourhajibagher, M.; Alikhani, M.Y. Antimicrobial photodynamic therapy: An effective alternative approach to control bacterial infections. J. Lasers Med. Sci. 2018, 9, 154. [Google Scholar] [CrossRef]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy–what we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef]
- Ghosh, S.; Qi, R.; Carter, K.A.; Zhang, G.; Pfeifer, B.A.; Lovell, J.F. Loading and releasing ciprofloxacin in photoactivatable liposomes. Biochem. Eng. J. 2019, 141, 43–48. [Google Scholar] [CrossRef]
- Jeong, S.; Lee, J.; Im, B.N.; Park, H.; Na, K. Combined photodynamic and antibiotic therapy for skin disorder via lipase-sensitive liposomes with enhanced antimicrobial performance. Biomaterials 2017, 141, 243–250. [Google Scholar] [CrossRef]
- Domalaon, R.; Ammeter, D.; Brizuela, M.; Gorityala, B.K.; Zhanel, G.G.; Schweizer, F. Repurposed antimicrobial combination therapy: Tobramycin-ciprofloxacin hybrid augments activity of the anticancer drug mitomycin C against multidrug-resistant Gram-negative bacteria. Front. Microbiol. 2019, 10, 1556. [Google Scholar] [CrossRef]
- Xu, X.; Xu, L.; Yuan, G.; Wang, Y.; Qu, Y.; Zhou, M. Synergistic combination of two antimicrobial agents closing each other’s mutant selection windows to prevent antimicrobial resistance. Sci. Rep. 2018, 8, 7237. [Google Scholar] [CrossRef]
- Walvekar, P.; Gannimani, R.; Govender, T. Combination drug therapy via nanocarriers against infectious diseases. Eur. J. Pharm. Sci. 2019, 127, 121–141. [Google Scholar] [CrossRef]
- Chai, G.; Park, H.; Yu, S.; Zhou, F.; Li, J.; Xu, Q.; Zhou, Q.T. Evaluation of co-delivery of colistin and ciprofloxacin in liposomes using an in vitro human lung epithelial cell model. Int. J. Pharm. 2019, 569, 118616. [Google Scholar] [CrossRef]
- Ye, T.; Sun, S.; Sugianto, T.D.; Tang, P.; Parumasivam, T.; Chang, Y.K.; Astudillo, A.; Wang, S.; Chan, H.-K. Novel combination proliposomes containing tobramycin and clarithromycin effective against Pseudomonas aeruginosa biofilms. Int. J. Pharm. 2018, 552, 130–138. [Google Scholar] [CrossRef]
- Fumakia, M.; Ho, E.A. Nanoparticles encapsulated with LL37 and serpin A1 promotes wound healing and synergistically enhances antibacterial activity. Mol. Pharm. 2016, 13, 2318–2331. [Google Scholar] [CrossRef]
- Shimanovich, U.; Lipovsky, A.; Eliaz, D.; Zigdon, S.; Knowles, T.P.; Nitzan, Y.; Michaeli, S.; Gedanken, A. Tetracycline Nanoparticles as Antibacterial and Gene-Silencing Agents. Adv. Healthc. Mater. 2015, 4, 723–728. [Google Scholar] [CrossRef]
- Morakul, B.; Suksiriworapong, J.; Chomnawang, M.T.; Langguth, P.; Junyaprasert, V.B. Dissolution enhancement and in vitro performance of clarithromycin nanocrystals produced by precipitation–lyophilization–homogenization method. Eur. J. Pharm. Biopharm. 2014, 88, 886–896. [Google Scholar] [CrossRef] [PubMed]
- Havel, H.A. Where are the nanodrugs? An industry perspective on development of drug products containing nanomaterials. AAPS J. 2016, 18, 1351–1353. [Google Scholar] [CrossRef] [PubMed]
- Romero-Calle, D.; Guimarães Benevides, R.; Góes-Neto, A.; Billington, C. Bacteriophages as Alternatives to Antibiotics in Clinical Care. Antibiotics 2019, 8, 138. [Google Scholar] [CrossRef] [PubMed]
- Chadha, P.; Katare, O.P.; Chhibber, S. Liposome loaded phage cocktail: Enhanced therapeutic potential in resolving Klebsiella pneumoniae mediated burn wound infections. Burns 2017, 43, 1532–1543. [Google Scholar] [CrossRef]
- Haworth, C.S.; Bilton, D.; Chalmers, J.D.; Davis, A.M.; Froehlich, J.; Gonda, I.; Thompson, B.; Wanner, A.; O’Donnell, A.E. Inhaled liposomal ciprofloxacin in patients with non-cystic fibrosis bronchiectasis and chronic lung infection with Pseudomonas aeruginosa (ORBIT-3 and ORBIT-4): Two phase 3, randomised controlled trials. Lancet Respir. Med. 2019, 7, 213–226. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Phase 3 Study With Ciprofloxacin Dispersion for Inhalation in Non-CF Bronchiectasis (ORBIT-3). Available online: https://clinicaltrials.gov/ct2/show/NCT01515007 (accessed on 21 November 2019).
- ClinicalTrials.gov. Liposomal Amikacin for Inhalation (LAI) for Nontuberculous Mycobacteria. Available online: https://clinicaltrials.gov/ct2/show/NCT01315236 (accessed on 4 November 2019).
- ClinicalTrials.gov. Study of Dose Escalation of Liposomal Amikacin for Inhalation (ARIKAYCE™)—Extension Phase. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03905642 (accessed on 18 November 2019).
- ClinicalTrials.gov. Liposomal Amikacin for Inhalation (LAI) in the Treatment of Mycobacterium Abscessus Lung Disease. Available online: https://clinicaltrials.gov/ct2/show/NCT03038178 (accessed on 4 November 2019).
- ClinicalTrials.gov. Extension Study of Liposomal Amikacin for Inhalation in Cystic Fibrosis (CF) Patients With Chronic Pseudomonas Aeruginosa (Pa) Infection. Available online: https://clinicaltrials.gov/ct2/show/NCT01316276 (accessed on 12 November 2019).
- Mullard, A. FDA approves antitoxin antibody. Nat. Rev. Drug Discov. 2016, 15, 811. [Google Scholar] [CrossRef]
- Azeredo da Silveira, S.; Perez, A. Improving the fate of severely infected patients: The promise of anti-toxin treatments and superiority trials. Expert Rev. Anti-Infect. Ther. 2017, 15, 973–975. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Laterre, P.-F.; Colin, G.; Dequin, P.-F.; Dugernier, T.; Boulain, T.; Azeredo da Silveira, S.; Lajaunias, F.C.; Perez, A.; François, B. CAL02, a novel antitoxin liposomal agent, in severe pneumococcal pneumonia: A first-in-human, double-blind, placebo-controlled, randomised trial. Lancet Infect. Dis. 2019, 19, 620–630. [Google Scholar] [CrossRef]
- Molchanova, N.; Hansen, P.R.; Franzyk, H. Advances in Development of Antimicrobial Peptidomimetics as Potential Drugs. Molecules 2017, 22, 1430. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Study to Evaluate Efficacy of LAI When Added to Multi-drug Regimen Compared to Multi-drug Regimen Alone (CONVERT). Available online: https://clinicaltrials.gov/ct2/show/NCT02344004 (accessed on 25 October 2019).
- ClinicalTrials.gov. Study of the Clinical Effectiveness of a Human Monoclonal Antibody to C. Difficile Toxin A and Toxin B in Patients With Clostridium Difficile Associated Disease. Available online: https://clinicaltrials.gov/ct2/show/NCT00350298?term=anti+toxin&draw=3&rank=13 (accessed on 16 November 2019).
- Ghobrial, O.G.; Derendorf, H.; Hillman, J.D. Pharmacodynamic activity of the lantibiotic MU1140. Int. J. Antimicrob. Agents 2009, 33, 70–74. [Google Scholar] [CrossRef]
- Andrä, J.; Jakovkin, I.; Grötzinger, J.; Hecht, O.; Krasnosdembskaya, A.D.; Goldmann, T.; Gutsmann, T.; Leippe, M. Structure and mode of action of the antimicrobial peptide arenicin. Biochem. J. 2008, 410, 113–122. [Google Scholar] [CrossRef]
- Gebhart, D.; Lok, S.; Clare, S.; Tomas, M.; Stares, M.; Scholl, D.; Donskey, C.; Lawley, T.; Govoni, A. Modified R-Type Bacteriocin Specifically Targeting Clostridium Difficile Prevents Colonization of Mice without Affecting Gut Microbiota Diversity. mBio 2015, 6. [Google Scholar] [CrossRef]
- Crowther, G.S.; Baines, S.D.; Todhunter, S.L.; Freeman, J.; Chilton, C.H.; Wilcox, M.H. Evaluation of NVB302 versus vancomycin activity in an in vitro human gut model of Clostridium difficile infection. J. Antimicrob. Chemother. 2012, 68, 168–176. [Google Scholar] [CrossRef]
- van der Velden, W.J.F.M.; van Iersel, T.M.P.; Blijlevens, N.M.A.; Donnelly, J.P. Safety and tolerability of the antimicrobial peptide human lactoferrin 1-11 (hLF1-11). BMC Med. 2009, 7, 44. [Google Scholar] [CrossRef]
- Zhang, W.; Li, Y.; Qian, G.; Wang, Y.; Chen, H.; Wu, Z.-H.; Liu, F.; Shen, Y.; Du, L. Identification and Characterization of the Anti-Methicillin-Resistant Staphylococcus aureus WAP-8294A2 Biosynthetic Gene Cluster from Lysobacter enzymogenes OH11. Antimicrob. Agents Chemother. 2011, 55, 5581–5589. [Google Scholar] [CrossRef]
- Kaplan, C.; Sim, J.; Shah, K.; Kaplan, A.; Shi, W.; Eckert, R. Selective Membrane Disruption: Mode of Action of C16G2, a Specifically Targeted Antimicrobial Peptide. Antimicrob. Agents Chemother. 2011, 55, 3446–3452. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. A Study of DPK-060 to Investigate Clinical Safety and Efficacy in Patients With Acute External Otitis. Available online: https://clinicaltrials.gov/ct2/show/NCT01447017 (accessed on 10 December 2019).
- ClinicalTrials.gov. A Phase II Study to Evaluate the Efficacy and Safety of Two Doses of LTX-109 in Impetigo. Available online: https://clinicaltrials.gov/ct2/show/NCT01803035 (accessed on 10 December 2019).
- Nilsson, A.C.; Janson, H.; Wold, H.; Fugelli, A.; Andersson, K.; Håkangård, C.; Olsson, P.; Olsen, W.M. LTX-109 is a novel agent for nasal decolonization of methicillin-resistant and -sensitive Staphylococcus aureus. Antimicrob. Agents Chemother. 2015, 59, 145–151. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Phase III Efficacy and Safety Study of AB103 in the Treatment of Patients With Necrotizing Soft Tissue Infections (ACCUTE). Available online: https://clinicaltrials.gov/ct2/show/NCT02469857 (accessed on 11 December 2019).
- Knight-Connoni, V.; Mascio, C.; Chesnel, L.; Silverman, J. Discovery and development of surotomycin for the treatment of Clostridium difficile. J. Ind. Microbiol. Biotechnol. 2016, 43, 195–204. [Google Scholar] [CrossRef]
- Stiefel, U.; Davis, N.; Helfand, M.; Donskey, C. Efficacy of Oral Ramoplanin for Inhibition of Intestinal Colonization by Vancomycin-Resistant Enterococci in Mice. Antimicrob. Agents Chemother. 2004, 48, 2144–2148. [Google Scholar] [CrossRef] [PubMed]
- Narang, A.; Chang, R.-K.; Hussain, M.A. Pharmaceutical development and regulatory considerations for nanoparticles and nanoparticulate drug delivery systems. J. Pharm. Sci. 2013, 102, 3867–3882. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; De Matos, M.B.C.; Metselaar, J.M.; Storm, G. Current trends and challenges in the clinical translation of nanoparticulate nanomedicines: Pathways for translational development and commercialization. Front. Pharmacol. 2018, 9, 790. [Google Scholar] [CrossRef]
- Tinkle, S.; McNeil, S.E.; Mühlebach, S.; Bawa, R.; Borchard, G.; Barenholz, Y.; Tamarkin, L.; Desai, N. Nanomedicines: Addressing the scientific and regulatory gap. Ann. N. Y. Acad. Sci. 2014, 1313, 35–56. [Google Scholar] [CrossRef]
- Kumar Teli, M.; Mutalik, S.; Rajanikant, G.K. Nanotechnology and nanomedicine: Going small means aiming big. Curr. Pharm. Des. 2010, 16, 1882–1892. [Google Scholar] [CrossRef]
- Accomasso, L.; Cristallini, C.; Giachino, C. Risk assessment and risk minimization in nanomedicine: A need for predictive, alternative, and 3Rs strategies. Front. Pharmacol. 2018, 9, 228. [Google Scholar] [CrossRef]
- Murday, J.S.; Siegel, R.W.; Stein, J.; Wright, J.F. Translational nanomedicine: Status assessment and opportunities. Nanomed. Nanotechnol. Biol. Med. 2009, 5, 251–273. [Google Scholar] [CrossRef]
- Hafner, A.; Lovric, J.; Lakos, G.P.; Pepic, I. Nanotherapeutics in the EU: An overview on current state and future directions. Int. J. Nanomed. 2014, 9, 1005. [Google Scholar]

| Liposome Type | Lipid Composition | Anti-Bacterial Drug/Nutraceutical Agents | Size | Biofilm | Findings | Reference Year |
|---|---|---|---|---|---|---|
| Conventional Liposomes | DPPC (Dipalmitoylphosphatidylcholine) | Calcifediol (25 (OH)D) | 151.2 ± 0.3 nm | P. aeruginosa |
| 2017 [37] |
| Conventional Liposomes | Egg yolk soybean L-α phosphatidyl choline |
| 280 nm–1.76 µm | Ocular post-surgery infections |
| 2018 [78] |
| Conventional Liposomes | pH-sensitive lipids (PSLs):Phosphatidylcholine (PCS 100):Cholesterol (1:3:1 w/w/w) | Vancomycin | 99.38 ± 0.59–105.60 ± 5.38 nm | MRSA |
| 2018 [79] |
| Lipoid Lipoid + SDch Lipoid + PG DPPCT (Dipalmitoylphosphatidylcholine), DODAB (Dimethyldioctadecylammonium bromide) | Azithromycin | 132–217 nm | MRSA |
| 2018 [35] |
| Conventional Liposomes | Hydrogenated soy phosphatidylcholine and cholesterol (7:3 w/w) | Ciprofloxacin nanocrystals | ∼130 nm | P. aeruginosa. |
| 2019 [80] |
| Conventional Liposomes | P90 G (Phospholipon 90 G), cholesterol |
| ∼200 nm | K. pneumoniae |
| 2019 [81] |
| Conventional Liposomes | Phospholipon 90 G | Biosurfactants isolated from Lactobacillus gasseri Bc9 | <200 nm | MRSA |
| 2019 [82] |
| Conventional Liposomes | Egg yolk Lecithin | Cinnamaldehyde | 75–92.14 nm | S. aureus |
| 2019 [83] |
| Fusogenic Liposomes | Dope/Dppc/CHe MS (4:2:4 molar ratios) | Fusidic acid | 98.77–99 nm | S. epidermidis (Staphylococcus epidermidis), Acinetobacter baumannii |
| 2015 [84] |
| Surface-Modified Liposomes | PC: DSPE PEG: Chol: SA (6.5:0.5:2:1 mole %) | β-Lapachone | 88.7–112.4 nm | MRSA C. neoformans (Cryptococcus neoformans) |
| 2015 [85] |
| Conventional liposomes Surface-Modified Liposomes Immobilized at surface of chitosan nanofiber mesh | Dipalmitoylphosphatidylcholine (DPPC)- cholesterol DPPC, cholesterol, DSPE-PEG-Mal and PE-Rho). Mal; (maleimide), Rho; (Rhamnolipids) | Gentamicin | 126.25–140.26 nm | E. coli P. aeruginosa S. aureus |
| 2015 [86] |
| Surface- Modified Liposomes | Phospholipid, cholesterol, tween 80, vitamin E (6:1:1.8:0.12 mass ratios). | Gallic acid | 153.2 ± 1.4 nm | E. Coli S. aureus |
| 2019 [87] |
| Surface- Modified Liposomes | Phospholipids + Rhamnolipids (1:0, 10:1, 5:1, 2:1, 1:1 w/w). | Curcumin | 46.4–251 nm | - |
| 2019 [88] |
| Surface-Modified Liposomes |
| Ciprofloxacin Betamethasone | ∼100 nm | Aggregatibacter actinomycetemcomitans |
| 2020 [44] |
| Liposomes-in-Hydrogel | Phospholipids (PC) | Isoniazide N-Dodecanoyl isonicotinohydrazide (DINH) | ∼130 nm | M. tuberculosis |
| 2019 [89] |
| Reactive Liposomes Encapsulating Enzyme (s) | DPPC, cholesterol, hexadecylamine | Endolysins | 303 nm |
|
| 2019 [63] |
| Lipid Composition | Drug | Size | Biofilm | Findings | Reference Year |
|---|---|---|---|---|---|
| SLNs | |||||
| Compritol 888 ATO (a lipid excipient) | Vancomycin- Linoleic acid complex | 102.7 ± 1.01 | S. aureus & MRSA |
| 2014 [101] |
| Compritol 888 ATO | Clotrimazole- silver complex | 124.1 ± 2.5 nm | S. aureus & MRSA |
| 2015 [102] |
| Glyceryl monostearate Precirol Stearylamine | Rifampin | 101 ± 4.7 nm | S. epidermidis |
| 2016 [103] |
| Stearic acid | Levofloxacin | 237.82 nm | S. aureus E. coli |
| 2016 [104] |
| Glyceryl behenate Tripalmitin Stearic acid | Clarithromycin | 318–526 nm. | S. aureus |
| 2019 [90] |
| Glycerol monostearate | Furosemide-silver complex (Ag-FSE) | 129.8 ± 38.5 nm | P. aeruginosa S. aureus |
| 2019 [105] |
| NLCs | |||||
| pH responsive NLC (Stearic acid and oleic acid) | Vancomycin | 225.2 ± 9.1 nm | S. aureus (Resistant and Sensitive) |
| 2019 [106] |
| Cetyl palmitate and caprylic acid | Mupirocin | 99.8–235 nm | MRSA |
| 2019 [107] |
Stearic acid and oleic acid
| Rifampicin |
| M. tuberculosis |
| 2019 [108] |
| Composition | Drug | Size | Biofilm | Findings | Reference Year |
|---|---|---|---|---|---|
| Cholesterol conjugated Poly (ethylene glycol) and anchored with transcriptional activator TAT (a trans-activator of transcription) peptide | Ciprofloxacin | 180 nm |
|
| 2008 [114] |
| Poly (lactic acid-co-glycolic acid)-block-poly (ethylene glycol)-alendronate copolymer | Vancomycin | 39.62–55.08 nm | S. aureus |
| 2015 [115] |
| Mixed-shell-polymeric-micelles consisting of a hydrophilic PEG-shell and pH-responsive poly (β-amino ester) | Triclosan | 160 nm | S. aureus |
| 2016 [116] |
| Silver decorated amphiphilic diblock copolymers, poly (ε-caprolactone)-block-poly(aspartic acid) | Curcumin | 90–95 nm | P. aeruginosa S. aureus |
| 2017 [117] |
| Ethylene oxide-propylene oxide triblock copolymers, Pluronics® (P84, P85, P103, P105, P123 and F127) | Rifampicin and Isoniazid | - | M. tuberculosis |
| 2018 [118] |
| Amphiphilic poly (ethylene glycol)-poly(ε-caprolactone) copolymers conjugated with vancomycin. | Ciprofloxacin | 77 nm | P. aeruginosa |
| 2018 [119] |
| Fatty acid grafted chitosan conjugates nanomicelles | Ciprofloxacin | 260 nm | P. aeroginosa K. pneumoniae S. pneumoniae |
| 2018 [120] |
| Carboxy methyl chitosan hydrophobically modified with stearic acid and conjugated with urea | Clarithromycin | 200 nm | H. pylori |
| 2019 [121] |
| D-α-tocopherol polyethylene glycol 1000 succinate polymeric micelles | Baicalin | 14.05 ± 4.52 nm, | E. coli |
| 2019 [122] |
| Type of Polymeric Nanoparticles | Drug | Size | Biofilm | Findings | Reference Year |
|---|---|---|---|---|---|
| Natural polymers | |||||
| Chitosan | Ciprofloxacin Chlortetracycline HCL. Gentamycin sulfate. | S. aureus E. coli |
| 2015 [131] | |
| pH-responsive chitosan coated iron oxide NPs | Ciprofloxacin | 30–80 nm | urinary tract and intestinal infections |
| 2016 [132] |
| Genipin cross-linked chitosan/heparin NPs | Ciprofloxacin | 250 nm | E. coli MTCC 443 |
| 2016 [133] |
| Chitosan/fucoidan NPs | Gentamicin | 270–300 nm | K. pneumoniae |
| 2016 [134] |
| pH-responsive chitosan nanoparticles with new anionic gemini surfactant (AGS) | Vancomycin | 220.57 ± 5.9 nm | MRSA |
| 2017 [135] |
| Chitosan nanoparticles and fucoidan coated chitosan NPs | Ciprofloxacin | Chitosan NPs: 124 ± 7 nm. Fucoidan coated chitosan CNPs: 320 ± 18 nm. | Salmonella |
| 2017 [136] |
| Chitosan-Dextran sulphate NPs | Ciprofloxacin | 350 nm | Gm +ve and Gm -ve ophthalmic microorganisms. |
| 2017 [137] |
| Alginate lyase functionalized chitosan NPs | Ciprofloxacin | 205.5 ± 9.0 nm | P. aeruginosa |
| 2019 [138] |
| Synthetic polymers | |||||
| PLGA functionalized with DNase I | Ciprofloxacin | 251.9 nm | P. aeruginosa |
| 2015 [139] |
| PLGA | Amikacin | 447 ± 7 | P. aeruginosa |
| 2016 [140] |
| PLGA | Ciprofloxacin-SDS complex (ciprofloxacin complex loaded PLGA) | 190.4 ± 28.6 nm | P. aeruginosa |
| 2017 [141] |
| PLGA (poly(lactic-co-glycolic acid) | Gentamicin | 227 nm | K. pneumonia |
| 2018 [142] |
| PLGA and PEG-PLGA di-block NPs | Tobramycin | NPs: 225–231 nm MPs: 896–902 nm | P. aeruginosa and Burkholderia cepacia complex (Bcc) |
| 2018 [143] |
| Polyethylenimine/diazeniumdiolate (PEI/NONOate)-doped PLGA nanoparticles | Nitric oxide (NO) | 240 ± 20 | MRSA |
| 2019 [144] |
| Alginate modified-PLGA nanoparticles | Amikacin and moxifloxacin | Alginate coated PLGA NPs: 640 ± 32 nm Alginate entrapped PLGA NPs: 312–365 nm | M. tuberculosis (H37Ra) |
| 2019 [145] |
| Antibiotic | Clinical Trial | Medical Condition/Indication | Trial Phase | Intervention Treatment |
|---|---|---|---|---|
| Ciprofloxacin | Inhaled ciprofloxacin loaded-liposome: Once a day management of respiratory infections [54]. | P. aeruginosa | Phase 1 | Ciprofloxacin |
| Ciprofloxacin | Inhaled ciprofloxacin loaded-liposome: Once a day management of respiratory infections [54]. | P. aeruginosa | Phase 2a | Ciprofloxacin |
| Ciprofloxacin | Inhaled ciprofloxacin loaded-liposome in patients with non-cystic fibrosis bronchiectasis and chronic lung infection with Pseudomonas aeruginosa (ORBIT-3 and ORBIT-4): two phase 3, randomised controlled trials [208]. | Bronchiectasis and Chronic P. Aeruginosa Infection | Phase 3 | Inhaled Liposomal Ciprofloxacin |
| Ciprofloxacin | Phase 3 Study with Ciprofloxacin Dispersion for Inhalation in Non-Cystic Fibrosis Bronchiectasis (ORBIT-3) [209]. | Non-Cystic Fibrosis Bronchiectasis | Phase 3 | Ciprofloxacin dispersion for inhalation (Liquid mixture of liposomally encapsulated and un encapsulated ciprofloxacin) Placebo: Liquid formulation of empty liposomes. |
| Amikacin | Liposomal Amikacin for Inhalation (LAI) for Nontuberculous Mycobacteria [210]. | Mycobacterium Infections, Nontuberculous | Phase 2 | Liposomal amikacin for inhalation (LAI) Drug: placebo |
| Amikacin | Extension Study of Liposomal Amikacin for Inhalation in Cystic Fibrosis (CF) Patients with Chronic Pseudomonas Aeruginosa (Pa) Infection [213]. | Cystic Fibrosis Patients with Chronic Pseudomonas aeruginosa Infection | Phase 3 | Amikacin |
| Amikacin | Inhaled amikacin loaded-liposome for treating Mycobacterium Abscesses Lung Disease [212]. | Mycobacterium Infections, Nontuberculous Mycobacteria, Atypical | Phase 2 | Liposomal amikacin for inhalation (LAI) plus multi-drug regimen |
| Amikacin | Study to Evaluate Efficacy of inhaled amikacin loaded-liposome combined with multi-drug regimen, Compared to Multi-drug Regimen Alone (CONVERT) [218]. | Mycobacterium Infections, Nontuberculous | Phase 3 | Liposomal Amikacin for Inhalation, 590 mg |
| Amikacin | Study of Dose Escalation of Liposomal Amikacin for Inhalation (ARIKAYCE™)-Extension Phase [211]. | Cystic Fibrosis | Phase 2 | Drug: Arikayce™ |
| Biological: CAL02 | CAL02; a liposomal adjunctive anti-toxin therapy in infections. A new therapeutic approach for severe community-acquired pneumonia [216]. | Severe community-acquired pneumonia | Phase 2 and 3 | CAL02 anti-toxin |
| Biological: GS-CDA1 Biological: MDX-1388 | Study of the Clinical Effectiveness of a Human Monoclonal Antibody to C. Difficile Toxin A and Toxin B in Patients with Clostridium Difficile Associated Disease [219]. | Clostridium Difficile Associated Disease | Phase 2 | Biological: (GS-CDA1) Biological: MDX-1388 |
| Antimicrobial Peptides | Medical Condition/Indication | Clinical Trial Phase | Antimicrobial Peptides Source |
|---|---|---|---|
| Mutacin 1140 (MU1140) [220] | Gm +ve bacteria (MRSA, C. difficile (Clostridium difficile)) | Preclinical | Streptococcus mutans |
| lipohexapeptides 1345 (HB1345) [217] | Broad-spectrum antibiotic, acne | Preclinical | Lipopeptide |
| Novarifyn (NP432) [50] | MRSA, P. aeruginosa, C. difficile, A. baumannii, E. coli | Preclinical | Synthetic antimicrobial |
| Arenicin (AP139) [221] | Gm−ve bacteria, UTI | Preclinical | Lugworm Arenicol marina |
| Arenicin (AP138) [217] | MRSA implant infections | Preclinical | Arenicin analog |
| Arenicin (AP114) [217] | C. difficile | Preclinical | Arenicin analog |
| Avidocin and purocin [222] | Gm+ve and Gm−ve bacteria | Preclinical | Modified R-type bacteriocin |
| Novacta biosystems (NVB-302) [223] | C. difficile | Phase 1 | Lantibiotic |
| Human lactoferrin (hlf1-11) [224] | Infection following transplantation | Phase 1 and phase 2 | Lactoferricin analog |
| (a potent cyclic lipodepsipeptides antibiotic) Wap-8294A2 [225] | Gm+ve bacteria (VRE and MRSA) | Phase 1 and phase 2 | Lysobactor spp. |
| The specifically targeted antimicrobial peptide (C16G2) [226] | Prevention of tooth decay caused by Streptococcus mutans | Phase 2 | Synthetic peptide |
| Antimicrobial Peptide (DPK-060) [227] | Acute external otitis | Phase 2 | Human protein kininogen |
| LTX-109 (Lytixar) [228,229] | Nasal decolonization of MRSA Impetigo | Phase 1 & 2 Phase 2 | Synthetic peptidomimetic |
| p2TA (AB 103) [230] (A CD28 mimetic peptide) | Necrotizing soft tissue infections | Phase 3 | Synthetic peptide |
| Surotomycin [231] | C. difficile (diarrhea) | Phase 3 | Cyclic lipopeptide |
| Ramoplanin (NTI-851) [232] | C. difficile | Phase 2 | Actinoplanes spp |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eleraky, N.E.; Allam, A.; Hassan, S.B.; Omar, M.M. Nanomedicine Fight against Antibacterial Resistance: An Overview of the Recent Pharmaceutical Innovations. Pharmaceutics 2020, 12, 142. https://doi.org/10.3390/pharmaceutics12020142
Eleraky NE, Allam A, Hassan SB, Omar MM. Nanomedicine Fight against Antibacterial Resistance: An Overview of the Recent Pharmaceutical Innovations. Pharmaceutics. 2020; 12(2):142. https://doi.org/10.3390/pharmaceutics12020142
Chicago/Turabian StyleEleraky, Nermin E., Ayat Allam, Sahar B. Hassan, and Mahmoud M. Omar. 2020. "Nanomedicine Fight against Antibacterial Resistance: An Overview of the Recent Pharmaceutical Innovations" Pharmaceutics 12, no. 2: 142. https://doi.org/10.3390/pharmaceutics12020142
APA StyleEleraky, N. E., Allam, A., Hassan, S. B., & Omar, M. M. (2020). Nanomedicine Fight against Antibacterial Resistance: An Overview of the Recent Pharmaceutical Innovations. Pharmaceutics, 12(2), 142. https://doi.org/10.3390/pharmaceutics12020142