Conjugation of Native-Like HIV-1 Envelope Trimers onto Liposomes Using EDC/Sulfo-NHS Chemistry: Requirements and Limitations
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Buffers
2.3. Liposome Preparation
2.3.1. Liposome Composition
2.3.2. Thin-Film Hydration
2.3.3. Cross-Flow Injection
2.3.4. Purification by Tangential Flow Filtration (TFF)
2.4. Particle Size and Zeta Potential
2.4.1. General
2.4.2. Titrations
2.5. Quantification of Peptides and Lipids by HPLC
2.6. Peptide Recovery and Peptide Encapsulation Efficiency
2.7. Protein Bioconjugation
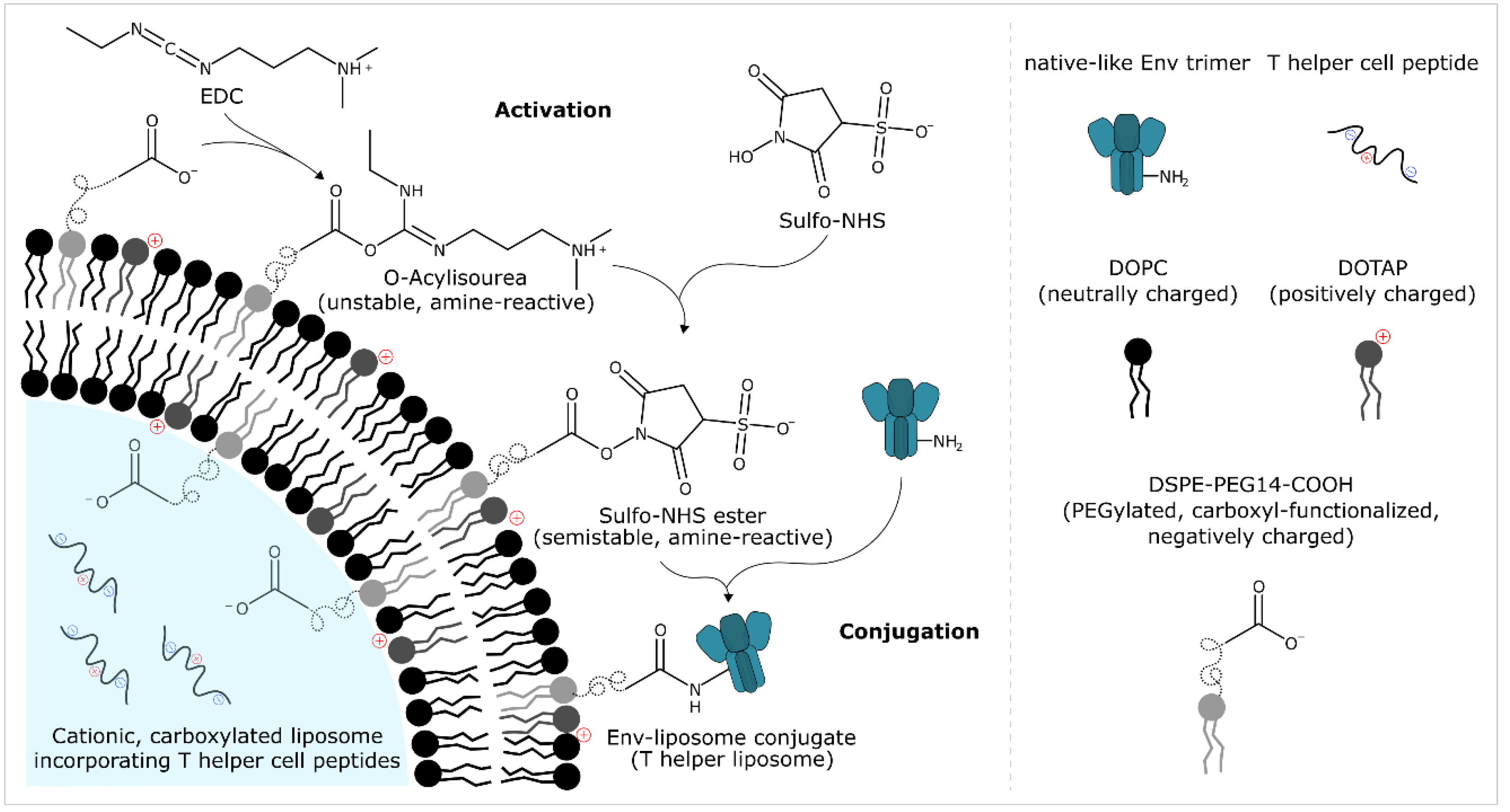
2.7.1. General
2.7.2. Activation
2.7.3. Removal/Inactivation of Excess Activation Reagents
2.7.4. Conjugation
2.8. Ultracentrifugation
2.9. Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.10. Enzyme-Linked Immunosorbent Assay (ELISA): Quantitative Analyses and Antigenicity Analyses
2.11. Antibody Biotinylation
2.12. Conjugation Efficiency
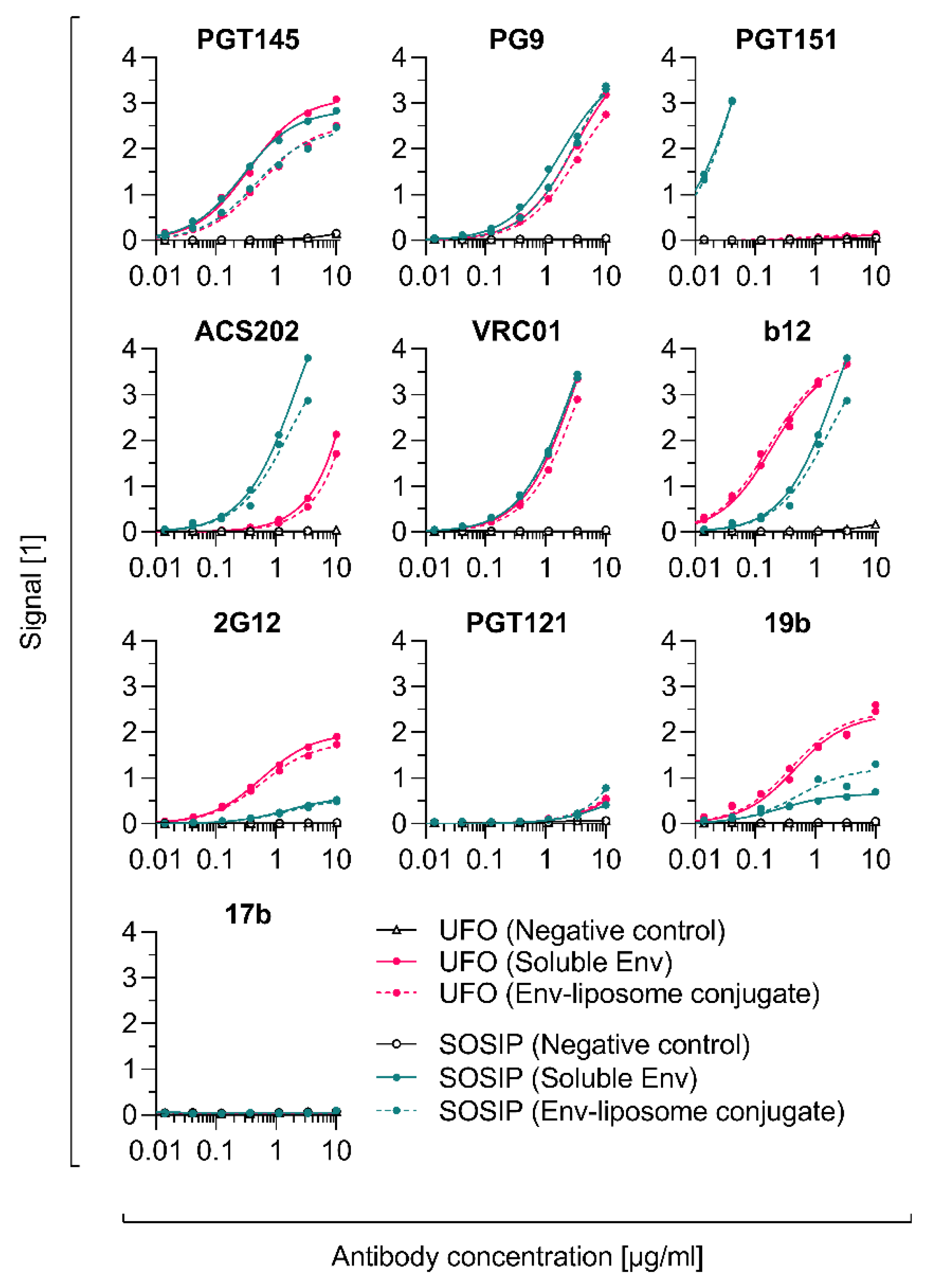
2.13. PGT145 Reactivity
3. Results and Discussion
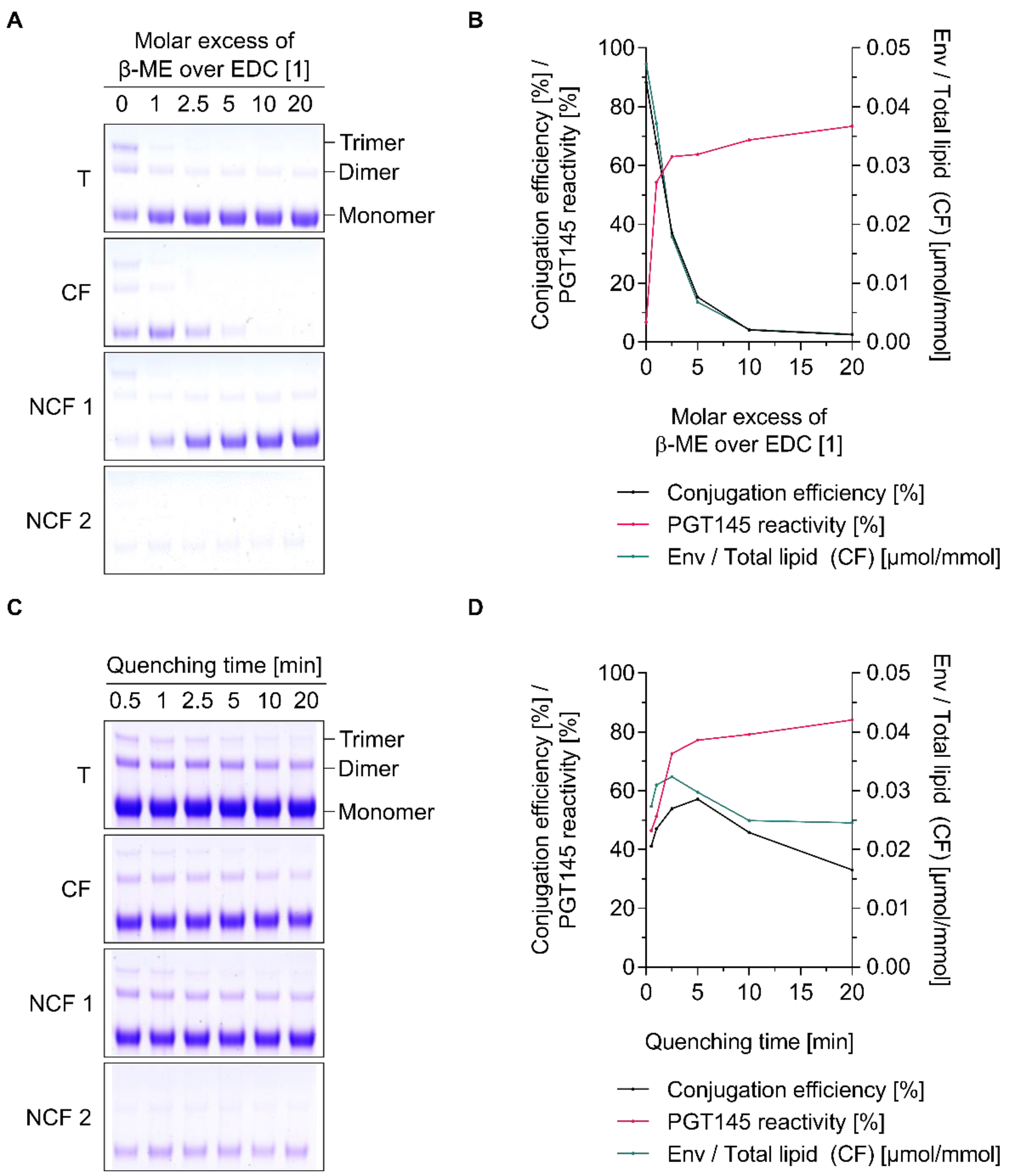
3.1. Efficient Removal of Residual EDC Improves the PGT145 Reactivity of Liposome-Displayed Env Trimers
3.2. The Poor Aqueous Stability of Sulfo-NHS Esters Impedes the Scalability of EDC/Sulfo-NHS-Based Approaches for the Preparation of Env-Liposome Conjugates
3.3. Chemical Inactivation of Residual EDC by β-Mercaptoethanol Is a Time-Saving and Potentially Scalable Alternative to the Removal by Filtration-Based Methods
3.4. The Charge of Both Liposomes and Env Trimers Determines the Extent of Conjugation
3.5. The Extent of Conjugation Is a Function of Ionic Strength
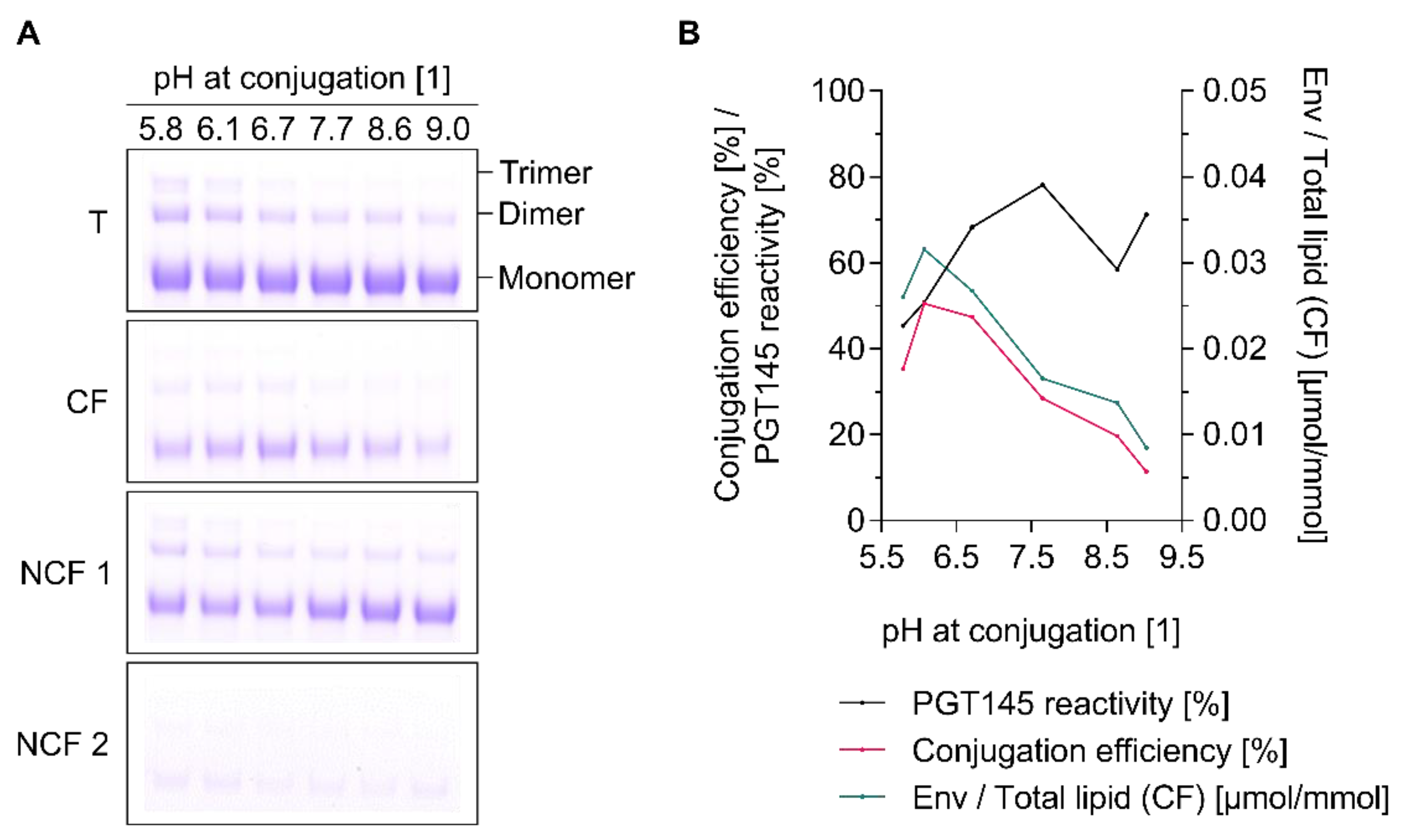
3.6. Conjugation Efficiency and PGT145 Reactivity of Liposome-Displayed Env Trimers Are a Function of pH
3.7. Efficient Conjugation Is Limited to Particular Reaction Conditions
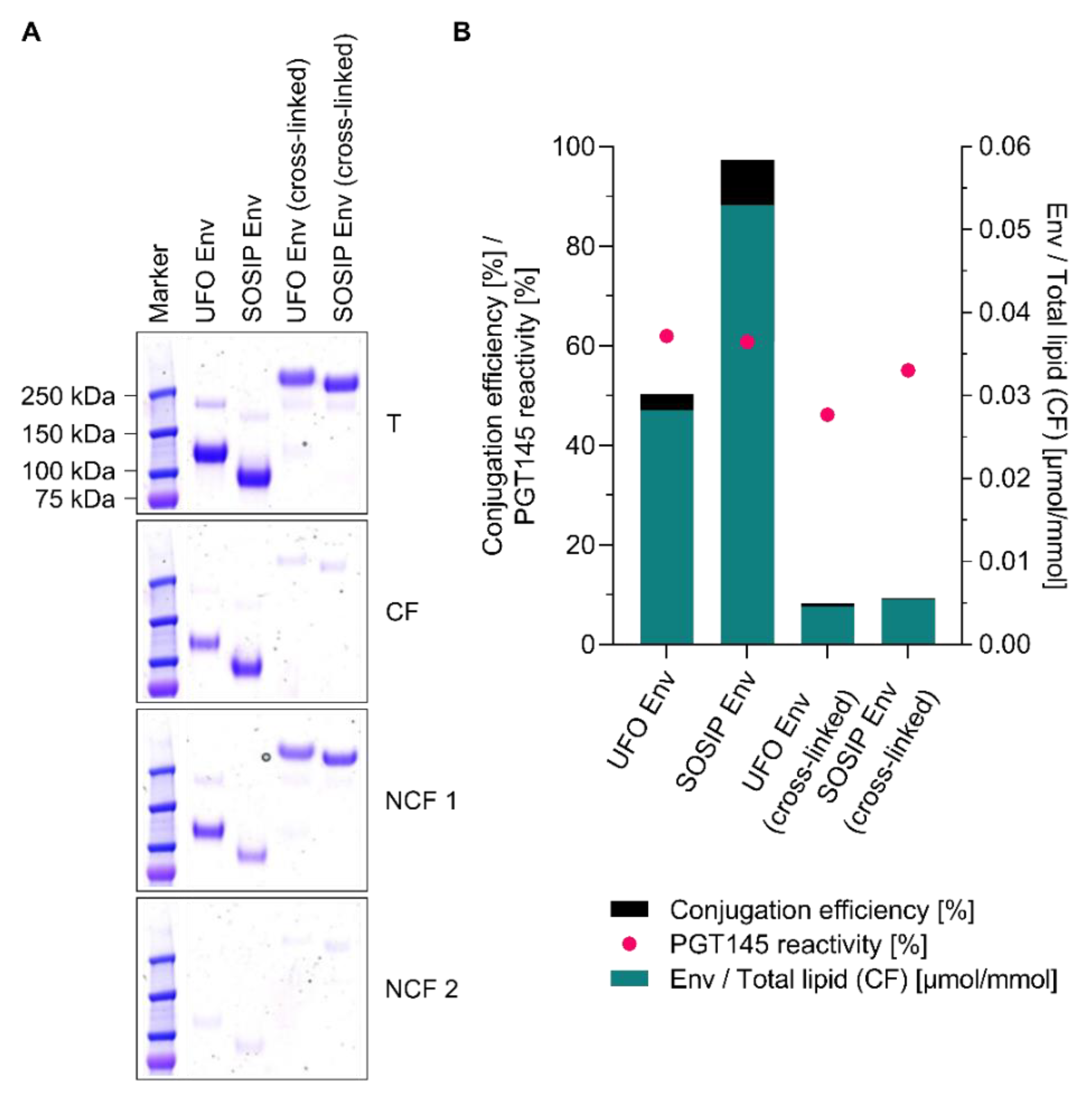
3.8. Conjugation of Other Tag-Free, Non-Functionalised Next-Generation HIV-1 Immunogens
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanders, R.W.; Moore, J.P. Native-like Env trimers as a platform for HIV-1 vaccine design. Immunol. Rev. 2017, 275, 161–182. [Google Scholar] [CrossRef]
- Klasse, P.J.; Ozorowski, G.; Sanders, R.W.; Moore, J.P. Env Exceptionalism: Why Are HIV-1 Env Glycoproteins Atypical Immunogens? Cell Host Microbe 2020, 27, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, K.E.; Wagh, K.; Korber, B.; Barouch, D.H. Vaccines and Broadly Neutralizing Antibodies for HIV-1 Prevention. Annu. Rev. Immunol. 2020, 38, 673–703. [Google Scholar] [CrossRef] [PubMed]
- Irvine, D.J.; Read, B.J. Shaping humoral immunity to vaccines through antigen-displaying nanoparticles. Curr. Opin. Immunol. 2020, 65, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Thalhauser, S.; Peterhoff, D.; Wagner, R.; Breunig, M. Critical design criteria for engineering a nanoparticulate HIV-1 vaccine. J. Control. Release 2020, 317, 322–335. [Google Scholar] [CrossRef] [PubMed]
- Irvine, D.J.; Aung, A.; Silva, M. Controlling timing and location in vaccines. Adv. Drug Deliv. Rev. 2020. [Google Scholar] [CrossRef]
- Brouwer, P.J.M.; Sanders, R.W. Presentation of HIV-1 envelope glycoprotein trimers on diverse nanoparticle platforms. Curr. Opin. HIV AIDS 2019, 14, 302–308. [Google Scholar] [CrossRef]
- Brinkkemper, M.; Sliepen, K. Nanoparticle Vaccines for Inducing HIV-1 Neutralizing Antibodies. Vaccines 2019, 7, 76. [Google Scholar] [CrossRef]
- del Moral-Sánchez, I.; Sliepen, K. Strategies for inducing effective neutralizing antibody responses against HIV-1. Expert Rev. Vaccines 2019, 18, 1127–1143. [Google Scholar] [CrossRef]
- Fries, C.N.; Curvino, E.J.; Chen, J.; Permar, S.R.; Fouda, G.G.; Collier, J.H. Advances in nanomaterial vaccine strategies to address infectious diseases impacting global health. Nat. Nanotechnol. 2020. [Google Scholar] [CrossRef]
- Dubrovskaya, V.; Tran, K.; Ozorowski, G.; Guenaga, J.; Wilson, R.; Bale, S.; Cottrell, C.A.; Turner, H.L.; Seabright, G.; O’Dell, S.; et al. Vaccination with Glycan-Modified HIV NFL Envelope Trimer-Liposomes Elicits Broadly Neutralizing Antibodies to Multiple Sites of Vulnerability. Immunity 2019, 51, 915–929. [Google Scholar] [CrossRef] [PubMed]
- Pauthner, M.; Havenar-Daughton, C.; Sok, D.; Nkolola, J.P.; Bastidas, R.; Boopathy, A.V.; Carnathan, D.G.; Chandrashekar, A.; Cirelli, K.M.; Cottrell, C.A.; et al. Elicitation of Robust Tier 2 Neutralizing Antibody Responses in Nonhuman Primates by HIV Envelope Trimer Immunization Using Optimized Approaches. Immunity 2017, 46, 1073–1088. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Moon, J.J.; Cheng, W. Quantitation and Stability of Protein Conjugation on Liposomes for Controlled Density of Surface Epitopes. Bioconjug. Chem. 2018, 29, 1251–1260. [Google Scholar] [CrossRef]
- Martinez-Murillo, P.; Tran, K.; Guenaga, J.; Lindgren, G.; Àdori, M.; Feng, Y.; Phad, G.E.; Vázquez Bernat, N.; Bale, S.; Ingale, J.; et al. Particulate Array of Well-Ordered HIV Clade C Env Trimers Elicits Neutralizing Antibodies that Display a Unique V2 Cap Approach. Immunity 2017, 46, 804–817. [Google Scholar] [CrossRef] [PubMed]
- Ingale, J.; Stano, A.; Guenaga, J.; Kumar Sharma, S.; Nemazee, D.; Zwick, M.B.; Wyatt, R.T. High-Density Array of Well-Ordered HIV-1 Spikes on Synthetic Liposomal Nanoparticles Efficiently Activate B Cells. Cell Rep. 2016, 15, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bale, S.; Goebrecht, G.; Stano, A.; Wilson, R.; Ota, T.; Tran, K.; Ingale, J.; Zwick, M.B.; Wyatt, R.T. Covalent Linkage of HIV-1 Trimers to Synthetic Liposomes Elicits Improved B Cell and Antibody Responses. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Pejawar-Gaddy, S.; Kovacs, J.M.; Barouch, D.H.; Chen, B.; Irvine, D.J. Design of Lipid Nanocapsule Delivery Vehicles for Multivalent Display of Recombinant Env Trimers in HIV Vaccination. Bioconjug. Chem. 2014, 25, 1470–1478. [Google Scholar] [CrossRef]
- Tokatlian, T.; Kulp, D.W.; Mutafyan, A.A.; Jones, C.A.; Menis, S.; Georgeson, E.; Kubitz, M.; Zhang, M.H.; Melo, M.B.; Silva, M.; et al. Enhancing Humoral Responses Against HIV Envelope Trimers via Nanoparticle Delivery with Stabilized Synthetic Liposomes. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Steichen, J.M.; Kulp, D.W.; Tokatlian, T.; Escolano, A.; Dosenovic, P.; Stanfield, R.L.; McCoy, L.E.; Ozorowski, G.; Hu, X.; Kalyuzhniy, O.; et al. HIV Vaccine Design to Target Germline Precursors of Glycan-Dependent Broadly Neutralizing Antibodies. Immunity 2016, 45, 483–496. [Google Scholar] [CrossRef]
- Tokatlian, T.; Read, B.J.; Jones, C.A.; Kulp, D.W.; Menis, S.; Chang, J.Y.H.; Steichen, J.M.; Kumari, S.; Allen, J.D.; Dane, E.L.; et al. Innate immune recognition of glycans targets HIV nanoparticle immunogens to germinal centers. Science (80-) 2019, 363, 649–654. [Google Scholar] [CrossRef]
- Kirchhof, S.; Strasser, A.; Wittmann, H.J.; Messmann, V.; Hammer, N.; Goepferich, A.M.; Brandl, F.P. New insights into the cross-linking and degradation mechanism of Diels-Alder hydrogels. J. Mater. Chem. B 2015, 3, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Oswald, M.; Geissler, S.; Goepferich, A. Determination of the activity of maleimide-functionalized phospholipids during preparation of liposomes. Int. J. Pharm. 2016, 514, 93–102. [Google Scholar] [CrossRef]
- Shah, S.; Dhawan, V.; Holm, R.; Nagarsenker, M.S.; Perrie, Y. Liposomes: Advancements and innovation in the manufacturing process. Adv. Drug Deliv. Rev. 2020. [Google Scholar] [CrossRef]
- Aldon, Y.; McKay, P.F.; Allen, J.; Ozorowski, G.; Felfödiné Lévai, R.; Tolazzi, M.; Rogers, P.; He, L.; de Val, N.; Fábián, K.; et al. Rational Design of DNA-Expressed Stabilized Native-Like HIV-1 Envelope Trimers. Cell Rep. 2018, 24, 3324–3338. [Google Scholar] [CrossRef]
- Kong, L.; He, L.; De Val, N.; Vora, N.; Morris, C.D.; Azadnia, P.; Sok, D.; Zhou, B.; Burton, D.R.; Ward, A.B.; et al. Uncleaved prefusion-optimized gp140 trimers derived from analysis of HIV-1 envelope metastability. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef]
- Hermanson, G.T. Zero-Length Crosslinkers. In Bioconjugate Techniques; Academic Press (Elsevier Inc.): Cambridge, MA, USA, 2013; pp. 259–273. ISBN 978-0-12-382239-0. [Google Scholar]
- Totaro, K.A.; Liao, X.; Bhattacharya, K.; Finneman, J.I.; Sperry, J.B.; Massa, M.A.; Thorn, J.; Ho, S.V.; Pentelute, B.L. Systematic Investigation of EDC/sNHS-Mediated Bioconjugation Reactions for Carboxylated Peptide Substrates. Bioconjug. Chem. 2016, 27, 994–1004. [Google Scholar] [CrossRef] [PubMed]
- Mädler, S.; Bich, C.; Touboul, D.; Zenobi, R. Chemical cross-linking with NHS esters: A systematic study on amino acid reactivities. J. Mass Spectrom. 2009, 44, 694–706. [Google Scholar] [CrossRef]
- Marques, A.C.; Costa, P.J.; Velho, S.; Amaral, M.H. Functionalizing nanoparticles with cancer-targeting antibodies: A comparison of strategies. J. Control. Release 2020, 320, 180–200. [Google Scholar] [CrossRef]
- Bogdanov, A.A.; Klibanov, A.L.; Torchilin, V.P. Protein immobilization on the surface of liposomes via carbodiimide activation in the presence of N -hydroxysulfosuccinimide. FEBS Lett. 1988, 231, 381–384. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, J. Surface engineering of nanomaterials with phospholipid-polyethylene glycol-derived functional conjugates for molecular imaging and targeted therapy. Biomaterials 2020, 230. [Google Scholar] [CrossRef] [PubMed]
- Ringe, R.P.; Cruz Portillo, V.M.; Dosenovic, P.; Ketas, T.J.; Ozorowski, G.; Nogal, B.; Perez, L.; LaBranche, C.C.; Lim, J.; Francomano, E.; et al. Neutralizing Antibody Induction by HIV-1 Envelope Glycoprotein SOSIP Trimers on Iron Oxide Nanoparticles May Be Impaired by Mannose Binding Lectin. J. Virol. 2020, 94. [Google Scholar] [CrossRef]
- Risse, F.; Gedig, E.T.; Gutmann, J.S. Carbodiimide-mediated immobilization of acidic biomolecules on reversed-charge zwitterionic sensor chip surfaces. Anal. Bioanal. Chem. 2018, 410, 4109–4122. [Google Scholar] [CrossRef]
- Fischer, M.J.E. Amine Coupling Through EDC/NHS: A Practical Approach. In Surface Plasmon Resonance. Methods in Molecular Biology (Methods and Protocols); de Mol, N.J., Fischer, M.J.E., Eds.; Humana Press Inc.: Totowa, NJ, USA, 2010; pp. 55–73. ISBN 978-1-60761-669-6. [Google Scholar]
- Gedig, E.T. Surface Chemistry in SPR Technology. In Handbook of Surface Plasmon Resonance; Schasfoort, R.B.M., Ed.; Royal Society of Chemistry: London, UK, 2017; pp. 200–211. ISBN 978-1-78801-028-3. [Google Scholar]
- Ranishenka, B.; Ulashchik, E.; Radchanka, A.; Shmanai, V.; Artemyev, M. Electrostatic Repulsion Controls Efficiency of Cu-Free Click-Reaction with Azide-Modified Semiconductor Quantum Dots. ChemNanoMat 2020, 6, 292–297. [Google Scholar] [CrossRef]
- Jans, H.; Jans, K.; Stakenborg, T.; Van de Broek, B.; Lagae, L.; Maes, G.; Borghs, G. Impact of pre-concentration to covalently biofunctionalize suspended nanoparticles. Nanotechnology 2010, 21. [Google Scholar] [CrossRef]
- Pejawar-Gaddy, S.; Rajawat, Y.; Hilioti, Z.; Xue, J.; Gaddy, D.F.; Finn, O.J.; Viscidi, R.P.; Bossis, I. Generation of a tumor vaccine candidate based on conjugation of a MUC1 peptide to polyionic papillomavirus virus-like particles. Cancer Immunol. Immunother. 2010, 59, 1685–1696. [Google Scholar] [CrossRef]
- Bartczak, D.; Kanaras, A.G. Preparation of Peptide-Functionalized Gold Nanoparticles Using One Pot EDC/Sulfo-NHS Coupling. Langmuir 2011, 27, 10119–10123. [Google Scholar] [CrossRef] [PubMed]
- Posse, E.; López Viñals, A.; de Arcuri, B.F.; Farías, R.N.; Morero, R.D. Lysozyme induced fusion of negatively charged phospholipid vesicles. Biochim. Biophys. Acta - Biomembr. 1990, 1024, 390–394. [Google Scholar] [CrossRef]
- Zschörnig, O.; Paasche, G.; Thieme, C.; Korb, N.; Arnold, K. Modulation of lysozyme charge influences interaction with phospholipid vesicles. Colloids Surfaces B Biointerfaces 2005, 42, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Bergers, J.J.; Vingerhoeds, M.H.; van Bloois, L.; Herron, J.N.; Janssen, L.H.M.; Fischer, M.J.E.; Crommelin, D.J.A. The Role of Protein Charge in Protein–Lipid Interactions. pH-Dependent Changes of the Electrophoretic Mobility of Liposomes through Adsorption of Water-Soluble, Globular Proteins. Biochemistry 1993, 32, 4641–4649. [Google Scholar] [CrossRef] [PubMed]
- Temchura, V.; Überla, K. Intrastructural help: Improving the HIV-1 envelope antibody response induced by virus-like particle vaccines. Curr. Opin. HIV AIDS 2017, 12, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Suleiman, E.; Damm, D.; Batzoni, M.; Temchura, V.; Wagner, A.; Überla, K.; Vorauer-Uhl, K. Electrostatically Driven Encapsulation of Hydrophilic, Non-Conformational Peptide Epitopes into Liposomes. Pharmaceutics 2019, 11, 619. [Google Scholar] [CrossRef] [PubMed]
- Sliepen, K.; Han, B.W.; Bontjer, I.; Mooij, P.; Garces, F.; Behrens, A.J.; Rantalainen, K.; Kumar, S.; Sarkar, A.; Brouwer, P.J.M.; et al. Structure and immunogenicity of a stabilized HIV-1 envelope trimer based on a group-M consensus sequence. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef]
- Schiffner, T.; de Val, N.; Russell, R.A.; de Taeye, S.W.; Torrents de la Peña, A.; Ozorowski, G.; Kim, H.J.; Nieusma, T.; Brod, F.; Cupo, A.; et al. Chemical Cross-Linking Stabilizes Native-Like HIV-1 Envelope Glycoprotein Trimer Antigens. J. Virol. 2016, 90, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.M.; Huber, M.; Doores, K.J.; Falkowska, E.; Pejchal, R.; Julien, J.-P.; Wang, S.-K.; Ramos, A.; Chan-Hui, P.-Y.; Moyle, M.; et al. Broad neutralization coverage of HIV by multiple highly potent antibodies. Nature 2011, 477, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Asokan, M.; Rudicell, R.S.; Louder, M.; McKee, K.; O’Dell, S.; Stewart-Jones, G.; Wang, K.; Xu, L.; Chen, X.; Choe, M.; et al. Bispecific Antibodies Targeting Different Epitopes on the HIV-1 Envelope Exhibit Broad and Potent Neutralization. J. Virol. 2015, 89, 12501–12512. [Google Scholar] [CrossRef]
- Burton, D.R.; Pyati, J.; Koduri, R.; Sharp, S.J.; Thornton, G.B.; Parren, P.W.H.I.; Sawyer, L.S.W.; Hendry, R.M.; Dunlop, N.; Nara, P.L.; et al. Efficient neutralization of primary isolates of HIV-1 by a recombinant human monoclonal antibody. Science 1994, 266, 1024–1027. [Google Scholar] [CrossRef]
- Buchacher, A.; Predl, R.; Strutzenberger, K.; Steinfellner, W.; Trkola, A.; Purtscher, M.; Gruber, G.; Tauer, C.; Steindl, F.; Jungbauer, A.; et al. Generation of Human Monoclonal Antibodies against HIV-1 Proteins; Electrofusion and Epstein-Barr Virus Transformation for Peripheral Blood Lymphocyte Immortalization. AIDS Res. Hum. Retroviruses 1994, 10, 359–369. [Google Scholar] [CrossRef]
- van Gils, M.J.; van den Kerkhof, T.L.G.M.; Ozorowski, G.; Cottrell, C.A.; Sok, D.; Pauthner, M.; Pallesen, J.; De Val, N.; Yasmeen, A.; De Taeye, S.W.; et al. An HIV-1 antibody from an elite neutralizer implicates the fusion peptide as a site of vulnerability. Nat. Microbiol. 2016, 2. [Google Scholar] [CrossRef]
- Wu, X.; Yang, Z.Y.; Li, Y.; Hogerkorp, C.M.; Schief, W.R.; Seaman, M.S.; Zhou, T.; Schmidt, S.D.; Wu, L.; Xu, L.; et al. Rational design of envelope identifies broadly neutralizing human monoclonal antibodies to HIV-1. Science 2010, 329, 856–861. [Google Scholar] [CrossRef]
- Falkowska, E.; Le, K.M.; Ramos, A.; Doores, K.J.; Lee, J.H.; Blattner, C.; Ramirez, A.; Derking, R.; van Gils, M.J.; Liang, C.-H.; et al. Broadly Neutralizing HIV Antibodies Define a Glycan-Dependent Epitope on the Prefusion Conformation of gp41 on Cleaved Envelope Trimers. Immunity 2014, 40, 657–668. [Google Scholar] [CrossRef]
- Robinson, J.E.; Holton, D.; Pacheco-Morell, S.; Liu, J.; McMurdo, H. Identification of Conserved and Variant Epitopes of Human Immunodeficiency Virus Type 1 (HIV-1) gp120 by Human Monoclonal Antibodies Produced by EBV-Transformed Cell Lines. AIDS Res. Hum. Retroviruses 1990, 6, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Thali, M.; Moore, J.P.; Furman, C.; MacArthur, C.; Ho, D.D.; Robinson, J.; Sodroski, J. Characterization of conserved human immunodeficiency virus type 1 gp120 neutralization epitopes exposed upon gp120-CD4 binding. J. Virol. 1993, 67, 3978–3988. [Google Scholar] [CrossRef] [PubMed]
- Marsh, D. Handbook of Lipid Bilayers, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2013; ISBN 978-1-4200-8833-5. [Google Scholar]
- Simberg, D.; Weisman, S.; Talmon, Y.; Barenholz, Y. DOTAP (and other cationic lipids): Chemistry, biophysics, and transfection. Crit. Rev. Ther. Drug Carrier Syst. 2004, 21, 257–317. [Google Scholar] [CrossRef] [PubMed]
- Protein Calculator v3.4. Available online: http://protcalc.sourceforge.net/ (accessed on 28 May 2019).
- Katinger, H.; Vorauer-Uhl, K.; Wagner, A.; Kreismayr, G. Method and device for producing lipid vesicles. United States Patent No.: US 6,843,942 B2, 17 January 2005. [Google Scholar]
- Wagner, A.; Vorauer-Uhl, K.; Kreismayr, G.; Katinger, H. The crossflow injection technique: An improvement of the ethanol injection method. J. Liposome Res. 2002, 12, 259–270. [Google Scholar] [CrossRef]
- Garcon, N.M.J.; Six, H.R. Universal vaccine carrier. Liposomes that provide T-dependent help to weak antigens. J. Immunol. 1991, 146, 3697–3702. [Google Scholar]
- Gregoriadis, G.; Wang, Z.; Barenholz, Y.; Francis, M.J. Liposome-entrapped T-cell peptide provides help for a co-entrapped B-cell peptide to overcome genetic restriction in mice and induce immunological memory. Immunology 1993, 80, 535–540. [Google Scholar]
- Goodman-Snitkoff, G.; Good, M.F.; Berzofsky, J.A.; Mannino, R.J. Role of intrastructural/intermolecular help in immunization with peptide-phospholipid complexes. J. Immunol. 1991, 147, 410–415. [Google Scholar] [PubMed]
- Nabi, G.; Storcksdieck genannt Bonsmann, M.; Tenbusch, M.; Gardt, O.; Barouch, D.H.; Temchura, V.; Überla, K. GagPol-specific CD4+ T-cells increase the antibody response to Env by intrastructural help. Retrovirology 2013, 10. [Google Scholar] [CrossRef]
- Storcksdieck genannt Bonsmann, M.; Niezold, T.; Temchura, V.; Pissani, F.; Ehrhardt, K.; Brown, E.P.; Osei-Owusu, N.Y.; Hannaman, D.; Hengel, H.; Ackerman, M.E.; et al. Enhancing the Quality of Antibodies to HIV-1 Envelope by GagPol-Specific Th Cells. J. Immunol. 2015, 195, 4861–4872. [Google Scholar] [CrossRef]
- Elsayed, H.; Nabi, G.; McKinstry, W.J.; Khoo, K.K.; Mak, J.; Salazar, A.M.; Tenbusch, M.; Temchura, V.; Überla, K. Intrastructural Help: Harnessing T Helper Cells Induced by Licensed Vaccines for Improvement of HIV Env Antibody Responses to Virus-Like Particle Vaccines. J. Virol. 2018, 92. [Google Scholar] [CrossRef]
- Damm, D.; Rojas-Sánchez, L.; Theobald, H.; Sokolova, V.; Wyatt, R.T.; Überla, K.; Epple, M.; Temchura, V. Calcium Phosphate Nanoparticle-Based Vaccines as a Platform for Improvement of HIV-1 Env Antibody Responses by Intrastructural Help. Nanomaterials 2019, 9, 1389. [Google Scholar] [CrossRef] [PubMed]
- Heß, R.; Bonsmann, M.S.G.; Lapuente, D.; Maaske, A.; Kirschning, C.; Ruland, J.; Lepenies, B.; Hannaman, D.; Tenbusch, M.; Überla, K. Glycosylation of HIV env impacts IgG subtype responses to vaccination. Viruses 2019, 11, 153. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.C.; Abraham, W.; Crespo, M.P.; Chen, S.H.; Liu, H.; Szeto, G.L.; Kim, M.; Reinherz, E.L.; Irvine, D.J. Liposomal vaccines incorporating molecular adjuvants and intrastructural T-cell help promote the immunogenicity of HIV membrane-proximal external region peptides. Vaccine 2015, 33, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Zaman, M.; Ozberk, V.; Langshaw, E.L.; McPhun, V.; Powell, J.L.; Phillips, Z.N.; Ho, M.F.; Calcutt, A.; Batzloff, M.R.; Toth, I.; et al. Novel platform technology for modular mucosal vaccine that protects against streptococcus. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Hills, T.; Jakeman, P.G.; Carlisle, R.C.; Klenerman, P.; Seymour, L.W.; Cawood, R. A Rapid-Response Humoral Vaccine Platform Exploiting Pre-Existing Non-Cognate Populations of Anti-Vaccine or Anti-Viral CD4+ T Helper Cells to Confirm B Cell Activation. PLoS One 2016, 11, e0166383. [Google Scholar] [CrossRef]
- Wallis, J.; Katti, P.; Martin, A.M.; Hills, T.; Seymour, L.W.; Shenton, D.P.; Carlisle, R.C. A liposome-based cancer vaccine for a rapid and high-titre anti-ErbB-2 antibody response. Eur. J. Pharm. Sci. 2020, 152. [Google Scholar] [CrossRef]
- Tronchere, H.; Boal, F. Liposome Flotation Assays for Phosphoinositide-protein Interaction. Bio-protocol 2017, 7. [Google Scholar] [CrossRef]
- Schiffner, T.; Kong, L.; Duncan, C.J.A.; Back, J.W.; Benschop, J.J.; Shen, X.; Huang, P.S.; Stewart-Jones, G.B.; DeStefano, J.; Seaman, M.S.; et al. Immune Focusing and Enhanced Neutralization Induced by HIV-1 gp140 Chemical Cross-Linking. J. Virol. 2013, 87, 10163–10172. [Google Scholar] [CrossRef]
- Stieh, D.J.; Phillips, J.L.; Rogers, P.M.; King, D.F.; Cianci, G.C.; Jeffs, S.A.; Gnanakaran, S.; Shattock, R.J. Dynamic electrophoretic fingerprinting of the HIV-1 envelope glycoprotein. Retrovirology 2013, 10. [Google Scholar] [CrossRef]
- Sakulkhu, U.; Mahmoudi, M.; Maurizi, L.; Coullerez, G.; Hofmann-Amtenbrink, M.; Vries, M.; Motazacker, M.; Rezaee, F.; Hofmann, H. Significance of surface charge and shell material of superparamagnetic iron oxide nanoparticle (SPION) based core/shell nanoparticles on the composition of the protein corona. Biomater. Sci. 2015, 3, 265–278. [Google Scholar] [CrossRef]
- Lundqvist, M.; Stigler, J.; Elia, G.; Lynch, I.; Cedervall, T.; Dawson, K.A. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc. Natl. Acad. Sci. USA 2008, 105, 14265–14270. [Google Scholar] [CrossRef] [PubMed]
- Heuts, J.; Varypataki, E.M.; van der Maaden, K.; Romeijn, S.; Drijfhout, J.W.; van Scheltinga, A.T.; Ossendorp, F.; Jiskoot, W. Cationic Liposomes: A Flexible Vaccine Delivery System for Physicochemically Diverse Antigenic Peptides. Pharm. Res. 2018, 35. [Google Scholar] [CrossRef] [PubMed]
- Veneziano, R.; Moyer, T.J.; Stone, M.B.; Wamhoff, E.C.; Read, B.J.; Mukherjee, S.; Shepherd, T.R.; Das, J.; Schief, W.R.; Irvine, D.J.; et al. Role of nanoscale antigen organization on B-cell activation probed using DNA origami. Nat. Nanotechnol. 2020, 15, 716–723. [Google Scholar] [CrossRef]
- Schwendener, R.A. Liposomes as vaccine delivery systems: A review of the recent advances. Ther. Adv. Vaccines 2014, 2, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Ma, Y.; Wang, C.; Hai, L.; Yan, C.; Zhang, Y.; Liu, F.; Cai, L. PEGylated cationic liposomes robustly augment vaccine-induced immune responses: Role of lymphatic trafficking and biodistribution. J. Control. Release 2012, 159, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, P.; Zhuang, Y.; Li, P.; Jiang, B.; Pan, H.; Liu, L.; Cai, L.; Ma, Y. Lymphatic-targeted cationic liposomes: A robust vaccine adjuvant for promoting long-term immunological memory. Vaccine 2014, 32, 5475–5483. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suleiman, E.; Mayer, J.; Lehner, E.; Kohlhauser, B.; Katholnig, A.; Batzoni, M.; Damm, D.; Temchura, V.; Wagner, A.; Überla, K.; et al. Conjugation of Native-Like HIV-1 Envelope Trimers onto Liposomes Using EDC/Sulfo-NHS Chemistry: Requirements and Limitations. Pharmaceutics 2020, 12, 979. https://doi.org/10.3390/pharmaceutics12100979
Suleiman E, Mayer J, Lehner E, Kohlhauser B, Katholnig A, Batzoni M, Damm D, Temchura V, Wagner A, Überla K, et al. Conjugation of Native-Like HIV-1 Envelope Trimers onto Liposomes Using EDC/Sulfo-NHS Chemistry: Requirements and Limitations. Pharmaceutics. 2020; 12(10):979. https://doi.org/10.3390/pharmaceutics12100979
Chicago/Turabian StyleSuleiman, Ehsan, Julia Mayer, Elisabeth Lehner, Bianca Kohlhauser, Alexandra Katholnig, Mirjam Batzoni, Dominik Damm, Vladimir Temchura, Andreas Wagner, Klaus Überla, and et al. 2020. "Conjugation of Native-Like HIV-1 Envelope Trimers onto Liposomes Using EDC/Sulfo-NHS Chemistry: Requirements and Limitations" Pharmaceutics 12, no. 10: 979. https://doi.org/10.3390/pharmaceutics12100979
APA StyleSuleiman, E., Mayer, J., Lehner, E., Kohlhauser, B., Katholnig, A., Batzoni, M., Damm, D., Temchura, V., Wagner, A., Überla, K., & Vorauer-Uhl, K. (2020). Conjugation of Native-Like HIV-1 Envelope Trimers onto Liposomes Using EDC/Sulfo-NHS Chemistry: Requirements and Limitations. Pharmaceutics, 12(10), 979. https://doi.org/10.3390/pharmaceutics12100979





