Characteristics of Skin Deposition of Itraconazole Solubilized in Cream Formulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Solubility Measurement of ITZ in Excipients for Cream Formulations
2.3. HPLC of ITZ
2.4. Preparation of O/W Cream Formulation and Physical Mixture
2.5. Solubility Measurement of ITZ in Oil Phase and O/W Cream Base
2.6. Characterization of O/W Cream Formulation and Physical Mixture
2.6.1. Organoleptic Properties
2.6.2. pH
2.6.3. Polarized Light Microscopy
2.7. Differential Scanning Calorimetry
2.8. Stability Test of O/W Cream Formulation of ITZ
2.9. In Vitro Skin Deposition and Penetration Analyses
2.9.1. Preparation of Rat Skin
2.9.2. In Vitro Skin Penetration Analysis
2.9.3. In Vitro Skin Deposition Analysis
2.10. Statistical Analysis
3. Results and Discussion
3.1. Solubility of ITZ in Excipients and O/W Cream Formulation
3.2. Organoleptic Characteristics of O/W Cream Formulation and Physical Mixture
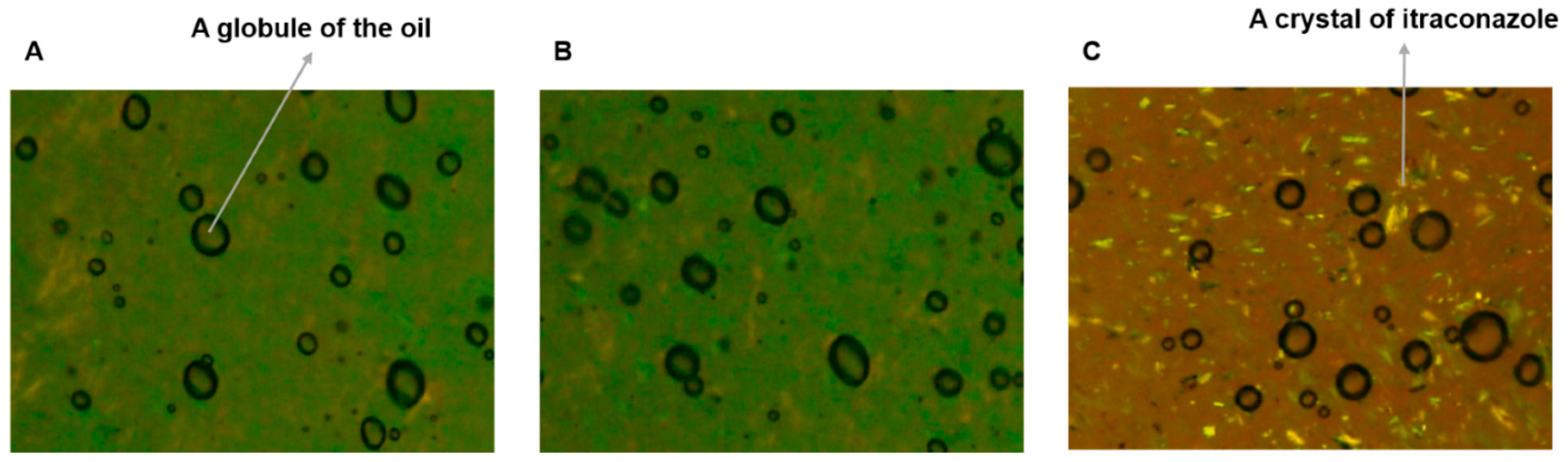
3.3. Polarized Light Microscopy
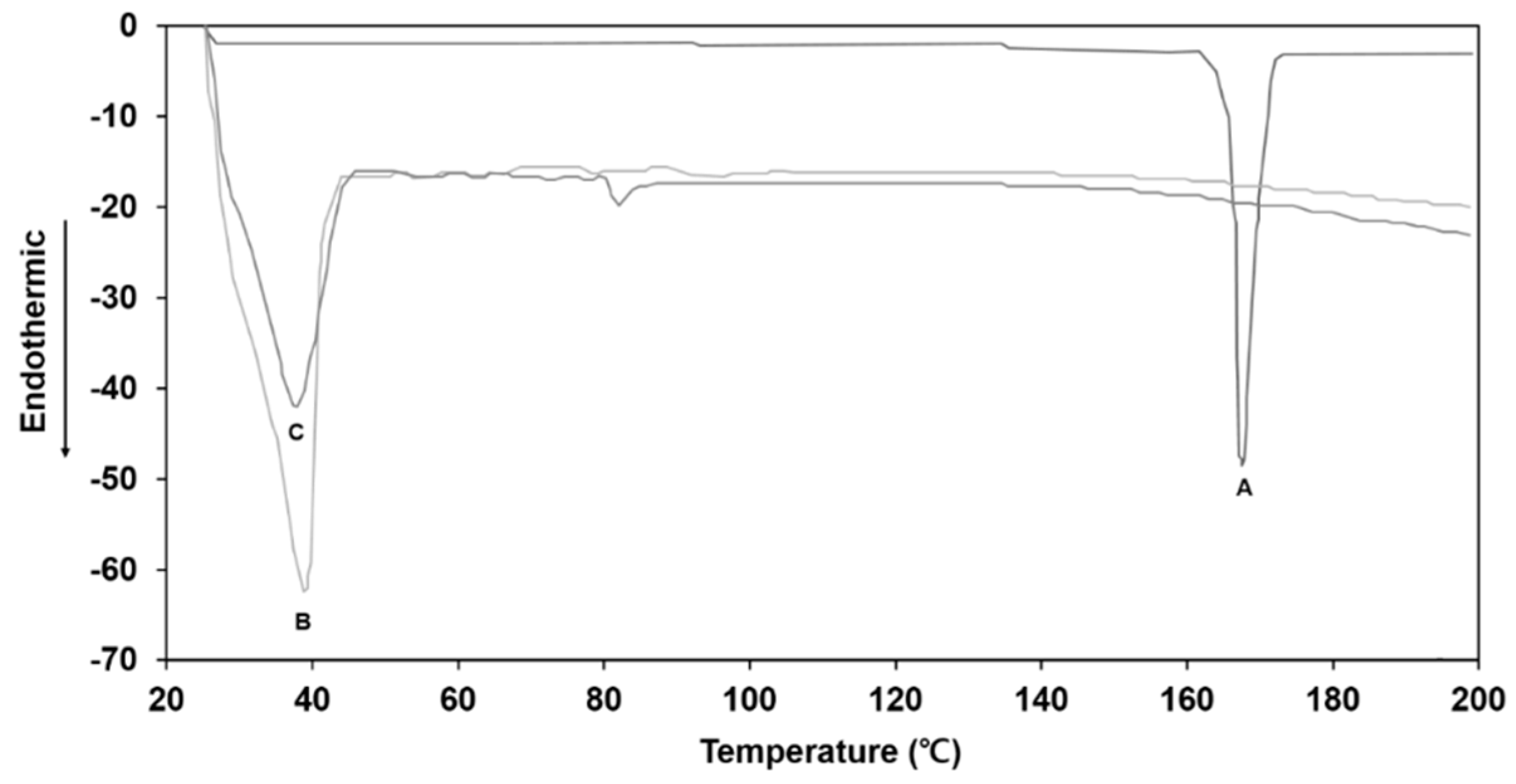
3.4. DSC Analysis
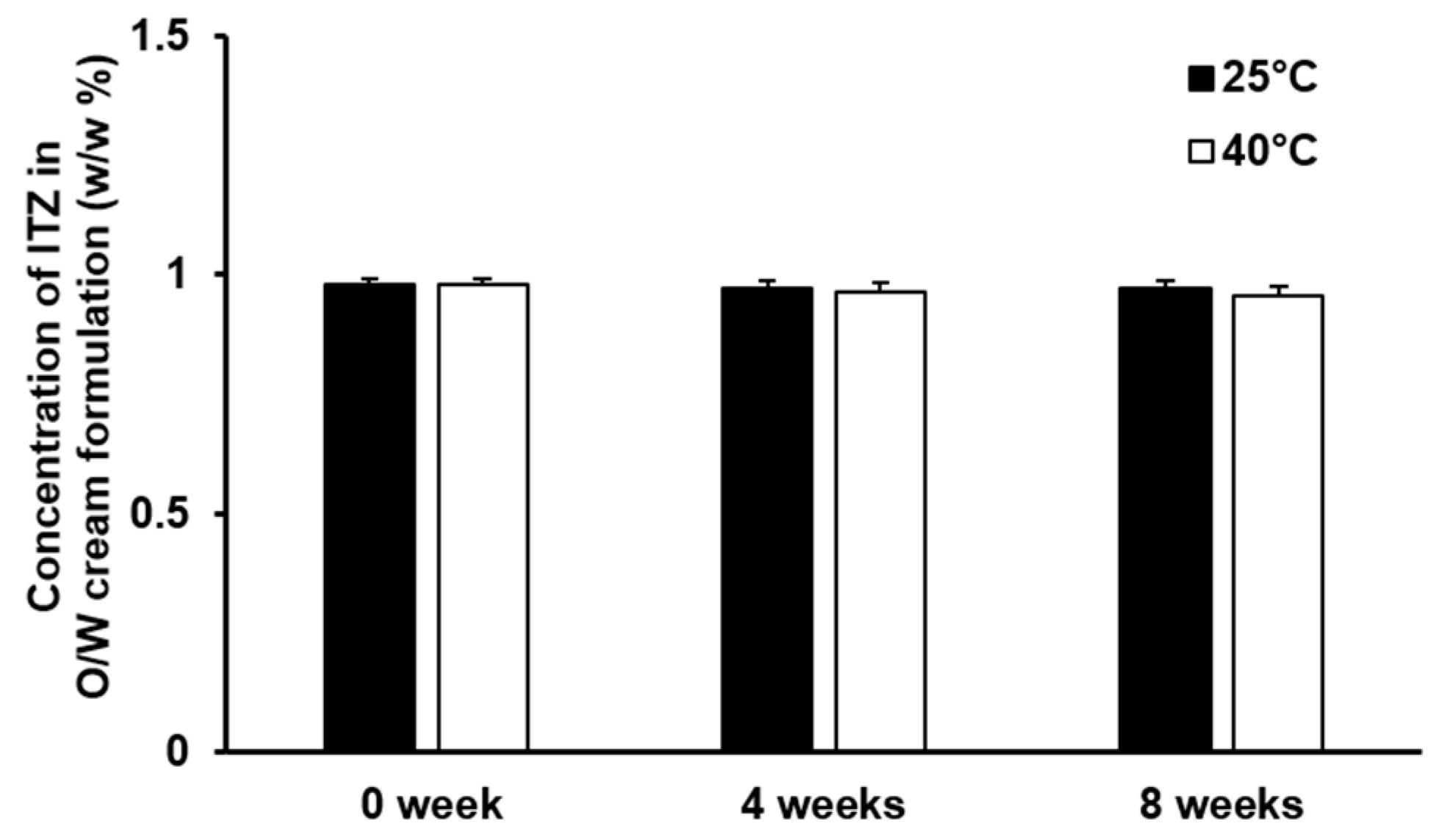
3.5. Stability of the Cream Formulation of ITZ
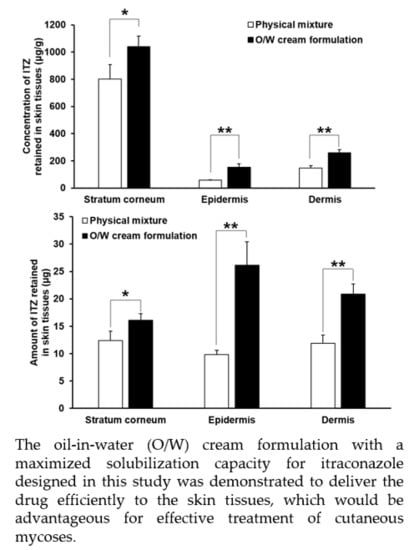
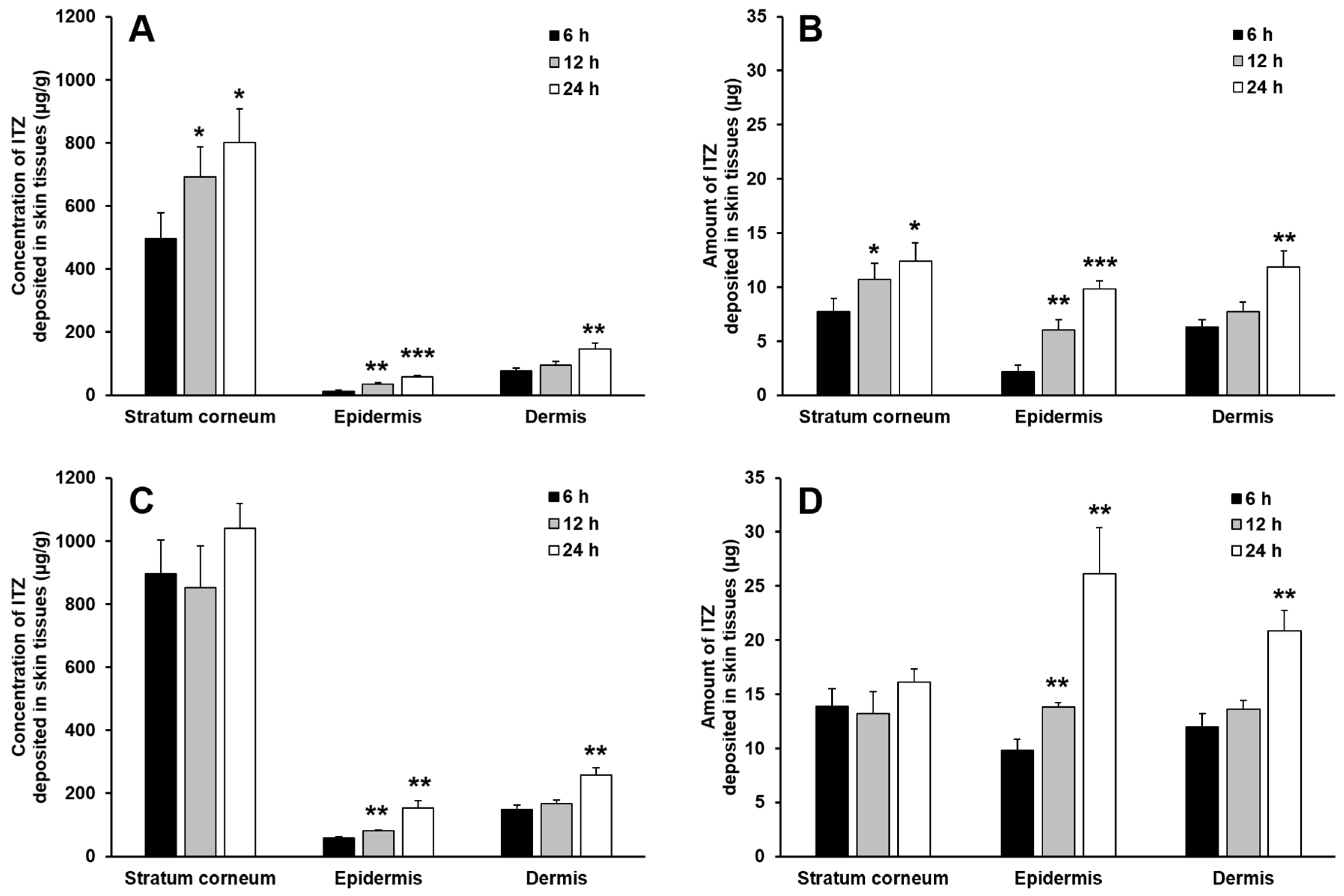
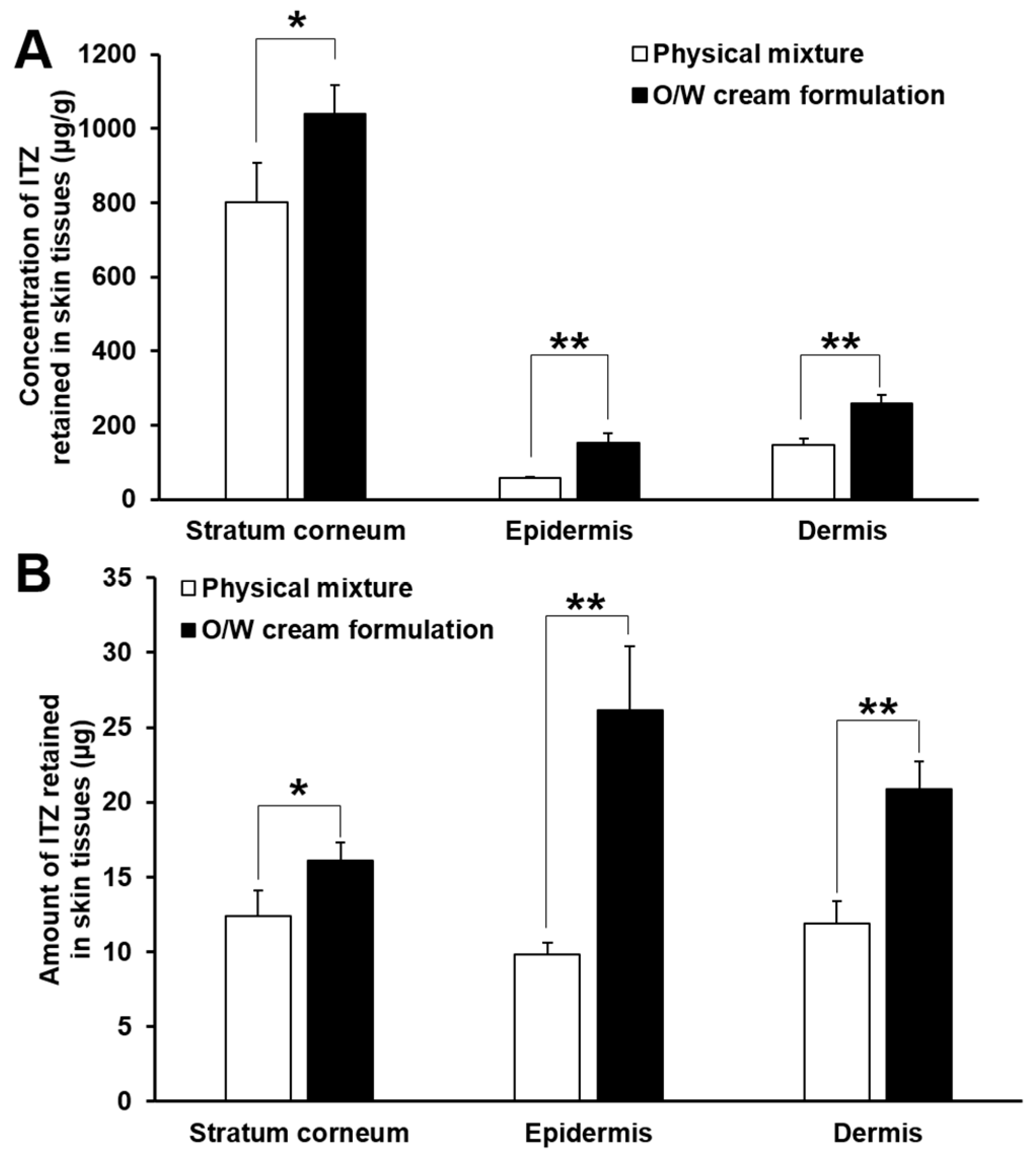
3.6. In Vitro Skin Deposition and Penetration Analyses
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hong, J.Y.; Kim, J.K.; Song, Y.K.; Park, J.S.; Kim, C.K. A new self-emulsifying formulation of itraconazole with improved dissolution and oral absorption. J. Control. Release 2006, 110, 332–338. [Google Scholar] [CrossRef] [PubMed]
- De Doncker, P.; Pande, S.; Richarz, U.; Garodia, N. Itraconazole: What clinicians should know? Indian J. Drugs Dermatol. 2017, 3, 4–10. [Google Scholar]
- Gupta, A.K.; Nolting, S.; de Prost, Y.; Delescluse, J.; Degreef, H.; Theissen, U.; Wallace, R.; Marynissen, G.; De Doncker, P. The use of itraconazole to treat cutaneous fungal infections in children. Dermatology 1999, 199, 248–252. [Google Scholar] [CrossRef]
- Debruyne, D.; Coquerel, A. Pharmacokinetics of antifungal agents in onychomycoses. Clin. Pharmacokinet. 2001, 40, 441–472. [Google Scholar] [CrossRef] [PubMed]
- Thiim, M.; Friedman, L.S. Hepatotoxicity of antibiotics and antifungals. Clin. Liver Dis. 2003, 7, 381–399. [Google Scholar] [CrossRef]
- Clinard, V.B.; Smith, J.D. Cutaneous fungal infections. US Pharm. 2015, 40, 35–39. [Google Scholar]
- Berben, P.; Mols, R.; Brouwers, J.; Tack, J.; Augustijns, P. Gastrointestinal behavior of itraconazole in humans—Part 2: The effect of intraluminal dilution on the performance of a cyclodextrin-based solution. Int. J. Pharm. 2017, 526, 235–243. [Google Scholar] [CrossRef]
- Pople, P.V.; Singh, K.K. Development and evaluation of topical formulation containing solid lipid nanoparticles of vitamin A. Aaps Pharmscitech 2006, 7, E63–E69. [Google Scholar] [CrossRef]
- Seo, J.; Kim, M.-J.; Jeon, S.-O.; Oh, D.-H.; Yoon, K.-H.; Choi, Y.W.; Bashyal, S.; Lee, S. Enhanced topical delivery of fish scale collagen employing negatively surface-modified nanoliposome. J. Pharm. Investig. 2018, 48, 243–250. [Google Scholar] [CrossRef]
- Choi, Y.H.; Han, H.-K. Nanomedicines: Current status and future perspectives in aspect of drug delivery and pharmacokinetics. J. Pharm. Investig. 2018, 48, 43–60. [Google Scholar] [CrossRef]
- Ghate, V.M.; Lewis, S.A.; Prabhu, P.; Dubey, A.; Patel, N. Nanostructured lipid carriers for the topical delivery of tretinoin. Eur. J. Pharm Biopharm. 2016, 108, 253–261. [Google Scholar] [CrossRef]
- Prow, T.W.; Grice, J.E.; Lin, L.L.; Faye, R.; Butler, M.; Becker, W.; Wurm, E.M.; Yoong, C.; Robertson, T.A.; Soyer, H.P.; et al. Nanoparticles and microparticles for skin drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 470–491. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Hajjar, B.; Zier, K.-I.; Khalid, N.; Azarmi, S.; Löbenberg, R. Evaluation of a microemulsion-based gel formulation for topical drug delivery of diclofenac sodium. J. Pharm. Investig. 2018, 48, 351–362. [Google Scholar] [CrossRef]
- Kelmann, R.G.; Colombo, M.; Nunes, R.J.; Simes, C.M.O.; Koester, L.S. Nanoemulsion-loaded hydrogels for topical administration of pentyl gallate. Aaps Pharmscitech 2018, 19, 2672–2678. [Google Scholar] [CrossRef] [PubMed]
- Mou, D.S.; Chen, H.B.; Du, D.R.; Mao, C.W.; Wan, H.L.; Xu, H.B.; Yang, X.L. Hydrogel-thickened nanoemulsion system for topical delivery of lipophilic drugs. Int. J. Pharm. 2008, 353, 270–276. [Google Scholar] [CrossRef]
- Zimmermann, T.; Yeates, R.A.; Laufen, H.; Pfaff, G.; Wildfeuer, A. Influence of concomitant food intake on the oral absorption of two triazole antifungal agents, itraconazole and fluconazole. Eur. J. Clin. Pharmacol. 1994, 46, 147–150. [Google Scholar] [CrossRef]
- Takahashi, H. Some problems in topical antifungals. Biomed. Ther. 1988, 20, 435–439. [Google Scholar]
- Tanuma, H.; Doi, M.; Yaguchi, A.; Ohta, Y.; Nishiyama, S.; Sekiguchi, K.; Katsuoka, K. Efficacy of oral fluconazole in tinea pedis of the hyperkeratotic type. Stratum corneum levels. Mycoses 1998, 41, 153–162. [Google Scholar] [CrossRef]
- Wildfeuer, A.; Faergemann, J.; Laufen, H.; Pfaff, G.; Zimmermann, T.; Seidl, H.P.; Lach, P. Bioavailability of fluconazole in the skin after oral medication. Mycoses 1994, 37, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Mano, Y.; Terasaka, S.; Sakurai, T.; Furuya, A.; Urano, H.; Sugibayashi, K. Usefulness of rat skin as a substitute for human skin in the in vitro skin permeation study. Exp. Anim. 2011, 60, 373–384. [Google Scholar] [CrossRef]
- Takahashi, H. Action of topical anti-trichophyton agents. Jpn. J. Dermatol. B 1972, 82, 421–437. [Google Scholar]
- Ariga, T. Activity of topical antifungals on infected sites-skin permeability and adsorption to horny materials. Nishinihon J. Dermatol. 1990, 52, 545–548. [Google Scholar] [CrossRef]
- Niwano, Y.; Kuzuhara, N.; Kanai, K.; Hamaguchi, H.; Ohmi, T. In vitro antifungal activity of lanoconazole, a topical antifungal agent. Kiso Rinsho Clin. Rep. 1996, 30, 123–130. [Google Scholar]
- Freedman, M.H.; Baxter, R.M.; Walker, G.C. In vitro sorption of griseofulvin by keratin substrates. J. Investig. Dermatol. 1962, 38, 199–208. [Google Scholar] [CrossRef]
- Hashiguchi, T.; Kodama, A.; Ryu, A.; Otagiri, M. Retention capacity of topical imidazole antifungal agents in the skin. Int. J. Pharm. 1998, 161, 195–204. [Google Scholar] [CrossRef]
- Abd, E.; Yousef, S.A.; Pastore, M.N.; Telaprolu, K.; Mohammed, Y.H.; Namjoshi, S.; Grice, J.E.; Roberts, M.S. Skin models for the testing of transdermal drugs. Clin. Pharmacol. 2016, 8, 163–176. [Google Scholar] [CrossRef]
- Wester, R.C.; Melendres, J.; Sedik, L.; Maibach, H.; Riviere, J.E. Percutaneous absorption of salicylic acid, theophylline, 2,4-dimethylamine, diethyl hexyl phthalic acid, and p-aminobenzoic acid in the isolated perfused porcine skin flap compared to man in vivo. Toxicol. Appl. Pharmacol. 1998, 151, 159–165. [Google Scholar] [CrossRef]
- Roberts, J.B. Use of Squamate Epidermis in Percutaneous-Absorption Studies—A Review. J. Toxicol. Cutan. Ocul. Toxicol. 1986, 5, 319–324. [Google Scholar] [CrossRef]
- Rigg, P.C.; Barry, B.W. Shed Snake Skin and Hairless Mouse Skin as Model Membranes for Human Skin during Permeation Studies. J. Investig. Dermatol. 1990, 94, 235–240. [Google Scholar] [CrossRef]
- Kuchler, S.; Struver, K.; Friess, W. Reconstructed skin models as emerging tools for drug absorption studies. Expert Opin. Drug Metab. Toxicol. 2013, 9, 1255–1263. [Google Scholar] [CrossRef]
- Flaten, G.E.; Palac, Z.; Engesland, A.; Filipovic-Grcic, J.; Vanic, Z.; Skalko-Basnet, N. In vitro skin models as a tool in optimization of drug formulation. Eur. J. Pharm. Sci. 2015, 75, 10–24. [Google Scholar] [CrossRef]
- Gray, G.M.; Yardley, H.J. Lipid compositions of cells isolated from pig, human, and rat epidermis. J. Lipid Res. 1975, 16, 434–440. [Google Scholar]
- Epstein, W.L. Biochemistry and Physiology of the Skin: Lowell Goldsmith, M.D., Editor, New York, 1983, Oxford University Press. Vol I, 684 Pages; vol II, 660 Pages; Elsevier: Amsterdam, The Netherlands, 1983; Volume 9, pp. 966–967. [Google Scholar]
- Lampe, M.A.; Williams, M.L.; Elias, P.M. Human epidermal lipids: Characterization and modulations during differentiation. J. Lipid Res. 1983, 24, 131–140. [Google Scholar]
- Sasaki, H.; Takahashi, T.; Mori, Y.; Nakamura, J.; Shibasaki, J. Transdermal delivery of 5-fluorouracil and its alkylcarbamoyl derivatives. Int. J. Pharm. 1990, 60, 1–9. [Google Scholar] [CrossRef]
| Excipients Tested to Evaluate the Solubility of ITZ | Solubility of ITZ (mg/mL) | ||
|---|---|---|---|
| 20 °C | 80 °C | ||
| Oils * | Labrafac® CC | 0.18 ± 0.01 | 3.62 ± 0.08 |
| Mineral oil * | Insoluble | Insoluble | |
| Paraffin oil * | Insoluble | Insoluble | |
| Emulsifiers * | Tween® 80 | 1.91 ± 0.19 | 37.53 ± 1.60 |
| Tween® 40 | 1.50 ± 0.08 | 23.35 ± 0.07 | |
| Span® 80 | 1.03 ± 0.07 | 23.68 ± 0.58 | |
| Span® 60 | Not measurable * | 22.08 ± 0.59 | |
| Glycerol esters of fatty acid * | Glyceryl mono-stearate | Not measurable * | 19.87 ± 1.11 |
| Polyglyceryl-3 methylglucose distearate | Not measurable * | 18.05 ± 1.83 | |
| Fatty acid * | Stearic acid * | Not measurable * | 14.50 ± 1.22 |
| Fatty alcohols * | Cetyl alcohol * | Not measurable * | 10.43 ± 0.63 |
| Cetostearyl alcohol * | Not measurable * | 8.32 ± 0.67 | |
| Oil phase of O/W cream formulation ** | 18.35 ± 0.12 | 32.62 ± 0.82 | |
| Phase of Cream Formulation | Ingredients | Content (%) |
|---|---|---|
| Oil phase | ITZ | 1.0 |
| Labrafac® CC | 23.0 | |
| Tween® 80 | 7.0 | |
| Glyceryl mono-stearate | 4.0 | |
| Stearic acid | 10.0 | |
| Cetyl alcohol | 6.0 | |
| Water phase | Propylene glycol | 9.0 |
| Distilled water | 40.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Jung, S.; Yeo, S.; Kim, D.; Na, Y.C.; Yun, G.; Lee, J. Characteristics of Skin Deposition of Itraconazole Solubilized in Cream Formulation. Pharmaceutics 2019, 11, 195. https://doi.org/10.3390/pharmaceutics11040195
Kim H, Jung S, Yeo S, Kim D, Na YC, Yun G, Lee J. Characteristics of Skin Deposition of Itraconazole Solubilized in Cream Formulation. Pharmaceutics. 2019; 11(4):195. https://doi.org/10.3390/pharmaceutics11040195
Chicago/Turabian StyleKim, Hyeongmin, Sukkyun Jung, Sooho Yeo, Dohyun Kim, Young Chae Na, Gyiae Yun, and Jaehwi Lee. 2019. "Characteristics of Skin Deposition of Itraconazole Solubilized in Cream Formulation" Pharmaceutics 11, no. 4: 195. https://doi.org/10.3390/pharmaceutics11040195
APA StyleKim, H., Jung, S., Yeo, S., Kim, D., Na, Y. C., Yun, G., & Lee, J. (2019). Characteristics of Skin Deposition of Itraconazole Solubilized in Cream Formulation. Pharmaceutics, 11(4), 195. https://doi.org/10.3390/pharmaceutics11040195