Comparison of Salbutamol Delivery Efficiency for Jet versus Mesh Nebulizer Using Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Nebulizers
2.2. Animals
2.3. Size of Nebulized Aerosol Droplets
2.4. Residual Volume, Nebulization Time and Output Rate in Each Nebulizer
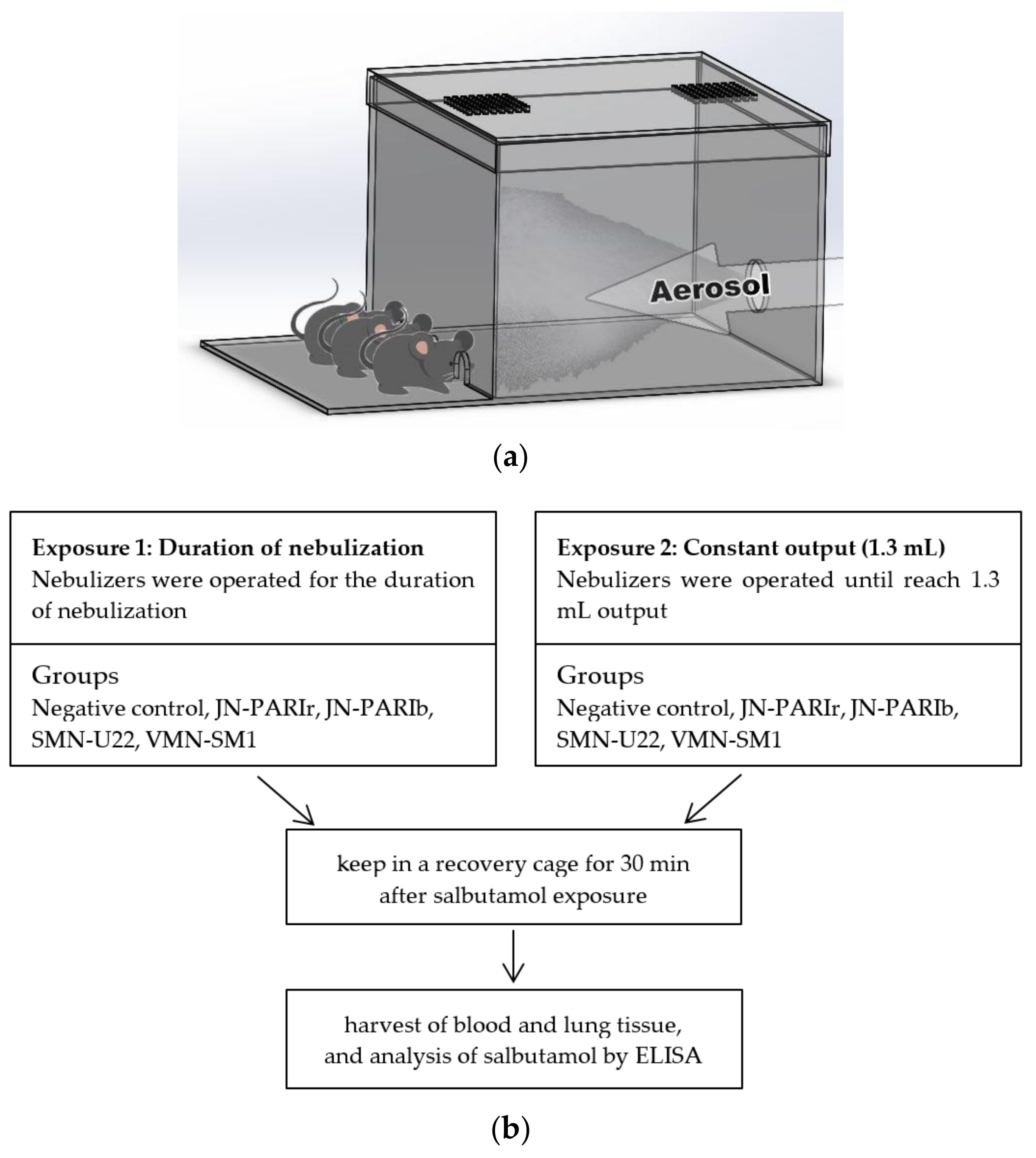
2.5. Exposure of Salbutamol to Mice
2.6. Blood and Tissue Collection
2.7. Determination of the Level of Salbutamol in Serum and Lung Tissues
2.8. In Vivo Lung Imaging Using ICG Nebulization
2.9. Statistical Analyses
3. Results
3.1. Measurement of Particle Size and Performance of Nebulizer
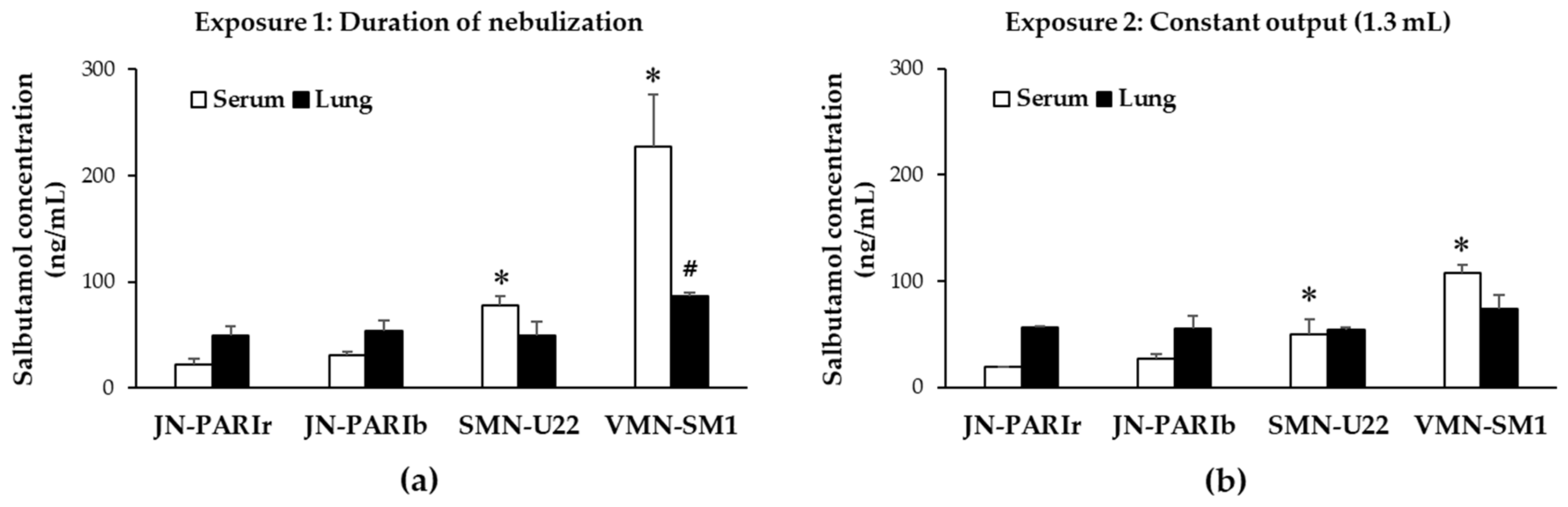
3.2. Delivery Efficiency of Salbutamol in Mice
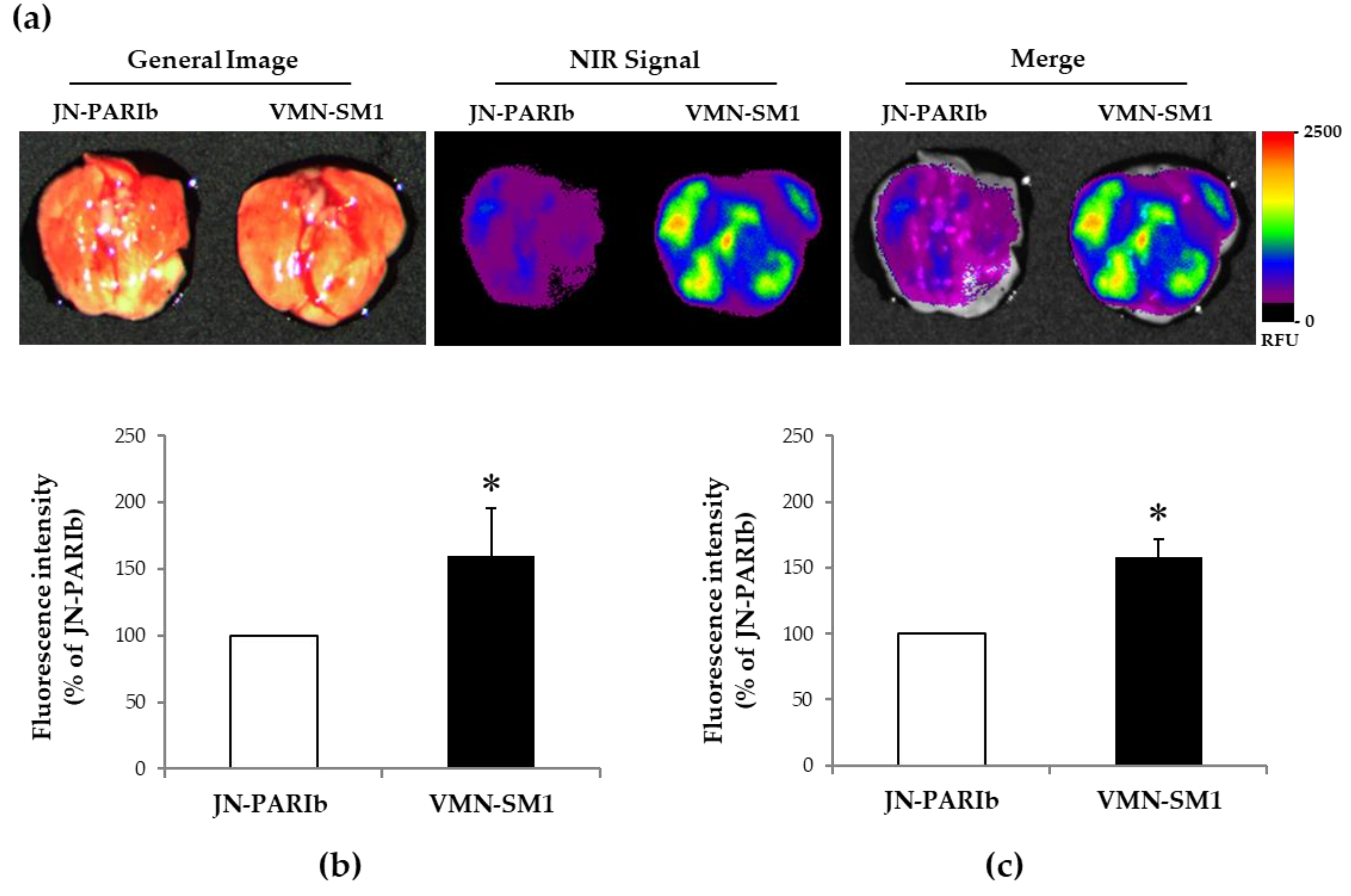
3.3. ICG Delivery in Mice Lung Tissue
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Goralski, J.L.; Davis, S.D. Breathing easier: Addressing the challenges of aerosolizing medications to infants and preschoolers. Respir. Med. 2014, 108, 1069–1074. [Google Scholar] [CrossRef]
- Mansour, M.M. Overcoming jet lag: Optimizing aerosol delivery with and without jet nebulizers. Respir. Care 2013, 58, 1255–1256. [Google Scholar] [CrossRef] [PubMed]
- Ari, A. Jet, Ultrasonic, and Mesh Nebulizers: An Evaluation of Nebulizers for Better Clinical Outcomes. Eurasian J. Pulmonol. 2014, 16, 1–7. [Google Scholar] [CrossRef]
- Ibrahim, M.; Verma, R.; Garcia-Contreras, L. Inhalation drug delivery devices: Technology update. Med. Devices 2015, 12, 131–139. [Google Scholar]
- Clay, M.M.; Pavia, D.; Newman, S.P.; Clarke, S.W. Factors influencing the size distribution of aerosols from jet nebulisers. Thorax 1983, 38, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.C.; Denyer, J.; Kendrick, A.H. Comparison of twenty three nebulizer/compressor combinations for domiciliary use. Eur. Respir. J. 1995, 8, 1214–1221. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, A.H.; Smith, E.C.; Denyer, J. Nebulizers-fill volume, residual volume and matching of nebulizer to compressor. Respir. Med. 1995, 89, 157–159. [Google Scholar] [CrossRef]
- Dhand, R. Nebulizers that use a vibrating mesh or plate with multiple apertures to generate aerosol. Respir. Care 2002, 47, 1406–1416. [Google Scholar] [PubMed]
- Waldrep, J.C.; Dhand, R. Advanced nebulizer designs employing vibrating mesh/aperture plate technologies for aerosol generation. Curr. Drug Deliv. 2008, 5, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.L.; Denk, O.; Leung, K. Higher tobramycin concentration and vibrating mesh technology can shorten antibiotic treatment time in cystic fibrosis. Pediatr. Pulmonol. 2011, 46, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Michotte, J.B.; Jossen, E.; Roeseler, J.; Liistro, G.; Reychler, G. In vitro comparison of five nebulizers during noninvasive ventilation: Analysis of inhaled and lost doses. J. Aerosol Med. Pulm. Drug Deliv. 2014, 27, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Vecellio, L. The mesh nebuliser: A recent technical innovation for aerosol delivery. Breathe 2006, 2, 252–260. [Google Scholar] [CrossRef]
- Rabea, H.; Ali, A.M.; Salah Eldin, R.; Abdelrahman, M.M.; Said, A.; Abdelrahim, M.E. Modelling of in-vitro and in-vivo performance of aerosol emitted from different vibrating mesh nebulisers in non-invasive ventilation circuit. Eur. J. Pharm. Sci. 2017, 97, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Ari, A.; Atalay, O.T.; Harwood, R.; Sheard, M.M.; Aljamhan, E.A.; Fink, J.B. Influence of nebulizer type, position, and bias flow on aerosol drug delivery in simulated pediatric and adult lung models during mechanical ventilation. Respir. Care 2010, 55, 845–851. [Google Scholar]
- Alhamad, B.R.; Fink, J.B.; Harwood, R.J.; Sheard, M.M.; Ari, A. Effect of Aerosol Devices and Administration Techniques on Drug Delivery in a Simulated Spontaneously Breathing Pediatric Tracheostomy Model. Respir. Care 2015, 60, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Ari, A.; de Andrade, A.D.; Sheard, M.; AIHamad, B.; Fink, J.B. Performance Comparisons of Jet and Mesh Nebulizers Using Different Interfaces in Simulated Spontaneously Breathing Adults and Children. J. Aerosol Med. Pulm. Drug Deliv. 2015, 28, 281–289. [Google Scholar] [CrossRef]
- Dugernier, J.; Hesse, M.; Jumetz, T.; Bialais, E.; Roeseler, J.; Depoortere, V.; Michotte, J.B.; Wittebole, X.; Ehrmann, S.; Laterre, P.F.; et al. Aerosol Delivery with Two Nebulizers Through High-Flow Nasal Cannula: A Randomized Cross-Over Single-Photon Emission Computed Tomography-Computed Tomography Study. J. Aerosol Med. Pulm. Drug Deliv. 2017, 30, 349–358. [Google Scholar] [CrossRef]
- Dubus, J.C.; Vecellio, L.; De Monte, M.; Fink, J.B.; Grimbert, D.; Montharu, J.; Valat, C.; Behan, N.; Diot, P. Aerosol deposition in neonatal ventilation. Pediatr. Res. 2005, 58, 10–14. [Google Scholar] [CrossRef]
- Galindo-Filho, V.C.; Ramos, M.E.; Rattes, C.S.; Barbosa, A.K.; Brandão, D.C.; Brandão, S.C.; Fink, J.B.; de Andrade, A.D. Radioaerosol Pulmonary Deposition Using Mesh and Jet Nebulizers During Noninvasive Ventilation in Healthy Subjects. Respir. Care 2015, 60, 1238–1246. [Google Scholar] [CrossRef]
- Murayama, N.; Murayama, K. Comparison of the Clinical Efficacy of Salbutamol with Jet and Mesh Nebulizers in Asthmatic Children. Pulm. Med. 2018, 2018, 6. [Google Scholar] [CrossRef]
- Dunne, R.B.; Shortt, S. Comparison of bronchodilator administration with vibrating mesh nebulizer and standard jet nebulizer in the emergency department. Am. J. Emerg. Med. 2018, 36, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Hu, J.; Zhan, S.; Zhang, R.; Tan, W. Effects of Temperature and Humidity on Laser Diffraction Measurements to Jet Nebulizer and Comparison with NGI. AAPS PharmSciTech 2016, 17, 380–388. [Google Scholar] [CrossRef]
- Technical Committee ISO/TC 121/SC 2. ISO 27427:2013, Anaesthetic and respiratory equipment-Nebulizing Systems and Components; International Organization for Standardization ISO Central Secretariat: Geneva, Switzerland, 2013. [Google Scholar]
- Tandon, R.; McPeck, M.; Smaldone, G.C. Measuring Nebulizer Output Aerosol Production vs Gravimetric Analysis. Chest 1997, 111, 1361–1365. [Google Scholar] [CrossRef] [PubMed]
- Sevick-Muraca, E.M. Translation of near-infrared fluorescence imaging technologies: Emerging clinical applications. Annu. Rev. Med. 2012, 63, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Marshall, M.V.; Rasmussen, J.C.; Tan, I.C.; Aldrich, M.B.; Adams, K.E.; Wang, X.; Fife, C.E.; Maus, E.A.; Smith, L.A.; Sevick-Muraca, E.M. Near-infrared fluorescence imaging in humans with indocyanine green: A review and update. Open Surg. Oncol. J. 2010, 2, 12. [Google Scholar] [CrossRef]
- Kassab, G.; Geralde, M.C.; Inada, N.M.; Achiles, A.E.; Guerra, V.G.; Bagnato, V.S. Nebulization as a tool for photosensitizer delivery to the respiratory tract. J. Biophotonics 2019, 12, 10. [Google Scholar] [CrossRef] [PubMed]
- Kassab, G.; Geralde, M.C.; Inada, N.M.; Bagnato, V.S. Fluorescence assessment of the delivery and distribution of nebulized. In Proceedings of the Photo-Optical Instrumentation Engineers (SPIE); SPIE: San Francisco, CA, USA, 2018; Volume 10475, p. 104750. [Google Scholar]
- Usmani, O.S.; Biddiscombe, M.F.; Barnes, P.J. Regional lung deposition and bronchodilator response as a function of beta2-agonist particle size. Am. J. Respir. Crit. Care Med. 2005, 172, 1497–1504. [Google Scholar] [CrossRef]
- Khilnani, G.C.; Banga, A. Aerosol therapy. Indian J. Chest Dis. Allied Sci. 2008, 50, 209. [Google Scholar]
- Nikander, K.; Denyer, J.; Everard, M.; Smaldone, G.C. Validation of a new breathing simulator generating and measuring inhaled aerosol with adult breathing patterns. J. Aerosol Med. 2000, 13, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Nikander, K.; Denyer, J.; Smith, N.; Wollmer, P. Breathing patterns and aerosol delivery: Impact of regular human patterns, and sine and square waveforms on rate of delivery. J. Aerosol Med. 2001, 14, 327–333. [Google Scholar] [CrossRef]
- Sangwan, S.; Condos, R.; Smaldone, G.C. Lung deposition and respirable mass during wet nebulization. J. Aerosol Med. 2003, 16, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Bosco, A.P.; Rhem, R.G.; Dolovich, M.B. In vitro estimations of in vivo jet nebulizer efficiency using actual and simulated tidal breathing patterns. J. Aerosol Med. 2005, 18, 427–438. [Google Scholar] [CrossRef]
- Rissler, J.; Gudmundsson, A.; Nicklasson, H.; Swietlicki, E.; Wollmer, P.; Löndahl, J. Deposition efficiency of inhaled particles (15-5000 nm) related to breathing pattern and lung function: An experimental study in healthy children and adults. Part. Fibre Toxicol. 2017, 14, 10. [Google Scholar] [CrossRef]
- Linner, R.; Perez-de-Sa, V.; Cunha-Goncalves, D. Lung deposition of nebulized surfactant in newborn piglets. Neonatology 2015, 107, 277–282. [Google Scholar] [CrossRef]
- Alander, J.T.; Kaartinen, I.; Laakso, A.; Pätilä, T.; Spillmann, T.; Tuchin, V.V.; Venermo, M.; Välisuo, P. A review of indocyanine green fluorescent imaging in surgery. Int. J. Biomed. Imaging 2012, 2012, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Landsman, M.L.; Kwant, G.; Mook, G.A.; Zijlstra, W.G. Light-absorbing properties, stability, and spectral stabilization of indocyanine green. J. Appl. Physiol. 1976, 40, 575–583. [Google Scholar] [CrossRef]
- Gawchik, S.M.; Saccar, C.L.; Noonan, M.; Reasner, D.S.; DeGraw, S.S. The safety and efficacy of nebulized levalbuterol compared with racemic albuterol and placebo in the treatment of asthma in pediatric patients. J. Allergy Clin. Immunol. 1999, 103, 615–621. [Google Scholar] [CrossRef]
- Usmani, O.S.; Biddiscombe, M.F.; Yang, S.; Meah, S.; Oballa, E.; Simpson, J.K.; Fahy, W.A.; Marshall, R.P.; Lukey, P.T.; Maher, T.M. The topical study of inhaled drug (salbutamol) delivery in idiopathic pulmonary fibrosis. Respir. Res. 2018, 19, 25. [Google Scholar] [CrossRef] [PubMed]
| Mode of Operation | Model | Abbreviation in the Study |
|---|---|---|
| Jet | PARI BOY SX-red nozzle (PARI GmbH, Starnberg, Germany) | JN-PARIr |
| PARI BOY SX-blue nozzle (PARI GmbH, Starnberg, Germany) | JN-PARIb | |
| Static mesh | NE-U22 (Omron Healthcare, Kyoto, Japan) | SMN-U22 |
| Vibrating mesh | NE-SM1 NEPLUS (KTMED Co., Seoul, Korea) | VMN-SM1 |
| Device | The Fifty Percent Volume Diameter (Dv(50) (μm)) | |
|---|---|---|
| 1 mg/mL Salbutamol | 1.25 mg/mL Salbutamol | |
| JN-PARIr | 3.40 ± 0.09 | 3.53 ± 0.05 |
| JN-PARIb | 4.54 ± 0.03 | 4.54 ± 0.01 |
| SMN-U22 | 6.83 ± 0.09 | 6.86 ± 0.07 |
| VMN-SM1 | 5.34 ± 0.18 | 5.51 ± 0.12 |
| Device | Residual Volume (mL) | Nebulization Time (min) | Output Rate (mL/min) | |||
|---|---|---|---|---|---|---|
| 1 mg/mL Salbutamol | 1.25 mg/mL Salbutamol | 1 mg/mL Salbutamol | 1.25 mg/mL Salbutamol | 1 mg/mL Salbutamol | 1.25 mg/mL Salbutamol | |
| JN-PARIr | 0.541 ± 0.021 | 0.536 ± 0.047 | 9.24 ± 0.05 | 9.33 ± 0.08 | 0.155 ± 0.002 | 0.153 ± 0.004 |
| JN-PARIb | 0.691 ± 0.011 | 0.684 ± 0.008 | 7.06 ± 0.05 | 7.10 ± 0.05 | 0.185 ± 0.002 | 0.184 ± 0.002 |
| SMN-U22 | 0.039 ± 0.006 | 0.034 ± 0.008 | 6.48 ± 0.03 | 6.50 ± 0.09 | 0.288 ± 0.002 | 0.288 ± 0.006 |
| VMN-SM1 | 0.041 ± 0.003 | 0.040 ± 0.002 | 8.16 ± 0.06 | 8.13 ± 0.06 | 0.237 ± 0.003 | 0.239 ± 0.003 |
| Device | Nebulization Time (min) |
|---|---|
| JN-PARIr | 8.12 ± 0.007 |
| JN-PARIb | 7.05 ± 0.007 |
| SMN-U22 | 4.34 ± 0.003 |
| VMN-SM1 | 5.45 ± 0.003 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, K.H.; Moon, S.-H.; Oh, J.Y.; Yoon, Y.-S.; Gu, N.; Lim, C.-Y.; Park, B.J.; Nam, K.C. Comparison of Salbutamol Delivery Efficiency for Jet versus Mesh Nebulizer Using Mice. Pharmaceutics 2019, 11, 192. https://doi.org/10.3390/pharmaceutics11040192
Chang KH, Moon S-H, Oh JY, Yoon Y-S, Gu N, Lim C-Y, Park BJ, Nam KC. Comparison of Salbutamol Delivery Efficiency for Jet versus Mesh Nebulizer Using Mice. Pharmaceutics. 2019; 11(4):192. https://doi.org/10.3390/pharmaceutics11040192
Chicago/Turabian StyleChang, Kyung Hwa, Sang-Hyub Moon, Jin Young Oh, Young-Soon Yoon, Namyi Gu, Chi-Yeon Lim, Bong Joo Park, and Ki Chang Nam. 2019. "Comparison of Salbutamol Delivery Efficiency for Jet versus Mesh Nebulizer Using Mice" Pharmaceutics 11, no. 4: 192. https://doi.org/10.3390/pharmaceutics11040192
APA StyleChang, K. H., Moon, S.-H., Oh, J. Y., Yoon, Y.-S., Gu, N., Lim, C.-Y., Park, B. J., & Nam, K. C. (2019). Comparison of Salbutamol Delivery Efficiency for Jet versus Mesh Nebulizer Using Mice. Pharmaceutics, 11(4), 192. https://doi.org/10.3390/pharmaceutics11040192





