In Vitro Methods to Study Colon Release: State of the Art and An Outlook on New Strategies for Better In-Vitro Biorelevant Release Media
Abstract
1. Introduction
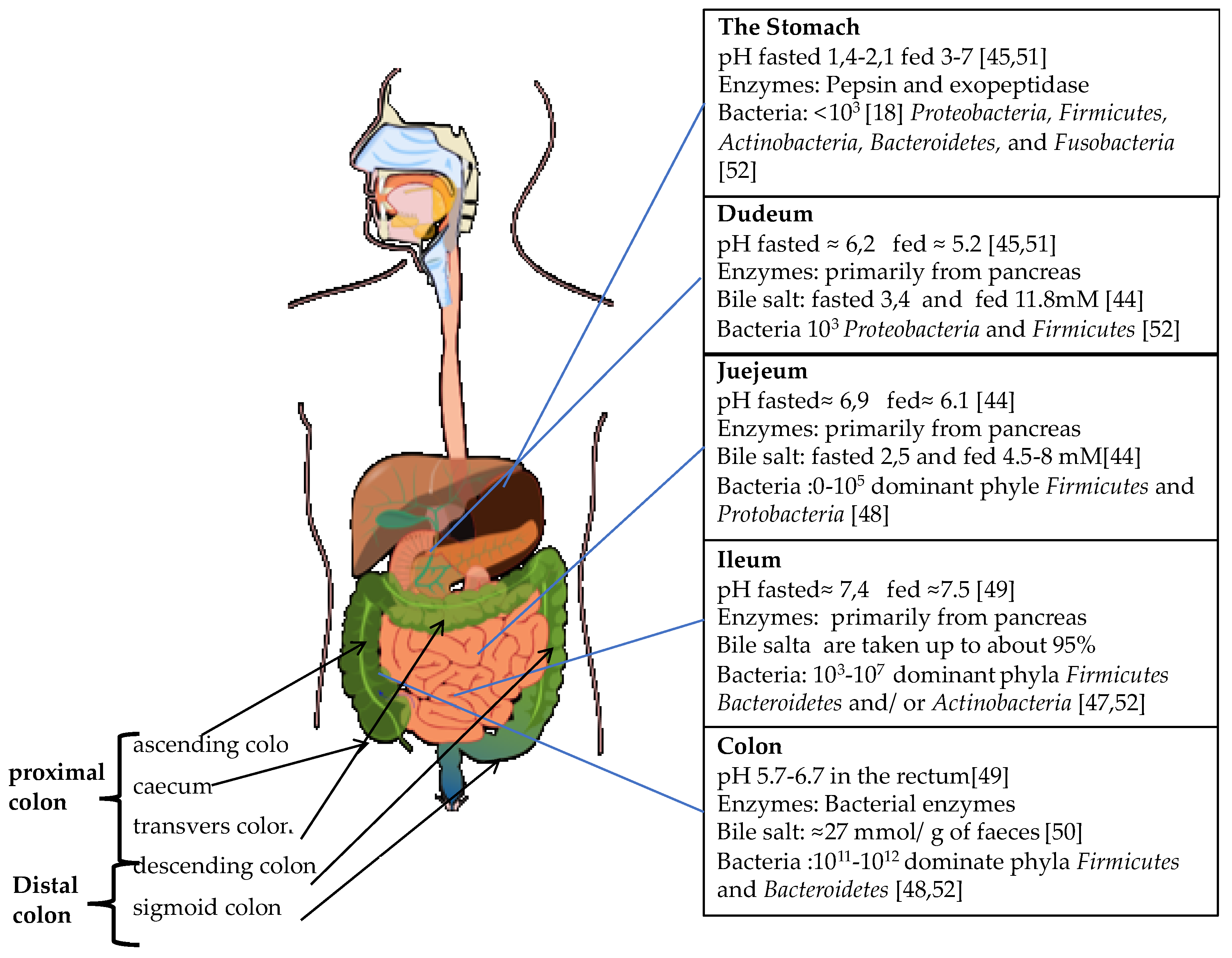
2. The GI Tract
2.1. The Upper GI-Tract
2.2. The Physiology of Colon
3. In Vitro Release Methods
Colon Release
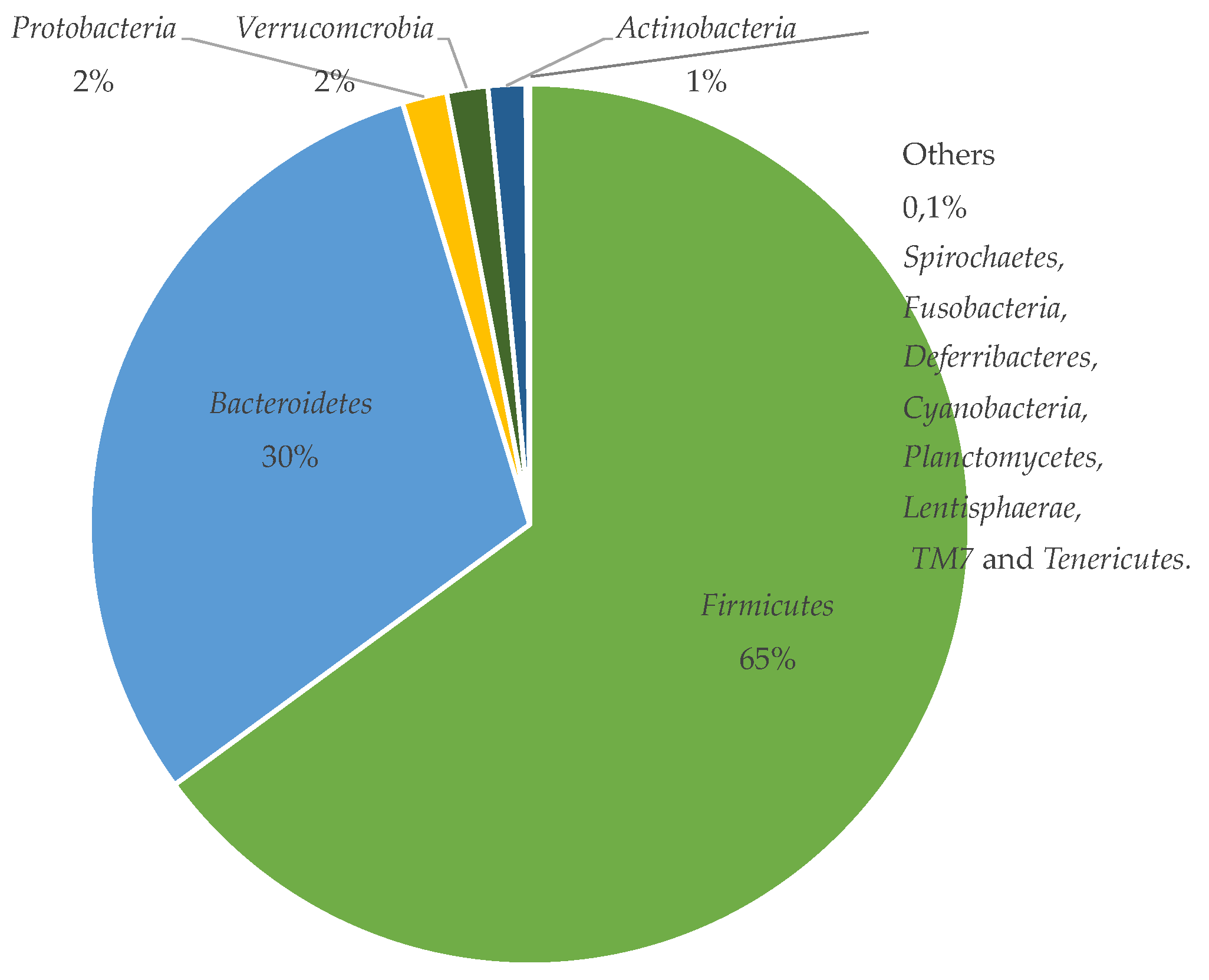
4. The Gut Microbiota
5. Can a Novel Media Based on the Microbiota be Developed?
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Kotla, N.G.; Rana, S.; Sivaraman, G.; Sunnapu, O.; Vemula, P.K.; Pandit, A.; Rochev, Y. Bioresponsive drug delivery systems in intestinal inflammation: State-of-the-art and future perspectives. Adv. Drug Deliv. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Sinha, V.R. Current pharmaceutical strategies for efficient site specific delivery in inflamed distal intestinal mucosa. J. Control. Release 2018, 272, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Tuleu, C.; Basit, A.; Waddington, W.; Ell, P.; Newton, J. Colonic delivery of 4-aminosalicylic acid using amylose-ethylcellulose-coated hydroxypropylmethylcellulose capsules. Aliment. Pharmacol. Ther. 2002, 16, 1771–1779. [Google Scholar] [CrossRef] [PubMed]
- Milojevic, S.; Newton, J.M.; Cummings, J.H.; Gibson, G.R.; Botham, R.L.; Ring, S.G.; Stockham, M.; Allwood, M.C. Amylose as a coating for drug delivery to the colon: Preparation and in vitro evaluation using 5-aminosalicylic acid pellets. J. Control. Release 1996, 38, 75–84. [Google Scholar] [CrossRef]
- Leopold, C.S.; Eikeler, D. Eudragit E as coating material for the pH-controlled drug release in the topical treatment of inflammatory bowel disease (IBD). J Drug Target 1998, 6, 85–94. [Google Scholar] [CrossRef]
- Wang, Q.S.; Wang, G.F.; Zhou, J.; Gao, L.N.; Cui, Y.L. Colon targeted oral drug delivery system based on alginate-chitosan microspheres loaded with icariin in the treatment of ulcerative colitis. Int. J. Pharm. 2016, 515, 176–185. [Google Scholar] [CrossRef]
- Andishmand, H.; Hamishehkar, H.; Babazadeh, A.; Taghvimi, A.; Mohammadifar, M.A.; Tabibiazar, M. A Colon Targeted Delivery System for Resveratrol Enriching in pH Responsive-Model. Pharm. Sci. 2017, 23, 42–49. [Google Scholar] [CrossRef]
- Gulbake, A.; Jain, A.; Jain, A.; Jain, A.; Jain, S.K. Insight to drug delivery aspects for colorectal cancer. World J. Gastroenterol. 2016, 22, 582–599. [Google Scholar] [CrossRef]
- Krishnaiah, Y.S.R.; Veer Raju, P.; Dinesh Kumar, B.; Bhaskar, P.; Satyanarayana, V. Development of colon targeted drug delivery systems for mebendazole. J. Control. Release 2001, 77, 87–95. [Google Scholar] [CrossRef]
- Blemur, L.; Le, T.C.; Marcocci, L.; Pietrangeli, P.; Mateescu, M.A. Carboxymethyl starch/alginate microspheres containing diamine oxidase for intestinal targeting. Biotechnol. Appl. Biochem. 2016, 63, 344–353. [Google Scholar] [CrossRef]
- Del Curto, M.; Maroni, A.; Foppoli, A.; Zema, L.; Gazzaniga, A.; Sangalli, M. Preparation and evaluation of an oral delivery system for time-dependent colon release of insulin and selected protease inhibitor and absorption enhancer compounds. J. Pharm. Sci. 2009, 98, 4661–4669. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Yu, H.; Zhang, Y.; Feng, H.; Hoag, S.W. A Multiparticulate Delivery System for Potential Colonic Targeting Using Bovine Serum Albumin as a Model Protein: Theme: Formulation and Manufacturing of Solid Dosage Forms Guest Editors: Tony Zhou and Tonglei Li. Pharm. Res. 2017, 34, 2663–2674. [Google Scholar] [CrossRef] [PubMed]
- Calinescu, C.; Mateescu, M. Carboxymethyl high amylose starch: Chitosan self-stabilized matrix for probiotic colon delivery. Eur. J. Pharm. Biopharm. 2008, 70, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Dodoo, C.C.; Wang, J.; Basit, A.W.; Stapleton, P.; Gaisford, S. Targeted delivery of probiotics to enhance gastrointestinal stability and intestinal colonisation. Int. J. Pharm. 2017, 530, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S. Microbiota Replacement Therapies: Innovation in Gastrointestinal Care. Clin. Pharmacol. Ther. 2018, 103, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Philip, A.K.; Philip, B. Colon targeted drug delivery systems: A review on primary and novel approaches. Oman Med. J. 2010, 25, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Chourasia, M.K.; Jain, S.K. Polysaccharides for colon targeted drug delivery. Drug Deliv. 2004, 11, 129–148. [Google Scholar] [CrossRef] [PubMed]
- Vandamme, T.F.; Lenourry, A.; Charreau, C.; Chaumeil, J.-C. The use of polysaccharides to target drugs to colon. Carbohydr. Polym. 2002, 48, 219–231. [Google Scholar] [CrossRef]
- Basit, A.; Short, M.; McConnell, E. Microbiota-triggered colonic delivery: Robustness of the polysaccharide approach. J. Drug Target. 2009, 17, 64–71. [Google Scholar] [CrossRef]
- Freire, C.; Podczeck, F.; Veiga, F.; Sousa, J. Starch-based coatings for colon-specific delivery. Part II: Physicochemical properties and in vitro drug release from high amylose maize starch films. Eur. J. Pharm. Biopharm. 2009, 72, 587–594. [Google Scholar] [CrossRef]
- Chen, J.; Li, X.; Chen, L.; Xie, F. Starch film-coated microparticles for oral colon-specific drug delivery. Carbohydr. Polym. 2018, 191, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Shi, Y.; Jiang, G.; Liu, W.; Han, H.; Feng, Q.; Ren, J.; Yuan, Y.; Wang, Y.; Shi, J.; et al. Smart nanocomposite hydrogels based on azo crosslinked graphene oxide for oral colon-specific drug delivery. Nanotechnology 2016, 27, 315105. [Google Scholar] [CrossRef] [PubMed]
- Sinha, V.R.; Kumria, R. Polysaccharides in colon-specific drug delivery. Int. J. Pharm. 2001, 224, 19–38. [Google Scholar] [CrossRef]
- Forbes, J.D.; Chen, C.Y.; Knox, N.C.; Marrie, R.A.; El-Gabalawy, H.; de Kievit, T.; Alfa, M.; Bernstein, C.N.; Van Domselaar, G. A comparative study of the gut microbiota in immune-mediated inflammatory diseases-does a common dysbiosis exist? Microbiome 2018, 6, 221. [Google Scholar] [CrossRef] [PubMed]
- Maroni, A.; Zema, L.; Cerea, M.; Foppoli, A.; Palugan, L.; Gazzaniga, A. Erodible drug delivery systems for time-controlled release into the gastrointestinal tract. J. Drug Deliv. Sci. Technol. 2016, 32, 229–235. [Google Scholar] [CrossRef]
- Sangalli, M.E.; Maroni, A.; Zema, L.; Busetti, C.; Giordano, F.; Gazzaniga, A. In vitro and in vivo evaluation of an oral system for time and/or site-specific drug delivery. J. Control. Release 2001, 73, 103–110. [Google Scholar] [CrossRef]
- Hu, M.Y.; Peppercorn, M.A. MMX mesalamine: A novel high-dose, once-daily 5-aminosalicylate formulation for the treatment of ulcerative colitis. Expert Opin. Pharmacother. 2008, 9, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Lentle, R.G.; Janssen, P.W.M. Physical characteristics of digesta and their influence on flow and mixing in the mammalian intestine: A review. J. Comp. Physiol. B 2008, 178, 673–690. [Google Scholar] [CrossRef]
- Shameem, M.; Katori, N.; Aoyagi, N.; Kojima, S. Oral Solid Controlled Release Dosage Forms: Role of GI-Mechanical Destructive Forces and Colonic Release in Drug Absorption Under Fasted and Fed Conditions in Humans. Pharm. Res. 1995, 12, 1049–1054. [Google Scholar] [CrossRef]
- Takahashi, T. Flow Behavior of Digesta and the Absorption of Nutrients in the Gastrointestine. J. Nutr. Sci. Vitaminol. 2011, 57, 265–273. [Google Scholar] [CrossRef]
- McConnell, E.; Short, M.; Basit, A. An in vivo comparison of intestinal pH and bacteria as physiological trigger. J. Control. Release 2008, 130, 154–160. [Google Scholar] [CrossRef]
- Li, J.; Yang, L.; Ferguson, S.M.; Hudson, T.J.; Watanabe, S.; Katsuma, M.; Fix, J.A. In vitro evaluation of dissolution behavior for a colon-specific drug delivery system (CODES™) in multi-pH media using United States Pharmacopeia apparatus II and III. AAPS PharmSciTech 2002, 3, 59. [Google Scholar] [CrossRef] [PubMed]
- Akhgari, A.; Heshmati, Z.; Afrasiabi Garekani, H.; Sadeghi, F.; Sabbagh, A.; Sharif Makhmalzadeh, B.; Nokhodchi, A. Indomethacin electrospun nanofibers for colonic drug delivery: In vitro dissolution studies. Colloids Surf. B 2017, 152, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Handali, S.; Moghimipour, E.; Rezaei, M.; Kouchak, M.; Ramezani, Z.; Dorkoosh, F.A. In vitro and in vivo evaluation of coated capsules for colonic delivery. J. Drug Deliv. Sci. Technol. 2018, 47, 492–498. [Google Scholar] [CrossRef]
- Oshi, M.A.; Naeem, M.; Bae, J.; Kim, J.; Lee, J.; Hasan, N.; Kim, W.; Im, E.; Jung, Y.; Yoo, J.-W. Colon-targeted dexamethasone microcrystals with pH-sensitive chitosan/alginate/Eudragit S multilayers for the treatment of inflammatory bowel disease. Carbohydr. Polym. 2018, 198, 434–442. [Google Scholar] [CrossRef]
- Linares, V.; Casas, M.; Caraballo, I. Printfills: 3D printed systems combining fused deposition modeling and injection volume filling. Application to colon-specific drug delivery. Eur. J. Pharm. Biopharm. 2019, 134, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Maroni, A.; Moutaharrik, S.; Zema, L.; Gazzaniga, A. Enteric coatings for colonic drug delivery: State of the art. Expert Opin. Drug Deliv. 2017, 14, 1027–1029. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Kimura, G.; Mawatari, S.-S.; Shimokawa, T.; Yoshikawa, Y.; Takada, K. New preparation method of intestinal pressure-controlled colon delivery capsules by coating machine and evaluation in beagle dogs. J. Control. Release 1998, 56, 293–302. [Google Scholar] [CrossRef]
- Ishibashi, T.; Pitcairn, G.R.; Yoshino, H.; Mizobe, M.; Wilding, I.R. Scintigraphic Evaluation of a New Capsule-Type Colon Specific Drug Delivery System in Healthy Volunteers. J. Pharm. Sci. 1998, 87, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, M.; Pagliaro, G.; Demedts, I.; Deloose, E.; Gevers, A.; Scheerens, C.; Rommel, N.; Tack, J. Pan-Colonic Pressurizations Associated With Relaxation of the Anal Sphincter in Health and Disease: A New Colonic Motor Pattern Identified Using High-Resolution Manometry. Am. J. Gastroenterol. 2017, 112, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Dinning, P.G.; Wiklendt, L.; Maslen, L.; Gibbins, I.; Patton, V.; Arkwright, J.W.; Lubowski, D.Z.; O’Grady, G.; Bampton, P.A.; Brookes, S.J.; et al. Quantification of in vivo colonic motor patterns in healthy humans before and after a meal revealed by high-resolution fiber-optic manometry. Neurogastroenterol. Motil. 2014, 26, 1443–1457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Langer, R.; Traverso, G. Nanoparticulate drug delivery systems targeting inflammation for treatment of inflammatory bowel disease. Nano Today 2017, 16, 82–96. [Google Scholar] [CrossRef]
- Hua, S.; Marks, E.; Schneider, J.J.; Keely, S. Advances in oral nano-delivery systems for colon targeted drug delivery in inflammatory bowel disease: Selective targeting to diseased versus healthy tissue. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1117–1132. [Google Scholar] [CrossRef] [PubMed]
- Vass, P.; Démuth, B.; Hirsch, E.; Nagy, B.; Andersen, S.K.; Vigh, T.; Verreck, G.; Csontos, I.; Nagy, Z.K.; Marosi, G. Drying technology strategies for colon-targeted oral delivery of biopharmaceuticals. J. Control. Release 2019, 296, 162–178. [Google Scholar] [CrossRef] [PubMed]
- Benshitrit, R.C.; Levi, C.S.; Tal, S.L.; Shimoni, E.; Lesmes, U. Development of oral food-grade delivery systems: Current knowledge and future challenges. Food Funct. 2012, 3, 10–21. [Google Scholar] [CrossRef]
- Sagalowicz, L.; Leser, M.E. Delivery systems for liquid food products. Curr. Opin. Colloid Interf. Sci. 2010, 15, 61–72. [Google Scholar] [CrossRef]
- Koziolek, M.; Grimm, M.; Becker, D.; Iordanov, V.; Zou, H.; Shimizu, J.; Wanke, C.; Garbacz, G.; Weitschies, W. Investigation of pH and Temperature Profiles in the GI Tract of Fasted Human Subjects Using the Intellicap((R)) System. J. Pharm. Sci. 2015, 104, 2855–2863. [Google Scholar] [CrossRef]
- Russell, T.L.; Berardi, R.R.; Barnett, J.L.; Dermentzoglou, L.C.; Jarvenpaa, K.M.; Schmaltz, S.P.; Dressman, J.B. Upper Gastrointestinal pH in Seventy-Nine Healthy, Elderly, North American Men and Women. Pharm. Res. 1993, 10, 187–196. [Google Scholar] [CrossRef]
- Helander, H.F.; Fändriks, L. Surface area of the digestive tract—Revisited. Scand. J. Gastroenterol. 2014, 49, 681–689. [Google Scholar] [CrossRef]
- Bergstrom, C.A.; Holm, R.; Jorgensen, S.A.; Andersson, S.B.; Artursson, P.; Beato, S.; Borde, A.; Box, K.; Brewster, M.; Dressman, J.; et al. Early pharmaceutical profiling to predict oral drug absorption: Current status and unmet needs. Eur. J. Pharm. Sci. 2014, 57, 173–199. [Google Scholar] [CrossRef]
- Pentafragka, C.; Symillides, M.; McAllister, M.; Dressman, J.; Vertzoni, M.; Reppas, C. The impact of food intake on the luminal environment and performance of oral drug products with a view to in vitro and in silico simulations: A PEARRL review. J. Pharm. Pharmacol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ridlon, J.M.; Kang, D.J.; Hylemon, P.B. Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 2006, 47, 241–259. [Google Scholar] [CrossRef] [PubMed]
- Villmones, H.C.; Haug, E.S.; Ulvestad, E.; Grude, N.; Stenstad, T.; Halland, A.; Kommedal, O. Species Level Description of the Human Ileal Bacterial Microbiota. Sci. Rep. 2018, 8, 4736. [Google Scholar] [CrossRef] [PubMed]
- Sundin, O.H.; Mendoza-Ladd, A.; Zeng, M.; Diaz-Arevalo, D.; Morales, E.; Fagan, B.M.; Ordonez, J.; Velez, P.; Antony, N.; McCallum, R.W. The human jejunum has an endogenous microbiota that differs from those in the oral cavity and colon. BMC Microbiol. 2017, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Fallingborg, J. Intraluminal pH of the human gastrointestinal tract. Dan. Med. Bull. 1999, 46, 183–196. [Google Scholar] [PubMed]
- Mudd, D.G.; Mckelvey, S.T.D.; Norwood, W.; Elmore, D.T.; Roy, A.D. Faecal bile acid concentrations of patients with carcinomaor increased risk of carcinoma in the large bowel. Gut 1980, 21, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Dressman, J.B.; Berardi, R.R.; Dermentzoglou, L.C.; Russell, T.L.; Schmaltz, S.P.; Barnett, J.L.; Jarvenpaa, K.M. Upper Gastrointestinal (GI) pH in Young, Healthy Men and Women. Pharm. Res. 1990, 7, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Hakansson, A.; Molin, G. Gut Microbiota and Inflammation. Nutrients 2011, 3, 637–682. [Google Scholar] [CrossRef]
- Walker, A.W.; Ince, J.; Duncan, S.H.; Webster, L.M.; Holtrop, G.; Ze, X.; Brown, D.; Stares, M.D.; Scott, P.; Bergerat, A.; et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011, 5, 220–230. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Nat. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Flint, H.J.; Scott, K.P.; Duncan, S.H.; Louis, P.; Forano, E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Andersson, H.; Tullberg, C.; Ahrné, S.; Hamberg, K.; Lazou Ahrén, I.; Molin, G.; Sonesson, M.; Håkansson, Å. Oral Administration of Lactobacillus plantarum 299v Reduces Cortisol Levels in Human Saliva during Examination Induced Stress: A Randomized, Double-Blind Controlled Trial. Int. J. Microbiol. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Håkansson, Å.; Bränning, C.; Molin, G.; Adawi, D.; Hagslätt, M.L.; Jeppsson, B.; Nyman, M.; Ahrné, S. Blueberry husks and probiotics attenuate colorectal inflammation and oncogenesis, and liver injuries in rats exposed to cycling DSS-treatment. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Ejtahed, H.S.; Angoorani, P.; Soroush, A.R.; Atlasi, R.; Hasani-Ranjbar, S.; Mortazavian, A.M.; Larijani, B. Probiotics supplementation for the obesity management; A systematic review of animal studies and clinical trials. J. Funct. Foods 2019, 52, 228–242. [Google Scholar] [CrossRef]
- Karlsson, C.L.J.; Molin, G.; Fåk, F.; Johansson Hagslätt, M.L.; Jakesevic, M.; Håkansson, Å.; Jeppsson, B.; Weström, B.; Ahrné, S. Effects on weight gain and gut microbiota in rats given bacterial supplements and a high-energy-dense diet from fetal life through to 6 months of age. Br. J. Nutr. 2011, 106, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Filip, M.; Tzaneva, V.; Dumitrascu, D.L. Fecal transplantation: Digestive and extradigestive clinical applications. Clujul Med. 2018, 91, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Geng, J.; Tang, X.; Fan, H.; Xu, J.; Wen, X.; Ma, Z.S.; Shi, P. Spatial heterogeneity and co-occurrence patterns of human mucosal-associated intestinal microbiota. ISME J. 2014, 8, 881–893. [Google Scholar] [CrossRef]
- Aguirre de Carcer, D.; Cuiv, P.O.; Wang, T.; Kang, S.; Worthley, D.; Whitehall, V.; Gordon, I.; McSweeney, C.; Leggett, B.; Morrison, M. Numerical ecology validates a biogeographical distribution and gender-based effect on mucosa-associated bacteria along the human colon. ISME J. 2011, 5, 801–809. [Google Scholar] [CrossRef]
- Tasse, L.; Bercovici, J.; Pizzut-Serin, S.; Robe, P.; Tap, J.; Klopp, C.; Cantarel, B.L.; Coutinho, P.M.; Henrissat, B.; Leclerc, M.; et al. Functional metagenomics to mine the human gut microbiome for dietary fiber catabolic enzymes. Genome Res. 2010, 20, 1605–1612. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Cummings, J.H.; Macfarlane, S.; Gibson, G.R. Influence of retention time on degradation of pancreatic enzymes by human colonic bacteria grown in a 3-stage continuous culture system. J. Appl. Bacteriol. 1989, 67, 521–527. [Google Scholar] [CrossRef]
- Reppas, C.; Karatza, E.; Goumas, C.; Markopoulos, C.; Vertzoni, M. Characterization of Contents of Distal Ileum and Cecum to Which Drugs/Drug Products are Exposed During Bioavailability/Bioequivalence Studies in Healthy Adults. Pharm. Res. 2015, 32, 3338–3349. [Google Scholar] [CrossRef] [PubMed]
- Persson, E.M.; Gustafsson, A.S.; Carlsson, A.S.; Nilsson, R.G.; Knutson, L.; Forsell, P.; Hanisch, G.; Lennernas, H.; Abrahamsson, B. The effects of food on the dissolution of poorly soluble drugs in human and in model small intestinal fluids. Pharm. Res. 2005, 22, 2141–2151. [Google Scholar] [CrossRef] [PubMed]
- Dokoumetzidis, A.; Macheras, P. A century of dissolution research: From Noyes and Whitney to the biopharmaceutics classification system. Int. J. Pharm. 2006, 321, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nelson, E. Solution rate of theophylline salts and effects from oral administration. J. Am. Pharm. Assoc. 1957, 46, 607–614. [Google Scholar] [CrossRef]
- Lindenbaum, J.; Mellow, M.H.; Blackstone, M.O.; Butler, V.P., Jr. Variation in biologic availability of digoxin from four preparations. N. Engl. J. Med. 1971, 285, 1344–1347. [Google Scholar] [CrossRef] [PubMed]
- Tyrer, J.H.; Eadie, M.J.; Sutherland, J.M.; Hooper, W.D. Outbreak of anticonvulsant intoxication in an Australian city. Br. Med. J. 1970, 4, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Kostewicz, E.S.; Abrahamsson, B.; Brewster, M.; Brouwers, J.; Butler, J.; Carlert, S.; Dickinson, P.A.; Dressman, J.; Holm, R.; Klein, S.; et al. In vitro models for the prediction of in vivo performance of oral dosage forms. Eur. J. Pharm. Sci. 2014, 57, 342–366. [Google Scholar] [CrossRef]
- Stamatopoulos, K.; Batchelor, H.K.; Simmons, M.J.H. Dissolution profile of theophylline modified release tablets, using a biorelevant Dynamic Colon Model (DCM). Eur. J. Pharm. Biopharm. 2016, 108, 9–17. [Google Scholar] [CrossRef] [PubMed]
- <1092> The Dissolution Procedure: Development and Validation. In The United States Pharmacopeia (USP); The United States Pharmacopeial Convention North Bethesda: Rockville, MD, USA, 2018.
- González-García, I.; Mangas-Sanjuán, V.; Merino-Sanjuán, M.; Bermejo, M. In vitro–in vivo correlations: General concepts, methodologies and regulatory applications. Drug Dev. Ind. Pharm. 2015, 41, 1935–1947. [Google Scholar] [CrossRef]
- Lu, Y.; Kim, S.; Park, K. In vitro-in vivo correlation: Perspectives on model development. Int. J. Pharm. 2011, 418, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. Prebiotics and Probiotics in Digestive Health. Clin. Gastroenterol. Hepatol. 2019, 17, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S.; Song, E.J.; Nam, Y.D.; Lee, S.Y. Probiotics in human health and disease: From nutribiotics to pharmabiotics. J. Microbiol. 2018, 56, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Ooi, S.L.; Correa, D.; Pak, S.C. Probiotics, prebiotics, and low FODMAP diet for irritable bowel syndrome—What is the current evidence? Complement. Ther. Med. 2019, 43, 73–80. [Google Scholar] [CrossRef]
- Kothari, D.; Patel, S.; Kim, S.K. Probiotic supplements might not be universally-effective and safe: A review. Biomed. Pharmacother. 2019, 111, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Astó, E.; Méndez, I.; Audivert, S.; Farran-Codina, A.; Espadaler, J. The Efficacy of Probiotics, Prebiotic Inulin-Type Fructans, and Synbiotics in Human Ulcerative Colitis: A Systematic Review and Meta-Analysis. Nutrients 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Recommendations on dissolution testing. In European Pharmacopoeia 9.0; EDQM-Council of Europe: Strasbourg, France, 2016.
- Dissolution test for solid dosage forms. In European Pharmacopoeia 9.0; EDQM-Council of Europe: Strasbourg, France, 2016.
- <711> Dissolution. In The United States Pharmacopeia (USP); The United States Pharmacopeial Convention North Bethesda: Rockville, MD, USA, 2018.
- SUPAC. Guidance for Industry Immediate Release Solid Oral Dosage Forms Scale-Up and Postapproval Changes: Chemistry, Manufacturing, and Controls, In Vitro Dissolution Testing, and In Vivo Bioequivalence Documentation. 1995. Available online: https://www.fda.gov/downloads/Drugs/Guidances/UCM456594.pdf (accessed on 2 February 2019).
- Dissolution Testing and Acceptance Criteria for Immediate-Release Solid Oral Dosage Form Drug Products Containing High Solubility Drug Substances Guidance for Industry. 2018. Available online: https://www.fda.gov/downloads/Drugs/Guidances/UCM456594.pdf (accessed on 2 February 2019).
- Guideline on the Investigation of Bioequivalence. 2010. Available online: https://www.ema.europa.eu/documents/scientific-guideline/guideline-investigation-bioequivalence-rev1_en.pdf (accessed on 2 February 2019).
- Reppas, C.; Vertzoni, M. Biorelevant in-vitro performance testing of orally administered dosage forms. J. Pharm. Pharmacol. 2012, 64, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Vertzoni, M.; Dressman, J.; Butler, J.; Hempenstall, J.; Reppas, C. Simulation of fasting gastric conditions and its importance for the in vivo dissolution of lipophilic compounds. Eur. J. Pharm. Biopharm. 2005, 60, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Christophersen, P.C.; Christiansen, M.L.; Holm, R.; Kristensen, J.; Jacobsen, J.; Abrahamsson, B.; Mullertz, A. Fed and fasted state gastro-intestinal in vitro lipolysis: In vitro in vivo relations of a conventional tablet, a SNEDDS and a solidified SNEDDS. Eur. J. Pharm. Sci. 2014, 57, 232–239. [Google Scholar] [CrossRef]
- Jantratid, E.; Janssen, N.; Reppas, C.; Dressman, J.B. Dissolution media simulating conditions in the proximal human gastrointestinal tract: An update. Pharm. Res. 2008, 25, 1663–1676. [Google Scholar] [CrossRef]
- Markopoulos, C.; Andreas, C.J.; Vertzoni, M.; Dressman, J.; Reppas, C. In-vitro simulation of luminal conditions for evaluation of performance of oral drug products: Choosing the appropriate test media. Eur. J. Pharm. Biopharm. 2015, 93, 173–182. [Google Scholar] [CrossRef]
- Granfeldt, Y.; Bjorck, I.; Drews, A.; Tovar, J. An in vitro procedure based on chewing to predict metabolic response to. Eur. J. Clin. Nutr. 1992, 46, 649–660. [Google Scholar]
- Egger, L.; Ménard, O.; Delgado-Andrade, C.; Alvito, P.; Assunção, R.; Balance, S.; Barberá, R.; Brodkorb, A.; Cattenoz, T.; Clemente, A.; et al. The harmonized INFOGEST in vitro digestion method: From knowledge to action. Food Res. Int. 2016, 88, 217–225. [Google Scholar] [CrossRef]
- Molly, K.; De Smet, I.; Nollet, L.; Vande Woestyne, M.; Verstraete, W. Effect of Lactobacilli on the Ecology of the Gastrointestinal Microbiota Cultured in the SHIME Reactor. Microb. Ecol. Health Dis. 1996, 2, 79–89. [Google Scholar] [CrossRef]
- Minekus, M.; Marteau, P.; Havenaar, R. Multicompartmental dynamic computer-controlled model simulating the stomach and small intestine. Altern. Lab. Anim. ATLA 1995. [Google Scholar]
- Macfarlane, G.T.; Macfarlane, S.; Gibson, G.R. Validation of a Three-Stage Compound Continuous Culture System for Investigating the Effect of Retention Time on the Ecology and Metabolism of Bacteria in the Human Colon. Microb. Ecol. 1998, 35, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Marefati, A.; Bertrand, M.; Sjöö, M.; Dejmek, P.; Rayner, M. Storage and digestion stability of encapsulated curcumin in emulsions based on starch granule Pickering stabilization. Food Hydrocolloids 2017, 63, 309–320. [Google Scholar] [CrossRef]
- Tikekar, R.V.; Pan, Y.; Nitin, N. Fate of curcumin encapsulated in silica nanoparticle stabilized Pickering emulsion during storage and simulated digestion. Food Res. Int. 2013, 51, 370–377. [Google Scholar] [CrossRef]
- Molly, K.; Vande Woestyne, M.; Verstraete, W. Development of a 5-step multi-chamber reactor as a simulation of the human intestinal microbial ecosystem. Appl. Microbiol. Biotechnol. 1993, 39, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Verwei, M.; Minekus, M.; Zeijdner, E.; Schilderink, R.; Havenaar, R. Evaluation of two dynamic in vitro models simulating fasted and fed state conditions in the upper gastrointestinal tract (TIM-1 and tiny-TIM) for investigating the bioaccessibility of pharmaceutical compounds from oral dosage forms. Int. J. Pharm. 2016, 498, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Claeys, B.; Vervaeck, A.; Hillewaere, X.K.; Possemiers, S.; Hansen, L.; De Beer, T.; Remon, J.P.; Vervaet, C. Thermoplastic polyurethanes for the manufacturing of highly dosed oral sustained release matrices via hot melt extrusion and injection molding. Eur. J. Pharm. Biopharm. 2015, 90, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Singh, R.P. A human gastric simulator (HGS) to study food digestion in human stomach. J. Food Sci. 2010, 75, E627–E635. [Google Scholar] [CrossRef]
- Tharakan, A.; Norton, I.; Fryer, P.; Bakalis, S. Mass transfer and nutrient absorption in a simulated model of small intestine. J. Food Sci. 2010, 75, E339–E346. [Google Scholar] [CrossRef] [PubMed]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carriere, F.; Boutrou, R.; Corredig, M.; Dupont, D. A standardised static in vitro digestion method suitable for food—An international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Vamanu, E.; Gatea, F.; Sarbu, I. In Vitro Ecological Response of the Human Gut Microbiome to Bioactive Extracts from Edible Wild Mushrooms. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Yang, L. Biorelevant dissolution testing of colon-specific delivery systems activated by colonic microflora. J. Control. Release 2008, 125, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Siew, L.F.; Man, S.M.; Newton, J.M.; Basit, A.W. Amylose formulations for drug delivery to the colon: A comparison of two fermentation models to assess colonic targeting performance in vitro. Int. J. Pharm. 2004, 273, 129–134. [Google Scholar] [CrossRef]
- Yadav, S.; Deka, S.R.; Tiwari, K.; Sharma, A.K.; Kumar, P. Multi-Stimuli Responsive Self-Assembled Nanostructures Useful for Colon Drug Delivery. IEEE Trans. NanoBiosci. 2017, 16, 764–772. [Google Scholar] [CrossRef]
- Rao, J.; Khan, A. Enzyme sensitive synthetic polymer micelles based on the azobenzene motif. J. Am. Chem. Soc. 2013, 135, 14056–14059. [Google Scholar] [CrossRef] [PubMed]
- Gliko-Kabir, I.; Yagen, B.; Baluom, M.; Rubinstein, A. Phosphated crosslinked guar for colon-specific drug delivery: II. In vitro and in vivo evaluation in the rat. J. Control. Release 2000, 63, 129–134. [Google Scholar] [CrossRef]
- Tuǧcu-Demiröz, F.; Acartürk, F.; Takka, S.; Konuş-Boyunaǧa, Ö. In-vitro and in-vivo evaluation of mesalazine-guar gum matrix tablets for colonic drug delivery. J. Drug Target. 2004, 12, 105–112. [Google Scholar] [CrossRef]
- Yamada, K.; Iwao, Y.; Bani-Jaber, A.; Noguchi, S.; Itai, S. Preparation and Evaluation of Newly Developed Chitosan Salt Coating Dispersions for Colon Delivery without Requiring Overcoating. Chem. Pharm. Bull. 2015, 63, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Jyoti, K.; Bhatia, R.K.; Martis, E.A.F.; Coutinho, E.C.; Jain, U.K.; Chandra, R.; Madan, J. Soluble curcumin amalgamated chitosan microspheres augmented drug delivery and cytotoxicity in colon cancer cells: In vitro and in vivo study. Colloids Surf. B 2016, 148, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Andishmand, H.; Tabibiazar, M.; Mohammadifar, M.A.; Hamishehkar, H. Pectin-zinc-chitosan-polyethylene glycol colloidal nano-suspension as a food grade carrier for colon targeted delivery of resveratrol. Int. J. Biol. Macromol. 2017, 97, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Gunter, E.A.; Popeyko, O.V. Calcium pectinate gel beads obtained from callus cultures pectins as promising systems for colon-targeted drug delivery. Carbohydr. Polym. 2016, 147, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Stubbe, B.; Maris, B.; Van den Mooter, G.; De Smedt, S.C.; Demeester, J. The in vitro evaluation of ‘azo containing polysaccharide gels’ for colon delivery. J. Control. Release 2001, 75, 103–114. [Google Scholar] [CrossRef]
- Krishnaiah, Y.S.R.; Bhaskar Reddy, P.R.; Satyanarayana, V.; Karthikeyan, R.S. Studies on the development of oral colon targeted drug delivery systems for metronidazole in the treatment of amoebiasis. Int. J. Pharm. 2002, 236, 43–55. [Google Scholar] [CrossRef]
- Imaoka, A.; Setoyama, H.; Takagi, A.; Matsumoto, S.; Umesaki, Y. Improvement of human faecal flora-associated mouse model for evaluation of the functional foods. J. Appl. Microbiol. 2004, 96, 656–663. [Google Scholar] [CrossRef]
- Savage, D.C.; Dubos, R.; Schaedler, R.W. The gastrointestinal epithelium and its autochthonous bacterial flora. J. Exp. Med. 1968, 127, 67–76. [Google Scholar] [CrossRef]
- Suau, A.; Bonnet, R.; Sutren, M.; Godon, J.J.; Gibson, G.R.; Collins, M.D.; Doré, J. Direct analysis of genes encoding 16S rRNA from complex communities reveals many novel molecular species within the human gut. Appl. Environ. Microbiol. 1999, 65, 4799–4807. [Google Scholar] [PubMed]
- Rama Prasad, Y.V.; Krishnaiah, Y.S.R.; Satyanarayana, V. In vitro evaluation of guar gum as a carrier for colon-specific drug delivery. J. Control. Release 1998, 51, 281–287. [Google Scholar] [CrossRef]
- Kumar Dev, R.; Bali, V.; Pathak, K. Novel microbially triggered colon specific delivery system of 5-Fluorouracil: Statistical optimization, in vitro, in vivo, cytotoxic and stability assessment. Int. J. Pharm. 2011, 411, 142–151. [Google Scholar] [CrossRef]
- Vaidya, A.; Jain, A.; Khare, P.; Agrawal, R.K.; Jain, S.K. Metronidazole Loaded Pectin Microspheres for Colon Targeting. J. Pharm. Sci. 2009, 98, 4229–4236. [Google Scholar] [CrossRef] [PubMed]
- Sirisha, V.N.l.; Eswariah, M.C.; Rao, A.S. A Novel Approach of Locust Bean Gum Microspheres for Colonic Delivery of Mesalamine. Int. J. Appl. Pharm. 2018, 10. [Google Scholar] [CrossRef][Green Version]
- Das, A.; Wadhwa, S.; Srivastava, A.K. Cross-Linked Guar Gum Hydrogel Discs for Colon-Specific. Delivery of Ibuprofen: Formulation and In Vitro Evaluation. Drug Deliv. 2006, 13, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Kalala, W.; Kinget, R.; Van den Mooter, G.; Samyn, C. Colonic drug-targeting: In vitro release of ibuprofen from capsules coated with poly(ether-ester) azopolymers. Int. J. Pharm. 1996, 139, 187–195. [Google Scholar] [CrossRef]
- Van den Mooter, G.; Samyn, C.; Kinget, R. The Relation Between Swelling Properties and Enzymatic Degradation of Azo Polymers Designed for Colon-Specific Drug Delivery. Pharm. Res. 1994, 11, 1737–1741. [Google Scholar] [CrossRef]
- Kopečková, P.; Rathi, R.; Takada, S.; Říhová, B.; Berenson, M.M.; Kopeček, J. Bioadhesive N-(2-hydroxypropyl) methacrylamide copolymers for colon-specific drug delivery. J. Control. Release 1994, 28, 211–222. [Google Scholar] [CrossRef]
- McConnell, E.L.; Tutas, J.; Mohamed, M.A.M.; Banning, D.; Basit, A.W. Colonic drug delivery using amylose films: The role of aqueous ethylcellulose dispersions in controlling drug release. Cellulose 2006, 14, 25–34. [Google Scholar] [CrossRef]
- Siew, L.F.; Basit, A.W.; Newton, J.M. The potential of organic-based amylose-ethylcellulose film coatings as oral colon-specific drug delivery systems. AAPS PharmSciTech 2000, 1, 53–61. [Google Scholar] [CrossRef]
- Macfarlane, S.; Quigley, M.E.; Hopkins, M.J.; Newton, D.F.; Macfarlane, G.T. Polysaccharide degradation by human intestinal bacteria during growth under multi-substrate limiting conditions in a three-stage continuous culture system. FEMS Microbiol. Ecol. 1998, 26, 231–243. [Google Scholar] [CrossRef]
- Karrout, Y.; Neut, C.; Wils, D.; Siepmann, F.; Deremaux, L.; Flament, M.P.; Dubreuil, L.; Desreumaux, P.; Siepmann, J. Novel polymeric film coatings for colon targeting: Drug release from coated pellets. Eur. J. Pharm. Sci. 2009, 37, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, L.; Hovgaard, L.; Mortensen, P.B.; Brøndsted, H. Dextran hydrogels for colon-specific drug delivery. V. Degradation in human intestinal incubation models. Eur. J. Pharm. Sci. 1995, 3, 329–337. [Google Scholar] [CrossRef]
- Wilson, P.J.; Basit, A.W. Exploiting gastrointestinal bacteria to target drugs to the colon: An in vitro study using amylose coated tablets. Int. J. Pharm. 2005, 300, 89–94. [Google Scholar] [CrossRef]
- Venema, K.; van den Abbeele, P. Experimental models of the gut microbiome. Best Pract. Res. Clin. Gastroenterol. 2013, 27, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Fooks, L.J.; Gibson, G.R. Mixed culture fermentation studies on the effects of synbiotics on the human intestinal pathogens Campylobacter jejuni and Escherichia coli. Anaerobe 2003, 9, 231–242. [Google Scholar] [CrossRef]
- O’Donnell, M.M.; Rea, M.C.; Shanahan, F.; Ross, R.P. The Use of a Mini-Bioreactor Fermentation System as a Reproducible, High-Throughput ex vivo Batch Model of the Distal Colon. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Yadav, A.K.; Prudhviraj, G.; Gulati, M.; Kaur, P.; Vaidya, Y. A novel dissolution method for evaluation of polysaccharide based colon specific delivery systems: A suitable alternative to animal sacrifice. Eur. J. Pharm. Sci. 2015, 73, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Sadora, M.; Singh, S.K.; Gulati, M.; Maharshi, P.; Sharma, A.; Kumar, B.; Rathee, H.; Ghai, D.; Malik, A.H.; et al. Novel biorelevant dissolution medium as a prognostic tool for polysaccharide-based colon-targeted drug delivery system. J. Adv. Pharm. Technol. Res. 2017, 8, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Kotla, N.G.; Singh, S.; Maddiboyina, B.; Sunnapu, O.; Webster, T.J. A novel dissolution media for testing drug release from a nanostructured polysaccharide-based colon specific drug delivery system: An approach to alternative colon media. Int. J. Nanomed. 2016, 11, 1089–1095. [Google Scholar] [CrossRef]
- Tap, J.; Mondot, S.; Levenez, F.; Pelletier, E.; Caron, C.; Furet, J.P.; Ugarte, E.; Muñoz-Tamayo, R.; Paslier, D.L.E.; Nalin, R.; et al. Towards the human intestinal microbiota phylogenetic core. Environ. Microbiol. 2009, 11, 2574–2584. [Google Scholar] [CrossRef]
- Yang, X.; Xie, L.; Li, Y.; Wei, C. More than 9,000,000 unique genes in human gut bacterial community: Estimating gene numbers inside a human body. PLoS ONE 2009, 4, e6074. [Google Scholar] [CrossRef] [PubMed]
- Kovatcheva-Datchary, P.; Egert, M.; Maathuis, A.; Rajilić-Stojanović, M.; De Graaf, A.A.; Smidt, H.; De Vos, W.M.; Venema, K. Linking phylogenetic identities of bacteria to starch fermentation in an in vitro model of the large intestine by RNA-based stable isotope probing. Environ. Microbiol. 2009, 11, 914–926. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.F.; Cao, W.W.; Cerniglia, C.E. PCR detection and quantitation of predominant anaerobic bacteria in human and animal fecal samples. Appl. Environ. microbiol. 1996, 62, 1242–1247. [Google Scholar] [PubMed]
- Moore, W.E.C.; Holdeman, L.V. Human fecal flora: The normal flora of 20 Japanese Hawaiians. J. Appl. Microbiol. 1974, 27, 961–979. [Google Scholar]
- Wang, M.; Ahrné, S.; Jeppsson, B.; Molin, G. Comparison of bacterial diversity along the human intestinal tract by direct cloning and sequencing of 16S rRNA genes. FEMS Microbiol. Ecol. 2005, 54, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Hold, G.L.; Pryde, S.E.; Russell, V.J.; Furrie, E.; Flint, H.J. Assessment of microbial diversity in human colonic samples by 16S rDNA sequence analysis. FEMS Microbiol. Ecol. 2002, 39, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Hold, G.L.; Schwiertz, A.; Aminov, R.I.; Blaut, M.; Flint, H.J. Oligonucleotide probes that detect quantitatively significant groups of butyrate-producing bacteria in human feces. Appl. Environ. Microbiol. 2003, 69, 4320–4324. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol. Lett. 2009, 294, 1–8. [Google Scholar] [CrossRef]
- Lopez-Siles, M.; Khan, T.M.; Duncan, S.H.; Harmsen, H.J.M.; Garcia-Gil, L.J.; Flint, H.J. Cultured representatives of two major phylogroups of human colonic Faecalibacterium prausnitzii can utilize pectin, uronic acids, and host-derived substrates for growth. Appl. Environ. Microbiol. 2012, 78, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Salyers, A.A.; Vercellotti, J.R.; West, S.E.H.; Wilkins, T.D. Fermentation of mucin and plant polysaccharides by strains of Bacteroides from the human colon. Appl. Environ. Microbiol. 1977, 33, 319–322. [Google Scholar]
- Davis, L.M.G.; Martínez, I.; Walter, J.; Goin, C.; Hutkins, R.W. Barcoded pyrosequencing reveals that consumption of galactooligosaccharides results in a highly specific bifidogenic response in humans. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Kleessen, B.; Schwarz, S.; Boehm, A.; Fuhrmann, H.; Richter, A.; Henle, T.; Krueger, M. Jerusalem artichoke and chicory inulin in bakery products affect faecal microbiota of healthy volunteers. Br. J. Nutr. 2007, 98, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Ze, X.; Duncan, S.H.; Louis, P.; Flint, H.J. Ruminococcus bromii is a keystone species for the degradation of resistant starch in the human colon. ISME J. 2012, 6, 1535–1543. [Google Scholar] [CrossRef] [PubMed]
- Abell, G.C.J.; Cooke, C.M.; Bennett, C.N.; Conlon, M.A.; McOrist, A.L. Phylotypes related to Ruminococcus bromii are abundant in the large bowel of humans and increase in response to a diet high in resistant starch. FEMS Microbiol. Ecol. 2008, 66, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Belenguer, A.; Holtrop, G.; Johnstone, A.M.; Flint, H.J.; Lobley, G.E. Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl. Environ. Microbiol. 2007, 73, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Scott, K.P.; Martin, J.C.; Duncan, S.H.; Flint, H.J. Prebiotic stimulation of human colonic butyrate-producing bacteria and bifidobacteria, in vitro. FEMS Microbiol. Ecol. 2014, 87, 30–40. [Google Scholar] [CrossRef]
- Benítez-Páez, A.; Gómez del Pulgar, E.M.; Sanz, Y. The glycolytic versatility of Bacteroides uniformis CECT 7771 and Its genome response to oligo and polysaccharides. Front. Cell. Infect. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Scher, J.U.; Sczesnak, A.; Longman, R.S.; Segata, N.; Ubeda, C.; Bielski, C.; Rostron, T.; Cerundolo, V.; Pamer, E.G.; Abramson, S.B.; et al. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. eLife 2013, 2013. [Google Scholar] [CrossRef]
- Kovatcheva-Datchary, P.; Nilsson, A.; Akrami, R.; Lee, Y.S.; De Vadder, F.; Arora, T.; Hallen, A.; Martens, E.; Björck, I.; Bäckhed, F. Dietary Fiber-Induced Improvement in Glucose Metabolism Is Associated with Increased Abundance of Prevotella. Cell Metab. 2015, 22, 971–982. [Google Scholar] [CrossRef]
| Bile Salt | Small Intestines | Colon |
|---|---|---|
| Cholic acid CA | 35% | 2% |
| Chenodeoxycholic acid CDCA | 35% | 2% |
| Deoxycholic acid (DCA) | 25% | 34% |
| Lithocholic acid LCA | 1% | 29% |
| Ursodeoxycholic acid UDCA | 2% | 2% |
| 12-oxy-LCA | 3% | |
| Others | 2% | 28% |
| GI Site Represented | Suggested Prandial State | Media | Complexity | References |
|---|---|---|---|---|
| Stomach | Fasted | pH 1.0–1.5 | Low | [87,89,90,91] |
| pH < 4.0 + Surfactant(s) | Medium | [79,87] | ||
| pH < 4.0 + Enzymes (pepsin) | High | [87,89] | ||
| Fed | Stomach pH + Physiological Surfactant(s) | Medium | [79] | |
| No specific site | Fasted | pH 4.5 | Low | [79,87,90,92] |
| Fed | pH > 4.0 and < 6.8 + Enzymes (Papain) | High | [89] | |
| Small intestines | Fasted | pH 5.5, 5.8, 6.5, 6.8 *, 7.2, 7.5 | Low | [87,89,90] |
| Intestinal pH + Physiological Surfactant(s) | Medium | [79] | ||
| Fed | Intestinal pH + Physiological Surfactant(s) | Medium | [79] | |
| pH ≥ 6.8 + Enzymes (Pancreatic powder) | High | [87,89] |
| Type of Formulation Components | Enzyme | Refences |
|---|---|---|
| Azo-structures, polymers and conjugates | Azoreductase | [114,115] |
| Guar gum | Galactomannanase, α-galactosidase | [116,117] |
| Chitosane | β-glucosidase | [118,119] |
| Pectin | Pectinase | [120,121] |
| Starch | Amylase | [113] |
| Dextrane | Dextranase | [122] |
| Inuline | Inulase | [122] |
| Singh, et.al., 2015 [140] | Kotla et al., 2016 [142] | |
|---|---|---|
| Composition | Bacterial Count/Amount (CFU) | Composition |
| Lactobacillus acidophilus | 0.75 × 1012 | Lactobacillus acidophilus |
| Lactobacillus rhamnosus | 0.75 × 1012 | Lactobacillus rhamnosus |
| Bifidobacterium longum | 0.75 × 1012 | Bifidobacterium longum |
| Bifidobacterium bifidum | 0.50 × 1012 | Bifidobacterium infantis |
| Saccharomyces boulardii | 0.10 × 1012 | Lactobacillus plantarum |
| Lactobacillus casei | ||
| Bifidobacterium breve | ||
| Streptococcus thermophilus | ||
| Saccharomyces boulardii | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wahlgren, M.; Axenstrand, M.; Håkansson, Å.; Marefati, A.; Lomstein Pedersen, B. In Vitro Methods to Study Colon Release: State of the Art and An Outlook on New Strategies for Better In-Vitro Biorelevant Release Media. Pharmaceutics 2019, 11, 95. https://doi.org/10.3390/pharmaceutics11020095
Wahlgren M, Axenstrand M, Håkansson Å, Marefati A, Lomstein Pedersen B. In Vitro Methods to Study Colon Release: State of the Art and An Outlook on New Strategies for Better In-Vitro Biorelevant Release Media. Pharmaceutics. 2019; 11(2):95. https://doi.org/10.3390/pharmaceutics11020095
Chicago/Turabian StyleWahlgren, Marie, Magdalena Axenstrand, Åsa Håkansson, Ali Marefati, and Betty Lomstein Pedersen. 2019. "In Vitro Methods to Study Colon Release: State of the Art and An Outlook on New Strategies for Better In-Vitro Biorelevant Release Media" Pharmaceutics 11, no. 2: 95. https://doi.org/10.3390/pharmaceutics11020095
APA StyleWahlgren, M., Axenstrand, M., Håkansson, Å., Marefati, A., & Lomstein Pedersen, B. (2019). In Vitro Methods to Study Colon Release: State of the Art and An Outlook on New Strategies for Better In-Vitro Biorelevant Release Media. Pharmaceutics, 11(2), 95. https://doi.org/10.3390/pharmaceutics11020095