Effect of Lactose Pseudopolymorphic Transition on the Aerosolization Performance of Drug/Carrier Mixtures
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Production of Micronized Lactose
2.2.2. Preparation of Hygroscopic Anhydrous α-Lactose (LαH)
2.2.3. Preparation of Stable Anhydrous α-lactose (LαS)
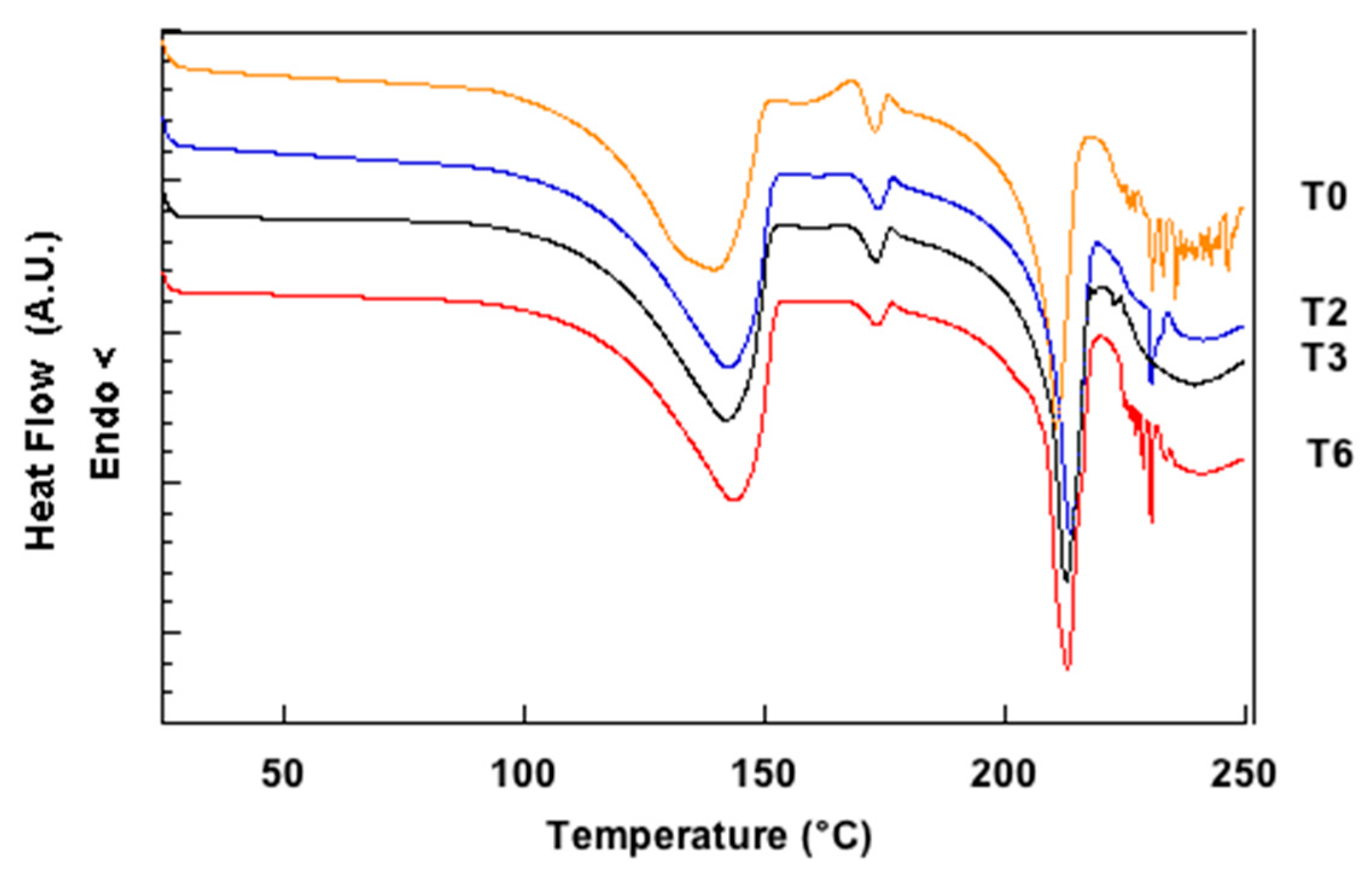
2.2.4. Differential Scanning Calorimetry (DSC)
2.2.5. Particle size Determination
2.2.6. Scanning Electron Microscopy (SEM)
2.2.7. Preparation of Drug/Carrier Mixtures
2.2.8. Salbutamol Sulphate Quantification
2.2.9. Budesonide Quantification
2.2.10. Homogeneity Test
2.2.11. In Vitro Deposition Tests
2.2.12. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Lactose Samples
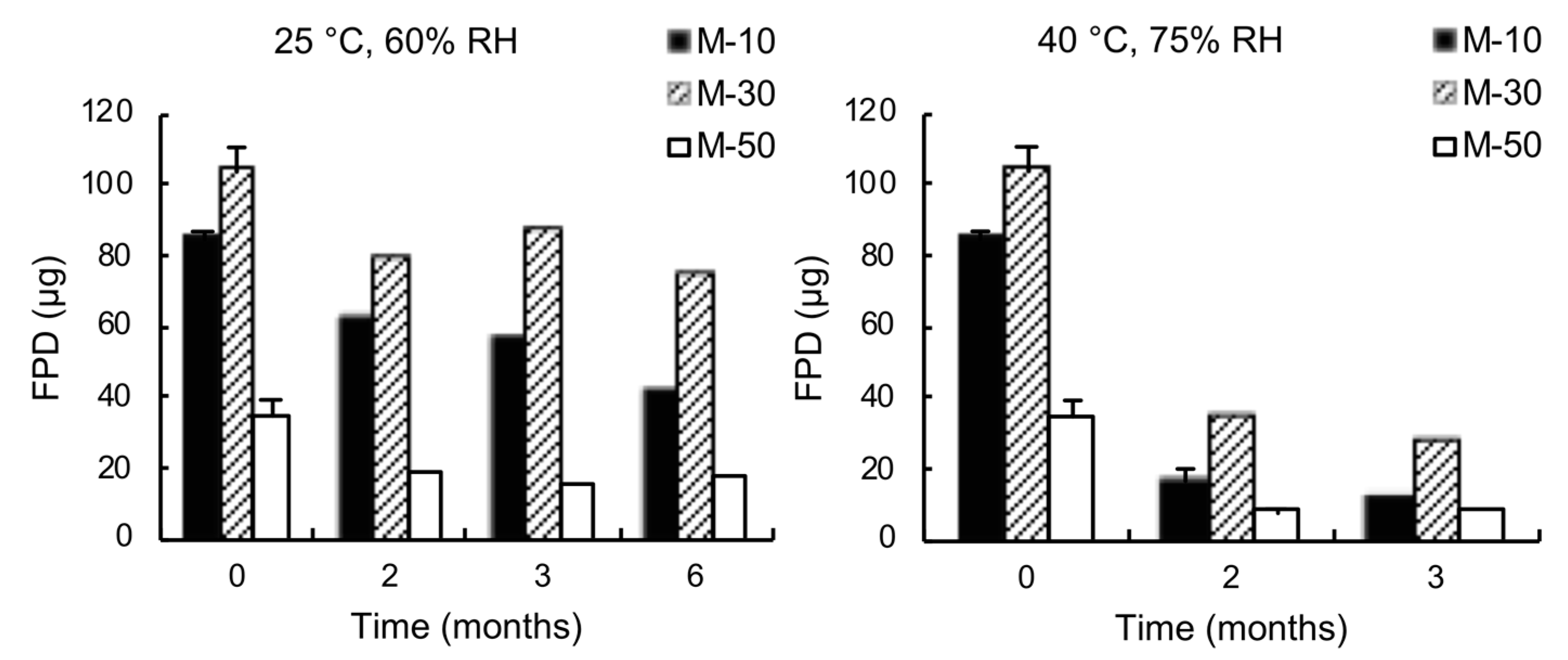
3.2. Effect of Lactose Solid-State on Drug Respirability
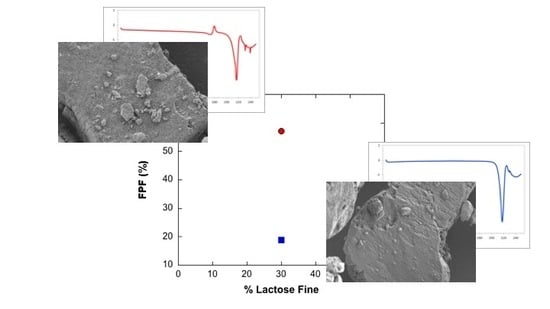
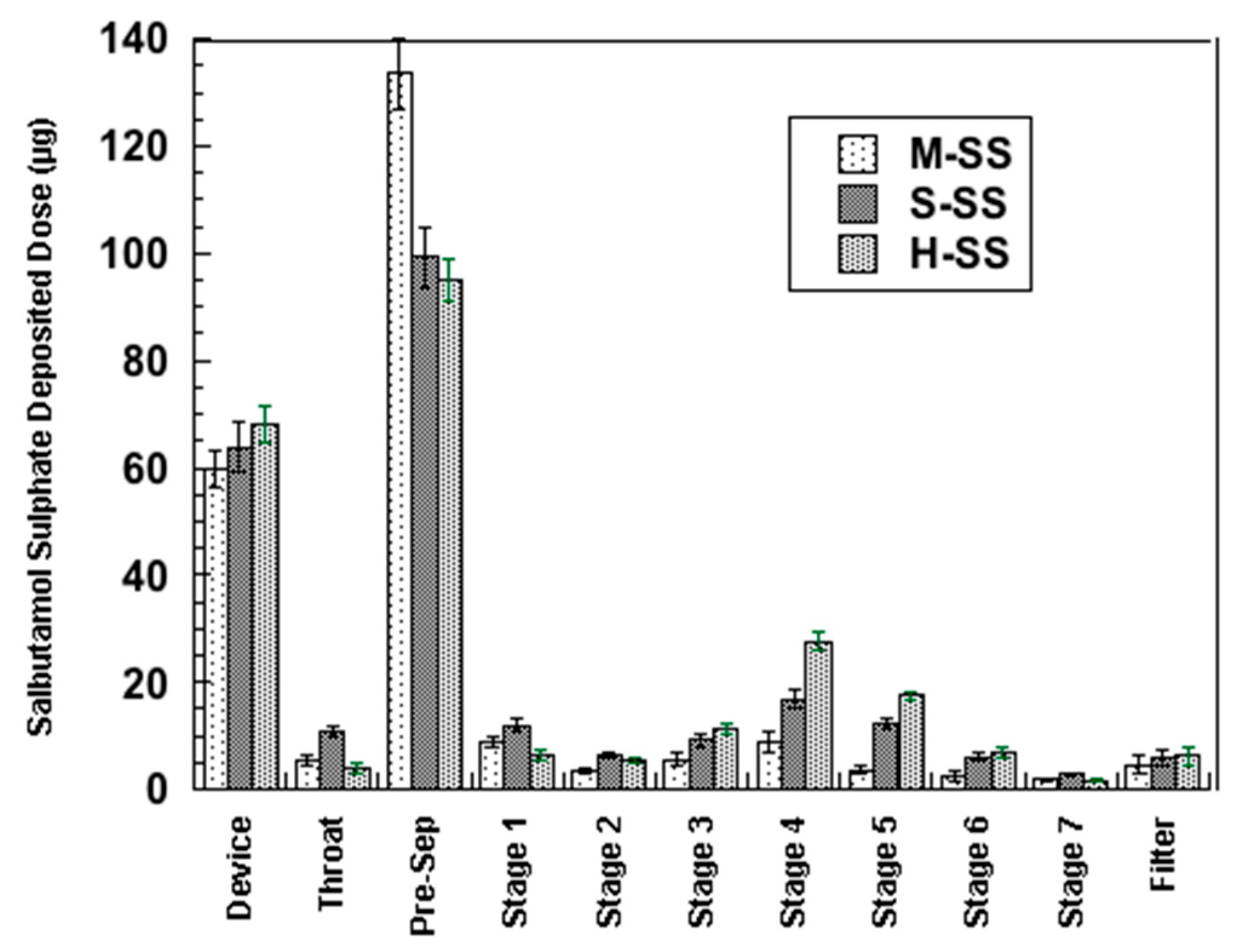
3.2.1. Ternary Mixtures
3.2.2. Binary Mixtures
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Frijlink, H.W.; De Boer, A.H. Dry powder inhalers for pulmonary drug delivery. Expert Opin. Drug Deliv. 2004, 1, 67–86. [Google Scholar] [CrossRef] [PubMed]
- Hoppentocht, M.; Hagedoorn, P.; Frijlink, H.; De Boer, A. Technological and practical challenges of dry powder inhalers and formulations. Adv. Drug Deliv. Rev. 2014, 75, 18–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pilcer, G.; Wauthoz, N.; Amighi, K. Lactose characteristics and the generation of the aerosol. Adv. Drug Deliv. Rev. 2012, 64, 233–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Boer, A.; Chan, H.K.; Price, R. A critical view on lactose-based drug formulation and device studies for dry powder inhalation: Which are relevant and what interactions to expect? Adv. Drug Deliv. Rev. 2012, 64, 257–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Della Bella, A.; Salomi, E.; Buttini, F.; Bettini, R. The role of the solid state and physical properties of the carrier in adhesive mixtures for lung delivery. Expert Opin. Drug Deliv. 2018, 15, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Kaialy, W.; Alhalaweh, A.; Velaga, S.P.; Nokhodchi, A. Influence of lactose carrier particle size on the aerosol performance of budesonide from a dry powder inhaler. Powder Technol. 2012, 227, 74–85. [Google Scholar] [CrossRef]
- Zeng, X.M.; Martin, G.P.; Marriot, C.; Pritchard, J. The Effect of carrier isize and morphology on the dispersion of salbutamol sulphate after aerosolization at different flow rates. J. Pharm. Pharmacol. 2000, 52, 1211–1221. [Google Scholar] [CrossRef]
- Donovan, M.J.; Smyth, H.D. Influence of size and surface roughness of large lactose carrier particles in dry powder inhaler formulations. Int. J. Pharm. 2010, 402, 1–9. [Google Scholar] [CrossRef]
- Zeng, X.M.; Martin, G.P.; Marriott, C.; Pritchard, J. The influence of carrier morphology on drug delivery by dry powder inhalers. Int. J. Pharm. 2000, 200, 93–106. [Google Scholar] [CrossRef]
- Shalash, A.O.; Molokhia, A.M.; Elsayed, M.M. Insights into the roles of carrier microstructure in adhesive/carrier-based dry powder inhalation mixtures: Carrier porosity and fine particle content. Eur. J. Pharm. Biopharm. 2015, 96, 291–303. [Google Scholar] [CrossRef]
- Heng, P.W.S.; Chan, L.W.; Lim, L.T. Quantification of the Surface Morphologies of Lactose Carriers and Their Effect on the in vitro Deposition of Salbutamol Sulphate. Chem. Pharm. Bull. 2000, 48, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Young, P.M.; Cocconi, D.; Colombo, P.; Bettini, R.; Price, R.; Steele, D.F.; Tobyn, M.J. Characterization of a surface modified dry powder inhalation carrier prepared by “particle smoothing”. J. Pharm. Pharmacol. 2002, 54, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, F.; Cocconi, D.; Bettini, R.; Giordano, F.; Santi, P.; Tobyn, M.; Price, R.; Young, P.; Caramella, C.; Colombo, P. The surface roughness of lactose particles can be modulated by wet-smoothing using a high-shear mixer. AAPS PharmSciTech 2004, 5, 69–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, B.M.J.; Chan, L.W.; Heng, P.W.S. Improving Dry Powder Inhaler Performance by Surface Roughening of Lactose Carrier Particles. Pharm. Res. 2016, 33, 1923–1935. [Google Scholar] [CrossRef]
- Larhrib, H.; Zeng, X.M.; Martin, G.P.; Marriott, C.; Pritchard, J. The use of different grades of lactose as a carrier for aerosolised salbutamol sulphate. Int. J. Pharm. 1999, 191, 1–14. [Google Scholar] [CrossRef]
- Louey, M. Influence of physico-chemical carrier properties on the in vitro aerosol deposition from interactive mixtures. Int. J. Pharm. 2003, 252, 87–98. [Google Scholar] [CrossRef]
- Traini, D.; Young, P.M.; Thielmann, F.; Acharya, M. The Influence of Lactose Pseudopolymorphic Form on Salbutamol Sulfate–Lactose Interactions in DPI Formulations. Drug Dev. Ind. Pharm. 2008, 34, 992–1001. [Google Scholar] [CrossRef]
- Zeng, X.M.; Martin, G.P.; Tee, S.K.; Marriot, C. The role of fine particle lactose on the dispersion and deaggregation of salbutamol sulphate in an air stream in vitro. Int. J. Pharm. 1998, 176, 99–110. [Google Scholar] [CrossRef]
- Zeng, X.M.; Martin, G.P.; Tee, S.K.; Abu Ghoush, A.; Marriott, C. Effects of particle size and adding sequence of fine lactose on the deposition of salbutamol sulphate from a dry powder formulation. Int. J. Pharm. 1999, 182, 133–144. [Google Scholar] [CrossRef]
- Jones, M.D.; Price, R. The Influence of Fine Excipient Particles on the Performance of Carrier-Based Dry Powder Inhalation Formulations. Pharm. Res. 2006, 23, 1665–1674. [Google Scholar] [CrossRef]
- Thalberg, K.; Berg, E.; Fransson, M. Modeling dispersion of dry powders for inhalation. The concepts of total fines, cohesive energy and interaction parameters. Int. J. Pharm. 2012, 427, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Martin, G.P.; Marriott, C.; Pritchard, J. Lactose as a carrier in dry powder formulations: The influence of surface characteristics on drug delivery. J. Pharm. Sci. 2001, 90, 1424–1434. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.; Chan, L.W.; Steckel, H.; Heng, P.W. Physico-chemical aspects of lactose for inhalation. Adv. Drug Deliv. Rev. 2012, 64, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Grasmeijer, F.; Lexmond, A.J.; Noort, M.V.D.; Hagedoorn, P.; Hickey, A.J.; Frijlink, H.W.; De Boer, A.H. New Mechanisms to Explain the Effects of Added Lactose Fines on the Dispersion Performance of Adhesive Mixtures for Inhalation. PLoS ONE 2014, 9, e87825. [Google Scholar] [CrossRef]
- Jawad, R.; Elleman, C.; Vermeer, L.; Drake, A.F.; Woodhead, B.; Martin, G.P.; Royall, P.G. The measurement of the β/α anomer composition within amorphous lactose prepared by spray and freeze drying using a simple 1H-NMR method. Pharm. Res. 2012, 29, 511–524. [Google Scholar] [CrossRef]
- Steckel, H.; Markefka, P.; Tewierik, H.; Kammelar, R. Effect of milling and sieving on functionality of dry powder inhalation products. Int. J. Pharm. 2006, 309, 51–59. [Google Scholar] [CrossRef]
- Young, P.M.; Chan, H.K.; Chiou, H.; Edge, S.; Tee, T.H.; Traini, D. The Influence of Mechanical Processing of Dry Powder Inhaler Carriers on Drug Aerosolization Performance. J. Pharm. Sci. 2007, 96, 1331–1341. [Google Scholar] [CrossRef]
- Pazesh, S.; Lazorova, L.; Berggren, J.; Alderborn, G.; Gråsjö, J. Considerations on the quantitative analysis of apparent amorphicity of milled lactose by Raman spectroscopy. Int. J. Pharm. 2016, 511, 488–504. [Google Scholar] [CrossRef]
- Della Bella, A.; Müller, M.; Soldati, L.; Elviri, L.; Bettini, R. Quantitative determination of micronization-induced changes in the solid state of lactose. Int. J. Pharm. 2016, 505, 383–393. [Google Scholar] [CrossRef]
- Pitchayajittipong, C.; Price, R.; Shur, J.; Kaerger, J.S.; Edge, S. Characterisation and functionality of inhalation anhydrous lactose. Int. J. Pharm. 2010, 390, 134–141. [Google Scholar] [CrossRef]
- Adi, H.; Larson, I.; Chiou, H.; Young, P.; Traini, D.; Stewart, P. Agglomerate Strength and Dispersion of Salmeterol Xinafoate from Powder Mixtures for Inhalation. Pharm. Res. 2006, 23, 2556–2565. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, G.A.; Raidal, S.; Robson, K.; Narkowicz, C.K.; Nichols, D.S.; Walters, E.H. Bronchopulmonary pharmacokinetics of (R)-salbutamol and (S)-salbutamol enantiomers in pulmonary epithelial lining fluid and lung tissue of horses. Br. J. Clin. Pharmacol. 2017, 83, 1436–1445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benet, L.Z.; Broccatelli, F.; Oprea, T.I. BDDCS Applied to Over 900 Drugs. AAPS J. 2011, 13, 519–547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guenette, E.; Barrett, A.; Kraus, D.; Brody, R.; Harding, L.; Magee, G. Understanding the effect of lactose particle size on the properties of DPI formulations using experimental design. Int. J. Pharm. 2009, 380, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Rudén, J.; Frenning, G.; Bramer, T.; Thalberg, K.; Alderborn, G. Relationships between surface coverage ratio and powder mechanics of binary adhesive mixtures for dry powder inhalers. Int. J. Pharm. 2018, 541, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Benassi, A.; Perazzi, I.; Bosi, R.; Cottini, C.; Bettini, R. Quantifying the loading capacity of a carrier-based DPI formulation and its dependence on the blending process. Powder Technol. 2019, 356, 607–617. [Google Scholar] [CrossRef]
- Carstensen, J.T. (Ed.) Advanced Pharmaceutical Solids; Marcel Dekker, Inc.: New York, NY, USA, 2001; Volume 110, p. 16. [Google Scholar]
- Cline, D.; Dalby, R.; Chow, A.H.L.; Tong, H.H.Y.; Shekunov, B.Y.; York, P. Use of inverse gas chromatography (IGC) to determine the surface energy and surface area of powdered materials. Pharm. Res. 2004, 21, 1718–1720. [Google Scholar] [CrossRef]
- Cline, D.; Dalby, R. Predicting the quality of powders for inhalation from surface energy and area. Pharm. Res. 2002, 19, 1274–1277. [Google Scholar] [CrossRef]

| Sample | dV10 (µm) | dV50 (µm) | dV90 (µm) |
|---|---|---|---|
| Lα·H2O | 10.2 ± 0.8 | 53.1 ± 2.0 | 103.3 ± 2.6 |
| LαH | 18.8 ± 1.1 | 54.7 ± 0.7 | 104.9 ± 0.6 |
| LαS | 20.7 ± 2.2 | 56.3 ± 0.4 | 105.7 ± 1.4 |
| Mixture | Lα·H2O (%) | Micronized Lactose (%) | SS (%) |
|---|---|---|---|
| M-SS | 99 | - | 1 |
| M-10 | 89 | 10 | 1 |
| M-30 | 69 | 30 | 1 |
| M-50 | 49 | 50 | 1 |
| Mixture | ED (µg) | FPD (µg) | FPF (%) |
|---|---|---|---|
| M-SS | 177.1 ± 3.4 | 26.8 ± 1.9 | 15.1 ± 0.8 |
| M-10 | 178.7 ± 4.6 | 84.3 ± 2.6 | 47.2 ± 0.7 |
| M-30 | 180.9 ± 9.7 | 103.4 ± 7.3 | 57.2 ± 2.5 |
| M-50 | 102.8 ± 5.5 | 34.9 ± 4.6 | 33.9 ± 2.8 |
| Mixture | ED (µg) | FPD (µg) | FPF (%) |
|---|---|---|---|
| H-SS | 182.7 ± 11.6 | 76.2 ± 3.6 | 41.7 ± 1.9 |
| S-SS | 173.0 ± 2.7 | 54.4 ± 2.2 | 31.5 ± 0.9 |
| Mixture | ED (µg) | FPD (µg) | FPF (%) |
|---|---|---|---|
| M-BUD | 154.1 ± 4.3 | 27.9 ± 0.1 | 18.1 ± 0.5 |
| S-BUD | 149.6 ± 4.0 | 17.9 ± 1.3 | 12.0 ± 0.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Della Bella, A.; Müller, M.; Danani, A.; Soldati, L.; Bettini, R. Effect of Lactose Pseudopolymorphic Transition on the Aerosolization Performance of Drug/Carrier Mixtures. Pharmaceutics 2019, 11, 576. https://doi.org/10.3390/pharmaceutics11110576
Della Bella A, Müller M, Danani A, Soldati L, Bettini R. Effect of Lactose Pseudopolymorphic Transition on the Aerosolization Performance of Drug/Carrier Mixtures. Pharmaceutics. 2019; 11(11):576. https://doi.org/10.3390/pharmaceutics11110576
Chicago/Turabian StyleDella Bella, Andrea, Michele Müller, Andrea Danani, Luciano Soldati, and Ruggero Bettini. 2019. "Effect of Lactose Pseudopolymorphic Transition on the Aerosolization Performance of Drug/Carrier Mixtures" Pharmaceutics 11, no. 11: 576. https://doi.org/10.3390/pharmaceutics11110576