Variability and Global Distribution of Subgenotypes of Bovine Viral Diarrhea Virus
Abstract
:1. Introduction
2. Genomic Organization
3. Mechanisms of Genetic Changes in BVDV Genomes
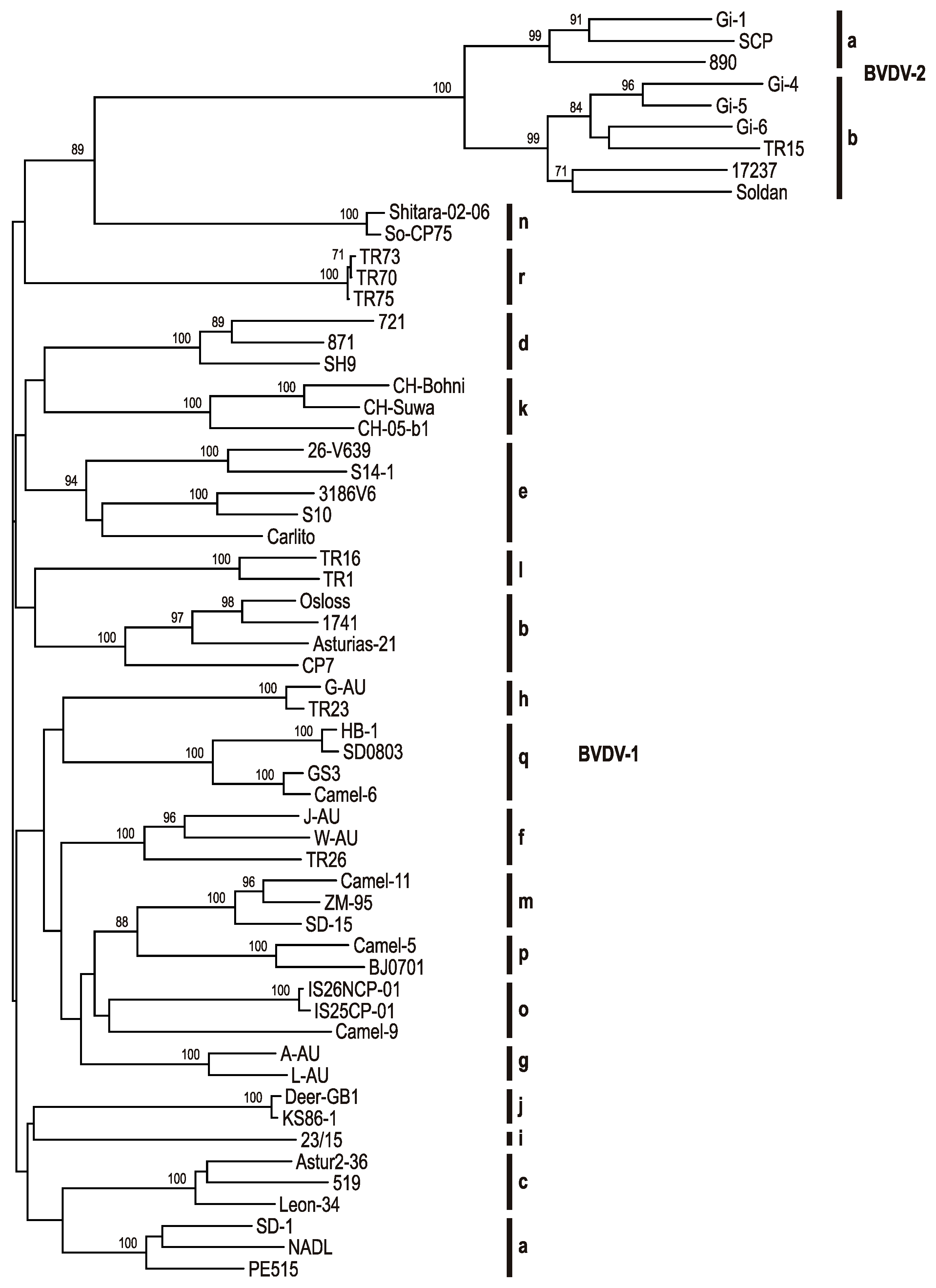
4. BVDV Variability
5. Global Distribution of BVDV Subgenotypes
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Houe, H. Epidemiological features and economical importance of bovine virus diarrhea virus BVDV infections. Vet. Microbiol. 1999, 64, 89–107. [Google Scholar] [CrossRef]
- Simmonds, P.; Becher, P.; Bukh, J.; Gould, E.A.; Meyers, G.; Monath, T.; Muerhoff, S.; Pletnev, A.; Rico-Hesse, R.; Smith, D.B.; et al. ICTV Report Consortium 2017. ICTV Virus Taxonomy Profile, Flaviviridae. J. Gen Virol. 2017, 98, 2–3. [Google Scholar] [PubMed]
- Becher, P.; Avalos-Ramirez, R.; Orlich, M.; Cedillo-Rosales, S.; König, M.; Schweizer, M.; Stalder, H.; Schirrmeier, H.; Thiel, H.J. Genetic and antigenic characterization of novel pestivirus genotypes, implications for classification. Virology 2003, 311, 96–104. [Google Scholar] [CrossRef]
- Becher, P.; Fischer, N.; Grundhoff, A.; Stalder, H.; Schweizer, M.; Postel, A. Complete genome sequence of bovine pestivirus strain PG-2, a second member of the tentative pestivirus species Giraffe. Genome Announc. 2014, 3, e00376-14. [Google Scholar] [CrossRef] [PubMed]
- Vilcek, S.; Ridpath, J.F.; Van Campen, H.; Cavender, J.L.; Warge, J. Characterization of a novel pestivirus originating from a pronghorn antelope. Virus Res. 2005, 108, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, P.D.; Read, A.J.; Frost, M.J.; Finlaison, D.S. Bungowannah virus a probable new species of pestivirus what have we found in the last 10 years? Anim. Health Res. Rev. 2015, 16, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Cortez, A.; Heinemann, M.B.; De Castro Am, M.G.; Soares, R.M.; Pinto, A.M.V.; Alfieri, A.A.; Flores, E.F.; Leite, R.C.; Richtzenhain, L.J. Genetic characterization of Brazilian bovine viral diarrhea virus isolates by partial nucleotide sequencing of the 5′UTR region. Pesq. Vet. Brasil 2006, 26, 211–216. [Google Scholar] [CrossRef]
- Schirrmeier, H.; Strebelow, G.; Depner, K.; Hoffmann, B.; Beer, M. Genetic and antigenic characterization of an atypical pestivirus isolate. A putative member of a novel pestivirus species. J. Gen. Virol. 2004, 8512, 3647–3652. [Google Scholar] [CrossRef] [PubMed]
- Stalder, H.P.; Meier, P.H.; Pfaffen, G.; Wageck-Canal, C.; Rüfenacht, J.; Schaller, P.; Bachofen, C.; Marti, S.; Vogt, H.R.; Peterhans, E. Genetic heterogeneity of pestiviruses of ruminants in Switzerland. Prev. Vet. Med. 2005, 72, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Postel, A.; Schmeiser, S.; Oguzoglu, T.C.; Indenbirken, D.; Alawi, M.; Fischer, N.; Grundhoff, A.; Becher, P. Close relationship of ruminant pestiviruses and classical swine fever virus. Emerg. Infect. Dis. 2015, 21, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Hause, B.M.; Collin, E.A.; Peddireddi, L.; Yuan, F.; Chen, Z.; Hesse, R.A.; Gauger, P.C.; Clement, T.; Fang, Y.; Anderson, G. Discovery of a novel putative atypical porcine pestivirus in pigs in the USA. J. Gen. Virol. 2015, 9610, 2994–2998. [Google Scholar] [CrossRef] [PubMed]
- Postel, A.; Hansmann, F.; Bächlein, C.; Fischer, N.; Alawi, M.; Grundhoff, A.; Derking, S.; Tenhündfeld, J.; Pfankuche, V.M.; Herder, V.; et al. Presence of atypical porcine pestivirus APPV genomes in newborn piglets correlates with congenital tremor. Sci. Rep. 2016, 6, 27735. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Ren, X.; Yang, L.; Hu, Y.; Yang, J.; He, G.; Zhang, J.; Dong, J.; Sun, L.; Du, J.; et al. Virome analysis for identification of novel mammalian viruses in bat species from Chinese provinces. J. Virol. 2012, 86, 10999–11012. [Google Scholar] [CrossRef] [PubMed]
- Firth, C.; Firth, C.; Bhat, M.; Firth, M.A.; Williams, S.H.; Frye, M.J.; Simmonds, P.; Conte, J.M.; Ng, J.; Garcia, J.; et al. Detection of zoonotic pathogens and characterization of novel viruses carried by commensal Rattus norvegicus in New York City. MBio 2014, 5, e01933-14. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, I.M.; Fuentes, R.; Celedón, M.O. Genotypic characterization of Chilean llama (Lama glama) and alpaca (Vicugna pacos) pestivirus isolates. Vet. Microbiol. 2014, 168, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Becher, P.; Orlich, M.; Kosmidou, A.; König, M.; Baroth, M.; Thiel, H.J. Genetic diversity of pestiviruses, Identification of novel groups and implications for classification. Virology 1999, 262, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Becher, P.; Orlich, M.; Shannon, AD.; Horner, G.; König, M.; Thiel, H.J. Phylogenetic analysis of pestiviruses from domestic and wild ruminants. J. Gen. Virol. 1997, 78, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Bauermann, F.V.; Ridpath, J.F.; Weiblen, R.; Flores, E.F. HoBi-like viruses an emerging group of pestiviruses. J. Vet. Diagn. Investig. 2013, 251, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Peletto, S.; Zuccon, F.; Pitti, M.; Gobbi, E.; Marco, L.D.; Caramelli, M.; Masoero, L.; Acutis, P.L. Detection and phylogenetic analysis of an atypical pestivirus, strain IZSPLV_To. Res. Vet. Sci. 2012, 921, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Ridpath, J.F. Practical significance of heterogeneity among BVDV strains, impact of biotype and genotype on U.S. control programs. Prev. Vet. Med. 2005, 721–722, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Vilcek, S.; Paton, D.J.; Durkovic, B.; Strojny, L.; Ibata, G.; Moussa, A.; Loitsch, A.; Rossmanith, W.; Vega, S.; Scicluna, M.T.; et al. Bovine viral diarrhoea virus genotype 1 can be separated into at least eleven genetic groups. Arc. Virol. 2001, 146, 99–115. [Google Scholar]
- Collett, M.S.; Larson, R.; Belzer, S.K.; Retzel, E. Proteins encoded by bovine viral diarrhea virus, the genomic organization of a pestivirus. Virology 1988, 1651, 200–208. [Google Scholar] [CrossRef]
- Becher, P.; Tautz, N. RNA recombination in pestiviruses, cellular RNA sequences in viral genomes highlight the role of host factors for viral persistence and lethal disease. RNA Biol. 2011, 8, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Tautz, N.; Tews, B.A.; Meyers, G. The molecular biology of pestiviruses. Adv. Virus Res. 2015, 93, 47–160. [Google Scholar] [CrossRef] [PubMed]
- Domingo, E.; Martínez-Salas, E.; Sobrino, F.; De La Torre, J.C.; Portela, A.; Ortín, J.; López Galindez, C.; Pérez Breña, P.; Villanueva, N.; et al. The quasispecies extremely heterogeneous nature of viral RNA genome populations, biological relevance. Gene 1985, 40, 1–8. [Google Scholar] [CrossRef]
- Becher, P.; Orlich, M.; König, M.; Thiel, H.J. Nonhomologous RNA recombination in bovine viral diarrhea virus, molecular characterization of a variety of subgenomic RNAs isolated during an outbreak of fatal mucosal disease. J. Virol. 1999, 73, 5646–5653. [Google Scholar] [PubMed]
- Luzzago, C.; Ebranati, E.; Sassera, D.; Lo Presti, A.; Lauzi, S.; Gabanelli, E.; Ciccozzi, M.; Zehender, G. Spatial and temporal reconstruction of bovine viral diarrhea virus genotype 1 dispersion in Italy. Infect. Genet. Evol. 2012, 122, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Chernick, A.; Godson, D.L.; van der Meer, F. Metadata beyond the sequence enables the phylodynamic inference of bovine viral diarrhea virus type 1a isolates from Western Canada. Infect. Genet. Evol. 2014, 28, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.R.; Weber, E.L. Homologous recombination in bovine pestiviruses phylogenetic and statistic evidence. Infect. Genet. Evol. 2004, 4, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.N.; Streck, A.F.; Silveira, S.; Mósena, A.C.; Silva, M.S.; Canal, C.W. Homologous recombination in pestiviruses, identification of three putative novel events between different subtypes/genogroups. Infect. Genet. Evol. 2015, 30, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Kovago, C.; Hornyak, A.; Kekesi, V.; Rusvai, M. Demonstration of homologous recombination events in the evolution of bovine viral diarrhoea virus by in silico investigations. Acta Vet. Hung 2016, 643, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Ridpath, J.F.; Bolin, S.R. Delayed onset postvaccinal mucosal disease as a result of genetic recombination between genotype 1 and genotype 2 BVDV. Virology 1995, 212, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Fulton, R.W.; Ridpath, J.F.; Confer, A.W.; Saliki, J.T.; Burge, L.J.; Payton, M.E. Bovine viral diarrhoea virus antigenic diversity, impact on disease and vaccination programmes. Biologicals 2003, 312, 89–95. [Google Scholar] [CrossRef]
- Zimmer, G.M.; Wentink, G.H.; Bruschke, C.; Westenbrink, F.J.; Brinkhof, J.; Goey, I. Failure of foetal protection after vaccination against an experimental infection with bovine viral diarrhea virus. Vet. Microbiol. 2002, 894, 255–265. [Google Scholar] [CrossRef]
- Becher, P.; König, M.; Paton, D.J.; Thiel, H.J. Further characterization of border disease virus isolates, evidence for the presence of more than three species within the genus pestivirus. Virology 1995, 209, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Ridpath, J.F.; Bolin, S.R.; Dubovi, E.J. Segregation of bovine viral diarrhea virus into genotypes. Virology 1994, 205, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Abe, Y.; Tamura, T.; Torii, S.; Wakamori, S.; Nagai, M.; Mitsuhashi, K.; Mine, J.; Fujimoto, Y.; Nagashima, N.; Yoshino, F.; et al. Genetic and antigenic characterization of bovine viral diarrhea viruses isolated from cattle in Hokkaido, Japan. J. Vet. Med. Sci. 2016, 781, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Nagai, M.; Ito, T.; Sugita, S.; Genno, A.; Takeuchi, K.; Ozawa, T.; Sakoda, Y.; Nishimori, T.; Takamura, K.; Akashi, H. Genomic and serological diversity of bovine viral diarrhea virus in Japan. Arch. Virol. 2001, 1464, 685–696. [Google Scholar] [CrossRef]
- Ridpath, J.F.; Fulton, R.W.; Kirkland, P.D.; Neil, J.D. Prevalence and antigenic differences observed between bovine viral diarrhea virus subgenotypes isolated from cattle in Australia and feedlots in the southwestern United States. J. Vet. Diagn. Investig. 2010, 22, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Arias, P.; Orlich, M.; Prieto, M.; Cedillo Rosales, S.; Thiel, H.J.; Alvarez, M.; Becher, P. Genetic heterogeneity of bovine viral diarrhoea viruses from Spain. Vet. Microbiol. 2003, 96, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, B.; Letellier, C.; Collard, A.; Quenon, P.; Dehan, P.; Hamers, C.; Pastoret, P.; Kerkhofs, P. Genetic and antigenic variability in bovine viral diarrhea virus BVDV isolates from Belgium. Virus Res. 2002, 85, 17–28. [Google Scholar] [CrossRef]
- Mingala, C.N.; Konnai, S.; Tajima, M.; Onuma, M.; Ohashi, K. Classification of new BVDV isolates from Philippine water buffalo using the viral E2 region. J. Basic Microb. 2009, 49, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Silveira, S.; Weber, M.N.; Mósena, A.C.; da Silva, M.S.; Streck, A.F.; Pescador, C.A.; Flores, E.F.; Weiblen, R.; Driemeier, D.; Ridpath, J.F.; et al. Genetic Diversity of Brazilian Bovine Pestiviruses Detected Between 1995 and 2014. Transbound Emerg. Dis. 2015, 64, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Tajima, M.; Dubovi, E.J. Genetic and clinical analyses of bovine viral diarrhea virus isolates from dairy operations in the United States of America. J. Vet. Diagn. Investig. 2005, 17, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Nagai, M.; Hayashi, M.; Sugita, S.; Sakoda, Y.; Mori, M.; Murakami, T.; Ozawa, T.; Yamada, N.; Akashi, H. Phylogenetic analysis of bovine viral diarrhea viruses using five different genetic regions. Virus Res. 2004, 992, 103–113. [Google Scholar] [CrossRef]
- Xia, H.; Liu, L.; Wahlberg, N.; Baule, C.; Belák, S. Molecular phylogenetic analysis of bovine viral diarrhoea virus, a Bayesian approach. Virus Res. 2007, 130, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Dubey, R.; Rajukumar, K.; Tosh, C.; Tiwari, A.; Pitale, S.S.; Pradhan, H.K. Genetic and antigenic characterization of bovine viral diarrhea virus type 2 isolated from Indian goats (Capra hircus). Vet. Microbiol. 2007, 124, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Craig, M.I.; König, G.A.; Benitez, D.F.; Draghi, M.G. Molecular analyses detect natural coinfection of water buffaloes Bubalus bubalis with bovine viral diarrhea viruses BVDV in serologically negative animals. Rev. Argent Microbiol. 2015, 472, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Giangaspero, M.; Harasawa, R.; Weber, L.; Belloli, A. Taxonomic and epidemiological aspect of the bovine viral diarrhoea virus 2 through the observation of the secondary structures in the 5′ genomic untranslated region. Vet. Ital. 2008, 44, 319–345. [Google Scholar] [PubMed]
- Jones, L.R.; Zandomeni, R.; Weber, E.L. Genetic typing of bovine viral diarrhea virus isolates from Argentina. Vet. Microbiol. 2001, 814, 367–375. [Google Scholar] [CrossRef]
- Pecora, A.; Malacari, D.A.; Ridpath, J.F.; Perez Aguirreburualde, M.S.; Combessies, G.; Odeón, A.C.; Romera, S.A.; Golemba, M.D.; Wigdorovitz, A. First finding of genetic and antigenic diversity in 1b-BVDV isolates from Argentina. Res. Vet. Sci. 2014, 961, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Vilcek, S.; Durkovic, B.; Kolesárová, M.; Greiser-Wilke, I.; Paton, D. Genetic diversity of international bovine viral diarrhoea virus BVDV isolates, identification of a new BVDV-1 genetic group. Vet. Res. 2004, 35, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.V.; Konradt, G.; de Souza, S.O.; Bassuino, D.M.; Silveira, S.; Mósena, A.C.; Canal, C.W.; Pavarini, S.P.; Driemeier, D. Natural Outbreak of BVDV-1d-Induced Mucosal Disease Lacking Intestinal Lesions. Vet. Pathol. 2016, 542, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Flores, E.F.; Ridpath, J.F.; Weiblen, R.; Vogel, F.S.; Gil, L.H. Phylogenetic analysis of Brazilian bovine viral diarrhea virus type 2 BVDV-2 isolates, evidence for a subgenotype with in BVDV-2. Virus Res. 2002, 87, 51–60. [Google Scholar] [CrossRef]
- Lunardi, M.; Headley, S.A.; Lisbôa, J.A.; Amude, A.M.; Alfieri, A.A. Outbreak of acute bovine viral diarrhea in Brazilian beef cattle, clinicopathological findings and molecular characterization of a wild-type BVDV strain subtype 1b. Res. Vet. Sci. 2008, 853, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Mósena, A.C.; Weber, M.N.; Cibulski, S.P.; Silveira, S.; Silva, M.S.; Mayer, F.Q.; Canal, C.W. Genomic characterization of a bovine viral diarrhea virus subtype 1i in Brazil. Arch. Virol. 2017, 1624, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Otonel, R.A.; Alfieri, A.F.; Dezen, S.; Lunardi, M.; Headley, S.A.; Alfieri, A.A. The diversity of BVDV subgenotypes in a vaccinated dairy cattle herd in Brazil. Trop. Anim. Health. Prod. 2014, 461, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.N.; Silveira, S.; Machado, G.; Groff, F.H.; Mósena, A.C.; Budaszewski, R.F.; Dupont, P.M.; Corbellini, L.G.; Canal, C.W. High frequency of bovine viral diarrhea virus type 2 in Southern Brazil. Virus Res. 2014, 191, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Pizarro Lucero, J.; Celedón, M.O.; Aguilera, M.; De Calisto, A. Molecular characterization of pestiviruses isolated from bovines in Chile. Vet. Microbiol. 2006, 115, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Ståhl, K.; Benito, A.; Felmer, R.; Zuñiga, J.; Reinhardt, G.; Rivera, H.; Baule, C.; Moreno-López, J. Genetic diversity of bovine viral diarrhoea virus BVDV from Peru and Chile. Pesq. Vet. Brasil 2009, 29, 41–44. (In Portuguese) [Google Scholar] [CrossRef]
- Ahn, C.B.; Walz, P.H.; Kennedy, G.A.; Kapil, S. Biotype Genotype and Clinical presentation associated with bovine viral diarrhea virus BVDV isolates from cattle. Int. J. Appl. Res. Vet. Med. 2005, 34, 319–325. [Google Scholar]
- Campen, H.V.; Ridpath, J.; Williams, E.; Cavender, J.; Edwards, J.; Smith, S.; Sawyer, H. Isolation of bovine viral diarrhea virus from a free-ranging mule deer in Wyoming. J. Wildlife Dis. 2001, 372, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Darweesh, M.F.; Rajput, M.K.S.; Braun, L.J.; Ridpath, J.F.; Neill, J.D.; Chase, C.C.L. Characterization of the cytopathic BVDV strains isolated from 13 mucosal disease cases arising in a cattle herd. Virus Res. 2015, 195, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Evermann, J.F.; Ridpath, J.F. Clinical and epidemiologic observations of bovine viral diarrhea virus in the northwestern United States. Vet. Microbiol. 2002, 89, 129–139. [Google Scholar] [CrossRef]
- Fulton, R.W.; Ridpath, J.F.; Ore, S.; Confer, A.W.; Saliki, J.T.; Burge, L.J.; Payton, M.E. Bovine viral diarrhoea virus BVDV subgenotypes in diagnostic laboratory accessions, distribution of BVDV1a, 1b, and 2a subgenotypes. Vet. Microbiol. 2005, 111, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Fulton, R.W.; d’Offay, J.M.; Landis, C.; Miles, D.G.; Smith, R.A.; Saliki, J.T.; Ridpath, J.F.; Confer, A.W.; Neill, J.D.; Eberle, R.; et al. Detection and characterization of viruses as field and vaccine strains in feedlot cattle with bovine respiratory disease. Vaccine 2016, 3430, 3478–3492. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.G.; Anderson, R.R.; Yu, J.Z.; Zylich, N.C.; Kinde, H.; Carman, S.; Bedenice, D.; Dubovi, E.J. Genotyping and phylogenetic analysis of bovine viral diarrhea virus isolates from BVDV infected alpacas in North America. Vet. Microbiol. 2009, 136, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Pogranichniy, R.M.; Schnur, M.E.; Raizman, E.A.; Murphy, D.A.; Negron, M.; Thacker, H.L. Isolation and Genetic Analysis of Bovine Viral Diarrhea Virus from Infected Cattle in Indiana. Vet. Med. Int. 2011, 2011, 925910. [Google Scholar] [CrossRef] [PubMed]
- Workman, A.M.; Harhay, G.P.; Heaton, M.P.; Grotelueschen, D.M.; Sjeklocha, D.; Smith, T.P. Full-length coding sequences for 12 bovine viral diarrhea virus isolates from persistently infected cattle in a feedyard in Kansas. Genome Announc. 2015, 3, e00487-15. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhang, S.; Pace, L.; Wilson, F.; Wan, H.; Zhang, M. Combination of reverse transcription real-time polymerase chain reaction and antigen capture enzyme-linked immunosorbent assay for the detection of animals persistently infected with Bovine viral diarrhea virus. J. Vet. Diagn. Investig. 2011, 231, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Deregt, D.; Tessaro, S.V.; Baxi, M.K.; Berezowski, J.; Ellis, J.A.; Wu, J.T.; Gilbert, S.A. Isolation of bovine viral diarrhoea viruses from bison. Vet. Rec. 2005, 15715, 448–450. [Google Scholar] [CrossRef]
- Maya, L.; Puentes, R.; Reolón, E.; Acuña, P.; Riet, F.; Rivero, R.; Cristina, J.; Colina, R. Molecular diversity of bovine viral diarrhea virus in Uruguay. Arch. Virol. 2016, 1613, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Mahony, T.J.; McCarthy, F.M.; Gravel, J.L.; Corney, B.; Young, P.L.; Vilcek, S. Genetic analysis of bovine viral diarrhoea viruses from Australia. Vet. Microbiol. 2005, 106, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Latif, A.O.; Goyal, S.M.; Chander, Y.; Abdel-Moneim, A.S.; Tamam, S.M.; Madbouly, H.M. Isolation and molecular characterisation of a pestivirus from goats in Egypt. Acta Vet. Hung 2013, 612, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Soltan, M.A.; Wilkes, R.P.; Elsheery, M.N.; Elhaig, M.M.; Riley, M.C.; Kennedy, M.A. Circulation of bovine viral diarrhea virus-1 BVDV-1 in dairy cattle and buffalo farms in Ismailia Province, Egypt. J. Infect. Dev. Ctries 2015, 9, 1331–1337. [Google Scholar] [CrossRef] [PubMed]
- Thabti, F.; Bakkali Kassimi, L.; M’zah, A.; Ben Romdane, S.; Russo, P.; Ben Said, M.S.; Hammami, S.; Pepin, M. First detection and genetic characterization of bovine viral diarrhoea viruses BVDV types 1 and 2 in Tunisia. Rev. Med. Vet-Toulouse 2005, 156, 419–422. [Google Scholar]
- Baule, C.; van Vuuren, M.; Lowings, J.P.; Belák, S. Genetic heterogeneity of bovine viral diarrhoea viruses isolated in Southern Africa. Virus Res. 1997, 522, 205–220. [Google Scholar] [CrossRef]
- Kabongo, N.; Baule, C.; Van Vuuren, M. Molecular analysis of bovine viral diarrhoea virus isolates from South Africa. Onderstepoort J. Vet. Res. 2003, 704, 273–279. [Google Scholar] [CrossRef]
- Ularamu, H.G.; Sibeko, K.P.; Bosman, A.B.; Venter, E.H.; van Vuuren, M. Genetic characterization of bovine viral diarrhoea BVD viruses, confirmation of the presence of BVD genotype 2 in Africa. Arch. Virol. 2013, 1581, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Sun, C.Q.; Cao, S.J.; Lin, T.; Yuan, S.S.; Zhang, H.B.; Zhai, S.L.; Huang, L.; Shan, T.L.; Zheng, H.; et al. High prevalence of bovine viral diarrhea virus 1 in Chinese swine herds. Vet. Microbiol. 2012, 159, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Ji, S.; Fei, W.; Raza, S.; He, C.; Chen, Y.; Chen, H.; Guo, A. Prevalence Study and Genetic Typing of Bovine Viral Diarrhea Virus BVDV in Four Bovine Species in China. PLoS ONE 2015, 104, e0121718. [Google Scholar] [CrossRef]
- Gao, S.; Luo, J.; Du, J.; Lang, Y.; Cong, G.; Shao, J.; Lin, T.; Zhao, F.; Belák, S.; Liu, L.; et al. Serological and molecular evidence for natural infection of Bactrian camels with multiple subgenotypes of bovine viral diarrhea virus in Western China. Vet. Microbiol. 2013, 163, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Du, J.; Shao, J.; Lang, Y.; Lin, T.; Cong, G.; Zhao, F.; Belák, S.; Liu, L.; Chang, H.; et al. Genome analysis reveals a novel genetically divergent subgenotype of bovine viral diarrhea virus in China. Infect. Genet. Evol. 2014, 21, 489–491. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Du, J.; Tian, Z.; Xing, S.; Luo, J.; Liu, G.; Chang, H.; Yin, H. Genome Sequence of a Subgenotype 1a Bovine Viral Diarrhea Virus in China. Genome Announc. 2016, 4, e01280-16. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Cao, X.; Zheng, F.; Chen, Q.; Zhou, J.; Yin, H.; Liu, L.; Cai, X. Identification and characterization of a novel subgenotype of bovine viral diarrhea virus isolated from dairy cattle in Northwestern China. Virus Genes 2013, 462, 375–376. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Liu, L.; Zheng, F.; Chen, Q.; Li, Z.; Cao, X.; Yin, H.; Zhou, J.; Cai, X. Molecular investigation of bovine viral diarrhea virus infection in yaks (Bos gruniens) from Qinghai, China. Virol. J. 2014, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Li, W.; Yang, L.; Wang, J.; Cheng, S.; Wei, Y.; Wang, Q.; Zhang, W.; Hao, F.; Ding, Y.; et al. Primary surveys on molecular epidemiology of bovine viral diarrhea virus 1 infecting goats in Jiangsu province, China. BMC Vet. Res. 2016, 12, 181. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Wang, Y.; Wang, J.; Wang, J.Y.; Zhu, G.Q. Identification and genetic characterization of new bovine viral diarrhea virus genotype 2 strains in pigs isolated in China. Virus Genes 2013, 46, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Fan, Q.; Xie, Z.; Liu, J.; Pang, Y.; Deng, X.; Xie, L.; Luo, S.; Khan, M.I. Complete genome sequence of a bovine viral diarrhea virus strain isolated in southern China. Genome Announc. 2014, 2, 512–514. [Google Scholar] [CrossRef] [PubMed]
- Xue, F.; Zhu, Y.M.; Li, J.; Zhu, L.C.; Ren, X.G.; Feng, J.K.; Shi, H.F.; Gao, Y.R. Genotyping of bovine viral diarrhea viruses from cattle in China between 2005 and 2008. Vet. Microbiol. 2010, 143, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Shi, X.; Chen, C.; Wu, H. Genetic characterization of a noncytopathic bovine viral diarrhea virus 2b isolated from cattle in China. Virus Genes 2014, 49, 339–341. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Shi, X.; Tong, Q.; Wu, Y.; Xia, M.Q.; Ji, Y.; Xue, W.; Wu, H. A bovine viral diarrhea virus type 1a strain in China, isolation, identification, and experimental infection in calves. Virol. J. 2014, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- Weng, X.G.; Song, Q.J.; Wu, Q.; Liu, M.C.; Wang, M.L.; Wang, J.F. Genetic characterization of bovine viral diarrhea virus strains in Beijing, China and innate immune responses of peripheral blood mononuclear cells in persistently infected dairy cattle. J. Vet. Sci. 2015, 16, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Ma, C.; Li, X.Z.; Liu, Y.X.; Huang, K.; Cao, J. Genetic diversity of bovine viral diarrhoea virus in Beijing region, China from 2009 to 2010. Afr. J. Microbiol. Res. 2013, 74, 4934–4939. [Google Scholar]
- Zhang, S.; Tan, B.; Ding, Y.; Wang, F.; Guo, L.; Wen, Y.; Cheng, S.; Wu, H. Complete genome sequence and pathogenesis of bovine viral diarrhea virus JL-1 isolate from cattle in China. Virol. J. 2014, 11, 67. [Google Scholar] [CrossRef] [PubMed]
- Zhong, F.; Li, N.; Huang, X.; Guo, Y.; Chen, H.; Wang, X.; Shi, C.; Zhang, X. Genetic typing and epidemiologic observation of bovine viral diarrhea virus in Western China. Virus Genes 2011, 42, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.Q.; Lin, Y.Q.; Ding, X.Y.; Ren, M.; Tao, J.; Wang, J.Y.; Zhang, G.P.; Zhu, G.Q. Genomic sequencing and characterization of a Chinese isolate of Bovine viral diarrhea virus 2. Acta Virol. 2009, 53, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.Q.; Ren, M.; Lin, Y.Q.; Ding, X.Y.; Zhang, G.P.; Zhao, X.; Zhu, G.Q. Identification of a bovine viral diarrhea virus 2 isolated from cattle in China. Acta Virol. 2009, 53, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Lu, H.; Cao, Y.; Gai, X.; Guo, C.; Liu, Y.; Liu, J.; Wang, X. Molecular Characterization of a Novel Bovine Viral Diarrhea Virus Isolate SD-15. PLoS ONE 2016, 11, e0165044. [Google Scholar] [CrossRef] [PubMed]
- Behera, S.P.; Mishra, N.; Vilcek, S.; Rajukumar, K.; Nema, R.K.; Prakash, A.; Kalaiyarasu, S.; Dubey, S.C. Genetic and antigenic characterization of bovine viral diarrhoea virus type 2 isolated from cattle in India. Comp. Immunol. Microbiol. Infect. Dis. 2011, 34, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Pattnaik, B.; Vilcek, S.; Patil, SS.; Jain, P.; Swamy, N.; Bhatia, S.; Pradhan, H.K. Genetic typing of bovine viral diarrhoea virus isolates from India. Vet. Microbiol. 2004, 10, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Pitale, S.S.; Rajukumar, K.; Prakash, A.; Behera, S.P.; Nema, R.K.; Dubey, S.C. Genetic variety of bovine viral diarrhea virus 1 strains isolated from sheep and goats in India. Acta Virol. 2012, 56, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Rajukumar, K.; Vilcek, S.; Tiwari, A.; Satav, J.S.; Dubey, S.C. Molecular characterization of bovine viral diarrhea virus type 2 isolate originating from a native Indian sheep (Ovies aries). Vet. Microbiol. 2008, 130, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Vilcek, S.; Rajukumar, K.; Dubey, R.; Tiwari, A.; Galav, V.; Pradhan, H.K. Identification of bovine viral diarrhea virus type 1 in yaks (Bos poephagus grunniens) in the Himalayan region. Res. Vet. Sci. 2008, 84, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Matsuno, K.; Sakoda, Y.; Kameyama, K.; Tamai, K.; Ito, A.; Kida, H. Genetic and pathobiological characterization of bovine viral diarrhea viruses recently isolated from cattle in Japan. J. Vet. Med. Sci. 2007, 68, 515–520. [Google Scholar] [CrossRef]
- Nagai, M.; Hayashi, M.; Itou, M.; Fukutomi, T.; Akashi, H.; Kida, H.; Sakoda, Y. Identification of new genetic subtypes of bovine viral diarrhea virus genotype 1 isolated in Japan. Virus Genes 2008, 36, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Tateishi, K.; Shinohara, M.; Naoi, Y.; Shiokawa, M.; Aoki, H.; Ohmori, K.; Mizutani, T.; Shirai, J.; Nagai, M. Complete Genome Sequencing of Bovine Viral Diarrhea Virus 1, Subgenotypes 1n and 1o. Genome Announc. 2016, 4, e01744-15. [Google Scholar] [CrossRef] [PubMed]
- Tajima, M. Bovine viral diarrhea virus 1 is classified into different subgenotypes depending on the analyzed region within the viral genome. Vet. Microbiol. 2004, 99, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Kozasa, T.; Aoki, H.; Sekiguchi, H.; Morino, S.; Nakamura, S. Genomic analyses of bovine viral diarrhea viruses isolated from cattle imported into Japan between 1991 and 2005. Vet. Microbiol. 2008, 127, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.J.; Chae, J.B.; Chae, J.S.; Yu, D.H.; Park, J.; Park, B.K.; Kim, H.C.; Yoo, J.G.; Choi, K.S. Identification of bovine viral diarrhea virus infection in Saanen goats in the Republic of Korea. Trop. Anim. Health Prod. 2016, 48, 1079–1082. [Google Scholar] [CrossRef] [PubMed]
- Oem, J.K.; Hyun, B.H.; Cha, S.H.; Lee, K.K.; Kim, S.H.; Kim, H.R.; Park, C.K.; Joo, Y.S. Phylogenetic analysis and characterization of Korean bovine viral diarrhea viruses. Vet. Microbiol. 2009, 139, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.K.; Kim, B.H.; Kweon, C.H.; Park, J.K.; Kim, H.Y.; So, B.J.; Kim, I.J. Genetic typing of bovine viral diarrhea viruses BVDV circulating in Korea. J. Bacteriol. Virol. 2007, 37, 147–152. [Google Scholar] [CrossRef]
- Ochirkhuu, N.; Konnai, S.; Odbileg, R.; Odzaya, B.; Gansukh, S.; Murata, S.; Ohashi, K. Molecular detection and characterization of bovine viral diarrhea virus in Mongolian cattle and yaks. Arch. Virol. 2016, 161, 2279–2283. [Google Scholar] [CrossRef] [PubMed]
- Hornberg, A.; Fernández, S.R.; Vogl, C.; Vilcek, S.; Matt, M.; Fink, M.; Köfer, J.; Schöpf, K. Genetic diversity of pestivirus isolates in cattle from Western Austria. Vet. Microbiol. 2009, 135, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Krametter-Froetscher, R.; Duenser, M.; Preyler, B.; Theiner, A.; Benetka, V.; Moestl, K.; Baumgartner, W. Pestivirus infection in sheep and goats in West Austria. Vet. J. 2010, 186, 342–346. [Google Scholar] [CrossRef]
- Vilcek, S.; Greiser-Wilke, I.; Durkovic, B.; Obritzhauser, W.; Deutz, A.; Köfer, J. Genetic diversity of recent bovine viral diarrhoea viruses from the southeast of Austria (Styria). Vet. Microbiol. 2003, 91, 285–291. [Google Scholar] [CrossRef]
- Bedekovi, T.; Lojki, I.; Lemo, N.; Čač, Ž.; Cvetni, Ž.; Lojki, M.; Madi, J. Genetic typing of Croatian bovine viral diarrhea virus isolates. Vet. Arch. 2012, 82, 449–462. [Google Scholar]
- Robesova, B.; Kovarcik, K.; Vilcek, S. Genotyping of bovine viral diarrhoea virus isolates from the Czech Republic. Vet. Med. 2009, 54, 393–398. [Google Scholar]
- Nagy, A.; Fahnøe, U.; Rasmussen, T.B.; Uttenthal, A. Studies on genetic diversity of bovine viral diarrhea viruses in Danish cattle herds. Virus Genes 2014, 48, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Uttenthal, Å.; Stadejek, T.; Nylin, B. Genetic diversity of bovine viral diarrhoea viruses BVDV in Denmark during a 10-year eradication period. Acta Path. Micro Im. 2005, 113, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Jackova, A.; Novackova, M.; Pelletier, C.; Audeval, C.; Gueneau, E.; Haffar, A.; Petit, E.; Rehby, L.; Vilcek, S. The extended genetic diversity of BVDV-1, Typing of BVDV isolates from France. Vet. Res. Commun. 2008, 32, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Beer, M.; Wolf, G.; Kaaden, O.R. Phylogenetic analysis of the 5′-untranslated region of German BVDV type II isolates. J. Vet. Med. B Infect. Dis. Vet. Public Health 2002, 49, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Gethmann, J.; Homeier, T.; Holsteg, M.; Schirrmeier, H.; Saßerath, M.; Hoffmann, B.; Beer, M.; Conraths, F.J. BVD-2 outbreak leads to high losses in cattle farms in Western Germany. Heliyon 2015, 21, e00019. [Google Scholar] [CrossRef] [PubMed]
- Jenckel, M.; Höper, D.; Schirrmeier, H.; Reimann, I.; Goller, K.V.; Hoffmann, B.; Beer, M. Mixed triple, allied viruses in unique recent isolates of highly virulent type 2 bovine viral diarrhea virus detected by deep sequencing. J. Virol. 2014, 88, 6983–6992. [Google Scholar] [CrossRef] [PubMed]
- Schirrmeier, H. Three years of mandatory BVDV control in Germany–lessons to be learned. In Proceedings of the XXVIII World Buiatric Congress, Cairns, Australia, 27 July–1 August 2014; pp. 245–248. [Google Scholar]
- Tajima, M.; Frey, H.R.; Yamato, O.; Maede, Y.; Moennig, V.; Scholz, H.; Greiser-Wilke, I. Prevalence of genotypes 1 and 2 of bovine viral diarrhea virus in Lower Saxony, Germany. Virus Res. 2001, 76, 31–42. [Google Scholar] [CrossRef]
- Grahama, D.A.; Larena, I.E.M.C.; Brittaina, D.; O’reilly, P.J. Genetic typing of ruminant pestivirus strains from Northern Ireland and the Republic of Ireland. Res. Vet. Sci. 2001, 71, 127–134. [Google Scholar] [CrossRef]
- Guelbenzu-Gonzalo, M.P.; Cooper, L.; Brown, C.; Leinster, S.; O’Neill, R.; Doyle, L.; Graham, D.A. Genetic diversity of ruminant Pestivirus strains collected in Northern Ireland between 1999 and 2011 and the role of live ruminant imports. Ir. Vet. J. 2016, 69, 7. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, E.; Garvey, M.; Walsh, C.; Arkins, S.; Cullinane, A. Genetic typing of bovine viral diarrhoea virus in cattle on Irish farms. Res. Vet. Sci. 2017, 111, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Bazzucchi, M.; Bertolotti, L.; Giammarioli, M.; Casciari, C.; Rossi, E.; Rosati, S.; De Mia, G.M. Complete Genome Sequence of a Bovine Viral Diarrhea Virus Subgenotype 1h Strain Isolated in Italy. Genome Announc. 2017, 5, e01697-16. [Google Scholar] [CrossRef] [PubMed]
- Cannella, V.; Giudice, E.; Ciulli, S.; Marco, P.D.; Purpari, G.; Cascone, G.; Annalisa, G. Genotyping of bovine viral diarrhea viruses BVDV isolated from cattle in Sicily. Comp. Clin. Pathol. 2012, 21, 1733–1738. [Google Scholar] [CrossRef]
- Cerutti, F.; Luzzago, C.; Lauzi, S.; Ebranati, E.; Caruso, C.; Masoero, L.; Moreno, A.; Acutis, P.L.; Zehender, G.; Peletto, S. Phylogeography, phylodynamics and transmission chains of bovine viral diarrhea virus subtype 1f in Northern Italy. Infect. Genet. Evol. 2016, 45, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Ciulli, S.; Purpari, G.; Agnello, S.; Di Marco, P.; Di Bella, S.; Volpe, E.; Mira, F.; de Aguiar Saldanha Pinheiro, A.C.; Vullo, S.; Guercio, A. Evidence for Tunisian-Like Pestiviruses Presence in Small Ruminants in Italy Since 2007. Transbound Emerg. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Decaro, N.; Lucente, M.S.; Lanave, G.; Gargano, P.; Larocca, V.; Losurdo, M.; Ciambrone, L.; Marino, P.A.; Parisi, A.; Casalinuovo, F.; et al. Evidence for Circulation of Bovine Viral Diarrhoea Virus Type 2c in Ruminants in Southern Italy. Transbound Emerg. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Falcone, E.; Cordioli, P.; Tarantino, M.; Muscillo, M.; Rosa, G.; Tollis, M. Genetic Heterogeneity of Bovine Viral Diarrhoea Virus in Italy. Vet. Res. Commun. 2003, 27, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Giammarioli, M.; Ceglie, L.; Rossi, E.; Bazzucchi, M.; Casciari, C.; Petrini, S.; De Mia, G.M. Increased genetic diversity of BVDV-1, recent findings and implications thereof. Virus Genes 2015, 50, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Giammarioli, M.; Pellegrini, C.; Casciari, C.; Rossi, E.; Mia, G.M. Genetic diversity of Bovine viral diarrhea virus 1, Italian isolates clustered in at least seven subgenotypes. J. Vet. Diagn. Investig. 2008, 20, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Giangaspero, M.; Harasawa, R.; Zecconi, A.; Luzzago, C. Genotypic characteristics of bovine viral diarrhea virus 2 strains isolated in northern Italy. J. Vet. Med. Sci. 2001, 63, 1045–1049. [Google Scholar] [CrossRef] [PubMed]
- Luzzago, C.; Bandi, C.; Bronzo, V.; Ruffo, G.; Zecconi, A. Distribution pattern of bovine viral diarrhoea virus strains in intensive cattle herds in Italy. Vet. Microbiol. 2001, 83, 265–274. [Google Scholar] [CrossRef]
- Luzzago, C.; Lauzi, S.; Ebranati, E.; Giammarioli, M.; Moreno, A.; Cannella, V.; Masoero, L.; Canelli, E.; Guercio, A.; Caruso, C.; et al. Extended Genetic Diversity of Bovine Viral Diarrhea Virus and Frequency of Genotypes and Subtypes in Cattle in Italy between 1995 and 2013. Biomed. Res. Int. 2014, 2014, 147145. [Google Scholar] [CrossRef] [PubMed]
- Goga, I.; Berxholi, K.; Hulaj, B.; Sylejmani, D.; Yakobson, B.; Stram, Y. Genotyping and phylogenetic analysis of bovine viral diarrhea virus BVDV isolates in Kosovo. Vet. Ital. 2014, 50, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Kuta, A.; Polak, M.P.; Larska, M.; Żmudziński, J.F. Predominance of bovine viral diarrhea virus 1b and 1d subtypes during eight years of survey in Poland. Vet. Microbiol. 2013, 166, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Polak, M.P.; Kuta, A.; Rybałtowski, W.; Rola, J.; Larska, M.; Zmudziński, J.F. First report of bovine viral diarrhoea virus-2 infection in cattle in Poland. Vet. J. 2014, 202, 643–645. [Google Scholar] [CrossRef] [PubMed]
- Barros, S.C.; Ramos, F.; Paupério, S.; Thompson, G.; Fevereiro, M. Phylogenetic analysis of Portuguese bovine viral diarrhoea virus. Virus Res. 2006, 118, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Toplak, I.; Sandvib, T.; Barlič-Maganja, D.; Grom, J.; Paton, D.J. Genetic typing of bovine viral diarrhoea virus, most Slovenian isolates are of genotypes 1d and 1f. Vet. Microbiol. 2004, 99, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Toplak, I.; Kuhar, U.; Kušar, D.; Papić, B.; Koren, S.; Toplak, N. Complete Genome Sequence of a Bovine Viral Diarrhea Virus Subgenotype 1e Strain, SLO/2407/2006, Isolated in Slovenia. Genome Announc. 2016, 17, e01310-16. [Google Scholar] [CrossRef]
- Aduriz, G.; Atxaerandio, R.; Cortabarria, N. First detection of bovine viral diarrhoea virus type 2 in cattle in Spain. Vet. Rec. Open 2015, 2, e000110. [Google Scholar] [CrossRef] [PubMed]
- Diéguez, F.J.; Eiras, C.; Sanjuán, M.L.; Vilar, M.; Arnaiz, I.; Yus, E. Variabilidad genética del virus de la diarrea vírica bovina BVDV en Galicia. Producción Animal 2008, 239, 4–7. [Google Scholar]
- Factor, C.; Yus, E.; Eiras, C.; Sanjuan, M.L.; Cerviño, M.; Arnaiz, I.; Diéguez, F.J. Genetic diversity of bovine viral diarrhea viruses from the Galicia region of Spain. Vet. Rec. Open 2016, 3, e000196. [Google Scholar] [CrossRef] [PubMed]
- Hurtado, A.; García-Pérez, A.L.; Aduriz, G.; Juste, R.A. Genetic diversity of ruminant pestiviruses from Spain. Virus Res. 2003, 92, 67–73. [Google Scholar] [CrossRef]
- Partida, E.L.; Fernández, M.; Gutiérrez, J.; Esnal, A.; Benavides, J.; Pérez, V.; de la Torre, A.; Álvarez, M.; Esperón, F. Detection of Bovine Viral Diarrhoea Virus 2 as the Cause of Abortion Outbreaks on Commercial Sheep Flocks. Transbound Emerg. Dis. 2016, 641. [Google Scholar] [CrossRef]
- Rodríguez-Prieto, V.; Kukielka, D.; Rivera-Arroyo, B.; Martínez-López, B.; de las Heras, A.I.; Sánchez-Vizcaíno, J.M.; Vicente, J. Evidence of shared bovine viral diarrhea infections between red deer and extensively raised cattle in south-central Spain. BMC Vet. Res. 2016, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Ståhl, K.; Kampa, J.; Baule, C.; Isaksson, M.; Moreno-Lopez, J.; Belak, S.; Alenius, S.; Lindberg, A. Molecular epidemiology of bovine viral diarrhoea during the final phase of the Swedish BVD-eradication programme. Prev. Vet. Med. 2005, 72, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Bachofen, C.; Stalder, H.; Braun, U.; Hilbe, M.; Ehrensperger, F.; Peterhans, E. Co-existence of genetically and antigenically diverse bovine viral diarrhoea viruses in an endemic situation. Vet. Microbiol. 2008, 13, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Stalder, H.; Schweizer, M.; Bachofen, C. Complete Genome Sequence of a Bovine Viral Diarrhea Virus Subgenotype 1e Strain Isolated in Switzerland. Genome Announc. 2015, 3, e00636-15. [Google Scholar] [CrossRef] [PubMed]
- Stalder, H.; Hug, C.; Zanoni, R.; Vogt, H.R.; Peterhans, E.; Schweizer, M.; Bachofen, C. A nationwide database linking information on the hosts with sequence data of their virus strains, A useful tool for the eradication of bovine viral diarrhea BVD in Switzerland. Virus Res. 2016, 218, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Oguzoglu, T.C.; Muz, D.; Yılmaz, V.; Alkan, F.; Akça, Y.; Burgu, I. Molecular characterization of Bovine virus diarrhea viruses species 2 BVDV-2 from cattle in Turkey. Trop. Anim. Health Prod. 2010, 42, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Oğuzoğlu, T.C.; Muz, D.; Yilmaz, V.; Timurkan, M.Ö.; Alkan, F.; Akça, Y.; Burgu, İ. Molecular Characteristics of Bovine Virus Diarrhoea Virus 1 Isolates from Turkey, Approaches for an Eradication Programme. Transbound Emerg. Dis. 2012, 59, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Sarikaya, B.; Azkur, A.K.; Gazyagci, S.; Aslan, M.E. Genetic Variability of Bovine Viral Diarrhea Virus in the 5′UTR in the Central Anatolia of Turkey. Acta Sci. Vet. 2012, 40, 1013. [Google Scholar]
- Yeşilbağ, K.; Förster, C.; Bank-Wolf, B.; Yilmaz, Z.; Alkan, F.; Ozkul, A.; Burgu, I.; Cedillo, R.S.; Thiel, H.J.; König, M. Genetic heterogeneity of bovine viral diarrhoea virus BVDV isolates from Turkey, Identification of a new subgroup in BVDV-1. Vet. Microbiol. 2008, 130, 258–267. [Google Scholar]
- Yeşilbağ, K.; Förster, C.; Ozyiğit, M.O.; Alpay, G.; Tuncer, P.; Thiel, H.J.; König, M. Characterisation of bovine viral diarrhoea virus BVDV isolates from an outbreak with haemorrhagic enteritis and severe pneumonia. Vet. Microbiol. 2014, 169, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, H.; Altan, E.; Ridpath, J.; Turan, N. Genetic diversity and frequency of bovine viral diarrhea virus BVDV detected in cattle in Turkey. Comp. Immunol. Microb. 2012, 35, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Booth, R.E.; Thomas, C.J.; El-Attar, L.M.; Gunn, G.; Brownlie, J. A phylogenetic analysis of Bovine Viral Diarrhoea Virus BVDV isolates from six different regions of the UK and links to animal movement data. Vet. Res. 2013, 44, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Strong, R.; Errington, J.; Cook, R.; Ross-Smith, N.; Wakeley, P.; Steinbach, F. Increased phylogenetic diversity of bovine viral diarrhoea virus type 1 isolates in England and Wales since 2001. Vet. Microbiol. 2013, 162, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Vilcek, S.; Drew, T.W.; McGoldrick, A.; Paton, D.J. Genetic typing of bovine pestiviruses from England and Wales. Vet. Microbiol. 1999, 69, 227–237. [Google Scholar] [CrossRef]
- Wakeley, P.R.; Turner, J.L.; Ibata, G.; King, D.P.; Sandvik, T.; Howard, P.; Drew, T.W. Characterisation of a type 2 bovine viral diarrhoea virus isolated from cattle in the UK. Vet. Microbiol. 2004, 102, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Sakoda, Y.; Ozawa, S.; Damrongwatanapokin, S.; Sato, M.; Ishikawa, K.; Fukusho, A. Genetic heterogeneity of porcine and ruminant pestiviruses mainly isolated in Japan. Vet. Microbiol. 1999, 65, 75–86. [Google Scholar] [CrossRef]
- Felsenstein, J. PHYLIP Phylogeny Inference Package version 3.5c. Distributed by the Author. 1993. [Google Scholar]
- Saitou, N.; Nei, M. The neighbor-joining method, a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. Confidence limits on phylogenies, an approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Becher, P.; Shannon, A.D.; Tautz, N.; Thiel, H.J. Molecular characterization of border disease virus, a pestivirus from sheep. Virology 1994, 198, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Vilcek, S.; Mojzisová, J.; Bajová, V.; Paulík, S.; Strojný, L.; Durkovic, B.; Hipíková, V. A survey for BVDV antibodies in cattle farms in Slovakia and genetic typing of BVDV isolates from imported animals. Acta Vet. Hung. 2003, 51, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Vilcek, S.; Durkovic, B.; Kolesarova, M.; Paton, D.J. Genetic diversity of BVDV, Consequences for classification and molecular epidemiology. Prev. Vet. Med. 2005, 72, 31–35. [Google Scholar] [CrossRef] [PubMed]
| Country | Genomic Region | Year of Isolation | BVDV-1 | BVDV-2 | Reference | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | k | l | m | n | o | p | q | r | s | t | u | ? | a | b | c | d | ? | ||||
| Argentina | 5′UTR, Npro, E2 | 1984–2010 | 23 | 36 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 2 | 5 | 4 | 1 | 1 | 4 | (I) |
| Brazil | 5′UTR, Npro, E2 | 1994–2016 | 54 | 20 | 4 | 24 | 1 | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | 2 | 2 | 50 | - | - | 3 | (II) |
| Peru and Chile | 5′UTR | 1993–2004 | 3 | 29 | 2 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 5 | (III) |
| USA | 5′UTR, Npro, E2 | 1971–2015 | 184 | 652 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 3 | 129 | 4 | - | - | 125 | (IV) |
| Canada | 5′UTR | 1990–1993 | 1 | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 3 | - | - | - | - | (V) |
| Uruguay | 5′UTR, Npro | 2014 | 12 | - | - | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 1 | - | - | - | (VI) |
| Total number | 277 | 738 | 6 | 24 | 1 | 2 | 7 | 139 | 59 | 1 | 1 | 137 | ||||||||||||||||||
| Country | Genomic Region | Year of Isolation | BVDV-1 | BVDV-2 | Reference | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | k | l | m | n | o | p | q | r | s | t | u | a | b | c | d | ||||
| Australia | 5′UTR, Npro | 1971–2005 | 13 | 1 | 425 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 4 | - | - | - | (I) |
| Total number | 13 | 1 | 425 | 4 | ||||||||||||||||||||||||
| Country | Genomic Region | Year of Isolation | BVDV-1 | BVDV-2 | Reference | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | k | l | m | n | o | p | q | r | s | t | u | ? | a | b | c | d | ? | ||||
| Egypt | 5′UTR, Npro | 1994–2004 | - | 4 | - | - | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (I) |
| Tunisia | 5′UTR, Npro | 2001–2002 | - | 2 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 3 | - | - | - | - | (II) |
| South Africa | 5′UTR | 1990–2009 | 31 | 13 | 20 | 20 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 20 | - | - | - | - | - | (III) |
| Total number | 31 | 19 | 20 | 20 | 1 | 20 | 3 | |||||||||||||||||||||||
| Country | Genomic Region | Year of Isolation | BVDV-1 | BVDV-2 | Reference | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | k | l | m | n | o | p | q | r | s | t | u | ? | a | b | c | d | ? | ||||
| China | 5′UTR, Npro, E2 | 2005–2013 | 15 | 113 | 17 | 13 | - | - | - | - | - | - | - | - | 116 | - | 5 | 9 | 14 | - | - | - | 22 | 10 | 2 | 1 | - | - | 12 | (I) |
| India | 5′UTR, Npro, Erns-E1, E2, NS5B | 2000–2010 | - | 23 | 6 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 3 | 1 | - | - | - | (II) |
| Philippine | E2 | - | - | 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (III) |
| Japan | 5′UTR, Npro | 1975–2006 | 216 | 558 | 226 | - | - | - | - | - | - | 4 | - | - | 1 | 2 | 2 | - | - | - | - | - | - | 2 | 315 | - | - | - | 2 | (IV) |
| Korea | 5′UTR | 2005–2015 | 21 | 6 | 2 | - | - | - | - | - | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | 18 | - | - | - | 1 | (V) |
| Mongolia | 5′UTR | 2014 | 4 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 4 | - | - | - | - | (VI) |
| Total number | 256 | 703 | 251 | 13 | 4 | 117 | 3 | 7 | 9 | 14 | 22 | 12 | 342 | 2 | 15 | |||||||||||||||
| Country | Genomic Region | Year of Isolation | BVDV-1 | BVDV-2 | Reference | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | k | l | m | n | o | p | q | r | s | t | u | ? | a | b | c | d | ? | ||||
| Austria | 5′UTR, Npro | 1997–2006 | 4 | 52 | - | 33 | 6 | 142 | 7 | 154 | - | - | 3 | - | - | - | - | - | - | - | - | - | - | 1 | 1 | - | - | - | 3 | (I) |
| Belgium | E2 | 1991–2002 | 1 | 19 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 4 | 6 | - | - | - | 7 | (II) |
| Croatia | 5′UTR, Npro | 2007–2011 | - | 11 | - | - | - | 7 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (III) |
| Czech Republic | 5′UTR, Npro | 2004–2007 | - | 16 | - | 16 | 2 | 7 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (IV) |
| Denmark | 5′UTR, E2 | 1962–2012 | - | 16 | - | 32 | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (V) |
| Finland | 5′UTR, Npro | 1994–2004 | - | - | - | 5 | - | 1 | - | - | - | 2 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (VI) |
| France | 5′UTR, Npro | 1993–2005 | 3 | 15 | - | 3 | 46 | - | - | - | - | - | - | 3▲ | - | - | - | - | - | - | - | - | - | - | 2 | - | - | - | 3 | (VII) |
| Germany | 5′UTR, E2 | 1960–2014 | 1 | 31 | - | 24 | 24 | 65 | 3 | 17 | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | 11 | - | 16 | - | - | (VIII) |
| Hungary | 5′UTR, Npro | 1971–1998 | - | 2 | - | - | - | 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (IX) |
| Ireland | 5′UTR | 1968–2014 | 428 | 19 | - | 1 | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 12 | - | - | (X) |
| Italy | 5′UTR, Npro | 1966–2016 | 16 | 193 | 2 | 27 | 141 | 55 | 8 | 20 | - | - | 3 | 1▲ | - | - | - | - | - | 2▲ | 1 | 1 | 2 | 8 | 10 | - | - | - | 5 | (XI) |
| Kosovo | 5′UTR | 2011 | - | 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (XII) |
| Poland | 5′UTR, Npro | 2004–2011 | - | 31 | - | 24 | - | 8 | 2 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 4 | - | - | - | - | (XIII) |
| Portugal | 5′UTR | - | 6 | 19 | - | 3 | 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 1 | 2 | - | - | - | (XIV) |
| Slovakia | 5′UTR, Npro | 1994–2004 | - | - | - | 1 | 1 | 1 | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 1 | - | - | - | - | (XV) |
| Slovenia | 5′UTR, Npro, C | 1997–2006 | - | 4 | - | 17 | 1 | 21 | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (XVI) |
| Spain | 5′UTR, Npro | 1989–2015 | 3 | 162 | 2 | 9 | 8 | 2 | - | 2 | - | - | 1 | 1 | - | - | - | - | - | - | - | - | - | - | 2 | 6 | - | - | - | (XVII) |
| Sweden | 5′UTR, Npro | 2002–2004 | 7 | 28 | - | 77 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | (XVIII) | |
| Switzerland | 5′UTR, Npro | 2008–2012 | - | 35 | - | - | 137 | 1 | - | 114 | - | - | 71 | - | - | - | - | - | - | - | - | - | - | 1 | - | - | - | - | - | (XIX) |
| Turkey | 5′UTR, Npro, C, Erns, E2 | 1997–2012 | 7 | 11 | - | 7 | - | 20 | - | 1 | 1 | - | - | 34▲ | - | - | - | - | - | 3▲ | - | - | - | 7 | 5 | 1 | - | - | 14 | (XX) |
| United Kingdom | 5′UTR, Npro | 1966–2011 | 390 | 65 | - | 2 | 5 | 1 | - | - | 23 | 1 | - | - | - | - | - | - | - | - | - | - | - | 3 | 7 | - | - | - | - | (XXI) |
| Total number | 866 | 732 | 4 | 281 | 376 | 334 | 21 | 309 | 24 | 3 | 79 | 39 | 5 | 1 | 1 | 2 | 24 | 50 | 9 | 28 | 32 | |||||||||
| Country | Genomic Region | Year of Isolation | BVDV-1 | BVDV-2 | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | k | l | m | n | o | p | q | r | s | t | u | ? | a | b | c | d | ? | |||
| Americas | 5′UTR, Npro, E2 | 1971–2010 | 277 | 738 | 6 | 24 | 1 | - | - | - | 2 | - | - | - | - | - | - | - | - | - | - | - | - | 7 | 139 | 59 | 1 | 1 | 137 |
| Australia | 5′UTR, Npro | 1971–2005 | 13 | 1 | 425 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 4 | - | - | - | - |
| Africa | 5′UTR, Npro | 1994–2004 | 31 | 19 | 20 | 20 | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | 20 | 3 | - | - | - | - |
| Asia | 5′UTR, Npro, Erns-E1, E2, NS5B | 1975–2012 | 256 | 703 | 251 | 13 | - | - | - | - | - | 4 | - | - | 117 | 3 | 7 | 9 | 14 | - | - | 22 | 12 | 342 | 2 | - | - | 15 | |
| Europe | 5′UTR, Npro, C, Erns, E2 | 1962–2012 | 866 | 732 | 4 | 281 | 376 | 334 | 21 | 309 | 24 | 3 | 79 | 39 | - | - | - | - | - | 5 | 1 | 1 | 2 | 24 | 50 | 9 | 28 | - | 32 |
| Total number/individual subgenotype | 1443 | 2193 | 706 | 338 | 377 | 334 | 21 | 309 | 26 | 8 | 79 | 39 | 117 | 3 | 7 | 9 | 14 | 5 | 1 | 1 | 24 | 63 | 538 | 70 | 29 | 1 | 184 | ||
| Total number/species | 6117 | 822 | |||||||||||||||||||||||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeşilbağ, K.; Alpay, G.; Becher, P. Variability and Global Distribution of Subgenotypes of Bovine Viral Diarrhea Virus. Viruses 2017, 9, 128. https://doi.org/10.3390/v9060128
Yeşilbağ K, Alpay G, Becher P. Variability and Global Distribution of Subgenotypes of Bovine Viral Diarrhea Virus. Viruses. 2017; 9(6):128. https://doi.org/10.3390/v9060128
Chicago/Turabian StyleYeşilbağ, Kadir, Gizem Alpay, and Paul Becher. 2017. "Variability and Global Distribution of Subgenotypes of Bovine Viral Diarrhea Virus" Viruses 9, no. 6: 128. https://doi.org/10.3390/v9060128
APA StyleYeşilbağ, K., Alpay, G., & Becher, P. (2017). Variability and Global Distribution of Subgenotypes of Bovine Viral Diarrhea Virus. Viruses, 9(6), 128. https://doi.org/10.3390/v9060128





