The Broad Host Range and Genetic Diversity of Mammalian and Avian Astroviruses
Abstract
:1. Introduction
1.1. Genome
1.2. Classification
2. Human Astroviruses
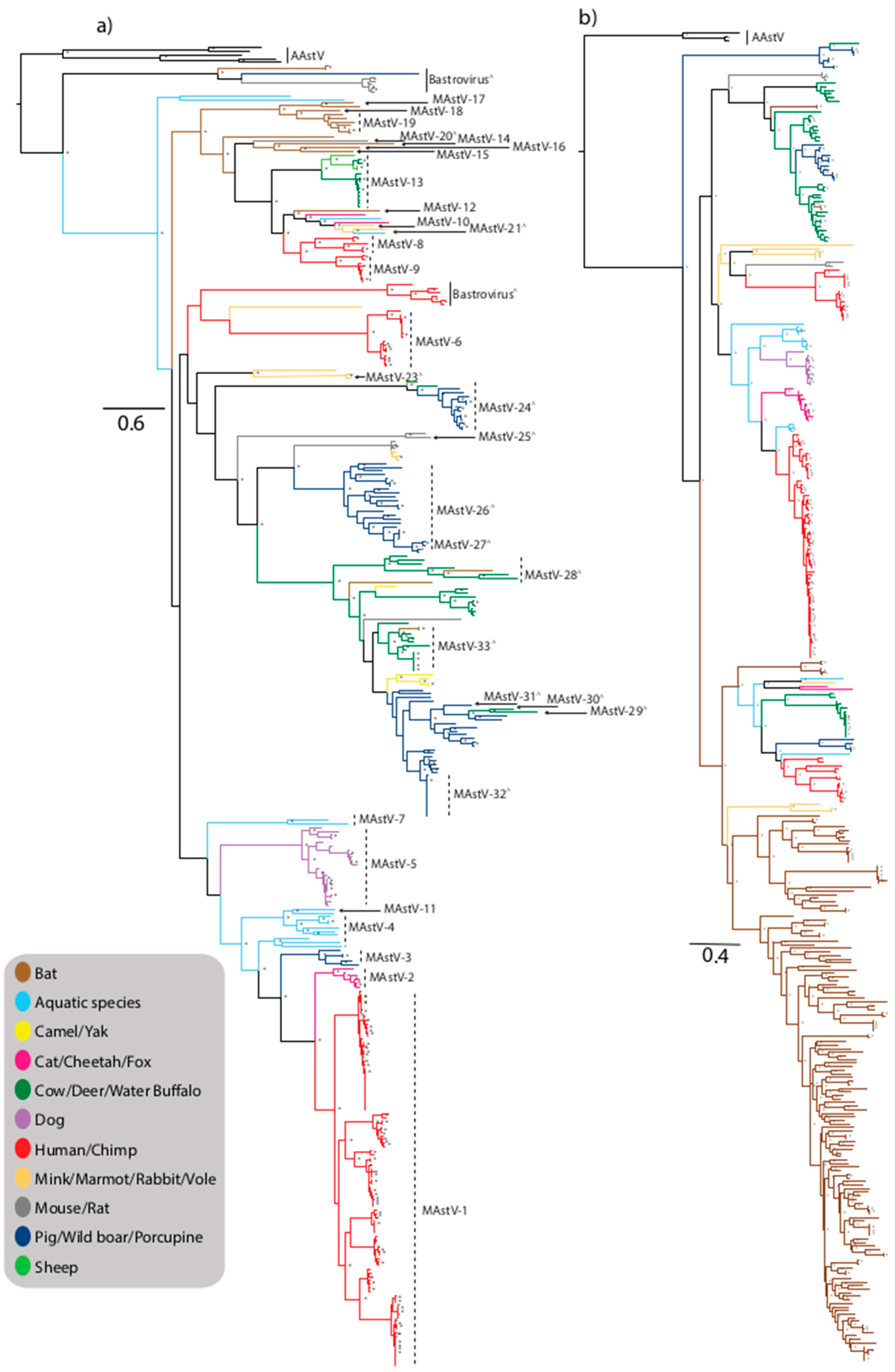
2.1. MAstV-1
2.2. MAstV-6
2.3. MAstV-8
2.4. MAstV-9
2.5. Bastrovirus
3. Non-Human MAstV
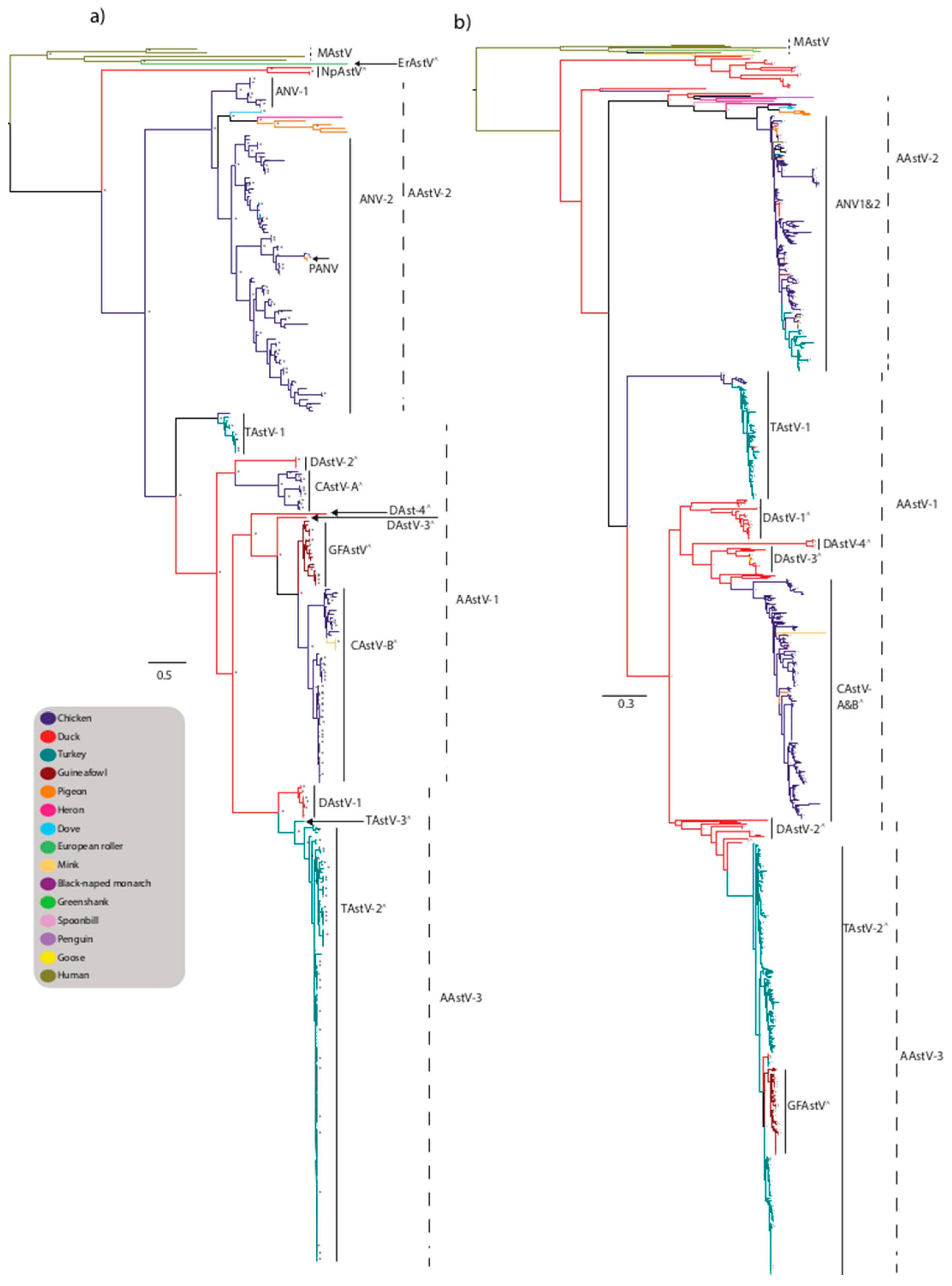
4. Avastrovirus
4.1. TAstV
4.2. CAstV
4.3. ANV
4.4. DAstV
4.5. Novel Astroviruses Detected in Wild Birds
5. Interspecies Transmission
6. Recombination
7. Future Considerations for Classification
Supplementary Materials
Conflicts of Interest
References
- Madeley, C.R.; Cosgrove, B.P. 28 nm Particles in fæces in infantile gastroenteritis. Lancet 1975, 306, 451–452. [Google Scholar] [CrossRef]
- Risco, C.; Carrascosa, J.L.; Pedregosa, A.M.; Humphrey, C.D.; Sanchez-Fauquier, A. Ultrastructure of human astrovirus serotype 2. J. Gen. Virol. 1995, 76, 2075–2080. [Google Scholar] [CrossRef] [PubMed]
- Appleton, H.; Higgins, P.G. Letter: Viruses and gastroenteritis in infants. Lancet 1975, 1, 1297. [Google Scholar] [CrossRef]
- Snodgrass, D.R.; Gray, E.W. Detection and transmission of 30 nm virus particles (astroviruses) in faeces of lambs with diarrhoea. Arch. Virol. 1977, 55, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Asplin, F.D. Duck hepatitis: Vaccination against two serological types. Vet. Rec. 1965, 77, 1529. [Google Scholar] [PubMed]
- Jeong, H.S.; Jeong, A.; Cheon, D.S. Epidemiology of astrovirus infection in children. Korean J. Pediatr. 2012, 55, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.I.; Noel, J.; Mitchell, D.; Herrmann, J.E.; Blacklow, N.R.; Pickering, L.K.; Dennehy, P.; Ruiz-Palacios, G.; de Guerrero, M.L.; Monroe, S.S. The changing epidemiology of astrovirus-associated gastroenteritis: A review. Arch. Virol. Suppl. 1996, 12, 287–300. [Google Scholar] [PubMed]
- Mitchell, D.K.; Matson, D.O.; Jiang, X.; Berke, T.; Monroe, S.S.; Carter, M.J.; Willcocks, M.M.; Pickering, L.K. Molecular epidemiology of childhood astrovirus infection in child care centers. J. Infect. Dis. 1999, 180, 514–517. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Li, L.; Victoria, J.; Oderinde, B.; Mason, C.; Pandey, P.; Zaidi, S.Z.; Delwart, E. Multiple novel astrovirus species in human stool. J. Gen. Virol. 2009, 90, 2965–2972. [Google Scholar] [CrossRef] [PubMed]
- Cordey, S.; Vu, D.L.; Schibler, M.; L’Huillier, A.G.; Brito, F.; Docquier, M.; Posfay-Barbe, K.M.; Petty, T.J.; Turin, L.; Zdobnov, E.M.; et al. Astrovirus MLB2, a new gastroenteric virus associated with meningitis and disseminated infection. Emerg. Infect. Dis. 2016, 22, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Quan, P.L.; Wagner, T.A.; Briese, T.; Torgerson, T.R.; Hornig, M.; Tashmukhamedova, A.; Firth, C.; Palacios, G.; Baisre-De-Leon, A.; Paddock, C.D.; et al. Astrovirus encephalitis in boy with X-linked agammaglobulinemia. Emerg. Infect. Dis. 2010, 16, 918–925. [Google Scholar] [CrossRef] [PubMed]
- De Benedictis, P.; Schultz-Cherry, S.; Burnham, A.; Cattoli, G. Astrovirus infections in humans and animals—Molecular biology, genetic diversity, and interspecies transmissions. Infect. Genet. Evol. 2011, 11, 1529–1544. [Google Scholar] [CrossRef] [PubMed]
- Moser, L.A.; Schultz-Cherry, S. Pathogenesis of astrovirus infection. Viral Immunol. 2005, 18, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Bouzalas, I.G.; Wuthrich, D.; Walland, J.; Drogemuller, C.; Zurbriggen, A.; Vandevelde, M.; Oevermann, A.; Bruggmann, R.; Seuberlich, T. Neurotropic astrovirus in cattle with nonsuppurative encephalitis in Europe. J. Clin. Microbiol. 2014, 52, 3318–3324. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Diab, S.; McGraw, S.; Barr, B.; Traslavina, R.; Higgins, R.; Talbot, T.; Blanchard, P.; Rimoldi, G.; Fahsbender, E.; et al. Divergent astrovirus associated with neurologic disease in cattle. Emerg. Infect. Dis. 2013, 19, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Schlottau, K.; Schulze, C.; Bilk, S.; Hanke, D.; Hoper, D.; Beer, M.; Hoffmann, B. Detection of a novel bovine astrovirus in a cow with encephalitis. Transbound. Emerg. Dis. 2016, 63, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Blomstrom, A.L.; Widen, F.; Hammer, A.S.; Belak, S.; Berg, M. Detection of a novel astrovirus in brain tissue of mink suffering from shaking mink syndrome by use of viral metagenomics. J. Clin. Microbiol. 2010, 48, 4392–4396. [Google Scholar] [CrossRef] [PubMed]
- Bosch, A.; Pintó, R.M.; Guix, S. Human Astroviruses. Clin. Microbiol. Rev. 2014, 27, 1048–1074. [Google Scholar] [CrossRef] [PubMed]
- Firth, A.E.; Atkins, J.F. Candidates in astroviruses, seadornaviruses, cytorhabdoviruses and coronaviruses for +1 frame overlapping genes accessed by leaky scanning. Virol. J. 2010, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Monroe, S.S.; Koonin, E.V.; Stine, S.E.; Glass, R.I. RNA sequence of astrovirus: Distinctive genomic organization and a putative retrovirus-like ribosomal frameshifting signal that directs the viral replicase synthesis. Proc. Natl. Acad. Sci. USA 1993, 90, 10539–10543. [Google Scholar] [CrossRef] [PubMed]
- Walter, J.E.; Mitchell, D.K.; Guerrero, M.L.; Berke, T.; Matson, D.O.; Monroe, S.S.; Pickering, L.K.; Ruiz-Palacios, G. Molecular epidemiology of human astrovirus diarrhea among children from a periurban community of Mexico City. J. Infect. Dis. 2001, 183, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.H.; Kakizawa, J.; Wen, L.Y.; Shimizu, M.; Nishio, O.; Fang, Z.Y.; Ushijima, H. Genetic analysis of the capsid region of astroviruses. J. Med. Virol. 2001, 64, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Dong, L.; Mendez, E.; Tao, Y. Crystal structure of the human astrovirus capsid spike. Proc. Natl. Acad. Sci. USA 2011, 108, 12681–12686. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, T. Novel human astroviruses: Challenges for developing countries. VirusDisease 2014, 25, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Bosch, A.; Guix, S.; Krishna, N.K.; Méndez, E.; Monroe, S.S.; Pantin-Jackwood, M.; Schultz-Cherry, S. Family—Astroviridae. In Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Lefkowitz, E., Adams, M.J., Carstens, E.B., Eds.; Elsevier: San Diego, CA, USA, 2012; pp. 953–959. [Google Scholar]
- Rivera, R.; Nollens, H.H.; Venn-Watson, S.; Gulland, F.M.D.; Wellehan, J.F., Jr. Characterization of phylogenetically diverse astroviruses of marine mammals. J. Gen. Virol. 2010, 91, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.; Lau, S.K.; Teng, J.L.; Tsang, A.K.; Joseph, S.; Xie, J.; Jose, S.; Fan, R.Y.; Wernery, U.; Yuen, K.Y. A novel astrovirus from dromedaries in the Middle East. J. Gen. Virol. 2015, 96, 2697–2707. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, E.A.; Small, C.T.; Freiden, P.; Feeroz, M.M.; Matsen, F.A.T.; San, S.; Hasan, M.K.; Wang, D.; Jones-Engel, L.; Schultz-Cherry, S. Non-human Primates harbor diverse mammalian and avian astroviruses including those associated with human infections. PLoS Pathog. 2015, 11, e1005225. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.A.; Hoang, L.; Pham le, D.; Hoang, K.T.; Mizuguchi, M.; Okitsu, S.; Ushijima, H. Identification of human astrovirus infections among children with acute gastroenteritis in the southern part of Vietnam during 2005–2006. J. Med. Virol. 2008, 80, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Wunderli, W.; Meerbach, A.; Guengoer, T.; Berger, C.; Greiner, O.; Caduff, R.; Trkola, A.; Bossart, W.; Gerlach, D.; Schibler, M.; et al. Astrovirus infection in hospitalized infants with severe combined immunodeficiency after allogeneic hematopoietic stem cell transplantation. PLoS ONE 2011, 6, e27483. [Google Scholar] [CrossRef] [PubMed]
- Shaukat, S.; Angez, M.; Alam, M.M.; Jebbink, M.F.; Deijs, M.; Canuti, M.; Sharif, S.; de Vries, M.; Khurshid, A.; Mahmood, T.; et al. Identification and characterization of unrecognized viruses in stool samples of non-polio acute flaccid paralysis children by simplified VIDISCA. Virol. J. 2014, 11, 146. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, J.B.; Lee, T.W. Human astrovirus serotypes. Lancet 1984, 2, 1405. [Google Scholar] [CrossRef]
- Herrmann, J.E.; Hudson, R.W.; Perron-Henry, D.M.; Kurtz, J.B.; Blacklow, N.R. Antigenic characterization of cell-cultivated astrovirus serotypes and development of astrovirus-specific monoclonal antibodies. J. Infect. Dis. 1988, 158, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Belliot, G.; Laveran, H.; Monroe, S.S. Detection and genetic differentiation of human astroviruses: Phylogenetic grouping varies by coding region. Arch. Virol. 1997, 142, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Gabbay, Y.B.; Leite, J.P.; Oliveira, D.S.; Nakamura, L.S.; Nunes, M.R.; Mascarenhas, J.D.; Heinemann, M.B.; Linhares, A.C. Molecular epidemiology of astrovirus type 1 in Belem, Brazil, as an agent of infantile gastroenteritis, over a period of 18 years (1982–2000): Identification of two possible new lineages. Virus Res. 2007, 129, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Guix, S.; Caballero, S.; Villena, C.; Bartolome, R.; Latorre, C.; Rabella, N.; Simo, M.; Bosch, A.; Pinto, R.M. Molecular epidemiology of astrovirus infection in Barcelona, Spain. J. Clin. Microbiol. 2002, 40, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.A.; Cardoso, D.D.; Schreier, E. Molecular characterization of human astroviruses isolated in Brazil, including the complete sequences of astrovirus genotypes 4 and 5. Arch. Virol. 2006, 151, 1405–1417. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Pinto, P.; Tummolo, F.; De Grazia, S.; Giammanco, G.M.; Medici, M.C.; Ganesh, B.; L’Homme, Y.; Farkas, T.; Jakab, F.; et al. Analysis of the ORF2 of human astroviruses reveals lineage diversification, recombination and rearrangement and provides the basis for a novel sub-classification system. Arch. Virol. 2014, 159, 3185–3196. [Google Scholar] [CrossRef] [PubMed]
- Finkbeiner, S.R.; Kirkwood, C.D.; Wang, D. Complete genome sequence of a highly divergent astrovirus isolated from a child with acute diarrhea. Virol. J. 2008, 5, 117. [Google Scholar] [CrossRef] [PubMed]
- Vu, D.-L.; Cordey, S.; Brito, F.; Kaiser, L. Novel human astroviruses: Novel human diseases? J. Clin. Virol. 2016, 82, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Holtz, L.R.; Bauer, I.K.; Jiang, H.; Belshe, R.; Freiden, P.; Schultz-Cherry, S.L.; Wang, D. Seroepidemiology of astrovirus MLB1. Clin. Vaccine Immunol. 2014, 21, 908–911. [Google Scholar] [CrossRef] [PubMed]
- Finkbeiner, S.R.; Holtz, L.R.; Jiang, Y.; Rajendran, P.; Franz, C.J.; Zhao, G.; Kang, G.; Wang, D. Human stool contains a previously unrecognized diversity of novel astroviruses. Virol. J. 2009, 6, 161. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Holtz, L.R.; Bauer, I.; Franz, C.J.; Zhao, G.; Bodhidatta, L.; Shrestha, S.K.; Kang, G.; Wang, D. Comparison of novel MLB-clade, VA-clade and classic human astroviruses highlights constrained evolution of the classic human astrovirus nonstructural genes. Virology 2013, 436, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Meyer, C.T.; Bauer, I.K.; Antonio, M.; Adeyemi, M.; Saha, D.; Oundo, J.O.; Ochieng, J.B.; Omore, R.; Stine, O.C.; Wang, D.; et al. Prevalence of classic, MLB-clade and VA-clade Astroviruses in Kenya and The Gambia. Virol. J. 2015, 12, 78. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.G.; Nordgren, J.; Ouermi, D.; Simpore, J.; Nitiema, L.W.; Deng, X.; Delwart, E. New astrovirus in human feces from Burkina Faso. J. Clin. Virol. 2014, 60, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Finkbeiner, S.R.; Li, Y.; Ruone, S.; Conrardy, C.; Gregoricus, N.; Toney, D.; Virgin, H.W.; Anderson, L.J.; Vinje, J.; Wang, D.; Tong, S. Identification of a novel astrovirus (astrovirus VA1) associated with an outbreak of acute gastroenteritis. J. Virol. 2009, 83, 10836–10839. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; Morfopoulou, S.; Hubb, J.; Emmett, W.A.; Ip, W.; Shah, D.; Brooks, T.; Paine, S.M.; Anderson, G.; Virasami, A.; et al. Astrovirus VA1/HMO-C: An increasingly recognized neurotropic pathogen in immunocompromised patients. Clin. Infect. Dis. 2015, 60, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Cordey, S.; Brito, F.; Vu, D.L.; Turin, L.; Kilowoko, M.; Kyungu, E.; Genton, B.; Zdobnov, E.M.; D’Acremont, V.; Kaiser, L. Astrovirus VA1 identified by next-generation sequencing in a nasopharyngeal specimen of a febrile Tanzanian child with acute respiratory disease of unknown etiology. Emerg. Microbes Infect. 2016, 5, e67. [Google Scholar] [CrossRef] [PubMed]
- Burbelo, P.D.; Ching, K.H.; Esper, F.; Iadarola, M.J.; Delwart, E.; Lipkin, W.I.; Kapoor, A. Serological studies confirm the novel astrovirus HMOAstV-C as a highly prevalent human infectious agent. PLoS ONE 2011, 6, e22576. [Google Scholar] [CrossRef] [PubMed]
- Oude Munnink, B.B.; Cotten, M.; Canuti, M.; Deijs, M.; Jebbink, M.F.; van Hemert, F.J.; Phan, M.V.T.; Bakker, M.; Jazaeri Farsani, S.M.; Kellam, P.; et al. A novel astrovirus-like RNA virus detected in human stool. Virus Evol. 2016, 2, vew005. [Google Scholar] [CrossRef] [PubMed]
- Pankovics, P.; Boros, A.; Kiss, T.; Delwart, E.; Reuter, G. Detection of a mammalian-like astrovirus in bird, European roller (Coracias garrulus). Infect. Genet. Evol. 2015, 34, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, Y.; Zimmer, J.F.; Moise, N.S.; Scott, F.W. Detection of astroviruses in feces of a cat with diarrhea. Brief report. Arch. Virol. 1981, 70, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Lukashov, V.V.; Goudsmit, J. Evolutionary relationships among Astroviridae. J. Gen. Virol. 2002, 83, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Atkins, A.; Wellehan, J.F.; Childress, A.L.; Archer, L.L.; Fraser, W.A.; Citino, S.B. Characterization of an outbreak of astroviral diarrhea in a group of cheetahs (Acinonyx jubatus). Vet. Microbiol. 2009, 136, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Moschidou, P.; Catella, C.; Larocca, V.; Pinto, P.; Losurdo, M.; Corrente, M.; Lorusso, E.; Banyai, K.; Decaro, N.; et al. Enteric disease in dogs naturally infected by a novel canine astrovirus. J. Clin. Microbiol. 2012, 50, 1066–1069. [Google Scholar] [CrossRef] [PubMed]
- Williams, F.P., Jr. Astrovirus-like, coronavirus-like, and parvovirus-like particles detected in the diarrheal stools of beagle pups. Arch. Virol. 1980, 66, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Bridger, J.C. Detection by electron microscopy of caliciviruses, astroviruses and rotavirus-like particles in the faeces of piglets with diarrhoea. Vet. Rec. 1980, 107, 532–533. [Google Scholar] [PubMed]
- Luo, Z.; Roi, S.; Dastor, M.; Gallice, E.; Laurin, M.A.; L’Homme, Y. Multiple novel and prevalent astroviruses in pigs. Vet. Microbiol. 2011, 149, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Wang, C.; Tong, W.; Zheng, H.; Hua, X.; Yang, S.; Guo, Y.; Zhang, W.; Tong, G. Complete genome of a novel porcine astrovirus. J. Virol. 2012, 86, 13820–13821. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.T.; Halbur, P.G.; Opriessnig, T. Complete genome sequence of a newly identified porcine astrovirus genotype 3 strain US-MO123. J. Virol. 2012, 86, 13126. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.T.; Gimenez-Lirola, L.G.; Gerber, P.F.; Jiang, Y.H.; Halbur, P.G.; Opriessnig, T. Identification and characterization of novel porcine astroviruses (PAstVs) with high prevalence and frequent co-infection of individual pigs with multiple PAstV types. J. Gen. Virol. 2013, 94, 570–582. [Google Scholar] [CrossRef] [PubMed]
- Ulloa, J.C.; Gutierrez, M.F. Genomic analysis of two ORF2 segments of new porcine astrovirus isolates and their close relationship with human astroviruses. Can. J. Microbiol. 2010, 56, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Reuter, G.; Nemes, C.; Boros, A.; Kapusinszky, B.; Delwart, E.; Pankovics, P. Astrovirus in wild boars (Sus scrofa) in Hungary. Arch. Virol. 2012, 157, 1143–1147. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Chmura, A.A.; Li, J.; Zhu, G.; Desmond, J.S.; Zhang, Y.; Zhang, W.; Epstein, J.H.; Daszak, P.; Shi, Z. Detection of diverse novel astroviruses from small mammals in China. J. Gen. Virol. 2014, 95, 2442–2449. [Google Scholar] [CrossRef] [PubMed]
- Reuter, G.; Pankovics, P.; Delwart, E.; Boros, A. Identification of a novel astrovirus in domestic sheep in Hungary. Arch. Virol. 2012, 157, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Woode, G.N.; Bridger, J.C. Isolation of small viruses resembling astroviruses and caliciviruses from acute enteritis of calves. J. Med. Microbiol. 1978, 11, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Wuthrich, D.; Boujon, C.L.; Truchet, L.; Selimovic-Hamza, S.; Oevermann, A.; Bouzalas, I.G.; Bruggmann, R.; Seuberlich, T. Exploring the virome of cattle with non-suppurative encephalitis of unknown etiology by metagenomics. Virology 2016, 493, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Seuberlich, T.; Wuthrich, D.; Selimovic-Hamza, S.; Drogemuller, C.; Oevermann, A.; Bruggmann, R.; Bouzalas, I. Identification of a second encephalitis-associated astrovirus in cattle. Emerg. Microbes Infect. 2016, 5, e5. [Google Scholar] [CrossRef] [PubMed]
- Selimovic-Hamza, S.; Bouzalas, I.G.; Vandevelde, M.; Oevermann, A.; Seuberlich, T. Detection of Astrovirus in Historical Cases of European Sporadic Bovine Encephalitis, Switzerland 1958–1976. Front. Vet. Sci. 2016, 3, 91. [Google Scholar] [CrossRef] [PubMed]
- Oem, J.K.; An, D.J. Phylogenetic analysis of bovine astrovirus in Korean cattle. Virus Genes 2014, 48, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Alfred, N.; Liu, H.; Li, M.L.; Hong, S.F.; Tang, H.B.; Wei, Z.Z.; Chen, Y.; Li, F.K.; Zhong, Y.Z.; Huang, W.J. Molecular epidemiology and phylogenetic analysis of diverse bovine astroviruses associated with diarrhea in cattle and water buffalo calves in China. J. Vet. Med. Sci. 2015, 77, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Woode, G.N.; Gourley, N.E.; Pohlenz, J.F.; Liebler, E.M.; Mathews, S.L.; Hutchinson, M.P. Serotypes of bovine astrovirus. J. Clin. Microbiol. 1985, 22, 668–670. [Google Scholar] [PubMed]
- Smits, S.L.; van Leeuwen, M.; Kuiken, T.; Hammer, A.S.; Simon, J.H.; Osterhaus, A.D. Identification and characterization of deer astroviruses. J. Gen. Virol. 2010, 91, 2719–2722. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.F.; Kondov, N.O.; Hayashimoto, N.; Uchida, R.; Cha, Y.; Beyer, A.I.; Wong, W.; Pesavento, P.A.; Suemizu, H.; Muench, M.O.; et al. Identification of an astrovirus commonly infecting laboratory mice in the US and Japan. PLoS ONE 2013, 8, e66937. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, C.C.; Loh, J.; Zhao, G.; Stappenbeck, T.S.; Wang, D.; Huang, H.V.; Virgin, H.W.; Thackray, L.B. Adaptive immunity restricts replication of novel murine astroviruses. J. Virol. 2012, 86, 12262–12270. [Google Scholar] [CrossRef] [PubMed]
- Englund, L.; Chriel, M.; Dietz, H.H.; Hedlund, K.O. Astrovirus epidemiologically linked to pre-weaning diarrhoea in mink. Vet. Microbiol. 2002, 85, 1–11. [Google Scholar] [CrossRef]
- Chu, D.K.W.; Poon, L.L.M.; Guan, Y.; Peiris, J.S.M. Novel astroviruses in insectivorous bats. J. Virol. 2008, 82, 9107–9114. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.C.; Chu, D.K.; Liu, W.; Dong, B.Q.; Zhang, S.Y.; Zhang, J.X.; Li, L.F.; Vijaykrishna, D.; Smith, G.J.; Chen, H.L.; et al. Detection of diverse astroviruses from bats in China. J. Gen. Virol. 2009, 90, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Li, J.; Hu, G.; Chen, Z.; Wu, Y.; Chen, Y.; Chen, Z.; Liao, Y.; Zhou, J.; Ke, X.; et al. Isolation and phylogenetic characterization of bat astroviruses in southern China. Arch. Virol. 2011, 156, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Victoria, J.G.; Wang, C.; Jones, M.; Fellers, G.M.; Kunz, T.H.; Delwart, E. Bat guano virome: Predominance of dietary viruses from insects and plants plus novel mammalian viruses. J. Virol. 2010, 84, 6955–6965. [Google Scholar] [CrossRef] [PubMed]
- Drexler, J.F.; Corman, V.M.; Wegner, T.; Tateno, A.F.; Zerbinati, R.M.; Gloza-Rausch, F.; Seebens, A.; Muller, M.A.; Drosten, C. Amplification of emerging viruses in a bat colony. Emerg. Infect. Dis. 2011, 17, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Kemenesi, G.; Dallos, B.; Gorfol, T.; Boldogh, S.; Estok, P.; Kurucz, K.; Oldal, M.; Nemeth, V.; Madai, M.; Banyai, K.; et al. Novel European lineages of bat astroviruses identified in Hungary. Acta Virol. 2014, 58, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Dufkova, L.; Strakova, P.; Sirmarova, J.; Salat, J.; Moutelikova, R.; Chrudimsky, T.; Bartonicka, T.; Nowotny, N.; Ruzek, D. Detection of diverse novel bat astrovirus sequences in the Czech Republic. Vector Borne Zoonotic Dis. 2015, 15, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Gough, R.E.; Collins, M.S.; Borland, E.; Keymer, L.F. Astrovirus-like particles associated with hepatitis in ducklings. Vet. Rec. 1984, 114, 279. [Google Scholar] [CrossRef] [PubMed]
- Pantin-Jackwood, M.J.; Strother, K.O.; Mundt, E.; Zsak, L.; Day, J.M.; Spackman, E. Molecular characterization of avian astroviruses. Arch. Virol. 2011, 156, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.K.; Leung, C.Y.; Perera, H.K.; Ng, E.M.; Gilbert, M.; Joyner, P.H.; Grioni, A.; Ades, G.; Guan, Y.; Peiris, J.S.; et al. A novel group of avian astroviruses in wild aquatic birds. J. Virol. 2012, 86, 13772–13778. [Google Scholar] [CrossRef] [PubMed]
- Bidin, M.; Lojkic, I.; Tisljar, M.; Bidin, Z.; Majnaric, D. Astroviruses associated with stunting and pre-hatching mortality in duck and goose embryos. Avian Pathol. 2012, 41, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Todd, D.; Wilkinson, D.S.; Jewhurst, H.L.; Wylie, M.; Gordon, A.W.; Adair, B.M. A seroprevalence investigation of chicken astrovirus infections. Avian Pathol. 2009, 38, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.I.; Icard, A.H.; Linnemann, E.; Sellers, H.S.; Mundt, E. Determination of the full length sequence of a chicken astrovirus suggests a different replication mechanism. Virus Genes 2012, 44, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Wang, F.; Zhang, D. Complete sequence of a novel duck astrovirus. Arch. Virol. 2014, 159, 2823–2827. [Google Scholar] [CrossRef] [PubMed]
- McNulty, M.S.; Curran, W.L.; McFerran, J.B. Detection of astroviruses in turkey faeces by direct electron microscopy. Vet. Rec. 1980, 106, 561. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, M.A.; Saif, Y.M.; Heggen-Peay, C.L.; Edens, F.W.; Havenstein, G.B. Induction of functional defects in macrophages by a poult enteritis and mortality syndrome-associated turkey astrovirus. Avian Dis. 2001, 45, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.L.; Saif, Y.M. Astrovirus: A cause of an enteric disease in turkey poults. Avian Dis. 1986, 30, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Koci, M.D.; Seal, B.S.; Schultz-Cherry, S. Molecular characterization of an avian astrovirus. J. Virol. 2000, 74, 6173–6177. [Google Scholar] [CrossRef] [PubMed]
- Pantin-Jackwood, M.J.; Day, J.M.; Jackwood, M.W.; Spackman, E. Enteric viruses detected by molecular methods in commercial chicken and turkey flocks in the United States between 2005 and 2006. Avian Dis. 2008, 52, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Cattoli, G.; De Battisti, C.; Toffan, A.; Salviato, A.; Lavazza, A.; Cerioli, M.; Capua, I. Co-circulation of distinct genetic lineages of astroviruses in turkeys and guinea fowl. Arch. Virol. 2007, 152, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Baxendale, W.; Mebatsion, T. The isolation and characterisation of astroviruses from chickens. Avian Pathol. 2004, 33, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Sajewicz-Krukowska, J.; Domanska-Blicharz, K. Nearly full-length genome sequence of a novel astrovirus isolated from chickens with ‘white chicks’ condition. Arch. Virol. 2016, 161, 2581–2587. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Imada, T.; Kawamura, H. Characterization of a picornavirus isolated from broiler chicks. Avian Dis. 1979, 23, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Imada, T.; Yamaguchi, S.; Mase, M.; Tsukamoto, K.; Kubo, M.; Morooka, A. Avian nephritis virus (ANV) as a new member of the family Astroviridae and construction of infectious ANV cDNA. J. Virol. 2000, 74, 8487–8493. [Google Scholar] [CrossRef] [PubMed]
- Takase, K.; Shinohara, K.; Tsuneyoshi, M.; Yamamoto, M.; Yamada, S. Isolation and characterisation of cytopathic avian enteroviruses from broiler chicks. Avian Pathol. 1989, 18, 631–642. [Google Scholar] [CrossRef] [PubMed]
- De Wit, J.J.; Dam, G.B.; de Laar, J.M.; Biermann, Y.; Verstegen, I.; Edens, F.; Schrier, C.C. Detection and characterization of a new astrovirus in chicken and turkeys with enteric and locomotion disorders. Avian Pathol. 2011, 40, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhu, A.L.; Yuan, C.L.; Yu, Y.; Zhu, C.X.; Lan, D.L.; Yang, Z.B.; Cui, L.; Hua, X.G. Detection of astrovirus infection in pigeons (Columbia livia) during an outbreak of diarrhoea. Avian Pathol. 2011, 40, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhu, A.L.; Yu, Y.; Yuan, C.L.; Zhu, C.X.; Yang, Z.B.; Cui, L.; Hua, X.G. Complete sequence and genetic characterization of pigeon avian nephritis virus, a member of the family Astroviridae. Arch. Virol. 2011, 156, 1559–1565. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Pan, M.; Wang, X.; Xu, Y.; Xie, X.; Knowles, N.J.; Yang, H.; Zhang, D. Complete sequence of a duck astrovirus associated with fatal hepatitis in ducklings. J. Gen. Virol. 2009, 90, 1104–1108. [Google Scholar] [CrossRef] [PubMed]
- Haider, S.A.; Calnek, B.W. In Vitro isolation, propagation, and characterization of duck hepatitis virus type III. Avian Dis. 1979, 23, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Liao, Q.; Liu, N.; Wang, X.; Wang, F.; Zhang, D. Genetic characterization of a novel astrovirus in Pekin ducks. Infect. Genet. Evol. 2015, 32, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Jiang, M.; Wang, M.; Wang, F.; Zhang, B.; Zhang, D. Isolation and detection of duck astrovirus CPH: Implications for epidemiology and pathogenicity. Avian Pathol. 2016, 45, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Honkavuori, K.S.; Briese, T.; Krauss, S.; Sanchez, M.D.; Jain, K.; Hutchison, S.K.; Webster, R.G.; Lipkin, W.I. Novel coronavirus and astrovirus in Delaware Bay shorebirds. PLoS ONE 2014, 9, e93395. [Google Scholar] [CrossRef] [PubMed]
- Mendenhall, I.H.; Yaung, K.N.; Joyner, P.H.; Keatts, L.; Borthwick, S.; Neves, E.S.; San, S.; Gilbert, M.; Smith, G.J. Detection of a novel astrovirus from a black-naped monarch (Hypothymis azurea) in Cambodia. Virol. J. 2015, 12, 182. [Google Scholar] [CrossRef] [PubMed]
- Kofstad, T.; Jonassen, C.M. Screening of feral and wood pigeons for viruses harbouring a conserved mobile viral element: Characterization of novel astroviruses and picornaviruses. PLoS ONE 2011, 6, e25964. [Google Scholar] [CrossRef] [PubMed]
- Mendenhall, I.H.; Smith, G.J.; Vijaykrishna, D. Ecological drivers of virus evolution: Astrovirus as a case study. J. Virol. 2015, 89, 6978–6981. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Yang, Y.; Wang, G.S.; Shao, X.Q.; Zhang, S.Q.; Wang, F.X.; Tan, B.; Tian, F.L.; Cheng, S.P.; Wen, Y.J. Detection and characterization of avastrovirus associated with diarrhea isolated from minks in China. Food Environ. Virol. 2014, 6, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Meliopoulos, V.A.; Kayali, G.; Burnham, A.; Oshansky, C.M.; Thomas, P.G.; Gray, G.C.; Beck, M.A.; Schultz-Cherry, S. Detection of antibodies against turkey astrovirus in humans. PLoS ONE 2014, 9, e96934. [Google Scholar] [CrossRef] [PubMed]
- Morsy El-Senousy, W.; Guix, S.; Abid, I.; Pinto, R.M.; Bosch, A. Removal of astrovirus from water and sewage treatment plants, evaluated by a competitive reverse transcription-PCR. Appl. Environ. Microbiol. 2007, 73, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Babkin, I.V.; Tikunov, A.Y.; Zhirakovskaia, E.V.; Netesov, S.V.; Tikunova, N.V. High evolutionary rate of human astrovirus. Infect. Genet. Evol. 2012, 12, 435–442. [Google Scholar] [CrossRef] [PubMed]
- De Grazia, S.; Martella, V.; Chironna, M.; Bonura, F.; Tummolo, F.; Calderaro, A.; Moschidou, P.; Giammanco, G.M.; Medici, M.C. Nationwide surveillance study of human astrovirus infections in an Italian paediatric population. Epidemiol. Infect. 2013, 141, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Jonassen, T.O.; Monceyron, C.; Lee, T.W.; Kurtz, J.B.; Grinde, B. Detection of all serotypes of human astrovirus by the polymerase chain reaction. J. Virol. Methods 1995, 52, 327–334. [Google Scholar] [CrossRef]
- Wolfaardt, M.; Kiulia, N.M.; Mwenda, J.M.; Taylor, M.B. Evidence of a recombinant wild-type human astrovirus strain from a Kenyan child with gastroenteritis. J. Clin. Microbiol. 2011, 49, 728–731. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Medici, M.C.; Terio, V.; Catella, C.; Bozzo, G.; Tummolo, F.; Calderaro, A.; Bonura, F.; Di Franco, M.; Banyai, K.; et al. Lineage diversification and recombination in type-4 human astroviruses. Infect. Genet. Evol. 2013, 20, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.F.; Sebeny, P.J.; Klena, J.D.; Pimentel, G.; Mansour, A.; Naguib, A.M.; Bruton, J.; Young, S.Y.; Holtz, L.R.; Wang, D. Novel astroviruses in children, Egypt. Emerg. Infect. Dis. 2011, 17, 2391–2393. [Google Scholar] [CrossRef] [PubMed]
- Matthijnssens, J.; Ciarlet, M.; McDonald, S.M.; Attoui, H.; Banyai, K.; Brister, J.R.; Buesa, J.; Esona, M.D.; Estes, M.K.; Gentsch, J.R.; et al. Uniformity of rotavirus strain nomenclature proposed by the Rotavirus Classification Working Group (RCWG). Arch. Virol. 2011, 156, 1397–1413. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donato, C.; Vijaykrishna, D. The Broad Host Range and Genetic Diversity of Mammalian and Avian Astroviruses. Viruses 2017, 9, 102. https://doi.org/10.3390/v9050102
Donato C, Vijaykrishna D. The Broad Host Range and Genetic Diversity of Mammalian and Avian Astroviruses. Viruses. 2017; 9(5):102. https://doi.org/10.3390/v9050102
Chicago/Turabian StyleDonato, Celeste, and Dhanasekaran Vijaykrishna. 2017. "The Broad Host Range and Genetic Diversity of Mammalian and Avian Astroviruses" Viruses 9, no. 5: 102. https://doi.org/10.3390/v9050102
APA StyleDonato, C., & Vijaykrishna, D. (2017). The Broad Host Range and Genetic Diversity of Mammalian and Avian Astroviruses. Viruses, 9(5), 102. https://doi.org/10.3390/v9050102





