A Decrease in Temperature and Humidity Precedes Human Rhinovirus Infections in a Cold Climate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Identification of Rhinovirus Infections
2.3. Exposure Assessment and Measures
2.4. Ethics Statement
2.5. Statistical Analyses
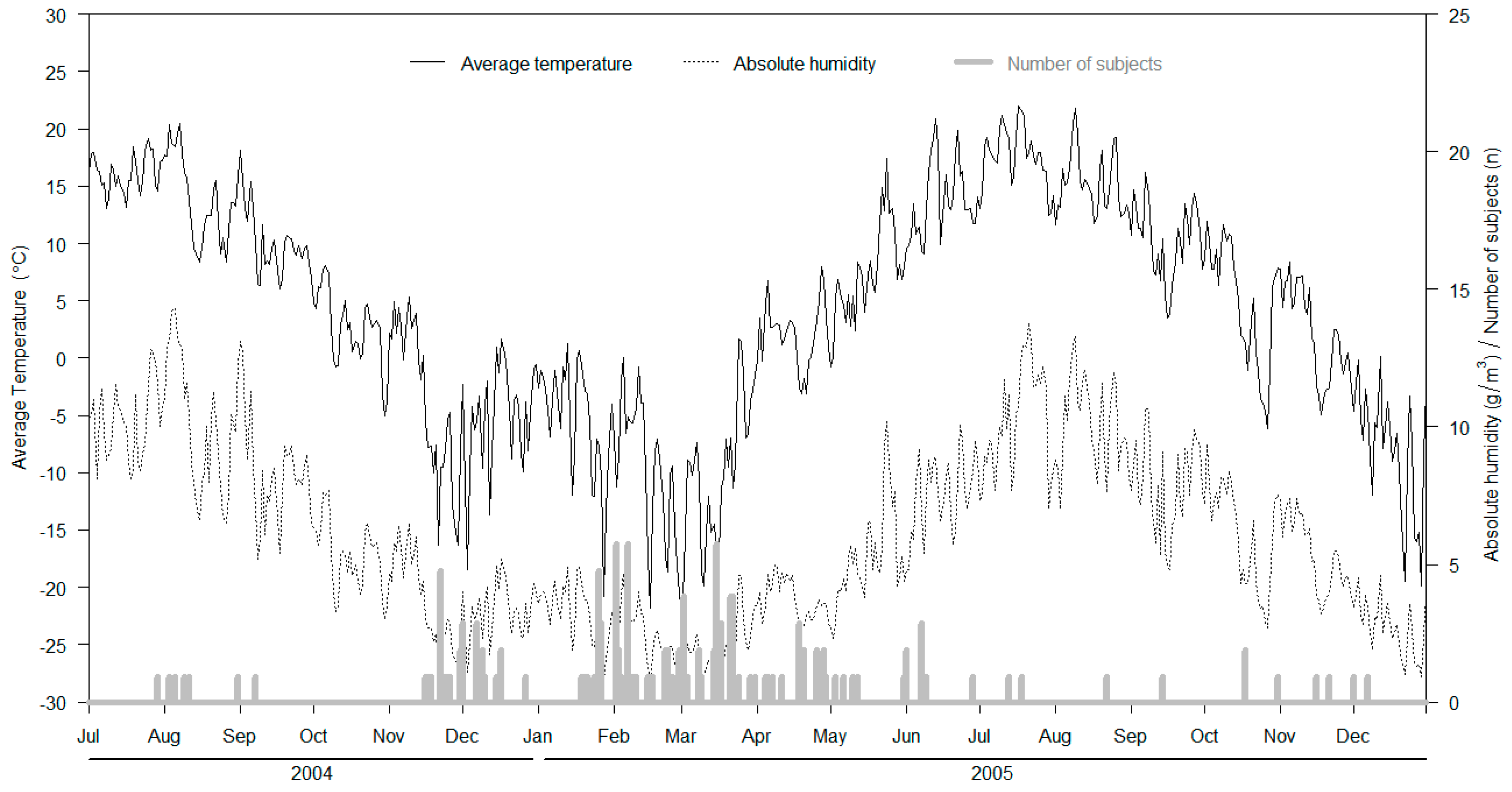
3. Results
4. Discussion
4.1. HRV Infections, Temperature and Humidity
4.2. Seasonality of HRV Infections
4.3. Strengths and Limitations of the Study
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jacobs, S.E.; Lamson, D.M.; St George, K.; Walsh, T.J. Human rhinoviruses. Clin. Microbiol. Rev. 2013, 26, 135–162. [Google Scholar] [CrossRef] [PubMed]
- Winther, B. Rhinovirus Infections in the upper airway. Proc. Am. Thorac. Soc. 2011, 8, 79–89. [Google Scholar] [CrossRef]
- Tanner, L.M.; Moffatt, S.; Milne, E.M.; Mills, S.D.H.; White, M. Socioeconomic and behavioural risk factors for adverse winter health and social outcomes in economically developed countries: A systematic review of quantitative observational studies. J. Epidemiol. Community Health 2013, 67, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Mourtzoukou, E.G.; Falagas, M.E. Exposure to cold and respiratory tract infections. Int. J. Tuberc. Lung Dis. 2007, 11, 938–943. [Google Scholar] [PubMed]
- Conlon, K.C.; Rajkovich, N.B.; White-Newsome, J.L.; Lasen, L.; O’Neill, M.S. Preventing cold-related morbidity and mortality in a changing climate. Maturitas 2011, 69, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Sloan, C.; Moore, M.L.; Hartert, T. Impact of pollution, climate, and sociodemographic factors on spatiotemporal dynamics of seasonal respiratory viruses. Clin. Transl. Sci. 2011, 4, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Shaman, J.; Kohn, M. Absolute humidity modulates influenza survival, transmission, and seasonality. Proc. Natl. Acad. Sci. USA 2009, 106, 3243–3248. [Google Scholar] [CrossRef] [PubMed]
- Du Prel, J.B.; Puppe, W.; Gröndahl, B.; Knuf, M.; Weigl, J.A.I.; Schaaff, F.; Schmitt, H. Are meteorological parameters associated with acute respiratory tract infections? Clin. Infect. Dis. 2009, 49, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Lowen, A.C.; Steel, J. Roles of humidity and temperature in shaping influenza seasonality. J. Virol. 2014, 88, 7692–7695. [Google Scholar] [CrossRef] [PubMed]
- Mäkinen, T.M.; Juvonen, R.; Jokelainen, J.; Harju, T.H.; Peitso, A.; Bloigu, A.; Silvennoinen-Kassinen, S.; Leinonen, M.; Hassi, J. Cold temperature and low humidity are associated with increased occurrence of respiratory tract infections. Respir. Med. 2009, 103, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Jaakkola, K.; Saukkoriipi, A.; Jokelainen, J.; Juvonen, R.; Kauppila, J.; Vainio, O.; Ziegler, T.; Rönkkö, E.; Jaakkola, J.J.; Ikäheimo, T.M.; et al. Decline in temperature and humidity increases the occurrence of influenza in cold climate. Environ. Health 2014, 13, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Liener, K.; Leiacker, R.; Lindemann, J.; Rettinger, G.; Keck, T. Nasal mucosal temperature after exposure to cold, dry air and hot, humid air. Acta Otolaryngol. 2003, 123, 851–856. [Google Scholar] [PubMed]
- Jaeger, J.J.; Deal, E.C., Jr.; Roberts, D.E., Jr.; Mcfadden, E.R., Jr. Cold air inhalation and esophageal temperature in exercising humans. Med. Sci. Sports Exerc. 1980, 12, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.A.; Naclerio, R.M.; Proud, D.; Toqias, A. Epithelial shedding is associated with reactions to cold, dry air. J. Allergy Clin. Immunol. 2006, 117, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Eccles, R. Acute cooling of the body surface and the common cold. Rhinology 2002, 40, 109–114. [Google Scholar] [PubMed]
- Johnson, C.; Eccles, R. Acute cooling of the feet and the onset of common cold symptoms. Fam. Pract. 2005, 22, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Ouédraogo, S.; Traoré, B.; Nene Bi, Z.A.; Yonli, F.T.; Kima, D.; Bonané, P.; Congo, L.; Traoré, R.O.; Yé, D.; Marguet, C.; et al. Viral etiology of respiratory tract infections in children at the pediatric hospital in Ouagadougou (Burkina Faso). PLoS ONE 2014, 31, e110435. [Google Scholar] [CrossRef] [PubMed]
- Gardinassi, L.G.; Marques Simas, P.V.; Salomão, J.B.; Durigon, E.L.; Trevisan, D.M.Z.; Cordeiro, J.A.; Lacerda, M.N.; Rahal, P.; de Souza, F.P. Seasonality of viral respiratory infections in southeast of Brazil: The influence of temperature and air humidity. Braz. J. Microbiol. 2012, 43, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Ge, W.Z.; Xu, F.; Zhao, Z.H.; kan, H.D. Association between diurnal temperature range and respiratory tract infections. Biomed. Environ. Sci. 2013, 26, 222–225. [Google Scholar] [PubMed]
- O’Shea, M.K.; Wilson, D. Respiratory infections in the military. J. R. Army Med. Corps 2013, 159, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Korzeniewski, K.; Nitsch-Osuch, A.; Chciałowski, A.; Korsak, J. Environmental factors, immune changes and respiratory diseases in troops during military activities. Respir. Physiol. Neurobiol. 2013, 187, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Shephard, R.J. Immune changes induced by exercise in an adverse environment. Can. J. Physiol. Pharmacol. 1998, 76, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Juvonen, R.; Bloigu, A.; Peitso, A.; Harju, T. Risk factors for acute respiratory tract illness in military conscripts. Respirology 2008, 13, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Holloway, B.; Dare, R.K.; Kuypers, J.; Yagi, S.; Williams, J.V.; Hall, C.B.; Erdman, D.D. Real-time reverse transcription-PCR assay for comprehensive detection of human rhinoviruses. J. Clin. Microbiol. 2008, 46, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Lessler, J.; Reich, N.G.; Brookmeyer, R.; Perl, T.M.; Nelson, K.E.; Cummings, D.A. Incubation periods of acute respiratory viral infections: A systematic review. Lancet Infect. Dis. 2009, 9, 291–300. [Google Scholar] [CrossRef]
- Gwaltney, J.M., Jr.; Hendley, J.O.; Patrie, J.T. Symptom severity patterns in experimental common colds and their usefulness in timing onset of illness in natural colds. Clin. Infect. Dis. 2003, 36, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Janes, H.; Sheppard, L.; Lumley, T. Reference selection strategies in case-crossover analyses of air pollution exposure data: Implications for bias. Epidemiology 2005, 16, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Savolainen-Kopra, C.; Blomqvist, S.; Kaijalainen, S.; Jounio, U.; Juvonen, R.; Peitso, A.; Saukkoriipi, A.; Vainio, O.; Hovi, T.; Roivainen, M. All known human rhinovirus species are present in sputum specimens of military recruits during respiratory infection. Viruses 2009, 1, 1178–1189. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, K.H. Sports in extreme conditions: The impact of exercise in cold temperatures on asthma and bronchial hyper-responsiveness in athletes. Br. J. Sports Med. 2012, 46, 796–799. [Google Scholar] [CrossRef] [PubMed]
- Douglas, R.G.; Lindgren, K.M.; Couch, R.B. Exposure to cold environment and Rhinovirus common cold-failure to demonstrate the effect. N. Engl. J. Med. 1968, 279, 742–747. [Google Scholar] [CrossRef]
- Graham, N.M.; Douglas, R.M.; Ryan, P. Stress and acute respiratory infection. Am. J. Epidemiol. 1986, 124, 389–401. [Google Scholar] [PubMed]
- Castellani, J.W.; M Brenner, I.K.; Rhind, S.G. Cold exposure: Human immune responses and intracellular cytokine expression. Med. Sci. Sports Exerc. 2002, 34, 2013–2020. [Google Scholar] [CrossRef] [PubMed]
- Yunus, A.S.; Jackson, T.P.; Crisafi, K.; Burimski, I.; Kilgore, N.R.; Zoumplis, D.; Allaway, G.P.; Wild, C.T.; Salzwedel, K. Elevated temperature triggers human respiratory syncytial virus F protein six-helix bundle formation. Virology 2010, 96, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, N.G.; Sanderson, G.; Hunter, J.; Johnston, S.L. Rhinoviruses replicate effectively at lower airway temperatures. J. Med. Virol. 1999, 58, 100–104. [Google Scholar] [CrossRef]
- Karim, Y.G.; Ijaz, M.K.; Sattar, S.A.; Johnson-Lussenburg, C.M. Effect of relative humidity on the airborne survival of rhinovirus-14. Can. J. Microbiol. 1985, 31, 1058–1061. [Google Scholar] [CrossRef] [PubMed]
- Sattar, S.A.; Karim, Y.G.; Springthorpe, V.S.; Johnson-Lussenburg, C.M. Survival of human rhinovirus type 14 dried onto nonporous inanimate surfaces: Effect of relative humidity and suspending medium. Can. J. Microbiol. 1987, 33, 802–806. [Google Scholar] [CrossRef] [PubMed]
- Tamerius, J.D.; Shaman, J.; Alonso, W.J.; Bloom-Feshbach, K.; Uejio, C.K.; Comrie, A.; Viboudet, C. Environmental predictors of seasonal influenza epidemics across temperate and tropical climates. PLoS Pathog. 2013, 9, e1003194. [Google Scholar] [CrossRef]
- Jackson, M.L. Confounding by season in ecologic studies of seasonal exposures and outcomes: Examples from estimates of mortality due to influenza. Ann. Epidemiol. 2009, 19, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Malanoski, A.P.; Lin, B.; Long, N.C.; Leski, T.A.; Blanay, K.M.; Hansen, C.J.; Brown, J.; Broderick, M.; Stenger, D.A.; et al. Broad spectrum respiratory pathogen analysis of throat swabs from military recruits reveals interference between rhinoviruses and adenoviruses. Microb. Ecol. 2010, 59, 623–634. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.E.; Bunning, M.L.; Lott, L.; Li, X.; Su, J.; Metzgar, D.; Brosch, L.; Panozzo, C.A.; Marconi, V.C.; Faix, D.J.; et al. Outbreak of severe respiratory disease associated with emergent human adenovirus serotype 14 at a US Air Force training facility in 2007. J. Infect. Dis. 2009, 199, 1419–1426. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Hazard | Reference 1 (Pre) | Reference 1 (Post) |
|---|---|---|---|
| Temperature (°C) | |||
| mean | −9.9 ± 4.9 | −9.7 ± 4.5 | −8.1 ± 4.8 |
| mean change | 1.5 ± 3.1 | −0.3 ± 4.6 | 1.9 ± 3.9 |
| maximum change | 4.1 ± 3.1 | 2.4 ± 4.4 | 6.0 ± 5.1 |
| Absolute humidity (AH) | |||
| mean | 2.2 ± 0.9 | 2.3 ± 0.9 | 2.4 ± 0.8 |
| mean change | 0.2 ± 0.6 | −0.1 ± 0.8 | 0.2 ± 0.6 |
| maximum change | 0.7 ± 0.6 | 0.4 ± 0.7 | 0.6 ± 0.7 |
| Parameter | OR (95% CI) 1 | Adjusted OR (95% CI) 2 |
|---|---|---|
| Absolute humidity (AH) | ||
| mean of three prior days | 0.94 (0.86–1.03) | 0.97 (0.80–1.16) |
| maximum change during three prior days | 1.09 (0.96–1.24) | 1.20 (1.03–1.40) |
| mean change during three prior days | 1.05 (0.91–1.21) | 1.13 (0.96–1.34) |
| Temperature (°C) | ||
| mean of three prior days | 0.96 (0.92–1.00) | 1.07 (1.00–1.15) |
| maximum change during three prior days | 1.04 (0.98–1.10) | 1.08 (1.01–1.17) |
| mean change during three prior days | 1.04 (0.97–1.11) | 1.08 (1.01–1.17) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ikäheimo, T.M.; Jaakkola, K.; Jokelainen, J.; Saukkoriipi, A.; Roivainen, M.; Juvonen, R.; Vainio, O.; Jaakkola, J.J.K. A Decrease in Temperature and Humidity Precedes Human Rhinovirus Infections in a Cold Climate. Viruses 2016, 8, 244. https://doi.org/10.3390/v8090244
Ikäheimo TM, Jaakkola K, Jokelainen J, Saukkoriipi A, Roivainen M, Juvonen R, Vainio O, Jaakkola JJK. A Decrease in Temperature and Humidity Precedes Human Rhinovirus Infections in a Cold Climate. Viruses. 2016; 8(9):244. https://doi.org/10.3390/v8090244
Chicago/Turabian StyleIkäheimo, Tiina M., Kari Jaakkola, Jari Jokelainen, Annika Saukkoriipi, Merja Roivainen, Raija Juvonen, Olli Vainio, and Jouni J.K. Jaakkola. 2016. "A Decrease in Temperature and Humidity Precedes Human Rhinovirus Infections in a Cold Climate" Viruses 8, no. 9: 244. https://doi.org/10.3390/v8090244
APA StyleIkäheimo, T. M., Jaakkola, K., Jokelainen, J., Saukkoriipi, A., Roivainen, M., Juvonen, R., Vainio, O., & Jaakkola, J. J. K. (2016). A Decrease in Temperature and Humidity Precedes Human Rhinovirus Infections in a Cold Climate. Viruses, 8(9), 244. https://doi.org/10.3390/v8090244






