Human Cytomegalovirus Nuclear Egress Proteins Ectopically Expressed in the Heterologous Environment of Plant Cells are Strictly Targeted to the Nuclear Envelope
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Plasmid Constructs
2.3. Transient Expression in Nicotiana benthamiana and Confocal Laser Scanning Microscopy
2.4. Transient Expression in Human Epithelial Hela Cells, Indirect Immunofluorescence Assay and Confocal Laser-Scanning Microscopy
2.5. Plant Protein PAGE and Western Blot Analysis
2.6. Yeast Two-Hybrid Screening
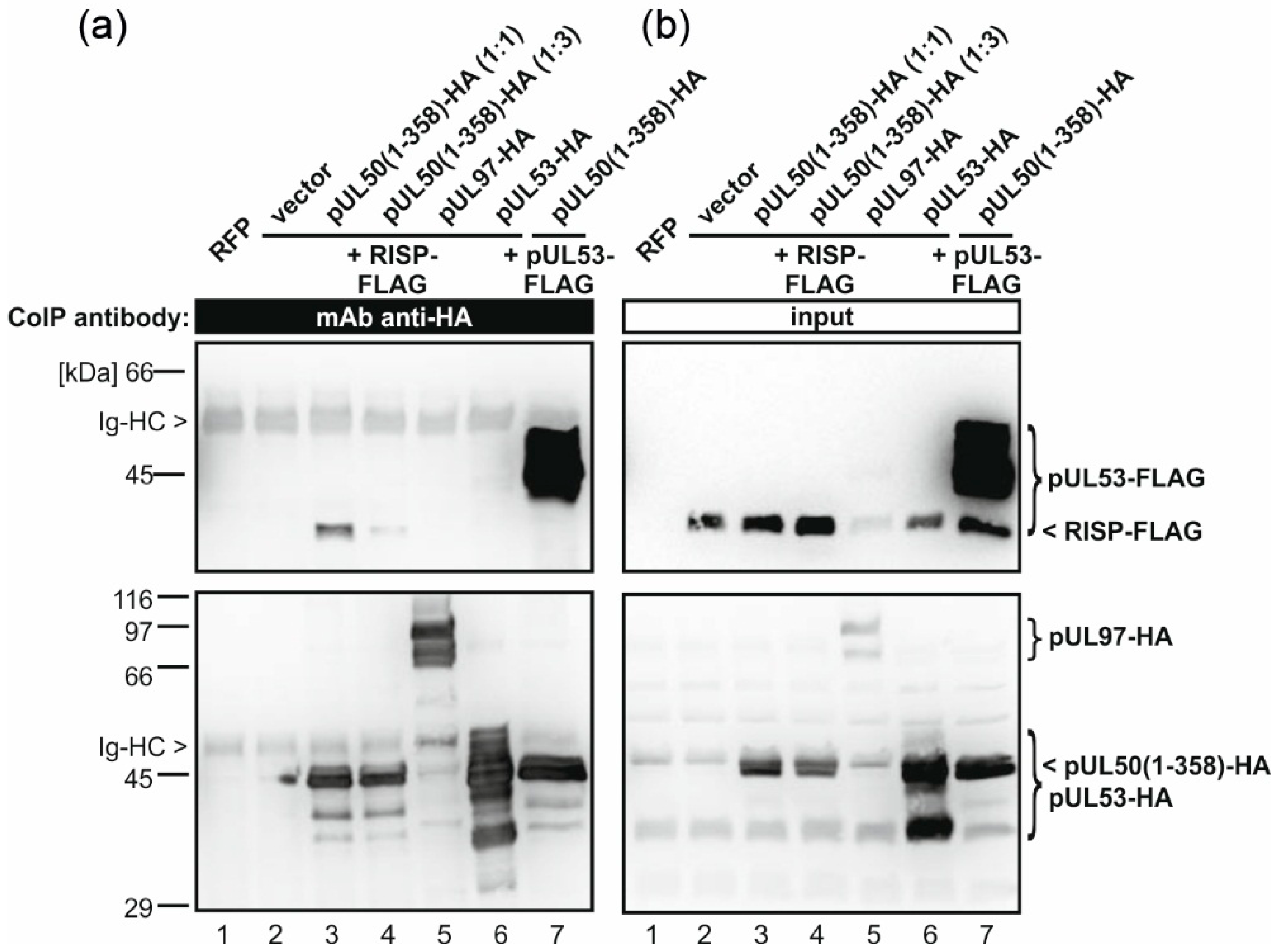
2.7. Coimmunoprecipitation Assay
3. Results
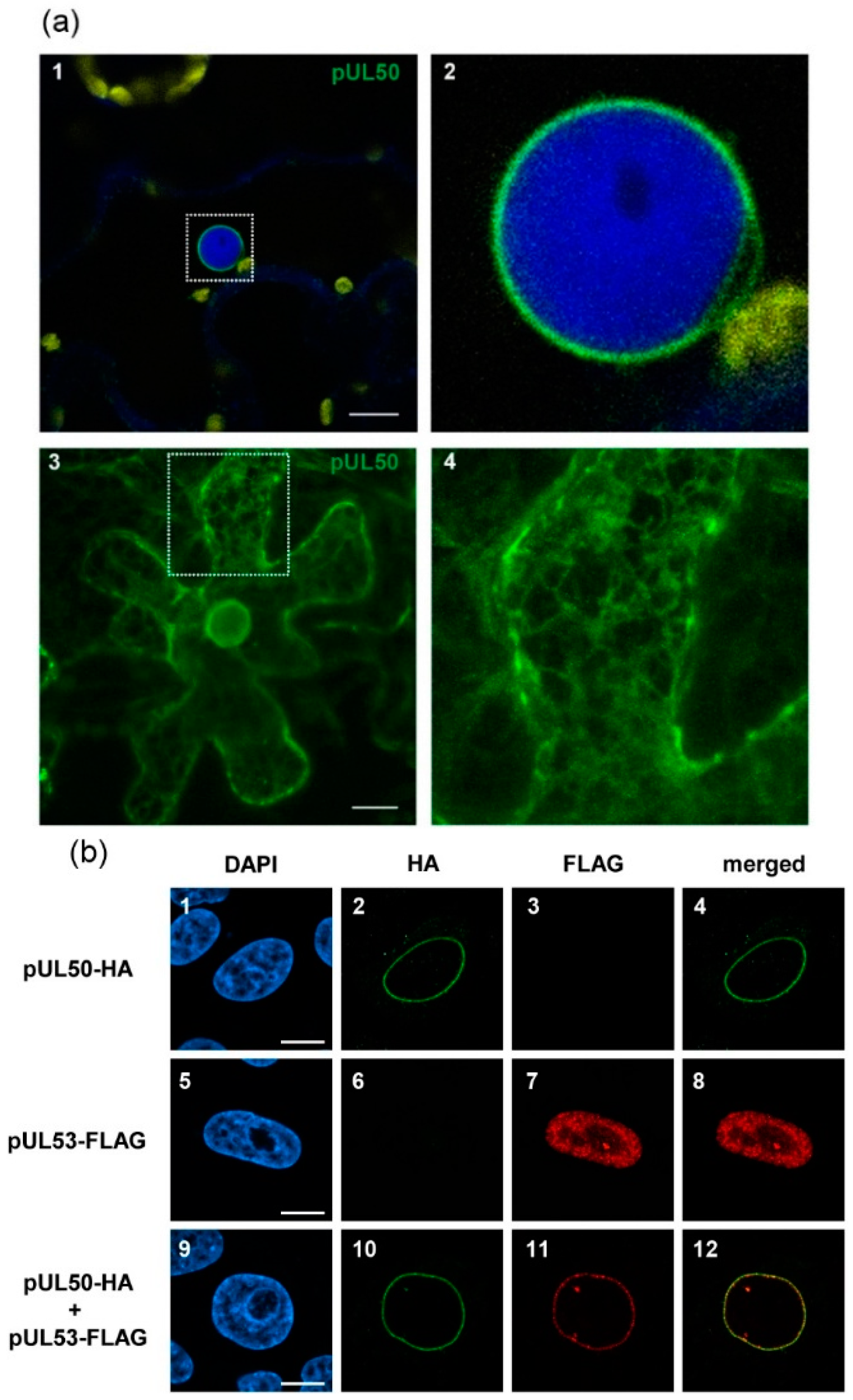
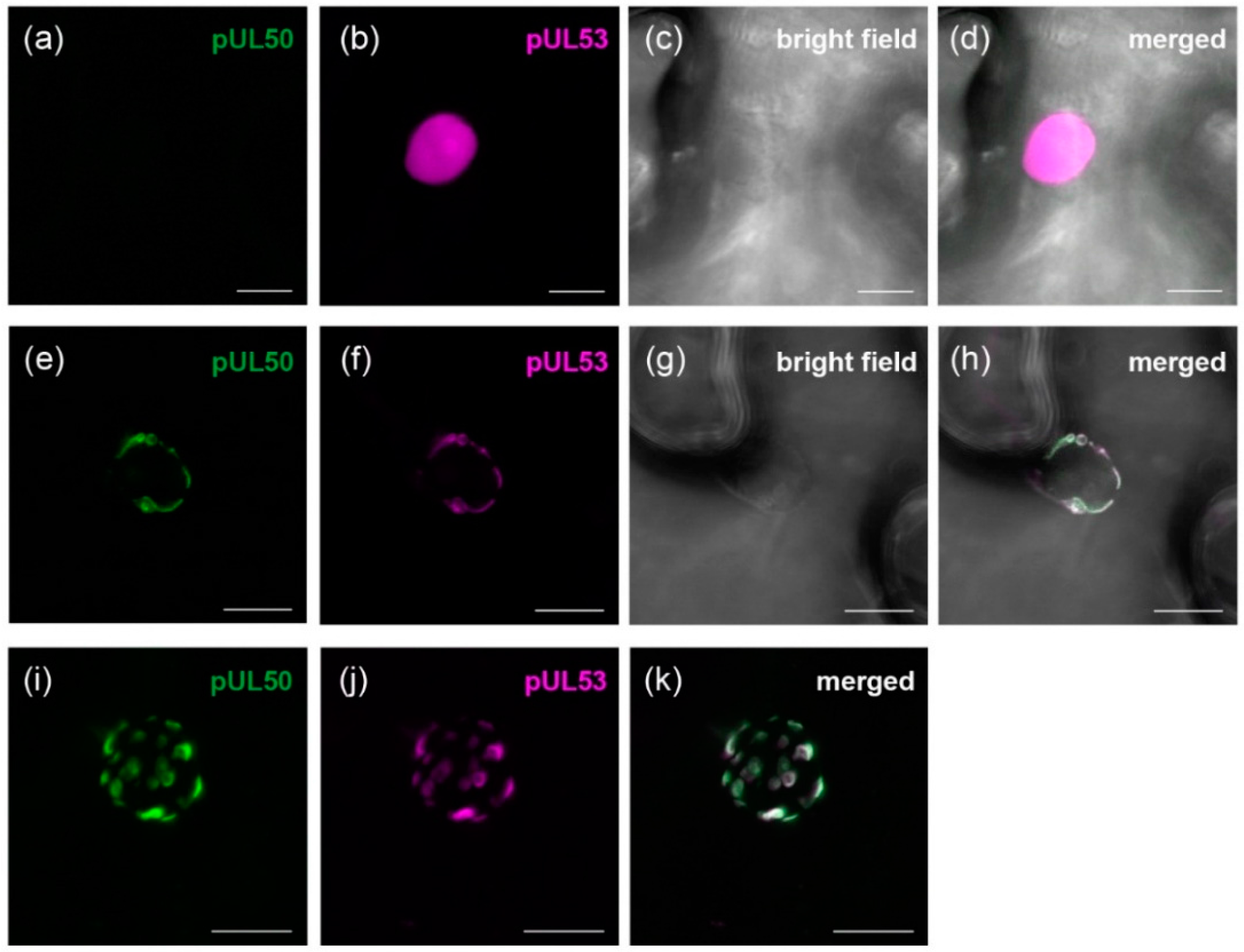
3.1. HCMV pUL50 Localizes to the Plant Nuclear Envelope
3.2. pUL50 is Able to Recruit HCMV pUL53 to the Nuclear Periphery in Nicotiana benthamiana
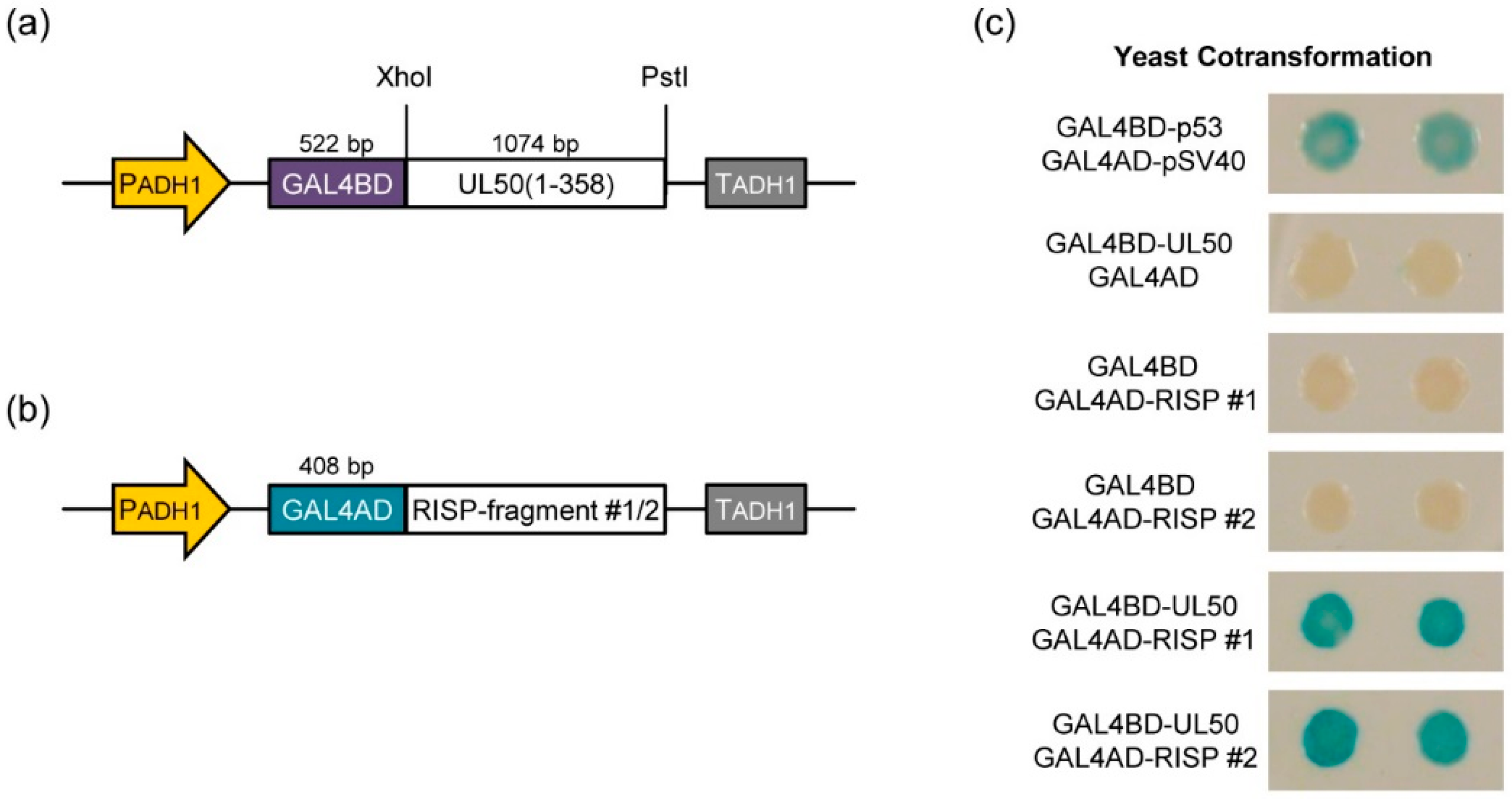
3.3. pUL50 Interacts with Arabidopsis thaliana re-Initiation Supporting Protein (RISP)
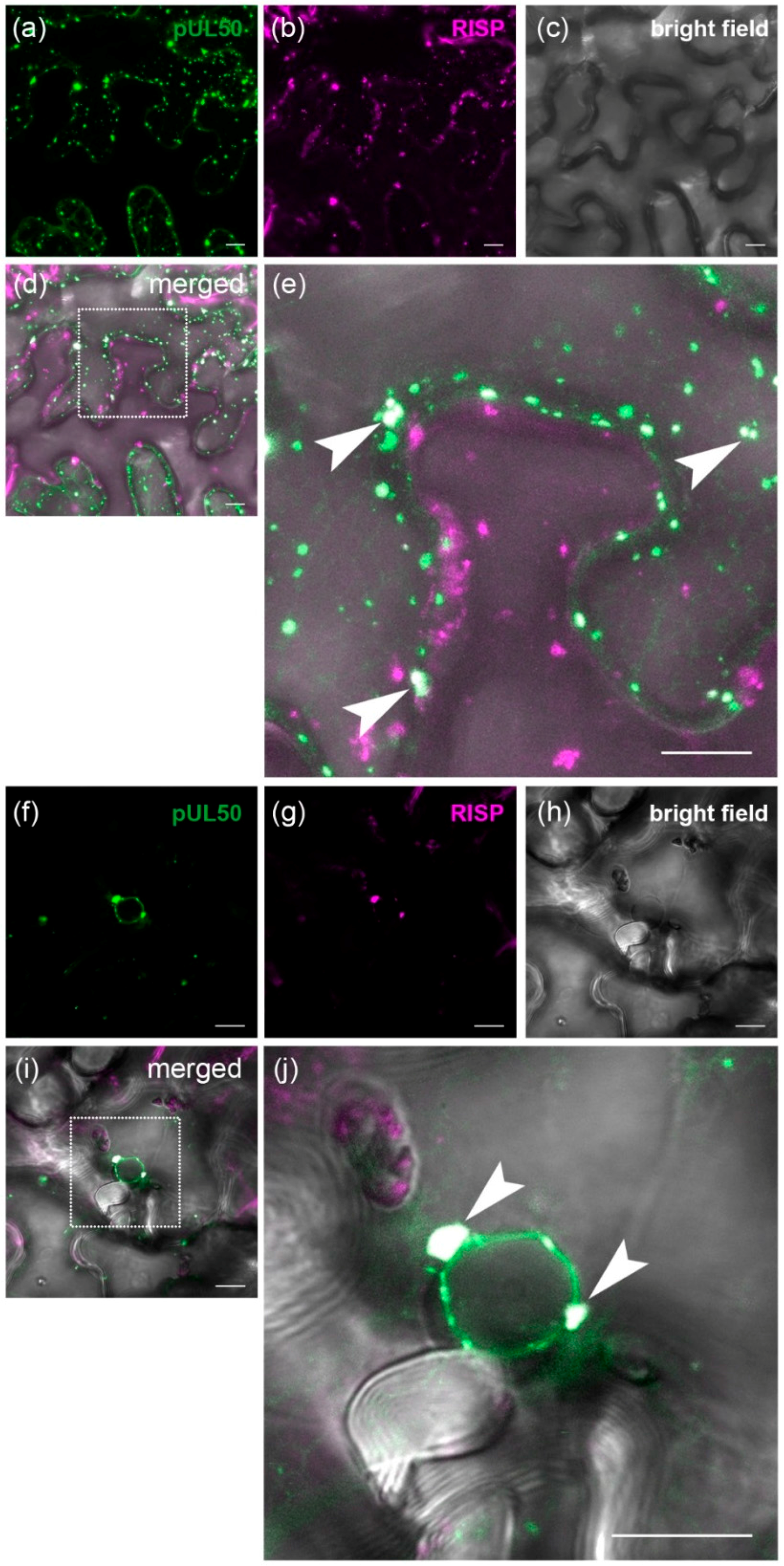
3.4. pUL50 Colocalizes with Arabidopsis thaliana Re-Initiation Supporting Protein (RISP)
4. Discussion
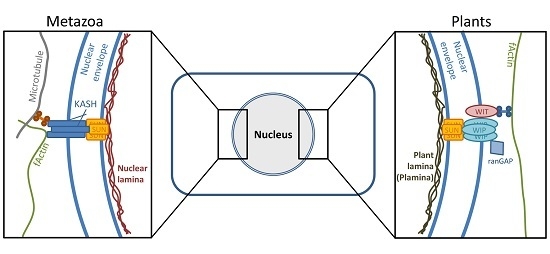
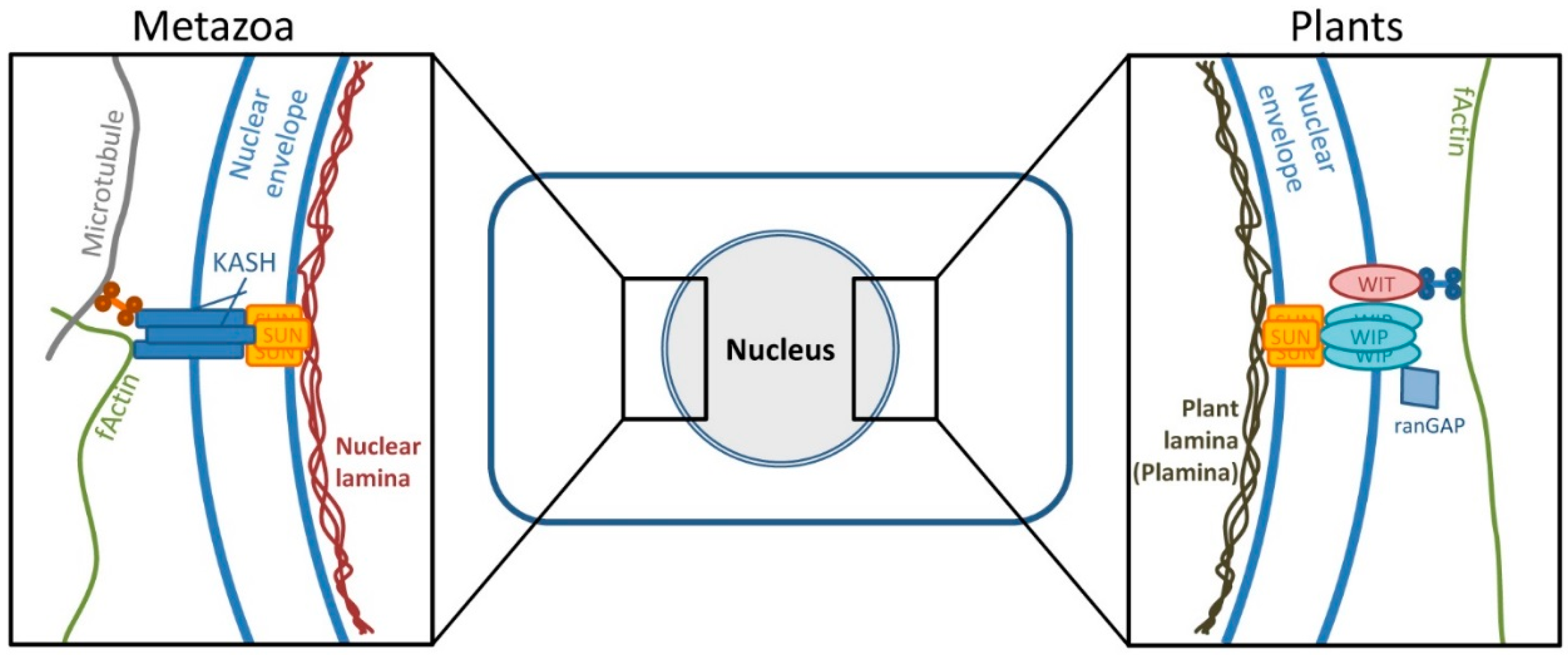
4.1. A Mechanism Similar to Herpesviral Nuclear Retention of pUL50 is Conserved in Plants
4.2. Interaction of pUL50 with the Arabidopsis thaliana protein RISP
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Terry, L.J.; Shows, E.B.; Wente, S.R. Crossing the nuclear envelope: Hierarchical regulation of nucleocytoplasmic transport. Science 2007, 318, 1412–1416. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Fukao, Y.; Iwamoto, M.; Haraguchi, T.; Hara-Nishimura, I. Identification and characterization of nuclear pore complex components in Arabidopsis thaliana. Plant Cell 2010, 22, 4084–4097. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.M.; Meier, I. The nuclear pore comes to the fore. Trends Plant Sci. 2008, 13, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Boruc, J.; Zhou, X.; Meier, I. Dynamics of the plant nuclear envelope and nuclear pore. Plant Physiol. 2012, 158, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Aebi, U.; Cohn, J.; Buhle, L.; Gerace, L. The nuclear lamina is a meshwork of intermediate-type filaments. Nature 1986, 323, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, D.W. On the occurrence of a fibrous lamina on the inner aspect of the nuclear envelope in certain cells of vertebrates. Am. J. Anat. 1966, 119, 129–145. [Google Scholar] [CrossRef] [PubMed]
- Aaronson, R.P.; Blobel, G. Isolation of nuclear pore complexes in association with a lamina. Proc. Natl. Acad. Sci. USA 1975, 72, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- Houben, F.; Ramaekers, F.C.; Snoeckx, L.H.; Broers, J.L. Role of nuclear lamina-cytoskeleton interactions in the maintenance of cellular strength. Biochim. Biophys. Acta 2007, 1773, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Pajerowski, J.D.; Dahl, K.N.; Zhong, F.L.; Sammak, P.J.; Discher, D.E. Physical plasticity of the nucleus in stem cell differentiation. Proc. Natl. Acad. Sci. USA 2007, 104, 15619–15624. [Google Scholar] [CrossRef] [PubMed]
- Favreau, C.; Dubosclard, E.; Ostlund, C.; Vigouroux, C.; Capeau, J.; Wehnert, M.; Higuet, D.; Worman, H.J.; Courvalin, J.C.; Buendia, B. Expression of lamin a mutated in the carboxyl-terminal tail generates an aberrant nuclear phenotype similar to that observed in cells from patients with dunnigan-type partial lipodystrophy and emery-dreifuss muscular dystrophy. Exp. Cell Res. 2003, 282, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Paddy, M.R.; Belmont, A.S.; Saumweber, H.; Agard, D.A.; Sedat, J.W. Interphase nuclear-envelope lamins form a discontinuous network that interacts with only a fraction of the chromatin in the nuclear periphery. Cell 1990, 62, 89–106. [Google Scholar] [CrossRef]
- Pickersgill, H.; Kalverda, B.; de Wit, E.; Talhout, W.; Fornerod, M.; van Steensel, B. Characterization of the Drosophila melanogaster genome at the nuclear lamina. Nat. Genet. 2006, 38, 1005–1014. [Google Scholar] [CrossRef] [PubMed]
- Andres, V.; Gonzalez, J.M. Role of A-type lamins in signaling, transcription, and chromatin organization. J. Cell Biol. 2009, 187, 945–957. [Google Scholar] [CrossRef] [PubMed]
- Dechat, T.; Adam, S.A.; Taimen, P.; Shimi, T.; Goldman, R.D. Nuclear lamins. Cold Spring Harb. Perspect. Biol. 2010, 2. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, T.A.; Misteli, T. The lamin protein family. Genome Biol. 2011, 12. [Google Scholar] [CrossRef] [PubMed]
- Fiserova, J.; Kiseleva, E.; Goldberg, M.W. Nuclear envelope and nuclear pore complex structure and organization in tobacco BY-2 cells. Plant J. 2009, 59, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Goto, C.; Tamura, K.; Fukao, Y.; Shimada, T.; Hara-Nishimura, I. The novel nuclear envelope protein KAKU4 modulates nuclear morphology in arabidopsis. Plant Cell 2014, 26, 2143–2155. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, T.A.; Stacey, N.J.; Sugimoto-Shirasu, K.; Richards, E.J. Little nuclei genes affecting nuclear morphology in Arabidopsis thaliana. Plant Cell 2007, 19, 2793–2803. [Google Scholar] [CrossRef] [PubMed]
- Ciska, M.; Masuda, K.; Moreno Diaz de la Espina, S. Lamin-like analogues in plants: The characterization of NMCP1 in Allium cepa. J. Exp. Bot. 2013, 64, 1553–1564. [Google Scholar] [CrossRef] [PubMed]
- Masuda, K.; Xu, Z.J.; Takahashi, S.; Ito, A.; Ono, M.; Nomura, K.; Inoue, M. Peripheral framework of carrot cell nucleus contains a novel protein predicted to exhibit a long α-helical domain. Exp. Cell Res. 1997, 232, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Fujino, K.; Ogawa, K.; Masuda, K. Localization of daucus carota NMCP1 to the nuclear periphery: The role of the N-terminal region and an NLS-linked sequence motif, RYNLRR, in the tail domain. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Graumann, K.; Meier, I. The plant nuclear envelope as a multifunctional platform linced by sun and kash. J. Exp. Bot. 2015, 66, 1649–1659. [Google Scholar] [CrossRef] [PubMed]
- Hodzic, D.M.; Yeater, D.B.; Bengtsson, L.; Otto, H.; Stahl, P.D. Sun2 is a novel mammalian inner nuclear membrane protein. J. Biol. Chem. 2004, 279, 25805–25812. [Google Scholar] [CrossRef] [PubMed]
- Padmakumar, V.C.; Libotte, T.; Lu, W.; Zaim, H.; Abraham, S.; Noegel, A.A.; Gotzmann, J.; Foisner, R.; Karakesisoglou, I. The inner nuclear membrane protein Sun1 mediates the anchorage of Nesprin-2 to the nuclear envelope. J. Cell Sci. 2005, 118, 3419–3430. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.K.; Starr, D.; Cohen, M.; Liu, J.; Han, M.; Wilson, K.L.; Gruenbaum, Y. Lamin-dependent localization of unc-84, a protein required for nuclear migration in Caenorhabditis elegans. Mol. Biol. Cell 2002, 13, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Haque, F.; Lloyd, D.J.; Smallwood, D.T.; Dent, C.L.; Shanahan, C.M.; Fry, A.M.; Trembath, R.C.; Shackleton, S. Sun1 interacts with nuclear lamin a and cytoplasmic nesprins to provide a physical connection between the nuclear lamina and the cytoskeleton. Mol. Cell. Biol. 2006, 26, 3738–3751. [Google Scholar] [CrossRef] [PubMed]
- Sosa, B.A.; Rothballer, A.; Kutay, U.; Schwartz, T.U. Linc complexes form by binding of three kash peptides to domain interfaces of trimeric sun proteins. Cell 2012, 149, 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Graumann, K.; Runions, J.; Evans, D.E. Characterization of SUN-domain proteins at the higher plant nuclear envelope. Plant J. 2010, 61, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Graumann, K. Evidence for LINC1-SUN associations at the plant nuclear periphery. PLoS ONE 2014, 9, e93406. [Google Scholar] [CrossRef] [PubMed]
- Crisp, M.; Liu, Q.; Roux, K.; Rattner, J.B.; Shanahan, C.; Burke, B.; Stahl, P.D.; Hodzic, D. Coupling of the nucleus and cytoplasm: Role of the LINC complex. J. Cell Biol. 2006, 172, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Starr, D.A.; Han, M. Role of ANC-1 in tethering nuclei to the actin cytoskeleton. Science 2002, 298, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Graumann, K.; Evans, D.E.; Meier, I. Novel plant SUN-KASH bridges are involved in RanGAP anchoring and nuclear shape determination. J. Cell Biol. 2012, 196, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Iwabuchi, K.; Fukao, Y.; Kondo, M.; Okamoto, K.; Ueda, H.; Nishimura, M.; Hara-Nishimura, I. Myosin XI-i links the nuclear membrane to the cytoskeleton to control nuclear movement and shape in arabidopsis. Curr. Biol. 2013, 23, 1776–1781. [Google Scholar] [CrossRef] [PubMed]
- Darlington, R.W.; Moss, L.H., 3rd. Herpesvirus envelopment. J. Virol. 1968, 2, 48–55. [Google Scholar] [PubMed]
- Stackpole, C.W. Herpes-type virus of the frog renal adenocarcinoma: I. Virus development in tumor transplants maintained at low temperature. J. Virol. 1969, 4, 75–93. [Google Scholar] [PubMed]
- Chen, D.H.; Jiang, H.; Lee, M.; Liu, F.; Zhou, Z.H. Three-dimensional visualization of tegument/capsid interactions in the intact human cytomegalovirus. Virology 1999, 260, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Pante, N.; Kann, M. Nuclear pore complex is able to transport macromolecules with diameters of about 39 nm. Mol. Biol. Cell 2002, 13, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Milbradt, J.; Webel, R.; Auerochs, S.; Sticht, H.; Marschall, M. Novel mode of phosphorylation-triggered reorganization of the nuclear lamina during nuclear egress of human cytomegalovirus. J. Biol. Chem. 2010, 285, 13979–13989. [Google Scholar] [CrossRef] [PubMed]
- Walzer, S.A.; Egerer-Sieber, C.; Sticht, H.; Sevvana, M.; Hohl, K.; Milbradt, J.; Muller, Y.A.; Marschall, M. Crystal structure of the human cytomegalovirus pUL50-pUL53 core nuclear egress complex provides insight into a unique assembly scaffold for virus-host protein interactions. J. Biol. Chem. 2015, 290, 27452–27458. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Kamil, J.P.; Coen, D.M. Preparation of the human cytomegalovirus nuclear egress complex and associated proteins. Methods Enzymol. 2016, 569, 517–526. [Google Scholar] [PubMed]
- Lye, M.F.; Sharma, M.; El Omari, K.; Filman, D.J.; Schuermann, J.P.; Hogle, J.M.; Coen, D.M. Unexpected features and mechanism of heterodimer formation of a herpesvirus nuclear egress complex. EMBO J. 2015, 34, 2937–2952. [Google Scholar] [CrossRef] [PubMed]
- Milbradt, J.; Auerochs, S.; Sevvana, M.; Muller, Y.A.; Sticht, H.; Marschall, M. Specific residues of a conserved domain in the N terminus of the human cytomegalovirus pUL50 protein determine its intranuclear interaction with pUL53. J. Biol. Chem. 2012, 287, 24004–24016. [Google Scholar] [CrossRef] [PubMed]
- Milbradt, J.; Auerochs, S.; Marschall, M. Cytomegaloviral proteins pUL50 and pUL53 are associated with the nuclear lamina and interact with cellular protein kinase C. J. Gen. Virol. 2007, 88, 2642–2650. [Google Scholar] [CrossRef] [PubMed]
- Lötzerich, M.; Ruzsics, Z.; Koszinowski, U.H. Functional domains of murine cytomegalovirus nuclear egress protein M53/P38. J. Virol. 2006, 80, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Camozzi, D.; Pignatelli, S.; Valvo, C.; Lattanzi, G.; Capanni, C.; Dal Monte, P.; Landini, M.P. Remodelling of the nuclear lamina during human cytomegalovirus infection: Role of the viral proteins pUL50 and pUL53. J. Gen. Virol. 2008, 89, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Milbradt, J.; Kraut, A.; Hutterer, C.; Sonntag, E.; Schmeiser, C.; Ferro, M.; Wagner, S.; Lenac, T.; Claus, C.; Pinkert, S.; et al. Proteomic analysis of the multimeric nuclear egress complex of human cytomegalovirus. Mol. Cell. Proteomics 2014, 13, 2132–2146. [Google Scholar] [CrossRef] [PubMed]
- Lemnitzer, F.; Raschbichler, V.; Kolodziejczak, D.; Israel, L.; Imhof, A.; Bailer, S.M.; Koszinowski, U.; Ruzsics, Z. Mouse cytomegalovirus egress protein pM50 interacts with cellular endophilin-A2. Cell. Microbiol. 2013, 15, 335–351. [Google Scholar] [CrossRef] [PubMed]
- Milbradt, J.; Auerochs, S.; Sticht, H.; Marschall, M. Cytomegaloviral proteins that associate with the nuclear lamina: Components of a postulated nuclear egress complex. J. Gen. Virol. 2009, 90, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Marschall, M.; Marzi, A.; aus dem Siepen, P.; Jochmann, R.; Kalmer, M.; Auerochs, S.; Lischka, P.; Leis, M.; Stamminger, T. Cellular p32 recruits cytomegalovirus kinase pUL97 to redistribute the nuclear lamina. J. Biol. Chem. 2005, 280, 33357–33367. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Inze, D.; Depicker, A. GATEWAY™ vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci. 2002, 7, 193–195. [Google Scholar] [CrossRef]
- Nakamura, S.; Mano, S.; Tanaka, Y.; Ohnishi, M.; Nakamori, C.; Araki, M.; Niwa, T.; Nishimura, M.; Kaminaka, H.; Nakagawa, T.; et al. GATEWAY binary vectors with the bialaphos resistance gene, bar, as a selection marker for plant transformation. Biosci. Biotechnol. Biochem. 2010, 74, 1315–1319. [Google Scholar] [CrossRef] [PubMed]
- Bartetzko, V.; Sonnewald, S.; Vogel, F.; Hartner, K.; Stadler, R.; Hammes, U.Z.; Bornke, F. The xanthomonas campestris pv. Vesicatoria type III effector protein XopJ inhibits protein secretion: Evidence for interference with cell wall-associated defense responses. Mol. Plant Microbe Interact. 2009, 22, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Marschall, M.; Stein-Gerlach, M.; Freitag, M.; Kupfer, R.; van Den Bogaard, M.; Stamminger, T. Inhibitors of human cytomegalovirus replication drastically reduce the activity of the viral protein kinase pUL97. J. Gen. Virol. 2001, 82, 1439–1450. [Google Scholar] [CrossRef] [PubMed]
- Voinnet, O.; Rivas, S.; Mestre, P.; Baulcombe, D. An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein of tomato bushy stunt virus. Plant J. 2003, 33, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Fiji Is Just Image J. Available online: http://fiji.sc/Fiji (accessed on 3 March 2016).
- Fields, S.; Song, O. A novel genetic system to detect protein-protein interactions. Nature 1989, 340, 245–246. [Google Scholar] [CrossRef] [PubMed]
- Harper, J.W.; Adami, G.R.; Wei, N.; Keyomarsi, K.; Elledge, S.J. The p21 Cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 1993, 75, 805–816. [Google Scholar] [CrossRef]
- Schiestl, R.H.; Gietz, R.D. High-efficiency transformation of intact yeast-cells using single stranded nucleic-acids as a carrier. Curr. Genet. 1989, 16, 339–346. [Google Scholar] [CrossRef] [PubMed]
- The Arabidopsis Information Resource. Available online: https://www.arabidopsis.org/servlets/TairObject?accession=Stock:4775513 (accessed on 3 March 2016).
- Bartel, P.L.; Fields, S. Analyzing protein-protein interactions using two-hybrid system. Methods Enzymol. 1994, 254, 241–263. [Google Scholar]
- Schregel, V.; Auerochs, S.; Jochmann, R.; Maurer, K.; Stamminger, T.; Marschall, M. Mapping of a self-interaction domain of the cytomegalovirus protein kinase pUL97. J. Gen. Virol. 2007, 88, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Schmeiser, C.; Borst, E.; Sticht, H.; Marschall, M.; Milbradt, J. The cytomegalovirus egress proteins pUL50 and pUL53 are translocated to the nuclear envelope through two distinct modes of nuclear import. J. Gen. Virol. 2013, 94, 2056–2069. [Google Scholar] [CrossRef] [PubMed]
- Thiebeauld, O.; Schepetilnikov, M.; Park, H.S.; Geldreich, A.; Kobayashi, K.; Keller, M.; Hohn, T.; Ryabova, L.A. A new plant protein interacts with eIF3 and 60S to enhance virus-activated translation re-initiation. EMBO J. 2009, 28, 3171–3184. [Google Scholar] [CrossRef] [PubMed]
- Zinchuk, V.; Wu, Y.; Grossenbacher-Zinchuk, O. Bridging the gap between qualitative and quantitative colocalization results in fluorescence microscopy studies. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Parry, D.A.D.; Conway, J.F.; Steinert, P.M. Structural studies on lamin – similarities and differences between lamin and intermediate-filament proteins. Biochem J 1986, 238, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Heitlinger, E.; Peter, M.; Haner, M.; Lustig, A.; Aebi, U.; Nigg, E.A. Expression of lamin b2 in escherichia coli: Characterization of its structure, assembly, and molecular interactions. J Cell Biol 1991, 113, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Antonin, W.; Ungricht, R.; Kutay, U. Traversing the npc along the pore membrane: Targeting of membrane proteins to the INM. Nucleus 2011, 2, 87–91. [Google Scholar] [CrossRef] [PubMed]
| Primer Name | Sequence (5’ to 3’) |
|---|---|
| Primers for RFP Vector Construction | |
| RFP fw SalI | GTCGACGGATCTGGTGTGTCTAAGGGCGAAGAGCTG |
| RFP rev PstI | CTGCAGCTATCAATTAAGTTTGTGCCCCAGTTTG |
| Primers for Localization Studies | |
| in Plants | |
| pUL50 fw | CACCATGGAGATGAACAAGGTTCTCCATC |
| pUL50 rev | TCAGTCGCGGTGTGCGGAG |
| pUL53 fw | CACCATGTCTAGCGTGAGCGGC |
| pUL53 rev | AGGCGCACGAATGCTGTTGAG |
| RISP fw | GGATCCAACAATGTCGAGTAATTGGGGAAGTAGCTCG |
| RISP rev | GTCGACTACAGCAGGAAGAGGAACTAAGCAAGTTG |
| in Human Cells | |
| 5-RISP_EcoRV-Acc65I | TCAGGTACCGATATCATGAACGACCTGAGTGAACATGTAC |
| 3-RISP-FLAG_NotI-ApaI | TCAGGGCCCGCGGCCGCTTACTTGTCGTCATCGTCTTTGTAGTCTACAGCAGGAAGAGGAACTAAGC |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lamm, C.E.; Link, K.; Wagner, S.; Milbradt, J.; Marschall, M.; Sonnewald, U. Human Cytomegalovirus Nuclear Egress Proteins Ectopically Expressed in the Heterologous Environment of Plant Cells are Strictly Targeted to the Nuclear Envelope. Viruses 2016, 8, 73. https://doi.org/10.3390/v8030073
Lamm CE, Link K, Wagner S, Milbradt J, Marschall M, Sonnewald U. Human Cytomegalovirus Nuclear Egress Proteins Ectopically Expressed in the Heterologous Environment of Plant Cells are Strictly Targeted to the Nuclear Envelope. Viruses. 2016; 8(3):73. https://doi.org/10.3390/v8030073
Chicago/Turabian StyleLamm, Christian E., Katrin Link, Sabrina Wagner, Jens Milbradt, Manfred Marschall, and Uwe Sonnewald. 2016. "Human Cytomegalovirus Nuclear Egress Proteins Ectopically Expressed in the Heterologous Environment of Plant Cells are Strictly Targeted to the Nuclear Envelope" Viruses 8, no. 3: 73. https://doi.org/10.3390/v8030073
APA StyleLamm, C. E., Link, K., Wagner, S., Milbradt, J., Marschall, M., & Sonnewald, U. (2016). Human Cytomegalovirus Nuclear Egress Proteins Ectopically Expressed in the Heterologous Environment of Plant Cells are Strictly Targeted to the Nuclear Envelope. Viruses, 8(3), 73. https://doi.org/10.3390/v8030073