The Role of Merkel Cell Polyomavirus and Other Human Polyomaviruses in Emerging Hallmarks of Cancer
Abstract
:1. Introduction
2. Human Polyomaviruses and Cancer
| Number of samples | Method | Number of positive samples | Comments | Reference | |
|---|---|---|---|---|---|
| Melanoma (st-age IV) | 18 | PCR and IHC (HPyV6 VP1moAb) | HPyV6: 18 HPyV7: 17 TSPyV: 4 HPyV9: 1 HPyV10: 12 | Low viral DNA loads, but higher for HPyV6 | [54] |
| Mucosal melanoma | 37 | PCR | KIPyV: 0 WUPyV: 0 HPyV6:0 HPyV7:0 TSPyV: 0 HPyV9:0 MWPyV: 0 | [55] | |
| Squamous cell carcinoma | 63 | PCR | HPyV6: 2 HPyV7: 1 | Low viral DNA loads | [56] |
| Basal cell carcinoma | 50 | PCR | HPyV6: 1 HPyV7: 2 | Low viral DNA loads | [56] |
| Melanoma | 47 | PCR | HPyV6: 2 HPyV7: 2 | Low viral DNA loads | [56] |
| Basal cell carcinoma | 41 | PCR | HPyV6:3 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| Squamous cell carcinoma | 52 | PCR | HPyV6:2 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| SCC in situ | 8 | PCR | HPyV6:1 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| Keratoacanthoma | 42 | PCR | HPyV6:2 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| Microcystic adnexal carcinoma | 5 | PCR | HPyV6:0 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| Atypical fibroxanthoma | 14 | PCR | HPyV6:0 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| Actinic keratosis | 31 | PCR | HPyV6:1 HPyV7:0 TSPyV: 0 HPyV9:0 | [57] | |
| Breast cancer | 54 | PCR | HPyV6: 1 HPyV7:1 | [58] | |
| Merkel cell carcinoma | deep sequencing | HPyV6: 1 HPyV7:1 HPyV9:1 | [59] | ||
| Extracutaneous melanoma | 38 | PCR | KIPyV: 0 WUPyV: 0 | [60] | |
| SCC+AK | 142 | deep sequencing | HPyV6: 1 | [61] | |
| Chronic lymphocytic leukaemia | 27 | PCR | HPyV9: 0 | [62] | |
| Primary cutaneous B-cell lymphomas (CBCLs) or cutaneous T-cell lymphomas (CTCLs) | 130 | PCR | HPyV6: 6 HPyV7: 1 TSPyV: 0 | [63] | |
| MCC | 28 | PCR | HPyV6: 0 HPyV7:0 | [64] | |
| Pilomatricomas (benign skin tumor associated with hair follicles | ? | ? | TSPyV: 0 | [65] | |
| Lung cancer | 20 | PCR | KIPyV:9 | [66] | |
| CNS tumors | 25 | PCR | KIPyV: 0 WUPyV: 0 | [67] | |
| Neuroblastoma | 31 | PCR | KIPyV: 0 WUPyV: 0 | [67] | |
| Acute lymphoblastic leukaemia | 50 | PCR | KIPyV: 0 WUPyV: 0 | [68] | |
| Lung cancer | 30 32 | PCR PCR | KIPyV: 0 WUPyV: 0 KIPyV: 0 WUPyV: 0 | [69] [70] | |
| Neuroendocrine tumors | 50 | PCR | KIPyV: 0 WUPyV: 0 HPyV6:0 HPyV7:0 TSPyV: 0 | [71] | |
| Skin lesions from CTCL patients | 39 | PCR | HPyV6:11 HPyV7:5 TSPyV: 0 HPyV9:0 | [72] | |
| Blood from CTCL patients | 39 | PCR | HPyV6:0 HPyV7:0 TSPyV: 0 HPyV9:0 | [72] | |
| Glioblastoma multiforme | 39 | PCR | HPyV6:0 HPyV7:0 HPyV9:0 | [73] | |
Thymic epithelial tumors Thymic hyperplasias Foetal thymus tissue | 37 20 20 | PCR, FISH, IHC | PCR FISH IHC HPyV7: 20 23 17 HPyV6: 0 HPyV7: 8 14 6 HPyV7: 0 | [74] |
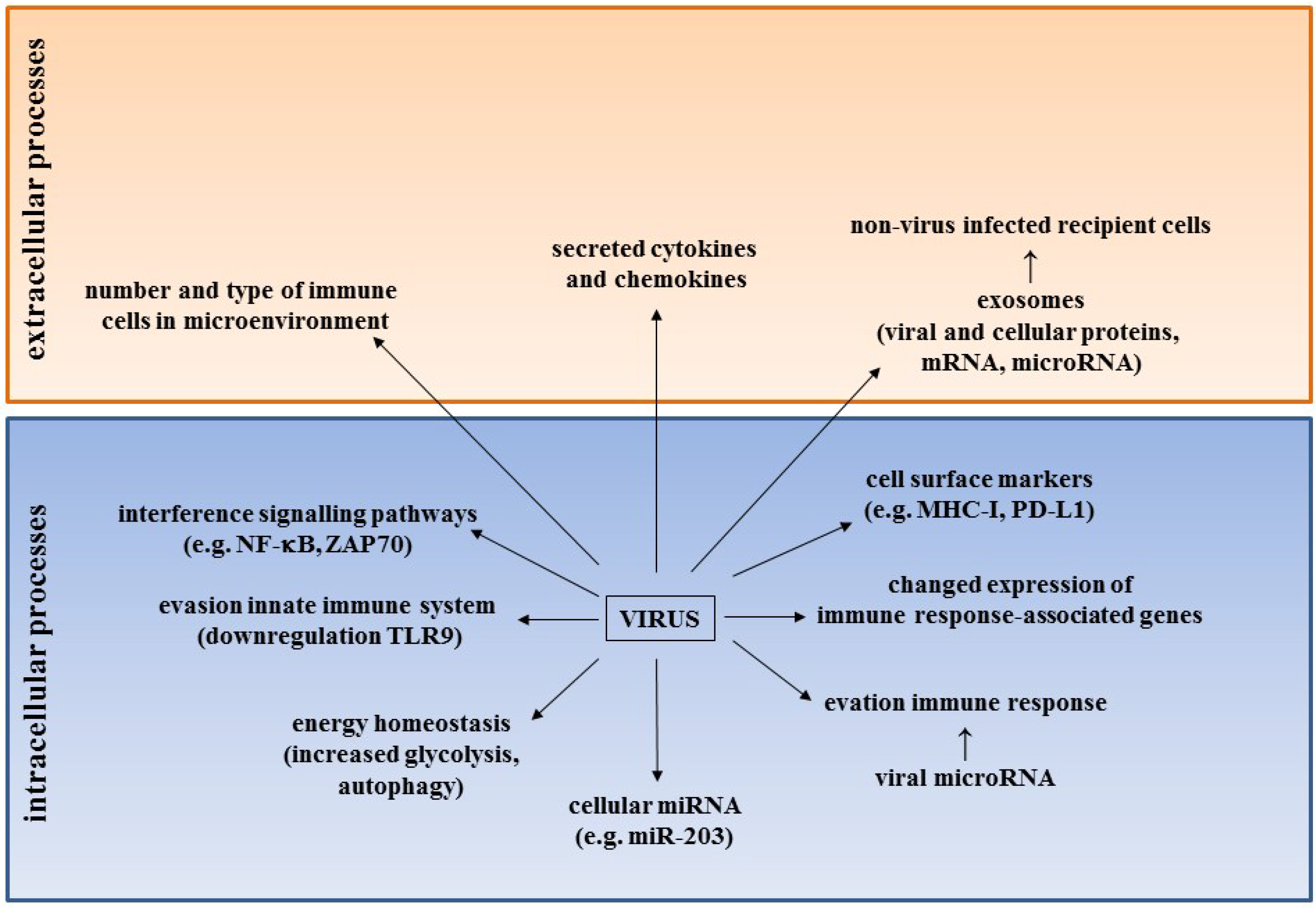
3. HPyV and Emerging Hallmarks of Cancer
3.1. The Immune System and HPyV in Cancer
3.1.1. HPyV and Evasion of the Innate Immune System
3.1.2. Immune Cells in the Microenvironment of MCC
| Component | MCPyV-positive versus MCPyV-negative MCC | Reference |
|---|---|---|
| Cells in tumor microenvironment | ||
| -CD3+ T-cells | ||
| higher number in MCPyV-positive MCC | [88,89,90] | |
| -CD4+ T-cells | ||
| high number associated with high LT-ag expression | [90] | |
| -CD8+ T-cells | higher number in MCPyV-positive MCC | |
| [89,91,92] | ||
| - CD16+ natural killer cell | higher number in MCPyV-positive MCC | |
| [88,90] | ||
| -CD20+ B cells | more common in MCPyV-positive MCC; | |
| no significant difference between MCPyV-positive and –negative MCC | [93] [89] | |
| -CD68+ macrophages | higher number in MCPyV-positive MCC | |
| [88,90,94,95] | ||
| -CD69+ macrophages | higher number in MCPyV-positive MCC | |
| [90,94,95] | ||
| -FoxP3+ regulatory T-cells | more common in MCPyV-positive MCC | |
| [93] | ||
| Cell surface markers: | ||
| -CD3D | enrichment of transcripts in MCPyV-positive MCC | [89] |
| enrichment of transcripts in MCPyV-positive MCC | ||
| -CD3G | lacking in CD8+ T-cells | [89] |
| -CXCR3 | lower levels in MCPyV-positive MCC | [93] |
| -MHC-I | higher in MCPyV-positive MCC | [96] |
| -PD1 | higher in MCPyV-positive MCC | [95,97,98] |
| -Tim-3 | [97] | |
| Signal transduction proteins | ||
| -NFκB levels | lower in MCPyV-positive MCC | [99] |
| -IκB levels | lower in MCPyV-positive MCC | [99] |
| -TANK | ||
| reduction in MCPyV st-ag expressing cells MCC13 cells | [99] | |
| - ZAP70 | compared to virus-negative cells | |
| enrichment of transcripts in MCPyV-positive MCC | [89] | |
| Cytokines/chemokines | ||
| -CCL20 | reduction in MCPyV st-ag expressing cells MCC13 cells | [99] |
| compared to virus-negative cells | ||
| -CXCL-9 | reduction in MCPyV st-ag expressing cells MCC13 cells | [99] |
| compared to virus-negative cells | ||
| -IL-2 | reduction in MCPyV st-ag expressing cells MCC13 cells | [99] |
| compared to virus-negative cells | ||
| -IL-8 | reduction in MCPyV st-ag expressing cells MCC13 cells | [99] |
| compared to virus-negative cells | ||
| -Prokineticin 1 mRNA | higher in MCPyV-negative MCC | [90] |
| -Prokineticin 2 mRNA | higher in MCPyV-positive MCC | [90] |
| Other differentially expressed proteins | ||
| -granzyme B (role in apoptosis) | ||
| Expression was rare in CD8+ cells | [93] |
3.1.3. Changes in Expression of Cell Surface Markers on MCC Cells
3.1.4. Expression Profile of Genes Associated with the Immune Response in MCC
3.1.5. Effect of st-ag on the NF-κB Pathway
3.1.6. Viral Microrna and Evation of the Immune Response
3.2. The Role of HPyV microRNA and HPyV-induced microRNA in Cancer
3.3. Effect of HPyV on Energy Homeostasis
3.4. HPyV and Exosomes
4. Therapeutic Strategies against Emerging Hallmarks of Cancer
5. Conclusion and Future Perspectives
Author Contributions
Conflicts of Interest
References
- Johne, R.; Buck, C.B.; Allander, T.; Atwood, W.J.; Garcea, R.L.; Imperiale, M.J.; Major, E.O.; Ramqvist, T.; Norkin, L.C. Taxonomical developments in the family Polyomaviridae. Arch. Virol. 2011, 156, 1627–1634. [Google Scholar] [CrossRef] [PubMed]
- Peretti, A.; FitzGerald, P.C.; Bliskovsky, V.; Pastrana, D.V.; Buck, C.B. Genome Sequence of a Fish-Associated Polyomavirus, Black Sea Bass (Centropristis striata) Polyomavirus 1. Genome Announc. 2015, 3, e01476–14. [Google Scholar] [CrossRef] [PubMed]
- DeCaprio, J.A.; Imperiale, M.J.; Major, E.O. Polyomaviruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins, and Wolters Kluwer: Philadelphia, PA, USA, 2013; Volume 2, pp. 1633–1661. [Google Scholar]
- Carter, J.J.; Daugherty, M.D.; Qi, X.; Bheda-Malge, A.; Wipf, G.C.; Robinson, K.; Roman, A.; Malik, H.S.; Galloway, D.A. Identification of an overprinting gene in Merkel cell polyomavirus provides evolutionary insight into the birth of viral genes. Proc. Natl. Acad. Sci. USA 2013, 110, 12744–12749. [Google Scholar] [CrossRef] [PubMed]
- Schowalter, R.W.; Buck, C.B. The merkel cell polyomavirus minor capsid protein. PLOS Pathog. 2013, 9, e1003558. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, B.; Moens, U. Genome analysis of non-human primate polyomaviruses. Infect. Genet. Evol. 2014, 26, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Fulghieri, C.; Bloom, S. SarahElizabeth Stewart. Emerg. Infect. Dis. 2014, 20, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Sweet, B.H.; Hilleman, M.R. The vacuolating virus, SV40. Proc. Soc. Exp. Biol. Med. 1960, 105, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; DeCaprio, J.A.; Fluck, M.M.; Schaffhausen, B.S. Cellular transformation by Simian Virus 40 and Murine Polyomavirus T antigens. Semin. Cancer Biol. 2009, 19, 218–228. [Google Scholar] [CrossRef] [PubMed]
- An, P.; Sáenz Robles, M.T.; Pipas, J.M. Large T antigens of polyomaviruses: Amazing molecular machines. Annu. Rev. Microbiol. 2012, 66, 213–236. [Google Scholar] [CrossRef] [PubMed]
- Valis, J.D.; Strandberg, J.D.; Shah, K.V. Transformation of hamster kidney cells by simian papovavirus SA12. Proc. Soc. Exp. Biol. Med. 1979, 160, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Bastien, C.; Feunteun, J. The hamster polyomavirus transforming properties. Oncogene 1988, 2, 129–135. [Google Scholar] [PubMed]
- Chen, J.D.; Neilson, K.; van Dyke, T. Lymphotropic papovavirus early region is specifically regulated in transgenic mice and efficiently induces neoplasia. J. Virol. 1989, 63, 2204–2214. [Google Scholar] [PubMed]
- Dela Cruz, F.N., Jr.; Giannitti, F.; Li, L.; Woods, L.W.; del Valle, L.; Delwart, E.; Pesavento, P.A. Novel polyomavirus associated with brain tumors in free-ranging raccoons, western United States. Emerg. Infect Dis. 2013, 19, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Simmons, J.H.; Riley, L.K.; Franklin, C.L.; Besch-Williford, C.L. Hamster polyomavirus infection in a pet Syrian hamster (Mesocricetus auratus). Vet. Pathol. 2001, 38, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Colegrove, K.M.; Wellehan, J.F., Jr.; Rivera, R.; Moore, P.F.; Gulland, F.M.; Lowenstine, L.J.; Nordhausen, R.W.; Nollens, H.H. Polyomavirus infection in a free-ranging California sea lion (Zalophus californianus) with intestinal T-cell lymphoma. J. Vet. Diagn. Invest. 2010, 22, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Stevens, H.; Bertelsen, M.F.; Sijmons, S.; van Ranst, M.; Maes, P. Characterization of a novel polyomavirus isolated from a fibroma on the trunk of an African elephant (Loxodonta africana). PLOS ONE 2013, 8, e77884. [Google Scholar] [CrossRef] [PubMed]
- Johne, R.; Müller, H. Polyomaviruses of birds: Etiological agents of inflammatory diseases in a tumor virus family. J. Virol. 2007, 81, 11554–11559. [Google Scholar] [CrossRef] [PubMed]
- Gardner, S.D.; Field, A.M.; Coleman, D.V.; Humle, B. New human papovavirus (B.K.) isolated from urine after renal transplantation. Lancet 1971, 1, 1253–1257. [Google Scholar] [CrossRef] [PubMed]
- Padgett, B.L.; Walker, D.L.; ZuRhein, G.M.; Eckroade, R.J.; Dessel, B.H. Cultivation of papova-like virus from human brain with progressive multifocal leukoencephalopathy. Lancet 1971, 1, 1257–1260. [Google Scholar] [CrossRef] [PubMed]
- Atkin, S.J.; Griffin, B.E.; Dilworth, S.M. Polyoma virus and simian virus 40 as cancer models: History and perspectives. Semin. Cancer Biol. 2009, 19, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Pipas, J.M. SV40: Cell transformation and tumorigenesis. Virology 2009, 384, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Allander, T.; Andreasson, K.; Gupta, S.; Bjerkner, A.; Bogdanovic, G.; Persson, M.A.; Dalialis, T.; Ramqvist, T.; Andersson, B. Identification of a third human polyomavirus. J. Virol. 2007, 81, 4130–4136. [Google Scholar] [CrossRef] [PubMed]
- Gaynor, A.; Nissen, M.D.; Whiley, D.M.; Mackay, I.M.; Lambert, S.B.; Wu, G.; Brennan, D.C.; Storch, G.A.; Wang, D. Identification of a novel polyomavirus from patients with acute respiratory tract infections. PLOS Pathog. 2007, 3, 595–604. [Google Scholar] [CrossRef]
- Feng, H.; Shuda, M.; Chang, Y.; Moore, P.S. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science 2008, 319, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Schowalter, R.M.; Pastrana, D.V.; Pumphrey, K.A.; Moyer, A.L.; Buck, C.B. Merkel cell polyomavirus and two previously unknown polyomaviruses are chronically shed from human skin. Cell Host Microbe 2010, 7, 509–515. [Google Scholar] [CrossRef] [PubMed]
- van der Meijden, E.; Janssens, R.W.; Lauber, C.; Bouwens Bavinck, J.N.; Gorbalenya, A.E.; Feltkamp, M.C. Discovery of a new human polyomavirus associated with trichodysplasia spinulosa in an immunocompromized patient. PLOS Pathog. 2010, 6, e1001024. [Google Scholar] [CrossRef] [PubMed]
- Scuda, N.; Hofmann, J.; Calvignac-Spencer, S.; Ruprecht, K.; Liman, P.; Kühn, J.; Hengel, H.; Ehlers, B. A novel human polyomavirus closely related to the African green monkey-derived lymphotropic polyomavirus. J. Virol. 2011, 85, 4586–4590. [Google Scholar] [CrossRef] [PubMed]
- Sauvage, V.; Foulongne, V.; Cheval, J.; Pariente, K.; Le Gouil, M.; Burguiere, A.M.; Manuguerra, J.C.; Caro, V.; Eloit, M. Human polyomavirus related to the green monkey lymphotropic polyomavirus. Emerg. Infect. Dis. 2011, 17, 1364–1370. [Google Scholar] [PubMed]
- Buck, C.B.; Phan, G.Q.; Raiji, M.T.; Murphy, P.M.; McDermott, D.H.; McBride, A.A. Complete genome sequence of a tenth human polyomavirus. J. Virol. 2012, 86, 10887. [Google Scholar] [CrossRef] [PubMed]
- Siebrasse, E.A.; Reyes, A.; Lim, E.S.; Zhao, G.; Mkakosya, R.S.; Manary, M.J.; Gordon, J.I.; Wang, D. Identification of MW polyomavirus, a novel polyomavirus in human stool. J. Virol. 2012, 86, 10321–10326. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Greninger, A.L.; Isa, P.; Phan, T.G.; Martinez, M.A.; de la Luz Sanchez, M.; Contreras, J.F.; Santos-Preciado, J.I.; Parsonnet, J.; Miller, S.; et al. Discovery of a novel polyomavirus in acute diarrheal samples from children. PLOS ONE 2012, 7, e49449. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.S.; Reyes, A.; Antonio, M.; Saha, D.; Ikumapayi, U.N.; Adeyemi, M.; Stine, O.C.; Skelton, R.; Brennan, D.C.; Mkakosya, R.S.; et al. Discovery of STL polyomavirus, a polyomavirus of ancestral recombinant origin that encodes a unique T antigen by alternative splicing. Virology 2013, 439, 163–164, Erratum in: Virology 2013, 439, 163–164. [Google Scholar]
- Korup, S.; Rietscher, J.; Calvignac-Spencer, S.; Trusch, F.; Hofmann, J.; Moens, U.; Sauer, I.; Voight, S.; Schmuck, R.; Ehlers, B. Identification of a novel human polyomavirus in organs of the gastrointestinal tract. PLOS ONE 2013, 8, e58021. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Pereira, M.; Rhodes, R.H.; An, P.; Pipas, J.M.; Jain, K.; Kapoor, A.; Briese, T.; Faust, P.L.; Lipkin, W.I. Identification of a novel polyomavirus in a pancreatic transplant recipient with retinal blindness and vascular myopathy. J. Infect. Dis. 2014, 210, 1595–1599. [Google Scholar] [CrossRef] [PubMed]
- Moens, U.; van Ghelue, M.; Song, X.; Ehlers, B. Serological cross-reactivity between human polyomaviruses. Rev. Med. Virol. 2013, 23, 250–264. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.S.; Meinerz, N.M.; Primi, B.; Wang, D.; Garcea, R.L. Common exposure to STL polyomavirus during childhood. Emerg. Infect. Dis. 2014, 20, 1559–1561. [Google Scholar] [CrossRef] [PubMed]
- Nicol, J.T.; Leblond, V.; Arnold, F.; Guerra, G.; Mazzoni, E.; Tognon, M.; Coursaget, P.; Touzé, A. Seroprevalence of human Malawi polyomavirus. J. Clin. Microbiol. 2014, 52, 321–323. [Google Scholar] [CrossRef] [PubMed]
- Moens, U.; van Ghelue, M.; Johannessen, M. Oncogenic potentials of the human polyomavirus regulatory proteins. Cell Mol. Life Sci. 2007, 64, 1656–1678. [Google Scholar] [CrossRef] [PubMed]
- Borchert, S.; Czech-Sioli, M.; Neumann, F.; Schmidt, C.; Wimmer, P.; Dobner, T.; Grundhoff, A.; Fischer, N. High-affinity Rb binding, p53 inhibition, subcellular localization, and transformation by wild-type or tumor-derived shortened Merkel cell polyomavirus large T antigens. J. Virol. 2014, 88, 3144–3160. [Google Scholar] [CrossRef] [PubMed]
- Verhaegen, M.E.; Mangelberger, D.; Harms, P.W.; Vozheiko, T.D.; Weick, J.W.; Wilbert, D.M.; Saunders, T.L.; Ermilov, A.N.; Bichakjian, C.K.; Johnson, T.M.; Imperiale, M.J.; Dlugosz, A.A. Merkel cell polyomavirus small t antigen is oncogenic in transgenic mice. J. Invest. Dermatol. 2014. [Google Scholar] [CrossRef]
- Spurgeon, M.E.; Cheng, J.; Bronson, R.T.; Lambert, P.F.; DeCaprio, J.A. Tumorigenic activity of Merkel cell polyomavirus T antigens expressed in the stratified epithelium of mice. Cancer Res. 2015. [Google Scholar] [CrossRef]
- Chang, Y.; Moore, P.S. Merkel cell carcinoma: A virus-induced human cancer. Annu. Rev. Pathol. 2012, 7, 123–144. [Google Scholar] [CrossRef] [PubMed]
- Moens, U.; Johannessen, M. Human polyomaviruses and cancer: Expanding repertoire. J. Dtsch. Dermatol. Ges. 2008, 6, 704–708. [Google Scholar] [CrossRef] [PubMed]
- Abend, J.R.; Jiang, M.; Imperiale, M.J. BK virus and cancer: Innocent until proven guilty. Semin. Cancer Biol. 2009, 19, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Maginnis, M.S.; Atwood, W.J. JC virus: An oncogenic virus in animals and humans? Semin. Cancer Biol. 2009, 19, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Enam, S.; Del Valle, L.; Lara, C.; Gan, D.D.; Ortiz-Hidalgo, C.; Palazzo, J.P.; Khalili, K. Association of human polyomavirus JCV with colon cancer: Evidence for interaction of viral T-antigen and beta-catenin. Cancer Res. 2002, 62, 7093–7101. [Google Scholar] [PubMed]
- Bulut, Y.; Ozdemir, E.; Ozercan, H.I.; Etem, E.O.; Aker, F.; Toraman, Z.A.; Seyrek, A.; Firdolas, F. Potential relationship between BK virus and renal cell carcinoma. J. Med. Virol. 2013, 85, 1085–1089. [Google Scholar] [CrossRef] [PubMed]
- Delbue, S.; Ferrante, P.; Provenzano, M. Polyomavirus BK and prostate cancer: An unworthy scientific effort? Oncoscience 2014, 1, 296–303. [Google Scholar] [PubMed]
- Carluccio, S.; Signorini, L.; Elia, F.; Villani, S.; Delbue, S.; Ferrante, P. A potential linkage between the JC and BK polyomaviruses and brain and urinary tract tumors: A review of the literature. Adv. Tumor. Virol. 2014, 4, 17–24. [Google Scholar]
- zur Hausen, H. Red meat consumption and cancer: Reasons to suspect involvement of bovine infectious factors in colorectal cancer. Int. J. Cancer 2012, 130, 2475–2483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, L.; Deng, X.; Kapusinszky, B.; Delwart, E. What is for dinner? Viral metagenomics of US store bought beef, pork and chicken. Virology 2014, 468–470, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Peretti, A.; FitzGerald, P.C.; Bliskovsky, V.; Buck, C.B.; Pastrana, D.V. Hamburger polyomaviruses. J. Gen. Virol. 2015. [Google Scholar] [CrossRef]
- Schrama, D.; Groesser, L.; Ugurel, S.; Hafner, C.; Pastrana, D.V.; Buck, C.B.; Cerroni, L.; Theiler, A.; Becker, J.C. Presence of human polyomavirus 6 in mutation-specific BRAF inhibitor-induced epithelial proliferation. JAMA Dermatol. 2014, 150, 1180–1186. [Google Scholar] [CrossRef] [PubMed]
- Ramqvist, T.; Nordfors, C.; Dalianis, T.; Ragnarsson-Olding, B. DNA from human polyomaviruses, TSPyV, MWPyV, HPyV6, 7, and 9 was not detected in primary mucosal melanomas. Anticancer Res. 2014, 34, 639–643. [Google Scholar] [PubMed]
- Imajoh, M.; Hashida, Y.; Nakajima, H.; Sano, S.; Daibata, M. Prevalence and viral DNA loads of three novel human polyomaviruses in skin cancers from Japanese patients. J. Dermatol. 2013, 40, 657–660. [Google Scholar] [CrossRef] [PubMed]
- Scola, N.; Wieland, U.; Silling, S.; Altmeyer, P.; Stücker, M.; Kreuter, A. Prevalence of human polyomaviruses in common and rare types of non-Merkel cell carcinoma skin cancer. Br. J. Dermatol. 2012, 167, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Antonsson, A.; Bialasiewicz, S.; Rockett, R.J.; Jacob, K.; Bennett, I.C.; SLoots, T.P. Exploring the Prevalence of Ten Polyomaviruses and Two Herpes Viruses in Breast Cancer. PLOS ONE 2012, 7, e39842. [Google Scholar] [CrossRef] [PubMed]
- Foulongne, V.; Sauvage, V.; Hebert, C.; Dereure, O.; Cheval, J.; Gouilh, M.A.; Pariente, K.; Segondy, M.; Burguière, A.; Manuguerra, J.C.; Caro, V.; Eloit, M. Human skin microbiota: High diversity of DNA viruses identified on the human skin by high throughput sequencing. PLOS ONE 2012, 7, e38499. [Google Scholar] [CrossRef] [PubMed]
- Giraud, G.; Ramqvist, T.; Ragnarsson-Olding, B.; Dalialis, T. DNA from BK virus and JC virus and from KI, WU, and MC polyomaviruses as well as from simian virus 40 is not detected in non-UV-light-associated primary malignant melanomas of mucous membranes. J. Clin. Microbiol. 2008, 46, 3595–3598. [Google Scholar] [CrossRef] [PubMed]
- Bzhalava, D.; Johansson, H.; Ekström, J.; Faust, H.; Möller, B.; Eklund, C.; Nordin, P.; Stenquist, B.; Paoli, J.; Persson, B.; Forslund, O.; Dillner, J. Unbiased approach for virus detection in skin lesions. PLOS ONE 2013, 8, e655953. [Google Scholar] [CrossRef]
- Imajoh, M.; Hashida, Y.; Taniguchi, A.; Kamioka, M.; Daibata, M. Novel human polyomaviruses, Merkel cell polyomavirus and human polyomavirus 9, in Japanese chronic lymphocytic leukemia cases. J. Hematol. Oncol. 2012, 5, e25. [Google Scholar] [CrossRef]
- Kreuter, A.; Silling, S.; Dewan, M.; Stücker, M.; Wieland, U. Evaluation of 4 recently discovered human polyomaviruses in primary cutaneous B-cell and T-cell lymphoma. Arch. Dermatol. 2011, 147, 1449–1451. [Google Scholar] [CrossRef] [PubMed]
- Duncavage, E.J.; Pfeifer, J.D. Human polyomaviruses 6 and 7 are not detectable in Merkel cell polyomavirus-negative Merkel cell carcinoma. J. Cutan. Pathol. 2011, 38, 790–796. [Google Scholar] [PubMed]
- Kanitakis, J.; Kazem, S.; van Der Meijden, E.; Feltkamp, M. Absence of the trichodysplasia spinulosa-associated polyomavirus in human pilomatricomas. Eur. J. Dermatol. 2011, 21, 453–454. [Google Scholar] [PubMed]
- Babakir-Mina, M.; Ciccozzi, M.; Campitelli, L.; Aquaro, S.; Lo Coco, A.; Perno, C.F.; Ciotti, M. Identification of the novel KI Polyomavirus in paranasal and lung tissues. J. Med. Virol. 2009, 81, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Giraud, G.; Ramqvist, T.; Pastrana, D.V.; Pavot, V.; Lindau, C.; Kogner, P.; Orrego, A.; Buck, C.B.; Allander, T.; Holm, S.; Gustavsson, B.; Dalianis, T. DNA from KI, WU and Merkel cell polyomavirus is not detected in childhood central nervous system tumors or neuroblastomas. PLOS ONE 2009, 4, e8239. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, B.; Honkaniemi, E.; Goh, S.; Giraud, G.; Forestier, E.; von Döbeln, U.; Allander, T.; Dalianis, T.; Bogdanovic, G. KI, WU, and Merkel cell polyomavirus DNA was not detected in guthrie cards of children who later developed acute lymphoblastic leukemia. J. Pediatr. Hematol. Oncol. 2012, 34, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Teramoto, S.; Kaiho, M.; Takano, Y.; Endo, R.; Kikuta, H.; Sawa, H.; Ariga, T.; Ishiguro, N. Detection of KI polyomavirus and WU polyomavirus DNA by real-time polymerase chain reaction in nasopharyngeal swabs and in normal lung and lung adenocarcinoma tissues. Microbiol. Immunol. 2011, 55, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Duncavage, E.J.; Le, B.M.; Wang, D.; Pfeifer, J.D. Merkel cell polyomavirus: A specific marker for Merkel cell carcinoma in histologically similar tumors. Am. J. Surg. Pathol. 2009, 33, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.; Höfler, D.; Koleganova, N.; Pawlita, M. Human polyomaviruses and other human viruses in neuroendocrine tumors. Cancer Epidemiol. Biomarkers Prev. 2011, 20, 1558–1561. [Google Scholar] [CrossRef] [PubMed]
- Du-Thanh, A.; Foulongne, V.; Guillot, B.; Dereure, O. Recently discovered human polyomaviruses in lesional and non-lesional skin of patients with primary cutaneous T-cell lymphomas. J. Dermatol. Sci. 2013, 71, 140–142. [Google Scholar] [CrossRef] [PubMed]
- Hashida, Y.; Taniguchi, A.; Yawata, T.; Hosokawa, S.; Murakami, M.; Hiroi, M.; Ueba, T.; Daibata, M. Prevalence of human cytomegalovirus, polyomaviruses, and oncogenic viruses in glioblastoma among Japanese subjects. Infect Agent Cancer 2015, 10, e3. [Google Scholar]
- Rennspiess, D.; Pujari, S.; Keijzers, M.; Abdul-Hamid, M.A.; Hochstenbag, M.; Dingemans, A.; Kurz, A.K.; Speel, E.; Haugg, A.; Pastrana, D.V.; et al. Detection of Human Polyomavirus 7 in human thymic epithelial tumors. J. Thorac. Oncol. 2015, 10, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Gjoerup, O.; Chang, Y. Update on human polyomaviruses and cancer. Adv. Cancer Res. 2010, 106, 1–51. [Google Scholar] [PubMed]
- DeCaprio, J.A.; Garcea, R.L. A cornucopia of human polyomaviruses. Nat. Rev. Microbiol. 2013, 11, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Spurgeon, M.E.; Lambert, P.F. Merkel cell polyomavirus: A newly discovered human virus with oncogenic potential. Virology 2013, 435, 118–130. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.S.; Chang, Y. Why do viruses cause cancer? Highlights of the first century of human tumor virology. Nat. Rev. Cancer 2010, 10, 878–889. [Google Scholar] [CrossRef] [PubMed]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Vasanthakumar, A.; Grigoriadis, G. Modulating T regulatory cells in cancer: How close are we? Immunol. Cell Biol. 2013, 91, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Vossen, M.T.; Westerhout, E.M.; Söderberg, N.C.; Wiertz, E.J. Viral immune evasion: A masterpiece of evolution. Immunogenetics 2002, 54, 527–542. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Afanasiev, O.; Nghiem, P. Immunobiology of Merkel cell carcinoma: Implications for immunotherapy of a polyomavirus-associated cancer. Curr. Oncol. Rep. 2011, 13, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, N.; Shuda, M.; Gheit, T.; Kwun, H.J.; Cornet, I.; Saidj, D.; Zannetti, C.; Hasan, U.; Chang, Y.; Moore, P.S.; et al. The T Antigen Locus of Merkel Cell Polyomavirus downregulates Human Toll-Like Receptor 9 Expression. J. Virol. 2013, 87, 13009–13019. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Gade, P.; Xiao, W.; Kalvakolanu, D.V. The interferon signaling network and transcription factor C/EBP-beta. Cell Mol. Immunol. 2007, 4, 407–418. [Google Scholar] [PubMed]
- Johnson, P.F. Molecular stop signs: Regulation of cell-cycle arrest by C/EBP transcription factors. J. Cell Sci. 2005, 118, 2545–2555. [Google Scholar] [CrossRef] [PubMed]
- Nerlov, C. The C/EBP family of transcription factors: A paradigm for interaction between gene expression and proliferation control. Trends Cell Biol. 2007, 17, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Sihto, H.; Joensuu, H. Tumor-infiltrating lymphocytes and outcome in Merkel cell carcinoma, a virus-associated cancer. Oncoimmunology 2012, 1, 1420–1421. [Google Scholar] [CrossRef] [PubMed]
- Harms, P.W.; Patel, R.M.; Verhaegen, M.E.; Giordano, T.J.; Nash, K.T.; Johnson, C.N.; Daignault, S.; Thomas, D.G.; Gudjonsson, J.E.; Elder, J.T.; et al. Distinct gene expression profiles of viral- and nonviral-associated merkel cell carcinoma revealed by transcriptome analysis. J. Invest. Dermatol. 2013, 133, 936–945. [Google Scholar] [CrossRef] [PubMed]
- Lauttia, S.; Sihto, H.; Kavola, H.; Koljonen, V.; Böhling, T.; Joensuu, H. Prokineticins and Merkel cell polyomavirus infection in Merkel cell carcinoma. Br. J. Cancer 2014, 110, 1446–1455. [Google Scholar] [CrossRef] [PubMed]
- Andea, A.A.; Coit, D.G.; Amin, B.; Busam, K.J. Merkel cell carcinoma: Histologic features and prognosis. Cancer 2008, 113, 2549–2558. [Google Scholar] [CrossRef] [PubMed]
- Paulson, K.G.; Iyer, J.G.; Tegeder, A.R.; Thibodeau, R.; Schelter, J.; Koba, S.; Schrama, D.; Simonson, W.T.; Lemos, B.D.; Byrd, D.R.; et al. Transcriptome-wide studies of Merkel cell carcinoma and validation of intratumoral CD8+ lymphocyte invasion as an independent predictor of survival. J. Clin. Oncol. 2011, 29, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Wheat, R.; Roberts, C.; Waterboer, T.; Steele, J.; Marsden, J.; Steven, N.M.; Blackbourn, D.J. Inflammatory cell distribution in primary merkel cell carcinoma. Cancers 2014, 6, 1047–1064. [Google Scholar] [CrossRef] [PubMed]
- Sihto, H.; Böhling, T.; Kavola, H.; Koljonen, V.; Salmi, M.; Jalkanen, S.; Joensuu, H. Tumor infiltrating immune cells and outcome of Merkel cell carcinoma: A population-based study. Clin. Cancer Res. 2012, 18, 2872–2881. [Google Scholar] [CrossRef] [PubMed]
- Triozzi, P.L.; Fernandez, A.P. The role of the immune response in merkel cell carcinoma. Cancers (Basel) 2013, 5, 234–254. [Google Scholar] [CrossRef]
- Paulson, K.G.; Tegeder, A.; Willmes, C.; Iyer, J.G.; Afanasiev, O.K.; Schrama, D.; Koba, S.; Thibodeau, R.; Nagase, K.; Simonson, W.T.; et al. Downregulation of MHC-I expression is prevalent but reversible in Merkel cell carcinoma. Cancer Immunol. Res. 2014, 2, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Afanasiev, O.K.; Yelistratova, L.; Miller, N.; Nagase, K.; Paulson, K.; Iyer, J.G.; Ibrani, D.; Koelle, D.M.; Nghiem, P. Merkel polyomavirus-specific T cells fluctuate with merkel cell carcinoma burden and express therapeutically targetable PD-1 and Tim-3 exhaustion markers. Clin. Cancer Res. 2013, 19, 5351–5360. [Google Scholar] [CrossRef] [PubMed]
- Lipson, E.J.; Vincent, J.G.; Loyo, M.; Kagohara, L.T.; Luber, B.S.; Wang, H.; Xu, H.; Nayar, S.K.; Wang, T.S.; Sidransky, D.; et al. PD-L1 expression in the Merkel cell carcinoma microenvironment: Association with inflammation, Merkel cell polyomavirus and overall survival. Cancer Immunol. Res. 2013, 1, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, D.A.; Abdul-Sada, H.; Knight, L.M.; Jackson, B.R.; Richards, K.; Prescott, E.L.; Peach, A.H.; Blair, G.E.; Macdonald, A.; Whitehouse, A. Merkel cell polyomavirus small T antigen targets the NEMO adaptor protein to disrupt inflammatory signaling. J. Virol. 2013, 87, 13853–13867. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.A.; Huang, S.J.; Murphy, G.F.; Mollet, I.G.; Hijnen, D.; Muthukuru, M.; Schanbacher, C.F.; Edwards, V.; Miller, D.M.; Kim, J.E.; et al. Human squamous cell carcinomas evade the immune response by down-regulation of vascular E-selectin and recruitment of regulatory T cells. J. Exp. Med. 2008, 205, 2221–2234. [Google Scholar] [CrossRef] [PubMed]
- Gehad, A.E.; Lichtman, M.K.; Schmults, C.D.; Teaque, J.E.; Calarese, A.W.; Jiang, Y.; Watanabe, R.; Clark, R.A. Nitric oxide-producing myeloid-derived suppressor cells inhibit vascular E-selectin expression in human squamous cell carcinomas. J. Invest Dermatol. 2012, 132, 2642–2651. [Google Scholar] [CrossRef] [PubMed]
- Afanasiev, O.K.; Nagase, K.; Simonson, W.; Vandeven, N.; Blom., A.; Koelle, D.M.; Clark, R.; Nghiem, P. Vascular E-selectin expression correlates with CD8 lymphocyte infiltration and improved outcome in Merkel cell carcinoma. J. Invest. Dermatol. 2013, 133, 2065–2073. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, H.; Tanaka, K.; Jay, F.; Khoury, G.; Jay, G. Modulation of the tumorigenicity of human adenovirus-12-transformed cells by interferon. Cell 1985, 43, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Cromme, F.V.; van Bommel, P.F.; Walboomers, J.M.; Gallee, M.P.; Stern, P.L.; Kenemans, P.; Helmerhorst, T.J.; Stukart, M.J.; Meijer, C.J. Differences in MHC and TAP-1 expression in cervical cancer lymph node metastases as compared with the primary tumors. Br. J. Cancer 1994, 69, 1176–1181. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Jugovic, P.; York, I.; Russ, G.; Bennink, J.; Yewdell, J.; Ploeggh, H.; Johnson, D. Herpes simplex virus turns off the TAP to evade host immunity. Nature 1995, 375, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Koopman, L.A.; van Der Slik, A.R.; Giphart, M.J.; Fleuren, G.J. Human leukocyte antigen class I gene mutations in cervical cancer. J. Natl. Cancer Inst. 1999, 91, 1669–1677. [Google Scholar] [CrossRef] [PubMed]
- Haque, M.; Ueda, K.; Nakano, K.; Hirata, Y.; Parravicini, C.; Corbellino, M.; Yamanishi, K. Major histocompatibility complex class I molecules are down-regulated at the cell surface by the K5 protein encoded by Kaposi’s sarcoma-associated herpesvirus/human herpesvirus-8. J. Gen. Virol. 2001, 82, 1175–1180. [Google Scholar] [PubMed]
- Hansen, T.H.; Bouvier, M. MHC class I antigen presentation: Learning from viral evasion strategies. Nat. Rev. Immunol. 2009, 9, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Chretien, A.S.; Le Roy, A.; Vey, N.; Prebet, T.; Blaise, D.; Fauriat, C.; Olive, D. Cancer-Induced Alterations of NK-Mediated Target Recognition: Current and Investigational Pharmacological Strategies Aiming at Restoring NK-Mediated Anti-Tumor Activity. Front. Immunol. 2014, 5, e122. [Google Scholar] [CrossRef]
- Riella, L.V.; Paterson, A.M.; Sharpe, A.H.; Chandraker, A. Role of the PD-1 pathway in the immune response. Am. J. Transplant. 2012, 12, 2575–2587. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, H.; Kishimoto, S.; Shibagaki, T.; Nagata, M.; Yasuno, H. Merkel cell carcinoma with spontaneous regression: An immunohistochemical, ultrastructural, and TUNEL labeling study. Am. J. Dermatopathol. 1997, 19, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, N.; Nakanishi, G.; Kabuto, M.; Nakano, T.; Eto, H.; Nakajima, H.; Sano, S.; Tanaka, T. Merkel cell carcinoma showing regression after biopsy: Evaluation of programmed cell death 1-positive cells. J. Dermatol. 2015. [Google Scholar] [CrossRef]
- Arany, I.; Tyring, S.K. Status of cytokine and antigen presentation genes in Merkel cell carcinoma of the skin. J. Cutan. Med. Surg. 1998, 2, 138–141. [Google Scholar] [PubMed]
- Lord, S.J.; Rajotte, R.V.; Korbutt, G.S.; Bleackley, R.C. Granzyme B: A natural born killer. Immunol. Rev. 2003, 193, 31–38. [Google Scholar] [CrossRef]
- Pahl, H.L. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene 1999, 18, 6853–6866. [Google Scholar] [CrossRef] [PubMed]
- Hiroi, M.; Ohmori, Y. Constitutive nuclear factor kappaB activity is required to elicit interferon-gamma-induced expression of chemokine CXC ligand 9 (CXCL9) and CXCL10 in human tumor cell lines. Biochem. J. 2003, 376, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Arora, R.; Shuda, M.; Guastafierro, A.; Feng, H.; Toptan, T.; Tolstov, Y.; Normolle, D.; Vollmer, L.L.; Vogt, A.; Dömling, A.; et al. Survivin is a therapeutic target in Merkel cell carcinoma. Sci. Transl. Med. 2012, 4, 133ra56. [Google Scholar] [CrossRef] [PubMed]
- Van Ghelue, M.; Khan, M.T.; Ehlers, B.; Moens, U. Genome analysis of the new human polyomaviruses. Rev. Med. Virol. 2012, 22, 354–377. [Google Scholar] [CrossRef] [PubMed]
- Youlden, D.R.; Youl, P.H.; Peter Soyer, H.; Fritschi, L.; Baade, P.D. Multiple primary cancers associated with Merkel cell carcinoma in Queensland, Australia, 1982–2011. J. Invest. Dermatol. 2014, 134, 2883–2889. [Google Scholar] [CrossRef] [PubMed]
- Mogha, A.; Fautrel, A.; Mouchet, N.; Guo, N.; Corre, S.; Adamski, H.; Watier, E.; Misery, L.; Galibert, M.D. Merkel cell polyomavirus small T-antigen mRNA level is increased following in vivo UV-radiation. PLOS ONE 2010, 5, e11423. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, C.S.; Grundhoff, A.T.; Tevethia, S.; Pipas, J.M.; Ganem, D. SV40-encoded microRNAs regulate viral gene expression and reduce susceptibility to cytotoxic T cells. Nature 2005, 435, 682–686. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.E.; McClure, L.V.; Goga, A.; Sullivan, C.S. Pan-viral-microRNA screening identifies interferon inhibition as a common function of diverse viruses. Proc. Natl. Acad. Sci. USA 2015, 112, 1856–1861. [Google Scholar] [CrossRef] [PubMed]
- Bauman, Y.; Mandelboim, O. MicroRNA based immunoevasion mechanism of human polyomaviruses. RNA Biol. 2011, 8, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Bauman, Y.; Nachmani, D.; Vitenshtein, A.; Tsukerman, P.; Drayman, N.; Stern-Ginossar, N.; Lankry, D.; Gruda, R.; Mandelboim, O. An identical miRNA of the human JC and BK polyoma viruses targets the stress-induced ligand ULBP3 to escape immune elimination. Cell Host Microbe 2011, 9, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Paulson, K.G.; Murchison, E.P.; Afanasiev, O.K.; Alkan, C.; Leonard, J.H.; Byrd, D.R.; Hannon, G.J.; Nghiem, P. Identification and validation of a novel mature microRNA encoded by the Merkel cell polyomavirus in human Merkel cell carcinomas. J. Clin. Virol. 2011, 52, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Groettrup, M.; Soza, A.; Eggers, M.; Kuehn, L.; Dick, T.P.; Schild, H. A role for the proteasome regulator PA28alpha in antigen presentation. Nature 1996, 381, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Okkenhaug, K.; Bilancio, A.; Farjot, G.; Priddle, H.; Sancho, S.; Peskett, E.; Pearce, W.; Meek, S.E.; Salpekar, A.; Waterfield, M.D.; Smith, A.J.; Vanhaesevroeck, B. Impaired B and T cell antigen receptor signaling in p110delta PI 3-kinase mutant mice. Science 2002, 297, 1031–1034. [Google Scholar] [PubMed]
- Yan, Q.; Sharma-Kuinkel, B.K.; Deshmukh, H.; Tsalik, E.L.; Cyr, D.D.; Lucas, J.; Woods, C.W.; Scott, W.K.; Sempowski, G.D.; Thaden, J.T.; et al. Dusp3 and Psme3 Are Associated with Murine Susceptibility to Staphylococcus aureus Infection and Human Sepsis. PLOS Pathog. 2014, 10, e1004149. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.J.; Cox, J.E.; Kincaid, R.P.; Martinez, A.; Sullivan, C.S. Divergent MicroRNA targetomes of closely related circulating strains of a polyomavirus. J. Virol. 2013, 87, 11135–11147. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Hsu, K.S.; Lim, J.H.; Bruggeman, L.A.; Kao, H.Y. α-Actinin 4 Potentiates Nuclear Factor κ-Light-chain-enhancer of Activated B-cell (NF-κB) Activity in Podocytes Independent of Its Cytoplasmic Actin Binding Function. J. Biol. Chem. 2015, 290, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Jiao, S.; Zhang, Z.; Li, C.; Huang, M.; Shi, Z.; Wang, Y.; Song, X.; Liu, H.; Li, C.; Chen, M.; et al. The kinase MST4 limits inflammatory responses through direct phosphorylation of the adaptor TRAF6. Nat. Immunol. 2015, 16, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, R.P.; Sullivan, C.S. Virus-Encoded microRNAs: An Overview and a Look to the Future. PLOS Pathog. 2012, 8, e1003018. [Google Scholar] [CrossRef] [PubMed]
- Lagatie, O.; Tritsmans, L.; Stuyver, L.J. The miRNA world of polyomaviruses. Virol. J. 2013, 10, e268. [Google Scholar] [CrossRef]
- Chen, C.J.; Cox, J.E.; Azarm, K.D.; Wylie, K.N.; Woolard, K.D.; Pesavento, P.A.; Sullivan, C.S. Identification of a polyomavirus microRNA highly expressed in tumors. Virology 2015, 476, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Haecker, I.; Yang, Y.; Gao, S.J.; Renne, R. γ-Herpesvirus-encoded miRNAs and their roles in viral biology and pathogenesis. Curr. Opin. Virol. 2013, 3, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Renwick, N.; Cekan, P.; Masry, P.A.; McGeary, S.E.; Miller, J.B.; Hafner, M.; Li, Z.; Mihailovic, A.; Morozov, P.; Brown, M.; et al. Multicolor microRNA FISH effectively differentiates tumor types. J. Clin. Invest. 2013, 123, 2694–2702. [Google Scholar] [CrossRef] [PubMed]
- Grundhoff, A.; Sullivan, C.S. Virus-encoded microRNAs. Virology 2011, 411, 325–343. [Google Scholar] [CrossRef] [PubMed]
- Cullen, B.R. MicroRNAs as mediators of viral evasion of the immune system. Nat. Immunol. 2013, 14, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Fimia, G.M.; Corazzari, M.; Antonioli, M.; Piacentini, M. Ambra1 at the crossroad between autophagy and cell death. Oncogene 2013, 32, 3311–3318. [Google Scholar] [CrossRef] [PubMed]
- Sung, C.K.; Yim, H.; Andrews, E.; Benjamin, T.L. A mouse polyomavirus-encoded microRNA targets the cellular apoptosis pathway through Smad2 inhibition. Virology 2014, 468–470, 57–62. [Google Scholar]
- Skalsky, R.L.; Cullen, B.R. Viruses, microRNAs, and host interactions. Annu. Rev. Microbiol. 2010, 64, 123–141. [Google Scholar] [CrossRef] [PubMed]
- Moens, U.; Seternes, O.M.; Johansen, B.; Rekvig, O.P. Mechanisms of transcriptional regulation of cellular genes by SV40 large T- and small t-antigens. Virus Genes 1997, 15, 135–154. [Google Scholar] [CrossRef] [PubMed]
- Cullen, B.R. Transcription and processing of human microRNA precursors. Mol. Cell 2004, 16, 861–865. [Google Scholar] [CrossRef] [PubMed]
- Seo, G.J.; Fink, L.H.; O’Hara, B.; Atwood, W.J.; Sullivan, C.S. Evolutionary conserved function of a viral microRNA. J. Virol. 2008, 82, 9823–9828. [Google Scholar] [CrossRef] [PubMed]
- Broekema, N.M.; Imperiale, M.J. miRNA regulation of BK polyomavirus replication during early infection. Proc. Natl. Acad. Sci. USA 2013, 110, 8200–8205. [Google Scholar] [CrossRef] [PubMed]
- Shuda, M.; Arora, R.; Kwun, H.J.; Feng, H.; Sarid, R.; Fernández-Figueras, M.T.; Tolstov, Y.; Gjoerup, O.; Mansukhani, M.M.; Swerdlow, S.H.; et al. Human Merkel cell polyomavirus infection I. MCV T antigen expression in Merkel cell carcinoma, lymphoid tissues and lymphoid tumors. Int. J. Cancer 2009, 125, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Brostoff, T.; Dela Cruz, F.N., Jr.; Church, M.E.; Woolard, K.D.; Pesavento, P.A. The raccoon polyomavirus genome and tumor antigen transcription are stable and abundant in neuroglial tumors. J. Virol. 2014, 88, 12816–12824. [Google Scholar] [CrossRef] [PubMed]
- Braconi, C.; Valeri, N.; Gasparini, P.; Huang, N.; Tacciolo, C.; Nuovo, G.; Suzuki, T.; Croce, C.M.; Patel, T. Hepatitis C virus proteins modulate microRNA expression and chemosensitivity in malignant hepatocytes. Clin. Cancer Res. 2010, 16, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.H.; Hu, T.F.; Chen, Y.C.; Tsai, Y.N.; Tsai, Y.H.; Cheng, C.C.; Wang, H.W. The manipulation of miRNA-gene regulatory networks by KSHV induces endothelial cell motility. Blood 2011, 118, 2896–2905. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Tilahun, Y.; Taha, O.; Alao, H.; Kodys, K.; Catalano, D.; Szabo, G. Increased microRNA-155 expression in the serum and peripheral monocytes in chronic HCV infection. J. Transl. Med. 2012, 10, e151. [Google Scholar] [CrossRef]
- Lin, L.; Yin, X.; Hu, X.; Wang, Q.; Zheng, L. The impact of hepatitis B virus x protein and microRNAs in hepatocellular carcinoma: A comprehensive analysis. Tumor. Biol. 2014, 35, 11695–11700. [Google Scholar] [CrossRef]
- Ma, L.; Deng, X.; Wu, M.; Zhang, G.; Huang, J. Down-regulationof miRNA204 by LMP-1 enhances CDC2 activity and facilitates invasion of EBV-associated nasopharyngeal carcinoma cells. FEBS Lett. 2014, 588, 1562–1570. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, S.; Pan, Q.; Blencowe, B.J.; Claycomb, J.M.; Frappier, L. Epstein-Barr virus EBNA1 protein regulates viral latency through effects on let-7 microRNA and dicer. J. Virol. 2014, 88, 11166–11177. [Google Scholar] [CrossRef] [PubMed]
- Vernin, C.; Thenoz, M.; Pinatel, C.; Gessain, A.; Gout, O.; Delfau-Larue, M.H.; Nazaret, N.; Legras-Lachuer, C.; Wattel, E.; Mortreux, F. HTLV-1 bZIP factor HBZ promotes cell proliferation and genetic instability by activating OncomiRs. Cancer Res. 2014, 74, 6082–6093. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Lee, L.; Caramuta, S.; Höög, A.; Browaldh, N.; Björnhagen, V.; Larsson, C.; Lui, W.O. MicroRNA expression patterns related to merkel cell polyomavirus infection in human merkel cell carcinoma. J. Invest. Dermatol. 2014, 134, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On respiratory impairment in cancer cells. Science 1956, 124, 269–270. [Google Scholar] [PubMed]
- Zhao, Y.; Liu, H.; Riker, A.I.; Fodstad, O.; Ledoux, S.P.; Wilson, G.L.; Tan, M. Emerging metabolic targets in cancer therapy. Front. Biosci. 2011, 16, 1844–1860. [Google Scholar] [CrossRef]
- Kinnaird, A.; Michelakis, E.D. Metabolic modulation of cancer: A new frontier with great translational potential. J. Mol. Med. 2015, 93, 127–142. [Google Scholar] [CrossRef] [PubMed]
- George, A.; Girault, S.; Testard, A.; Delva, R.; Soulié, P.; Couturier, O.F.; Morel, O. The impact of (18)F-FDG-PET/CT on Merkel cell carcinoma management: A retrospective study of 66 scans from a single institution. Nucl. Med. Commun. 2014, 35, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, K.; Nishino, H.; Iwashima, A. Analysis of hexose transport in untransformed and sarcoma virus-transformed mouse 3T3 cells by photoaffinity binding of cytochalasin B. Biochim. Biophys Acta 1985, 821, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Bravard, A.; Beaumatin, J.; Luccioni, C.; Fritsch, P.; Lefrançois, D.; Thenet, S.; Adolphe, M.; Dutrillaux, B. Chromosomal, mitochondrial and metabolic alterations in SV40-transformed rabbit chondrocytes. Carcinogenesis 1992, 13, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Lachaise, F.; Martin, G.; Drougard, C.; Perl, A.; Vuillaume, M.; Wegnez, M.; Sarasin, A.; Daya-Grosjean, L. Relationship between posttranslational modification of transaldolase and catalase deficiency in UV-sensitive repair-deficient xeroderma pigmentosum fibroblasts and SV40-transformed human cells. Free Radic. Biol. Med. 2001, 30, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Noch, E.; Sariyer, I.K.; Gordon, J.; Khalili, K. JC virus T-antigen regulates glucose metabolic pathways in brain tumor cells. PLOS ONE 2012, 7, e35054. [Google Scholar] [CrossRef] [PubMed]
- Duracher, D.; Wyczechowska, D.; Wilk, A.; Lassak, A.; Zea, A.; Estrada, J.; Reis, K. The Effects of JCV T-antigen on Tumor Cell Metabolism. Available online: http://www.medschool.lsuhsc.edu/cancer_center/docs/Dustin%20Duracher%20Poster%2036x60.pdf (accessed on 10 March 2015).
- Ramanathan, A.; Wang, C.; Schreiber, S.L. Perturbational profiling of a cell-line model of tumorigenesis by using metabolic measurements. Proc. Natl. Acad. Sci. USA 2005, 102, 5992–5997. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Veldman, T.; Rundell, K.; Schlegel, R. Simian virus 40 small tumor antigen activates AKT and telomerase and induces anchorage-independent growth of human epithelial cells. J. Virol. 2002, 76, 10685–10691. [Google Scholar] [CrossRef] [PubMed]
- Elstrom, R.L.; Bauer, D.E.; Buzzai, M.; Karnauskas, R.; Harris, M.H.; Plas, D.R.; Zhuang, H.; Cinalli, R.M.; Alavi, A.; Rudin, C.M.; Thompson, C.B. Akt stimulates aerobic glycolysis in cancer cells. Cancer Res. 2004, 64, 3892–3899. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Viciana, P.; Collins, C.; Fried, M. Polyoma and SV40 proteins differentially regulate PP2A to activate distinct cellular signaling pathways involved in growth control. Proc. Natl. Acad. Sci. USA 2006, 103, 19290–19295. [Google Scholar] [CrossRef] [PubMed]
- Sontag, E.; Fedorov, S.; Kamibayashi, C.; Robbins, D.; Cobb, M.; Mumby, M. The interaction of SV40 small tumor antigen with protein phosphatase 2A stimulates the map kinase pathway and induces cell proliferation. Cell 1993, 75, 887–897. [Google Scholar] [CrossRef] [PubMed]
- Agnetti, G.; Maraldi, T.; Giorentini, D.; Giordano, E.; Prata, C.; Hakim, G.; Muscari, C.; Guarnieri, C.; Caldarera, C.M. Activation of glucose transport during simulated ischemia in H9c2 cardiac myoblasts is mediated by protein kinase C isoforms. Life Sci. 2005, 78, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Madan, E.; Gogna, R.; Bhatt, M.; Pati, U.; Kuppusamy, P.; Mahdi, A.A. Regulation of glucose metabolism by p53: Emerging new roles for the tumor suppressor. Oncotarget 2011, 2, 948–957. [Google Scholar] [PubMed]
- Shivakumar, C.V.; Das, G.C. Interaction of human polyomavirus BK with the tumor-suppressor protein p53. Oncogene 1996, 13, 323–332. [Google Scholar] [PubMed]
- Staib, C.; Pesch, J.; Gerwig, R.; Gerber, J.K.; Brehm, U.; Stangl, A.; Grummt, F. p53 inhibits JC virus DNA replication in vivo and interacts with JC virus large T-antigen. Virology 1996, 219, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Kenific, C.M.; Debnath, J. Cellular and metabolic functions for autophagy in cancer cells. Trends Cell Biol. 2015, 25, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.M.; Jung, J.U. Modulation of the Autophagy Pathway by Human Tumor Viruses. Semin. Cancer Biol. 2013, 23, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Bouley, S.J.; Maginnis, M.S.; Derdowski, A.; Gee, G.V.; O’Hara, B.A.; Nelson, C.D.; Bara, A.M.; Atwood, W.J.; Dugan, A.S. Host cell autophagy promotes BK virus infection. Virology 2014, 456–457, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.H.; Rangarajan, A. Simian virus 40 small T antigen activates AMPK and triggers autophagy to protect cancer cells from nutrient deprivation. J. Virol. 2009, 83, 8565–8574. [Google Scholar] [CrossRef] [PubMed]
- Rundell, K.; Major, E.O.; Lampert, M. Association of cellular 56,000- and 32,000-molecular-weight protein with BK virus and polyoma virus t-antigens. J. Virol. 1981, 37, 1090–1093. [Google Scholar] [PubMed]
- Bollag, B.; Hofstetter, C.A.; Reviriego-Mendoza, M.M.; Frisque, R.J. JC virus small T antigen binds phosphatase PP2A and Rb family proteins and is required for efficient viral DNA replication activity. PLOS ONE 2010, 5, e10606. [Google Scholar] [CrossRef] [PubMed]
- Shuda, M.; Kwun, H.J.; Feng, H.; Chang, Y.; Moore, P.S. Human Merkel cell polyomavirus small T antigen is an oncoprotein targeting the 4E-BP1 translation regulator. J. Clin. Invest. 2011, 121, 3623–3634. [Google Scholar] [CrossRef] [PubMed]
- Berrios, C.; Jung, J.; Primi, B.; Wang, M.; Pedamallu, C.; Duke, F.; Marcelus, C.; Cheng, J.; Garcea, R.L.; Meyerson, M.; DeCaprio, J.A. Malawi polyomavirus is a prevalent human virus that interacts with known tumor suppressors. J. Virol. 2015, 89, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Kwun, H.J.; Shuda, M.; Camacho, C.J.; Gamper, A.M.; Thant, M.; Chang, Y.; Moore, P.S. Restricted Protein Phosphatase 2A Targeting by Merkel Cell Polyomavirus Small T Antigen. J. Virol. 2015, 89, 4191–4200. [Google Scholar] [CrossRef] [PubMed]
- Basile, A.; Darbinian, N.; Kaminski, R.; White, M.K.; Gentilella, A.; Turco, M.C.; Khalili, K. Evidence for modulation of BAG3 by polyomavirus JC early protein. J. Gen. Virol. 1999, 90, 1629–1640. [Google Scholar] [CrossRef]
- Sariyer, I.K.; Merabova, N.; Patel, P.K.; Knezevic, T.; Rosati, A.; Turco, M.C.; Khalili, K. Bag3-induced autophagy is associated with degradation of JCV oncoprotein, T-Ag. PLOS ONE 2012, 7, e45000. [Google Scholar] [CrossRef] [PubMed]
- Ribbens, J.J.; Moser, A.B.; Hubbard, W.C.; Bongarzone, E.R.; Maegawa, G.H. Characterization and application of a disease-cell model for a neurodegenerative lysosomal disease. Mol. Genet. Metab. 2014, 111, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Meckes, D.G., Jr.; Raab-Traub, N. Microvesicles and viral infection. J. Virol. 2011, 85, 12844–12854. [Google Scholar] [CrossRef] [PubMed]
- Brinton, L.T.; Sloane, H.S.; Kester, M.; Kelly, K.A. Formation and role of exosomes in cancer. Cell Mol. Life Sci. 2015, 72, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Schorey, J.S.; Cheng, Y.; Singh, P.P.; Smith, V.L. Exosomes and other extracellular vesicles in host-pathogen interactions. EMBO Rep. 2015, 16, 24–43. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnaiah, V.; Thumann, C.; Fofana, I.; Habersetzer, F.; Pan, Q.; de Ruiter, P.E.; Willemsen, R.; Demmers, J.A.; Stalin Raj, V.; Jenster, G.; et al. Exosome-mediated transmission of hepatitis C virus between hepatoma Huh7.5 cells. Proc. Natl. Acad. Sci. USA 2013, 110, 13109–13113. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, E.; Narayanan, A.; van Duyne, R.; Shabbeer-Meyering, S.; Iordanskiy, S.; Saifuddin, M.; Das, R.; Afonso, P.V.; Sampey, G.C.; Chung, M.; et al. Human T-lymphotropic virus type 1-infected cells secrete exosomes that contain Tax protein. J. Biol. Chem. 2014, 289, 22284–22305. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, Y.; Duan, J.; Ma, Y.; Shen, Z.; Wei, L.; Cui, X.; Zhang, J.; Xie, Y.; Liu, J. Quantitative proteomic analysis of exosome protein content changes induced by hepatitis B virus in Huh-7 cells using SILAC labeling and LC-MS/MS. J. Proteome Res. 2014, 13, 5391–5402. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Chennakrishnaiah, S.; Audemard, E.; Montermini, L.; Meehan, B.; Rak, J. Oncogenic ras-driven cancer cell vesiculation leads to emission of double-stranded DNA capable of interacting with target cells. Biochem. Biophys. Res. Commun. 2014, 451, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Giovannelli, I.; Martelli, F.; Repice, A.; Massacesi, L.; Azzi, A.; Giannecchini, S. Detection of JCPyV microRNA in blood and urine samples of multiple sclerosis patients under natalizumab therapy. J. Neurovirol. 2015. [Google Scholar] [CrossRef]
- Meckes, D.G., Jr.; Shair, K.H.; Marquitz, A.R.; Kung, C.P.; Edwards, R.H.; Raab-Traub, N. Human tumor virus utilizes exosomes for intercellular communication. Proc. Natl. Acad. Sci. USA 2010, 107, 20370–20375. [Google Scholar] [CrossRef] [PubMed]
- Dreux, M.; Garaigorta, U.; Boyd, B.; Décembre, E.; Chung, J.; Whitten-Bauer, C.; Wieland, S.; Chisari, F.V. Short-range exosomal transfer of viral RNA from infected cells to plasmacytoid dendritic cells triggers innate immunity. Cell Host Microbe 2012, 12, 558–570. [Google Scholar] [CrossRef] [PubMed]
- Chugh, P.E.; Sin, S.H.; Ozgur, S.; Henry, D.H.; Menezes, P.; Griffith, J.; Eron, J.J.; Damania, B.; Dittmer, D.P. Systemically circulating viral and tumor-derived microRNAs in KSHV-associated malignancies. PLOS Pathog. 2013, 9, e1003484. [Google Scholar] [CrossRef] [PubMed]
- Meckes, D.G., Jr.; Gunawardena, H.P.; Dekroon, R.M.; Heaton, P.R.; Edwards, R.H.; Ozgur, S.; Griffith, J.D.; Damania, B.; Raab-Traub, N. Modulation of B-cell exosome proteins by gamma herpesvirus infection. Proc. Natl. Acad. Sci. USA 2013, 110, E2925–E2933. [Google Scholar] [CrossRef] [PubMed]
- Velders, M.P.; Macedo, M.F.; Provenzano, M.; Elmishad, A.G.; Holzhütter, H.G.; Carbone, M.; Kwast, W.M. Human T cell responses to endogenously presented HLA-A*0201 restricted peptides of Simian virus 40 large T antigen. J. Cell Biochem. 2001, 82, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Vlastos, A.; Andreasson, K.; Tegerstedt, K.; Holländerová, D.; Heidari, S.; Forstová, J.; Ramqvist, T.; Dalianis, T. VP1 pseudocapsids, but not a glutathione-S-transferase VP1 fusion protein, prevent polyomavirus infection in a T-cell immune deficient experimental mouse model. J. Med. Virol. 2003, 70, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.B.; Shearer, M.H.; Tarbox, J.A.; Kang, H.S.; Jumper, C.A.; Bright, R.K.; Kennedy, R.C. In vitro simian virus 40 large tumor antigen expression correlates with differential immune responses following DNA immunization. Virology 2005, 332, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Gomez, B.P.; Viscidi, R.P.; Peng, S.; He, L.; Ma, B.; Wu, T.C.; Hung, C.F. Development of a DNA vaccine targeting Merkel cell polyomavirus. Vaccine 2012, 30, 1322–1329. [Google Scholar] [CrossRef] [PubMed]
- Sospedra, M.; Schippling, S.; Yousef, S.; Jelcic, I.; Bofill-Mas, S.; Planas, R.; Stellmann, J.P.; Demina, V.; Cinque, P.; Garcea, R.; et al. Treating progressive multifocal leukoencephalopathy with interleukin 7 and vaccination with JC virus capsid protein VP1. Clin. Infect Dis. 2014, 59, 1588–1592. [Google Scholar] [CrossRef] [PubMed]
- Willmes, C.; Adam, C.; Alb, M.; Völkert, L.; Houben, R.; Becker, J.C.; Schrama, D. Type I and II IFNs inhibit Merkel cell carcinoma via modulation of the Merkel cell polyomavirus T antigens. Cancer Res. 2012, 72, 2120–2128. [Google Scholar] [CrossRef] [PubMed]
- Nakajima., H.; Takaishi, M.; Yamamoto, M.; Kamijima, R.; Kodama, H.; Tarutani, M.; Sano, S. Screening of the specific polyoma virus as diagnostic and prognostic tools for Merkel cell carcinoma. J. Dermatol. Sci. 2009, 56, 211–213. [Google Scholar] [CrossRef] [PubMed]
- Biver-Dalle, C.; Nguyen, T.; Touzé, A.; Saccomani, C.; Penz, S.; Cunat-Peultier, S.; Riou-Gotta, M.O.; Humbert, P.; Coursaget, P.; Aubin, F. Use of interferon-alpha in two patients with Merkel cell carcinoma positive for Merkel cell polyomavirus. Acta Oncol. 2011, 50, 479–480. [Google Scholar] [CrossRef] [PubMed]
- Chapuis, A.G.; Afanasiev, O.K.; Iyer, J.G.; Paulson, K.G.; Parvathaneni, U.; Hwang, J.H.; Lai, I.; Roberts, I.M.; Sloan, H.L.; Bhatia, S.; et al. Regression of metastatic Merkel cell carcinoma following transfer of polyomavirus-specific T cells and therapies capable of re-inducing HLA class-I. Cancer Immunol. Res. 2014, 2, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Hata, Y.; Matsuka, K.; Ito, O.; Matsuda, H.; Furuichi, H.; Konstantinos, A.; Nuri, B. Two cases of Merkel cell carcinoma cured by intratumor injection of natural human tumor necrosis factor. Plast Reconstr. Surg. 1997, 99, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Lampreave, J.L.; Bénard, F.; Alavi, A.; Jimeneze-Hoyuela, J.; Fraker, D. PET evaluation of therapeutic limb perfusion in Merkel’s cell carcinoma. J. Nucl. Med. 1998, 39, 2087–2090. [Google Scholar] [PubMed]
- Olieman, A.F.; Liénard, D.; Eggermont, A.M.; Kroon, B.B.; Lejeune, F.J.; Hoekstra, H.J.; Koops, H.S. Hyperthermic isolated limb perfusion with tumor necrosis factor alpha, interferon gamma, and melphalan for locally advanced nonmelanoma skin tumors of the extremities: A multicenter study. Arch. Surg. 1999, 134, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Krishna, S.M.; Kim, C.N. Merkel cell carcinoma in a patient treated with adalimumab: Case report. Cutis 2011, 87, 81–84, Erratum in Cutis 2011, 87, 185. [Google Scholar]
- Linn-Rasker, S.P.; van Albada-Kuipers, G.A.; Dubois, S.V.; Janssen, K.; Zweers, P.G. Merkel cell carcinoma during treatment with TNF-alpha inhibitors: Coincidence or warning? Ned. Tijdschr. Geneeskd. 2012, 156, A44–A64. [Google Scholar]
- Reiss, K.A.; Forde, P.M.; Brahmer, J.R. Harnessing the power of the immune system via blockade of PD-1 and PD-L1: A promising new anticancer strategy. Immunotherapy 2014, 6, 459–475. [Google Scholar] [CrossRef] [PubMed]
- Orba, Y.; Sawa, H.; Iwata, H.; Tanaka, S.; Nagashima, K. Inhibition of virus production in JC virus-infected cells by postinfection RNA interference. J. Virol. 2004, 78, 7270–7273. [Google Scholar] [CrossRef] [PubMed]
- Sabbioni, S.; Callegari, E.; Spizzo, R.; Veronese, A.; Altavilla, G.; Corallini, A.; Negrini, M. Anticancer activity of an adenoviral vector expressing short hairpin RNA against BK virus T-ag. Cancer Gene Ther. 2007, 14, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Houben, R.; Shuda, M.; Weinkam, R.; Schrama, D.; Feng, H.; Chang, Y.; Moore, P.S.; Becker, J.C. Merkel cell polyoma virus-infected Merkel cell carcinoma cells require expression of viral T antigens. J. Virol. 2010, 84, 7064–7072. [Google Scholar] [CrossRef] [PubMed]
- Angermeyer, S.; Hesbacher, S.; Becker, J.C.; Schrama, D.; Houben, R. Merkel cell polyomavirus-positive Merkel cell carcinoma cells do not require expression of the viral small T antigen. J. Invest Dermatol. 2013, 133, 2059–2064. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.C.; Wang, M.; Fang, C.Y.; Chen, P.L.; Shen, C.H.; Chang, D. Inhibition of BK virus replication in human kidney cells by BK virus large tumor antigen-specific shRNA delivered by JC virus-like particles. Antiviral Res. 2014, 103, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Hartman, Z.C.; Wei, J.; Glass, O.K.; Guo, H.; Lei, G.; Yang, X.Y.; Osada, T.; Hobeika, A.; Delcayre, A.; Le Pecq, J.B.; Morse, M.A.; Clay, T.M.; Lyerly, H.K. Increasing vaccine potency through exosome antigen targeting. Vaccine 2011, 29, 9361–9367. [Google Scholar] [CrossRef] [PubMed]
- Shuda, M.; Feng, H.; Kwun, H.J.; Rosen, S.T.; Gjoerup, O.; Moore, P.S.; Chang, Y. T antigen mutations are a human tumor-specific signature for Merkel cell polyomavirus. Proc. Natl. Acad. Sci. USA 2008, 105, 16272–16277. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moens, U.; Rasheed, K.; Abdulsalam, I.; Sveinbjørnsson, B. The Role of Merkel Cell Polyomavirus and Other Human Polyomaviruses in Emerging Hallmarks of Cancer. Viruses 2015, 7, 1871-1901. https://doi.org/10.3390/v7041871
Moens U, Rasheed K, Abdulsalam I, Sveinbjørnsson B. The Role of Merkel Cell Polyomavirus and Other Human Polyomaviruses in Emerging Hallmarks of Cancer. Viruses. 2015; 7(4):1871-1901. https://doi.org/10.3390/v7041871
Chicago/Turabian StyleMoens, Ugo, Kashif Rasheed, Ibrahim Abdulsalam, and Baldur Sveinbjørnsson. 2015. "The Role of Merkel Cell Polyomavirus and Other Human Polyomaviruses in Emerging Hallmarks of Cancer" Viruses 7, no. 4: 1871-1901. https://doi.org/10.3390/v7041871
APA StyleMoens, U., Rasheed, K., Abdulsalam, I., & Sveinbjørnsson, B. (2015). The Role of Merkel Cell Polyomavirus and Other Human Polyomaviruses in Emerging Hallmarks of Cancer. Viruses, 7(4), 1871-1901. https://doi.org/10.3390/v7041871





