Anti-CMV-IgG Positivity of Donors Is Beneficial for alloHSCT Recipients with Respect to the Better Short-Term Immunological Recovery and High Level of CD4+CD25high Lymphocytes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Characteristics
| Patients | |
| Number (n) | 99 |
| Age (median, range) | 45, 6–65 |
| Gender (female/male) | 54/45 |
| Type of transplantation (alternative/SIB) | 59/40 |
| Transplant material (PBPCs/BM) | 94/4 1 PBPCs + BM |
| Diagnosis | |
| AML | 53 |
| ALL | 23 |
| Other lymphoproliferative disorders | 3 |
| Myeloproliferative disorders | 9 |
| Myelodysplastic syndrome | 11 |
| Conditioning regimen: | |
| Myeloablative | 67 |
| Reduced intensity | 32 |
| aGvHD grade | |
| 0 | 55 |
| I | 14 |
| II | 13 |
| III | 5 |
| IV | 12 |
| CMV IgG serostatus | |
| anti-CMV-IgG negative | 20 |
| anti-CMV-IgG positive | 78 |
| Data not available | 1 |
| Donors | |
| Age (median, range) | 37, 19–62 |
| SIB donors (median, range) | 44, 19–62 |
| MUD donors (median, range) | 31, 20–54 |
| Gender (female/male) | 31, 48/51 |
| CMV IgG serostatus | |
| anti-CMV-IgG negative | 33 |
| anti-CMV-IgG positive | 64 |
| Data not available | 2 |
2.2. Phenotypic Analysis
2.3. EBV, CMV, and HHV6 Quantification
2.4. Anti-CMV IgG Antibody Quantification
2.5. CRP Quantification
2.6. Statistical Analysis
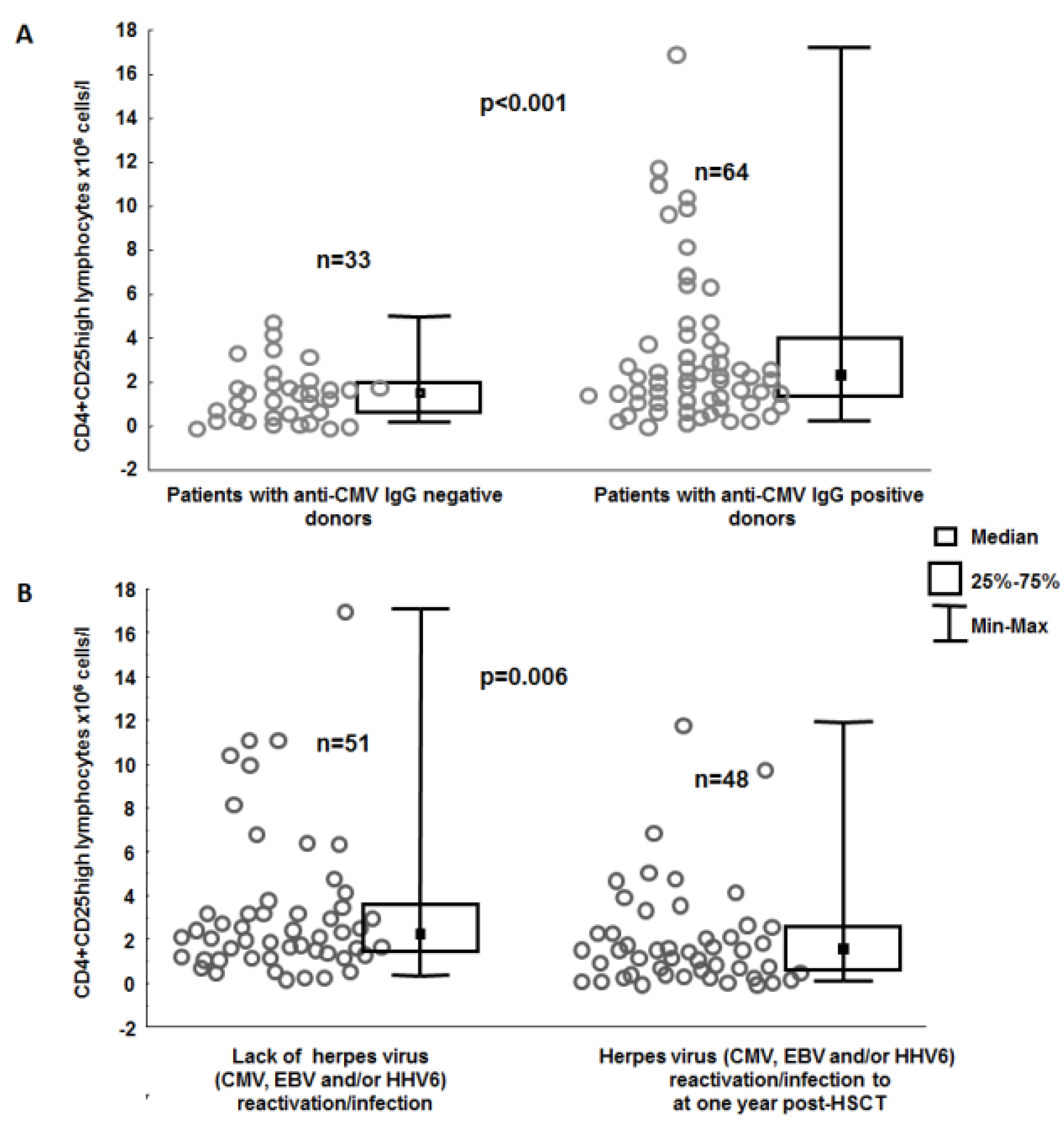
3. Results
| Parameters | Percentage of CD4+CD25high lymphocytes | HLA mismatch | Anti-CMV IgG serostatus (D−/R+) | |
|---|---|---|---|---|
| Entire group, n = 99 | ||||
| Coefficient | −2.421 | 1.147 | 1.555 | |
| p value | 0.022 | 0.051 | 0.010 | |
| Odds ratio | 0.089 | 3.150 | 4.737 | |
| −95% Cl | −4.489 | −0.004 | 0.389 | |
| +95% Cl | −0.353 | 2.298 | 2.722 | |
| In vivo T cell-depleted group (patients receiving ATG or Campath), n = 73 | ||||
| Coefficient | −2.779 | 1.196 | 1.755 | |
| p value | 0.033 | 0.061 | 0.010 | |
| Odds ratio | 0.062 | 3.306 | 5.784 | |
| −95% Cl | 0.005 | 0.945 | 1.537 | |
| +95% Cl | 0.794 | 11.570 | 21.764 | |

| Entire group, n = 99 | ||||
| Parameters | Number of CD4+ lymphocytes (×103 cells/L) | Percentage of CD4+CD25high lymphocytes | Number of CD4+CD25high lymphocytes (×103 cells/L) | MUD donor |
| Coefficient | 0.001 | 1.394 | −0.529 | 0.963 |
| p value | 0.496 | 0.223 | 0.031 | 0.047 |
| Odds ratio | 1.001 | 4.031 | 0.589 | 2.619 |
| −95% Cl | −0.003 | −0.863 | −1.009 | 0.014 |
| +95% Cl | 0.005 | 3.651 | −0.049 | 1.911 |
| In vivo T cell-depleted group (patients receiving ATG or Campath), n = 73 | ||||
| Parameters | HLA mismatch | Anti-CMV IgG serostatus (D−/R+) | Number of CD4+CD25high lymphocytes (×103 cells/L) | MUD donor |
| Coefficient | 0.600 | −0.423 | −0.363 | 1.970 |
| p value | 0.347 | 0.487 | 0.048 | 0.029 |
| Odds ratio | 1.822 | 0.655 | 0.696 | 7.173 |
| −95% Cl | 0.515 | 0.196 | 0.485 | 1.224 |
| +95% Cl | 6.450 | 2.194 | 0.997 | 42.016 |
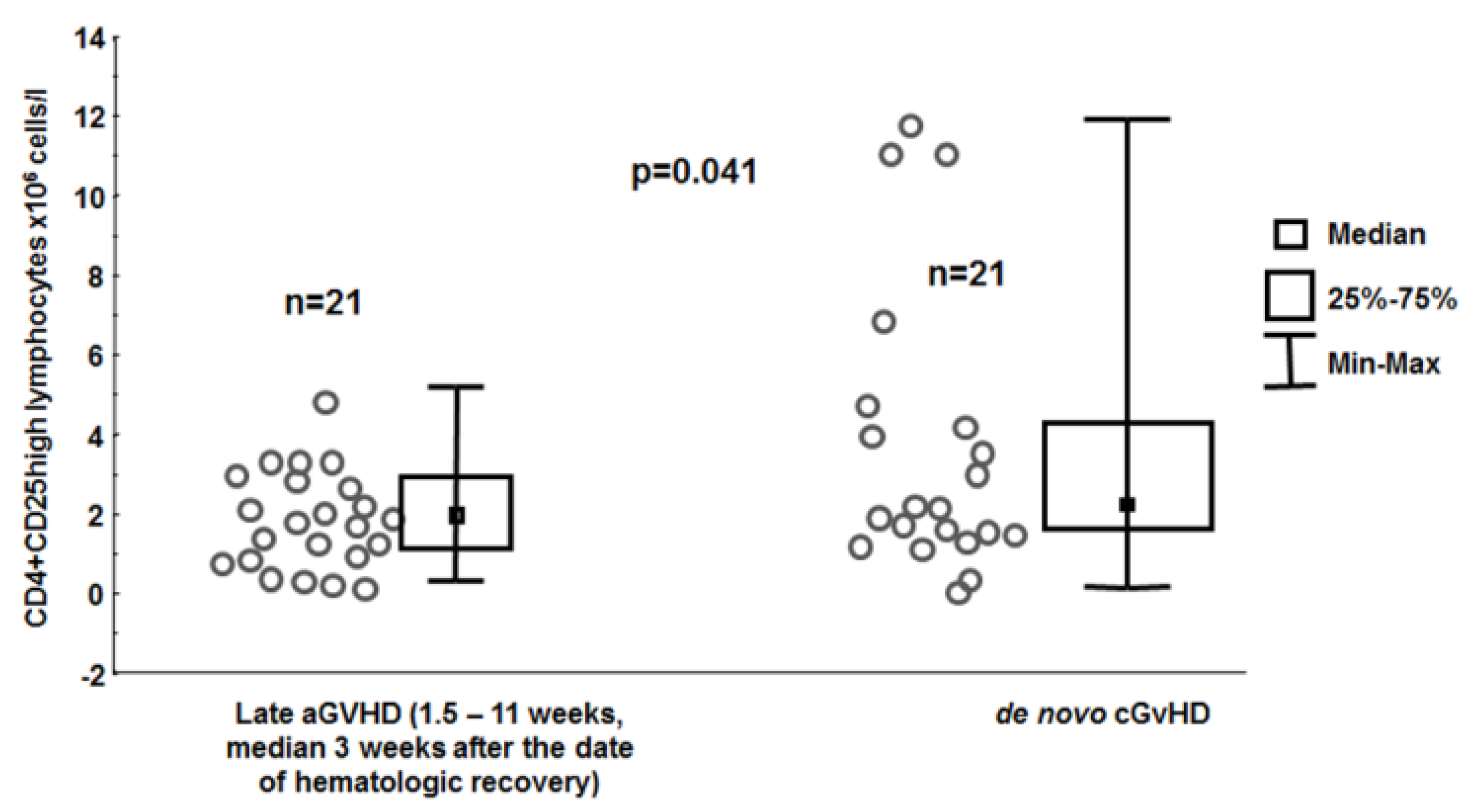
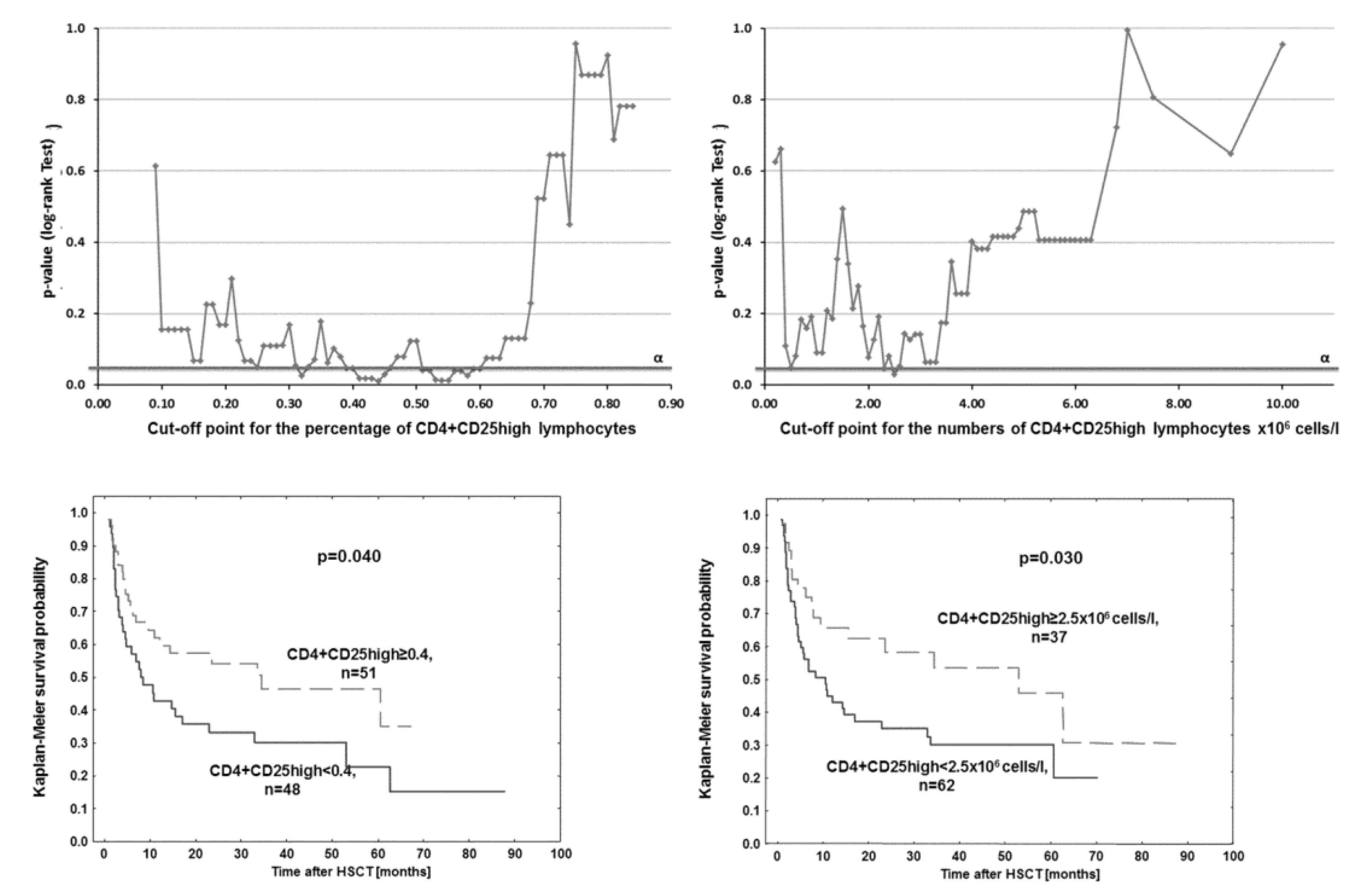
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ovsyannikova, I.G.; Jacobson, R.M.; Ryan, J.E.; Dhiman, N.; Vierkant, R.A.; Poland, G.A. Relationship between HLA polymorphisms and gamma interferon and interleukin-10 cytokine production in healthy individuals after rubella vaccination. Clin. Vaccine Immunol. 2007, 14, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Walldén, J.; Ilonen, J.; Roivainen, M.; Ludvigsson, J.; Vaarala, O. Effect of HLA genotype or CTLA-4 polymorphism on cytokine response in healthy children. Scand. J. Immunol. 2008, 68, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Vasto, S.; Colonna-Romano, G.; Larbi, A.; Wikby, A.; Caruso, C.; Pawelec, G. Role of persistent CMV infection in configuring T cell immunity in the elderly. Immun. Ageing 2007, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Caruso, C.; Candore, G.; Modica, M.; Bonanno, C.; Sireci, G.; Dieli, F.; Salerno, A. HLA and cytkine production. J. Interferon Cytokine Res. 1996, 16, 983–988. [Google Scholar] [PubMed]
- Fürst, D.; Müller, C.; Vucinic, V.; Bunjes, D.; Herr, W.; Gramatzki, M.; Schwerdtfeger, R.; Arnold, R.; Einsele, H.; Wulf, G.; et al. High-resolution HLA matching in hematopoietic stem cell transplantation: A retrospective collaborative analysis. Blood 2013, 122, 3220–3229. [Google Scholar] [CrossRef] [PubMed]
- Duquesnoy, R.J. HLA epitope based matching for transplantation. Transpl. Immunol. 2014, 31, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Sakaguchi, S. The role of regulatory T cells in controlling immunologic self-tolerance. Int. Rev. Cytol. 2003, 225, 1–32. [Google Scholar] [PubMed]
- Takahashi, T.; Sakaguchi, S. Naturally arising CD25+CD4+ regulatory T cells in maintaining immunologic self-tolerance and preventing autoimmune disease. Curr. Mol. Med. 2003, 3, 693–706. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, E.; Sakihama, T.; Setoguchi, R.; Tanaka, K.; Sakaguchi, S. Induction of antigen-specific immunologic tolerance by in vivo and in vitro antigen-specific expansion of naturally arising Foxp3+CD25+CD4+ regulatory T cells. Int. Immunol. 2004, 16, 1189–1201. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Sakaguchi, N.; Asano, M.; Itoh, M.; Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 1995, 155, 1151–1164. [Google Scholar] [PubMed]
- Zhang, Y.; Maksimovic, J.; Naselli, G.; Qian, J.; Chopin, M.; Blewitt, M.E.; Oshlack, A.; Harrison, L.C. Genome-wide DNA methylation analysis identifies hypomethylated genes regulated by FOXP3 in human regulatory T cells. Blood 2013, 122, 2823–2836. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, D.A.; Zheng, S.G.; Gray, J.D. The role of the combination of IL-2 and TGF-beta or IL-10 in the generation and function of CD4+ CD25+ and CD8+ regulatory T cell subsets. J. Leukoc. Biol. 2003, 74, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.G.; Wang, J.H.; Gray, J.D.; Soucier, H.; Horwitz, D.A. Natural and induced CD4+CD25+ cells educate CD4+CD25- cells to develop suppressive activity: The role of IL-2, TGF-beta, and IL-10. J. Immunol. 2004, 172, 5213–5221. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Putnam, A.L.; Xu-Yu, Z.; Szot, G.L.; Lee, M.R.; Zhu, S.; Gottlieb, P.A.; Kapranov, P.; Gingeras, T.R.; Fazekas de St Groth, B.; et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006, 203, 1701–1711. [Google Scholar] [CrossRef] [PubMed]
- Seddiki, N.; Santner-Nanan, B.; Martinson, J.; Zaunders, J.; Sasson, S.; Landay, A.; Solomon, M.; Selby, W.; Alexander, S.I.; Nanan, R.; et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 2006, 203, 1693–700. [Google Scholar] [CrossRef] [PubMed]
- Sojka, D.K.; Huang, Y.H.; Fowell, D.J. Mechanisms of regulatory T-cell suppression—A diverse arsenal for a moving target. Immunology 2008, 124, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Wing, K.; Onishi, Y.; Prieto-Martin, P.; Yamaguchi, T. Regulatory T cells: How do they suppress immune responses? Int. Immunol. 2009, 21, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Engelhardt, B.G.; Crowe, J.E. Homing in on acute graft vs. host disease: Tissue-specific T regulatory and Th17 cells. Curr. Top. Microbiol. Immunol. 2010, 341, 121–146. [Google Scholar] [PubMed]
- Thornton, A.M.; Donovan, E.E.; Piccirillo, C.A.; Shevach, E.M. Cutting Edge: IL-2 Is Critically Required for the In Vitro Activation of CD4+CD25+ T Cell Suppressor Function. J. Immunol. 2004, 172, 6519–6523. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, N.; Lönnqvist, B.; Ringdén, O.; Rajantie, J.; Siimes, M.A.; Volin, L.; Ruutu, T.; Nikoskelainen, J.; Toivanen, A.; Ryder, L. Graft-versus-leukaemia activity associated with cytomegalovirus seropositive bone marrow donors but separated from graft-versus-host disease in allograft recipients with AML. Eur. J. Haematol. 1987, 38, 350–255. [Google Scholar] [CrossRef] [PubMed]
- Remberger, M.; Mattsson, J.; Hassan, Z.; Karlsson, N.; LeBlanc, K.; Omazic, B.; Okas, M.; Sairafi, D.; Ringdén, O. Risk factors for acute graft-versus-host disease grades II-IV after reduced intensity conditioning allogeneic stem cell transplantation with unrelated donors: A single centre study. Bone Marrow Transplant. 2008, 41, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Jaskula, E.; Bochenska, J.; Kocwin, E.; Tarnowska, A.; Lange, A. CMV Serostatus of Donor-Recipient Pairs Influences the Risk of CMV Infection/Reactivation in HSCT Patients. Bone Marrow Res. 2012, 2012, 375075. [Google Scholar] [CrossRef] [PubMed]
- Jaskula, E.; Dlubek, D.; Sedzimirska, M.; Duda, D.; Tarnowska, A. Lange, a Reactivations of cytomegalovirus, human herpes virus 6, and Epstein-Barr virus differ with respect to risk factors and clinical outcome after hematopoietic stem cell transplantation. Transplant. Proc. 2010, 42, 3273–3276. [Google Scholar] [CrossRef] [PubMed]
- Jaskula, E.; Lange, A.; Dlubek, D.; Kyrcz-Krzemień, S.; Markiewicz, M.; Dzierzak-Mietla, M.; Jedrzejczak, W.W.; Gronkowska, A.; Nowak, J.; Warzocha, K.; et al. IL-10 promoter polymorphisms influence susceptibility to aGvHD and are associated with proportions of CD4+FoxP3+ lymphocytes in blood after hematopoietic stem cell transplantation. Tissue Antigens 2013, 82, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Herndler-Brandstetter, D.; Landgraf, K.; Tzankov, A.; Jenewein, B.; Brunauer, R.; Laschober, G.T.; Parson, W.; Kloss, F.; Gassner, R.; Lepperdinger, G.; Grubeck-Loebenstein, B. The impact of aging on memory T cell phenotype and function in the human bone marrow. J. Leukoc. Biol. 2012, 91, 197–205. [Google Scholar] [CrossRef] [PubMed]
- McIver, Z.; Melenhorst, J.J.; Wu, C.; Grim, A.; Ito, S.; Cho, I.; Hensel, N.; Battiwalla, M.; Barrett, A.J. Donor lymphocyte count and thymic activity predict lymphocyte recovery and outcomes after matched-sibling hematopoietic stem cell transplant. Haematologica 2013, 98, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Azuma, E.; Hirayama, M.; Yamamoto, H.; Komada, Y. The role of donor age in naive T-cell recovery following allogeneic hematopoietic stem cell transplantation: The younger the better. Leuk. Lymphoma 2002, 43, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Hanley, P.J.; Bollard, C.M. Controlling cytomegalovirus: Helping the immune system take the lead. Viruses 2014, 6, 2242–2258. [Google Scholar] [CrossRef] [PubMed]
- Snydman, D.R. Editorial commentary: The complexity of latent cytomegalovirus infection in stem cell donors. Clin. Infect. Dis. 2014, 59, 482–483. [Google Scholar] [CrossRef] [PubMed]
- Boeckh, M.; Nichols, W.G. The impact of cytomegalovirus serostatus of donor and recipient before hematopoietic stem cell transplantation in the era of antiviral prophylaxis and preemptive therapy. Blood 2004, 103, 2003–2008. [Google Scholar] [CrossRef] [PubMed]
- Ljungman, P.; Brand, R.; Hoek, J.; de la Camara, R.; Cordonnier, C.; Einsele, H.; Styczynski, J.; Ward, K.N.; Cesaro, S. Donor cytomegalovirus status influences the outcome of allogeneic stem cell transplant: A study by the European group for blood and marrow transplantation. Clin. Infect. Dis. 2014, 59, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Ljungman, P. The role of cytomegalovirus serostatus on outcome of hematopoietic stem cell transplantation. Curr. Opin. Hematol. 2014, 21, 466–469. [Google Scholar] [CrossRef] [PubMed]
- Ljungman, P.; Brand, R.; Einsele, H.; Frassoni, F.; Niederwieser, D.; Cordonnier, C. Donor CMV serologic status and outcome of CMV-seropositive recipients after unrelated donor stem cell transplantation: An EBMT megafile analysis. Blood 2003, 102, 4255–4260. [Google Scholar] [CrossRef] [PubMed]
- Jaskula, E.; Dlubek, D.; Duda, D.; Bogunia-Kubik, K.; Mlynarczewska, A.; Lange, A. Interferon gamma 13-CA-repeat homozygous genotype and a low proportion of CD4+ lymphocytes are independent risk factors for cytomegalovirus reactivation with a high number of copies in hematopoietic stem cell transplantation recipients. Biol. Blood Marrow Transplant. 2009, 15, 1296–305. [Google Scholar] [CrossRef] [PubMed]
- CMV IgG Test System. Available online: http://www.zeusscientific.com/products/technology-systems/elisa/ (accessed on 19 March 2015).
- Jaskula, E.; Lange, A.; Kyrcz-Krzemien, S.; Markiewicz, M.; Dzierzak-Mietla, M.; Jedrzejczak, W.W.; Czajka, P.; Mordak-Domagala, M.; Lange, J.; Gronkowska, A.; et al. NOD2/CARD15 single nucleotide polymorphism 13 (3020insC) is associated with risk of sepsis and single nucleotide polymorphism 8 (2104C>T) with herpes viruses reactivation in patients after allogeneic hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2014, 20, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Ljungman, P.; Hakki, M.; Boeckh, M. Cytomegalovirus in hematopoietic stem cell transplant recipients. Infect. Dis. Clin. N. Am. 2010, 24, 319–337. [Google Scholar] [CrossRef]
- Ljungman, P.; Hakki, M.; Boeckh, M. Cytomegalovirus in hematopoietic stem cell transplant recipients. Hematol. Oncol. Clin. N. Am. 2011, 25, 151–169. [Google Scholar] [CrossRef]
- George, B.; Pati, N.; Gilroy, N.; Ratnamohan, M.; Huang, G.; Kerridge, I.; Hertzberg, M.; Gottlieb, D.; Bradstock, K. Pre-transplant cytomegalovirus (CMV) serostatus remains the most important determinant of CMV reactivation after allogeneic hematopoietic stem cell transplantation in the era of surveillance and preemptive therapy. Transpl. Infect. Dis. 2010, 12, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Ringdén, O.; Labopin, M.; Solders, M.; Beelen, D.; Arnold, R.; Ehninger, G.; Milpied, N.; Niederwieser, D.; Hamladji, R.M.; Kyrcz-Krzemien, S.; et al. Who Is the Best Hematopoietic Stem-Cell Donor for a Male Patient With Acute Leukemia? Transplantation 2014, 98, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Gratwohl, A.; Brand, R.; Apperley, J.; Biezen, A.V.; Bandini, G.; Devergie, A.; Schattenberg, A.; Frassoni, F.; Guglielmi, C.; Iacobelli, S.; et al. Graft-versus-host disease and outcome in HLA-identical sibling transplantations for chronic myeloid leukemia. Blood 2002, 100, 3877–3886. [Google Scholar] [CrossRef] [PubMed]
- Jagasia, M.; Arora, M.; Flowers, M.E.D.; Chao, N.J.; McCarthy, P.L.; Cutler, C.S.; Urbano-Ispizua, A.; Pavletic, S.Z.; Haagenson, M.D.; Zhang, M.J.; et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood 2012, 119, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Cicin-Sain, L.; Brien, J.D.; Uhrlaub, J.L.; Drabig, A.; Marandu, T.F.; Nikolich-Zugich, J. Cytomegalovirus infection impairs immune responses and accentuates T-cell pool changes observed in mice with aging. PLoS Pathog. 2012, 8, e1002849. [Google Scholar] [CrossRef] [PubMed]
- Strioga, M.; Pasukoniene, V.; Characiejus, D. CD8+ CD28− and CD8+ CD57+ T cells and their role in health and disease. Immunology 2011, 134, 17–32. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A.; Sinclair, E.; Hatano, H.; Hsue, P.Y.; Epling, L.; Hecht, F.M.; Bangsberg, D.R.; Martin, J.N.; McCune, J.M.; Deeks, S.G.; et al. Impact of HIV on CD8+ T cell CD57 expression is distinct from that of CMV and aging. PLoS One 2014, 9, e89444. [Google Scholar] [CrossRef] [PubMed]
- Terrazzini, N.; Bajwa, M.; Vita, S.; Cheek, E.; Thomas, D.; Seddiki, N.; Smith, H.; Kern, F. A novel cytomegalovirus-induced regulatory-type T-cell subset increases in size during older life and links virus-specific immunity to vascular pathology. J. Infect. Dis. 2014, 209, 1382–1392. [Google Scholar] [CrossRef] [PubMed]
- Magenau, J.M.; Qin, X.; Tawara, I.; Rogers, C.E.; Kitko, C.; Schlough, M.; Bickley, D.; Braun, T.M.; Jang, P.S.; Lowler, K.P.; et al. Frequency of CD4+CD25hiFOXP3+ regulatory T cells has diagnostic and prognostic value as a biomarker for acute graft-versus-host-disease. Biol. Blood Marrow Transplant. 2010, 16, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.; Munder, M.; Karakhanova, S.; Ho, A.D.; Goerner, M. The initial phase of graft-versus-host disease is associated with a decrease of CD4+CD25+ regulatory T cells in the peripheral blood of patients after allogeneic stem cell transplantation. Clin. Lab. Haematol. 2006, 28, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Z.; Sun, Z.; Li, Q.; Zhang, A.; Liu, H.; Xu, J.; Xu, X.; Geng, L.; Harris, D.; Hu, S.; Wang, Y. Correlation of the CD4+CD25high T-regulatory cells in recipients and their corresponding donors to acute GVHD. Transpl. Int. 2007, 20, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Baecher-Allan, C.; Brown, J.A.; Freeman, G.J.; Hafler, D.A. CD4+CD25high Regulatory Cells in Human Peripheral Blood. J. Immunol. 2001, 167, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Roncador, G.; Brown, P.J.; Maestre, L.; Hue, S.; Martínez-Torrecuadrada, J.L.; Ling, K.L.; Pratap, S.; Toms, C.; Fox, B.C.; Cerundolo, V.; et al. Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level. Eur. J. Immunol. 2005, 35, 1681–1691. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.G.; Wang, J.; Wang, P.; Gray, J.D.; Horwitz, D.A. IL-2 is essential for TGF-beta to convert naive CD4+CD25- cells to CD25+Foxp3+ regulatory T cells and for expansion of these cells. J. Immunol. 2007, 178, 2018–2027. [Google Scholar] [CrossRef] [PubMed]
- Zorn, E.; Nelson, E.A.; Mohseni, M.; Porcheray, F.; Kim, H.; Litsa, D.; Bellucci, R.; Raderschall, E.; Canning, C.; Soiffer, R.J.; et al. IL-2 regulates FOXP3 expression in human CD4+CD25+ regulatory T cells through a STAT-dependent mechanism and induces the expansion of these cells in vivo. Blood 2006, 108, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Boyman, O.; Sprent, J. The role of interleukin-2 during homeostasis and activation of the immune system. Nat. Rev. Immunol. 2012, 12, 180–190. [Google Scholar] [PubMed]
- Sester, M.; Gärtner, B.C.; Sester, U.; Girndt, M.; Mueller-Lantzsch, N.; Köhler, H. Is the cytomegalovirus serologic status always accurate? A comparative analysis of humoral and cellular immunity. Transplantation 2003, 76, 1229–1230. [Google Scholar] [CrossRef] [PubMed]
- Alcami, A.; Koszinowski, U.H. Viral mechanisms of immune evasion. Trends Microbiol. 2000, 21, 3542–3547. [Google Scholar]
- Jones, R.P. Infectious-like Spread of an Agent Leading to Increased Medical Admissions and Deaths in Wigan ( England ), during 2011 and 2012. Br. J. Med. Med. Res. 2014, 4, 4723–4741. [Google Scholar] [CrossRef]
- Alonso Arias, R.; Moro-García, M.A.; Echeverría, A.; Solano-Jaurrieta, J.J.; Suárez-García, F.M.; López-Larrea, C. Intensity of the humoral response to cytomegalovirus is associated with the phenotypic and functional status of the immune system. J. Virol. 2013, 87, 4486–4495. [Google Scholar] [CrossRef] [PubMed]
- Bour-Jordan, H.; Blueston, J. CD28 function: A balance of costimulatory and regulatory signals. J. Clin. Immunol 2002, 22, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Joe, G.; Hexner, E.; Zhu, J.; Emerson, S.G. Alloreactive Memory T Cells Are Responsible for the Persistence of Graft-versus-Host Disease. J. Immunol. 2005, 174, 3051–3058. [Google Scholar] [CrossRef] [PubMed]
- Paz Morante, M.; Briones, J.; Canto, E.; Sabzevari, H.; Martino, R.; Sierra, J.; Rodriguez-Sanchez, J.L.; Vidal, S. Activation-associated phenotype of CD3 T cells in acute graft-versus-host disease. Clin. Exp. Immunol. 2006, 145, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.W.; Levine, J.E.; Ferrara, J.L. M. Pathogenesis and management of graft-versus-host disease. Immunol. Allergy Clin. N. Am. 2010, 30, 75–101. [Google Scholar] [CrossRef]
- Ferrara, J.L.M.; Levine, J.E.; Reddy, P.; Holler, E. Graft-versus-host disease. Lancet 2009, 373, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Ball, L.M.; Egeler, R.M. Acute GvHD: Pathogenesis and classification. Bone Marrow Transplant. 2008, 41, S58–S64. [Google Scholar] [CrossRef] [PubMed]
- Issa, F.; Wood, K.J. CD4+ regulatory T cells in solid organ transplantation. Curr. Opin. Organ Transplant. 2010, 15, 757–764. [Google Scholar] [CrossRef] [PubMed]
- San Segundo, D.; Millán, O.; Muñoz-Cacho, P.; Boix, F.; Paz-Artal, E.; Talayero, P.; Morales, J.M.; Muro, M.; de Cos, M.Á.; Guirado, L.; et al. High Proportion of Pretransplantation Activated Regulatory T cells (CD4+CD25highCD62L+CD45RO+) Predicts Acute Rejection in Kidney Transplantation. Transplantation 2014, 98, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Lindenberg, M.; Solmaz, G.; Puttur, F.; Sparwasser, T. Mouse Cytomegalovirus infection overrules T regulatory cell suppression on natural killer cells. Virol J. 2014, 9, 145–157. [Google Scholar] [CrossRef]
- Potena, L.; Valantine, H. a Cytomegalovirus-associated allograft rejection in heart transplant patients. Curr. Opin. Infect. Dis. 2007, 20, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Perez-Sola, M.J.; Caston, J.J.; Solana, R.; Rivero, A.; Torre-Cisneros, J. Indirect effects of cytomegalovirus infection in solid organ transplant recipients. Enferm. Infecc. Microbiol. Clin. 2008, 26, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Zorn, E.; Kim, H.T.; Lee, S.J.; Floyd, B.H.; Litsa, D.; Arumugarajah, S.; Bellucci, R.; Alyea, E.P.; Antin, J.H.; Soiffer, R.J.; et al. Reduced frequency of FOXP3 + CD4 + CD25 + regulatory T cells in patients with chronic graft-versus-host disease. 2005, 106, 2903–2911. [Google Scholar]
- Anderson, B.E.; Mcniff, J.M.; Matte, C.; Athanasiadis, I.; Shlomchik, W.D.; Shlomchik, M.J. Recipient CD4 + T cells that survive irradiation regulate chronic graft-versus-host disease. 2004, 104, 1565–1573. [Google Scholar]
- Rieger, K.; Loddenkemper, C.; Maul, J.; Fietz, T.; Wolff, D.; Terpe, H.; Steiner, B.; Berg, E.; Miehlke, S.; Bornha, M.; et al. Mucosal FOXP3 + regulatory T cells are numerically deficient in acute and chronic GvHD. 2006, 107, 1717–1723. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaskula, E.; Dlubek, D.; Tarnowska, A.; Lange, J.; Mordak-Domagala, M.; Suchnicki, K.; Sedzimirska, M.; Borowik, A.; Mizia, S.; Lange, A. Anti-CMV-IgG Positivity of Donors Is Beneficial for alloHSCT Recipients with Respect to the Better Short-Term Immunological Recovery and High Level of CD4+CD25high Lymphocytes. Viruses 2015, 7, 1391-1408. https://doi.org/10.3390/v7031391
Jaskula E, Dlubek D, Tarnowska A, Lange J, Mordak-Domagala M, Suchnicki K, Sedzimirska M, Borowik A, Mizia S, Lange A. Anti-CMV-IgG Positivity of Donors Is Beneficial for alloHSCT Recipients with Respect to the Better Short-Term Immunological Recovery and High Level of CD4+CD25high Lymphocytes. Viruses. 2015; 7(3):1391-1408. https://doi.org/10.3390/v7031391
Chicago/Turabian StyleJaskula, Emilia, Dorota Dlubek, Agnieszka Tarnowska, Janusz Lange, Monika Mordak-Domagala, Krzysztof Suchnicki, Mariola Sedzimirska, Agata Borowik, Sylwia Mizia, and Andrzej Lange. 2015. "Anti-CMV-IgG Positivity of Donors Is Beneficial for alloHSCT Recipients with Respect to the Better Short-Term Immunological Recovery and High Level of CD4+CD25high Lymphocytes" Viruses 7, no. 3: 1391-1408. https://doi.org/10.3390/v7031391
APA StyleJaskula, E., Dlubek, D., Tarnowska, A., Lange, J., Mordak-Domagala, M., Suchnicki, K., Sedzimirska, M., Borowik, A., Mizia, S., & Lange, A. (2015). Anti-CMV-IgG Positivity of Donors Is Beneficial for alloHSCT Recipients with Respect to the Better Short-Term Immunological Recovery and High Level of CD4+CD25high Lymphocytes. Viruses, 7(3), 1391-1408. https://doi.org/10.3390/v7031391







