Silencing of a Germin-Like Protein Gene (CchGLP) in Geminivirus-Resistant Pepper (Capsicum chinense Jacq.) BG-3821 Increases Susceptibility to Single and Mixed Infections by Geminiviruses PHYVV and PepGMV
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Growth
2.2. Construction of Viral Vector for Silencing CchGLP
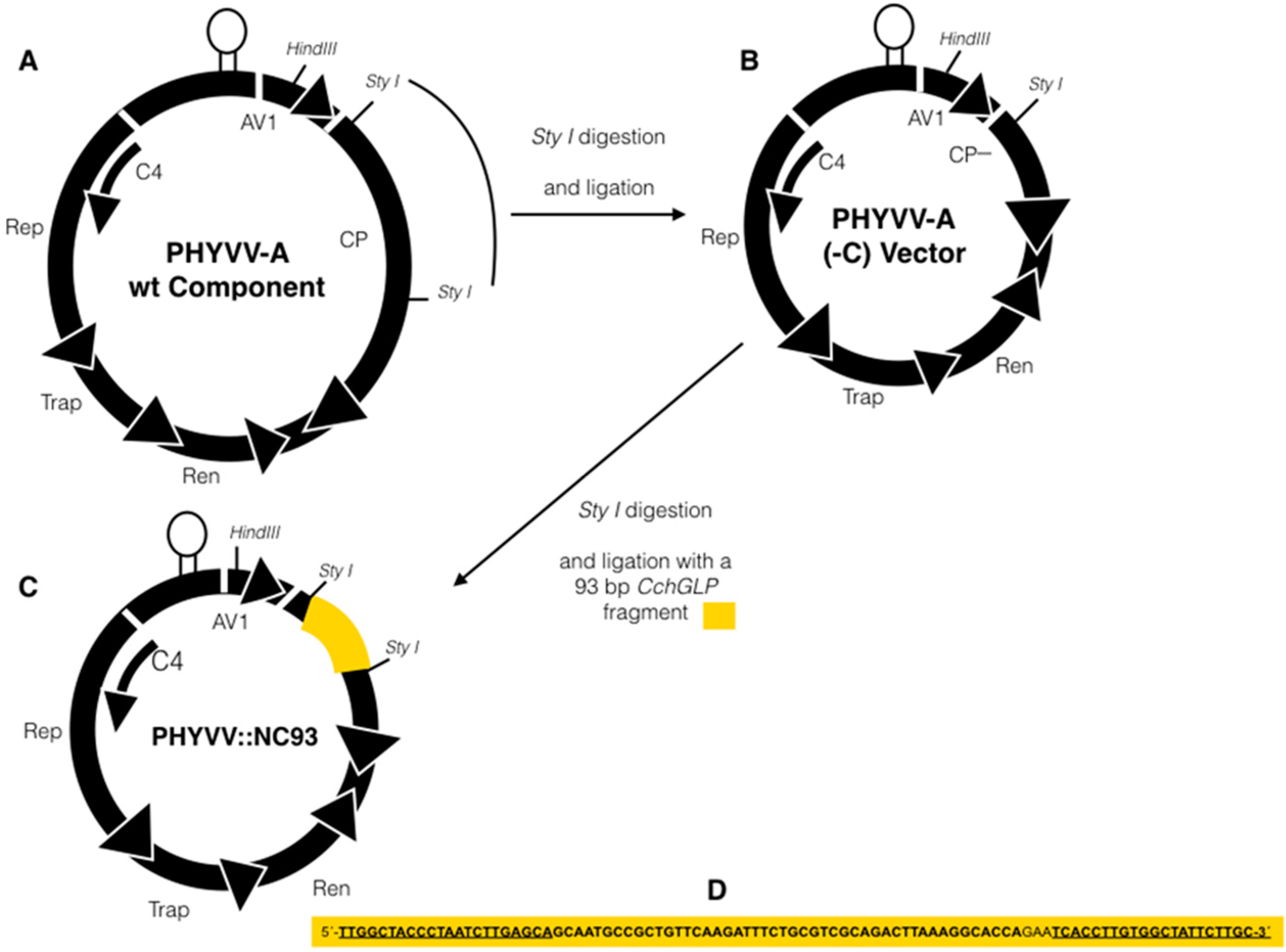
2.3. Inoculation of C. chinense BG-3821 Plants with Silencing Vector and Geminivirus Infections
2.4. Detection of Geminiviruses by PCR
2.5. Assessment CchGLP Gene Silencing in C. chinense BG-3821
2.6. Evaluation of the Phenotypic Response
2.7. Statistical Analysis
3. Results
3.1. Phenotypic Response in C. chinense BG-3821 CchGLP-Silenced Plants



3.2. Analysis of Systemic Silencing of CchGLP

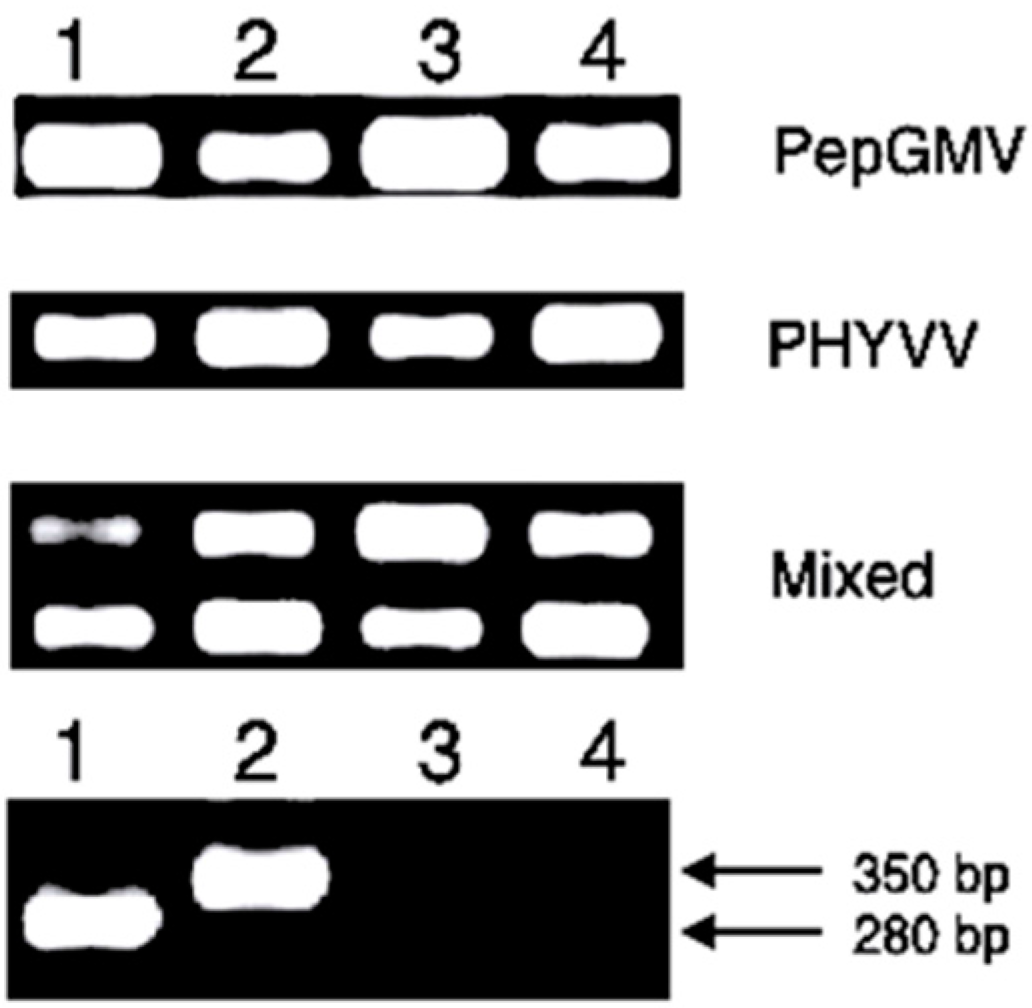
3.3. Severity of Disease Analysis
| Treatment | Severity in CchGLP Silenced C. chinense BG-3821 | Severity in CchGLP Non-Silenced C. chinense BG-3821 Plants | Severity in Susceptible C. chinense Plants (Accession UX-SMH-1) |
|---|---|---|---|
| 30 dpi 90 dpi | 30 dpi 90 dpi | 30 dpi 90 dpi | |
| PHYVV | 3b 5b | 0a 0b | 5c 8b |
| PepGMV | 3b 6a | 0a 1a | 7a 9a |
| Mixed: PHYVV + PepGMV | 4a 6a | 0a 1a | 6b 9a |
| Mock-inoculated (empty viral vector + wt PHYVV component B) | 0c 0c | 0a 0b | 0d 0c |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Góngora-Castillo, E.; Ibarra-Laclette, E.; Diana, L.; Trejo-Saavedra, D.L.; Rivera-Bustamante, R.F. Transcriptome analysis of symptomatic and recovered leaves of geminivirus-infected pepper (Capsicum annuum). Virol. J. 2012, 9, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Hanley-Bowdoin, L.; Bejarano, E.R.; Dominique Robertson, D.; Mansoor, S. Geminiviruses: Masters at redirecting and reprogramming plant processes. Nat. Rev. Microbiol. 2013, 11, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Varsani, A.; Navas-Castillo, J.; Moriones, E.; Hernandez-Zepeda, C.; Idris, A.; Brown, J.K.; Murilo-Zerbini, F.; Martin, D.P. Establishment of three new genera in the family Geminiviridae: Becurtovirus, Eragrovirus and Turncurtovirus. Arch. Virol. 2014, 159, 2193–2203. [Google Scholar] [CrossRef] [PubMed]
- Lugo-Melchor, O.Y.; Guzmán-Uriarte, R.; García-Estrada, R.S.; Félix, J.L. Geminivirus Transmitidos por Mosca Blanca (Bemisia tabaci) en Tomate, en el Valle Agrícola de Culiacán, Sinaloa. Rev. Mex. Fitopatol. 2011, 29, 109–118. [Google Scholar]
- Torres-Pacheco, I.; Garzón-Tiznado, J.A.; Brown, J.K.; Becerra-Flora, A.; Rivera-Bustamante, R.F. Detection and distribution of geminiviruses in Mexico and Southern United States. Phytopathology 1996, 86, 1186–1192. [Google Scholar] [CrossRef]
- De La Torre-Almaráz, R.; Valverde, R.; Méndez-Lozano, J.; Ascencio-Ibañez, J.T.; Rivera-Bustamante, R.F. Caracterización preliminar de un geminivirus en tomate de cáscara (Physalis ixocarpa B.) en la región centro de Mexico. Agrociencia 2002, 36, 471–481. [Google Scholar]
- Guevara-Olvera, L.; Ruíz-Nito, M.L.; Rangel-Cano, R.M.; Torres-Pacheco, I.; Rivera-Bustamante, R.F.; Muñoz-Sánchez, C.I.; González-Chavira, M.M.; Cruz-Hernandez, A.; Guevara-Gonzalez, R.G. Expression of a germin-like protein gene (CchGLP) from a geminivirus-resistant pepper (Capsicum chinense Jacq.) enhances tolerance to geminivirus infection in transgenic tobacco. Physiol. Mol. Plant Pathol. 2012, 78, 45–50. [Google Scholar] [CrossRef]
- Godínez-Hernández, Y.; Anaya-López, J.L.; Díaz-Plaza, R.; González-Chavira, M.; Torres-Pacheco, I.; Rivera-Bustamante, R.F.; Guevara-González, R.G. Characterization of resistance to pepper huasteco geminivirus in chili peppers from Yucatán, Mexico. Hortscience 2001, 36, 139–142. [Google Scholar]
- Anaya-López, J.L.; Godínez-Hernández, Y.; Muñoz-Sánchez, C.I.; Guevara-Olvera, L.; Guevara-González, R.G.; Rivera-Bustamante, R.F.; González-Chavira, M.M.; Torres-Pacheco, I. Identification of resistance to single and mixed infections of pepper golden mosaic virus PepGMV and the Huasteco pepper virus in chilli peppers (Capsicum chinense Jaq.). Rev. Chapingo Ser. Hortic. 2003, 9, 225–234. [Google Scholar]
- Anaya-López, J.L.; González-Chavira, M.; Pons-Hernández, J.L.; Garzón-Tiznado, J.A.; Torres-Pacheco, I.; Rivera-Bustamante, R.F.; Hernández-Verdugo, S.; Guevara-González, R.G.; Muñoz-Sánchez, C.I.; Guevara-Olvera, L. Resistance to geminivirus mixed infections in Mexican wild peppers. Horscience 2003, 38, 251–255. [Google Scholar]
- Anaya-López, J.L.; Pérez-Mora, E.; Torres-Pacheco, I.; Muñoz-Sánchez, C.I.; Guevara-Olvera, L.; González-Chavira, M.M.; Ochoa-Alejo, N.; Rivera-Bustamante, R.F.; Guevara-González, R.G. Inducible gene expresión by Pepper huasteco virus in Capsicum chinense plants with resistance to geminivirus infections. Can. J. Plant Pathol. 2005, 27, 276–282. [Google Scholar] [CrossRef]
- Gasca-González, M.R.; Rivera-Herrera, Y.; Torres-Pacheco, I.; González-Chavira, M.M.; Guevara-Olvera, L.; Muñoz-Sánchez, C.I.; Guevara-González, R.G. Study on the transcripto in Capsicum chinense Jacq. resistant to Pepper Huasteca Yellow Vein Virus (PHYVV). Agrociencia 2008, 42, 107–117. [Google Scholar]
- García-Neria, M.A.; Rivera-Bustamante, R.F. Characterization of geminivirus resistance in an accession of Capsicum chinense Jacq. Mol. Plant-Microbe Int. 2011, 24, 172–182. [Google Scholar] [CrossRef]
- Barrera-Pacheco, A.; Joaquín-Ramos, A.; Torres-Pacheco, I.; Gonzalez-Chavira, M.; Perez-Perez, C.; Guevara-Olvera, L.; Guevara-Gonzalez, R.G. Analysis of transcriptional expression induced in Capsicum chinense BG-3821 under conditions of biotic and abiotic stress. Agrociencia 2008, 42, 95–106. [Google Scholar]
- León-Galvan, F.; de Jesus Joaquin-Ramos, A.; Torres-Pacheco, I.; Barba de la Rosa, A.P.; Guevara-Olvera, L.; Gonzalez-Chavira, M.M.; Ocampo-Velozquez, R.V.; Rico-García, E.; Guevara-González, R.G. A Germin-like protein gene (CchGLP) of Capsicum chinense Jacq. is induced during incompatible interactions and display Mn-superoxide dismutase activity. Int. J. Mol. Sci. 2011, 12, 7301–7313. [Google Scholar] [CrossRef] [PubMed]
- Dunwell, J.; Gibbings, J.G.; Mahmood, T.; Saqlan Naqvi, S.M. Germin and Germin-like Proteins: Evolution, Structure, and Function. Crit. Rev. Plant Sci. 2008, 27, 342–375. [Google Scholar] [CrossRef]
- Mejía-Teniente, L.; Torres-Pacheco, I.; González-Chavira, M.M.; Ocampo-Velázquez, R.V.; Herrera-Ruiz, G.; Chapa-Oliver, A.M.; Guevara-González, R.G. Use of elicitors as an approach for sustainable agriculture. Afr. J. Biotechnol. 2010, 9, 9155–9162. [Google Scholar]
- Mejía-Teniente, L.; Durán-Flores, F.D.; Chapa-Oliver, A.M.; Torres-Pacheco, I.; Cruz-Hernández, A.; González-Chavira, M.M.; Ocampo-Velozquez, R.V.; Guevara-González, R.G. Oxidative and molecular responses in Capsicum annuum L. after hydrogen peroxide, salicylic acid and chitosan foliar applications. Int. J. Mol. Sci. 2013, 14, 10178–10196. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.; Saha, J.; Sengupta, A.; Gupta, K. Recent advances on Virus Induced Gene silencing (VIGS): Plant functional genomics. Plant Biochem. Physiol. 2013, 1, 1–2. [Google Scholar] [CrossRef]
- Carrillo-Tripp, J.; Shimada-Beltrán, H.; Rivera-Bustamante, R.F. Use of geminiviral vectors for functional genomics. Curr. Opin. Plant Biol. 2006, 9, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Godge, M.R.; Purkayastha, A.; Dasgupta, I.; Kumar, P. Virus-Induced gene silencing for functional analysis of selected genes. Plant Cell Rep. 2008, 27, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, O.; Bartholmes, C.; Gleisberg, S. Virus-induced gene silencing (VIGS) in Cysticapnos vesicaria, a zygomorphic-flowered Papaveraceae (Ranunculales, basal eudicots). Ann. Bot. 2012, 109, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, N.; Yoshikawa, N. Virus-induced gene silencing in soybean seeds and the emergence stage of soybean plants with apple latent spherical virus vectors. Plant Mol. Biol. 2009, 71, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Purkayastha, A.; Mathur, S.; Verma, V.; Sharma, S.; Dasgupta, I. Virus-induced gene silencing in rice using a vector derived from DNA virus. Planta 2010, 232, 1531–1540. [Google Scholar] [CrossRef] [PubMed]
- Turnage, M.A.; Muangsan, N.; Peele, C.G.; robertson, D. Geminivirus-based vectors for gene silencing in Arabidopsis. Plant J. 2002, 30, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Muangsan, N.; Beclin, C.; Vaucheret, H.; Robertson, D. Geminivirus VIGS of endogenous genes requires SGS2/SDE1 and SGS3 and defines a new branch in the genetic pathway for silencing in plants. Plant J. 2004, 38, 1004–1014. [Google Scholar] [CrossRef] [PubMed]
- Abraham-Juarez, M.R.; Rocha-Granados, M.C.; Lopez, M.G.; Rivera-Bustamante, R.F.; Ochoa-Alejo, N. Virus-induced silencing of Comt, pAmt and Kas genes results in a reduction of capsaicinoid accumulation in chili pepper fruits. Planta 2008, 227, 681–695. [Google Scholar] [CrossRef] [PubMed]
- pCR® 2.1-TOPO® TA Vector—Subcloning Vector (Invitrogen, Carlsbad, CA, USA). Available online: https://www.lifetechnologies.com.
- Dellaporta, S.L.; Wood, J.; Hicks, J.B. A plant DNA minipreparation: Version II. Plant Mol. Biol. Report. 1983, 1, 19–21. [Google Scholar] [CrossRef]
- SAS Institute. SAS/STAT User’s Guide, Version 6, 4th ed.; SAS Institute: Cary, NC, USA, 1990. [Google Scholar]
- Park, C.-J.; An, J.-M.; Shin, Y.-C.; Kim, K.-J.; Lee, B.-J.; Paek, K.-H. Molecular characterization of pepper germin-like protein as the novel PR-16 family of pathogenesis-related proteins isolated during the resistance response to viral and bacterial infection. Planta 2004, 219, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.; Baldwin, I.T. Silencing of a germin-like gene in Nicotiana attenuata improves performance of native herbivores. Plant Physiol. 2006, 140, 1126–1136. [Google Scholar] [CrossRef] [PubMed]
- Saucedo-García, M.; González-Solís, A.; Rodriguez-Mejía, P.; Olivera-Flores, T.J.; Vázquez-Santana, S.; Cahoon, E.B.; Gavilanes-Ruiz, M. Reactive oxygen species as transducers of sphinganine-mediated cell death pathway. Plant Signal. Behav. 2011, 6, 1616–1619. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mejía-Teniente, L.; Joaquin-Ramos, A.D.J.; Torres-Pacheco, I.; Rivera-Bustamante, R.F.; Guevara-Olvera, L.; Rico-García, E.; Guevara-Gonzalez, R.G. Silencing of a Germin-Like Protein Gene (CchGLP) in Geminivirus-Resistant Pepper (Capsicum chinense Jacq.) BG-3821 Increases Susceptibility to Single and Mixed Infections by Geminiviruses PHYVV and PepGMV. Viruses 2015, 7, 6141-6151. https://doi.org/10.3390/v7122930
Mejía-Teniente L, Joaquin-Ramos ADJ, Torres-Pacheco I, Rivera-Bustamante RF, Guevara-Olvera L, Rico-García E, Guevara-Gonzalez RG. Silencing of a Germin-Like Protein Gene (CchGLP) in Geminivirus-Resistant Pepper (Capsicum chinense Jacq.) BG-3821 Increases Susceptibility to Single and Mixed Infections by Geminiviruses PHYVV and PepGMV. Viruses. 2015; 7(12):6141-6151. https://doi.org/10.3390/v7122930
Chicago/Turabian StyleMejía-Teniente, Laura, Ahuizolt De Jesús Joaquin-Ramos, Irineo Torres-Pacheco, Rafael F. Rivera-Bustamante, Lorenzo Guevara-Olvera, Enrique Rico-García, and Ramon G. Guevara-Gonzalez. 2015. "Silencing of a Germin-Like Protein Gene (CchGLP) in Geminivirus-Resistant Pepper (Capsicum chinense Jacq.) BG-3821 Increases Susceptibility to Single and Mixed Infections by Geminiviruses PHYVV and PepGMV" Viruses 7, no. 12: 6141-6151. https://doi.org/10.3390/v7122930
APA StyleMejía-Teniente, L., Joaquin-Ramos, A. D. J., Torres-Pacheco, I., Rivera-Bustamante, R. F., Guevara-Olvera, L., Rico-García, E., & Guevara-Gonzalez, R. G. (2015). Silencing of a Germin-Like Protein Gene (CchGLP) in Geminivirus-Resistant Pepper (Capsicum chinense Jacq.) BG-3821 Increases Susceptibility to Single and Mixed Infections by Geminiviruses PHYVV and PepGMV. Viruses, 7(12), 6141-6151. https://doi.org/10.3390/v7122930






