Herpes Simplex Virus 1 Us3 Deletion Mutant is Infective Despite Impaired Capsid Translocation to the Cytoplasm
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
2.2. Virus Replication Assays
2.3. Cryo-Fixation for Transmission Electron Microscopy
2.4. Determination of Nuclear and Cell Volume
2.5. Phenotype Distribution
2.6. Cryo-Field Emission Scanning Electron Microscopy (cryo-FESEM)
3. Results
3.1. Budding of R7041(∆Us3) Capsids at the Inner Nuclear Membrane
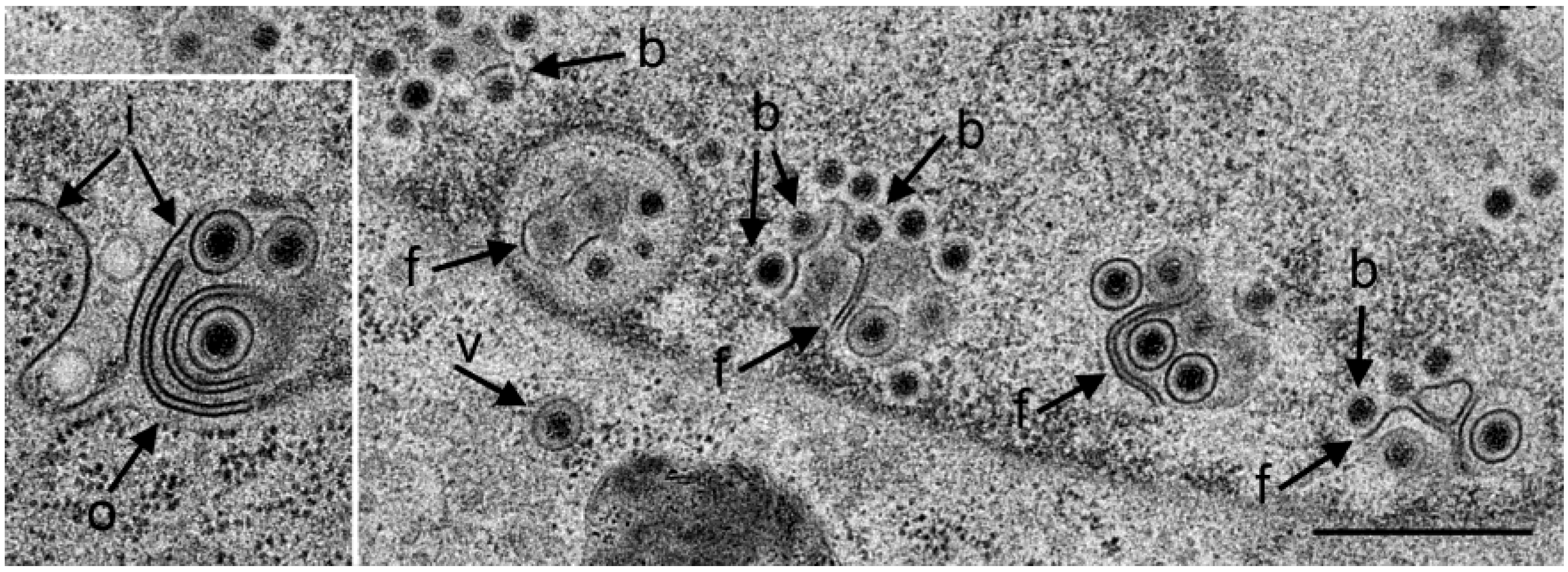
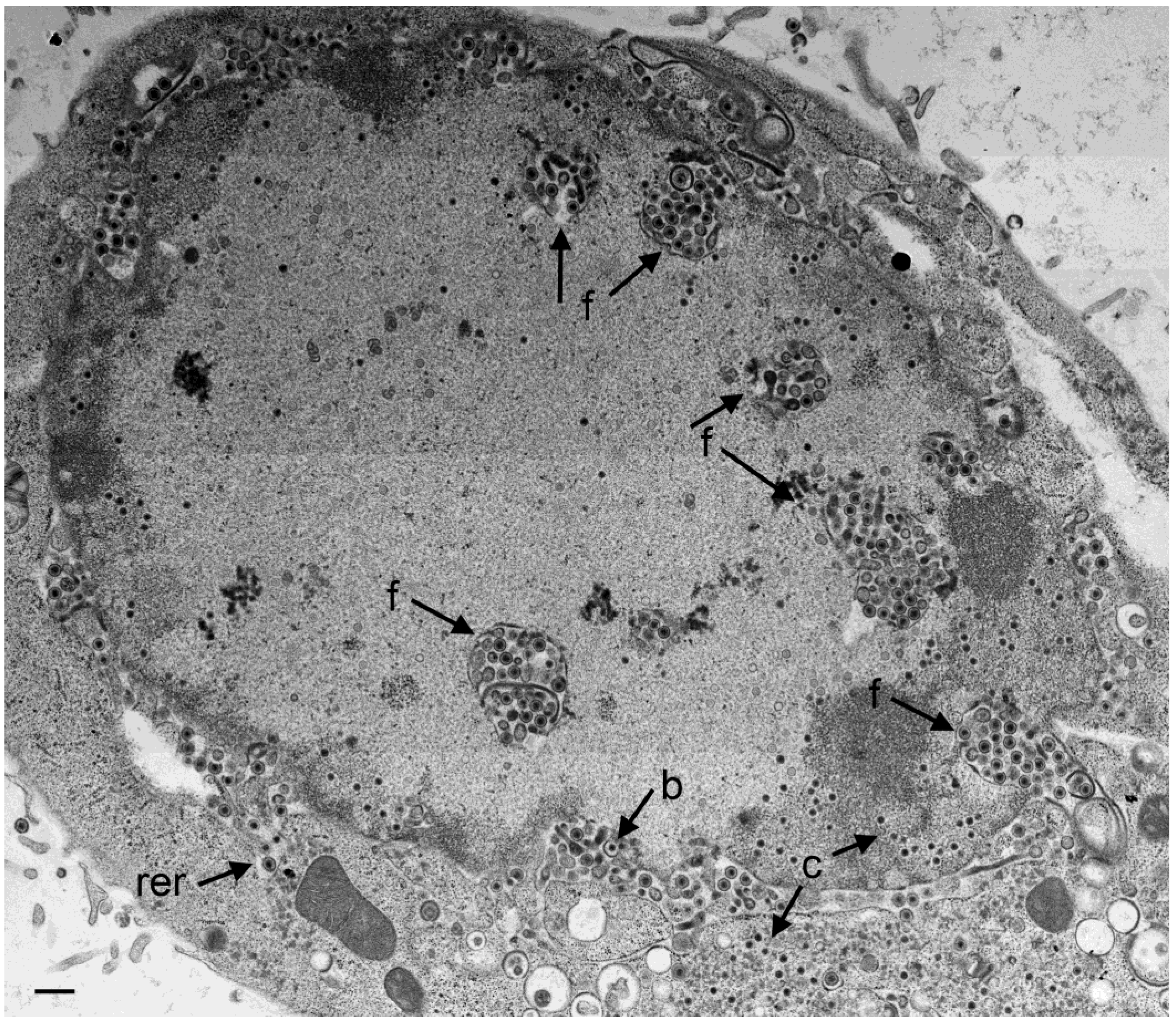
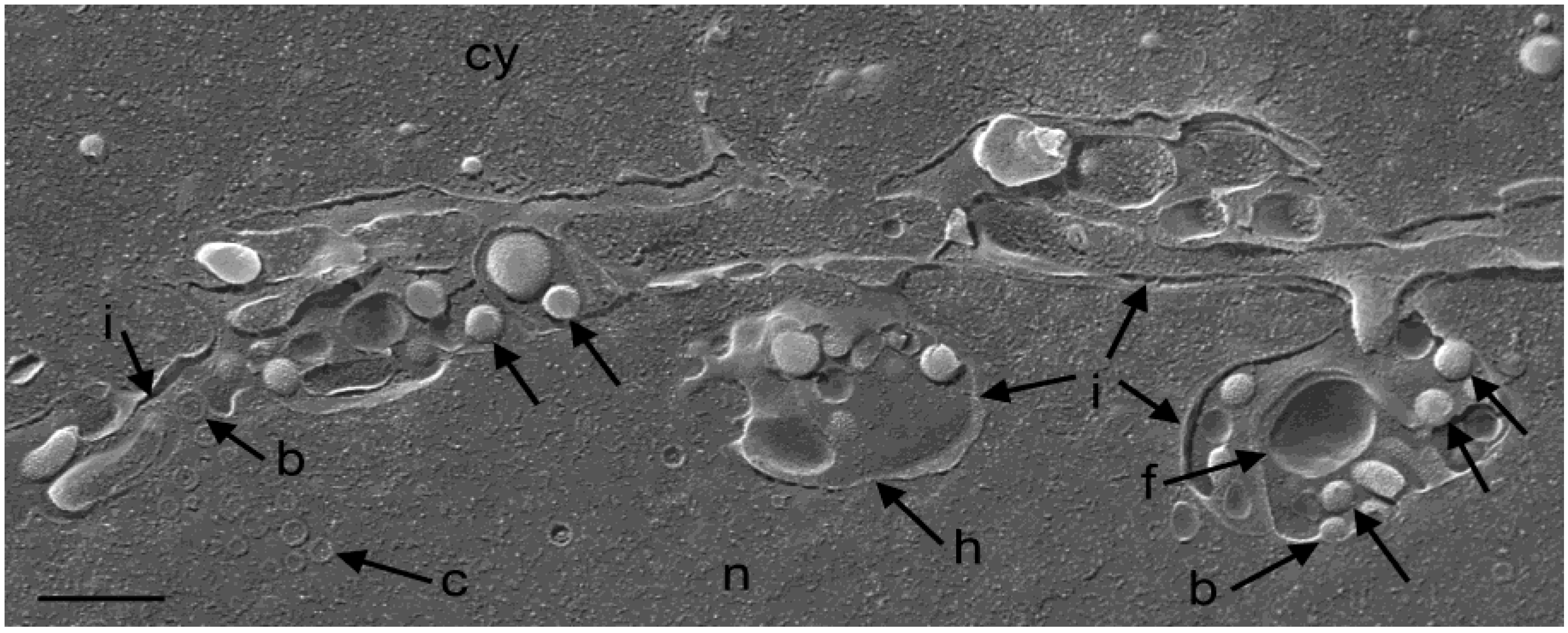
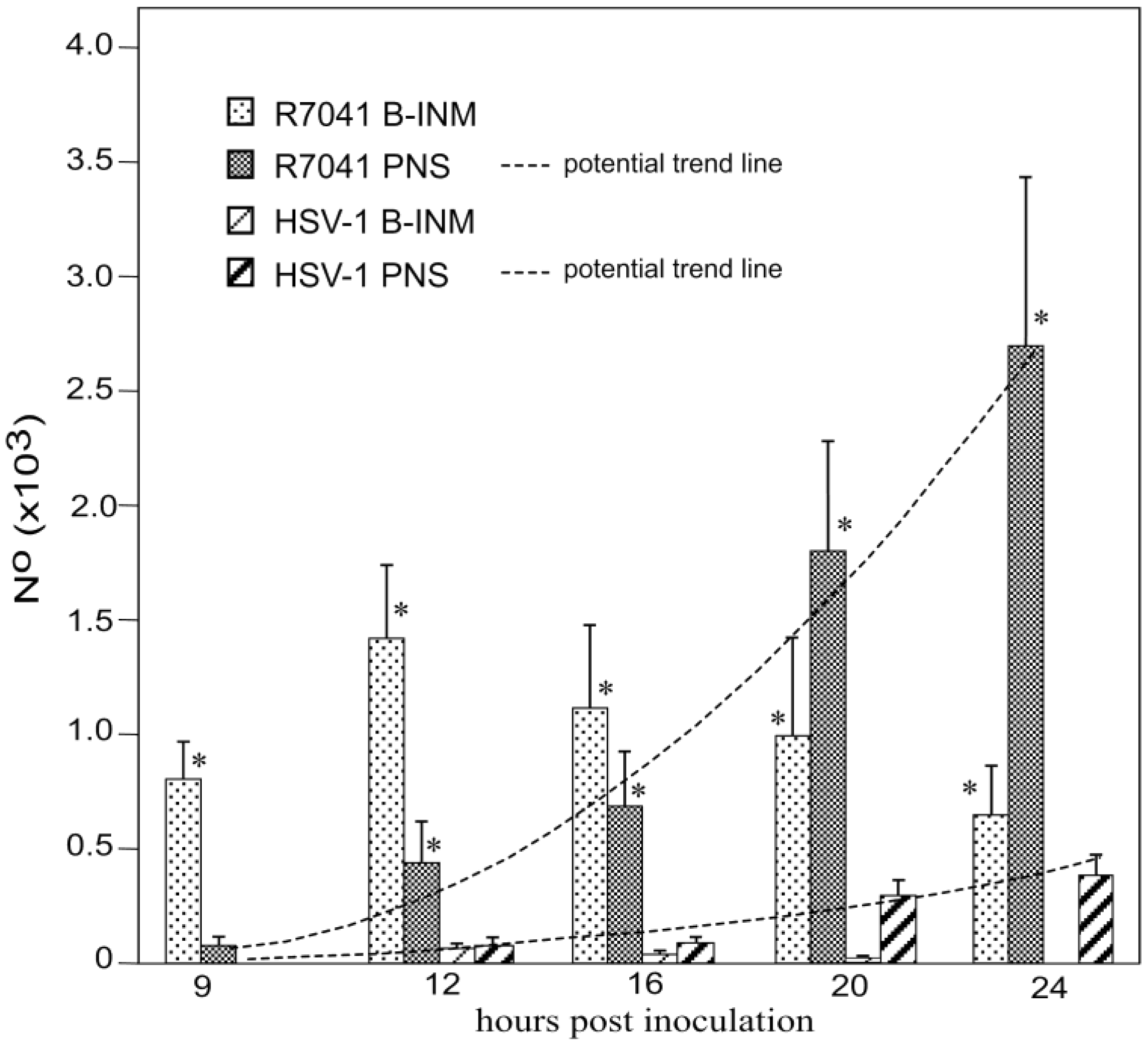
3.2. Retention of R7041(∆Us3) Virions in the PNS
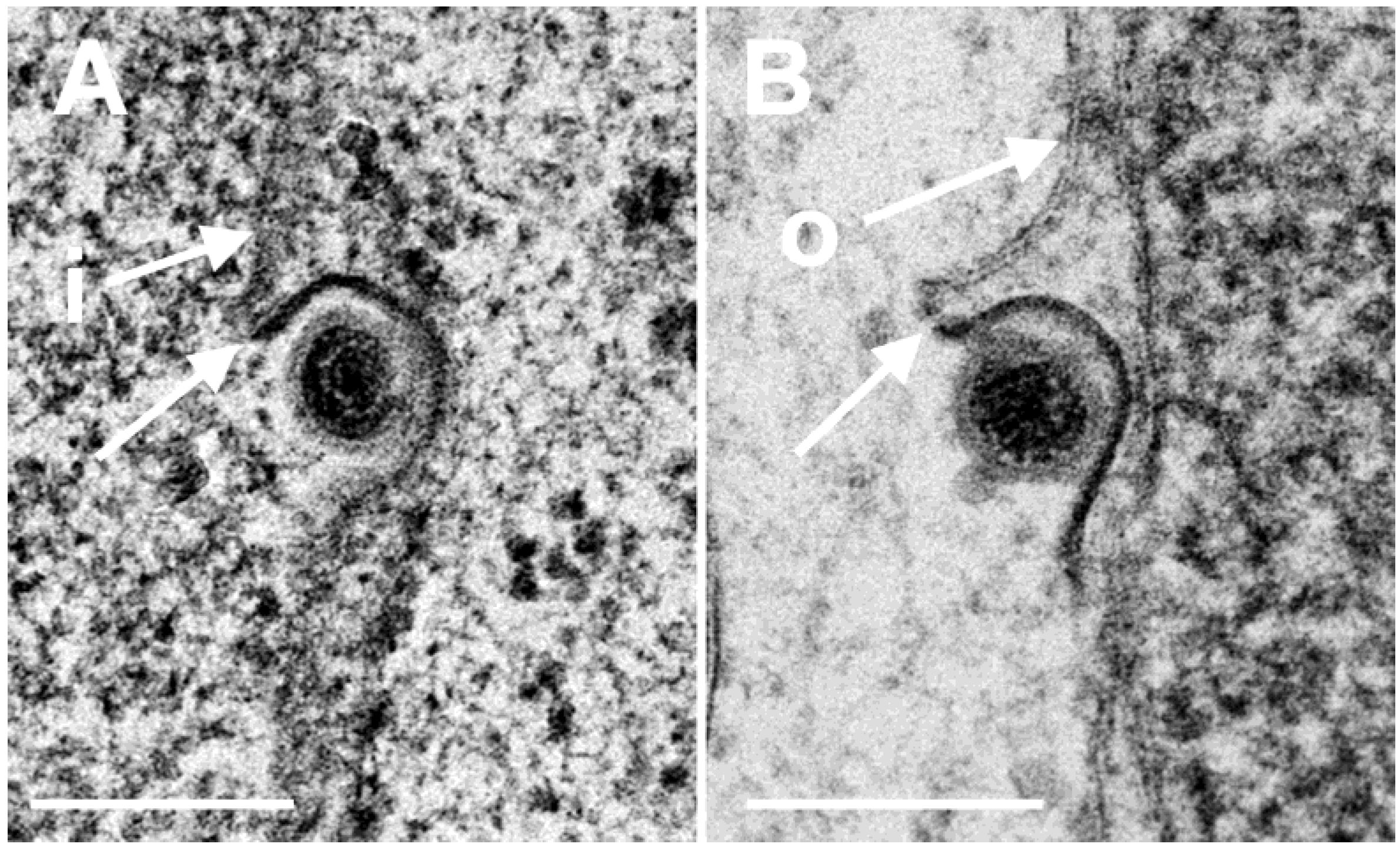
3.3. Interaction of Capsids with the Outer Nuclear Membrane
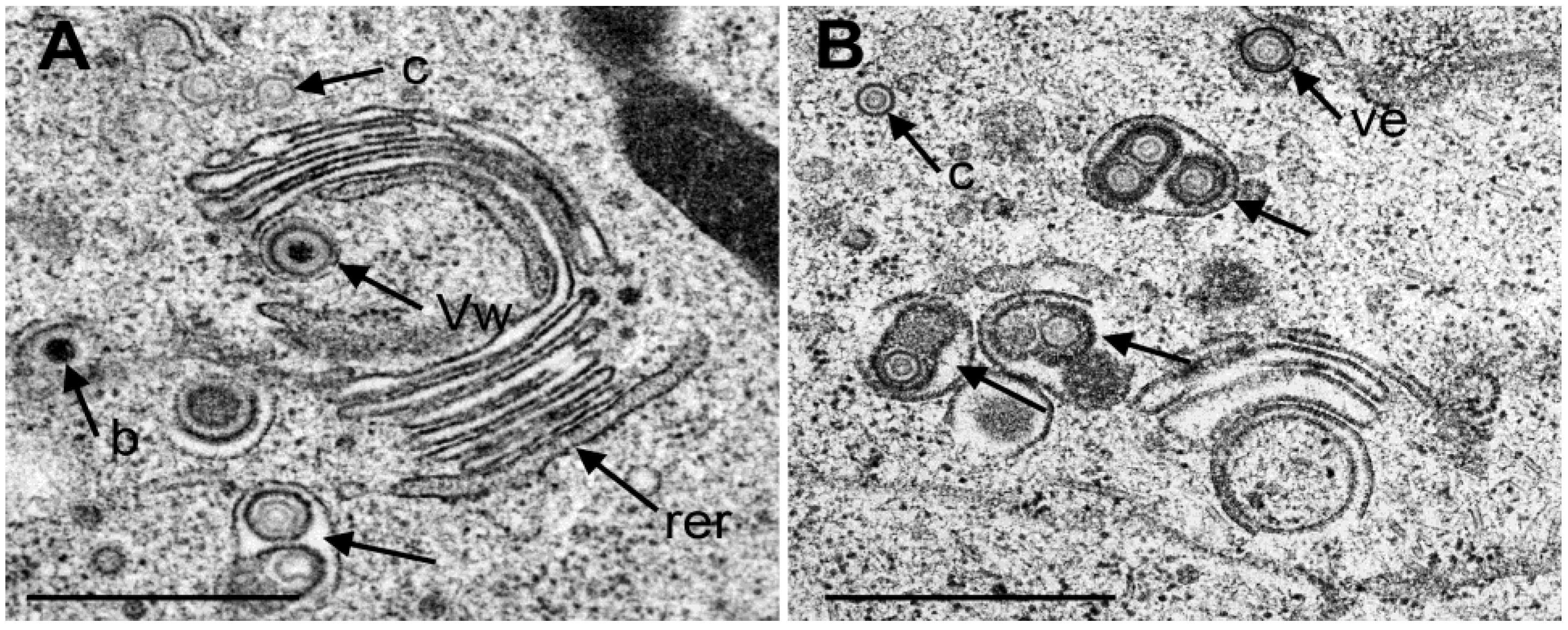
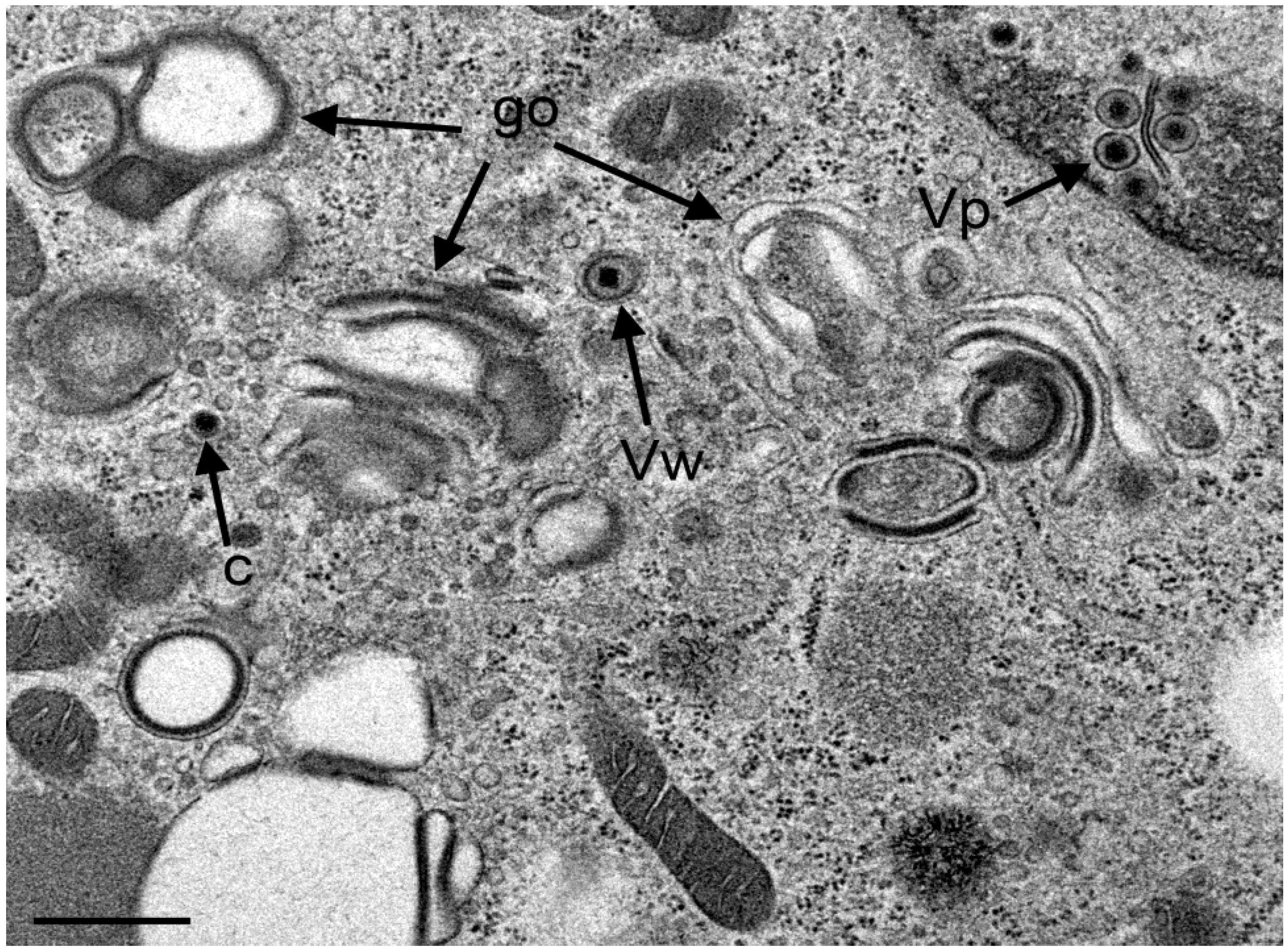
3.4. Envelopment of R7041(∆US3) Capsids at the Golgi Complex
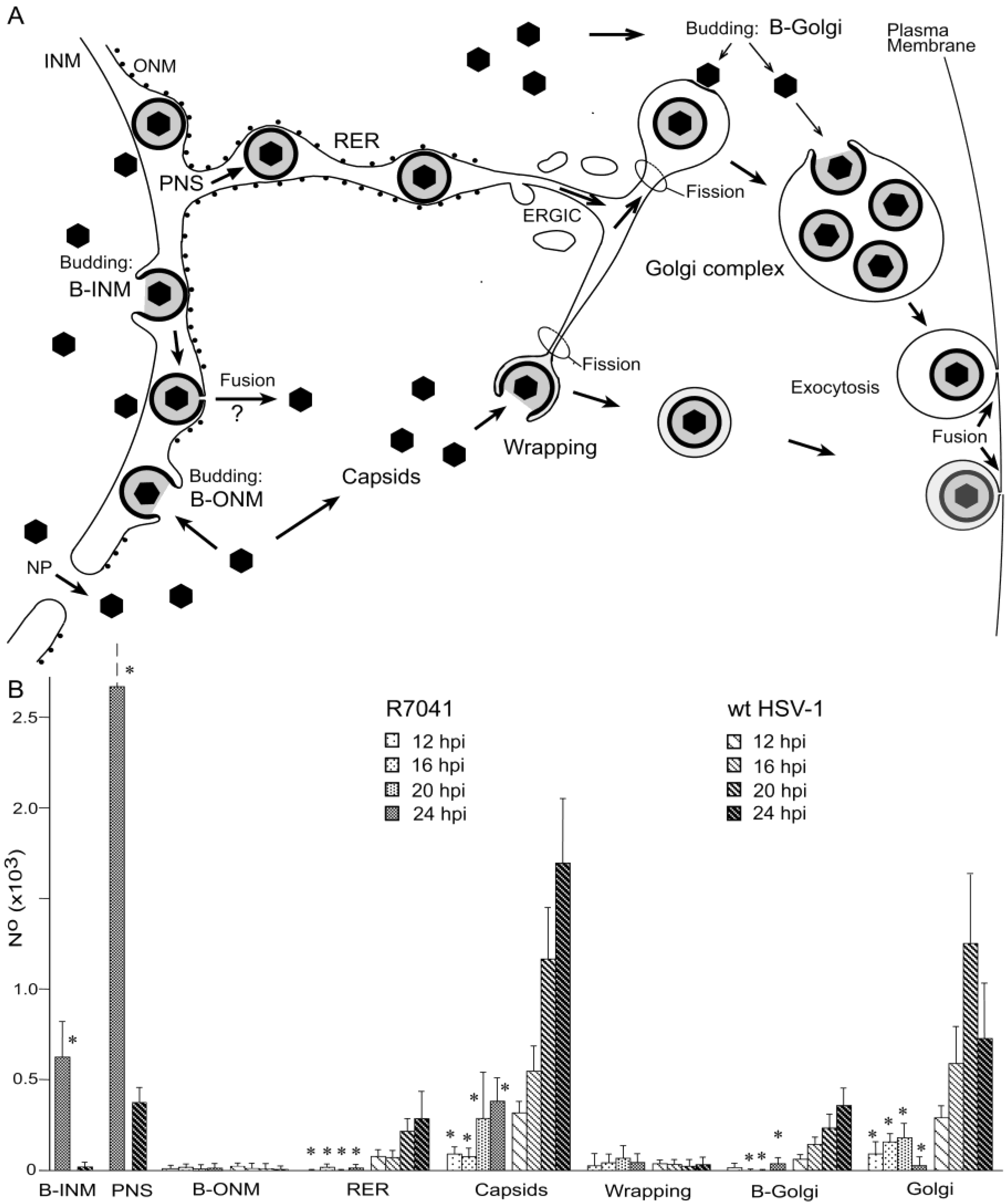

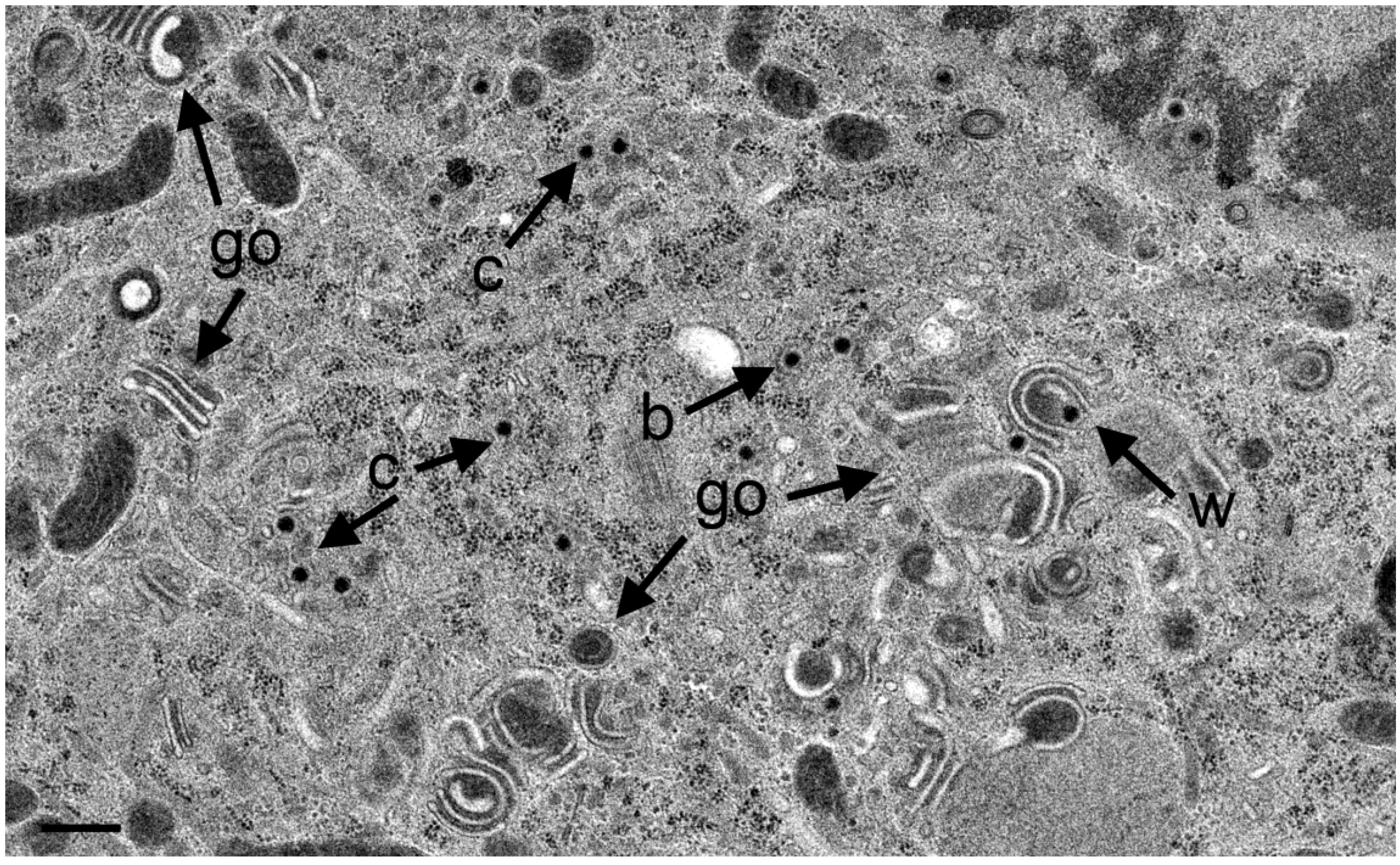
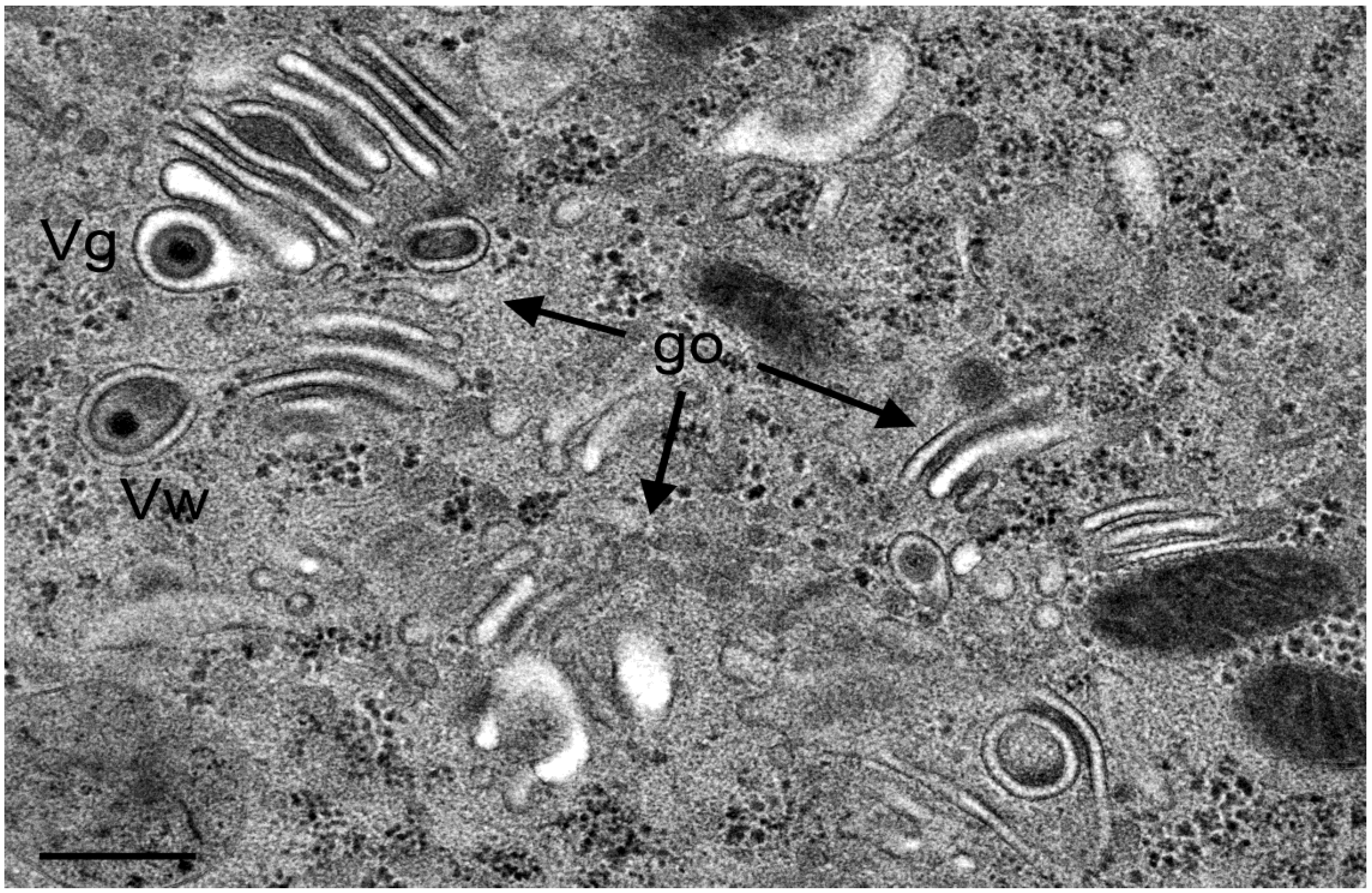
3.5. Infectious Progeny R7041(∆Us3) Virus
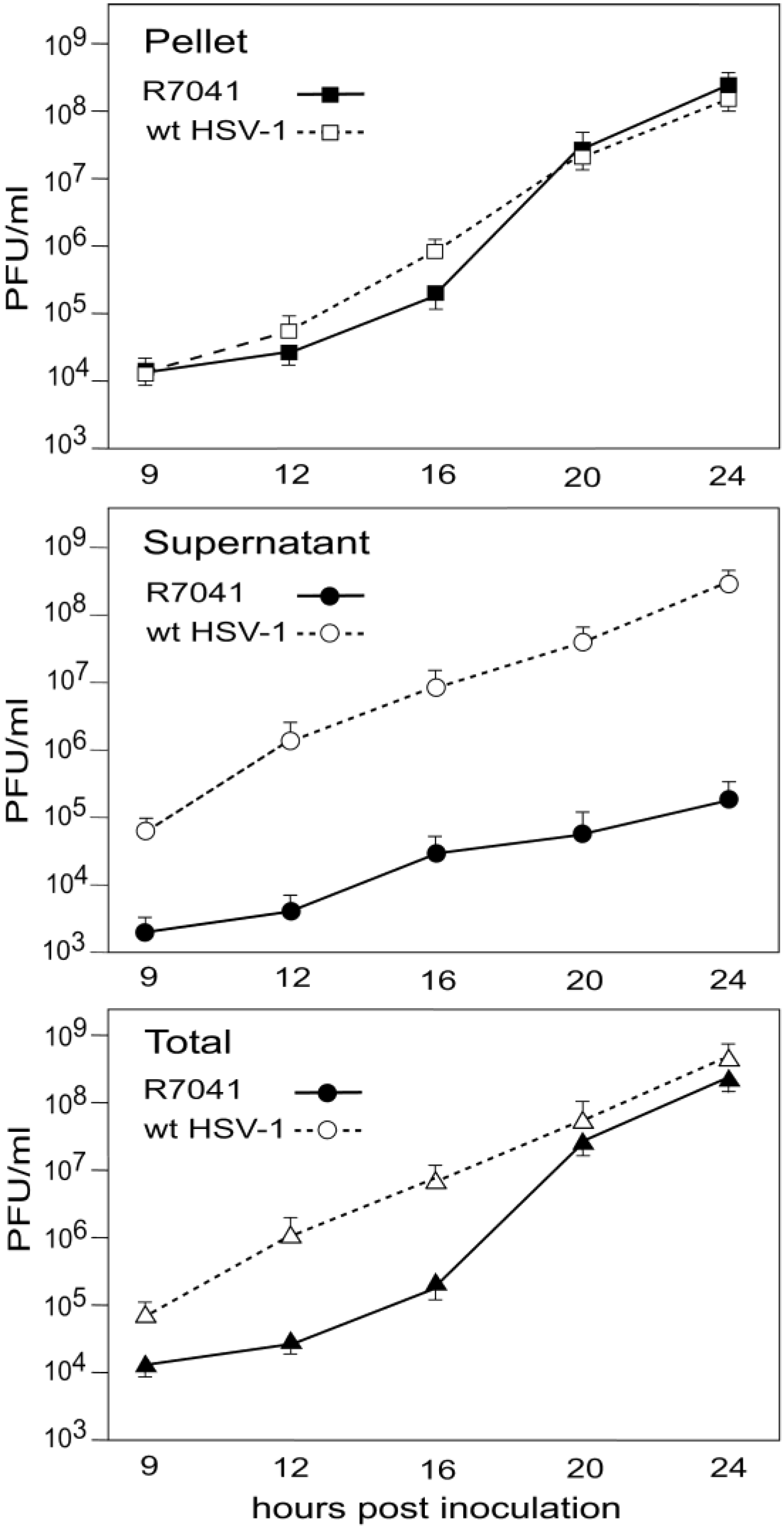
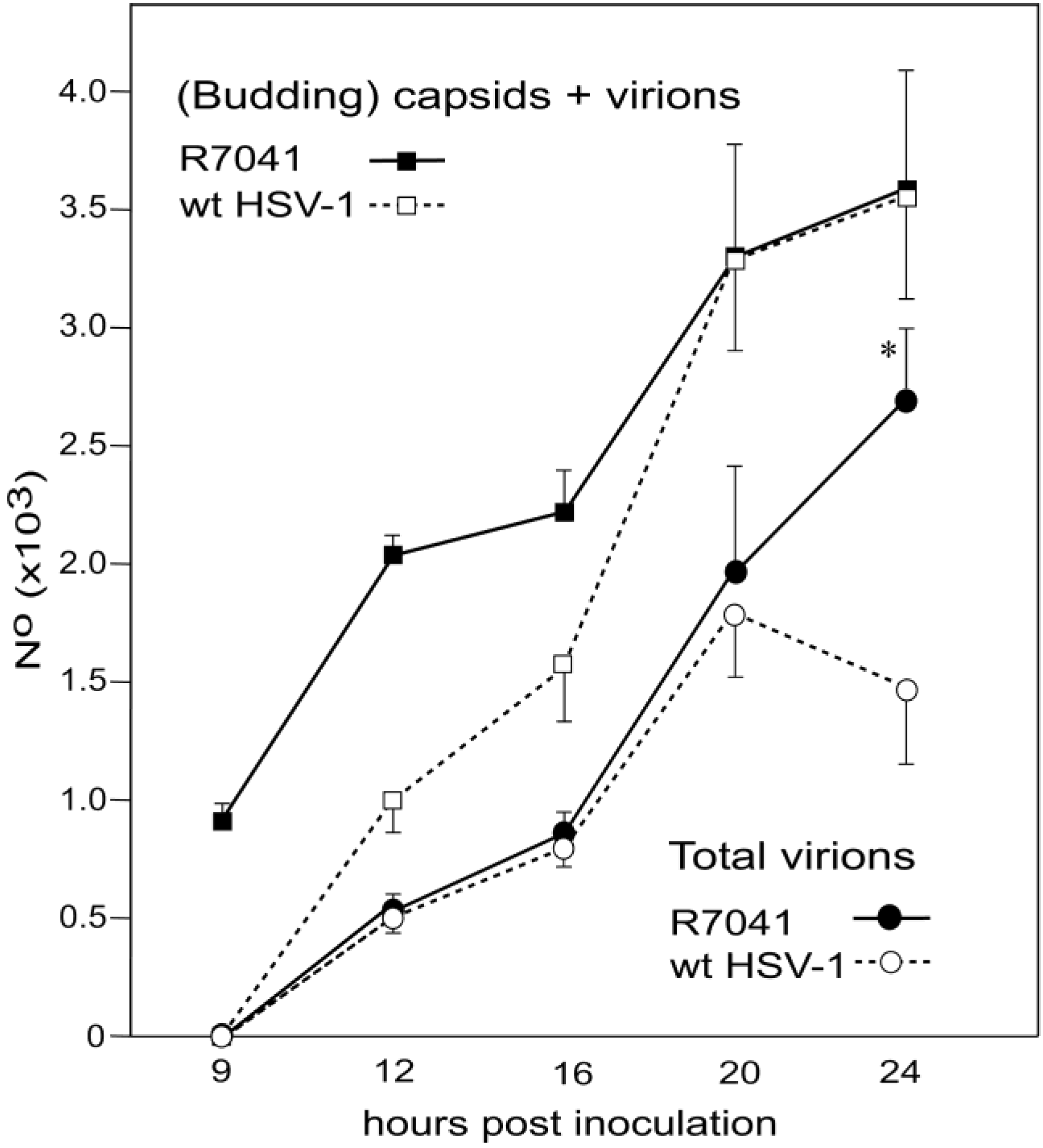
4. Discussion
4.1. Virions Derived by Budding at the INM
4.2. Virus Translocation to the Cytoplasm
4.3. Conclusion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roizman, B.; Knipe, D.M.; Whitley, R.J. Herpes simplex viruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Wolters Kluver/Lipincott Wiliams & Wilkins: Philadelphia, PA, USA, 2014; pp. 1823–1897. [Google Scholar]
- Quinlan, M.P.; Chen, L.B.; Knipe, D.M. The intranuclear location of a herpes simplex virus DNA-binding protein is determined by the status of viral DNA replication. Cell 1984, 36, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Granzow, H. Egress of alphaherpesviruses: Comparative ultrastructural study. J. Virol. 2001, 75, 3675–3684. [Google Scholar] [CrossRef] [PubMed]
- Skepper, J.N.; Whiteley, A.; Browne, H.; Minson, A. Herpes simplex virus nucleocapsids mature to progeny virions by an envelopment --> deenvelopment --> reenvelopment pathway. J. Virol. 2001, 75, 5697–5702. [Google Scholar] [CrossRef] [PubMed]
- Stackpole, C.W. Herpes-type virus of the frog renal adenocarcinoma. I. Virus development in tumor transplants maintained at low temperature. J. Virol. 1969, 4, 75–93. [Google Scholar] [PubMed]
- Klupp, B.G.; Granzow, H.; Mettenleiter, T.C. Nuclear envelope breakdown can substitute for primary envelopment-mediated nuclear egress of herpesviruses. J. Virol. 2011, 85, 8285–8292. [Google Scholar] [PubMed]
- Gilbert, R.; Ghosh, K.; Rasile, L.; Ghosh, H.P. Membrane anchoring domain of herpes simplex virus glycoprotein gB is sufficient for nuclear envelope localization. J. Virol. 1994, 68, 2272–2285. [Google Scholar] [PubMed]
- Granzow, H.; Weiland, F.; Jons, A.; Klupp, B.G.; Karger, A.; Mettenleiter, T.C. Ultrastructural analysis of the replication cycle of pseudorabies virus in cell culture: A reassessment. J. Virol. 1997, 71, 2072–2082. [Google Scholar] [PubMed]
- Radsak, K.; Eickmann, M.; Mockenhaupt, T.; Bogner, E.; Kern, H.; Eis-Hübinger, A.; Reschke, M. Retrieval of human cytomegalovirus glycoprotein B from the infected cell surface for virus envelopment. Arch. Virol. 1996, 141, 557–572. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Roizman, B. Concerning the egress of herpes simplex virus from infected cells: Electron and light microscope observations. Virology 1969, 38, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Stannard, L.M.; Himmelhoch, S.; Wynchank, S. Intra-nuclear localization of two envelope proteins, gB and gD, of herpes simplex virus. Arch. Virol. 1996, 141, 505–524. [Google Scholar] [CrossRef] [PubMed]
- Whealy, M.E.; Card, J.P.; Meade, R.P.; Robbins, A.K.; Enquist, L.W. Effect of brefeldin A on alphaherpesvirus membrane protein glycosylation and virus egress. J. Virol. 1991, 65, 1066–1081. [Google Scholar] [PubMed]
- Wild, P.; Schraner, E.M.; Cantieni, D.; Loepfe, E.; Walther, P.; Müller, M.; Engels, M. The significance of the Golgi complex in envelopment of bovine herpesvirus 1 (BHV-1) as revealed by cryobased electron microscopy. Micron 2002, 33, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Leuzinger, H.; Ziegler, U.; Schraner, E.M.; Fraefel, C.; Glauser, D.L.; Heid, I.; Ackermann, M.; Mueller, M.; Wild, P. Herpes simplex virus 1 envelopment follows two diverse pathways. J. Virol. 2005, 79, 13047–13059. [Google Scholar] [CrossRef] [PubMed]
- Farnsworth, A.; Wisner, T.W.; Webb, M.; Roller, R.; Cohen, G.; Eisenberg, R.; Johnson, D.C. Herpes simplex virus glycoproteins gB and gH function in fusion between the virion envelope and the outer nuclear membrane. Proc. Natl. Acad. Sci. USA 2007, 104, 10187–10192. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.E.; Wills, E.G.; Roller, R.J.; Ryckman, B.J.; Baines, J.D. Ultrastructural localization of the herpes simplex virus type 1 UL31, UL34, and US3 proteins suggests specific roles in primary envelopment and egress of nucleocapsids. J. Virol. 2002, 76, 8939–8952. [Google Scholar] [CrossRef] [PubMed]
- Poon, A.P.; Benetti, L.; Roizman, B. U(S)3 and U(S)3.5 protein kinases of herpes simplex virus 1 differ with respect to their functions in blocking apoptosis and in virion maturation and egress. J. Virol. 2006, 80, 3752–3764. [Google Scholar] [CrossRef] [PubMed]
- Wisner, T.W.; Wright, C.C.; Kato, A.; Kawaguchi, Y.; Mou, F.; Baines, J.D.; Roller, R.J.; Johnson, D.C. Herpesvirus gB-induced fusion between the virion envelope and outer nuclear membrane during virus egress is regulated by the viral US3 kinase. J. Virol. 2009, 83, 3115–3126. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.; Bruun, B.; Minson, T.; Browne, H. Glycoproteins gB, gD, and gHgL of herpes simplex virus type 1 are necessary and sufficient to mediate membrane fusion in a Cos cell transfection system. J. Virol. 1998, 72, 873–875. [Google Scholar] [PubMed]
- Ryckman, B.J.; Roller, R.J. Herpes simplex virus type 1 primary envelopment: UL34 protein modification and the US3-UL34 catalytic relationship. J. Virol. 2004, 78, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Purves, F.C.; Longnecker, R.M.; Leader, D.P.; Roizman, B. Herpes simplex virus 1 protein kinase is encoded by open reading frame US3 which is not essential for virus growth in cell culture. J. Virol. 1987, 61, 2896–2901. [Google Scholar] [PubMed]
- Purves, F.C.; Spector, D.; Roizman, B. The herpes simplex virus 1 protein kinase encoded by the US3 gene mediates posttranslational modification of the phosphoprotein encoded by the UL34 gene. J. Virol. 1991, 65, 5757–5764. [Google Scholar] [PubMed]
- Weibull, C.; Christiansson, A. Extraction of proteins and membrane lipids during low temperature embedding of biological material for electron microscopy. J. Microsc. 1986, 142, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M. The integrating power of cryofixation-based electron microscopy in biology. Acta Microsc. 1992, 1, 37–46. [Google Scholar]
- Harreveld, A.V.; Fifkova, E. Rapid freezing of deep cerebral structures for electron microscopy. Anat. Rec. 1975, 182, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Hawes, P.; Netherton, C.L.; Mueller, M.; Wileman, T.; Monaghan, P. Rapid freeze-substitution preserves membranes in high-pressure frozen tissue culture cells. J. Microsc. 2007, 226, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Wild, P. Electron Microscopy of Viruses and Virus-Cell Interactions. In Introduction to Electron Microscopy for Biologists; Methods in Cell Biology; Allen, T., Ed.; Elsevier: San Diego, USA, 2008; pp. 497–524. [Google Scholar]
- Wild, P.; Schraner, E.M.; Adler, H.; Humbel, B.M. Enhanced resolution of membranes in cultured cells by cryoimmobilization and freeze-substitution. Microsc. Res. Tech. 2001, 53, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Sutter, E.; de Oliveira, A.P.; Tobler, K.; Schraner, E.M.; Sonda, S.; Kaech, A.; Lucas, M.S.; Ackermann, M.; Wild, P. Herpes simplex virus 1 induces de novo phospholipid synthesis. Virology 2012, 429, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Weibel, E. Stereological methods. In Practical Methods for Biological Morphometry; Academic Press: London, UK, 1979; Volume 1. [Google Scholar]
- Zhou, Z.H.; Chiu, W.; Haskell, K.; Spears, H., Jr.; Jakana, J.; Rixon, F.J.; Scott, L.R. Refinement of herpesvirus B-capsid structure on parallel supercomputers. Biophys. J. 1998, 74, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, K.; Desai, P.; Winkler, D.C.; Heymann, J.B.; Belnap, D.M.; Baumeister, W.; Steven, A.C. Three-dimensional structure of herpes simplex virus from cryo-electron tomography. Science 2003, 302, 1396–1398. [Google Scholar] [CrossRef] [PubMed]
- Wild, P.; Käch, A.; Lucas, M.S. High resolution scanning electron microscopy of the nuclear surface in herpes simplex virus 1 infected cells. In Scanning Electron Microscopy for the Life Sciences; Schatten, H., Ed.; Cambridge University Press: New York, USA, 2012. [Google Scholar]
- Wild, P.; Senn, C.; Manera, C.L.; Sutter, E.; Schraner, E.M.; Tobler, K.; Ackermann, M.; Ziegler, U.; Lucas, M.S.; Kaech, A. Exploring the nuclear envelope of herpes simplex virus 1-infected cells by high-resolution microscopy. J. Virol. 2009, 83, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Wagenaar, F.; Pol, J.M.; Peeters, B.; Gielkens, A.L.J.; de Wind, N.; Kimman, T.G. The US3-encoded protein kinase from pseudorabies virus affects egress of virions from the nucleus. J. Gen. Virol. 1995, 76, 1851–1859. [Google Scholar] [CrossRef] [PubMed]
- Granzow, H.; Klupp, B.G.; Mettenleiter, T.C. The pseudorabies virus US3 protein is a component of primary and of mature virions. J. Virol. 2004, 78, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Wild, P.; de Oliveira, A.P.; Sonda, S.; Schraner, E.M.; Ackermann, M.; Tobler, K. The herpes simplex virus 1 Us3 regulates phospholipid synthesis. Virology 2012, 432, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Darlington, R.W.; Moss, L.H. Herpesvirus envelopment. J. Virol. 1968, 2, 48–55. [Google Scholar] [PubMed]
- Leabu, M. Membrane fusion in cells: Molecular machinery and mechanisms. J. Cell. Mol. Med. 2006, 10, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Haluska, C.K.; Riske, K.A.; Marchi-Artzner, V.; Lehn, J.-M.; Lipowsky, R.; Dimova, R. From the Cover: Time scales of membrane fusion revealed by direct imaging of vesicle fusion with high temporal resolution. Proc. Natl. Acad. Sci. USA 2006, 103, 15841–15846. [Google Scholar] [CrossRef] [PubMed]
- Kanaseki, T.; Kawasaki, K.; Murata, M.; Ikeuchi, Y.; Ohnishi, S. Structural features of membrane fusion between influenza virus and liposome as revealed by quick-freezing electron microscopy. J. Cell Biol. 1997, 137, 1041–1056. [Google Scholar] [CrossRef] [PubMed]
- Naldinho-Souto, R.; Browne, H.; Minson, T. Herpes Simplex Virus Tegument Protein VP16 Is a Component of Primary Enveloped Virions. J. Virol. 2006, 80, 2582–2584. [Google Scholar] [CrossRef] [PubMed]
- Campadelli, G.; Brandimarti, R.; di Lazzaro, C.; Ward, P.L.; Roizman, B.; Torrisi, M.R. Fragmentation and dispersal of Golgi proteins and redistribution of glycoproteins and glycolipids processed through the Golgi apparatus after infection with herpes simplex virus 1. Proc. Natl. Acad. Sci. USA 1993, 90, 2798–2802. [Google Scholar] [CrossRef] [PubMed]
- Senn, C.; Sutter, E.; Leisinger, S.; Schraner, E.M.; Ackermann, M.; Kaech, A.; Wild, P. The Golgi complex: Crossroad in alpha-herpesvirus envelopment. Proc. 31th Herpes Workshop, Seattle 2006, 11, 34. [Google Scholar]
- Mettenleiter, T.C.; Klupp, B.G.; Granzow, H. Herpesvirus assembly: A tale of two membranes. Curr. Opin. Microbiol. 2006, 9, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Mettenleiter, T.C. Budding events in herpesvirus morphogenesis. Virus Res. 2004, 106, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Baines, J.D.; Wills, E.; Jacob, R.J.; Pennington, J.; Roizman, B. Glycoprotein M of herpes simplex virus 1 is incorporated into virions during budding at the inner nuclear membrane. J. Virol. 2007, 81, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Padula, M.E.; Sydnor, M.L.; Wilson, D.W. Isolation and preliminary characterization of herpes simplex virus 1 primary enveloped virions from the perinuclear space. J. Virol. 2009, 83, 4757–4765. [Google Scholar] [CrossRef] [PubMed]
- Kotsakis, A.; Pomeranz, L.E.; Blouin, A.; Blaho, J.A. Microtubule reorganization during herpes simplex virus type 1 infection facilitates the nuclear localization of VP22, a major virion tegument protein. J. Virol. 2001, 75, 8697–8711. [Google Scholar] [CrossRef] [PubMed]
- Longnecker, R.; Roizman, B. Clustering of genes dispensable for growth in culture in the S component of the HSV-1 genome. Science 1987, 236, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Kato, A.; Liu, Z.; Minowa, A.; Imai, T.; Tanaka, M.; Sugimoto, K.; Nishiyama, Y.; Arii, J.; Kawaguchi, Y. Herpes simplex virus 1 protein kinase Us3 and major tegument protein UL47 reciprocally regulate their subcellular localization in infected cells. J. Virol. 2011, 85, 9599–9613. [Google Scholar] [CrossRef] [PubMed]
- Campadelli-Fiume, G.; Farabegoli, F.; di Gaeta, S.; Roizman, B. Origin of unenveloped capsids in the cytoplasm of cells infected with herpes simplex virus 1. J. Virol. 1991, 65, 1589–1595. [Google Scholar] [PubMed]
- Torrisi, M.R.; di Lazzaro, C.; Pavan, A.; Pereira, L.; Campadelli-Fiume, G. Herpes simplex virus envelopment and maturation studied by fracture label. J. Virol. 1992, 66, 554–561. [Google Scholar] [PubMed]
- Spring, S.B.; Roizman, B.; Schwartz, J. Herpes simplex virus products in productive and abortive infection. II. Electron microscopic and immunological evidence for failure of virus envelopment as a cause of abortive infection. J. Virol. 1968, 2, 384–392. [Google Scholar] [PubMed]
- Baines, J.D.; Ward, P.L.; Campadelli-Fiume, G.; Roizman, B. The UL20 gene of herpes simplex virus 1 encodes a function necessary for viral egress. J. Virol. 1991, 65, 6414–6424. [Google Scholar] [PubMed]
- Hauri, H.P.; Schweizer, A. The endoplasmic reticulum-Golgi intermediate compartment. Curr. Opin. Cell Biol. 1992, 4, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Han, H.M.; Bouchet-Marquis, C.; Huebinger, J.; Grabenbauer, M. Golgi apparatus analyzed by cryo-electron microscopy. Histochem. Cell Biol. 2013, 140, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Marko, M.; Hsieh, C.; Schalek, R.; Frank, J.; Mannella, C. Focused-ion-beam thinning of frozen-hydrated biological specimens for cryo-electron microscopy. Nat. Methods 2007, 4, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, K.; Okamoto, M.; Nakano, A. Contact of cis-Golgi with ER exit sites executes cargo capture and delivery from the ER. Nat. Commun. 2014, 5, 3653. [Google Scholar] [CrossRef] [PubMed]
- Borchers, K.; Oezel, M. Simian Agent 8 (SA8): Morphogenesis and ultrastructure. Zbl Bakt 1993, 279, 526–536. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wild, P.; Leisinger, S.; De Oliveira, A.P.; Schraner, E.M.; Kaech, A.; Ackermann, M.; Tobler, K. Herpes Simplex Virus 1 Us3 Deletion Mutant is Infective Despite Impaired Capsid Translocation to the Cytoplasm. Viruses 2015, 7, 52-71. https://doi.org/10.3390/v7010052
Wild P, Leisinger S, De Oliveira AP, Schraner EM, Kaech A, Ackermann M, Tobler K. Herpes Simplex Virus 1 Us3 Deletion Mutant is Infective Despite Impaired Capsid Translocation to the Cytoplasm. Viruses. 2015; 7(1):52-71. https://doi.org/10.3390/v7010052
Chicago/Turabian StyleWild, Peter, Sabine Leisinger, Anna Paula De Oliveira, Elisabeth M. Schraner, Andres Kaech, Mathias Ackermann, and Kurt Tobler. 2015. "Herpes Simplex Virus 1 Us3 Deletion Mutant is Infective Despite Impaired Capsid Translocation to the Cytoplasm" Viruses 7, no. 1: 52-71. https://doi.org/10.3390/v7010052
APA StyleWild, P., Leisinger, S., De Oliveira, A. P., Schraner, E. M., Kaech, A., Ackermann, M., & Tobler, K. (2015). Herpes Simplex Virus 1 Us3 Deletion Mutant is Infective Despite Impaired Capsid Translocation to the Cytoplasm. Viruses, 7(1), 52-71. https://doi.org/10.3390/v7010052




