Prevalence and Genotyping of High Risk Human Papillomavirus in Cervical Cancer Samples from Punjab, Pakistan
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. DNA Extraction from Formalin Fixed Paraffin Embedded (FFPE) and Fresh Tissue Samples
2.3. DNA Extraction from HeLa Cells
2.4. Polymerase Chain Reaction
| Primer | Sequence (5'-3') | Target | Product Size (bp) | Reference |
|---|---|---|---|---|
| PC03 | ACACAACTGTGTTCACTAGC | β-globin | 110 | [15] |
| PC04 | CAACTTCATCCACGTTCACC | |||
| GP5+ | TTTGTTACTGTGGTAGATACTAC | L1 | 150 | [16] |
| GP6+ | GAAAAATAAACTGTAAATCATATTC | |||
| New TS 16 | GGTCGGTGGACCGGTCGATG | E6 HPV 16 | 96 | [17] |
| GCAATGTAGGTGTATCTCCA | ||||
| New TS 18 | CCTTGGACGTAAATTTTTGG CACGCACACGCTTGGCAGGT | L1 HPV 18 | 115 | [17] |
| C16E7F | GGGGAATTCGCATGGAGATACACCTACATTC | E7 HPV 16 | 297 | Current study |
| C16E7R | GGGCTCGAGTGGTTTCTGAGAACAGATGG | |||
| C18E7F | GGATCCGCATGGACCTAAGGCAACATT | E7 HPV 18 | 318 | Current study |
| C18E7R | GAATTCGCTGCTGGGATGCACACCA |
| Primer Name | Hot Start | Denaturation | Annealing | Extension | Final Extension |
|---|---|---|---|---|---|
| 35 cycles | |||||
| PC03/PC04 | 94 °C, 4 min | 94 °C, 30 s | 54 °C, 45 s | 72 °C, 30 s | 7 min, 72 °C |
| GP5+/GP6+ | 94 °C, 4 min | 94 °C, 30 s | 45 °C, 45 s | 72 °C, 30 s | 7 min, 72 °C |
| New TS 16 | 94 °C, 4 min | 94 °C, 45 s | 58 °C, 45 s | 72 °C, 30 s | 7 min, 72 °C |
| New TS 18 | |||||
| C16E7 | |||||
| C18E7 | |||||
3. Results and Discussion
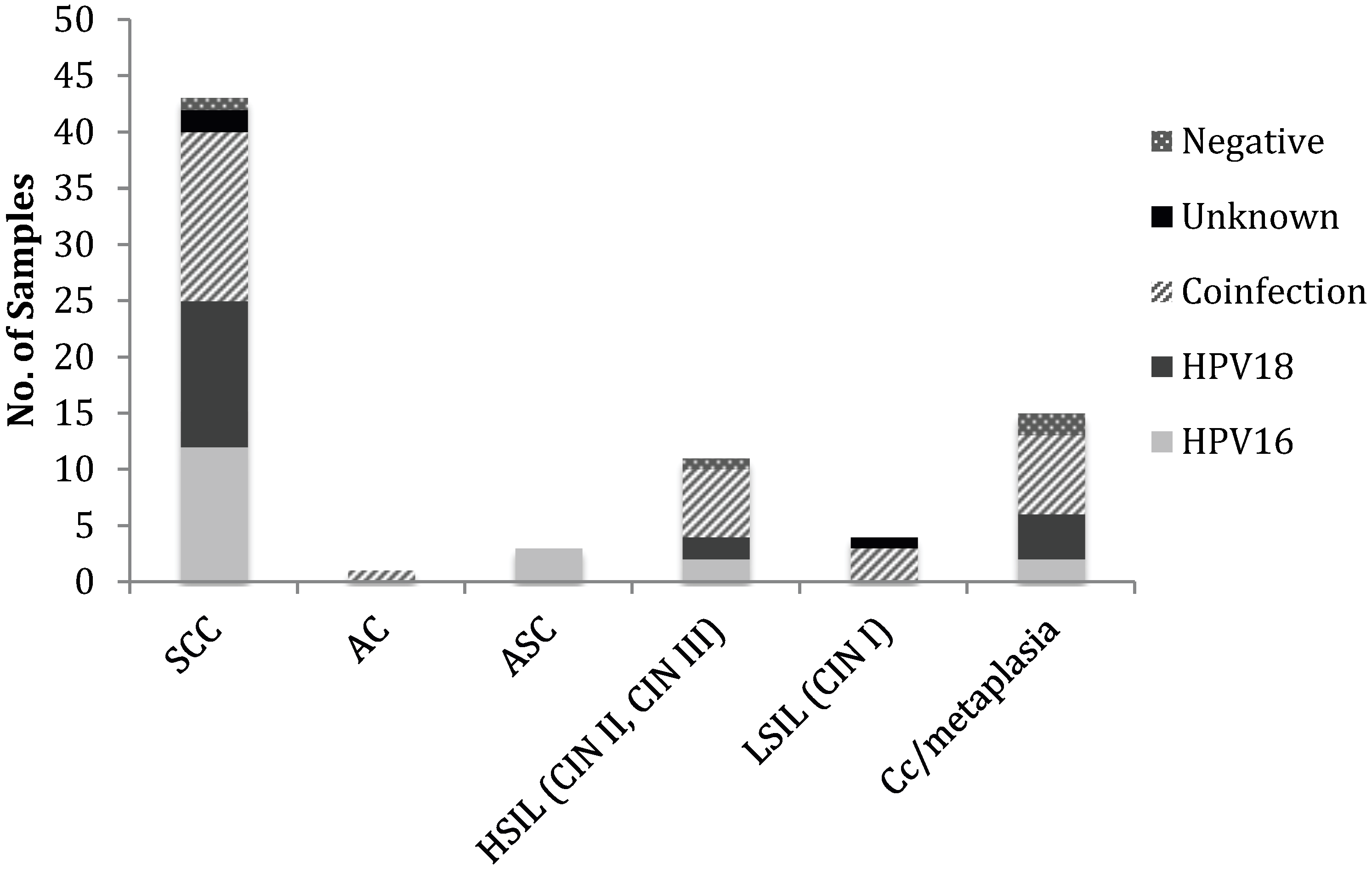
3.1. HPV Status of Cervical Cancer Biopsies
| Cytological diagnosis | HPV16 + | HPV18 + | Co infection | Unknown a | HPV − | Total |
|---|---|---|---|---|---|---|
| Invasive Cervical Cancer (ICC) | ||||||
| SCC | 12 | 13 | 15 | 2 | 1 | 43 |
| AC | - | - | 1 | - | - | 1 |
| ASC | 3 | - | - | - | - | 3 |
| High Grade Neoplasia | ||||||
| HSIL (CIN II, CIN III) | 2 | 2 | 6 | - | 1 | 11 |
| Low Grade Neoplasia | ||||||
| LSIL (CIN I) | - | - | 3 | 1 | - | 4 |
| Chronic cervicitis/metaplasia | 2 | 4 | 7 | - | 2 | 15 |
| Total | 19 | 19 | 32 | 3 | 4 | 77 |
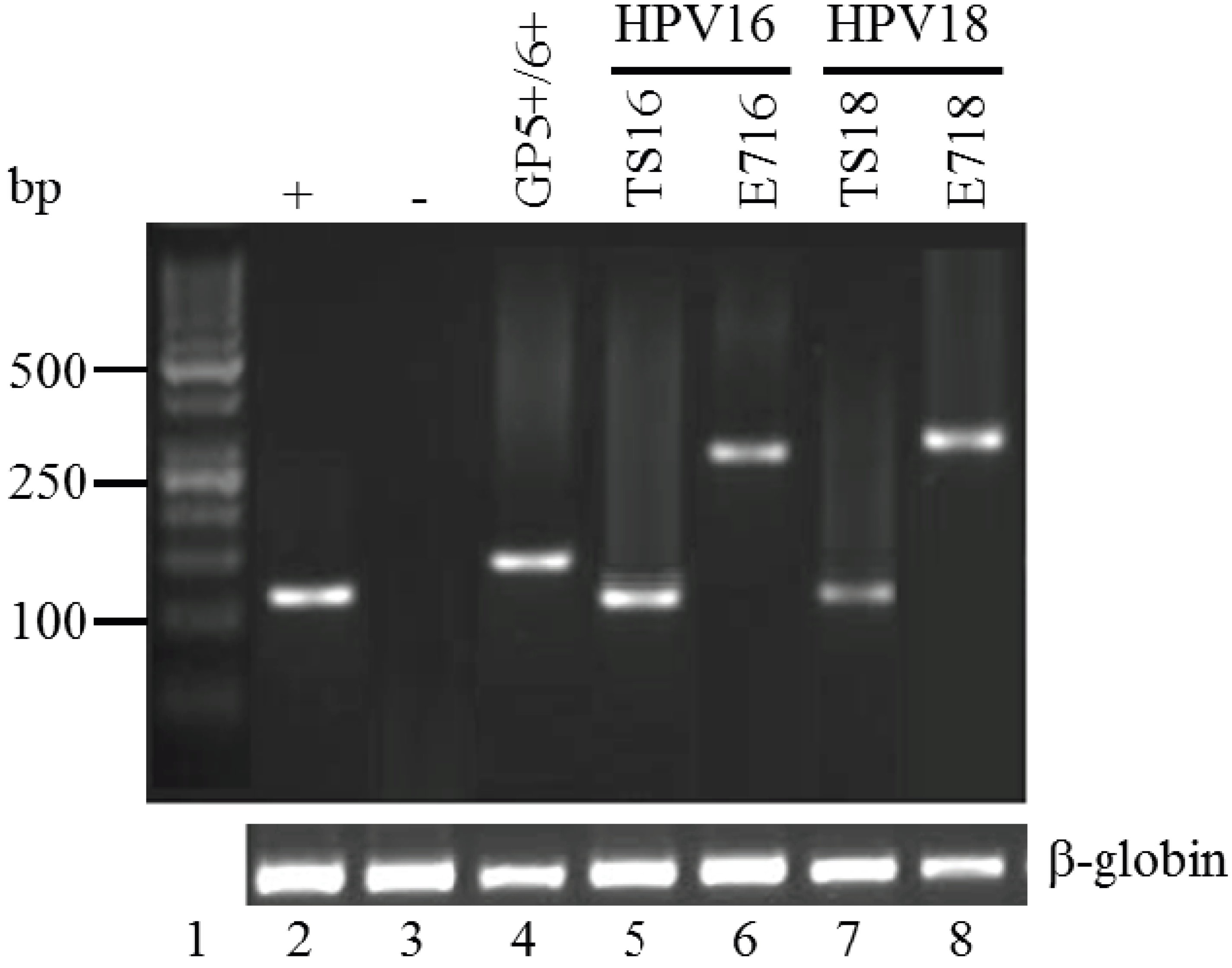
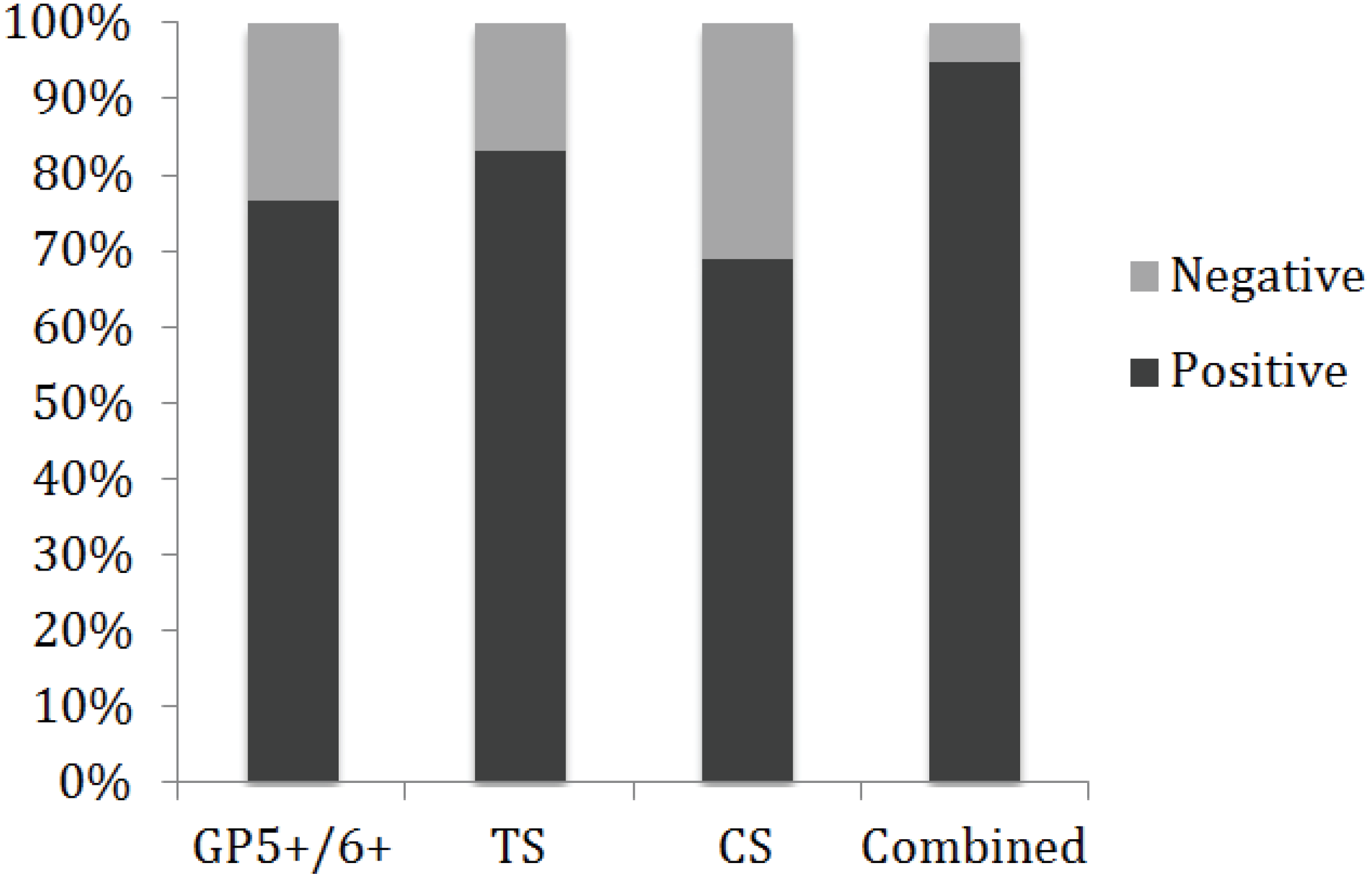
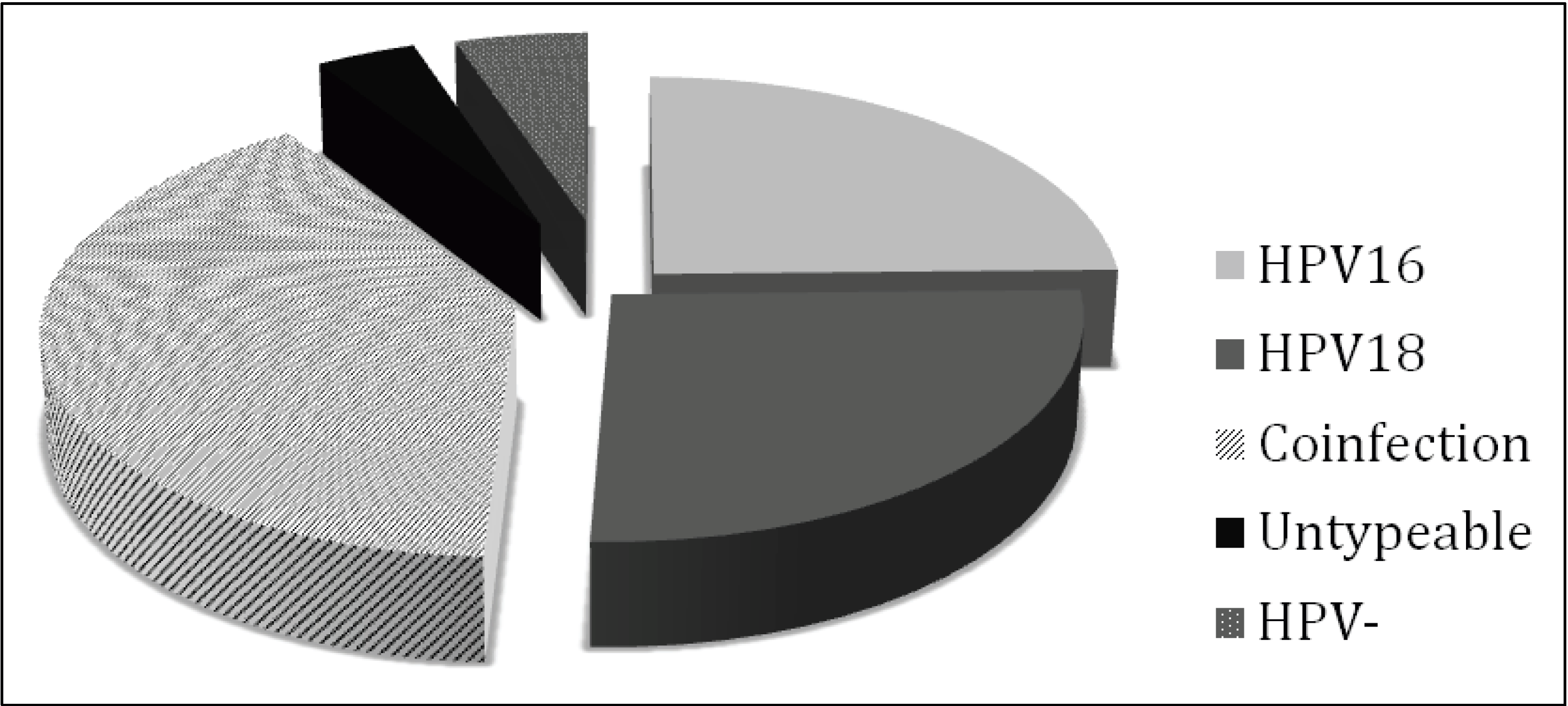
3.2. Histopathology, Age, and HPV Status
| Age Group | HPV16 + | HPV18 + | Co-infection | Genotype Unknown | HPV − | N |
|---|---|---|---|---|---|---|
| 21–30 | 1 | 1 | - | - | - | 2 |
| 31–40 | 4 | 3 | 5 | - | - | 12 |
| 41–50 | 4 | 4 | 7 | - | - | 15 |
| 51–60 | 5 | 4 | 3 | - | - | 12 |
| 61–70 | 3 | 2 | 3 | 2 | 1 | 11 |
| 71–80 | 1 | - | - | - | - | 1 |
| Unknown | 1 | 5 | 14 | 1 | 3 | 24 |
| Total | 19 | 19 | 32 | 3 | 4 | 77 |

4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA: A Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Zur Hausen, H. Papillomavirus infections: A major cause of human cancers. Biochem. Biophys. Acta 1996, 1288, F55–F78. [Google Scholar]
- Zur Hausen, H. Papillomaviruses in human cancers. Proc. Assoc. Am. Phys. 1999, 111, 581–587. [Google Scholar] [CrossRef]
- Burd, E.M. Human Papillomavirus and Cervical Cancer. Clin. Microbiol. Rev. 2003, 16, 1–17. [Google Scholar] [CrossRef]
- Muñoz, N.; Bosch, F.X.; de Sanjosé, S.; Herrero, R.; Castellsagué, X.; Shah, K.V.; Snijders, P.J.F.; Meijer, C.J.L.M. International Agency for Research on Cancer Multicenter Cervical Cancer Study Group Epidemiologic classification of human papillomavirus types associated with cervical cancer. N. Engl. J. Med. 2003, 348, 518–527. [Google Scholar] [CrossRef]
- Kjaer, S.K.; van den Brule, A.J.C.; Paull, G.; Svare, E.I.; Sherman, M.E.; Thomsen, B.L.; Suntum, M.; Bock, J.E.; Poll, P.A.; Meijer, C.J.L.M. Type specific persistence of high risk human papillomavirus (HPV) as indicator of high grade cervical squamous intraepithelial lesions in young women: Population based prospective follow up study. BMJ 2002, 325, 572. [Google Scholar] [CrossRef]
- Snijders, P.J.F.; van den Brule, A.J.C.; Meijer, C.J.L.M. The clinical relevance of human papillomavirus testing: Relationship between analytical and clinical sensitivity. J. Pathol. 2003, 201, 1–6. [Google Scholar] [CrossRef]
- Cancer Incidence in Five Continents; Curado, M.P.; Edwards, B.; Shin, H.R.; Storm, H.; Ferlay, J.; Heanue, M.; Boyle, P. (Eds.) IARC Scietinifc Publications No. 160; IARC: Lyon, France, 2007; Volume IX.
- Vaccarella, S.; Bruni, L.; Seoud, M. Burden of human papillomavirus infections and related diseases in the extended Middle East and North Africa region. Vaccine 2013, 31, G32–G44. [Google Scholar] [CrossRef]
- Yousuf, S.; Syed, S.; Moazzam, A.; Lucky, M.H. Frequency of high risk human papillomavirus types in squamous cell carcinoma of cervix among women. J. Pak. Med. Assoc. 2010, 60, 193–196. [Google Scholar]
- Khan, S.; Jaffer, N.N.; Khan, M.N.; Rai, M.A.; Shafiq, M.; Ali, A.; Pervez, S.; Khan, N.; Aziz, A.; Ali, S.H. Human papillomavirus subtype 16 is common in Pakistani women with cervical carcinoma. Int. J. Infect. Dis. 2007, 11, 313–317. [Google Scholar] [CrossRef]
- Raza, S.A.; Franceschi, S.; Pallardy, S.; Malik, F.R.; Avan, B.I.; Zafar, A.; Ali, S.H.; Pervez, S.; Serajuddaula, S.; Snijders, P.J.F.; et al. Human papillomavirus infection in women with and without cervical cancer in Karachi, Pakistan. Br. J. Cancer 2010, 102, 1657–1660. [Google Scholar] [CrossRef]
- Hamidi, A.E.L.; Liu, H.; Zhang, Y.; Hamoudi, R.; Kocjan, G.; Du, M.-Q. Archival cervical smears: A versatile resource for molecular investigations. Cytopathology 2002, 13, 291–299. [Google Scholar] [CrossRef]
- Jacobs, M.V.; Zielinski, D.; Meijer, C.J.; Pol, R.P.; Voorhorst, F.J.; de Schipper, F.A.; Runsink, A.P.; Snijders, P.J.; Walboomers, J.M. A simplified and reliable HPV testing of archival Papanicolaou-stained cervical smears: Application to cervical smears from cancer patients starting with cytologically normal smears. Br. J. Cancer 2000, 82, 1421–1426. [Google Scholar] [CrossRef]
- Saiki, R.K.; Gelfand, D.H.; Stoffel, S.; Scharf, S.J.; Higuchi, R.; Horn, G.T.; Mullis, K.B.; Erlich, H.A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 1988, 239, 487–491. [Google Scholar]
- De Roda Husman, A.M.; Walboomers, J.M.; van den Brule, A.J.; Meijer, C.J.; Snijders, P.J. The use of general primers GP5 and GP6 elongated at their 3' ends with adjacent highly conserved sequences improves human papillomavirus detection by PCR. J. Gen. Virol. 1995, 76, 1057–1062. [Google Scholar] [CrossRef]
- Baay, M.F.; Quint, W.G.; Koudstaal, J.; Hollema, H.; Duk, J.M.; Burger, M.P.; Stolz, E.; Herbrink, P. Comprehensive study of several general and type-specific primer pairs for detection of human papillomavirus DNA by PCR in paraffin-embedded cervical carcinomas. J. Clin. Microbiol. 1996, 34, 745–747. [Google Scholar]
- Bhurgri, Y.; Nazir, K.; Shaheen, Y.; Usman, A.; Faridi, N.; Bhurgri, H.; Malik, J.; Bashir, I.; Bhurgri, A.; Kayani, N.; et al. Patho-epidemiology of Cancer Cervix in Karachi South. Asian Pac. J. Cancer Prev. 2007, 8, 357–362. [Google Scholar]
- Bosch, F.X.; Manos, M.M.; Muñoz, N.; Sherman, M.; Jansen, A.M.; Peto, J.; Schiffman, M.H.; Moreno, V.; Kurman, R.; Shah, K.V. Prevalence of human papillomavirus in cervical cancer: A worldwide perspective. International biological study on cervical cancer (IBSCC) Study Group. J. Natl. Cancer Inst. 1995, 87, 796–802. [Google Scholar] [CrossRef]
- Walboomers, J.M.; Jacobs, M.V.; Manos, M.M.; Bosch, F.X.; Kummer, J.A.; Shah, K.V.; Snijders, P.J.; Peto, J.; Meijer, C.J.; Muñoz, N. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 1999, 189, 12–19. [Google Scholar] [CrossRef]
- Böhmer, G.; van den Brule, A.J.C.; Brummer, O.; Meijer, C.L.M.; Petry, K.U. No confirmed case of human papillomavirus DNA-negative cervical intraepithelial neoplasia grade 3 or invasive primary cancer of the uterine cervix among 511 patients. Am. J. Obstet. Gynecol. 2003, 189, 118–120. [Google Scholar] [CrossRef]
- Ghaffari, S.R.; Sabokbar, T.; Mollahajian, H.; Dastan, J.; Ramezanzadeh, F.; Ensani, F.; Yarandi, F.; Mousavi-Jarrahi, A.; Mohagheghi, M.A.; Moradi, A. Prevalence of human papillomavirus genotypes in women with normal and abnormal cervical cytology in Iran. Asian Pac. J. Cancer Prev. 2006, 7, 529–532. [Google Scholar]
- Shahsiah, R.; Khademalhosseini, M.; Mehrdad, N.; Ramezani, F.; Nadji, S.A. Human papillomavirus genotypes in Iranian patients with cervical cancer. Pathol. Res. Pract. 2011, 207, 754–757. [Google Scholar] [CrossRef]
- Esmaeili, M.; Bonyadi, M.; Dastranj, A.; Alizadeh, M.; Melli, M.S.; Shobeiri, M.J. HPV Typing in Women with Cervical Precancerous and Cancerous Lesions in Northwestern Iran. Gynecol. Obstet. Investig. 2008, 66, 68–72. [Google Scholar] [CrossRef]
- Alsbeih, G.; Ahmed, R.; Al-Harbi, N.; Venturina, L.A.; Tulbah, A.; Balaraj, K. Prevalence and genotypes’ distribution of human papillomavirus in invasive cervical cancer in Saudi Arabia. Gynecol. Oncol. 2011, 121, 522–526. [Google Scholar] [CrossRef]
- Haghshenas, M.; Golini-moghaddam, T.; Rafiei, A.; Emadeian, O.; Shykhpour, A.; Ashrafi, G.H. Prevalence and type distribution of high-risk human papillomavirus in patients with cervical cancer: A population-based study. Infect. Agents Cancer 2013, 8, 20. [Google Scholar] [CrossRef]
- Schellekens, M.C.; Dijkman, A.; Aziz, M.F.; Siregar, B.; Cornain, S.; Kolkman-Uljee, S.; Peters, L.A.W.; Fleuren, G.J. Prevalence of single and multiple HPV types in cervical carcinomas in Jakarta, Indonesia. Gynecol. Oncol. 2004, 93, 49–53. [Google Scholar] [CrossRef]
- Suthipintawong, C.; Siriaunkgul, S.; Tungsinmunkong, K.; Pientong, C.; Ekalaksananan, T.; Karalak, A.; Kleebkaow, P.; Vinyuvat, S.; Triratanachat, S.; Khunamornpong, S.; et al. Human Papilloma Virus Prevalence, Genotype Distribution, and Pattern of Infection in Thai Women. Asian Pac. J. Cancer Prev. 2011, 12, 853–856. [Google Scholar]
- Muñoz, N. Human papillomavirus and cancer: The epidemiological evidence. J. Clin. Virol. 2000, 19, 1–5. [Google Scholar] [CrossRef]
- Baldez da Silva, M.F.P.T.; Guimarães, V.; Silva, M.A.R.; Medeiros do Amaral, C.M.; Beçak, W.; Stocco, R.C.; Freitas, A.C.; Crovella, S. Frequency of human papillomavirus types 16, 18, 31, and 33 and sites of cervical lesions in gynecological patients from Recife, Brazil. Genet. Mol. Res. 2012, 11, 462–466. [Google Scholar] [CrossRef]
- Liu, B.; Lu, Z.; Wang, P.; Basang, Z.; Rao, X. Prevalence of high-risk human papillomavirus types (HPV-16, HPV-18) and their physical status in primary laryngeal squamous cell carcinoma. Neoplasma 2010, 57, 594–600. [Google Scholar] [CrossRef]
- Zur Hausen, H. Papillomaviruses and cancer: From basic studies to clinical application. Nat. Rev. Cancer 2002, 2, 342–350. [Google Scholar] [CrossRef]
- Bosch, F.X.; Lorincz, A.; Muñoz, N.; Meijer, C.J.L.M.; Shah, K.V. The causal relation between human papillomavirus and cervical cancer. J. Clin. Pathol. 2002, 55, 244–265. [Google Scholar] [CrossRef]
- Nanda, K.; McCrory, D.C.; Myers, E.R.; Bastian, L.A.; Hasselblad, V.; Hickey, J.D.; Matchar, D.B. Accuracy of the Papanicolaou test in screening for and follow-up of cervical cytologic abnormalities: A systematic review. Ann. Intern. Med. 2000, 132, 810–819. [Google Scholar] [CrossRef]
- Seabrook, J.M.; Hubbard, R.A. Achieving quality reproducible results and maintaining compliance in molecular diagnostic testing of human papillomavirus. Arch. Pathol. Lab. Med. 2003, 127, 978–983. [Google Scholar]
- Forman, D.; de Martel, C.; Lacey, C.J.; Soerjomataram, I.; Lortet-Tieulent, J.; Bruni, L.; Vignat, J.; Ferlay, J.; Bray, F.; Plummer, M.; et al. Global Burden of Human Papillomavirus and Related Diseases. Vaccine 2012, 30, F12–F23. [Google Scholar] [CrossRef]
- Tayyeb, R.; Khawaja, N.P.; Malik, N. Comparison of visual inspection of cervix and Pap smear for cervical cancer screening. J. Coll. Phys. Surg. Pak. 2003, 13, 201–203. [Google Scholar]
- Lăără, E.; Day, N.E.; Hakama, M. Trends in mortality from cervical cancer in the Nordic countries: Association with organised screening programmes. Lancet 1987, 1, 1247–1249. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Siddiqa, A.; Zainab, M.; Qadri, I.; Bhatti, M.F.; Parish, J.L. Prevalence and Genotyping of High Risk Human Papillomavirus in Cervical Cancer Samples from Punjab, Pakistan. Viruses 2014, 6, 2762-2777. https://doi.org/10.3390/v6072762
Siddiqa A, Zainab M, Qadri I, Bhatti MF, Parish JL. Prevalence and Genotyping of High Risk Human Papillomavirus in Cervical Cancer Samples from Punjab, Pakistan. Viruses. 2014; 6(7):2762-2777. https://doi.org/10.3390/v6072762
Chicago/Turabian StyleSiddiqa, Abida, Maidah Zainab, Ishtiaq Qadri, Muhammad Faraz Bhatti, and Joanna L. Parish. 2014. "Prevalence and Genotyping of High Risk Human Papillomavirus in Cervical Cancer Samples from Punjab, Pakistan" Viruses 6, no. 7: 2762-2777. https://doi.org/10.3390/v6072762
APA StyleSiddiqa, A., Zainab, M., Qadri, I., Bhatti, M. F., & Parish, J. L. (2014). Prevalence and Genotyping of High Risk Human Papillomavirus in Cervical Cancer Samples from Punjab, Pakistan. Viruses, 6(7), 2762-2777. https://doi.org/10.3390/v6072762






