Characterization of Cytomegalovirus Lung Infection in Non-HIV Infected Children
Abstract
:1. Introduction
2. Experimental
2.1. Patients
2.2. Clinical Assessment
2.3. Diagnostic Studies
3. Results
| N | 15 |
|---|---|
| Male, n (%) | 8 (53) |
| Age (year), median (IQR) | 3 (0.25–4.9) |
| Comorbidity | |
| Immunocompromised, n (%) | 13/15 (87) |
| Systemic steroid use, n (%) | 11 (73) |
| Hypogammaglobulinemia | 8 (53) |
| Malnutrition, n (%) | 6 (40) |
| Acute leukemia, n (%) | 3 (20) |
| Autoimmune disease, n (%) | 2 (13) |
| Allogeneic bone marrow/stem cell transplant, n (%) | 2 (13) |
| Renal transplant, n (%) | 1 (6) |
3.1. Clinical Characterization of CMV Lung Infection
| Duration | |
|---|---|
| Onset of symptoms (days), median (IQR) | 14 (3–20) |
| Time to diagnosis (days), median (IQR) | 26 (11–37) |
| Presenting symptoms | |
| Cough, n (%) | 15 (100) |
| Increased breathing effort, n (%) | 14 (93) |
| Fever, n (%) | 10 (67) |
| Weight loss, n (%) | 8 (53) |
| Laboratory findings | |
| Anemia, n (%) | 8 (53) |
| Thrombocytopenia, n (%) | 6 (40) |
| Leukocytosis, n (%) | 6 (40) |
| Leukocytopenia, n (%) | 4 (27) |
| Abnormal liver function, n (%) | 7 (47) |
| Clinical variables | |
|---|---|
| Abnormal lung sounds, n (%) | 15 (100) |
| Wheezing, n (%) | 12 (80) |
| Rales, n (%) | 11 (73) |
| Rhonchus, n (%) | 10 (67) |
| Hypoxemia, n (%) | 15 (100) |
| Mechanical ventilation (MV), n (%) | 7 (47) |
| Days of MV, median (IQR) | 8 (7–26) |
| Death, n (%) | 2 (13) |
| Radiological variables | |
| Abnormal lung images, n (%) | 15 (100) |
| Ground-glass opacity, n (%) | 12 (80) |
| Consolidation, n (%) | 12 (80) |
| Atelectasis, n (%) | 6 (40) |
| Air trapping, n (%) | 5 (33) |
| Nodular pattern, n (%) | 3 (20) |
| Reticular pattern, n (%) | 1 (6) |
| Tree-in-bud pattern, n (%) | 1 (6) |
3.2. Radiological Findings in CMV Lung Infection
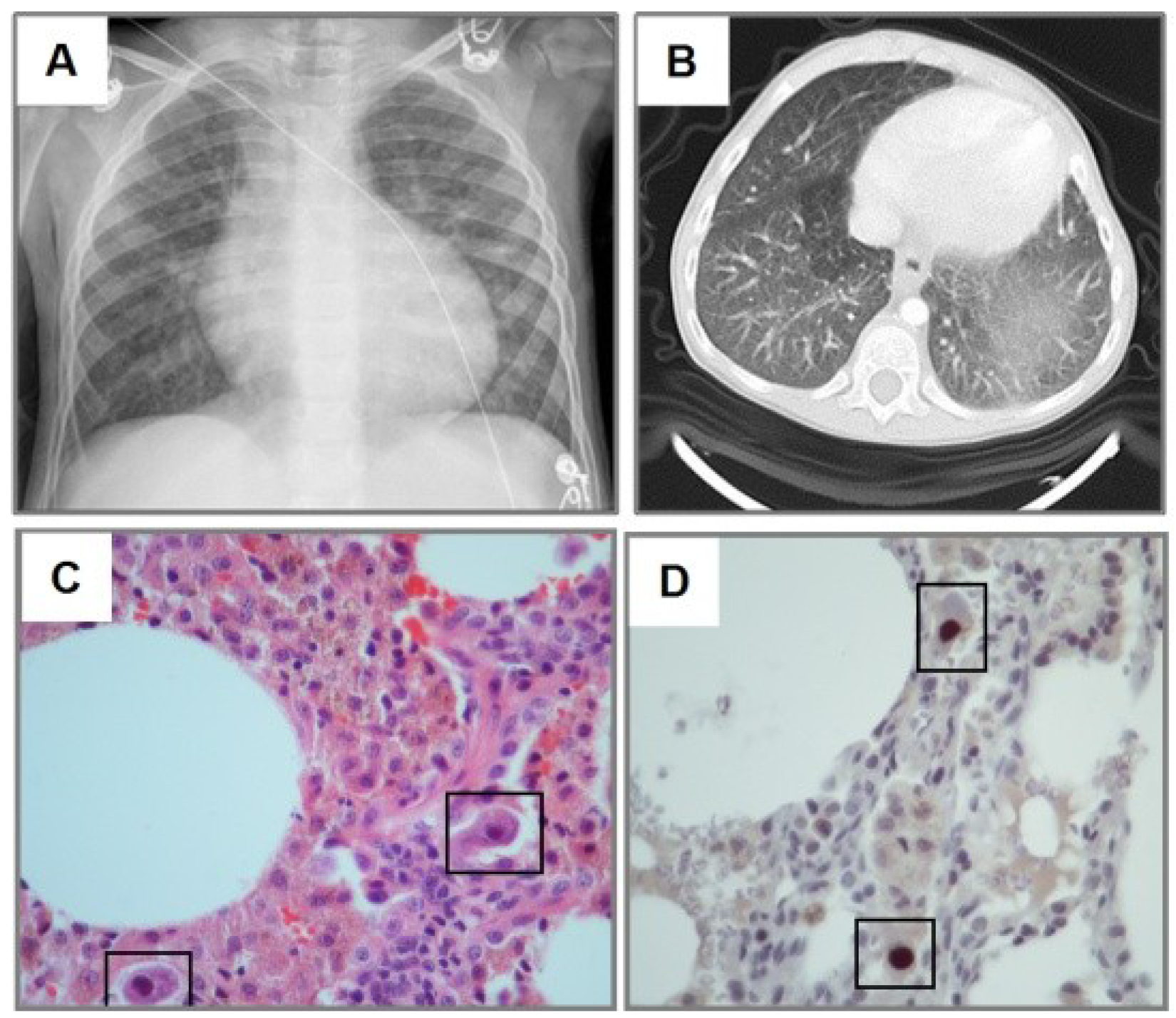
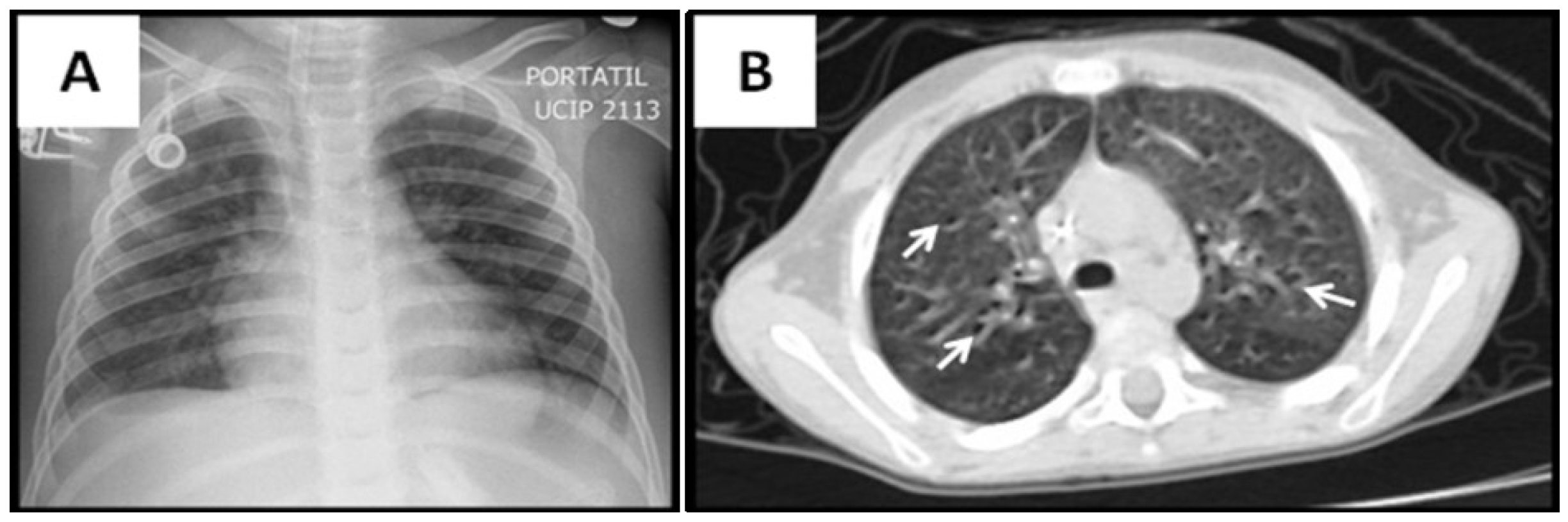
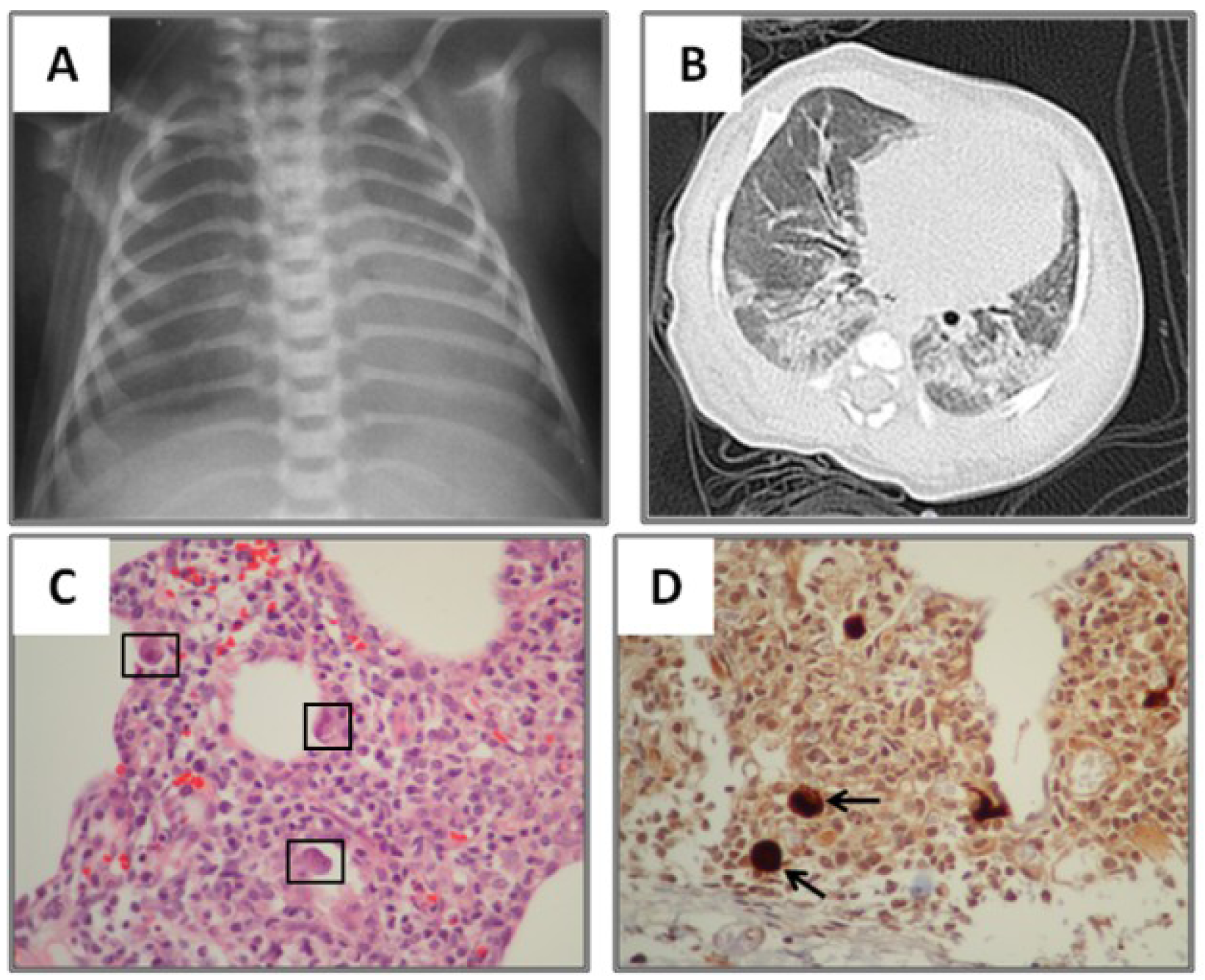
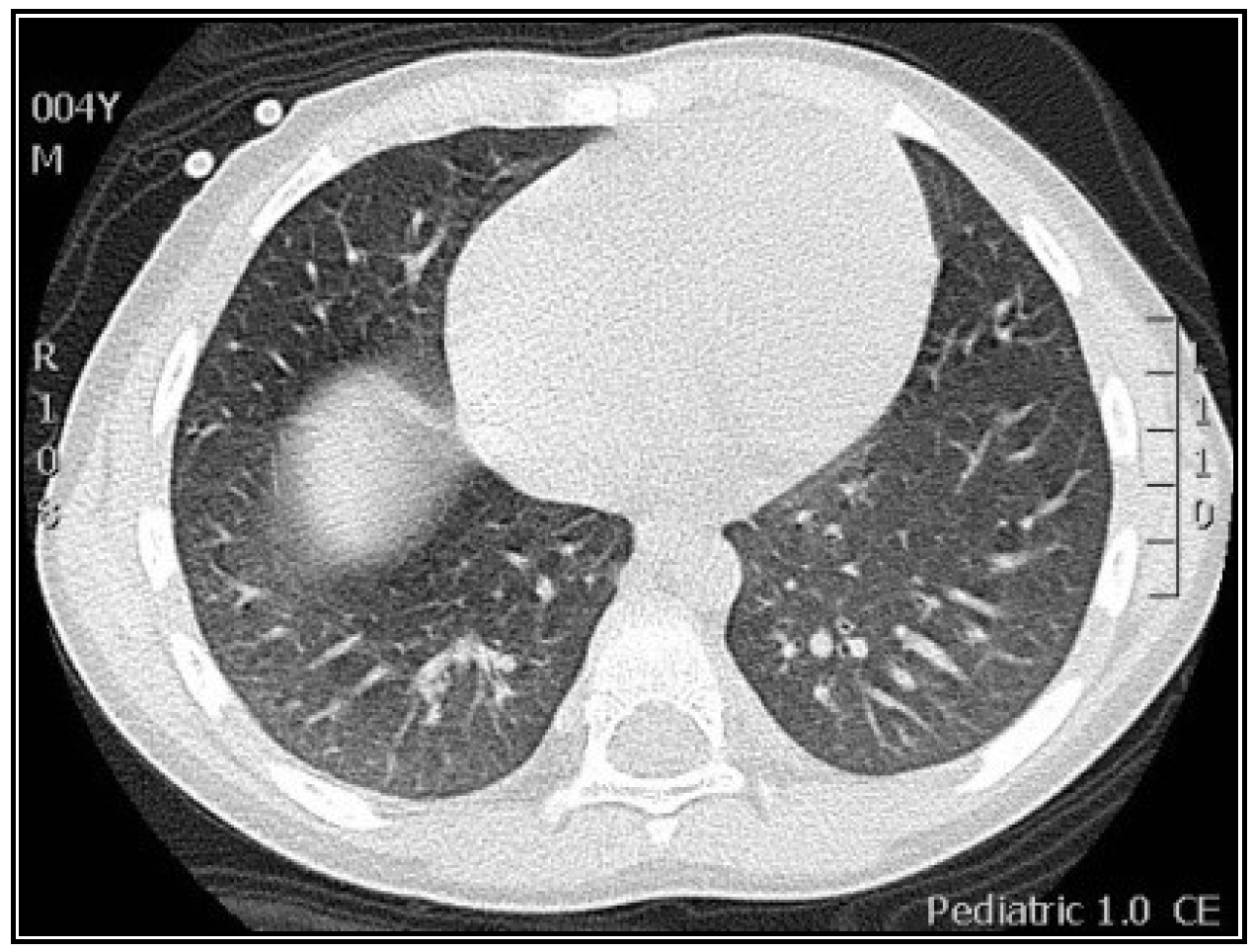
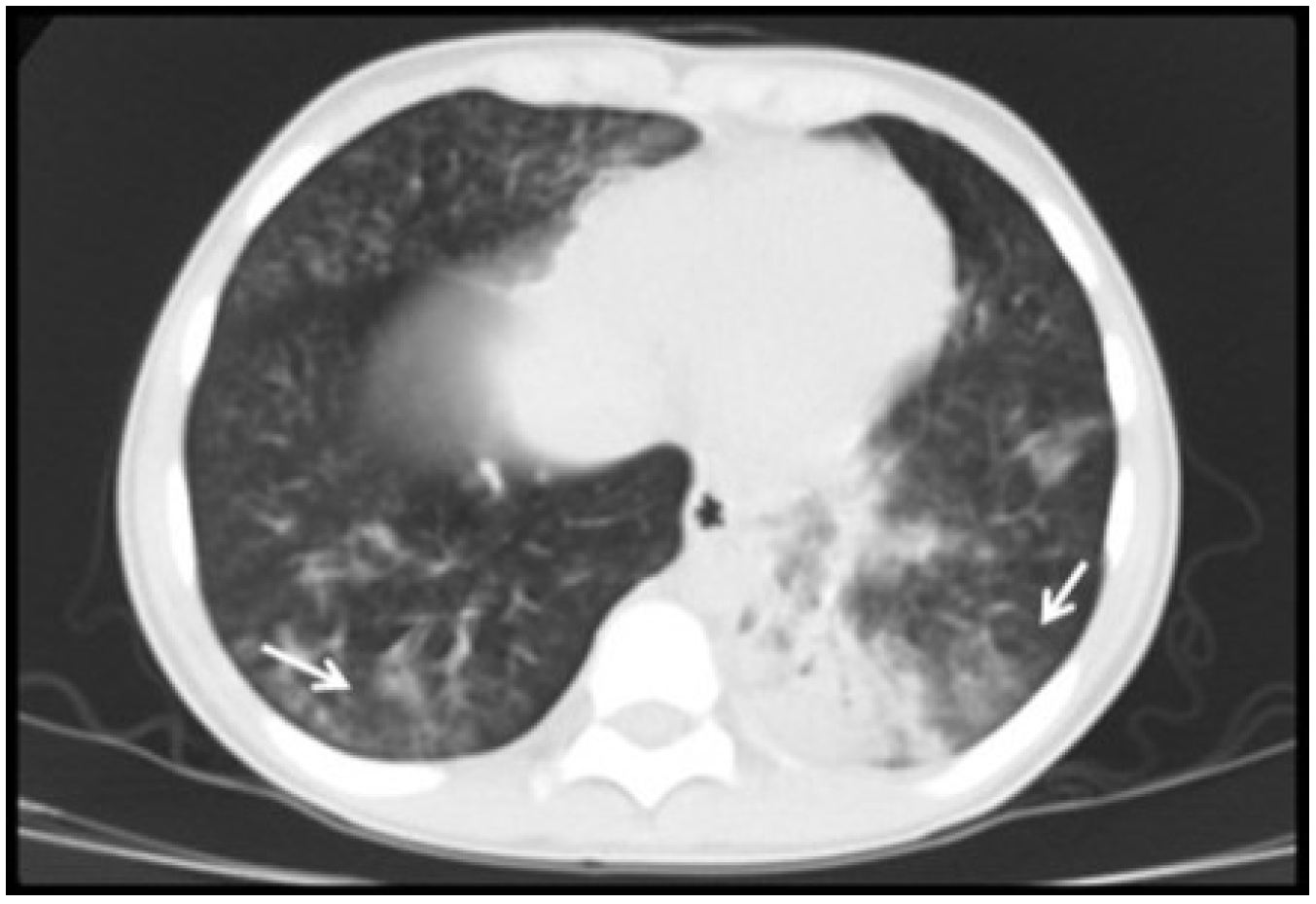
3.3. Diagnostic and Treatment Approaches of CMV Lung Infection in Children
| Intervention | |
|---|---|
| Bronchoalveolar lavage (BAL), n (%) | 12 (80) |
| CMV PCR (+) in BAL, n (%) | 9/11(82) |
| Neutrophilia in BAL, n (%) | 11/12(92) |
| Lung biopsy, n (%) | 5 (33) |
| Biopsy confirmed CMV, n (%) | 5/5 (100) |
| CMV serum PCR viral load, n (%) | 14/15 (93) |
| CMV PCR viral load (+) in serum | 7/14 (50) |
| CMV copies/μL, median (IQR) | 268 (20–20,000) |
| CMV serology, n (%) | 5/15 (33) |
| CMV IgM (+) in serum | 4/5 (80) |
| CMV treatment, n (%) | 14/15 (93) |
| Ganciclovir, n (%) | 14/14 (100) |
| Valganciclovir, n (%) | 4/14 |
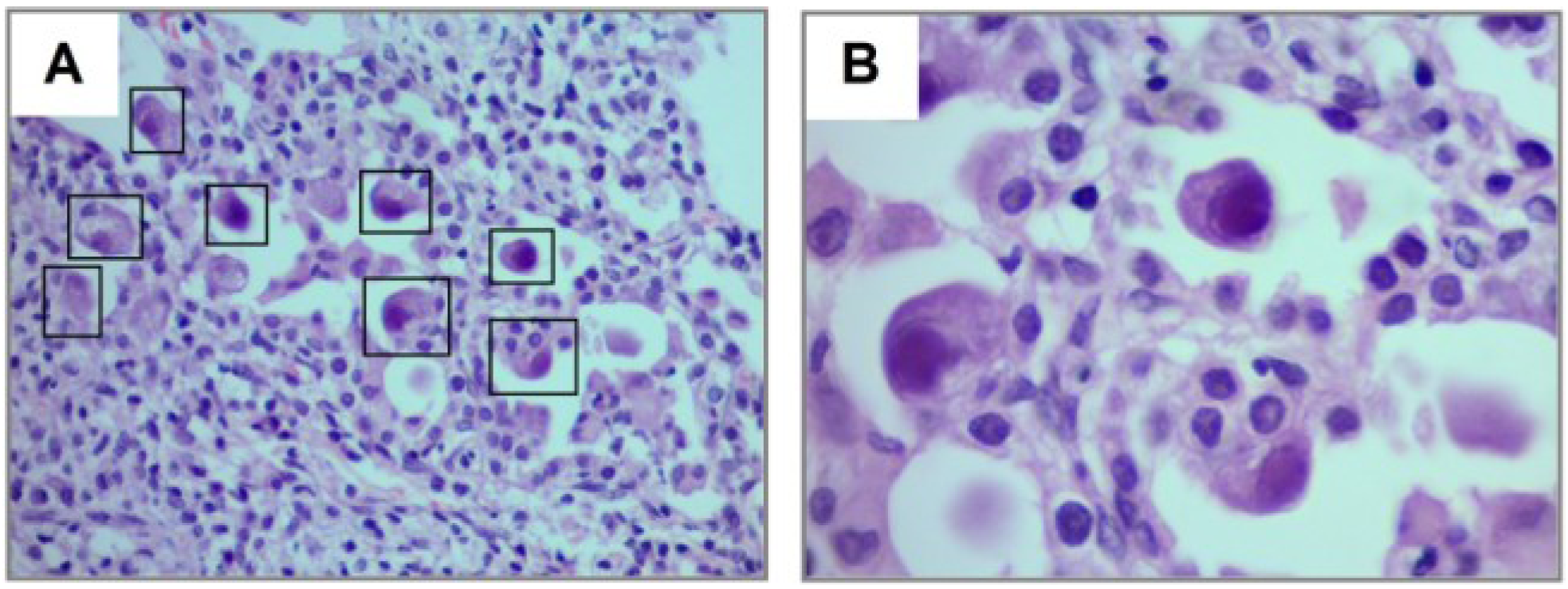
4. Discussion
| Immunosupression |
| Systemic steroid use |
| Malnutrition |
| Hypogammaglobulinemia |
| Hematologic malignancy |
| Post-transplantation |
| Clinical |
| Cough |
| Increased breathing effort |
| Hypoxemia |
| Diffuse adventitious lung sounds (i.e., rales, wheezing) |
| Imaging |
| Ground-glass opacity/consolidation |
| Bronchoscopy |
| CMV PCR in BAL |
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Manicklal, S.; Emery, V.C.; Lazzarotto, T.; Boppana, S.B.; Gupta, R.K. The “silent” global burden of congenital citomegalovirus. Clin. Microbiol. Rev. 2013, 26, 86–102. [Google Scholar] [CrossRef]
- Russel, M.; Palmer, A.; Michaels, M. Citomegalovirus infection in pediatric inmunocompromised hosts. Infect. Disord. Drug Targets 2011, 11, 437–448. [Google Scholar] [CrossRef]
- Gandhi, M.; Khanna, R. Human cytomegalovirus: Clinical aspects, immune regulation, and emerging treatments. Lancet Infect. Dis. 2004, 4, 725–738. [Google Scholar] [CrossRef]
- Radigan, K.; Wunderink, R. Epidemic viral pneumonia and other emerging pathogens. Clin. Chest Med. 2011, 32, 451–467. [Google Scholar] [CrossRef]
- Smith, S.D.; Cho, C.T.; Brahmacupta, N.; Lenahan, M.F. Pulmonary involvement with cytomegalovirus infections in children. Arch. Dis. Child. 1977, 52, 441–446. [Google Scholar] [CrossRef]
- Zhou, W.; Lin, F.; Teng, L.; Li, H.; Hou, J.; Tong, R.; Zheng, C.; Lou, Y.; Tan, W. Prevalence of herpes and respiratory viruses in induced sputum among hospitalized children with non typical bacterial community-acquired pneumonia. PLoS One 2013, 8, e79477. [Google Scholar]
- Jouneau, S.; Poineuf, J.S.; Minjolle, S.; Tattevin, P.; Uhel, F.; Kerjouan, M.; le Hô, H.; Desrues, B. Wich patients should be tested for viruses on bronchoalveolar lavage fluid? Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 671–677. [Google Scholar] [CrossRef]
- Cunha, B.A. Cytomegalovirus pneumonia: Community-acquired pneumonia in immunocompetent hosts. Infect. Dis. Clin. North Am. 2010, 24, 147–158. [Google Scholar] [CrossRef]
- De Blic, J.; Midulla, F.; Barbato, A.; Clement, A.; Dab, I.; Eber, E.; Green, C.; Grigg, J.; Kotecha, S.; Kurland, G.; et al. Bronchoalveolar lavage in children. ERS Task Force on bronchoalveolar lavage in children. European Respiratory Society. Eur. Respir. J. 2000, 15, 217–231. [Google Scholar] [CrossRef]
- Hansel, D.M.; Bankier, A.A.; MacMahon, H.; McLoud, T.C.; Müller, N.L.; Remy, J. Fleischner society: Glossary of terms for thoracic imaging. Radiology 2008, 246, 697–722. [Google Scholar] [CrossRef]
- Doan, T.; Phung, T.T.; Pham, H.V.; Pham, S.H.; Nguyen, L.T. Effect of ganciclovir for the treatment of severe cytomegalovirus-associated pneumonia in children without a specific immunocompromised state. BMC Infect. Dis. 2013, 13, e424. [Google Scholar] [CrossRef]
- Jogeesvaran, K.H.; Owens, C. Chronic diseases of lung parenchyma in children: The role of imaging. Pediatr. Radiol. 2010, 40, 850–858. [Google Scholar] [CrossRef]
- Alarcon, A.A.; Baquero-Artigao, F.; Grupo de estudio de la infección por citomegalovirus de la Sociedad Española de Infectología Pediátrica. Review and guidelines on the prevention, diagnosis and treatment of post-natal cytomegalovirus infection. An. Pediatr. 2011, 74. [Google Scholar] [CrossRef]
- Escribano, A.; Chilet, M.; Clari, M.Á.; Lucas, R.; Costa, E.; Bravo, D.; Muñoz-Cobo, B.; Borrás, R.; Navarro, D. Frequent detection of cytomegalovirus (CMV) DNA in the lower respiratory tract in CMV-seropositive pediatric patients with underlying chronic bronchopulmonary diseases lacking canonical immunosuppression. J. Med. Virol. 2013, 85, 888–892. [Google Scholar] [CrossRef]
- Khan, N. The immunological burden of human cytomegalovirus infection. Arch. Immunol. Ther. Exp. 2007, 55, 299–308. [Google Scholar] [CrossRef]
- Lidehäll, A.K.; Engman, M.L.; Sund, F.; Malm, G.; Lewensohn-Fuchs, I.; Ewald, U.; Tötterman, T.H.; Karltorp, E.; Korsgren, O.; Eriksson, B.M. Cytomegalovirus-specific CD4 and CD8 T cell responses in infants and children. Scand. J. Immunol. 2013, 77, 135–143. [Google Scholar] [CrossRef]
- La Rosa, C.; Diamond, D.J. The immune response to human CMV. Future Virol. 2012, 7, 279–293. [Google Scholar] [CrossRef]
- Corrin, B.; Nicholson, A.G. Pathology of the Lungs, 2nd ed.; Elsevier: London, UK, 2006; Chapter 5; pp. 159–160. [Google Scholar]
- Griffiths, M.H.; Miller, R.F.; Semple, S.J. Interstitial pneumonitis in patients infected with the human immunodeficiency virus. Thorax 1995, 50, 1141–1146. [Google Scholar] [CrossRef]
- Jeena, P.M.; Coovadia, H.M.; Chrystal, V. Pneumocystis carinii and cytomegalovirus infections in severely ill, HIV-infected African infants. Ann. Trop. Paediatr. 1996, 16, 361–368. [Google Scholar]
- Avila-Agüero, M.L.; Paris, M.M.; Alfaro, W.; Avila-Agüero, C.R.; Faingezicht, I. Ganciclovir therapy in cytomegalovirus (CMV) infection in immunocompetent pediatric patients. Int. J. Infect. Dis. 2003, 7, 278–281. [Google Scholar] [CrossRef]
- Honda, J.; Yonemitsu, J.; Kitajima, H.; Yosida, N.; Fumirori, T.; Oizumi, K. Clinical utility of capillary polymerase chain reaction for diagnosis of Cyto-megalovirus pneumonia. Scand. J. Infect. Dis. 2001, 33, 702–705. [Google Scholar] [CrossRef]
- Ljungman, P. Cytomegalovirus pneumonia: Presentation, diagnosis, and treatment. Semin. Respir. Infect. 1995, 10, 209–215. [Google Scholar]
- Moon, J.H.; Kim, E.A.; Lee, K.S.; Kim, T.S.; Jung, K.J.; Song, J.H. Cytomegalovirus pneumonia: High-resolution CT findings in ten non-AIDS immunocompromised patients. Korean J. Radiol. 2000, 1, 73–78. [Google Scholar] [CrossRef]
- West, B.W. Respiratory Physiology the Essentials: Ventilation-Perfusion Relationships, 9th ed.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2012; pp. 56–76. [Google Scholar]
- Agrawal, A.; Agrawal, A.; Bansal, V.; Pandit, M. A systematic approach to interpretation of heterogeneous lung attenuation on computed tomography of the chest. Lung India 2013, 30, 327–334. [Google Scholar] [CrossRef]
- Popler, J.; Gower, W.A.; Mogayzel, P.J., Jr.; Nogee, L.M.; Langston, C.; Wilson, A.C.; Hay, T.C.; Deterding, R.R. Familial neuroendocrine cell hyperplasia of infancy. Pediatr. Pulmonol. 2010, 45, 749–755. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Restrepo-Gualteros, S.M.; Jaramillo-Barberi, L.E.; Gonzalez-Santos, M.; Rodriguez-Martinez, C.E.; Perez, G.F.; Gutierrez, M.J.; Nino, G. Characterization of Cytomegalovirus Lung Infection in Non-HIV Infected Children. Viruses 2014, 6, 2038-2051. https://doi.org/10.3390/v6052038
Restrepo-Gualteros SM, Jaramillo-Barberi LE, Gonzalez-Santos M, Rodriguez-Martinez CE, Perez GF, Gutierrez MJ, Nino G. Characterization of Cytomegalovirus Lung Infection in Non-HIV Infected Children. Viruses. 2014; 6(5):2038-2051. https://doi.org/10.3390/v6052038
Chicago/Turabian StyleRestrepo-Gualteros, Sonia M., Lina E. Jaramillo-Barberi, Monica Gonzalez-Santos, Carlos E. Rodriguez-Martinez, Geovanny F. Perez, Maria J. Gutierrez, and Gustavo Nino. 2014. "Characterization of Cytomegalovirus Lung Infection in Non-HIV Infected Children" Viruses 6, no. 5: 2038-2051. https://doi.org/10.3390/v6052038
APA StyleRestrepo-Gualteros, S. M., Jaramillo-Barberi, L. E., Gonzalez-Santos, M., Rodriguez-Martinez, C. E., Perez, G. F., Gutierrez, M. J., & Nino, G. (2014). Characterization of Cytomegalovirus Lung Infection in Non-HIV Infected Children. Viruses, 6(5), 2038-2051. https://doi.org/10.3390/v6052038




