Heparan Sulfate Proteoglycan: An Arbovirus Attachment Factor Integral to Mosquito Salivary Gland Ducts
Abstract
:1. Introduction
2. Results
2.1. Virus-Associated Pathology in Salivary Glands
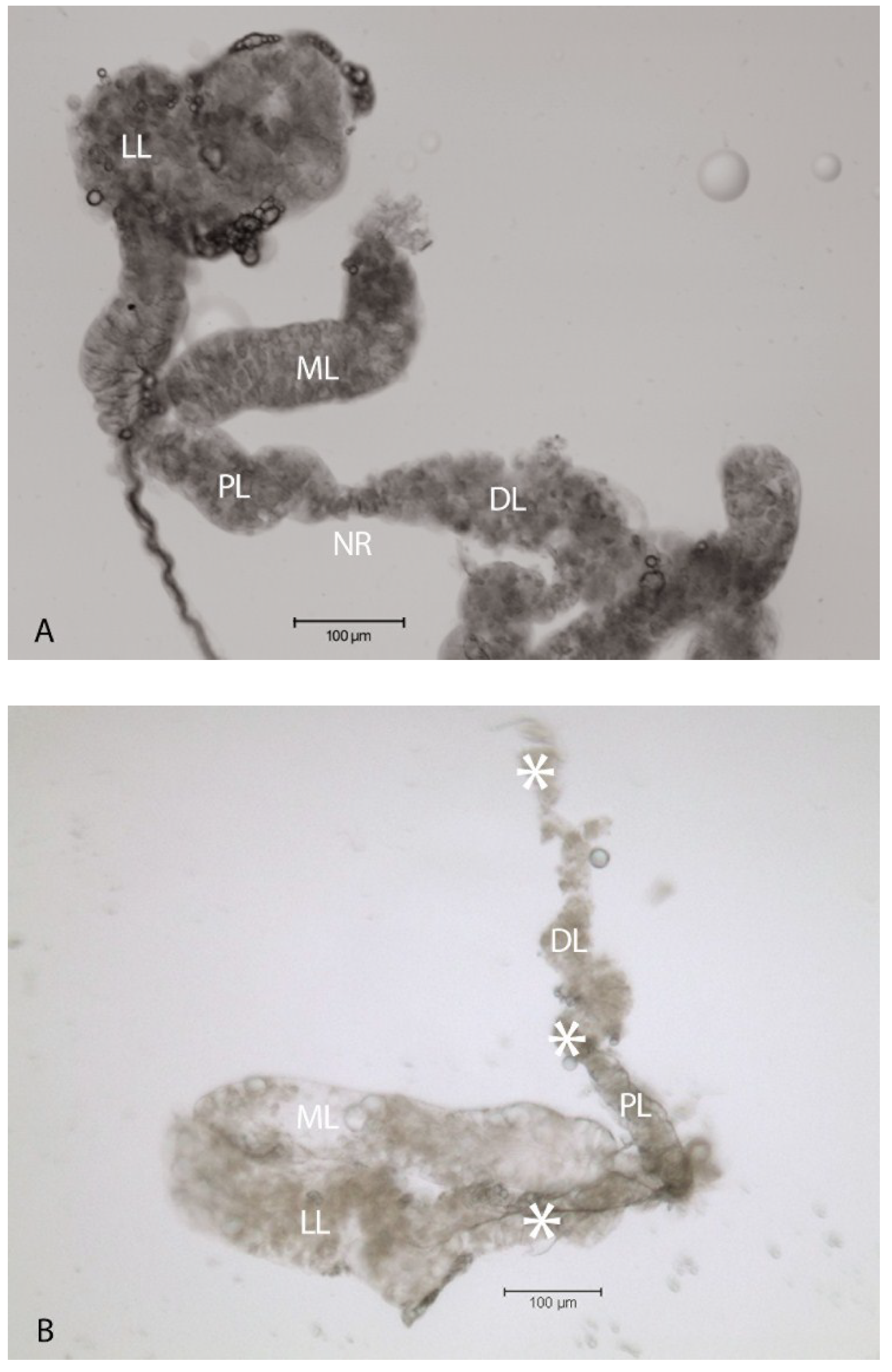
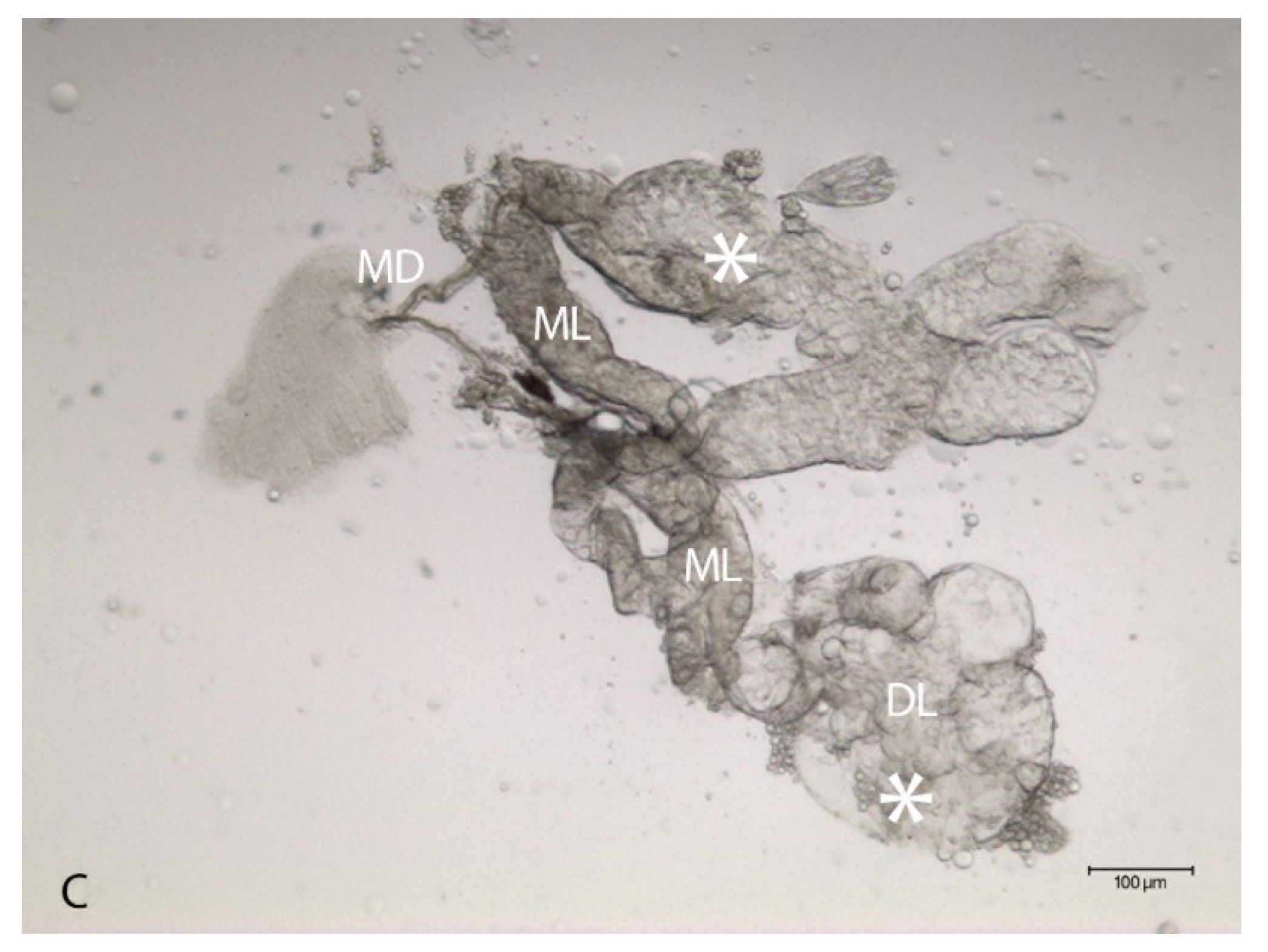
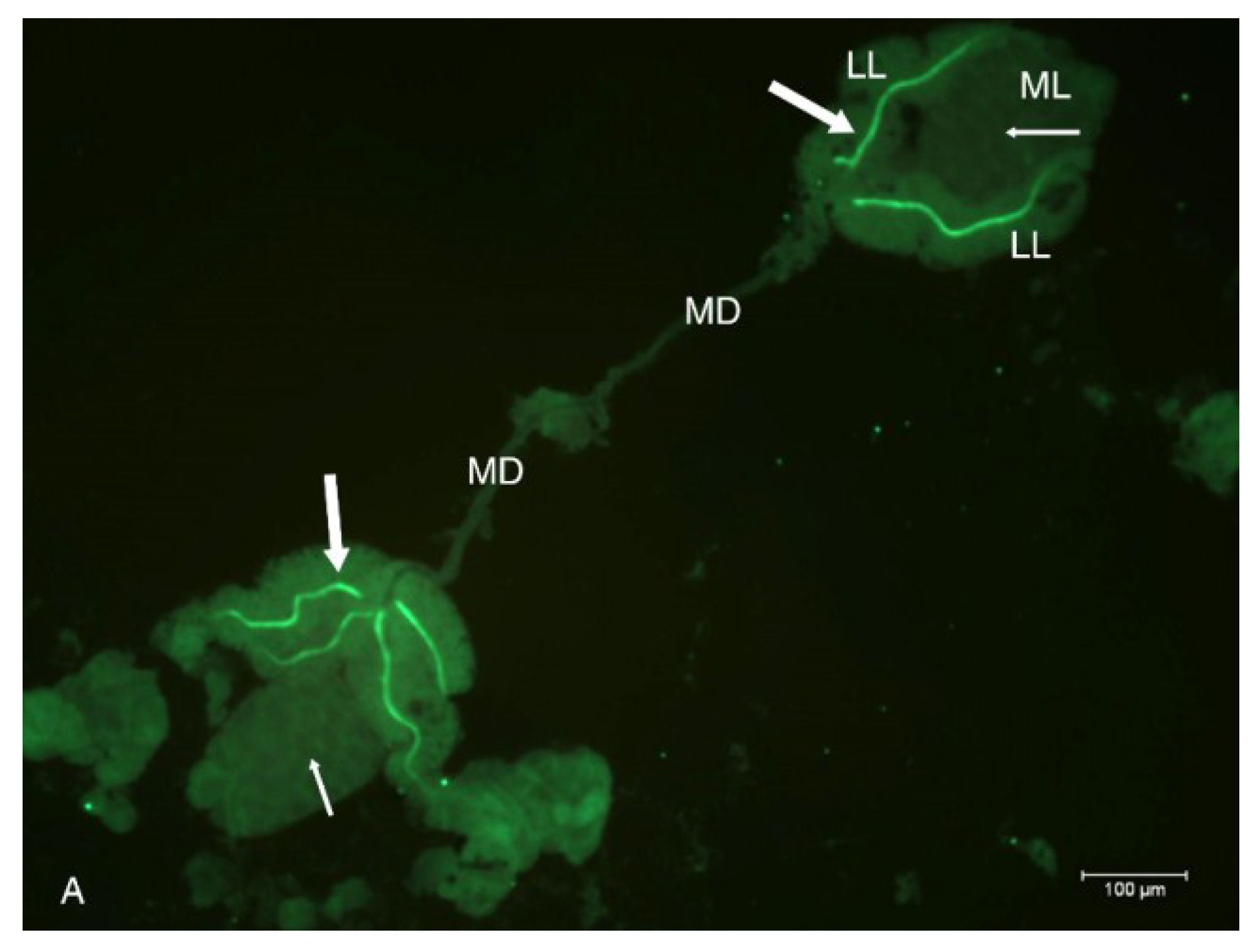
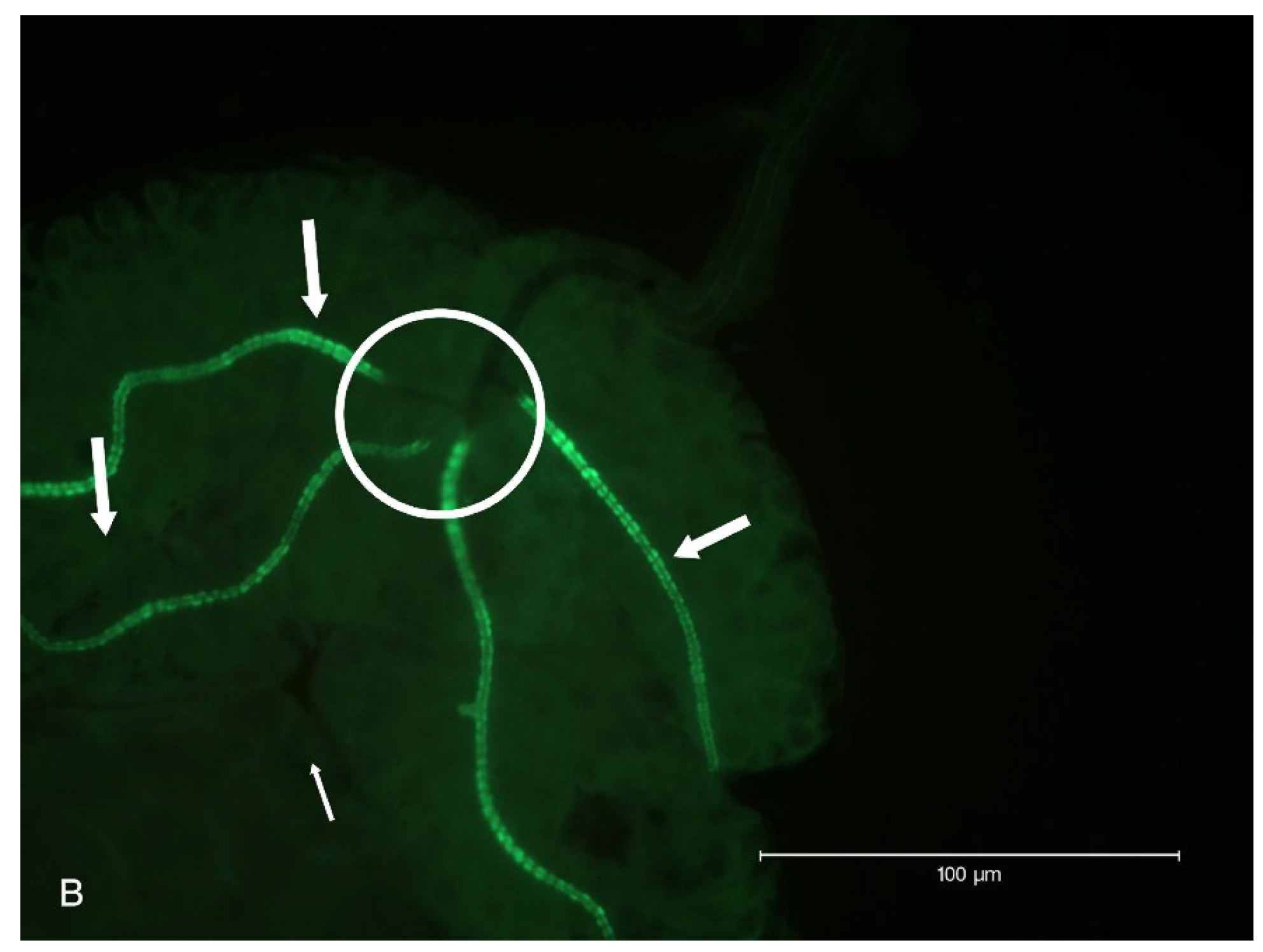
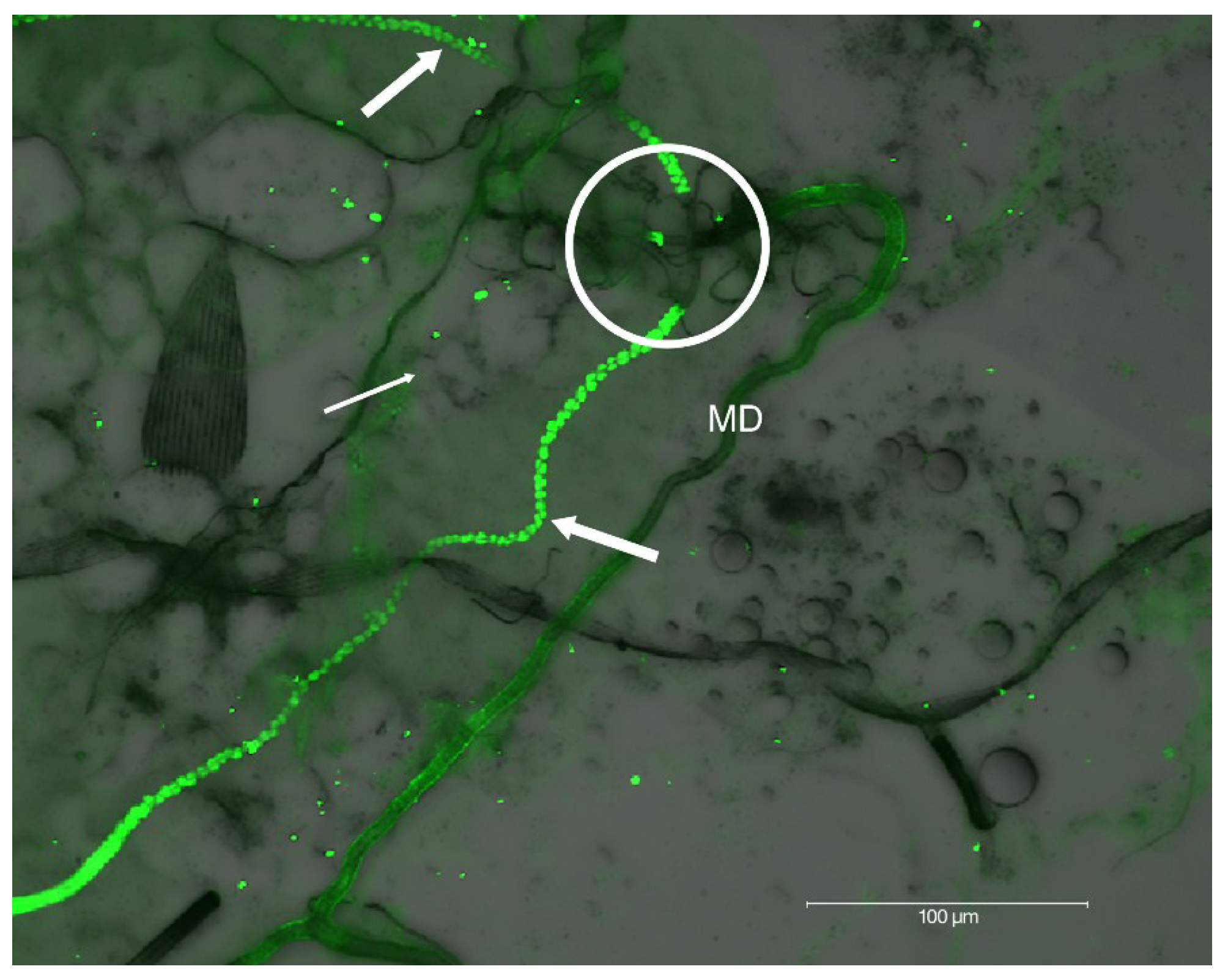
2.2. Localization of HSPG to Salivary Glands
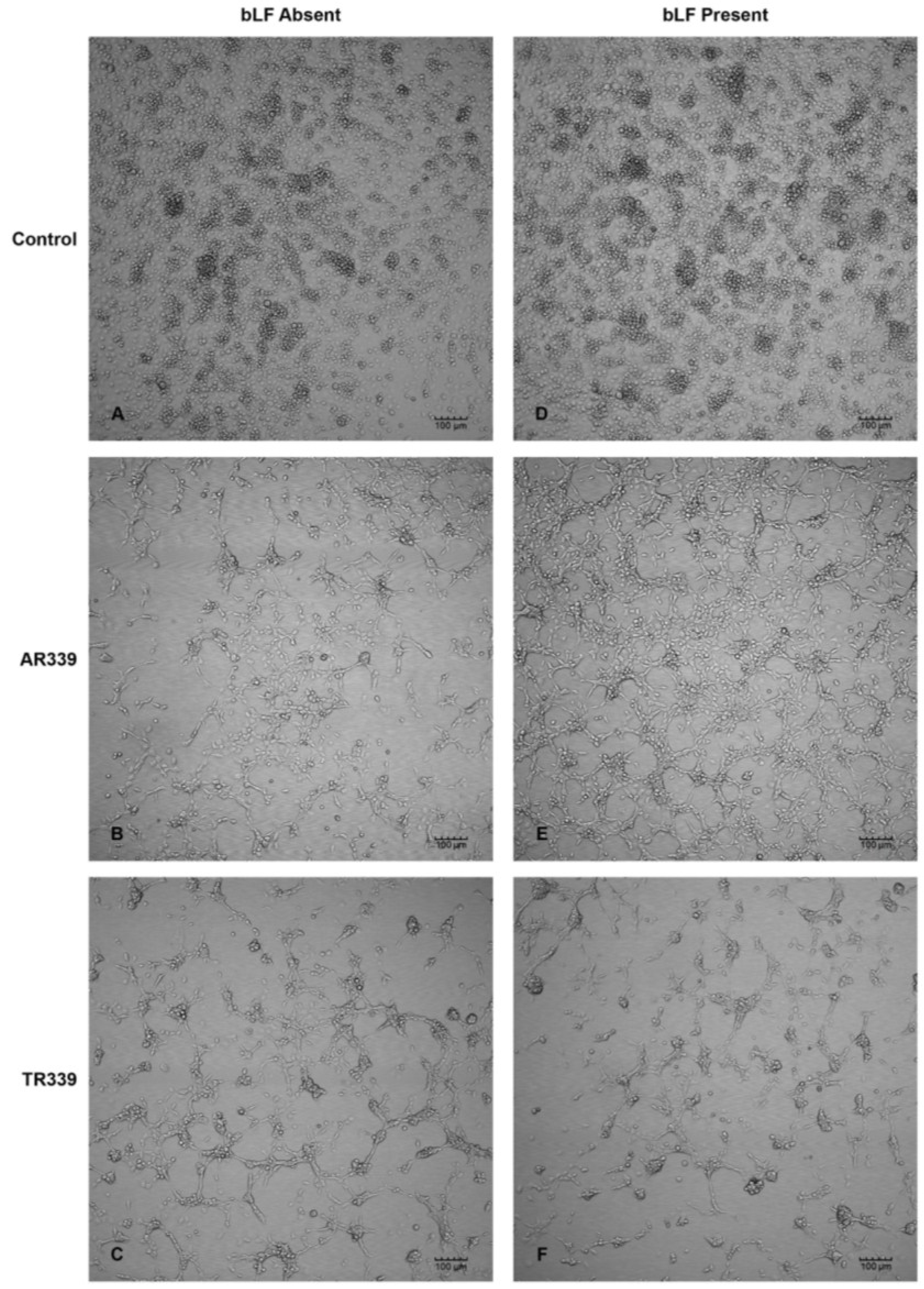
2.3. Lactoferrin Inhibition of SINV Infection in C7-10 Cells
2.4 Confocal Analysis of Plaque Assays and Salivary Glands
| 24 h p.i. | 48 h p.i. | 120 h p.i. | |
|---|---|---|---|
| No bLF | 1 small foci * | 3 medium plaques | 3 large plaques |
| 200 µg/mL bLF | No plaques | No plaques | 4 small plaques |
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Virus Production
4.3. Growth and Maintenance of Colony Mosquitoes
4.4. Artificial Membrane Feeding and Virus Infection of Mosquitoes
4.5. Leg Assay for Determination of Virus Dissemination
4.6. Salivary Gland Isolation and Localization of HSPG
4.7. Lactoferrin Inhibition by CPE Assay in C7-10 Cells
4.8. Lactoferrin Inhibition by Plaque Assay in C7-10 Cells
4.9. Fluorescent Virus Infection of Mosquitoes and Confocal Microscopy
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Kurkela, S.; Ratti, O.; Huhtamo, E.; Ucategui, N.; Nuotri, P.; Laakkonen, J.; Manni, T.; Helle, P.; Vaheri, A.; Vapalahti, O.; et al. Sindbis virus infection in resident birds, migratory birds, and humans, Finland. Emerg. Infect. Dis. 2008, 14, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Brummer-Korvenkontio, M.; Kuusisto, P. Has Western Finland been spared the “Pogosta”? Suom. Laak. 1981, 32, 2606–2607. [Google Scholar]
- Strauss, E.G.; Rice, C.M.; Strauss, J.H. Complete nucleotide sequence of the genomic RNA of Sindbis virus. Virology 1984, 133, 92–110. [Google Scholar] [CrossRef] [PubMed]
- Kurkela, S.; Manni, T.; Vaheri, A.; Vapalahti, O. Causative agent of Pogosta disease isolated from blood and skin lesions. Emerg. Infect. Dis. 2004, 10, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Doherty, R.L.; Carley, J.G.; Filippich, C.; Kay, B.H.; Gorman, B.M.; Rajapaksa, F. Isolation of Sindbis (Alphavirus) and Leanyer viruses from mosquitoes collected in the Northern Territory of Australia, 1974. Aust. J. Exp. Biol. Med. Sci. 1977, 55, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Yuill, T.M. The ecology of tropical arthropod-borne viruses. Ann. Rev. Ecol. Syst. 1986, 17, 189–219. [Google Scholar] [CrossRef]
- Schiefer, B.A.; Smith, J.R. Comparative susceptibility of eight mosquito species to Sindbis virus. Am. J. Trop. Med. Hyg. 1974, 23, 131–134. [Google Scholar] [PubMed]
- Chamberlin, R.W. Epidemiology of arthropod-borne Togaviruses: The role of arthropods as hosts and vectors and of vertebrate hosts in natural transmission cycles. In Togaviridae and Flaviridae; Schleisinger, S., Schleisinger, J.J., Eds.; Plenum Press: New York, NY, USA, 1980; pp. 175–227. [Google Scholar]
- Clements, A.N. Adult salivary glands and their secretions. In The Biology of Mosquitoes; Chapman and Hall: London, UK, 1996; pp. 251–262. [Google Scholar]
- Kelly, E.M.; Moon, D.C.; Bowers, D.F. Apoptosis in mosquito salivary glands: Sindbis virus-associated and tissue homeostasis. J. Gen. Virol. 2012, 93, 2419–2424. [Google Scholar] [CrossRef] [PubMed]
- Dhar, R.; Kumar, N. Role of mosquito salivary glands. Curr. Sci. 2003, 85, 1308–1313. [Google Scholar]
- Ribeiro, J.M.; Arca, B.; Lombardo, F.; Calvo, E.; Phan, V.M.; Chandra, P.K.; Wikel, S.K. An annotated catalogue of salivary gland transcripts in the adult female mosquito, Aedes aegypti. BMC Genomics 2007, 8. [Google Scholar] [CrossRef] [PubMed]
- Orr, C.W.; Hudson, A.; West, A.S. The salivary glands of Aedes aegypti. Histological-histochemical studies. Can. J. Zool. 1961, 39, 265–272. [Google Scholar] [CrossRef]
- Janzen, H.G.; Wright, K.A. The salivary glands of Aedes aegypti (L.): An electron microscope study. Can. J. Zool. 1971, 49, 1343–1346. [Google Scholar] [CrossRef] [PubMed]
- Bowers, D.F.; Abell, B.A.; Brown, D.T. Replication and tissue tropism of the alphavirus Sindbis in the mosquito Aedes albopictus. Virology 1995, 212, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bowers, D.F.; Coleman, C.G.; Brown, D.T. Sindbis virus-associated pathology in Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2003, 40, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.N.; Howley, P.M.; Griffin, D.E.; Lamb, R.A.; Martin, M.A.; Roizman, B.; Strausm, B.; Snipe, D.M. Fields Virology; Lippincott, Williams & Wilkins: New York, NY, USA, 2001. [Google Scholar]
- Klimstra, W.B.; Ryman, K.D.; Johnston, R.E. Adaptation of Sindbis virus to BHK cells selects for use of heparan sulfate as an attachment receptor. J. Virol. 1998, 72, 7357–7366. [Google Scholar] [PubMed]
- Waarts, B.L.; Onwuchekwa, J.C.; Aneke, J.M.; Kimata, K.; Bittman, R.; Meijer, K.F.; Wilschut, J. Antiviral activity of human lactoferrin: Inhibition of alphavirus interaction with heparan sulfate. Virology 2005, 333, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Sinnis, P.; Alida, C.; Toida, T.; Toyoda, H.; Kinoshita-Toyoda, A.; Xie, J.; Kemp, M.; Linhardt, R.J. Mosquito heparan sulfate and its potential role in malaria infection and transmission. J. Biol. Chem. 2007, 282, 25376–25384. [Google Scholar] [CrossRef] [PubMed]
- Van der Strate, B.W.; Beljaars, L.; Molema, G.; Harmsen, M.C.; Meijer, D.K. Antiviral activities of lactoferrin. Antivir. Res. 2001, 52, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Kanyshkova, T.G.; Buneva, V.N.; Nevinsky, G.A. Lactoferrin and its biological functions. Biochem. Biokhimiia 2001, 66, 1–7. [Google Scholar] [CrossRef]
- Karpf, A.R.; Lenches, E.; Strauss, E.G.; Strauss, J.H.; Brown, D.T. Superinfection exclusion of alphaviruses in three mosquito cell lines persistently infected with Sindbis virus. J. Virol. 1997, 71, 7119–7123. [Google Scholar] [PubMed]
- Sun, C.; Gardner, C.L.; Watson, A.M.; Ryman, K.D.; Klimstra, W.B. Stable, high-level expression of reporter proteins from improved alphavirus expression vectors to track replication and dissemination during encephalitic and arthritogenic disease. J. Virol. 2014, 88, 2035–2046. [Google Scholar] [CrossRef] [PubMed]
- Mims, C.A.; Day, M.F.; Marshall, I.D. Cytopathic effect of Semliki Forest Virus in the mosquito Aedes aegypti. Am. J. Trop. Med. Hyg. 1966, 15, 775–784. [Google Scholar] [PubMed]
- James, A.A.; Rossingol, P.A. Mosquito salivary glands: Parasitological and molecular aspects. Parasitol. Today 1991, 7, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Marinotti, O.; James, A.A. An alpha-glucosidase in the salivary glands of the vector mosquito Aedes aegypti. Insect Biochem. 1990, 6, 619–623. [Google Scholar] [CrossRef]
- Ribeiro, J.M.C. Characterization of a vasodilator from the salivary glands of the yellow fever mosquito Aedes aegypti. J. Exp. Biol. 1992, 165, 61–71. [Google Scholar] [PubMed]
- Byrnes, A.P.; Griffin, D.E. Binding of Sindbis virus to cell surface heparan sulfate. J. Virol. 1998, 72, 7349–7356. [Google Scholar] [PubMed]
- Bernard, K.A.; Klimstra, W.B.; Johnston, R.E. Mutations in the E2 glycoprotein of Venezuelan equine encephalitis virus confer heparan sulfate interaction, low morbidity, and rapid clearance from blood of mice. Virology 2000, 276, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Heil, M.L.; Albee, A.; Strauss, J.H.; Kuhn, R.J. An amino acid substitution in the coding region of the E2 glycoprotein adapts Ross River virus to utilize heparan sulfate as an attachment moiety. J. Virol. 2001, 75, 6303–6309. [Google Scholar] [CrossRef] [PubMed]
- Jackson, T.; Ellard, F.M.; Ghazaleh, R.A.; Brookes, S.M.; Blakemore, W.E.; Corteyn, A.H.; Stuart, D.I.; Newman, J.W.; King, A.M. Efficient infection of cells in culture by type O foot-and-mouth disease virus requires binding to cell surface heparan sulfate. J. Virol. 1996, 70, 5282–5287. [Google Scholar] [PubMed]
- Goodfellow, I.G.; Sioofy, A.B.; Powell, R.M.; Evans, D.J. Echoviruses bind heparan sulfate at the cell surface. J. Virol. 2001, 75, 4918–4921. [Google Scholar] [CrossRef] [PubMed]
- Trybala, E.; Roth, A.; Johansson, M.; Liljeqvist, J.A.; Rekabdar, E.; Larm, O.; Bergstrom, T. Glycosaminoglycan-binding ability is a feature of wild-type strains of herpes simplex virus type 1. Virology 2002, 302, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Da Cunha Sais, T.; de Moraes, R.M.; Ribolla, P.E.; de Bianchi, A.G.; Marinotti, O.; Bijovsky, A.T. Morphological aspects of Culex quinquefasciatus salivary glands. Arthropod Struct. Dev. 2003, 32, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, P.A.; Spielman, A. Fluid transport across the ducts of the salivary glands of a mosquito. J. Insect Physiol. 1982, 28, 579–581. [Google Scholar] [CrossRef]
- Muangman, D. Studies on discharged Sindbis virus from the mouthparts and anus of infected Aedes aegypti. D.P.H. Thesis, Harvard School of Public Health, Boston, MA, USA, 1968. [Google Scholar]
- Chien, Y.-J.; Chen, W.-J.; Hsu, W.-L.; Chiou, S.-S. Bovine lactoferrin inhibits Japanese encephalitis virus by binding to heparan sulfate and receptor for low density lipoprotein. Virology 2008, 19, 143–151. [Google Scholar] [CrossRef]
- Carvalho, C.A.M.; Sousa, I.P., Jr.; Silva, J.L.; Oliveira, A.C.; Goncalves, R.B.; Gomes, A.M.O. Inhibition of Mayaro virus infection by bovine lactoferrin. Virology 2014, 452, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Renz, D.; Brown, D.T. Characteristics of Sindbis virus temperature-sensitive mutants in cultured BHK-21 and Aedes albopictus (mosquito) cells. J. Virol. 1976, 19, 775–781. [Google Scholar] [PubMed]
- Miller, M.L.; Brown, D.T. Morphogenesis of Sindbis virus in three subclones of Aedes albopictus (mosquito) cells. J. Virol. 1992, 66, 4180–4190. [Google Scholar] [PubMed]
- Gerberg, E.J. Manual for Mosquito Rearing and Experimental Techniques; American Mosquito Control Association, Inc.: Baltimore, MD, USA, 1970; pp. 2–53. [Google Scholar]
- Mustermann, L.E.; Wasmuth, L.M. Handbook of Insect Rearing; Singh, P., Moore, R.F., Eds.; Elsevier: Amsterdam, The Netherlands, 1985; Volume 2, pp. 15–24. [Google Scholar]
- Lyski, Z.L.; Saredy, J.J.; Ciano, K.A.; Stem, J.; Bowers, D.F. Blood feeding position increases success of recalcitrant mosquitoes. Vector-Borne Zoonotic Dis. 2011, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Turell, M.J.; Bailey, C.L.; Beaman, J.R. Vector competence of a Houston, Texas strain of Aedes albopictus for Rift Valley fever virus. J. Am. Mosq. Cont. Assoc. 1988, 4, 94–96. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciano, K.A.; Saredy, J.J.; Bowers, D.F. Heparan Sulfate Proteoglycan: An Arbovirus Attachment Factor Integral to Mosquito Salivary Gland Ducts. Viruses 2014, 6, 5182-5197. https://doi.org/10.3390/v6125182
Ciano KA, Saredy JJ, Bowers DF. Heparan Sulfate Proteoglycan: An Arbovirus Attachment Factor Integral to Mosquito Salivary Gland Ducts. Viruses. 2014; 6(12):5182-5197. https://doi.org/10.3390/v6125182
Chicago/Turabian StyleCiano, Kristen A., Jason J. Saredy, and Doria F. Bowers. 2014. "Heparan Sulfate Proteoglycan: An Arbovirus Attachment Factor Integral to Mosquito Salivary Gland Ducts" Viruses 6, no. 12: 5182-5197. https://doi.org/10.3390/v6125182
APA StyleCiano, K. A., Saredy, J. J., & Bowers, D. F. (2014). Heparan Sulfate Proteoglycan: An Arbovirus Attachment Factor Integral to Mosquito Salivary Gland Ducts. Viruses, 6(12), 5182-5197. https://doi.org/10.3390/v6125182




