CD8 and CD4 T Cells in West Nile Virus Immunity and Pathogenesis
Abstract
:1. Introduction
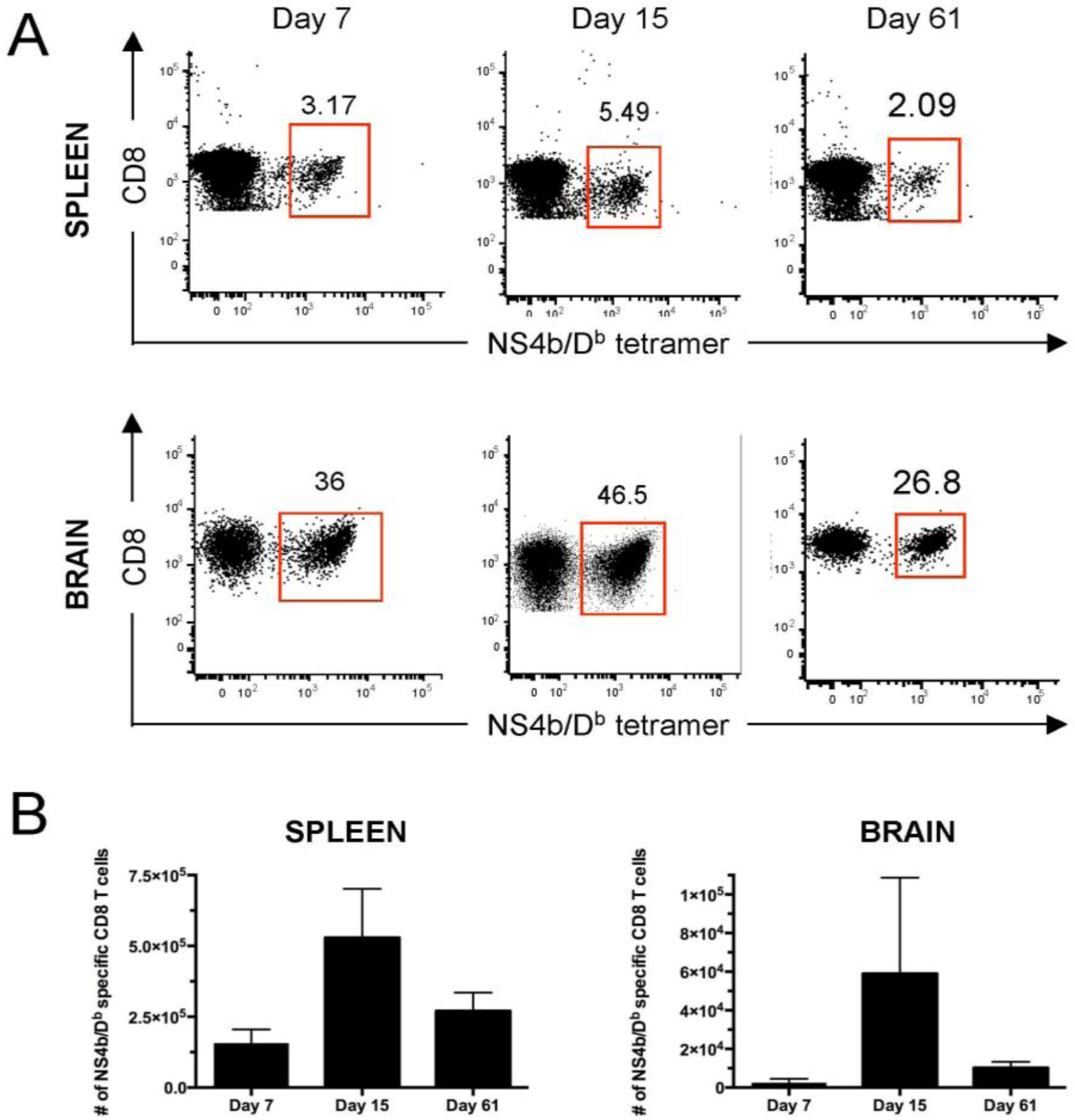
2. CD8 T Cells
3. CD4 T Cells
4. Innate Immune Signaling and T Cells
5. Conclusions
Conflicts of Interest
References and Notes
- Asnis, D.S.; Conetta, R.; Teixeira, A.A.; Waldman, G.; Sampson, B.A. The West Nile Virus outbreak of 1999 in New York: The Flushing Hospital experience. Clin. Infect. Dis. 2000, 30, 413–418. [Google Scholar] [CrossRef]
- Dauphin, G.; Zientara, S.; Zeller, H.; Murgue, B. West Nile: Worldwide current situation in animals and humans. Comp. Immunol. Microbiol. Infect. Dis. 2004, 27, 343–355. [Google Scholar] [CrossRef]
- Hubalek, Z.; Halouzka, J. West Nile fever—A reemerging mosquito-borne viral disease in Europe. Emerg. Infect. Dis. 1999, 5, 643–650. [Google Scholar] [CrossRef]
- Murray, K.; Baraniuk, S.; Resnick, M.; Arafat, R.; Kilborn, C.; Cain, K.; Shallenberger, R.; York, T.L.; Martinez, D.; Hellums, J.S.; et al. Risk factors for encephalitis and death from West Nile virus infection. Epidemiol. Infect. 2006, 134, 1325–1332. [Google Scholar] [CrossRef]
- Nash, D.; Mostashari, F.; Fine, A.; Miller, J.; O'Leary, D.; Murray, K.; Huang, A.; Rosenberg, A.; Greenberg, A.; Sherman, M.; et al. The outbreak of West Nile virus infection in the New York City area in 1999. N. Engl. J. Med. 2001, 344, 1807–1814. [Google Scholar] [CrossRef]
- Suthar, M.S.; Diamond, M.S.; Gale, M., Jr. West Nile virus infection and immunity. Nat. Rev. Microbiol. 2013, 11, 115–128. [Google Scholar] [CrossRef]
- Engle, M.J.; Diamond, M.S. Antibody prophylaxis and therapy against West Nile virus infection in wild-type and immunodeficient mice. J. Virol. 2003, 77, 12941–12949. [Google Scholar] [CrossRef]
- Brien, J.D.; Uhrlaub, J.L.; Nikolich-Zugich, J. Protective capacity and epitope specificity of CD8(+) T cells responding to lethal West Nile virus infection. Eur. J. Immunol. 2007, 37, 1855–1863. [Google Scholar]
- Shrestha, B.; Diamond, M.S. Role of CD8+ T cells in control of West Nile virus infection. J. Virol. 2004, 78, 8312–8321. [Google Scholar] [CrossRef]
- Wang, Y.; Lobigs, M.; Lee, E.; Mullbacher, A. CD8+ T cells mediate recovery and immunopathology in West Nile virus encephalitis. J. Virol. 2003, 77, 13323–13334. [Google Scholar] [CrossRef]
- Szretter, K.J.; Daniels, B.P.; Cho, H.; Gainey, M.D.; Yokoyama, W.M.; Gale, M., Jr.; Virgin, H.W.; Klein, R.S.; Sen, G.C.; Diamond, M.S. 2'-O methylation of the viral mRNA cap by West Nile virus evades ifit1-dependent and -independent mechanisms of host restriction in vivo. PLoS Pathog. 2012, 8, e1002698. [Google Scholar] [CrossRef]
- Friese, M.A.; Fugger, L. Pathogenic CD8(+) T cells in multiple sclerosis. Ann. Neurol. 2009, 66, 132–141. [Google Scholar] [CrossRef]
- Hafalla, J.C.; Cockburn, I.A.; Zavala, F. Protective and pathogenic roles of CD8+ T cells during malaria infection. Parasite Immunol. 2006, 28, 15–24. [Google Scholar] [CrossRef]
- Guidotti, L.G.; Chisari, F.V. Immunobiology and pathogenesis of viral hepatitis. Annu. Rev. Pathol. 2006, 1, 23–61. [Google Scholar] [CrossRef]
- Harty, J.T.; Badovinac, V.P. Influence of effector molecules on the CD8(+) T cell response to infection. Curr. Opin. Immunol. 2002, 14, 360–365. [Google Scholar] [CrossRef]
- Russell, J.H.; Ley, T.J. Lymphocyte-mediated cytotoxicity. Annu. Rev. Immunol. 2002, 20, 323–370. [Google Scholar]
- Shresta, S.; Pham, C.T.; Thomas, D.A.; Graubert, T.A.; Ley, T.J. How do cytotoxic lymphocytes kill their targets? Curr. Opin. Immunol. 1998, 10, 581–587. [Google Scholar] [CrossRef]
- Shrestha, B.; Samuel, M.A.; Diamond, M.S. CD8+ T cells require perforin to clear West Nile virus from infected neurons. J. Virol. 2006, 80, 119–129. [Google Scholar] [CrossRef]
- Shrestha, B.; Diamond, M.S. Fas ligand interactions contribute to CD8+ T-cell-mediated control of West Nile virus infection in the central nervous system. J. Virol. 2007, 81, 11749–11757. [Google Scholar] [CrossRef]
- Shrestha, B.; Pinto, A.K.; Green, S.; Bosch, I.; Diamond, M.S. CD8+ T cells use TRAIL to restrict West Nile virus pathogenesis by controlling infection in neurons. J. Virol. 2012, 86, 8937–8948. [Google Scholar] [CrossRef]
- Wang, Y.; Lobigs, M.; Lee, E.; Mullbacher, A. Exocytosis and Fas mediated cytolytic mechanisms exert protection from West Nile virus induced encephalitis in mice. Immunol. Cell. Biol. 2004, 82, 170–173. [Google Scholar] [CrossRef]
- Brien, J.D.; Uhrlaub, J.L.; Hirsch, A.; Wiley, C.A.; Nikolich-Zugich, J. Key role of T cell defects in age-related vulnerability to West Nile virus. J. Exp. Med. 2009, 206, 2735–2745. [Google Scholar] [CrossRef]
- Bartholdy, C.; Christensen, J.P.; Wodarz, D.; Thomsen, A.R. Persistent virus infection despite chronic cytotoxic T-lymphocyte activation in gamma interferon-deficient mice infected with lymphocytic choriomeningitis virus. J. Virol. 2000, 74, 10304–10311. [Google Scholar] [CrossRef]
- Cantin, E.; Tanamachi, B.; Openshaw, H. Role for gamma interferon in control of herpes simplex virus type 1 reactivation. J. Virol. 1999, 73, 3418–3423. [Google Scholar]
- Guidotti, L.G.; McClary, H.; Loudis, J.M.; Chisari, F.V. Nitric oxide inhibits hepatitis B virus replication in the livers of transgenic mice. J. Exp. Med. 2000, 191, 1247–1252. [Google Scholar] [CrossRef]
- Karupiah, G.; Chen, J.H.; Mahalingam, S.; Nathan, C.F.; MacMicking, J.D. Rapid interferon gamma-dependent clearance of influenza A virus and protection from consolidating pneumonitis in nitric oxide synthase 2-deficient mice. J. Exp. Med. 1998, 188, 1541–1546. [Google Scholar] [CrossRef]
- Nansen, A.; Jensen, T.; Christensen, J.P.; Andreasen, S.O.; Ropke, C.; Marker, O.; Thomsen, A.R. Compromised virus control and augmented perforin-mediated immunopathology in IFN-gamma-deficient mice infected with lymphocytic choriomeningitis virus. J. Immunol. 1999, 163, 6114–6122. [Google Scholar]
- Ramshaw, I.A.; Ramsay, A.J.; Karupiah, G.; Rolph, M.S.; Mahalingam, S.; Ruby, J.C. Cytokines and immunity to viral infections. Immunol. Rev. 1997, 159, 119–135. [Google Scholar] [CrossRef]
- Kitaura, K.; Fujii, Y.; Hayasaka, D.; Matsutani, T.; Shirai, K.; Nagata, N.; Lim, C.K.; Suzuki, S.; Takasaki, T.; Suzuki, R.; et al. High clonality of virus-specific T lymphocytes defined by TCR usage in the brains of mice infected with West Nile virus. J. Immunol. 2011, 187, 3919–3930. [Google Scholar] [CrossRef]
- Lelic, A.; Verschoor, C.P.; Ventresca, M.; Parsons, R.; Evelegh, C.; Bowdish, D.; Betts, M.R.; Loeb, M.B.; Bramson, J.L. The polyfunctionality of human memory CD8+ T cells elicited by acute and chronic virus infections is not influenced by age. PLoS Pathog. 2012, 8, e1003076. [Google Scholar] [CrossRef]
- Shrestha, B.; Wang, T.; Samuel, M.A.; Whitby, K.; Craft, J.; Fikrig, E.; Diamond, M.S. Gamma interferon plays a crucial early antiviral role in protection against West Nile virus infection. J. Virol. 2006, 80, 5338–5348. [Google Scholar]
- Wang, T.; Scully, E.; Yin, Z.; Kim, J.H.; Wang, S.; Yan, J.; Mamula, M.; Anderson, J.F.; Craft, J.; Fikrig, E. IFN-gamma-producing gamma delta T cells help control murine West Nile virus infection. J. Immunol. 2003, 171, 2524–2531. [Google Scholar]
- Miller, R.A. The aging immune system: Primer and prospectus. Science 1996, 273, 70–74. [Google Scholar]
- Nikolich-Zugich, J. T cell aging: Naive but not young. J. Exp. Med. 2005, 201, 837–840. [Google Scholar] [CrossRef]
- Uhrlaub, J.L.; Brien, J.D.; Widman, D.G.; Mason, P.W.; Nikolich-Zugich, J. Repeated in vivo stimulation of T and B cell responses in old mice generates protective immunity against lethal West Nile virus encephalitis. J. Immunol. 2011, 186, 3882–3891. [Google Scholar] [CrossRef]
- Piazza, P.; McMurtrey, C.P.; Lelic, A.; Cook, R.L.; Hess, R.; Yablonsky, E.; Borowski, L.; Loeb, M.B.; Bramson, J.L.; Hildebrand, W.H.; et al. Surface phenotype and functionality of WNV specific T cells differ with age and disease severity. PLoS One 2010, 5, e15343. [Google Scholar] [CrossRef]
- Parsons, R.; Lelic, A.; Hayes, L.; Carter, A.; Marshall, L.; Evelegh, C.; Drebot, M.; Andonova, M.; McMurtrey, C.; Hildebrand, W.; et al. The memory T cell response to West Nile virus in symptomatic humans following natural infection is not influenced by age and is dominated by a restricted set of CD8+ T cell epitopes. J. Immunol. 2008, 181, 1563–1572. [Google Scholar]
- Stewart, B.S.; Demarest, V.L.; Wong, S.J.; Green, S.; Bernard, K.A. Persistence of virus-specific immune responses in the central nervous system of mice after West Nile virus infection. BMC Immunol. 2011, 12, e6. [Google Scholar] [CrossRef]
- Larsen, M.V.; Lelic, A.; Parsons, R.; Nielsen, M.; Hoof, I.; Lamberth, K.; Loeb, M.B.; Buus, S.; Bramson, J.; Lund, O. Identification of CD8+ T cell epitopes in the West Nile virus polyprotein by reverse-immunology using NetCTL. PLoS One 2010, 5, e12697. [Google Scholar] [CrossRef]
- McMurtrey, C.P.; Lelic, A.; Piazza, P.; Chakrabarti, A.K.; Yablonsky, E.J.; Wahl, A.; Bardet, W.; Eckerd, A.; Cook, R.L.; Hess, R.; et al. Epitope discovery in West Nile virus infection: Identification and immune recognition of viral epitopes. Proc. Natl. Acad. Sci. USA 2008, 105, 2981–2986. [Google Scholar] [CrossRef]
- Kaabinejadian, S.; Piazza, P.A.; McMurtrey, C.P.; Vernon, S.R.; Cate, S.J.; Bardet, W.; Schafer, F.B.; Jackson, K.W.; Campbell, D.M.; Buchli, R.; et al. Identification of class I HLA T cell control epitopes for West Nile virus. PLoS One 2013, 8, e66298. [Google Scholar]
- Gardner, I.A.; Wong, S.J.; Ferraro, G.L.; Balasuriya, U.B.; Hullinger, P.J.; Wilson, W.D.; Shi, P.Y.; MacLachlan, N.J. Incidence and effects of West Nile virus infection in vaccinated and unvaccinated horses in California. Vet. Res. 2007, 38, 109–116. [Google Scholar] [CrossRef]
- Ng, T.; Hathaway, D.; Jennings, N.; Champ, D.; Chiang, Y.W.; Chu, H.J. Equine vaccine for West Nile virus. Dev. Biol. (Basel) 2003, 114, 221–227. [Google Scholar]
- Kim, S.; Li, L.; McMurtrey, C.P.; Hildebrand, W.H.; Weidanz, J.A.; Gillanders, W.E.; Diamond, M.S.; Hansen, T.H. Single-chain HLA-A2 MHC trimers that incorporate an immundominant peptide elicit protective T cell immunity against lethal West Nile virus infection. J. Immunol. 2010, 184, 4423–4430. [Google Scholar] [CrossRef]
- Shrestha, B.; Ng, T.; Chu, H.J.; Noll, M.; Diamond, M.S. The relative contribution of antibody and CD8+ T cells to vaccine immunity against West Nile encephalitis virus. Vaccine 2008, 26, 2020–2033. [Google Scholar] [CrossRef]
- Brien, J.D.; Uhrlaub, J.L.; Nikolich-Zugich, J. West Nile virus-specific CD4 T cells exhibit direct antiviral cytokine secretion and cytotoxicity and are sufficient for antiviral protection. J. Immunol. 2008, 181, 8568–8575. [Google Scholar]
- Durrant, D.M.; Robinette, M.L.; Klein, R.S. IL-1R1 is required for dendritic cell-mediated T cell reactivation within the CNS during West Nile virus encephalitis. J. Exp. Med. 2013, 210, 503–516. [Google Scholar] [CrossRef]
- Keynan, Y.; Card, C.M.; McLaren, P.J.; Dawood, M.R.; Kasper, K.; Fowke, K.R. The role of regulatory T cells in chronic and acute viral infections. Clin. Infect. Dis. 2008, 46, 1046–1052. [Google Scholar] [CrossRef]
- Lanteri, M.C.; O'Brien, K.M.; Purtha, W.E.; Cameron, M.J.; Lund, J.M.; Owen, R.E.; Heitman, J.W.; Custer, B.; Hirschkorn, D.F.; Tobler, L.H.; et al. Tregs control the development of symptomatic West Nile virus infection in humans and mice. J. Clin. Invest. 2009, 119, 3266–3277. [Google Scholar]
- Suthar, M.S.; Ma, D.Y.; Thomas, S.; Lund, J.M.; Zhang, N.; Daffis, S.; Rudensky, A.Y.; Bevan, M.J.; Clark, E.A.; Kaja, M.K.; et al. IPS-1 is essential for the control of West Nile virus infection and immunity. PLoS Pathog. 2010, 6, e1000757. [Google Scholar] [CrossRef]
- Belkaid, Y.; Piccirillo, C.A.; Mendez, S.; Shevach, E.M.; Sacks, D.L. CD4+CD25+ regulatory T cells control Leishmania major persistence and immunity. Nature 2002, 420, 502–507. [Google Scholar] [CrossRef]
- Rouse, B.T.; Sarangi, P.P.; Suvas, S. Regulatory T cells in virus infections. Immunol. Rev. 2006, 212, 272–286. [Google Scholar] [CrossRef]
- Belkaid, Y.; Rouse, B.T. Natural regulatory T cells in infectious disease. Nat. Immunol. 2005, 6, 353–360. [Google Scholar] [CrossRef]
- Lund, J.M.; Hsing, L.; Pham, T.T.; Rudensky, A.Y. Coordination of early protective immunity to viral infection by regulatory T cells. Science 2008, 320, 1220–1224. [Google Scholar] [CrossRef]
- Daffis, S.; Suthar, M.S.; Szretter, K.J.; Gale, M., Jr.; Diamond, M.S. Induction of IFN-beta and the innate antiviral response in myeloid cells occurs through an IPS-1-dependent signal that does not require IRF-3 and IRF-7. PLoS Pathog. 2009, 5, e1000607. [Google Scholar] [CrossRef]
- Kohlmeier, J.E.; Cookenham, T.; Roberts, A.D.; Miller, S.C.; Woodland, D.L. Type I interferons regulate cytolytic activity of memory CD8(+) T cells in the lung airways during respiratory virus challenge. Immunity 2010, 33, 96–105. [Google Scholar] [CrossRef]
- Quigley, M.; Huang, X.; Yang, Y. STAT1 signaling in CD8 T cells is required for their clonal expansion and memory formation following viral infection in vivo. J. Immunol. 2008, 180, 2158–2164. [Google Scholar]
- Pinto, A.K.; Daffis, S.; Brien, J.D.; Gainey, M.D.; Yokoyama, W.M.; Sheehan, K.C.; Murphy, K.M.; Schreiber, R.D.; Diamond, M.S. A temporal role of type I interferon signaling in CD8+ T cell maturation during acute West Nile virus infection. PLoS Pathog. 2011, 7, e1002407. [Google Scholar] [CrossRef]
- Ramos, H.J.; Lanteri, M.C.; Blahnik, G.; Negash, A.; Suthar, M.S.; Brassil, M.M.; Sodhi, K.; Treuting, P.M.; Busch, M.P.; Norris, P.J.; et al. 1L-1beta signaling promotes CNS-intrinsic immune control of West Nile virus infection. PLoS Pathog. 2012, 8, e1003039. [Google Scholar] [CrossRef]
- Alexopoulou, L.; Holt, A.C.; Medzhitov, R.; Flavell, R.A. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 2001, 413, 732–738. [Google Scholar] [CrossRef]
- Bieback, K.; Lien, E.; Klagge, I.M.; Avota, E.; Schneider-Schaulies, J.; Duprex, W.P.; Wagner, H.; Kirschning, C.J.; Ter Meulen, V.; Schneider-Schaulies, S. Hemagglutinin protein of wild-type measles virus activates toll-like receptor 2 signaling. J. Virol. 2002, 76, 8729–8736. [Google Scholar] [CrossRef]
- Edelmann, K.H.; Richardson-Burns, S.; Alexopoulou, L.; Tyler, K.L.; Flavell, R.A.; Oldstone, M.B. Does Toll-like receptor 3 play a biological role in virus infections? Virology 2004, 322, 231–238. [Google Scholar] [CrossRef]
- Kurt-Jones, E.A.; Popova, L.; Kwinn, L.; Haynes, L.M.; Jones, L.P.; Tripp, R.A.; Walsh, E.E.; Freeman, M.W.; Golenbock, D.T.; Anderson, L.J.; et al. Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nat. Immunol. 2000, 1, 398–401. [Google Scholar] [CrossRef]
- Rassa, J.C.; Meyers, J.L.; Zhang, Y.; Kudaravalli, R.; Ross, S.R. Murine retroviruses activate B cells via interaction with toll-like receptor 4. Proc. Natl. Acad. Sci. USA 2002, 99, 2281–2286. [Google Scholar] [CrossRef]
- Town, T.; Jeng, D.; Alexopoulou, L.; Tan, J.; Flavell, R.A. Microglia recognize double-stranded RNA via TLR3. J. Immunol. 2006, 176, 3804–3812. [Google Scholar]
- Wang, T.; Town, T.; Alexopoulou, L.; Anderson, J.F.; Fikrig, E.; Flavell, R.A. Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat. Med. 2004, 10, 1366–1373. [Google Scholar] [CrossRef]
- Szretter, K.J.; Daffis, S.; Patel, J.; Suthar, M.S.; Klein, R.S.; Gale, M., Jr.; Diamond, M.S. The innate immune adaptor molecule MyD88 restricts West Nile virus replication and spread in neurons of the central nervous system. J. Virol. 2010, 84, 12125–12138. [Google Scholar]
- Town, T.; Bai, F.; Wang, T.; Kaplan, A.T.; Qian, F.; Montgomery, R.R.; Anderson, J.F.; Flavell, R.A.; Fikrig, E. Toll-like receptor 7 mitigates lethal West Nile encephalitis via interleukin 23-dependent immune cell infiltration and homing. Immunity 2009, 30, 242–253. [Google Scholar] [CrossRef]
- Lazear, H.M.; Pinto, A.K.; Ramos, H.J.; Vick, S.C.; Shrestha, B.; Suthar, M.S.; Gale, M., Jr.; Diamond, M.S. The pattern recognition receptor MDA5 modulates CD8+ T cell-dependent clearance of West Nile virus from the central nervous system. J. Virol. 2013, 87, 11401–11415. [Google Scholar] [CrossRef]
- Brien, J.D.; Daffis, S.; Lazear, H.M.; Cho, H.; Suthar, M.S.; Gale, M., Jr.; Diamond, M.S. Interferon regulatory factor-1 (IRF-1) shapes both innate and CD8(+) T cell immune responses against West Nile virus infection. PLoS Pathog. 2011, 7, e1002230. [Google Scholar] [CrossRef]
- Suthar, M.S.; Ramos, H.J.; Brassil, M.M.; Netland, J.; Chappell, C.P.; Blahnik, G.; McMillan, A.; Diamond, M.S.; Clark, E.A.; Bevan, M.J.; et al. The RIG-I-like receptor LGP2 controls CD8(+) T cell survival and fitness. Immunity 2012, 37, 235–248. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Netland, J.; Bevan, M.J. CD8 and CD4 T Cells in West Nile Virus Immunity and Pathogenesis. Viruses 2013, 5, 2573-2584. https://doi.org/10.3390/v5102573
Netland J, Bevan MJ. CD8 and CD4 T Cells in West Nile Virus Immunity and Pathogenesis. Viruses. 2013; 5(10):2573-2584. https://doi.org/10.3390/v5102573
Chicago/Turabian StyleNetland, Jason, and Michael J. Bevan. 2013. "CD8 and CD4 T Cells in West Nile Virus Immunity and Pathogenesis" Viruses 5, no. 10: 2573-2584. https://doi.org/10.3390/v5102573
APA StyleNetland, J., & Bevan, M. J. (2013). CD8 and CD4 T Cells in West Nile Virus Immunity and Pathogenesis. Viruses, 5(10), 2573-2584. https://doi.org/10.3390/v5102573



