Abstract
As an early response to infection, cells induce a profile of the early inflammatory proteins including antiviral cytokines and chemokines. Two families of transcriptional factors play a major role in the transcriptional activation of the early inflammatory genes: The well-characterized family of NFkB factors and the family of interferon regulatory factors (IRF). The IRFs play a critical role in the induction of type I interferon (IFN) and chemokine genes, as well as genes mediating antiviral, antibacterial, and inflammatory responses. Type I IFNs represent critical components of innate antiviral immunity. These proteins not only exert direct antiviral effects, but also induce maturation of dendritic cells (DC), and enhance functions of NK, T and B cells, and macrophages. This review will summarize the current knowledge of the mechanisms leading to the innate antiviral response with a focus on its role in the regulation of HIV-1 infection and pathogenicity. We would like this review to be both historical and a future perspective.
1. Introduction
The ability of the host to detect invasion by a pathogenic intruder and to activate defense mechanisms to eliminate infection is essential for survival. The innate immune response has developed as a rapid and regulated defense mechanism against invading pathogens. Remarkable progress has been made in recent years into the identification of cellular receptors detecting the invading pathogens as well as in understanding the signaling pathways leading to the induction of Type I IFN genes and inflammatory cytokines. It has been shown that the recognition of an invading pathogenic organism can occur upon binding of viral nucleic acids or envelope proteins to specific membrane associate receptors, denoted Toll-like (TLR) receptors, which can recognize conserved patterns of proteins, lipoproteins, and viral RNA [1]. In addition, cytoplasmic sensors can recognize viral RNA or DNA in the B configuration of the invading pathogens. In addition, the cellular polymerase III that converts cytoplasmic DNA into 5’phosphorylated RNA recognized by RIGI is also an important component of the innate virus detection system. The binding of viral ligands to host sensors induces multiple signaling pathways that activate cellular transcription factors controlling the expression of a diverse set of genes, which in turn coordinate both the innate and adaptive immune responses. Although cascades of multiple kinases and ubiquitination steps usually mediate activation of these transcription factors, their functional diversity is modulated by interaction with other transcription factors and cofactors. Indeed, these regulatory networks are critical components of the host defense against invading pathogens, including viruses.
In response to infection, cells induce early inflammatory proteins including type I IFN and inflammatory cytokines. Two families of transcriptional factors play a major role in the stimulation of expression of these proteins: the well-characterized family of NFkB factors and the family of interferon regulatory factors (IRF). The IRFs play a critical role in the induction of Type I interferon (IFN) and chemokines as well as proteins mediating antiviral, antibacterial, and inflammatory responses [2,3]. Type I IFNs are critical components of the innate antiviral response. These proteins not only exert direct antiviral effects, but also induce maturation of dendritic cells (DC), recruitment of the immune cells to the sites of infection, and enhance the functions of macrophages, NK, T and B cells, and macrophages [4]. The importance of Type I IFN to the activation of effector cell populations and adaptive immunity is also emerging. This chapter will review the current knowledge of the molecular mechanisms leading to the innate antiviral response with a focus on its role in HIV-1 infection. We would like this review to have both a historical and a future perspective.
2. Transcription Factors of the IRF Family
The IRFs are transcriptional mediators of virus, bacteria and IFN-induced signaling pathways and as such play a critical role in antiviral defense, immune response, cell growth regulation and apoptosis. To date, nine human cellular IRF genes (IRF-1, IRF-2, IRF-3, IRF-4/Pip/ICSAT, IRF-5, IRF-6, IRF-7, ICSBP/IRF-8 and ISGF3γ/p48/IRF-9), as well as virus-encoded analogues of cellular IRFs have been identified [5,6]. These factors can function as transcriptional activators (e.g., IRF-1, IRF-3, and IRF-9), repressors (e.g., IRF-8) or both (e.g., IRF-2, IRF-4, IRF-5). They all share significant homology in the N-terminal 115 amino acids, which comprise the DNA-binding domain, characterized by five tryptophan repeats. Three of these repeats contact DNA with specific recognition of 5’GAAA3’ and 5’AANNGAAA3’ sequences [7]. However, the unique function of a particular IRF is accounted for by cell type-specific expression, intrinsic transactivation potential, and an ability to interact with other members of the IRF family or other transcription factors and co-factors [8]. All IRFs, except IRF-1 and IRF-2, contain the IRF- associated domain (IAD) in the 3’ terminal part of the protein, which mediates these interactions.
The first IRFs, IRF-1 and IRF-2, were identified through their ability to bind to the virus responsive element (VRE) of the IfnB gene and were proposed to function as an activator and repressor of the IfnB gene, respectively [9]. However, homozygous deletion of IRF-1 in mice did not impair activation of IfnA or IfnB genes in infected mouse embryo fibroblasts (MEFs), while dsRNA-mediated induction of Type I IFN was down-regulated [10]. Subsequent studies have revealed that IRF-1 is involved in a broad spectrum of antiviral defenses mediated by IFN-γ by activation of the IfnG genes. Furthermore, analysis of the repertoire of lymphoid cells from mice devoid of IRF-1(Irf1 null) has shown defects in the maturation of CD8+ T cells as well as a defective Th1 response, impaired production of IL-12 in macrophages, and defective NK cell development.
Although IRF-1 stimulates expression of the IfnG gene, it does not have a critical role in viral mediated stimulation of Type I IfnB genes. However, subsequent identification of three IRFs (IRF-3, IRF-5 and IRF-7) showed that they are direct transducers of virus-mediated signaling and demonstrated their crucial role in the expression of Type I Ifn genes and some chemokines [6,11,12,13]. The identification of IRF-3 and IRF-7 and their role in the transcriptional activation of type I Ifn genes had a major impact on the understanding of the molecular mechanism of the pathogen-induced innate antiviral response [14,15,16,17]. In human cells, multiple spliced variants of these IRFs can be detected, and some of these variants can function as dominant negative mutants. In infected cells, the ubiquitously expressed IRF-3 mediates induction of IFNβ and of some IFN induced genes (ISG), whereas expression of IRF-5 and IRF-7 is limited largely to lymphoid cells, where they are required for the expression of the IfnA genes [3,14]. Reconstitution of IRF-5 or IRF-7 expression in infected fibroblasts that express only IFNβ confirmed expression of IFNα subtypes [18]. In many cells, IRF-3 and IRF-7 are involved in the amplification of the interferon response: antiviral response is generally induced through two sequential steps: (1) virus activates IRF-3, which leads to synthesis of IFNβ. (2) IFNβ stimulates transcription of IRF-7, which results in synthesis of IFNα and further enhancement of IFNβ synthesis [12].
High constitutive levels of both IRF-5 and IRF-7 were detected in plasmacytoid dendritic cells (PDC), which are high IFNα producers [19,20]. Subtypes of IfnA genes induced by IRF-5 and IRF-7 in B cells are distinct, indicating that these two factors have both essential and non-redundant functions [21]. IRF-7 expression is critical for induction of IfnA genes both in vitro and in vivo. Virus infection or CpG DNA was not able to stimulate expression of Type I Ifn genes in Irf7−/− mice. The authors concluded, therefore, that IRF-7 is the master regulator of type I IFN [22], although residual IFN production, mediated by IRF-3, could still be induced in cells of non-lymphoid origin. In contrast Irf5−/− mice showed not only a decrease in virus mediated induction of Type I IFN, but also a significant decrease in expression of inflammatory cytokines such as TNFα, IL-6 and IL-12 [23,24]. Furthermore, recent data indicate that IRF-5 has a critical role in the development of TH-1 responses to Leishmania donovani infection [25] as well as in the differentiation and function of B cells [26] and macrophages [27]. Thus, while the role of IRF-7 is critical for the innate antiviral response, IRF-5 function is broader, and can mediate the cross-talk between innate and adaptive immune responses.
2.1. The Viral IRF: KSHV-Encoded Viral IRF
Kaposi’s sarcoma-associated herpes virus (KSHV) is a member of the γ herpes virus family and is genetically similar to EBV and monkey Herpes Virus Saimiri (HVS) [28]. Sequence analysis of the KSHV genome revealed the presence of about 80 open reading frames (ORFs) and a number of ORFs showing homology to cellular genes including a cluster of four ORFs with homology to the cellular IRF family transcription factors [29], three of which have been cloned and characterized. The open reading frame 9 of KSHV genome (ORF K9)-encoded vIRF-1, has been studied most extensively and was shown to inhibit both virus-mediated induction of Type I Ifn genes and IFN-induced genes (ISG) [30,31,32,33,34].
vIRF-2 (ORF K11.1) encodes a small nuclear protein (163 aa) that unlike the cellular IRFs, binds dsRNA-activated protein kinase (PKR), inhibits its kinase activity and blocks the phosphorylation of the PKR substrate, eukaryotic translation initiation factor 2α [34,35].
vIRF-3/LANA2, encoded by ORFs K10.5 and K10.6 [36,37], is a multifunctional nuclear protein constitutively expressed in KSHV-positive B cell lymphoma cells (PEL) and Castleman’s disease tumors [38]. The vIRF-3 protein binds to IRF-3, IRF-7 and IRF-5 [37,38] vIRF-3 also interacts with two tumor suppressor genes, p53 and MM-1 [39].
2.2. IRF Mediated Cross Talk between Innate and Adaptive Immune Response
The observation that some IRFs also have an essential role in the differentiation and function of lymphoid cells emerged mainly from a large number of studies in genetically modified mice. Thus, IRF-1 participates not only in the induction of the IfnG gene, but also in several autocrine loops that induce and enhance the Th1 response [40,41]. IRF-4 has been also associated with Th cell development and the differentiation of B cells to plasma cells [42]; Irf4−/− mice show a complete absence of plasma cells [43]. Irf8−/− mice show major defects in CD8+DC and plasmacytoid dendritic cells (PDC). These mice also display increased susceptibility to infection, which is due to a defect in the Th1 immune response and an inability to express IL-12 [44,45]. IRF-3 and IRF-7 play essential roles in the antiviral response, however IRF-7 also has an important role in the differentiation of monocytes to macrophages [46]. The Th1 and Th2 responses of Irf3−/− or Irf7−/− mice have yet to be characterized, however a DNA-mediated Th1 immune response was enhanced by co-expression of IRF-3 and IRF-7 [47]. IRF-5 plays an important role in B cell differentiation and function in vitro. Infected B cells over expressing IRF-5 show up-regulated expression of several immune response genes, including CD80, MyD88 and ISG [21], and IRF-5 promotes macrophage polarization and TH-1 response [25,27]. Irf5−/− mice show a defect in differentiation of B cells to plasma cells and an attenuated response to antigenic stimulation [26]. IRF-9 is an important component of the ISGF-3 complex with an essential role in the type I IFN signaling pathway and induction of many ISGs including IRF-7 and IRF-5 [48]. The Ifr9−/− mice show a significant defect in Type I IFN signaling and share many characteristics with the Stat1−/− and InfR1−/− mice. These mice are also predicted to be defective in expression of many ISGs including both IRF-7 and IRF-5. However, whether they show modulation of the adaptive immune response similar to Irf5−/− mice has yet to be evaluated.
6. Role of the Innate Immune Response in HIV-1 Infection
It has been well established that HIV-1 infection is associated with the modulation and activation of the immune system [117], characterized by a progressive decline and dysfunction of T cells, impaired function of NK cells, and polyclonal activation of B cells. The levels of immune activation are an indication of the progression of the HIV-1 disease. When the cytokine response induced during the early stages of HIV-1 was evaluated by the analysis of cytokine plasma levels or cytokine expression in PBMC, in most cases there was up-regulation of cytokines and chemokines including IFN, IL-1 and TNFα and decreased levels of IL-6 and IL-2. Furthermore, the expression of Type I IFNβ was transient and two consecutive peaks in cytokine levels were observed. The first peak, which occurred at about 6 days after the start of viremia, was transient and showed an increase in IFN-α, IL-15, TNF-α, IP-10, and MCP-1, and the second peak, associated with production of IL-6, IL-8, IL-10, IL-18, and IFN-γ, was detected 7 to 12 days post infection [118]. This data indicate that the early steps of HIV1 infection are associated with activation of the innate antiviral and inflammatory response prior to development of the adaptive immune response. Potential cross talk between innate and adaptive immunity in HIV-1 pathogenicity and its mechanism has not been investigated in detail. The profound innate inflammatory response to HIV-1 infection leading to the induction of an antiviral response may also have detrimental effects. The expression of pro-inflammatory cytokines may promote HIV-1 replication and suppress the antiviral effect of Type I IFN. Stimulation of HIV-1 replication by TNFα and IL-6 was shown in vitro and may also occur in vivo [119]. Another factor of the innate immune system, IRF-1, was recently implicated in the regulation of HIV-1 infection. IRF-1 is expressed constitutively at low levels, but its expression is strongly up-regulated by IFN γ. IRF-1 was also induced in T cells during early steps of HIV-1 infection [120] and the expression of IRF-1 was also observed in non-permissive HIV-1 infection of DC. In these cells, HIV-1 induced increased expression of IRF-1 and IRF-7, but the activation of IRF-3 was suppressed [121]. The 5’LTR region of HIV-1 contains an IRF-1 binding site, and its deletion impairs HIV-1 replication in vitro [122]. Interestingly, specific IRF1 polymorphisms have been found in a small group of people showing resistance to HIV-1 infection and pathogenicity (non progressors). This polymorphism was associated with the reduced levels of IRF-1 in PBMC, suggesting that the decrease in IRF1 expression may limit the susceptibility to HIV infection as IRF-1 contributes to stimulation of HIV-1 replication [123].
The detrimental role for immune activation in HIV infection is supported by animal studies. In the pathogenic SIV animal model of HIV, induction of the strong innate inflammatory response is characterized by production of Type I IFN during the very early stages of SIV infection; in contrast, in nonpathogenic SIV infection the induction of inflammation and Type I IFN is only transient [124].
6.1. HIV-1 Infection in vitro Induces Interferon Signature
In vitro, HIV-1 infects a number of diverse cell types; however the characteristics of the infections in these cells are distinct. Thus while infection in activated CD4+ T cells is productive and cytopathic, in naïve T cells HIV-1 is present in latent state. In macrophages HIV-1 replicates at low levels and does not induce cytopathic effects. Thus the innate antiviral response to HIV-1 infection in different cell types may be distinct. Several cell types, including CD4+ T cells, primary macrophages, and PBMC, infected with HIV-1 in vitro induced interferon signature, while induction of either IFNα or IFNβ was generally difficult to detect. Analysis of this typical interferon signature transcription profile in HIV infected CD4+T cells showed significant up-regulation of expression of interferon stimulated genes with increasing viral load, including genes of the intrinsic antiretroviral defense. Upon successful antiretroviral treatment, the signature profile of previously viremic individuals reverted to a pattern comparable to that of non progressors and uninfected individuals [125]. In contrast, when the transcription profile of lymph nodes from HIV-1 infected patients was analyzed, only about 5% of the identified genes were associated with the antiviral response and the majority of the genes were negatively associated with HIV-1 replication. Based on these data the authors concluded that the antiviral response is not sufficient to attenuate HIV-1 replication [126]. Recent data have shown up-regulation of type I IFN induced ISG in activated HIV-1+CD4+ T cells [127], indicating that only a subset of CD4+ cells can induce IFNα. However the IFN signature induced in PBMC can be also induced by exogenous Type I IFN produced by other types of cells such as PDC, which are high producers of IFNα. The interaction between HIV-1 and DC is complex and not yet fully understood. It was shown that PDC are recruited to the site of HIV-1 replication in HIV-positive lymph nodes and this recruitment has been facilitated by expression of increased levels of lymphocyte homing marker CCR7 on circulating DC of HIV-1 infected individuals [128]. Activated DC from HIV-1 infected individuals expressed high levels of IFNα and other inflammatory cytokines and these could well contribute to increased HIV-1 replication through immune activation.
The mechanism and the signaling pathway by which HIV-1 activates IFN production in dendritic cells has been subject of intensive studies. The PDC are an essential part of the innate immune system and the major producer of IFNα It was shown that the co-culture of PBMC with HIV-1 infected lymphocytes or infected CD4+ cells resulted in production of relative high levels of Type I IFN and that the major producers were the DC [118]. Production of IFNα was observed in lymph nodes of AIDS patients [128] and has been associated with disease progression [129,130]. Recent clinical data suggest that production of IFNα may drive the hyper activation of the immune system resulting in CD4 T cell depletion and AIDS [127]. Presently, it is assumed that IFNα alone contributes to the progression of AIDS disease, however a decrease in CD4+T cells is also associated with a significant increase in serum levels of inflammatory cytokines including TNFα. IL-6, IL-18 and IL-7 which may contribute to the observed hyper activation of the immune system.
The DC are generally resistant to HIV-1 infection, and co-culture of HIV-1 particles with monocyte derived dendritic cells (MDDC) neither infects DC or stimulates their activation. However when the freshly isolated DCs were infected with various HIV-1 strains in vivo, they were permissive for productive infection with the macrophage tropic HIV-1 (R5 HIV-1) and the infected DCs transferred HIV-1 to CD4 T cells [131]. It was shown that DC and T cells can form conjugates that facilitate transfer of HIV-1 from DC to T cells [132]. It was also shown that this transfer is bi-phasic [133].
As described above, cell recognize RNA viruses either by the membrane bound receptors such as TOLL receptors or cytoplasmic receptors such as RNA helicase RigI and MDA5. Which of these receptors are engaged upon HIV-1 infection has not been fully explored. However it was shown that the peptide inhibitors of TLR7 ligands inhibited IFN production in PBMC co-cultivated with HIV-1 infected T cells, indicating that the HIV-1 recognition is at least partially mediated by TLR7 [130,134]. The TLR7 independent pathway, which depends on IRF-3, was also induced in the co-culture experiments of HIV-1 infected T cells with TLR negative cells. Interestingly the induction of Type I IFN did not depend on HIV-1 replication in the infected cells, but was facilitated by the fusion of the infected T cells with DC [134]. It is possible that the fusion transfers large amounts of viral nucleic acids to the cytoplasm of DC, where the viral RNA may be recognized either by TLR7 or one of the cytoplasmic sensors such as Rig I or MDA5. Alternatively, the un-integrated proviral DNA could be recognized by one of the recently identified cytoplasmic DNA sensors. Further studies will certainly address these questions.
Another very interesting and novel aspect of the mechanism of IFNα induction in DC is the observation that the restriction of HIV-1 replication in DC can be overcome by the SIV encoded protein Vpx. Expression of this protein or co-infection with SIV (that encodes Vpx) allowed HIV-1 replication in DC, stimulated production of IFNα and activated DC [135]. It was also shown recently that Vpx induces proteasomal degradation of SAMHD1, a negative regulator of the interferon response. Silencing of SAMHD1 in non-permissive cell lines alleviated HIV-1 restriction and overexpression of SAMHD1 in sensitive cells attenuated HIV-1 replication [136]. Further experiments demonstrated requirement of an interaction of HIV-1 capsid with CYPA for IFN production [135]. The induction of IFN was also dependent on nuclear localization of phosphorylated IRF-3. Whether the phosphorylation and activation of IRF-3 in these cells depends entirely on the capsid interaction with cellular proteins or whether the requirement for the capsid interaction is connected to the TLR3 or RIG I- IRF-3 axis needs yet to be clarified. However it is unlikely that the SAMHD1 is the only factor that can modulate HIV-1 replication in DC. The attenuation of HIV-1 replication by the intrinsic cellular antiviral proteins such as APOBECC3G, TRIM and others [95] may also contribute to this inhibition.
6.2. Factors Counteracting the Antiviral Response to HIV-1 Infection
In addition to the antiviral ISG described earlier, type I IFN induces expression of several ISGs that interfere with different stages of HIV-1 and retroviral replication. These proteins are constitutively expressed at low levels in a cells type specific manner, and unlike the antiviral enzymes PKR or 2’5’OAS (already described above), they do not seem to require virus mediated activation (reviewed in [137,138]. The first of these cellular factors, dubbed intrinsic immunity factors was APOBEC3G [139]. APOBEC3G is a cytidine deaminase that represses reverse transcription of viral RNA, acts on CC dinucleotide motifs and induces deamination of cytidine to uridine during synthesis of negative strand proviral DNA transcripts. This change leads to a G to A transition in proviral DNA and decreased ability (fitness) in virus replication. The expression of APOBEC3G is stimulated by Type I IFN [140], and depends on NFATc1/NFATc2 and IRF-4. When either NFATc1 or NFATc2 and IRF-4 were co-expressed, activity of the APOBEC3G promoter was observed in cells that normally lack APOBEC3G expression. In addition to its effect on virus fitness APOBEC3G editing results in misfolding of viral proteins and enhances their recognition by CTL cells, thereby stimulating the antiviral adaptive immune response [141].
Type I IFN effectively induces the family of tripartite motif (TRIM) proteins that also restrict HIV-1 replication. TRIM5a from rhesus macaque (RhTRIM5a) blocks the early replication steps of HIV-1 and other retroviruses [142] by targeting the incoming viral capsid (CA) protein for degradation. Anti-HIV-1 activity has also been reported for TRIM1 that also targets the CA protein at an early stage of pre-reverse transcription. TRIM19 was suggested to affect trafficking of viral proteins [143], and TRIM22 blocked the release of HIV-1 Gag particles [144].
It was shown that Type I IFN inhibits both acute and chronic retroviral (MuLV) infection and that the inhibition was at the posttranslational level affecting virus assembly and release [145]. HIV-1 replication was found to be also sensitive to IFN inhibition, and the block was mapped to the late stages of HIV-1 replication [146]. IFNα induced antiretroviral activities also include a decrease in proviral DNA accumulation [147], restriction of viral entry and viral nucleic acid synthesis [148], and in macrophages Type I IFN restricts HIV-1 replication during the initial stages of infection [149].
At least three ISGs were shown to affect HIV-1 assembly and release. ISG15, a ubiquitin-like protein inhibits HIV-1 infection [150] by interfering with the ubiquitination of HIV-1 Gag, blocking the interaction of TSG101 with Gag and consequently inhibiting the endosomal trafficking pathway, used by HIV-1 virions to exit cells [103]. The ISG 15 block is not specific for HIV-1, but also affects release of the Ebola virus particles [104]. TRIM22 blocks the intracellular trafficking of the viral structural protein Gag to the surface of the cell. The antiviral activity of TRIM22 is dependent on the E3 ligase activity of the RING-containing region of TRIM22 [144]. Finally, the cellular protein BST-2, also known as tetherin, has been shown to be a very effective inhibitor of HIV-1 release [138]. This protein is expressed constitutively in DC and B cells, but can be also effectively induced by Type I IFN in other cell types. Unlike the trapped retroviral virions in IFN treated cells, which have decreased infectivity, the HIV-1 virions retained on the cell surface by tetherin are infectious. The tetherin mediated inhibition is not specific but inhibits release of a large group of retroviruses, filoviruses and herpes viruses [151,152].
Altogether these results indicate that HIV-1 can induce a strong innate immune response, associated with type I IFN mediated induction of several ISGs with a profound anti HIV-1 activity. Therefore the absence of the innate antiviral control of HIV-1 replication has been somewhat puzzling. One reason is that HIV-1, like the other RNA viruses, developed mechanisms by which it down-regulates the action of the antiviral ISG. HIV-1 encodes three small accessory proteins Vif, Vpr and Vpu, which circumvent the innate immune response by targeting the cellular antiviral proteins APOBEC3G and tetherin for ubiquitin dependent proteasome degradation. The expression of Virion infectivity factor, Vif, is essential for HIV-1 replication in primary T cells [137]. In the absence of Vif, APOBEC3G is incorporated into HIV-1 virions; thus Vif prevents packaging of APOBEC3G and its availability during the very early stages of HV-1 infection [153]. Virion associated viral protein R (Vpr) stimulates proviral transcription, and similar to SIV encoding Vpx, facilitates infection of macrophages. Both Vpr and Vif interact with cullin associated ubiquitin complex [137]. Vpr and Vif also target IRF-3 for degradation in T cells, which results in inhibition of the antiviral response and IFNβ synthesis [113]. Finally, the membrane protein, Vpu, is required for efficient release of virus particles. Vpu interacts with the trans membrane domain of tetherin and targets it for proteasomal or lyosomal degradation [154,155]. Virions of HIV-1 mutants lacking the vpu gene accumulate on the plasma membrane of infected cells and are not released to the medium. Vpu also interacts with CD4 and targets it for degradation by the cullin associated ubiquitin complex [156].
Other viral encoded functions may also counteract innate immunity. Interestingly, the negative factor Nef that has multiple functions, mostly in the modulation of adaptive immunity, can overlap Vpu and antagonize tetherin. In fact, primate SIV variants that lack Vpu use Nef to block the function of tetherin [157] (Figure 1).
The role of HIV-1 encoded protease was recently shown to play a role in the inhibition of Rig I mediated signaling induced by in vitro synthesized HIV-1 RNA. Ectopic expression of HIV-1 protease resulted in inhibition of IRF-3 phosphorylation and induction of ISG [158]. Since it is not clear whether ss HIV-1 RNA induces an interferon response in infected cells by the Rig I pathway, the in vivo relevance of this observation is yet to be confirmed.
In addition to virus encoded antiviral factors, several cellular proteins also contribute to down-regulation of the HIV-1 induced antiviral response. Thus the cellular DNAse Trex1 degrades the unintegrated proviral DNA in infected cells, thus preventing its detection by TLR9 or by the cytoplasmic DNA sensors. In TREX1 deficient cells HIV-1 DNA accumulates, which leads to induction of the IFN response [159]. Interestingly induction of IFN required the cellular adaptor STING and IRF-3, suggesting that proviral DNA is recognized by one of the cytoplamic DNA sensors, rather than by TLR9 [78]. Finally it was shown in SIV infected CNS macrophages, that IFNβ signaling is attenuated by induction of Suppressor of interferon signaling, SOCS3 [160]. Also the virus induced miRNA may modulate the antiviral response. In vitro studies have shown that several IFNβ induced miRNA including miR-26a, -34a, -145 targeted IFNB mRNA and attenuated the antiviral response [161]. Whether these miRNA also regulate the Type I IFN system in HIV-1 infected cells in vivo is yet to be determined.
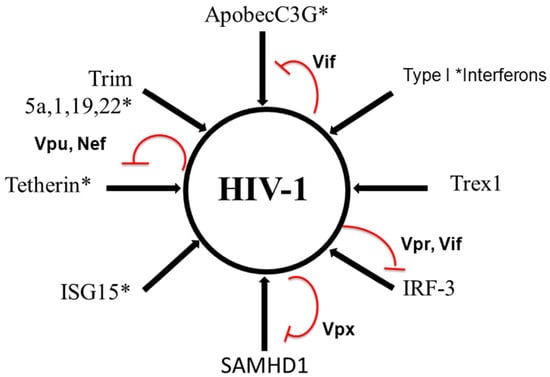
Figure 1.
Schematic illustration of regulation of HIV-1 infection during innate antiviral response.
7. Reflections and Future Considerations
Recent studies have clearly established that type I IFN has not only a critical role in the innate antiviral response but also in the pathogenicity of viral infection. Presently, some aspects of virus mediated induction of type I IFN and cytokine genes are understood at molecular levels, while others are still at a descriptive stage. The availability of genetically modified mice lacking essential components of the IFN signaling pathways confirmed that IFN is the earliest cellular defense against viral infection and a potent stimulant for the subsequent adaptive immune response. Rather unexpected was the observation that type I IFN induced large de-repression of the cellular genome stimulating expression of a large number of cellular genes, that are not only antiviral but are also involved in many different cellular functions. While it is easy to see the advantage generated by redundancy of antiviral proteins in their function against a wide variety of viruses, it is less evident why IFN evolved to control not only immune, but also many cellular functions. One has to wonder whether the multiplicity of IFN controlled genes reflect the number of cellular functions used by viruses for replication and survival. Important insights into viral pathogenicity have emerged from the observation than some of the nonstructural viral proteins target components of the IFN system, thereby decreasing or eliminating the antiviral response.
The innate antiviral response leads to expression of a large number of inflammatory cytokines in addition to interferon. The increased understanding of the cross talk of IFN pathway with the other inflammatory cytokines, illustrates an increasing degree of complexity in the mechanism of IFN action. This is well demonstrated in HIV-1 infection, where the early response to infection leads to a massive inflammatory response consisting of production of type I IFN, and many inflammatory cytokines. Thus while transient expression of type IFN may limit viral spread in the initial stages of infection, the cytokines stimulate HIV-1 infection either directly or by activation of immune cells. Since the ectopic expression of IFN inhibits HIV-1 replication in T cells [162], future studies will no doubt explore the means not only how to eliminate HIV-1 mimicry, but also how to generate the IFN response in the absence of inflammatory cytokines.
Acknowledgements
The author wishes to thanks Karen Beemon, Frank Maldarelli and Simona Stager for valuable input into this review and apologize to authors whose work was not cited, since any omission of relevant publications was not intentional.
References and Notes
- Akira, S.; Takeda, K.; Kaisho, T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat. Immunol. 2001, 2, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, T.; Ogasawara, K.; Takaoka, A.; Tanaka, N. IRF family of transcription factors as regulators of host defense. Annu. Rev. Immunol. 2001, 19, 623–655. [Google Scholar] [CrossRef] [PubMed]
- Barnes, B.J.; Moore, P.A.; Pitha, P.M. Virus-specific activation of a novel interferon regulatory factor, IRF-5, results in the induction of distinct interferon alpha genes. J. Biol. Chem. 2001, 276, 23382–23390. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Medzhitov, R. Toll-like receptor control of the adaptive immune responses. Nat. Immunol. 2004, 5, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, A.; Yanai, H.; Kondo, S.; Duncan, G.; Negishi, H.; Mizutani, T.; Kano, S.; Honda, K.; Ohba, Y.; Mak, T.W.; Taniguchi, T. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature 2005, 434, 243–249. [Google Scholar] [CrossRef]
- Barnes, B.; Lubyova, B.; Pitha, P.M. On the role of IRF in host defense. J. Interferon Cytokine Res. 2002, 22, 59–71. [Google Scholar] [CrossRef]
- Escalante, C.R.; Yie, J.; Thanos, D.; Aggarwal, A.K. Expression, purification, and co-crystallization of IRF-I bound to the interferon-beta element PRDI. FEBS Lett. 1997, 414, 219–220. [Google Scholar]
- Takaoka, A.; Taniguchi, T. Cytosolic DNA recognition for triggering innate immune responses. Adv. Drug Deliv. Rev. 2008, 60, 847–857. [Google Scholar] [CrossRef]
- Fujita, T.; Sakakibara, J.; Sudo, Y.; Miyamoto, M.; Kimura, Y.; Taniguchi, T. IRF-1, mediating induction and silencing properties to human IFN-á gene regulatory elements. EMBO J. 1988, 7, 3397–3405. [Google Scholar] [CrossRef]
- Reis, L.F.; Ruffner, H.; Stark, G.; Aguet, M.; Weissmann, C. Mice devoid of interferon regulatory factor 1 (IRF-1) show normal expression of type I interferon genes. EMBO J. 1994, 13, 4798–4806. [Google Scholar] [CrossRef]
- Au, W.C.; Yeow, W.S.; Pitha, P.M. Analysis of functional domains of interferon regulatory factor 7 and its association with IRF-3. Virology 2001, 280, 273–282. [Google Scholar] [CrossRef]
- Marie, I.; Durbin, J.E.; Levy, D.E.s. Differential viral induction of distinct interferon-alpha genes by positive feedback through interferon regulatory factor-7. EMBO J. 1998, 17, 6660–6669. [Google Scholar] [CrossRef]
- Au, W.C.; Pitha, P.M. Recruitment of multiple interferon regulatory factors and histone acetyltransferase to the transcriptionally active interferon a promoters. J. Biol. Chem. 2001, 276, 41629–41637. [Google Scholar] [CrossRef]
- Au, W.-C.; Moore, P.A.; Lowther, W.; Juang, Y.-T.; Pitha, P.M. Identification of a member of the interferon regulatory factor family that binds to the interferon-stimulated response element and activates expression of interferon-induced genes. Proc. Natl. Acad. Sci. U. S. A. 1995, 92, 11657–11661. [Google Scholar] [CrossRef]
- Ronco, L.V.; Karpova, A.Y.; Vidal, M.; Howley, P.M. Human papillomavirus 16 E6 oncoprotein binds to interferon regulatory factor-3 and inhibits its transcriptional activity. Genes Dev. 1998, 12, 2061–2072. [Google Scholar] [CrossRef]
- Au, W.C.; Moore, P.A.; LaFleur, D.W.; Tombal, B.; Pitha, P.M. Characterization of the interferon regulatory factor-7 and its potential role in the transcription activation of interferon A genes. J. Biol. Chem. 1998, 273, 29210–29217. [Google Scholar] [CrossRef]
- Akira, S. TLR signaling. Curr. Top. Microbiol. Immunol. 2006, 311, 1–16. [Google Scholar]
- Yeow, W.S.; Au, W.C.; Juang, Y.T.; Fields, C.D.; Dent, C.L.; Gewert, D.R.; Pitha, P.M. Reconstitution of virus-mediated expression of interferon alpha genes in human fibroblast cells by ectopic interferon regulatory factor-7. J. Biol. Chem. 2000, 275, 6313–6320. [Google Scholar] [CrossRef]
- Gibson, S.J.; Lindh, J.M.; Riter, T.R.; Gleason, R.M.; Rogers, L.M.; Fuller, A.E.; Oesterich, J.L.; Gorden, K.B.; Qiu, X.; McKane, S.W.; et al. Plasmacytoid dendritic cells produce cytokines and mature in response to the TLR7 agonists, imiquimod and resiquimod. Cell. Immunol. 2002, 218, 74–86. [Google Scholar] [CrossRef]
- Siegal, F.P.; Kadowaki, N.; Shodell, M.; Fitzgerald-Bocarsly, P.A.; Shah, K.; Ho, S.; Antonenko, S.; Liu, Y.J. The nature of the principal type 1 interferon-producing cells in human blood. Science 1999, 284, 1835–1837. [Google Scholar] [CrossRef]
- Barnes, B.J.; Richards, J.; Mancl, M.; Hanash, S.; Beretta, L.; Pitha, P.M. Global and distinct targets of IRF-5 and IRF-7 during innate response to viral infection. J. Biol. Chem. 2004, 279, 45194–45207. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Yanai, H.; Negishi, H.; Asagiri, M.; Sato, M.; Mizutani, T.; Shimada, N.; Ohba, Y.; Takaoka, A.; Yoshida, N.; Taniguchi, T. IRF-7 is the master regulator of type-I interferon-dependent immune responses. Nature 2005, 434, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Paun, A.; Pitha, P.M. The innate antiviral response: New insights into a continuing story. Adv. Virus Res. 2007, 69, 1–66. [Google Scholar] [PubMed]
- Takaoka, A.; Tamura, T.; Taniguchi, T. Interferon regulatory factor family of transcription factors and regulation of oncogenesis. Cancer Sci. 2008, 99, 467–478. [Google Scholar] [CrossRef]
- Paun, A.; Bankoti, R.; Joshi, T.; Pitha, P.M.; Stager, S. Critical role of IRF-5 in the development of T helper 1 responses to Leishmania donovani infection. PLoS Pathog. 2011, 7, e1001246. [Google Scholar] [CrossRef]
- Lien, C.; Fang, C.M.; Huso, D.; Livak, F.; Lu, R.; Pitha, P.M. Critical role of IRF-5 in regulation of B-cell differentiation. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 4664–4668. [Google Scholar] [CrossRef]
- Krausgruber, T.; Blazek, K.; Smallie, T.; Alzabin, S.; Lockstone, H.; Sahgal, N.; Hussell, T.; Feldmann, M.; Udalova, I.A. IRF5 promotes inflammatory macrophage polarization and TH1-TH17 responses. Nat. Immunol. 2011, 12, 231–238. [Google Scholar] [CrossRef]
- Moore, P.S.; Chang, Y. Antiviral activity of tumor-suppressor pathways: Clues from molecular piracy by KSHV. Trends Genet. 1998, 14, 144–150. [Google Scholar] [CrossRef]
- Cunningham, C.; Barnard, S.; Blackbourn, D.J.; Davison, A.J. Transcription mapping of human herpesvirus 8 genes encoding viral interferon regulatory factors. J. Gen. Virol. 2003, 84, 1471–1483. [Google Scholar] [CrossRef]
- Burysek, L.; Yeow, W.S.; Lubyova, B.; Kellum, M.; Schafer, S.L.; Huang, Y.Q.; Pitha, P.M. Functional analysis of human herpesvirus 8-encoded viral interferon regulatory factor 1 and its association with cellular interferon regulatory factors and p300. J. Virol. 1999, 73, 7334–7342. [Google Scholar] [CrossRef]
- Flowers, C.C.; Flowers, S.P.; Nabel, G.J. Kaposi's sarcoma-associated herpesvirus viral interferon regulatory factor confers resistance to the antiproliferative effect of interferon-alpha. Mol. Med. 1998, 4, 402–412. [Google Scholar] [CrossRef]
- Gao, S.J.; Boshoff, C.; Jayachandra, S.; Weiss, R.A.; Chang, Y.; Moore, P.S. KSHV ORF K9 (vIRF) is an oncogene which inhibits the interferon signaling pathway. Oncogene 1997, 15, 1979–1985. [Google Scholar] [CrossRef]
- Li, M.; Lee, H.; Guo, J.; Neipel, F.; Fleckenstein, B.; Ozato, K.; Jung, J.U. Kaposi's sarcoma-associated herpesvirus viral interferon regulatory factor. J. Virol. 1998, 72, 5433–5440. [Google Scholar] [CrossRef]
- Burysek, L.; Yeow, W.S.; Pitha, P.M. Unique properties of a second human herpesvirus 8-encoded interferon regulatory factor (vIRF-2). J. Hum. Virol. 1999, 2, 19–32. [Google Scholar]
- Burysek, L.; Pitha, P.M. Latently expressed human herpesvirus 8-encoded interferon regulatory factor 2 inhibits double-stranded RNA-activated protein kinase. J. Virol. 2001, 75, 2345–2352. [Google Scholar] [CrossRef]
- Lubyova, B.; Pitha, P.M. Characterization of a novel human herpesvirus 8-encoded protein, vIRF-3, that shows homology to viral and cellular interferon regulatory factors. J. Virol. 2000, 74, 8194–8201. [Google Scholar] [CrossRef]
- Rivas, C.; Thlick, A.E.; Parravicini, C.; Moore, P.S.; Chang, Y. Kaposi's sarcoma-associated herpesvirus LANA2 is a B-cell-specific latent viral protein that inhibits p53. J. Virol. 2001, 75, 429–438. [Google Scholar] [CrossRef]
- Lubyova, B.; Kellum, M.J.; Frisancho, A.J.; Pitha, P.M. Kaposi's sarcoma-associated herpesvirus-encoded vIRF-3 stimulates the transcriptional activity of cellular IRF-3 and IRF-7. J. Biol. Chem. 2004, 279, 7643–7654. [Google Scholar] [CrossRef]
- Lubyova, B.; Kellum, M.J.; Frisancho, J.A.; Pitha, P.M. Stimulation of c-Myc transcriptional activity by vIRF-3 of Kaposi sarcoma-associated herpesvirus. J. Biol. Chem. 2007, 282, 31944–31953. [Google Scholar] [CrossRef]
- Ducan, G.S.; Mittrucker, H.-W.; Kagi, D.; Matsuyama, T.; Mak, T.W. The transcription factor interferon regulatory factor-1 is essential for natural killer cell function in vivo. J. Exp. Med. 1996, 184, 2043–2048. [Google Scholar] [CrossRef]
- Lohoff, M.; Ferrick, D.; Mittrucker, H.W.; Duncan, G.S.; Bischof, S.; Rollinghoff, M.; Mak, T.W. Interferon regulatory factor-1 is required for a T helper 1 immune response in vivo. Immunity 1997, 6, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Mittrucker, H.-W.; Matasuyama, T.; Grossman, A.; Kundig, T.M.; Potter, J.; Shahinian, A.; Wakeham, A.; Patterson, B.; Ohashi, P.; Mak, T. Requirement for the transcription factor LSIRF/IRF4 for mature B and T lymphocyte function. Science 1997, 275, 540–543. [Google Scholar] [CrossRef] [PubMed]
- Klein, U.; Casola, S.; Cattoretti, G.; Shen, Q.; Lia, M.; Mo, T.; Ludwig, T.; Rajewsky, K.; Dalla-Favera, R. Transcription factor IRF4 controls plasma cell differentiation and class-switch recombination. Nat. Immunol. 2006, 7, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Nagamura-Inoue, T.; Shmeltzer, Z.; Kuwata, T.; Ozato, K. ICSBP directs bipotential myeloid progenitor cells to differentiate into mature macrophages. Immunity 2000, 13, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Thotakura, P.; Tanaka, T.S.; Ko, M.S.; Ozato, K. Identification of target genes and a unique cis element regulated by IRF-8 in developing macrophages. Blood 2005, 106, 1938–1947. [Google Scholar] [CrossRef]
- Lu, R.; Pitha, P.M. Monocyte differentiation to macrophage requires interferon regulatory factor 7. J. Biol. Chem. 2001, 276, 45491–45496. [Google Scholar] [CrossRef]
- Sasaki, S.; Amara, R.R.; Yeow, W.S.; Pitha, P.M.; Robinson, H.L. Regulation of DNA-raised immune responses by cotransfected interferon regulatory factors. J. Virol. 2002, 76, 6652–6659. [Google Scholar] [CrossRef]
- Maiwald, T.; Schneider, A.; Busch, H.; Sahle, S.; Gretz, N.; Weiss, T.S.; Kummer, U.; Klingmuller, U. Combining theoretical analysis and experimental data generation reveals IRF9 as a crucial factor for accelerating interferon alpha-induced early antiviral signalling. FEBS J. 2010, 277, 4741–4754. [Google Scholar] [CrossRef]
- Lin, C.H.; Hare, B.J.; Wagner, G.; Harrison, S.C.; Maniatis, T.; Fraenkel, E. A small domain of CBP/p300 binds diverse proteins: Solution structure and functional studies. Mol. Cell 2001, 8, 581–590. [Google Scholar] [CrossRef]
- Braganca, J.; Genin, P.; Bandu, M.T.; Darracq, N.; Vignal, M.; Casse, C.; Doly, J.; Civas, A. Synergism between multiple virus-induced factor-binding elements involved in the differential expression of interferon A genes. J. Biol. Chem. 1997, 272, 22154–22162. [Google Scholar] [CrossRef]
- Wathelet, M.G.; Lin, C.H.; Parekh, B.S.; Ronco, L.V.; Howley, P.M.; Maniatis, T. Virus infection induces the assembly of coordinately activated transcription factors on the IFN-beta enhancer in vivo. Mol. Cell 1998, 1, 507–518. [Google Scholar] [CrossRef]
- Medzhitov, R. CpG DNA: security code for host defense. Nat. Immunol. 2001, 2, 15–16. [Google Scholar] [CrossRef]
- Beutler, B.; Poltorak, A. Toll we meet again. Nat. Immunol. 2001, 2, 9–10. [Google Scholar] [CrossRef]
- Lin, S.C.; Lo, Y.C.; Wu, H. Helical assembly in the MyD88-IRAK4-IRAK2 complex in TLR/IL-1R signalling. Nature 2010, 465, 885–890. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Signaling to NF-kappaB by Toll-like receptors. Trends Mol. Med. 2007, 13, 460–469. [Google Scholar] [CrossRef]
- Izaguirre, A.; Barnes, B.J.; Amrute, S.; Yeow, W.S.; Megjugorac, N.; Dai, J.; Feng, D.; Chung, E.; Pitha, P.M.; Fitzgerald-Bocarsly, P. Comparative analysis of IRF and IFN-alpha expression in human plasmacytoid and monocyte-derived dendritic cells. J. Leukoc. Biol. 2003, 74, 1125–1138. [Google Scholar] [CrossRef]
- Matsumoto, M.; Oshiumi, H.; Seya, T. Antiviral responses induced by the TLR3 pathway. Rev. Med. Virol. 2011, 21, 67–77. [Google Scholar] [CrossRef]
- Fitzgerald, K.A.; McWhirter, S.M.; Faia, K.L.; Rowe, D.C.; Latz, E.; Golenbock, D.T.; Coyle, A.J.; Liao, S.M.; Maniatis, T. IKKepsilon and TBK1 are essential components of the IRF3 signaling pathway. Nat. Immunol. 2003, 4, 491–496. [Google Scholar] [CrossRef]
- Grandvaux, N.; Servant, M.J.; tenOever, B.; Sen, G.C.; Balachandran, S.; Barber, G.N.; Lin, R.; Hiscott, J. Transcriptional profiling of interferon regulatory factor 3 target genes: Direct involvement in the regulation of interferon-stimulated genes. J. Virol. 2002, 76, 5532–5539. [Google Scholar] [CrossRef]
- Fitzgerald, K.A.; Rowe, D.C.; Barnes, B.J.; Caffrey, D.R.; Visintin, A.; Latz, E.; Monks, B.; Pitha, P.M.; Golenbock, D.T. LPS-TLR4 signaling to IRF-3/7 and NF-kappaB involves the toll adapters TRAM and TRIF. J. Exp. Med. 2003, 198, 1043–1055. [Google Scholar] [CrossRef]
- Hoebe, K.; Du, X.; Georgel, P.; Janssen, E.; Tabeta, K.; Kim, S.O.; Goode, J.; Lin, P.; Mann, N.; Mudd, S.; et al. Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature 2003, 424, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.A.; Palsson-McDermott, E.M.; Bowie, A.G.; Jefferies, C.A.; Mansell, A.S.; Brady, G.; Brint, E.; Dunne, A.; Gray, P.; Harte, M.T.; et al. Mal (MyD88-adapter-like) is required for Toll-like receptor-4 signal transduction. Nature 2001, 413, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Takaoka, A.; Taniguchi, T. Type I interferon [corrected] gene induction by the interferon regulatory factor family of transcription factors. Immunity 2006, 25, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Haller, O.; Kochs, G.; Weber, F. The interferon response circuit: induction and suppression by pathogenic viruses. Virology 2006, 344, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.H.; Ulevitch, R.J. Cloning and characterization of a sub-family of human toll-like receptors: hTLR7, hTLR8 and hTLR9. Eur. Cytokine Netw. 2000, 11, 372–378. [Google Scholar]
- Hemmi, H.; Takeuchi, O.; Kawai, T.; Kaisho, T.; Sato, S.; Sanjo, H.; Matsumoto, M.; Hoshino, K.; Wagner, H.; Takeda, K.; et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000, 408, 740–745. [Google Scholar] [CrossRef]
- Diebold, S.S.; Kaisho, T.; Hemmi, H.; Akira, S.; Reis e Sousa, C. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 2004, 303, 1529–1531. [Google Scholar] [CrossRef]
- Lund, J.M.; Alexopoulou, L.; Sato, A.; Karow, M.; Adams, N.C.; Gale, N.W.; Iwasaki, A.; Flavell, R.A. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 5598–5603. [Google Scholar] [CrossRef]
- Schoenemeyer, A.; Barnes, B.J.; Mancl, M.E.; Latz, E.; Goutagny, N.; Pitha, P.M.; Fitzgerald, K.A.; Golenbock, D.T. The interferon regulatory factor, IRF5, is a central mediator of toll-like receptor 7 signaling. J. Biol. Chem. 2005, 280, 17005–17012. [Google Scholar] [CrossRef]
- Balkhi, M.Y.; Fitzgerald, K.A.; Pitha, P.M. Functional regulation of MyD88-activated interferon regulatory factor 5 by K63-linked polyubiquitination. Mol. Cell. Biol. 2008, 28, 7296–7308. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, M.; Kikuchi, M.; Matsumoto, K.; Imaizumi, T.; Miyagishi, M.; Taira, K.; Foy, E.; Loo, Y.M.; Gale, M., Jr.; Akira, S.; et al. Shared and unique functions of the DExD/H-box helicases RIG-I, MDA5, and LGP2 in antiviral innate immunity. J. Immunol. 2005, 175, 2851–2858. [Google Scholar] [CrossRef] [PubMed]
- Loo, Y.M.; Fornek, J.; Crochet, N.; Bajwa, G.; Perwitasari, O.; Martinez-Sobrido, L.; Akira, S.; Gill, M.A.; Garcia-Sastre, A.; Katze, M.G.; et al. Distinct RIG-I and MDA5 signaling by RNA viruses in innate immunity. J. Virol. 2008, 82, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. Antiviral signaling through pattern recognition receptors. J. Biochem. 2007, 141, 137–145. [Google Scholar] [CrossRef]
- Sato, A.; Linehan, M.M.; Iwasaki, A. Dual recognition of herpes simplex viruses by TLR2 and TLR9 in dendritic cells. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 17343–17348. [Google Scholar] [CrossRef]
- Sorensen, L.N.; Reinert, L.S.; Malmgaard, L.; Bartholdy, C.; Thomsen, A.R.; Paludan, S.R. TLR2 and TLR9 synergistically control herpes simplex virus infection in the brain. J. Immunol. 2008, 181, 8604–8612. [Google Scholar] [CrossRef]
- Compton, T.; Kurt-Jones, E.A.; Boehme, K.W.; Belko, J.; Latz, E.; Golenbock, D.T.; Finberg, R.W. Human cytomegalovirus activates inflammatory cytokine responses via CD14 and Toll-like receptor 2. J. Virol. 2003, 77, 4588–4596. [Google Scholar] [CrossRef]
- Rathinam, V.A.; Jiang, Z.; Waggoner, S.N.; Sharma, S.; Cole, L.E.; Waggoner, L.; Vanaja, S.K.; Monks, B.G.; Ganesan, S.; Latz, E.; et al. The AIM2 inflammasome is essential for host defense against cytosolic bacteria and DNA viruses. Nat. Immunol. 2010, 11, 395–402. [Google Scholar] [CrossRef]
- Ishikawa, H.; Ma, Z.; Barber, G.N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 2009, 461, 788–792. [Google Scholar] [CrossRef]
- Ishii, K.J.; Akira, S. Innate immune recognition of, and regulation by, DNA. Trends Immunol. 2006, 27, 525–532. [Google Scholar] [CrossRef]
- Wang, Z.; Choi, M.K.; Ban, T.; Yanai, H.; Negishi, H.; Lu, Y.; Tamura, T.; Takaoka, A.; Nishikura, K.; Taniguchi, T. Regulation of innate immune responses by DAI (DLM-1/ZBP1) and other DNA-sensing molecules. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 5477–5482. [Google Scholar] [CrossRef]
- Unterholzner, L.; Keating, S.E.; Baran, M.; Horan, K.A.; Jensen, S.B.; Sharma, S.; Sirois, C.M.; Jin, T.; Latz, E.; Xiao, T.S.; et al. IFI16 is an innate immune sensor for intracellular DNA. Nat. Immunol. 2010, 11, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Ablasser, A.; Bauernfeind, F.; Hartmann, G.; Latz, E.; Fitzgerald, K.A.; Hornung, V. RIG-I-dependent sensing of poly(dA:dT) through the induction of an RNA polymerase III-transcribed RNA intermediate. Nat. Immunol. 2009, 10, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Kanneganti, T.D. Central roles of NLRs and inflammasomes in viral infection. Nat. Rev. Immunol. 2010, 10, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Muller, U.; Steinhoff, U.; Reis, L.F.; Hemmi, S.; Pavlovic, J.; Zinkernagel, R.M.; Aguet, M. Functional role of type I and type II interferons in antiviral defense. Science 1994, 264, 1918–1921. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, S.; Jouanguy, E.; Al-Hajjar, S.; Fieschi, C.; Al-Mohsen, I.Z.; Al-Jumaah, S.; Yang, K.; Chapgier, A.; Eidenschenk, C.; Eid, P.; et al. Impaired response to interferon-alpha/beta and lethal viral disease in human STAT1 deficiency. Nat. Genet. 2003, 33, 388–391. [Google Scholar] [CrossRef]
- Bouloy, M.; Janzen, C.; Vialat, P.; Khun, H.; Pavlovic, J.; Huerre, M.; Haller, O. Genetic evidence for an interferon-antagonistic function of rift valley fever virus nonstructural protein NSs. J. Virol. 2001, 75, 1371–1377. [Google Scholar] [CrossRef]
- Darnell, J.E.J.; Kerr, I.M.; Stark, G.R. Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 1994, 264, 1415–1421. [Google Scholar] [CrossRef]
- Der, S.D.; Zhou, A.; Williams, B.R.; Silverman, R.H. Identification of genes differentially regulated by interferon alpha, beta, or gamma using oligonucleotide arrays. Proc. Natl. Acad. Sci. U. S. A. 1998, 95, 15623–15628. [Google Scholar] [CrossRef]
- Li, X.; Leung, S.; Qureshi, S.; Darnell, J.E.J.; Stark, G.R. Formation of STAT1-STAT2 heterodimers and their role in the activation of IRF-1 gene transcription by interferon-alpha. J. Biol.Chem. 1996, 271, 5790–5794. [Google Scholar] [CrossRef]
- Schindler, C.; Fu, X.-Y.; Improta, T.; Aebersold, R.; Darnell, J.E.J. Proteins of transcription factor ISGF-3: one gene encodes the 91- and 84-kDa ISGF-3 proteins that are activated by interferon alpha. Proc. Natl. Acad. Sci. U. S. A. 1992, 89, 7836–7839. [Google Scholar] [CrossRef]
- Darnell, J.E., Jr. STATs and gene regulation. Science 1997, 277, 1630–1635. [Google Scholar] [CrossRef]
- Levy, D.E.; Darnell, J.E., Jr. Stats: Transcriptional control and biological impact. Nat. Rev. Mol. Cell Biol. 2002, 3, 651–662. [Google Scholar] [CrossRef]
- Platanias, L.C. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat. Rev. Immunol. 2005, 5, 375–386. [Google Scholar] [CrossRef]
- Samuel, C.E. Antiviral actions of interferons. Clin. Microbiol. Rev. 2001, 14, 778–809, table of contents. [Google Scholar] [CrossRef]
- Schoggins, J.W.; Wilson, S.J.; Panis, M.; Murphy, M.Y.; Jones, C.T.; Bieniasz, P.; Rice, C.M. A diverse range of gene products are effectors of the type I interferon antiviral response. Nature 2011, 472, 481–485. [Google Scholar] [CrossRef]
- Kerr, I.M.; Brown, R.E. pppA2'p5'A2'p5'A: An inhibitor of protein synthesis synthesized with an enzyme fraction from interferon-treated cells. Proc. Natl. Acad. Sci. U. S. A. 1978, 75, 256–260. [Google Scholar] [CrossRef]
- Samuel, C.E. The eIF-2 alpha protein kinases, regulators of translation in eukaryotes from yeasts to humans. J. Biol. Chem. 1993, 268, 7603–7606. [Google Scholar] [CrossRef]
- Kumar, A.; Haque, J.; Lacoste, J.; Hiscott, J.; Williams, B. Double stranded RNA-dependent protein kinase activates transcription factor NF-kB by phosphorylating IkB. Proc. Natl. Acad. Sci. U. S. A. 1994, 91, 6288–6292. [Google Scholar] [CrossRef]
- Staeheli, P.; Grob, R.; Meier, E.; Sutcliffe, J.G.; Haller, O. Influenza virus-susceptible mice carry Mx genes with a large deletion or a nonsense mutation. Mol. Cell. Biol. 1988, 8, 4518–4523. [Google Scholar]
- Haller, O.; Arnheiter, H.; Gresser, I.; Lindenmann, J. Virus-specific interferon action. Protection of newborn Mx carriers against lethal infection with influenza virus. J. Exp. Med. 1981, 154, 199–203. [Google Scholar] [CrossRef]
- Zhou, A.; Paranjape, J.M.; Der, S.D.; Williams, B.R.; Silverman, R.H. Interferon action in triply deficient mice reveals the existence of alternative antiviral pathways. Virology 1999, 258, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Harty, R.N.; Pitha, P.M.; Okumura, A. Antiviral activity of innate immune protein ISG15. J. Innate. Immun. 2009, 1, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Okumura, A.; Lu, G.; Pitha-Rowe, I.; Pitha, P.M. Innate antiviral response targets HIV-1 release by the induction of ubiquitin-like protein ISG15. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 1440–1445. [Google Scholar] [CrossRef] [PubMed]
- Okumura, A.; Pitha, P.M.; Harty, R.N. ISG15 inhibits Ebola VP40 VLP budding in an L-domain-dependent manner by blocking Nedd4 ligase activity. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 3974–3979. [Google Scholar] [CrossRef]
- Murphy, D.G.; Dimock, K.; Kang, C.Y. Numerous transitions in human parainfluenza virus 3 RNA recovered from persistently infected cells. Virology 1991, 181, 760–763. [Google Scholar] [CrossRef]
- Chen, K.; Huang, J.; Zhang, C.; Huang, S.; Nunnari, G.; Wang, F.X.; Tong, X.; Gao, L.; Nikisher, K.; Zhang, H. Alpha interferon potently enhances the anti-human immunodeficiency virus type 1 activity of APOBEC3G in resting primary CD4 T cells. J. Virol. 2006, 80, 7645–7657. [Google Scholar] [CrossRef]
- Yu, Q.; Chen, D.; Konig, R.; Mariani, R.; Unutmaz, D.; Landau, N.R. APOBEC3B and APOBEC3C are potent inhibitors of simian immunodeficiency virus replication. J. Biol. Chem. 2004, 279, 53379–53386. [Google Scholar] [CrossRef]
- Hou, J.; Wang, P.; Lin, L.; Liu, X.; Ma, F.; An, H.; Wang, Z.; Cao, X. MicroRNA-146a feedback inhibits RIG-I-dependent Type I IFN production in macrophages by targeting TRAF6, IRAK1, and IRAK2. J. Immunol. 2009, 183, 2150–2158. [Google Scholar] [CrossRef]
- O'Connell, R.M.; Taganov, K.D.; Boldin, M.P.; Cheng, G.; Baltimore, D. MicroRNA-155 is induced during the macrophage inflammatory response. Proc. Natl. Acad. Sci. U. S. A. 2007, 104, 1604–1609. [Google Scholar] [CrossRef]
- Andrejeva, J.; Childs, K.S.; Young, D.F.; Carlos, T.S.; Stock, N.; Goodbourn, S.; Randall, R.E. The V proteins of paramyxoviruses bind the IFN-inducible RNA helicase, mda-5, and inhibit its activation of the IFN-beta promoter. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 17264–17269. [Google Scholar] [CrossRef]
- Precious, B.; Childs, K.; Fitzpatrick-Swallow, V.; Goodbourn, S.; Randall, R.E. Simian virus 5 V protein acts as an adaptor, linking DDB1 to STAT2, to facilitate the ubiquitination of STAT1. J. Virol. 2005, 79, 13434–13441. [Google Scholar] [CrossRef]
- Basler, C.F.; Mikulasova, A.; Martinez-Sobrido, L.; Paragas, J.; Muhlberger, E.; Bray, M.; Klenk, H.D.; Palese, P.; Garcia-Sastre, A. The Ebola virus VP35 protein inhibits activation of interferon regulatory factor 3. J. Virol. 2003, 77, 7945–7956. [Google Scholar] [CrossRef]
- Okumura, A.; Alce, T.; Lubyova, B.; Ezelle, H.; Strebel, K.; Pitha, P.M. HIV-1 accessory proteins VPR and Vif modulate antiviral response by targeting IRF-3 for degradation. Virology 2008, 373, 85–97. [Google Scholar] [CrossRef]
- Ulane, C.M.; Kentsis, A.; Cruz, C.D.; Parisien, J.P.; Schneider, K.L.; Horvath, C.M. Composition and assembly of STAT-targeting ubiquitin ligase complexes: Paramyxovirus V protein carboxyl terminus is an oligomerization domain. J. Virol. 2005, 79, 10180–10189. [Google Scholar] [CrossRef]
- Alcami, A.; Symons, J.A.; Smith, G.L. The vaccinia virus soluble alpha/beta interferon (IFN) receptor binds to the cell surface and protects cells from the antiviral effects of IFN. J. Virol. 2000, 74, 11230–11239. [Google Scholar] [CrossRef]
- Ahmed, M.; McKenzie, M.O.; Puckett, S.; Hojnacki, M.; Poliquin, L.; Lyles, D.S. Ability of the matrix protein of vesicular stomatitis virus to suppress beta interferon gene expression is genetically correlated with the inhibition of host RNA and protein synthesis. J. Virol. 2003, 77, 4646–4657. [Google Scholar] [CrossRef]
- Haase, A.T. Targeting early infection to prevent HIV-1 mucosal transmission. Nature 2010, 464, 217–223. [Google Scholar] [CrossRef]
- Stacey, A.R.; Norris, P.J.; Qin, L.; Haygreen, E.A.; Taylor, E.; Heitman, J.; Lebedeva, M.; DeCamp, A.; Li, D.; Grove, D.; et al. Induction of a striking systemic cytokine cascade prior to peak viremia in acute human immunodeficiency virus type 1 infection, in contrast to more modest and delayed responses in acute hepatitis B and C virus infections. J. Virol. 2009, 83, 3719–3733. [Google Scholar] [CrossRef]
- Popik, W.; Pitha, P.M. Role of tumor necrosis factor alpha in activation and replication of the tat-defective human immunodeficiency virus type 1. J.Virol. 1993, 67, 1094–1099. [Google Scholar] [CrossRef]
- Sgarbanti, M.; Remoli, A.L.; Marsili, G.; Ridolfi, B.; Borsetti, A.; Perrotti, E.; Orsatti, R.; Ilari, R.; Sernicola, L.; Stellacci, E.; et al. IRF-1 is required for full NF-kappaB transcriptional activity at the human immunodeficiency virus type 1 long terminal repeat enhancer. J. Virol. 2008, 82, 3632–3641. [Google Scholar] [CrossRef]
- Harman, A.N.; Lai, J.; Turville, S.; Samarajiwa, S.; Gray, L.; Marsden, V.; Mercier, S.; Jones, K.; Nasr, N.; Cumming, H.; et al. HIV infection of dendritic cells subverts the interferon induction pathway via IRF1 and inhibits type 1 interferon production. Blood 2011. [Google Scholar] [CrossRef] [PubMed]
- Van Lint, C.; Amella, C.A.; Emiliani, S.; John, M.; Jie, T.; Verdin, E. Transcription factor binding sites downstream of the human immunodeficiency virus type 1 transcription start site are important for virus infectivity. J. Virol. 1997, 71, 6113–6127. [Google Scholar] [CrossRef] [PubMed]
- Su, R.C.; Sivro, A.; Kimani, J.; Jaoko, W.; Plummer, F.A.; Ball, T.B. Epigenetic control of IRF1 responses in HIV-exposed seronegative versus HIV-susceptible individuals. Blood 2011, 117, 2649–2657. [Google Scholar] [CrossRef] [PubMed]
- Sodora, D.L.; Ross, T.M. Simian immunodeficiency virus pathogenesis. Curr. HIV Res. 2009, 7, 1. [Google Scholar] [CrossRef]
- Rotger, M.; Dang, K.K.; Fellay, J.; Heinzen, E.L.; Feng, S.; Descombes, P.; Shianna, K.V.; Ge, D.; Gunthard, H.F.; Goldstein, D.B.; et al. Genome-wide mRNA expression correlates of viral control in CD4+ T-cells from HIV-1-infected individuals. PLoS Pathog. 2010, 6, e1000781. [Google Scholar] [CrossRef]
- Smith, A.J.; Li, Q.; Wietgrefe, S.W.; Schacker, T.W.; Reilly, C.S.; Haase, A.T. Host genes associated with HIV-1 replication in lymphatic tissue. J. Immunol. 2010, 185, 5417–5424. [Google Scholar] [CrossRef]
- Sedaghat, A.R.; German, J.; Teslovich, T.M.; Cofrancesco, J., Jr.; Jie, C.C.; Talbot, C.C., Jr.; Siliciano, R.F. Chronic CD4+ T-cell activation and depletion in human immunodeficiency virus type 1 infection: Type I interferon-mediated disruption of T-cell dynamics. J. Virol. 2008, 82, 1870–1883. [Google Scholar] [CrossRef]
- Lehmann, C.; Lafferty, M.; Garzino-Demo, A.; Jung, N.; Hartmann, P.; Fatkenheuer, G.; Wolf, J.S.; van Lunzen, J.; Romerio, F. Plasmacytoid dendritic cells accumulate and secrete interferon alpha in lymph nodes of HIV-1 patients. PLoS ONE 2010, 5, e11110. [Google Scholar] [CrossRef]
- Hyrcza, M.D.; Kovacs, C.; Loutfy, M.; Halpenny, R.; Heisler, L.; Yang, S.; Wilkins, O.; Ostrowski, M.; Der, S.D. Distinct transcriptional profiles in ex vivo CD4+ and CD8+ T cells are established early in human immunodeficiency virus type 1 infection and are characterized by a chronic interferon response as well as extensive transcriptional changes in CD8+ T cells. J. Virol. 2007, 81, 3477–3486. [Google Scholar] [CrossRef]
- Mandl, J.N.; Barry, A.P.; Vanderford, T.H.; Kozyr, N.; Chavan, R.; Klucking, S.; Barrat, F.J.; Coffman, R.L.; Staprans, S.I.; Feinberg, M.B. Divergent TLR7 and TLR9 signaling and type I interferon production distinguish pathogenic and nonpathogenic AIDS virus infections. Nat. Med. 2008, 14, 1077–1087. [Google Scholar] [CrossRef]
- Cameron, P.U.; Handley, A.J.; Baylis, D.C.; Solomon, A.E.; Bernard, N.; Purcell, D.F.; Lewin, S.R. Preferential infection of dendritic cells during human immunodeficiency virus type 1 infection of blood leukocytes. J. Virol. 2007, 81, 2297–2306. [Google Scholar] [CrossRef]
- Pope, M.; Betjes, M.G.; Romani, N.; Hirmand, H.; Cameron, P.U.; Hoffman, L.; Gezelter, S.; Schuler, G.; Steinman, R.M. Conjugates of dendritic cells and memory T lymphocytes from skin facilitate productive infection with HIV-1. Cell 1994, 78, 389–398. [Google Scholar] [CrossRef]
- Turville, S.G.; Santos, J.J.; Frank, I.; Cameron, P.U.; Wilkinson, J.; Miranda-Saksena, M.; Dable, J.; Stossel, H.; Romani, N.; Piatak, M., Jr.; et al. Immunodeficiency virus uptake, turnover, and 2-phase transfer in human dendritic cells. Blood 2004, 103, 2170–2179. [Google Scholar] [CrossRef]
- Lepelley, A.; Louis, S.; Sourisseau, M.; Law, H.K.; Pothlichet, J.; Schilte, C.; Chaperot, L.; Plumas, J.; Randall, R.E.; Si-Tahar, M.; et al. Innate sensing of HIV-infected cells. PLoS Pathog. 2011, 7, e1001284. [Google Scholar] [CrossRef]
- Manel, N.; Hogstad, B.; Wang, Y.; Levy, D.E.; Unutmaz, D.; Littman, D.R. A cryptic sensor for HIV-1 activates antiviral innate immunity in dendritic cells. Nature 2010, 467, 214–217. [Google Scholar] [CrossRef]
- Laguette, N.; Sobhian, B.; Casartelli, N.; Ringeard, M.; Chable-Bessia, C.; Segeral, E.; Yatim, A.; Emiliani, S.; Schwartz, O.; Benkirane, M. SAMHD1 is the dendritic- and myeloid-cell-specific HIV-1 restriction factor counteracted by Vpx. Nature 2011, 474, 654–657. [Google Scholar] [CrossRef]
- Malim, M.H.; Emerman, M. HIV-1 accessory proteins—Ensuring viral survival in a hostile environment. Cell Host Microbe 2008, 3, 388–398. [Google Scholar] [CrossRef]
- Neil, S.; Bieniasz, P. Human immunodeficiency virus, restriction factors, and interferon. J. Interferon Cytokine Res. 2009, 29, 569–580. [Google Scholar] [CrossRef]
- Sheehy, A.M.; Gaddis, N.C.; Choi, J.D.; Malim, M.H. Isolation of a human gene that inhibits HIV-1 infection and is suppressed by the viral Vif protein. Nature 2002, 418, 646–650. [Google Scholar] [CrossRef]
- Trapp, S.; Derby, N.R.; Singer, R.; Shaw, A.; Williams, V.G.; Turville, S.G.; Bess, J.W., Jr.; Lifson, J.D.; Robbiani, M. Double-stranded RNA analog poly(I:C) inhibits human immunodeficiency virus amplification in dendritic cells via type I interferon-mediated activation of APOBEC3G. J. Virol. 2009, 83, 884–895. [Google Scholar] [CrossRef]
- Casartelli, N.; Sourisseau, M.; Feldmann, J.; Guivel-Benhassine, F.; Mallet, A.; Marcelin, A.G.; Guatelli, J.; Schwartz, O. Tetherin restricts productive HIV-1 cell-to-cell transmission. PLoS Pathog. 2010, 6, e1000955. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.R. Are viruses inhibited by APOBEC3 molecules from their host species? PLoS Pathog. 2009, 5, e1000347. [Google Scholar] [CrossRef] [PubMed]
- Nisole, S.; Stoye, J.P.; Saib, A. TRIM family proteins: Retroviral restriction and antiviral defence. Nat. Rev. Microbiol. 2005, 3, 799–808. [Google Scholar] [CrossRef]
- Gack, M.U.; Shin, Y.C.; Joo, C.H.; Urano, T.; Liang, C.; Sun, L.; Takeuchi, O.; Akira, S.; Chen, Z.; Inoue, S.; Jung, J.U. TRIM25 RING-finger E3 ubiquitin ligase is essential for RIG-I-mediated antiviral activity. Nature 2007, 446, 916–920. [Google Scholar] [CrossRef]
- Pitha, P.M.; Rowe, W.P.; Oxman, M.N. Effect of interferon on exogenous, endogenous, and chroniv murine leukemia virus infection. Virology 1976, 70, 324–338. [Google Scholar] [CrossRef] [PubMed]
- Baca-Regen, L.; Heinzinger, N.; Stevenson, M.; Gendelman, H.E. Alpha interferon-induced antiretroviral activities: Restriction of viral nucleic acid synthesis and progeny virion production in human immunodeficiency virus type 1-infected monocytes. J. Virol. 1994, 68, 7559–7565. [Google Scholar] [CrossRef]
- Goujon, C.; Malim, M.H. Characterization of the alpha interferon-induced postentry block to HIV-1 infection in primary human macrophages and T cells. J. Virol. 2010, 84, 9254–9266. [Google Scholar] [CrossRef]
- Shirazi, Y.; Pitha, P.M. Alpha interferon inhibits early stages of the human immunodeficiency virus type 1 replication cycle. J. Virol. 1992, 66, 1321–1328. [Google Scholar] [CrossRef]
- Cheney, K.M.; McKnight, A. Interferon-alpha mediates restriction of human immunodeficiency virus type-1 replication in primary human macrophages at an early stage of replication. PLoS ONE 2010, 5, e13521. [Google Scholar] [CrossRef]
- Kunzi, M.S.; Pitha, P.M. Role of interferon-stimulated gene ISG-15 in the interferon-omega-mediated inhibition of human immunodeficiency virus replication. J. Interferon Cytokine Res. 1996, 16, 919–927. [Google Scholar] [CrossRef]
- Kaletsky, R.L.; Francica, J.R.; Agrawal-Gamse, C.; Bates, P. Tetherin-mediated restriction of filovirus budding is antagonized by the Ebola glycoprotein. Proc. Natl. Acad. Sci. U. S. A. 2009, 106, 2886–2891. [Google Scholar] [CrossRef]
- Jouvenet, N.; Neil, S.J.; Zhadina, M.; Zang, T.; Kratovac, Z.; Lee, Y.; McNatt, M.; Hatziioannou, T.; Bieniasz, P.D. Broad-spectrum inhibition of retroviral and filoviral particle release by tetherin. J. Virol. 2009, 83, 1837–1844. [Google Scholar] [CrossRef]
- Chiu, Y.L.; Greene, W.C. APOBEC3G: an intracellular centurion. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 689–703. [Google Scholar] [CrossRef]
- Douglas, J.L.; Viswanathan, K.; McCarroll, M.N.; Gustin, J.K.; Fruh, K.; Moses, A.V. Vpu directs the degradation of the human immunodeficiency virus restriction factor BST-2/Tetherin via a {beta}TrCP-dependent mechanism. J. Virol. 2009, 83, 7931–7947. [Google Scholar] [CrossRef]
- Goffinet, C.; Allespach, I.; Homann, S.; Tervo, H.M.; Habermann, A.; Rupp, D.; Oberbremer, L.; Kern, C.; Tibroni, N.; Welsch, S.; et al. HIV-1 antagonism of CD317 is species specific and involves Vpu-mediated proteasomal degradation of the restriction factor. Cell Host Microbe 2009, 5, 285–297. [Google Scholar] [CrossRef]
- Bour, S.; Schubert, U.; Strebel, K. The human immunodeficiency virus type 1 Vpu protein specifically binds to the cytoplasmic domain of CD4: implications for the mechanism of degradation. J. Virol. 1995, 69, 1510–1520. [Google Scholar] [CrossRef]
- Sauter, D.; Schindler, M.; Specht, A.; Landford, W.N.; Munch, J.; Kim, K.A.; Votteler, J.; Schubert, U.; Bibollet-Ruche, F.; Keele, B.F.; et al. Tetherin-driven adaptation of Vpu and Nef function and the evolution of pandemic and nonpandemic HIV-1 strains. Cell Host Microbe 2009, 6, 409–421. [Google Scholar] [CrossRef]
- Solis, M.; Nakhaei, P.; Jalalirad, M.; Lacoste, J.; Douville, R.; Arguello, M.; Zhao, T.; Laughrea, M.; Wainberg, M.A.; Hiscott, J. RIG-I-mediated antiviral signaling is inhibited in HIV-1 infection by a protease-mediated sequestration of RIG-I. J. Virol. 2011, 85, 1224–1236. [Google Scholar] [CrossRef]
- Yan, N.; Regalado-Magdos, A.D.; Stiggelbout, B.; Lee-Kirsch, M.A.; Lieberman, J. The cytosolic exonuclease TREX1 inhibits the innate immune response to human immunodeficiency virus type 1. Nat. Immunol. 2010, 11, 1005–1013. [Google Scholar] [CrossRef]
- Akhtar, L.N.; Benveniste, E.N. Viral exploitation of host SOCS protein functions. J. Virol. 2011, 85, 1912–1921. [Google Scholar] [CrossRef]
- Witwer, K.W.; Sisk, J.M.; Gama, L.; Clements, J.E. MicroRNA regulation of IFN-beta protein expression: rapid and sensitive modulation of the innate immune response. J. Immunol. 2010, 184, 2369–2376. [Google Scholar] [CrossRef] [PubMed]
- Bednarik, D.P.; Mosca, J.D.; Raj, N.B.K.; Pitha, P.M. Inhibition of human immunodeficiency virus (HIV) replication by HIV-trans-activated à2-interferon. Proc. Natl. Acad. Sci. U. S. A. 1989, 86, 4958–4962. [Google Scholar] [CrossRef] [PubMed]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/)