Efficient Sensing of Avian Influenza Viruses by Porcine Plasmacytoid Dendritic Cells
Abstract
1. Introduction
2. Results
2.1. General Characteristics of pDC Responses Induced by AIV
| Host | Subtype | Complete Name | Designationa | HAU | TCID50/mL | CT Valuesb |
|---|---|---|---|---|---|---|
| avian | HP H5N1 | A/turkey/Turkey/05 | H5N1 TT05 | 1280 | 7.9 × 106 | 12.51 ± 0.05 |
| HP H5N1 | A/cygnus/Italy/742/06 | H5N1 CI06 | 1280 | 5.0 × 107 | 11.76 ± 0.04 | |
| HP H5N1 | A/mallard/Italy/835/06 | H5N1 MI06 | 640 | 3.2 × 104 | 13.11 ± 0.02 | |
| HP H7N1 | A/turkey/Italy/4580/98 | H7N1 TI98 | 1280 | 3.2 × 106 | 14.24 ± 0.09 | |
| HP H7N1 | A/ostrich/Italy/2332/00 | H7N1 OI00 | 1280 | 2.0 × 107 | 13.17 ± 0.10 | |
| LP H7N1 | A/turkey/Italy/3675/99 | H7N1 TI99 | 320 | 3.2 × 104 | 14.51 ± 0.02 | |
| human | H1N1 | A/New Caledonia/20/99 | H1N1 NC99 | 320 | 3.2 × 106 | 18.28 ± 0.18 |
| H3N2 | A/Wisconsin/67/05 | H3N2 WS05 | 1280 | 3.2 × 106 | 12.86 ± 0.06 | |
| H3N2 | A/Hong Kong/8/68 | H3N2 HK68 | 320 | 1.3 × 106 | 17.17 ± 0.15 | |
| porcine | H1N1 | A/swine/Iowa/1976/31 | H1N1 SI76 | 640 | 5.0 × 106 | 16.54 ± 0.06 |
| H1N1 | A/swine/Belgium/1/98 | H1N1 SB98 | 320 | 5.0 × 106 | 16.32 ± 0.06 | |
| H3N2 | A/swine/Flanders/1/98 | H3N2 SF98 | 640 | 3.2 × 106 | 17.50 ± 0.05 |
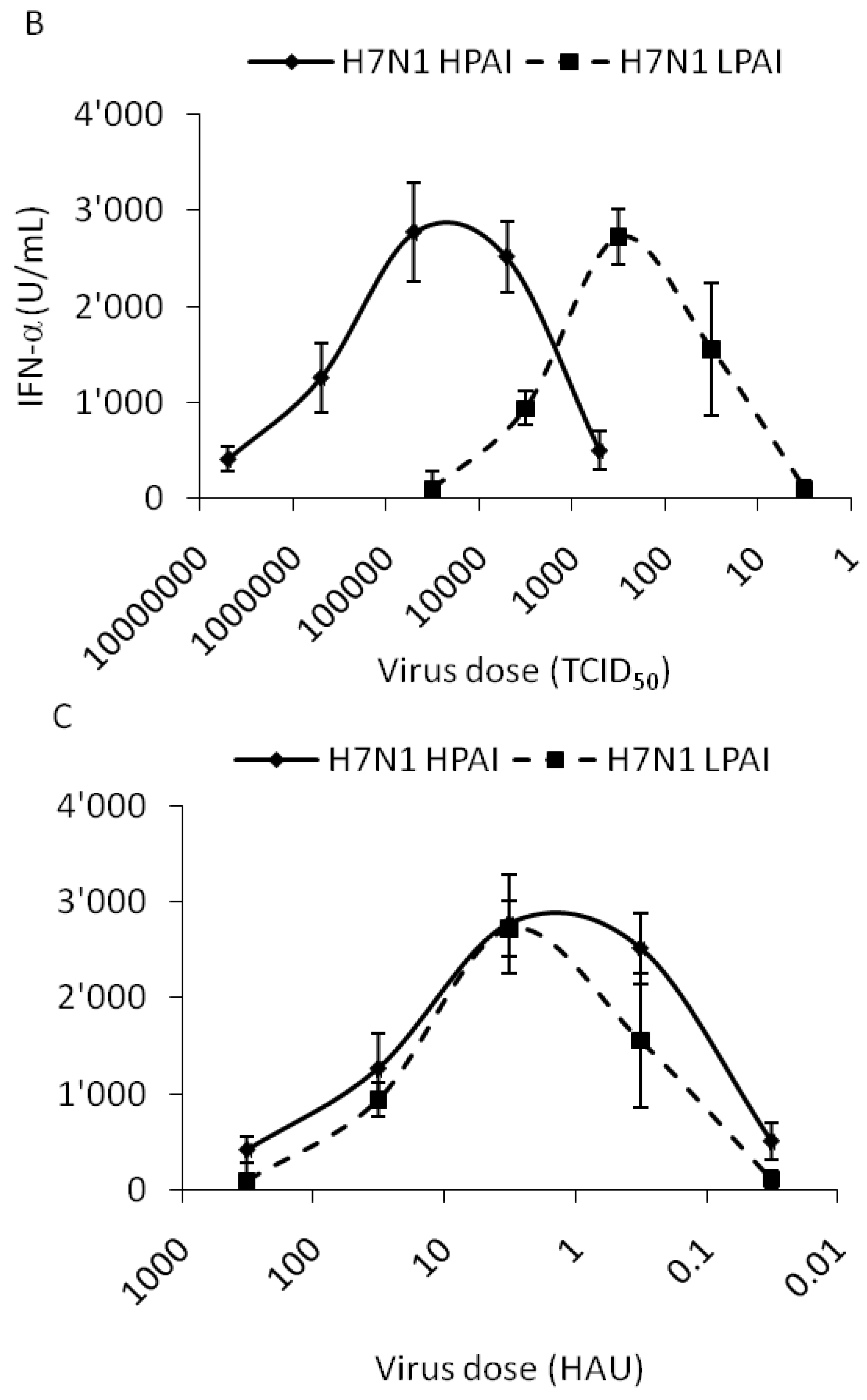
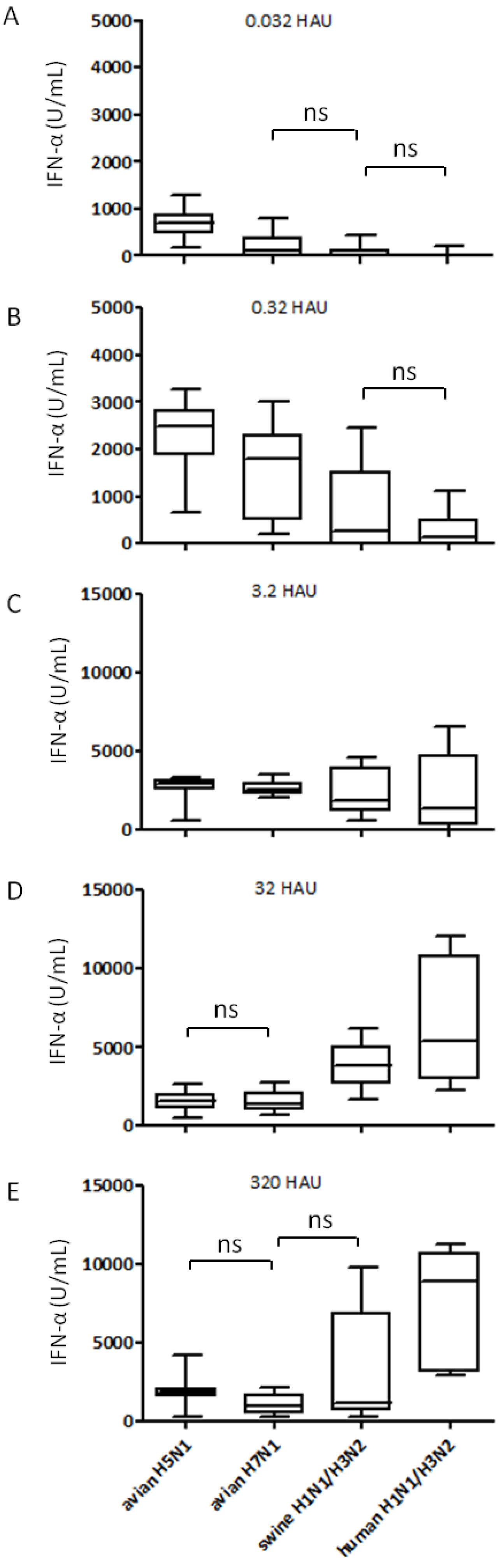
2.2. Interferon Response of Avian and Mammalian Strains
2.3. IFN-α Responses are not Correlated to Viral RNA Content
2.4. Relation between pDC Percentage and IFN-α
2.5. Infection of pDC and Monocytic Cells
2.6. Sialic-Acid Dependant Entry of IAV in pDC

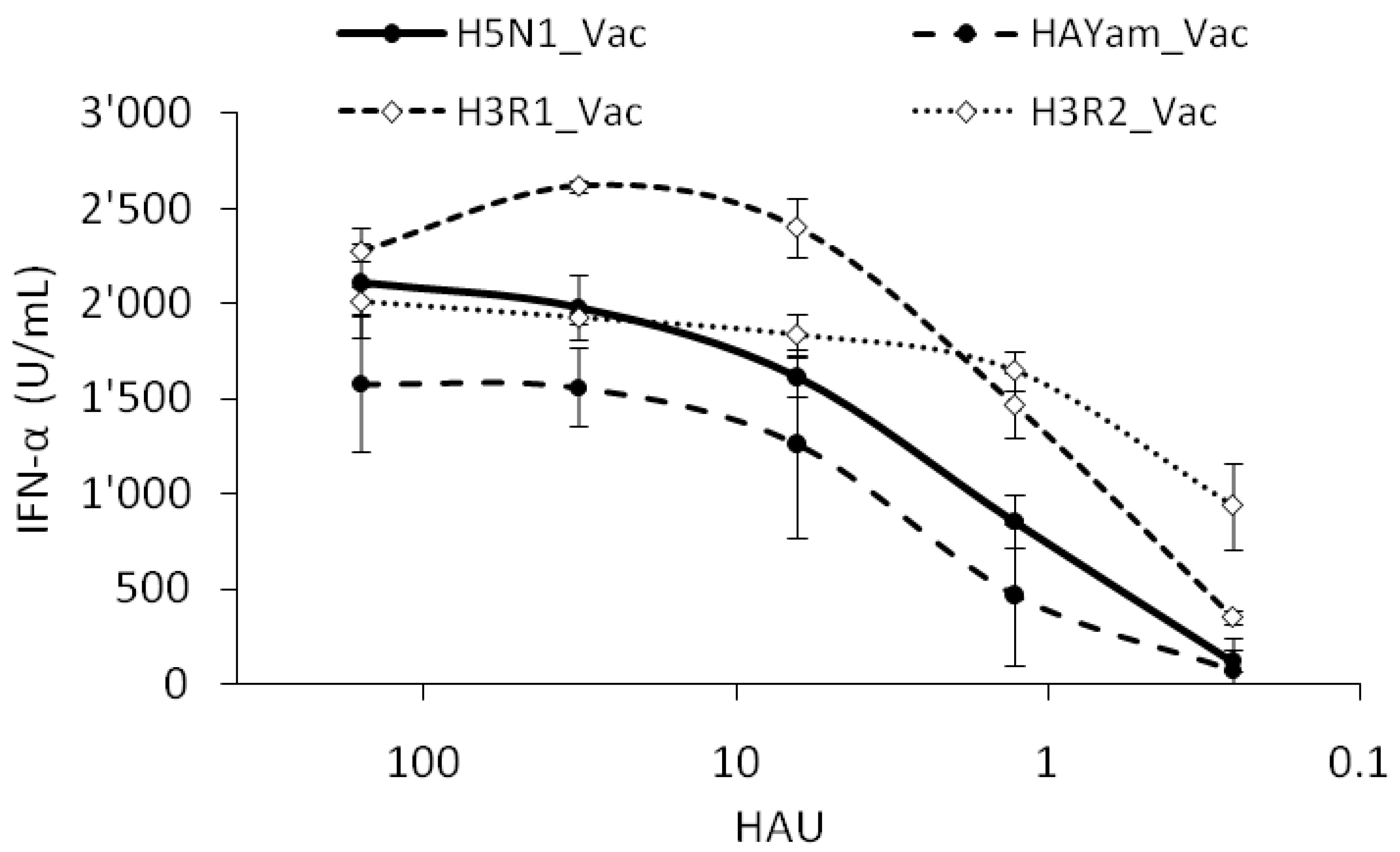
2.7. Reverse Genetic Viruses and Interferogenic Profile

| Designation | Subtype | Segment Substitution | Mutations | SA Binding Properties | Backbone | Cleavage Site |
|---|---|---|---|---|---|---|
| R1 | H3N2 | H3/N2 from HK68 | None | α-2,6 | H1N1 WS33 | Monobasic |
| R2 | H3N2 | H3/N2 from HK68 | L226Q/S228Gd | α-2,3 | H1N1 WS33 | Monobasic |
| Vac | H5N1 | N1/NS from H7N1a | None | α-2,3 | H5N2b | Monobasic |
| HAYam_Vac | H5N1 | H5 from H5N1c | None | α-2,3 | Vac | Polybasic |
| H3R1_Vac | H3N1 | H3 (R1) from HK68 | None | α-2,6 | Vac | Monobasic |
| H3R2_Vac | H3N1 | H3 (R2) from HK68 | L226Q/S228Gd | α-2,3 | Vac | Monobasic |
3. Experimental Section
3.1. Viruses and Cell Lines
3.2. Virus Inactivation
3.3. Reverse Genetic Viruses
3.4. Viral RNA Quantification
3.5. pDC Enrichment and Stimulation with Influenza
3.6. Flow Cytometry
3.7. Statistical analyses
4. Discussion and Conclusions
Acknowledgements
References and Notes
- de Jong, M.D.; Simmons, C.P.; Thanh, T.T.; Hien, V.M.; Smith, G.J.; Chau, T.N.; Hoang, D.M.; Chau, N.V.; Khanh, T.H.; Dong, V.C.; et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat. Med. 2006, 12, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Beigel, J.H.; Farrar, J.; Han, A.M.; Hayden, F.G.; Hyer, R.; de Jong, M.D.; Lochindarat, S.; Nguyen, T.K.; Nguyen, T.H.; Tran, T.H.; et al. Avian influenza A (H5N1) infection in humans. N. Engl. J. Med. 2005, 353, 1374–1385. [Google Scholar] [PubMed]
- Chan, M.C.; Cheung, C.Y.; Chui, W.H.; Tsao, S.W.; Nicholls, J.M.; Chan, Y.O.; Chan, R.W.; Long, H.T.; Poon, L.L.; Guan, Y.; et al. Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells. Respir. Res. 2005, 6, 135. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Poon, L.L.; Lau, A.S.; Luk, W.; Lau, Y.L.; Shortridge, K.F.; Gordon, S.; Guan, Y.; Peiris, J.S. Induction of proinflammatory cytokines in human macrophages by influenza A (H5N1) viruses: A mechanism for the unusual severity of human disease? Lancet 2002, 360, 1831–1837. [Google Scholar] [CrossRef] [PubMed]
- Lindenmann, J.; Burke, D.C.; Isaacs, A. Studies on the production, mode of action and properties of interferon. Br. J. Exp. Pathol. 1957, 38, 551–562. [Google Scholar] [PubMed]
- Siegal, F.P.; Kadowaki, N.; Shodell, M.; Fitzgerald-Bocarsly, P.A.; Shah, K.; Ho, S.; Antonenko, S.; Liu, Y.J. The nature of the principal type 1 interferon-producing cells in human blood. Science. 1999, 284, 1835–1837. [Google Scholar] [CrossRef]
- Jewell, N.A.; Vaghefi, N.; Mertz, S.E.; Akter, P.; Peebles, R.S., Jr.; Bakaletz, L.O.; Durbin, R.K.; Flano, E.; Durbin, J.E. Differential type I interferon induction by respiratory syncytial virus and influenza a virus in vivo. J. Virol. 2007, 81, 9790–9800. [Google Scholar] [CrossRef]
- Tsoumakidou, M.; Tzanakis, N.; Papadaki, H.A.; Koutala, H.; Siafakas, N.M. Isolation of myeloid and plasmacytoid dendritic cells from human bronchoalveolar lavage fluid. Immunol. Cell. Biol. 2006, 84, 267–273. [Google Scholar] [CrossRef]
- GraphPad Prism, version 5.03; GraphPad Software: La Jolla, CA, USA, 2010.[Green Version]
- Guzylack-Piriou, L.; Alves, M.P.; McCullough, K.C.; Summerfield, A. Porcine Flt3 ligand and its receptor: Generation of dendritic cells and identification of a new marker for porcine dendritic cells. Dev. Comp. Immunol. 2010, 34, 455–464. [Google Scholar] [CrossRef]
- Matrosovich, M.; Matrosovich, T.; Uhlendorff, J.; Garten, W.; Klenk, H.D. Avian-virus-like receptor specificity of the hemagglutinin impedes influenza virus replication in cultures of human airway epithelium. Virology 2007, 361, 384–390. [Google Scholar] [CrossRef]
- Soda, K.; Sakoda, Y.; Isoda, N.; Kajihara, M.; Haraguchi, Y.; Shibuya, H.; Yoshida, H.; Sasaki, T.; Sakamoto, R.; Saijo, K.; et al. Development of vaccine strains of H5 and H7 influenza viruses. Jpn. J. Vet. Res. 2008, 55, 93–98. [Google Scholar] [PubMed]
- Isoda, N.; Sakoda, Y.; Kishida, N.; Bai, G.R.; Matsuda, K.; Umemura, T.; Kida, H. Pathogenicity of a highly pathogenic avian influenza virus, A/chicken/Yamaguchi/7/04 (H5N1) in different species of birds and mammals. Arch. Virol. 2006, 151, 1267–1279. [Google Scholar] [CrossRef] [PubMed]
- Bahnemann, H.G. Binary ethylenimine as an inactivant for foot-and-mouth disease virus and its application for vaccine production. Arch. Virol. 1975, 47, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Bahnemann, H.G. Inactivation of viral antigens for vaccine preparation with particular reference to the application of binary ethylenimine. Vaccine 1990, 8, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Aarthi, D.; Ananda Rao, K.; Robinson, R.; Srinivasan, V.A. Validation of binary ethyleneimine (BEI) used as an inactivant for foot and mouth disease tissue culture vaccine. Biologicals 2004, 32, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, E.; Krauss, S.; Perez, D.; Webby, R.; Webster, R.G. Eight-plasmid system for rapid generation of influenza virus vaccines. Vaccine 2002, 20, 3165–3170. [Google Scholar] [CrossRef] [PubMed]
- Ocana-Macchi, M.; Bel, M.; Guzylack-Piriou, L.; Ruggli, N.; Liniger, M.; McCullough, K.C.; Sakoda, Y.; Isoda, N.; Matrosovich, M.; Summerfield, A. Hemagglutinin-dependent tropism of H5N1 avian influenza virus for human endothelial cells. J. Virol. 2009, 83, 12947–12955. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, M.A.; Renzullo, S.; Baumer, A. Phylogenetic characterization of H5N1 highly pathogenic avian influenza viruses isolated in Switzerland in 2006. Virus Genes. 2008, 37, 407–413. [Google Scholar] [CrossRef]
- Spackman, E.; Senne, D.A.; Myers, T.J.; Bulaga, L.L.; Garber, L.P.; Perdue, M.L.; Lohman, K.; Daum, L.T.; Suarez, D.L. Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. J. Clin. Microbiol. 2002, 40, 3256–3260. [Google Scholar] [CrossRef]
- Guzylack-Piriou, L.; Balmelli, C.; McCullough, K.C.; Summerfield, A. Type-A CpG oligonucleotides activate exclusively porcine natural interferon-producing cells to secrete interferon-alpha, tumour necrosis factor-alpha and interleukin-12. Immunology. 2004, 112, 28–37. [Google Scholar] [CrossRef]
- Summerfield, A.; Guzylack-Piriou, L.; Schaub, A.; Carrasco, C.P.; Tache, V.; Charley, B.; McCullough, K.C. Porcine peripheral blood dendritic cells and natural interferon-producing cells. Immunology. 2003, 110, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Summerfield, A.; McCullough, K.C. Porcine bone marrow myeloid cells: Phenotype and adhesion molecule expression. J. Leukoc. Biol. 1997, 62, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Sandbulte, M.R.; Boon, A.C.; Webby, R.J.; Riberdy, J.M. Analysis of cytokine secretion from human plasmacytoid dendritic cells infected with H5N1 or low-pathogenicity influenza viruses. Virology. 2008, 381, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Thitithanyanont, A.; Engering, A.; Ekchariyawat, P.; Wiboon-ut, S.; Limsalakpetch, A.; Yongvanitchit, K.; Kum-Arb, U.; Kanchongkittiphon, W.; Utaisincharoen, P.; Sirisinha, S.; et al. High susceptibility of human dendritic cells to avian influenza H5N1 virus infection and protection by IFN-alpha and TLR ligands. J. Immunol. 2007, 179, 5220–5227. [Google Scholar] [CrossRef]
- Summerfield, A.; McCullough, K.C. Dendritic Cells in Innate and Adaptive Immune Responses against Influenza Virus. Viruses. 2009, 1, 1022–1034. [Google Scholar] [CrossRef]
- Peiris, J.S.; Cheung, C.Y.; Leung, C.Y.; Nicholls, J.M. Innate immune responses to influenza A H5N1: Friend or foe? Trends Immunol. 2009, 30, 574–584. [Google Scholar] [CrossRef]
- Wolf, A.I.; Buehler, D.; Hensley, S.E.; Cavanagh, L.L.; Wherry, E.J.; Kastner, P.; Chan, S.; Weninger, W. Plasmacytoid dendritic cells are dispensable during primary influenza virus infection. J. Immunol. 2009, 182, 871–879. [Google Scholar] [CrossRef]
- Fonteneau, J.F.; Gilliet, M.; Larsson, M.; Dasilva, I.; Munz, C.; Liu, Y.J.; Bhardwaj, N. Activation of influenza virus-specific CD4+ and CD8+ T cells: A new role for plasmacytoid dendritic cells in adaptive immunity. Blood 2003, 101, 3520–3526. [Google Scholar] [CrossRef]
- Lui, G.; Manches, O.; Angel, J.; Molens, J.P.; Chaperot, L.; Plumas, J. Plasmacytoid dendritic cells capture and cross-present viral antigens from influenza-virus exposed cells. PLoS ONE 2009, 4, e7111. [Google Scholar] [CrossRef]
- GeurtsvanKessel, C.H.; Willart, M.A.; van Rijt, L.S.; Muskens, F.; Kool, M.; Baas, C.; Thielemans, K.; Bennett, C.; Clausen, B.E.; Hoogsteden, H.C.; et al. Clearance of influenza virus from the lung depends on migratory langerin+CD11b- but not plasmacytoid dendritic cells. J. Exp. Med. 2008, 205, 1621–1634. [Google Scholar] [CrossRef]
- Langlois, R.A.; Legge, K.L. Plasmacytoid dendritic cells enhance mortality during lethal influenza infections by eliminating virus-specific CD8 T cells. J. Immunol. 2010, 184, 4440–4446. [Google Scholar] [CrossRef] [PubMed]
- Tumpey, T.M.; Szretter, K.J.; Van Hoeven, N.; Katz, J.M.; Kochs, G.; Haller, O.; Garcia-Sastre, A.; Staeheli, P. The Mx1 gene protects mice against the pandemic 1918 and highly lethal human H5N1 influenza viruses. J. Virol. 2007, 81, 10818–10821. [Google Scholar] [CrossRef] [PubMed]
- Haller, O.; Frese, M.; Kochs, G. Mx proteins: Mediators of innate resistance to RNA viruses. Rev. Sci. Tech. 1998, 17, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Hayden, F.G.; Fritz, R.; Lobo, M.C.; Alvord, W.; Strober, W.; Straus, S.E. Local and systemic cytokine responses during experimental human influenza A virus infection. Relation to symptom formation and host defense. J. Clin. Invest. 1998, 101, 643–649. [Google Scholar] [PubMed]
- Van Reeth, K.; Van Gucht, S.; Pensaert, M. Correlations between lung proinflammatory cytokine levels, virus replication, and disease after swine influenza virus challenge of vaccination-immune pigs. Viral Immunol. 2002, 15, 583–594. [Google Scholar] [CrossRef]
- Baskin, C.R.; Bielefeldt-Ohmann, H.; Tumpey, T.M.; Sabourin, P.J.; Long, J.P.; Garcia-Sastre, A.; Tolnay, A.E.; Albrecht, R.; Pyles, J.A.; Olson, P.H.; et al. Early and sustained innate immune response defines pathology and death in nonhuman primates infected by highly pathogenic influenza virus. Proc. Natl. Acad. Sci. U. S. A. 2009, 106, 3455–3460. [Google Scholar] [CrossRef]
- Choi, Y.K.; Nguyen, T.D.; Ozaki, H.; Webby, R.J.; Puthavathana, P.; Buranathal, C.; Chaisingh, A.; Auewarakul, P.; Hanh, N.T.; Ma, S.K.; et al. Studies of H5N1 influenza virus infection of pigs by using viruses isolated in Vietnam and Thailand in 2004. J. Virol. 2005, 79, 10821–10825. [Google Scholar] [CrossRef]
- Lipatov, A.S.; Kwon, Y.K.; Sarmento, L.V.; Lager, K.M.; Spackman, E.; Suarez, D.L.; Swayne, D.E. Domestic pigs have low susceptibility to H5N1 highly pathogenic avian influenza viruses. PLoS Pathog. 2008, 4, e1000102. [Google Scholar] [CrossRef]
- Londrigan, S.L.; Turville, S.G.; Tate, M.D.; Deng, Y.M.; Brooks, A.G.; Reading, P.C. N-linked Glycosylation Facilitates Sialic Acid-Independent Attachment and Entry of Influenza A Viruses into Cells Expressing DC-SIGN or L-SIGN. J. Virol. 2011, 85, 2990–3000. [Google Scholar] [CrossRef]
- Wang, S.F.; Huang, J.C.; Lee, Y.M.; Liu, S.J.; Chan, Y.J.; Chau, Y.P.; Chong, P.; Chen, Y.M. DC-SIGN mediates avian H5N1 influenza virus infection in cis and in trans. Biochem. Biophys. Res. Commun. 2008, 373, 561–566. [Google Scholar] [CrossRef]

© 2011 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bel, M.; Ocaña-Macchi, M.; Liniger, M.; McCullough, K.C.; Matrosovich, M.; Summerfield, A. Efficient Sensing of Avian Influenza Viruses by Porcine Plasmacytoid Dendritic Cells. Viruses 2011, 3, 312-330. https://doi.org/10.3390/v3040312
Bel M, Ocaña-Macchi M, Liniger M, McCullough KC, Matrosovich M, Summerfield A. Efficient Sensing of Avian Influenza Viruses by Porcine Plasmacytoid Dendritic Cells. Viruses. 2011; 3(4):312-330. https://doi.org/10.3390/v3040312
Chicago/Turabian StyleBel, Michael, Manuela Ocaña-Macchi, Matthias Liniger, Kenneth C. McCullough, Mikhail Matrosovich, and Artur Summerfield. 2011. "Efficient Sensing of Avian Influenza Viruses by Porcine Plasmacytoid Dendritic Cells" Viruses 3, no. 4: 312-330. https://doi.org/10.3390/v3040312
APA StyleBel, M., Ocaña-Macchi, M., Liniger, M., McCullough, K. C., Matrosovich, M., & Summerfield, A. (2011). Efficient Sensing of Avian Influenza Viruses by Porcine Plasmacytoid Dendritic Cells. Viruses, 3(4), 312-330. https://doi.org/10.3390/v3040312






