Ecopathology of Ranaviruses Infecting Amphibians
Abstract
1. Global Impact
1.1. Distribution of Ranaviruses in Amphibians
1.2. Species Susceptibility
2. Pathology
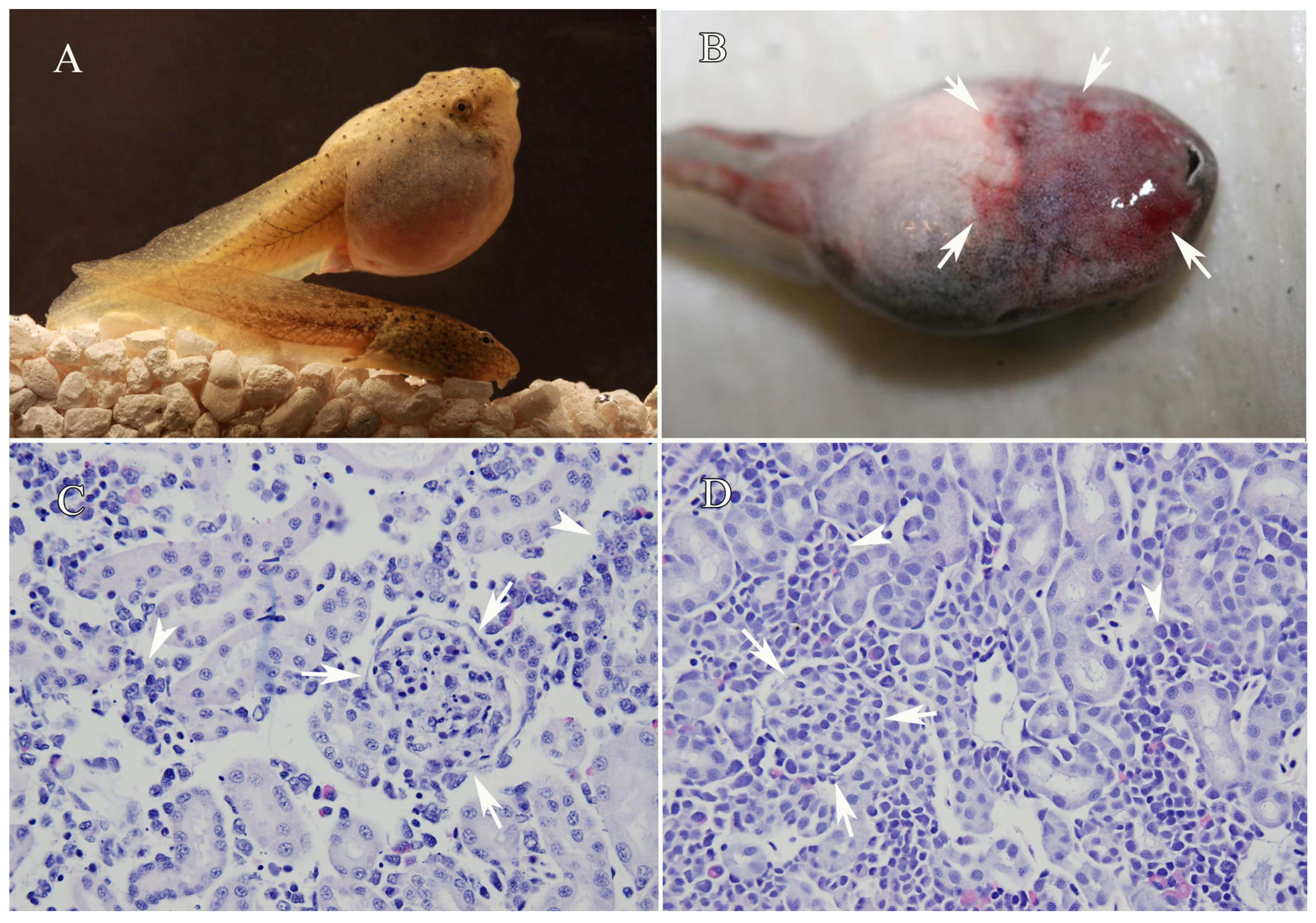
2.1. Clinical/Field Signs of Ranaviral Disease
2.2. Histopathological Findings
2.3. Evidence of Subclinical Infections
3. Ecology
3.1. Population Impacts
3.2. Coevolution
3.3. Transmission
4. Conservation
4.1. Anthropogenic Stressors
4.2. Captive Breeding and Amphibian Trade
4.3. Management Options
5. Future Directions
Acknowledgments
Conflicts of Interest
References and Notes
- Gray, M.J.; Miller, D.L.; Hoverman, J.T. Ecology and pathology of amphibian ranaviruses. Dis. Aquat. Organ. 2009, 87, 243–266. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, A.A.; Langton, T.E.S.; Bennett, P.M.; Lewin, J.F.; Drury, S.E.N.; Gough, R.E.; MacGregor, S.K. Pathological and Microbiological Findings from Incidents of Unusual Mortality of the Common Frog (Rana temporaria). Philos. Trans. R. Soc. Lon. B. Biol. Sci. 1996, 351, 1539–1557. [Google Scholar]
- Green, D.E.; Converse, K.A.; Schrader, A.K. Epizootiology of Sixty-Four Amphibian Morbidity and Mortality Events in the USA, 1996–2001. Ann. NY Acad. Sci. 2002, 969, 323–339. [Google Scholar] [CrossRef]
- Schock, D.M.; Bollinger, T.K. An Apparent Decline of Northern Leopard Frogs (Rana pipiens) on the Rafferty Dam Mitigation Lands near Esteven, Saskatchewan. Blue Jay. 2005, 63, 144–154. [Google Scholar] [CrossRef]
- Ariel, E.; Kielgast, J.; Svart, H.E.; Larsen, K.; Tapiovaara, H.; Bang Jensen, B.; Holopainen, R. Ranavirus in Wild Edible Frogs, Pelophylax kl. Esculentus, in Denmark. Dis. Aquat. Organ. 2009, 85, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Teacher, A.G.F.; Cunningham, A.A.; Garner, W.J. Assessing the Long-Term Impact of Ranavirus Infection in Wild Common Frog Populations. Anim. Con. 2010, 13, 514–522. [Google Scholar] [CrossRef]
- Schock, D.M.; Bollinger, T.K.; Chinchar, V.G.; Jancovich, J.K.; Collins, J.P. Experimental Evidence that Amphibian Ranaviruses Are Multi-Host Pathogens. Copeia 2008, 1, 133–143. [Google Scholar] [CrossRef]
- Hoverman, J.T.; Gray, M.J.; Miller, D.L. Anuran Susceptibilities to Ranavirus: The Role of Species Identity, Exposure Route, and Novel Virus Isolates. Dis. Aquat. Organ. 2010, 89, 97–107. [Google Scholar] [CrossRef]
- Hoverman, J.T.; Gray, M.J.; Haislip, J.T.; Miller, D.L. Phylogeny, life history, and ecology contribute to differences in amphibian susceptibility to ranaviruses. EcoHealth 2011. [Google Scholar] [CrossRef]
- Haislip, N.A.; Gray, M.J.; Hoverman, J.T.; Miller, D.L. Development and Disease: How Susceptibility to an Emerging Pathogen Changes through Anuran Development. PLoS One 2011, 6, e22307. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Wang, K.Y.; Zhou, Z.Y.; Li, C.W.; Wang, J.; He, M.; Yin, Z.Q.; Lai, W.M. First Report of a Ranavirus Associated with Morbidity and Mortality in Farmed Chinese Gian Salamanders (Andrias davidianus). J. Comp. Pathol. 2011, 145, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Zhu, D.; Wei, Y.; Schloegel, L.M.; Chen, X.; Wang, X. Broad Distribution of Ranavirus in Free-Ranging Rana dybowskii in Heilongjiang, China. EcoHealth 2010, 7, 18–23. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, Z.; Jiang, Y.; Liang, S.; Gui, J. Preliminary Studies on Virus Isolation and Cell Infection from Diseased Frog Rana grylio. Acta Hydrobiologica Sinica 1996, 4, 390–392. [Google Scholar]
- Zhang, Q.Y.; Xiao, F.; Li, Z.Q.; Gui, J.F.; Mao, J.; Chinchar, V.G. Characterization of an Iridovirus from the Cultured Pig Frog, Rana grylio, with Lethal Syndrome. Dis. Aquat. Organ. 2001, 48, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Weng, S.P.; He, J.G.; Wang, X.H.; Lü, L.; Deng, M.; Chan, S.-M. Outbreaks of an Iridovirus Disease in Cultured Tiger Frog, Rana tigrina rugulosa, in Southern China. J. Fish. Dis. 2002, 25, 423–427. [Google Scholar] [CrossRef]
- Une, Y.; Nakajinma, K.; Taharaguchi, S.; Ogihara, K.; Murakami, M. Ranavirus Infection Outbreak in the Salamander (Hynobius nebulosus) in Japan. J. Comp. Pathol. 2009, 141, 310. [Google Scholar] [CrossRef]
- Une, Y.; Sakuma, A.; Matsueda, H.; Nakai, K.; Murakami, M. Ranavirus Outbreak in North American Bullfrogs (Rana catesbeiana), Japan. Emerg. Infect. Dis. 2009, 15, 1146–1147. [Google Scholar] [CrossRef]
- Kanchanakhan, S. An Ulcerative Disease of the Cultured Tiger Frog, Rana tigrina, in Thailand: Virological Examination. AAHRI News 1998, 7, 1–2. [Google Scholar]
- Speare, R.; Smith, J.R. An Iridovirus-Like Agent Isolated from the Ornate Burrowing Frog Limnodynastes ornatus in Northern Australia. Dis. Aquat. Organ. 1992, 14, 51–57. [Google Scholar] [CrossRef]
- Pasmans, F.; Blahak, S.; Martel, A.; Pantchev, N.; Zwart, P. Ranavirus-Associated Mass Mortality in Imported Red Tailed Knobby Newts (Tylototriton kweichowensis): A Case Report. Vet. J. 2008, 176, 257–259. [Google Scholar] [CrossRef]
- Fijan, N.; Matasin, Z.; Petrinec, Z.; Valpotic, I.; Zillenberg, L.O. Isolation of an Iridovirus-Like Agent from the Green Frog (Rana esculenta L.). Vet. Arch. 1991, 61, 151–158. [Google Scholar]
- Ariel, E.; Kielgast, J.; Svart, H.E.; Larsen, K.; Tapiovaara, H.; Bang Jensen, B.; Holopainen, R. Ranavirus in Wild Edible Frogs, Pelophylax kl. esculentus in Denmark. Dis. Aquat. Organ. 2009, 85, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Milstein, D.; King, R.; Leader, N. Israel Parks and Nature Authority, Jerusalem, Israel. Unpublished work, 2011.
- Kik, M.; Martel, A.; Spitzen-van der Sluijs, A.; Pasmans, F.; Wohlsein, P.; Gröne, A.; Rijks, J.M. Ranavirus-Associated Mass Mortality in Wild Amphibians, the Netherlands, 2010: A First Report. Vet. J. 2011. [Google Scholar] [CrossRef] [PubMed]
- Alves de Matos, A.P.; Caeiro, M.F.; Marschang, R.E.; Papp, T.; Soares, C.; Marçal, M.R.; Carretero, M.A. Adaptation of Ranaviruses from Peneda-Gerês National Park (Portugal) to Cell Cultures and Their Characterization. Microsc. Microanal. 2008, 14, 139–140. [Google Scholar] [CrossRef]
- Balseiro, A.; Dalton, K.P.; Cerro1, A.; Marquez, I.; Cunningham, A.A.; Parra, F.; Prieto, J.M.; Casais, R. Pathology, Isolation and Molecular Characterisation of a Ranavirus from the Common Midwife Toad Alytes obstetricans on the Iberian Peninsula. Dis. Aquat. Organ. 2009, 84, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Balseiro, A.; Dalton, K.P.; Del Cerro, A.; Marquez, I.; Parra, F.; Prieto, J.M.; Casias, R. Outbreak of Common Midwife Toad Virus in Alpine Newts (Mesotriton alpestris cyreni) and Common Midwife Toads (Alytes obstetricans) in Northern Spain: Comparative Pathological Study of an Emerging Ranavirus. Vet. J. 2010, 186, 256–258. [Google Scholar] [CrossRef]
- Marschang, R. Universität Hohenheim, Stuttgart, Germany. Unpublished work, 2011.
- Duffus, A.L.J. Ranavirus Ecology in Common Frogs (Rana temporaria) from the United Kingdom: Transmission Dynamics, Alternate Hosts and Host-Strain Interactions. Ph.D. Thesis, Queen Mary University of London, London, UK, 2010. [Google Scholar]
- Hyatt, A.D.; Gould, A.R.; Zupanovic, Z.; Cunningham, A.A.; Hengstberger, S.; Whittington, R.J.; Kattenbelt, J.; Coupar, B.E.H. Comparative Studies of Piscine and Amphibian Iridoviruses. Arch. Virol. 2010, 145, 301–331. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, A.A.; Langton, T.E.S.; Bennett, P.M.; Drury, S.E.N.; Gough, R.E.; Kirkwood, K.E. Unusual Mortality Associated with Poxvirus-Like Particles in Frogs (Rana temporaria). Vet. Rec. 1993, 133, 141–142. [Google Scholar] [CrossRef]
- Drury, S.E.N.; Gough, R.E.; Cunningham, A.A. Isolation of an Iridovirus-Like Agent from Common Frogs (Rana temporaria). Vet. Rec. 1995, 137, 72–73. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, T.K.; Mao, J.; Schock, D.; Brigham, R.M.; Chinchar, V.G. Pathology, Isolation, and Preliminary Molecular Characterization of a Novel Iridovirus from Tiger Salamanders in Saskatchewan. J. Wildl. Dis. 1999, 35, 413–429. [Google Scholar] [CrossRef] [PubMed]
- Schock, D.M.; Bollinger, T.K.; Collins, J.P. Mortality Rates Differ among Amphibian Populations Exposed to Three Strains of a Lethal Ranavirus. EcoHealth 2010, 6, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Goater, C. University of Lethbridge, Lethbridge, Alberta, Canada. Unpublished work, 2011.
- Duffus, A.L.J.; Pauli, B.D.; Wozney, K.; Brunetti, C.R.; Berrill, M. Frog Virus 3-Like Infections in Aquatic Amphibian Communities. J. Wildl. Dis. 2008, 44, 109–120. [Google Scholar] [CrossRef]
- Gahl, M. Bates College, Lewiston, ME, USA. Unpublished work, 2011.
- St-Amour, V.; Wong, W.M.; Garner, T.W.M.; Lesbarreres, D. Anthropogenic Influence on Prevalence of 2 Amphibian Pathogens. Emerg. Infect. Dis. 2008, 14, 1175–1176. [Google Scholar] [CrossRef] [PubMed]
- Forzán, M. Canadian Cooperative Wildlife Health Centre, Charlottetown, Prince Edward Island, Canada. Unpublished work, 2011.
- Greer, A.L.; Berrill, M.; Wilson, P.J. Five Amphibian Mortality Events Associated with Ranavirus Infection in South Central Ontario, Canada. Dis. Aquat. Organ. 2005, 67, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Echaubard, P.; Little, K.; Pauli, B.; Lesbarrères, D. Context-Dependent Effects of Ranaviral Infection on Northern Leopard Frog Life History Traits. PLoS One 2010, 5, e13723. [Google Scholar] [CrossRef]
- Paetow, L.J.; Pauli, B.D.; McLoughlin, J.D.; Bidulka, J.; Marcogliese, D.J. First Detection of Ranavirus in Lithobates pipiens in Quebec. Herpetol. Rev. 2011, 42, 211–214. [Google Scholar]
- Schock, D.M.; Ruthig, G.R.; Collins, J.P.; Kutz, S.J.; Carrière, S.; Gau, R.J.; Veitch, A.M.; Larter, N.C.; Tate, D.P.; Guthrie, G.; et al. Amphibian Chytrid Fungus and Ranaviruses in the Northwest Territories, Canada. Dis. Aquat. Organ. 2010, 92, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Speare, R.; Freeland, W.J.; Bolton, S.J. A Possible Iridovirus in Erythrocytes of Bufo marinus in Costa Rica. J. Wildl. Dis. 1991, 27, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Green, D.E.; Ip, H.S. US Geological Survey National Wildlife Health Center, Madison, Wisconsin, USA. Unpublished work, 2011.
- Brunner, J.L.; Barnett, K.E.; Gosier, C.J.; McNulty, S.A.; Rubbo, M.J.; Kolozsvary, M.B. Ranavirus Infection in Die-Offs of Vernal Pool Amphibians in New York, USA. Herpetol. Rev. 2011, 42, 76–79. [Google Scholar]
- Gahl, M.K.; Calhoun, A.J.K. The Role of Multiple Stressors in Ranavirus-Caused Amphibian Mortalities in Acadia National Park Wetlands. Can. J. Zool. 2010, 88, 108–118. [Google Scholar] [CrossRef]
- Petranka, J.W.; Murray, S.M.; Kennedy, C.A. Response of Amphibians to Restoration of a Southern Appalachian Wetland: Perturbations Confound Post-Restoration Assessment. Wetlands 2003, 23, 278–290. [Google Scholar] [CrossRef]
- Todd-Thompson, M. Seasonality, Variation in Species Prevalence, and Localized Disease for Ranavirus in Cades Cove (Great Smoky Mountains National Park) amphibians. Master Thesis, University of Tennessee, Knoxville, TN, USA, 2010. Available online: http://trace.tennessee.edu/utk_gradthes/665 (accessed on 17 November 2011).
- Docherty, D.E.; Meteyer, C.U.; Wang, J.; Mao, J.; Case, S.T.; Chinchar, V.G. Diagnostic and Molecular Evaluation of Three Iridovirus-Associated Salamander Mortality Events. J. Wildl. Dis. 2003, 39, 556–566. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Davidson, E.W.; Morado, J.F.; Jacobs, B.L.; Collins, J.P. Isolation of a Lethal Virus from the Endangered Tiger Salamander Ambystoma tigrinum stebbinsi. Dis. Aquat. Organ. 1997, 31, 161–167. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Davidson, E.W.; Parameswaran, N.; Mao, J.; Chinchar, V.G.; Collins, J.P.; Jacobs, B.L.; Storfer, A. Evidence for Emergence of an Amphibian Iridoviral Disease because of Human-Enhanced Spread. Mol. Ecol. 2005, 14, 213–224. [Google Scholar] [CrossRef]
- Picco, A.M.; Collins, J.P. Amphibian Commerce as a likely Source of Pathogen Pollution. Conserv. Biol. 2008, 22, 1582–1589. [Google Scholar] [CrossRef] [PubMed]
- Greer, A.L.; Brunner, J.L.; Collins, J.P. Spatial and Temporal Patterns of Ambystoma tigrinum Virus (ATV) Prevalence in Tiger Salamanders Ambystoma tigrinum nebulosum. Dis. Aquat. Organ. 2009, 85, 1–6. [Google Scholar] [CrossRef]
- Hoverman, J.T.; Gray, M.J.; Haislip, J.T.; Miller, D.L. Widespread occurrence of ranavirus in pond-breeding amphibian populations. EcoHealth in press, 2012.
- Converse, K.A.; Green, D.E. Diseases of Tadpoles. In Wildlife Diseases: Landscape Epidemiology, Spatial Distribution and Utilization of Remote Sensing Technology; Majumdar, S.K., Huffman, J.E., Brenner, F.J., Panah, A.I., Eds.; The Pennsylvania Academy of Science: Easton, PA, USA, 2005; Chapter 7; pp. 72–88. [Google Scholar]
- Jones, M.E.B.; Pessier, A.P. San Diego Zoo Global, San Diego, CA, USA. Unpublished work, 2011.
- Gray, M.; Miller, D.; Sousa, M. University of Tennessee, Knoxville, TN, USA. Unpublished work, 2011.
- Miller, D.L.; Rajeev, S.; Brookins, M.; Cook, J.; Whittington, L.; Baldwin, C.A. Concurrent Infection with Ranavirus, Batrachochytrium dendrobatidis, and Aeromonas in a Captive Anuran Colony. J. Zoo Wildl. Med. 2008, 39, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Driskell, E.A.; Miller, D.L.; Swist, S.L.; Gyimesi, Z.S. PCR Detection of Ranavirus in Adult Anurans from the Louisville Zoological Garden. J. Zoo Wildl. Med. 2009, 40, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Green, D.E.; Converse, K.A. Diseases of Frogs and Toads. In Wildlife Diseases: Landscape Epidemiology, Spatial Distribution and Utilization of Remote Sensing Technology; Majumdar, S.K., Huffman, J.E., Brenner, F.J., Panah, A.I., Eds.; The Pennsylvania Academy of Science: Easton, PA, USA, 2005; Chapter 8; pp. 89–117. [Google Scholar]
- Torrence, S.M.; Green, D.E.; Benson, C.J.; Ip, H.S.; Smith, L.M.; McMurray, S.T. A New Ranavirus Isolated from Pseudacris clarkii Tadpoles in Playa Wetlands in the Southern High Plains, Texas. J. Aquat. Anim. Health. 2010, 22, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Russell, D.M.; Goldberg, C.S.; Sprague, L.; Waits, L.P.; Green, D.E.; Schuler, K.L.; Rosenblum, E.B. Ranavirus Outbreaks in Amphibian Populations of Northern Idaho. Herpetol. Rev. 2011, 42, 223–225. [Google Scholar]
- Gray, M.J.; Miller, D.L.; Hoverman, J.T. First Report of Ranavirus Infecting Lungless Salamanders. Herpetol. Rev. 2009, 40, 316–319. [Google Scholar]
- Davidson, S.R.A.; Chambers, D.L. Ranavirus Prevalence in Amphibian Populations of Wise County, Virginia, USA. Herpetol. Rev. 2011, 42, 214–215. [Google Scholar]
- Gray, M.J.; Miller, D.L.; Schmutzer, A.C.; Baldwin, C.A. Frog Virus 3 Prevalence in Tadpole Populations at Cattle-Access and Non-Access Wetlands in Tennessee, U.S.A. Dis. Aquat. Organ. 2007, 77, 97–103. [Google Scholar] [CrossRef]
- Majji, S.; LaPatra, S.; Long, S.M.; Sample, R.; Bryan, L.; Sinning, A.; Chinchar, V.G. Rana catesbeiana Virus Z (RCV-Z): A Novel Pathogenic Ranavirus. Dis. Aquat. Organ. 2006, 73, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.L.; Rajeev, S.; Gray, M.J.; Baldwin, C. Frog Virus 3 Infection, Cultured American Bullfrogs. Emerg. Infect. Dis. 2007, 13, 342–343. [Google Scholar] [CrossRef]
- Miller, D.L.; Gray, M.J.; Rajeev, S.; Schmutzer, A.C.; Burton, E.C.; Merrill, A.; Baldwin, C. Pathological Findings in Larval and Juvenile Anurans Inhabiting Farm Ponds in Tennessee, U.S.A. J. Wildl. Dis. 2009, 45, 314–324. [Google Scholar] [CrossRef]
- Wolf, K.; Bullock, G.L.; Dunbar, C.E.; Quimby, M.C. Tadpole Edema Virus: A Viscerotropic Pathogen for Anuran Amphibians. J. Infect. Dis. 1969, 118, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.J.; Pessier, A.P.; Jacobson, E.R. Experimental Transmission and Induction of Ranaviral Disease in Western Ornate Box Turtles (Terrapene ornate ornate) and Red-eared Sliders (Trachemys scripta elegans). Vet. Path. 2007, 44, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Clark, H.F.; Brennan, J.C.; Zeigel, R.F.; Karzon, D.T. Isolation and Characterizatoin of Viruses from the Kidneys of Rana pipiens with Renal Adenocarcinoma before and after Passage in the Red Eft (Triturus viridescens). J. Virol. 1968, 2, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Granoff, A.; Came, P.E.; Rafferty, K.A. The Isolation and Properties of Viruses from Rana pipiens: Their Possible Relationship to the Renal Adenocarcinoma of the Leopard Frog. Ann. NY Acad. Sci. 1965, 126, 237–255. [Google Scholar] [CrossRef] [PubMed]
- Uyehara, I.K.; Gamble, T.; Cotner, S. The Presence of Ranavirus in Anuran Populations at Itasca State Park, Minnesota, USA. Herpetol. Rev. 2010, 41, 177–179. [Google Scholar]
- Harp, E.M.; Petranka, J.W. Ranavirus in Wood Frogs (Rana sylvatica): Potential Sources of Transmission within and between Ponds. J. Wildl. Dis. 2006, 42, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.D.; Green, E.; Fellers, G.; Chinchar, V.G. Molecular Characterization of Iridoviruses Isolated from Sympatric Amphibians and Fish. Virus Res. 1999, 63, 45–52. [Google Scholar] [CrossRef]
- Glenney, J.W.; Julian, J.T.; Quartz, W.M. Preliminary Amphibian Health Survey in the Delaware Water Gap National Recreation Area. J. Aquat. Anim. Health 2010, 22, 102–114. [Google Scholar] [CrossRef]
- Pessier, A.P. San Diego Zoo Global, San Diego, CA, USA. Unpublished work, 2011.
- Fox, S.F.; Greer, A.L.; Tores-Cervantes, R.; Collins, J.P. First Case of Ranavirus-Associated Morbidity and Mortality in Natural Populations of the South American Frog, Atelognathus patagonicus. Dis. Aquat. Organ. 2006, 72, 87–92. [Google Scholar] [CrossRef]
- Mazzoni, R.; José de Mesquita, A.; Fleury, L.F.F.; Diederichsen de Brito, W.M.E.; Nunes, I.A.; Robert, J.; Morales, H.; Coelho, A.S.G.; Barthasson, D.L.; Galli, L.; et al. Mass Mortality Associated with a Frog Virus 3-Like Ranavirus Infection in Farmed Tadpoles, Rana catesbeiana, from Brazil. Dis. Aquat. Organ. 2009, 86, 181–191. [Google Scholar] [CrossRef]
- Galli, L.; Pereira, A.; Márquez, A.; Mazzoni, R. Ranavirus Detection by PCR in Cultured Tadpoles (Rana catesbeiana Shaw, 1802) from South America. Aquaculture 2006, 257, 78–82. [Google Scholar] [CrossRef]
- Zupanovic, Z.; Lopz, G.; Hyatt, A.D.; Green, B.; Bartran, G.; Parkes, H.; Whittingtin, R.J.; Speare, R. Giant Toads, Bufo marinus, in Australia and Venezuela Have Antibodies against Ranaviruses. Dis. Aquat. Organ. 1998, 32, 1–8. [Google Scholar] [CrossRef]
- Pearman, P.B.; Garner, T.W.J.; Straub, M.; Greber, U.F. Response of the Italian Agile Frog (Rana latastei) to a Ranavirus, Frog Virus 3: A Model for Viral Emergence in Naïve Populations. J. Wildl. Dis. 2004, 40, 660–669. [Google Scholar] [CrossRef]
- Pearman, P.B.; Garner, T.W.J. Susceptibility of Italian Agile Frog Populations to an Emerging Strain of Ranavirus Parallels Population Genetic Diversity. Ecol. Lett. 2005, 8, 401–408. [Google Scholar] [CrossRef]
- Gantress, J.; Maniero, G.D.; Cohen, N.; Robert, J. Development and Characterization of a Model System to Study Amphibian Immune Responses to Iridoviruses. Virology 2003, 311, 254–262. [Google Scholar] [CrossRef]
- Ridenhour, B.J.; Storfer, A. Geographically Variable Selection in Ambystoma tigrinum Virus (Iridoviridae) throughout the Western United States. J. Evol. Biol. 2008, 21, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Gahl, M.J.; Calhoun, A.J.K. Landscape setting and Risk of Ranavirus Mortality Events. Biol. Cons. 2008, 141, 2679–2689. [Google Scholar] [CrossRef]
- Forson, D.; Storfer, A. Effects of Atrazine and Iridovirus Infection on Survival and Life History Characteristics in Long-Toed Salamanders, Ambystoma macrodactylum. Environ. Toxicol. Chem. 2006, 25, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Kerby, J.L.; Hart, J.A.; Storfer, A. Combined Effects of Virus, Pesticides, and Predator Cue on Larval Tiger Salamander (Ambystoma trigrinum). EcoHealth 2011. [Google Scholar] [CrossRef]
- Robert, J.; Morales, H.; Buck, W.; Cohen, N.; Marr, S.; Gantress, J. Adaptive Immunity and Histopathology in Frog Virus 3-Infected Xenopus. Virology 2005, 332, 667–675. [Google Scholar] [CrossRef]
- Duffus, A.L.J.; Cunningham, A.A. Major disease threats to European amphibians. Herpetol. J. 2010, 20, 117–127. [Google Scholar]
- Pasmans, F.; Blahak, S.; Martel, A.; Pantchev, N.; Zwart, P. Ranavirus-Associated Mass Mortality in Imported Red Tailed Knobby Newts (Tylototriton kweichowensis): A Case Report. Vet. J. 2008, 176, 257–259. [Google Scholar] [CrossRef]
- Green, D.E.; Gray, M.J.; Miller, D.L. Disease Monitoring and Biosecurity. In Amphibian Ecology and Conservation: A Handbook of Techniques; Dodd, C.K., Ed.; Oxford University Press: New York, NY, USA, 2010; Chapter 26; pp. 481–505. [Google Scholar]
- Rollins-Smith, L.A. Metamorphosis and the Amphibian Immune System. Immunol. Rev. 1998, 166, 221–230. [Google Scholar] [CrossRef]
- Rollins-Smith, L.A. Neuroendocrine-Immune System Interactions in Amphibians Implications for Understanding Global Amphibian Declines. Immunol. Res. 2001, 23, 273–280. [Google Scholar] [CrossRef]
- Robert, J. Emerging Ranaviral Infectious Diseases and Amphibian Decline. Diversity 2010, 2, 330. [Google Scholar] [CrossRef]
- Cullen, B.R.; Owens, L. Experimental Challenge and Clinical Cases of Bohle Iridovirus (BIV) in Native Australian Anurans. Dis. Aquat. Organ. 2002, 49, 83–92. [Google Scholar] [CrossRef]
- Warne, R.W.; Crespi, E.J.; Brunner, J.L. Escape from the Pond: Stress and Developmental Responses to Ranavirus Infection in Wood Frog Tadpoles. Funct. Ecol. 2011, 25, 139–146. [Google Scholar] [CrossRef]
- Tweedell, K.; Granoff, A. Viruses and Renal Carcinoma of Rana pipiens. V. Effect of Frog Virus 3 on Developing Frog Embryos and Larvae. J. Nat. Cancer Inst. 1968, 40, 407–410. [Google Scholar] [PubMed]
- Converse, K.A.; Green, D. Diseases of Salamanders. In Wildlife Diseases: Landscape Epidemiology, Spatial Distribution and Utilization of Remote Sensing Technology; Majumdar, S.K., Huffman, J.E., Brenner, F.J., Panah, A.I., Eds.; The Pennsylvania Academy of Science: Easton, PA, USA, 2005; Chapter 9; pp. 117–130. [Google Scholar]
- Brunner, J.L.; Richards, K.; Collins, J.P. Dose and Host Characteristics Influence Virulence of Ranavirus Infections. Oecologia 2005, 144, 399–406. [Google Scholar] [CrossRef]
- Robert, J.; George, E.; De Jesús Andino, F.; Chen, G. Waterborne Infectivity of the Ranavirus Frog Virus 3 in Xenopus laevis. Virology 2011, 2, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Burton, E.C.; Miller, D.L.; Styer, E.L.; Gray, M.J. Amphibian Ocular Malformation Associated with Frog Virus 3. Vet. J. 2008, 177, 442–444. [Google Scholar] [CrossRef]
- Miller, D. University of Tennessee, Knoxville, TN, USA. Unpublished work, 2011.
- Hsieh, C.Y. National Pingtung University of Science and Technology, Pingtung, Taiwan. Unpublished work, 2011.
- Cunningham, A.A.; Tems, C.A.; Russell, P.H. Immunohistochemical Demonstration of Ranavirus Antigen in the Tissues of Infected Frogs (Rana temporaria) with Systemic Haemorrhagic or Cutaneous Ulcerative Disease. J. Comp. Pathol. 2008, 138, 3–11. [Google Scholar] [CrossRef]
- Matos, A.P.A.; Silva Trabucho Caeiro, M.F.A.; Papp, T.; Cunha Almeida Matos, B.A.; Correia, A.C.L.; Marschang, R.E. New Viruses from Lacerta Monticola (Serra da Estrela, Portugal): Further Evidence for a New Group of Nucleo-Cytoplasmic Large Deoxyriboviruses. Microsc. Microanal. 2011, 17, 101–108. [Google Scholar] [CrossRef]
- Schmutzer, A.C. Influences of Cattle on Larval Amphibians and the Aquatic Environment in Cumberland Plateau Wetlands. Master Thesis, University of Tennessee, Knoxville, TN, USA, 2007; p. 216. [Google Scholar]
- Brunner, J.L.; Schock, D.M.; Davidson, E.W.; Collins, J.P. Intraspecific Reservoirs: Complex Life History and the Persistence of a Lethal Ranavirus. Ecology 2004, 85, 560–566. [Google Scholar] [CrossRef]
- Picco, A.M.; Brunner, J.L.; Collins, J.P. Susceptibility of the Endangered California Tiger Salamander, Ambystoma californiense, to Ranavirus Infection. J. Wildl. Dis. 2007, 43, 286–290. [Google Scholar] [CrossRef]
- Robert, J.; Abramowitz, L.; Gantress, J.; Morales, H.D. Xenopus laevis: A Possible Vector of Ranavirus Infection? J. Wildl. Dis. 2007, 43, 645–652. [Google Scholar] [CrossRef]
- Morales, H.D.; Abramowitz, L.; Gertz, J.; Sowa, J.; Vogel, A.; Robert, J. Innate Immune Responses and Permissiveness to Ranavirus Infection of Peritoneal Leukocytes in the Frog Xenopus leavis. J. Virol. 2010, 84, 4912–4922. [Google Scholar] [CrossRef] [PubMed]
- Chinchar, V.G. Ranaviruses (Family Iridoviridae): Emerging Cold-Blooded Killers. Arch. Virol. 2002, 147, 447–470. [Google Scholar] [CrossRef]
- Petranka, J.W.; Harp, E.M.; Holbrook, C.T.; Hamel, J.A. Long-Term Persistence of Amphibian Populations in a Restored Wetland Complex. Biol. Cons. 2007, 138, 371–380. [Google Scholar] [CrossRef]
- Anderson, R.M.; May, R.M. Population Biology of Infectious Disease. Nature 1979, 280, 361–461. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.J.S. An Introduction to Mathematical Biology; Prentice Hall: Upper Saddle River, NJ, USA, 2007; p. 368. [Google Scholar]
- McCallum, H.; Barlow, N.; Hone, J. How Should Pathogen Transmission Be Modeled? Trends Ecol. Evol. 2001, 16, 295–300. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Davidson, E.W.; Seiler, A.; Jacobs, B.L.; Collins, J.P. Transmission of the Ambystoma tigrinum Virus to Alternative Hosts. Dis. Aquat. Organ. 2001, 46, 159–163. [Google Scholar] [CrossRef]
- Moody, N.J.G.; Owens, L. Experimental Demonstration of the Pathogenicity of a Frog Virus, Bohle iridovirus, for a Fish Species, Barramundi Lates calcarifer. Dis. Aquat. Organ. 1994, 18, 95–102. [Google Scholar] [CrossRef]
- Ariel, E. Pathology and Serological Aspects of Bohle Iridovirus Infections in Six Selected Water-Associated Reptiles in North Queensland. Ph.D. Thesis, James Cook University, Queensland, Australia, 1997. [Google Scholar]
- Bang Jensen, B.B.; Ersboll, A.K.; Ariel, E. Susceptibility of Pike, Esox lucius, to a Panel of Ranavirus Isolates. Dis. Aquat. Organ. 2009, 83, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Gobbo, F.; Cappellozza, E.; Pastore, M.R.; Bovo, G. Susceptibility of Black Bullhead, Ameiurus melas, to a Panel of Ranavirus Isolates. Dis. Aquat. Organ. 2010, 90, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Jancovich, J.K.; Bremont, M.; Touchman, J.W.; Jacobs, B.L. Evidence for Multiple Recent Host Species Shifts among the Ranaviruses (Family Iridoviridae). J. Virol. 2010, 84, 2634–2647. [Google Scholar] [CrossRef] [PubMed]
- Picco, A.M.; Karam, A.P.; Collins, J.P. Pathogen Host Switching in Commercial Trade with Management Recommendations. EcoHealth 2010, 7, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Langdon, J.S. Experimental Transmission and Pathogenicity of Epizootic Haematopoietic Necrosis Virus (EHNV) in Redfin Perch, Perca fluviatilis L., and 11 Other Teleosts. J. Fish. Dis. 1989, 12, 295–310. [Google Scholar] [CrossRef]
- Nazir, J.; Spengler, M.; Marschang, R.E. Environmental Persistence of Amphibian and Reptilian Ranaviruses. Dis. Aquat. Organ. in press, 2012.
- Brunner, J.L.; Schock, D.M.; Collins, J.P. Transmission Dynamics of the Amphibian Ranavirus Ambystoma tigrinum Virus. Dis. Aquat. Organ. 2007, 77, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Greer, A.L.; Collins, J.P. Habitat Fragmentation as a Result of Biotic and Abiotic Factors Controls Pathogen Transmission Throughout a Host Population. J. Anim. Ecol. 2008, 77, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Wells, K.D. The Ecology and Behavior of Amphibians; University of Chicago Press: Chicago, IL, USA, 2007; p. 1148. [Google Scholar]
- Greer, A.L.; Briggs, C.I.; Collins, J.P. Testing a Key Assumption of Host-Pathogen Theory: Density and Disease Transmission. Oikos 2008, 117, 1667–1673. [Google Scholar] [CrossRef]
- Ryder, J.J.; Miller, M.R.; White, A.; Knell, R.J.; Boots, M. Host-Parasite Population Dynamics Under Combined Frequency- and Density-Dependent Transmission. Oikos 2007, 116, 2017–2026. [Google Scholar] [CrossRef]
- Storfer, A.; Alfaro, M.E.; Ridenhour, B.J.; Jancovich, J.K.; Mech, S.G.; Parris, M.J.; Collins, J.P. Phylogenetic Concordance Analysis Shows an Emerging Pathogen is Novel and Endemic. Ecol. Lett. 2007, 10, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Teacher, A.G.F.; Garner, T.W.J.; Nichols, R.A. Evidence for Directional Selection at a Novel Major Histocompatibility Class I Marker in Wild Common Frogs (Rana temporaria) Exposed to a Viral Pathogen (Ranavirus). PLoS One 2009, 4, e4616. [Google Scholar] [CrossRef]
- Pfennig, D.W.; Loeb, M.L.G.; Collins, J.P. Pathogens as a Factor Limiting the Spread of Cannibalism in Tiger Salamanders. Oecologia 1991, 99, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.P.; Zerba, K.E.; Sredl, M.J. Shaping Intraspecific Variation: Development, Ecology and the Evolution of Morphology and Life History Variation in Tiger Salamanders. Genetica 1993, 89, 167–183. [Google Scholar] [CrossRef]
- Bolker, B.M.; Castro, F.; Storfer, A.; Mech, S.; Harvey, E.; Collins, J.P. Disease as a Selective Force Precluding Widespread Cannibalism: A Case Study of an Iridovirus of Tiger Salamanders, Ambystoma tigrinum. Evol. Ecol. 2008, 14, 213–224. [Google Scholar]
- Parris, M.J.; Storfer, A.; Collins, J.P.; Davidson, E.W. Life History Responses to Pathogens in Tiger Salamander (Ambystoma tigrinum) Larvae. J. Herpetol. 2005, 39, 366–372. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Rannala, B.; Yang, Z. Statistical Tests of Host-Parasite Conspeciation. Evolution 1997, 51, 410–419. [Google Scholar] [CrossRef]
- Hunter, W.; Sinisterra, X.H.; McKenxie, C.L.; Shatters, R.G. Iridovirus Infection and Vertical Transmission in Citrus Aphids. Proc. Florida State Hort. Soc. 2001, 114, 70–72. [Google Scholar]
- Drennan, J.D.; LaPatra, S.E.; Siple, J.T.; Ireland, S.; Cain, K.D. Transmission of White Sturgeon Iridovirus in Kootenai River White Sturgeon, Acipenser transmontanus. Dis. Aquat. Organ. 2006, 70, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Gruia-Gray, J.; Desser, S.S. Cytopathological Observations and Epizootiology of Frog Erythrocytic Virus in Bullfrogs (Rana catesbeiana). J. Wildl. Dis. 1992, 28, 34–41. [Google Scholar] [CrossRef]
- Burton, E.C.; Gray, M.J.; Schmutzer, A.C.; Miller, D.L. Differential Responses of Postmetamorphic Amphibians to Cattle Grazing in Wetlands. J. Wildl. Manage. 2009, 73, 269–277. [Google Scholar] [CrossRef]
- Carey, C.D.; Cohen, N.; Rollins-Smith, L. Amphibian Declines: An Immunological Perspective. Dev. Comp. Immunol. 1999, 23, 459–472. [Google Scholar] [CrossRef] [PubMed]
- Blaustein, A.R.; Romansic, J.M.; Kiesecker, J.M.; Hatch, A.C. Ultraviolet Radiation, Toxic Chemicals, and Amphibian Population Declines. Diversity and Distribution 2003, 9, 123–140. [Google Scholar] [CrossRef]
- Forson, D.D.; Storfer, A. Atrazine Increases Ranavirus Susceptibility in the Tiger Salamander, Ambystoma tigrinum. Ecol. Appl. 2010, 16, 2325–2332. [Google Scholar] [CrossRef]
- Kerby, J.L.; Richards-Hrdlicka, K.L.; Storfer, A.; Skelly, D.K. An Examination of Amphibian Sensitivity to Environmental Contaminants: Are Amphibians Poor Canaries? Ecol. Lett. 2010, 13, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Ewald, P.W.; Mims, C.A.; Lachmann, P.J.; Hughes, A.L.; Gillett, J.D.; Parker, C.E. Evolution of Mutation Rate and Virulence among Human Retroviruses. Philos. Trans. R. Soc. London (Biol.) 1994, 346, 333–343. [Google Scholar]
- Schloegel, L.M.; Picco, A.M.; Kilpatrick, A.M.; Davies, A.J.; Hyatt, A.D.; Daszak, P. Magnitude of the US Trade in Amphibians and Presence of Batrachochytrium dendrobatidis and Ranavirus Infection in Imported North American Bullfrogs (Rana catesbeiana). Biol. Cons. 2009, 142, 1420–1426. [Google Scholar] [CrossRef]
- Schloegel, L.M.; Daszak, P.; Cunningham, A.A.; Speare, R.; Hill, B. Two Amphibian Diseases, Chytridiomycosis and Ranaviral Disease, are now Globally Notifiable to the World Organization for Animal Health (OIE): An Assessment. Dis. Aquat. Organ. 2010, 92, 101–108. [Google Scholar] [CrossRef]
- Mao, J.; Tham, T.N.; Gentry, G.A.; Aubertin, A.; Chinchar, V.G. Cloning, Sequence Analysis, and Expression of the Major Capsid Protein of the Iridovirus Frog Virus 3. Virology 1996, 216, 431–436. [Google Scholar] [CrossRef]
- Manual of Diagnostic Tests for Aquatic Animals 2011. Available online: http://www.oie.int/international-standard-setting/aquatic-manual/access-online/ (accessed on 31 October 2011).
- Aquatic Animal Health Code (2011). Available online: http://www.oie.int/international-standard-setting/aquatic-code/access-online/ (accessed on 31 October 2011).
- Bryan, L.K.; Baldwin, C.A.; Gray, M.J.; Miller, D.L. Efficacy of Select Disinfectants at Inactivating Ranavirus. Dis. Aquat. Organ. 2009, 84, 89–94. [Google Scholar] [CrossRef]
- Rojas, S.; Richards, K.; Jancovich, J.K.; Davidson, E.W. Influence of Temperature on Ranavirus Infection in Larval Salamanders, Ambystoma tigrinum. Dis. Aquat. Organ. 2005, 63, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, D.; Maeno, Y.; Honda, A.; Yokoyama, K.; Tooriyama, T.; Manabe, S. 1999. Effectiveness of a Vaccine Against Red Sea Bream Iridoviral Disease in a Field Trial Test. Dis. Aquat. Organ. 1999, 36, 73–75. [Google Scholar] [CrossRef]
- Caipang, C.M.; Takano, T.; Hirono, I.; Aoki, T. Genetic Vaccines Protect Red Seabream, Pagrus major, Upon Challenge with Red Sea Bream Iridovirus (RSIV). Fish Shellfish Immunol. 2006, 21, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Tamaru, Y.; Ohtsuka, M.; Kato, K.; Manabe, S.; Kuroda, K.; Sanada, M.; Ueda, M. Application of the Arming System for the Expression of the 380R Antigen from Red Sea Bream Iridovirus (RSIV) on the Surface of Yeast Cells: A First Step for the Development of an Oral Vaccine. Biotechnol. Prog. 2006, 22, 949–953. [Google Scholar] [CrossRef]
- Kerby, J. 1st International Symposium on Ranaviruses. Froglog 2011, 98, 35–37. [Google Scholar]
- Lesbarreres, D.; Balseiro, A.; Brunner, J.; Chinchar, V.; Duffus, A.; Kerby, J.; Miller, D.L.; Robert, J.; Schock, D.; Waltzek, T.; Gray, M. Ranavirus: Past, present and future. Biol. Lett. 2011. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.; Chinchar, V.G. Ranaviruses: An emerging threat to ectothermic vertebrates. Report of the First International Symposium on Ranaviruses, Minneapolis MN, USA, 8 July 2011. Dev. Comp. Immunol. 2011, 36, 259–261. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Mao, J.; Chinchar, V.G.; Wyatt, C.; Case, S.T.; Kumar, S.; Valente, G.; Subramanian, S.; Davidson, E.W.; Collins, J.P.; Jacobs, B.L. Genomic Sequence of a Ranavirus (Family Iridoviridae) Associated with Salamander Mortalities in North America. Virology 2003, 316, 90–103. [Google Scholar] [CrossRef]
- Williams, T.; Barbosa-Solomieu, V.; Chinchar, G.D. A Decade of Advances in Iridovirus Research. Adv. Virus Res. 2005, 65, 173–248. [Google Scholar] [PubMed]
- Sample, R.; Bryan, L.; Long, S.; Majii, S.; Hoskins, G.; Olivier, J.; Chinchar, V.G. Inhibition of Iridovirus Protein Synthesis and Virus Replication by Antisense Morpholino Oligonucleotides Targeted to the Major Capsid Protein, the 18 kDa Immediate-Early Protein, and a Viral Homolog of RNA Polymerase II. Virology 2007, 358, 311–320. [Google Scholar]
- Whitley, D.S.; Sample, R.C.; Sinning, A.R.; Henegar, J.; Chinchar, V.G. Antisense approaches for elucidating ranavirus gene function in an infected fish cell line. Dev. Comp. Immunol. 2010, 35, 937–948. [Google Scholar] [CrossRef]
- Chen, G.; Ward, B.M.; Yu, K.H.; Chinchar, V.G.; Robert, J. Improved Knockout Methodology Reveals That Frog Virus 3 Mutants Lacking either the 18K Immediate-Early Gene or the Truncated vIF-2{alpha} Gene Are Defective for Replication and Growth In Vivo. J. Virol. 2011, 85, 11131–11138. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Jacobs, B.L. Innate Immune Evasion Mediated by the Ambystoma tigrinum Virus Eukaryotic Translation Initiation Factor 2{alpha} Homologue. J. Virol. 2011, 85, 5061–5069. [Google Scholar] [CrossRef] [PubMed]
- Whittington, R.J.; Becker, J.A.; Dennis, M.M. Iridovirus Infections in Finfish—Critical Review with Emphasis on Ranaviruses. J. Fish. Dis. 2010, 33, 95–122. [Google Scholar] [CrossRef] [PubMed]
- Holopainen, R.; Honkanenb, J.; Bang-Jensen, B.; Ariel, E.; Tapiovaaraa, H. Quantitation of Ranaviruses in Cell Culture and Tissue Samples. J. Virol. Methods 2011, 171, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Global Ranavirus Consortium. Available online: http://fwf.ag.utk.edu/mgray/ranavirus/ranavirus.htm (accessed on 31 October 2011).
| Continent | Nation | Family | Scientific Name | I,M1 | W,C2 | References |
|---|---|---|---|---|---|---|
| Asia | China | Cryptobranchidae | Andrias davidianus | M | C | [11] |
| Ranidae | Rana dybowskii | I | W | [12] | ||
| Ranidae | Rana grylio | M | C | [13,14] | ||
| Ranidae | Rana tigrina | M | C | [15] | ||
| Japan | Hynobiidae | Hynobius nebulosus | M | C | [16] | |
| Ranidae | Lithobates catesbeianus | M | W | [17] | ||
| Thailand | Ranidae | Rana tigrina | M | C | [18] | |
| Australia | Australia | Myobatrachidae | Limnodynastes ornatus | M | C | [19] |
| Europe | Belgium | Salamandridae | Tylototriton kweichowensis | M | C | [20] |
| Croatia | Ranidae | Pelophylax esculenta | M | W | [21] | |
| Denmark | Ranidae | Pelophylax esculenta | M | W | [22] | |
| Israel | Bufonidae | Pseudepidalea viridis | I | W | [23] | |
| Netherlands | Ranidae | Pelophylax spp. | M | W | [24] | |
| Salamandridae | Lissotriton vulgaris | M | W | [24] | ||
| Portugal | Salamandridae | Triturus marmoratus | M | W | [25] | |
| Salamandridae | Triturus boscai | M | W | [25] | ||
| Spain | Alytidae | Alytes obstetricans | M | W | [26] | |
| Salamandridae | Ichthyosaura alpestris | M | W | [27] | ||
| Switzerland | Ranidae | Pelophylax esculenta | M | C | [28] | |
| Pelophylax ridibundus | M | C | [28] | |||
| UK | Alytidae | Alytes obstetricans | M | W | [29] | |
| Bufonidae | Bufo bufo | M | W | [29,30] | ||
| Ranidae | Rana temporaria | M | W | [2,6,29,31,32] | ||
| Salamandridae | Lissotriton vulgaris | I | W | [29] | ||
| North America | Canada | Ambystomatidae | Ambystoma mavortium | M | W | [33,34,35] |
| Ambystoma spp. | I | W | [36] | |||
| Hylidae | Hyla versicolor | I | W | [36] | ||
| Pseudacris crucifer | M | W | [37] | |||
| Pseudacris spp. | I | W | [36] | |||
| Ranidae | Lithobates clamitans | I, M | W | [37,38,39] | ||
| Lithobates pipiens | I, M | W, C | [4,39,40,41,42] | |||
| Lithobates sylvaticus | I, M | W | [7,36,37,39,40,43] | |||
| Salamandridae | Notophthalmus viridescens | I | W | [36] | ||
| Costa Rica | Bufonidae | Bufo marinus | I | C | [44] | |
| USA | Ambystomatidae | Ambystoma jeffersonianum | M | W | [45] | |
| Ambystoma macrodactylum | M | W | [45] | |||
| Ambystoma maculatum | I, M | W | [3,46,47,48,49] | |||
| Ambystoma mavortium | I, M | W | [50,51,52,53,54] | |||
| Ambystoma opacum | M | W | [49] | |||
| Ambystoma tigrium | I, M | W, C | [3,55] | |||
| Bufonidae | Anaxyrus americanus | I | W | [55] | ||
| Anaxyrus boreas boreas | I, M | W, C | [56,57] | |||
| Cryptobranchidae | Cryptobranchus alleganiensis alleganiensis | I | W | [58] | ||
| Dendrobatidae | Dendrobates auratus | I | C | [59] | ||
| Phyllobates terribilis | I | C | [59] | |||
| Hylidae | Acris crepitans | I | W | [55] | ||
| Hyla chrysoscelis | I, M | W, C | [45,60] | |||
| Hyla cinerea | M | W | [61] | |||
| Pseudacris clarkii | M | W | [62] | |||
| Pseudacris crucifer | M | W | [3,47,49] | |||
| Pseudacris feriarum | I, M | W | [49,55] | |||
| Pseudacris regilla | M | W | [45] | |||
| Pseudacris sierra | M | W | [63] | |||
| Plethodontidae | Desmognathus conanti | I | W | [64] | ||
| Desmognathus fuscus | I | W | [65] | |||
| Desmognathus imitator | I | W | [64] | |||
| Desmognathus monticola | I | W | [65,64] | |||
| Desmognathus ocoee | I | W | [64] | |||
| Desmognathus quadramaculatus | I | W | [65,64] | |||
| Desmognathus santeetlah | I | W | [64] | |||
| Desmognathus wrighti | I | W | [64] | |||
| Eurycea cirrigera | I | W | [65] | |||
| Eurycea longicauda | I | W | [65] | |||
| Eurycea lucifuga | I | W | [65] | |||
| Eurycea wilderae | I | W | [64] | |||
| Gyrinophilus porphyriticus | I | W | [64] | |||
| Plethodon glutinosus complex | I | W | [65] | |||
| Plethodon jordani | I | W | [64] | |||
| Ranidae | Lithobates blairi | M | W | [45] | ||
| Lithobates catesbeianus | I, M | W, C | [3,47,65,66,67,68,69,70] | |||
| Lithobates clamitans | I, M | W | [3,47,66,69,71] | |||
| Lithobates palustris | I, M | W | [3,55,65] | |||
| Lithobates pipiens | I, M | W | [3,72,73,74] | |||
| Lithobates septentrionalis | M | W | [3] | |||
| Lithobates sphenocephalus | I, M | W | [45,55,71] | |||
| Lithobates sylvaticus | I, M | W | [3,46,47,48,49,74,75] | |||
| Pyxicephalus adspersus | M | C | [59] | |||
| Rana aurora | M | W | [76] | |||
| Rana draytonii | M | W | [45] | |||
| Rana heckscheri | M | W | [45] | |||
| Rana luteiventris | M | W | [56,61,63] | |||
| Rana muscosa | M | W | [56] | |||
| Rhacophoridae | Rhacophorus dennysi | M | C | [59] | ||
| Salamandridae | Notophthalmus viridescens | I, M | W | [3,73,77] | ||
| Scaphiopodidae | Scaphiopus holbrookii | I, M | W | [78] | ||
| South America | Argentina | Leptodactylidae | Atelognathus patagonicus | M | W | [79] |
| Brazil | Ranidae | Lithobates catesbeianus | M | C | [80] | |
| Uruguay | Ranidae | Lithobates catesbeianus | I | C | [81] | |
| Venezuela | Bufonidae | Bufo marinus | I | W | [82] | |
| Leptodactylidae | Leptodactylus sp. | I | W | [82] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Miller, D.; Gray, M.; Storfer, A. Ecopathology of Ranaviruses Infecting Amphibians. Viruses 2011, 3, 2351-2373. https://doi.org/10.3390/v3112351
Miller D, Gray M, Storfer A. Ecopathology of Ranaviruses Infecting Amphibians. Viruses. 2011; 3(11):2351-2373. https://doi.org/10.3390/v3112351
Chicago/Turabian StyleMiller, Debra, Matthew Gray, and Andrew Storfer. 2011. "Ecopathology of Ranaviruses Infecting Amphibians" Viruses 3, no. 11: 2351-2373. https://doi.org/10.3390/v3112351
APA StyleMiller, D., Gray, M., & Storfer, A. (2011). Ecopathology of Ranaviruses Infecting Amphibians. Viruses, 3(11), 2351-2373. https://doi.org/10.3390/v3112351




