Herpesviruses that Infect Fish
Abstract
1. Introduction
2. Molecular Characteristics of Fish Herpesviruses
| Virus name (abbreviation) | Family, Clade 1 (Genus) | Common name (abbreviation) | Host(s) | Disease | Cell line 3- CPE, temperature | Ref. |
|---|---|---|---|---|---|---|
| Anguillid HV 1 (AngHV1) | Alloherpesviridae, 1 | HV anguillae (HVA) | Japanese eel Anguilla japonica and European eel A. Anguilla | Hemorrhages of skin, fins, gills, liver | EK, EO-1, EP-1, BF-2, FHM, RTG-2, 20–25 °C syncytia and rounded cells | [4,11] |
| Cyprinid HV 1 (CyHV1) | Alloherpesviridae, 1 (Cyprinivirus) | HV cyprini, carp pox HV, carp HV(CHV) | Common carp Cyprinus carpio | High losses in fry- exopthalmia hemorrhages, survivors have papillomas | KF-1, EPC, FHM 15–20 °C. Cells rounded and vacuolated | [12,13] |
| Cyprinid HV 2 (CyHV2) | Alloherpesviridae, 1 (Cyprinivirus) | Goldfish hematopoietic necrosis virus (GFHNV) | Goldfish Carassius auratus | High mortality all ages. Necrosis of hematopoietic tissue, spleen, pancreas, intestine | GF-1 (EPC, FHM)-unreliable, characterized by PCR and sequencing | [14,15,16] |
| Cyprinid HV 3 (CyHV3) | Alloherpesviridae, 1 (Cyprinivirus) | Koi HV (KHV), carp nephritis and gill necrosis virus (CNGV) | Common carp | gill inflammation, hyperplasia, and necrosis, hematopoietic tissue necrosis, high mortality,18–26 °C, all ages | KF-1, CCB, CFC, Au, Tol/FL Vacuolation after 4 days at 20 °C. | [17,18] |
| Ictalurid HV 1 (IcHV1) | Alloherpesviridae, 2 (Ictalurivirus) | channel catfish virus (CCV), Channel catfish herpesvirus | Channel catfish Ictalurus punctatus | Kidney, liver and intestinal necrosis, hemorrhages, high mortality in young fish at above 27 °C | CCO, BB 30 °C, syncytia | [19,20] |
| Ictalurid HV 2 (IcHV2) | Alloherpesviridae, 2 (Ictalurivirus) | Ictalurus melas HV (IcmHV) | Black bullhead Ameiurus melas | Kidney necrosis, hemorrhages, high mortality all ages | CCO, BF-2 | [21,22] |
| Acipenserid HV 1 (AciHV1) | Alloherpesviridae, 2 | White sturgeon HV 1 | White sturgeon Acipenser transmontanus | diffuse dermatitis, high losses in juveniles | WSSK-1 syncytia 15 °C | [23] |
| Acipenserid HV 2 (AciHV2) | Alloherpesviridae, 2 (Ictalurivirus) | White sturgeon HV 2 | White sturgeon | Epithelial hyperplasia | WSSK-1, WSS-2 rounded vacuolated cells 15 °C | [24] |
| Salmonid HV 1(SalHV1) | Alloherpesviridae, 2 (Salmonivirus) | HV salmonis (HPV) Steelhead herpesvirus (SHV) | Rainbow trout Oncorhynchus mykiss | Mild disease low losses at 10 °C. Adults- Virus shedding in ovarian fluid. No signs of disease. | RTG-2, CHSE 214, 10–15 °C extensive syncytia | [25] |
| Salmonid HV 2(SalHV2) | Alloherpesviridae, 2 (Salmonivirus) | Oncorhynchus masou virus (OMV) | Cherry salmon O. masou, coho salmon O. kisutch, sockeye salmon O. nerka, coho salmon O. keta, rainbow trout, | Viremia, external hemorrhages expthalmia, hepatic necrosis with high losses in young. Survivors- oral papillomas, virus shed in ovaran fluid | RTG, CHSE 214,15 °C syncytia | [26,27] |
| Salmonid HV 3 (SalHV3) | Alloherpesviridae, 2 (Salmonivirus) | Epizootic epitheliotropic disease virus (EEDV) | Lake trout Salvelinus namaycush, lake trout × brook trout S. fontinalis hybrids | Epithelial hyperplasia, hypertrophy, hemorrhages on eye and jaw. High losses in juveniles at 6–15 °C | EM, PCR and sequencing | [28,29] |
| Gadid herpesvirus 1 (GaHV1) | Alloherpesvirdae, 2 | Atlantic cod herpesvirus (ACHV) | Atlantic cod Gadus morhua | Hypertophy of cells in gills. High losses in adults. | EM, PCR and sequencing | [30] |
| Ranid HV 1 (RaHV1) | Alloherpesviridae, 2 2 (Batrachovirus) | Lucké tumor HV (LTHV) | Leopard frog Rana pipiens | Renal adenocarcinoma | EM, tumor explant culture | [31] |
| Ranid HV 2 (RaHV2) | Alloherpesviridae, 2 2 (Batrachovirus) | Frog virus 4 (FV-4) | Leopard frog | No known disease | ICR-2A | [32,33] |
| Pilchard HV | Alloherpesviridae, 2 | Australian pilchard Sardinops sagax | Acute losses with gill inflammation, epithelial hyperplasia and hypertrophy | EM, PCR and sequencing | [34,35,36,37] | |
| tilapia HV | Possible Herpesviridae | Tilapia larvae encephalitis virus (TLEV) | Blue tilapia (Oreochromis aureus) | Encephalitis and high loses in larvae | EM, PCR and Sequencing | [38] |
| Percid HV 1 (PeHV1) | HV vitreum, walleye HV | Walleye Stizostedion vitreum | diffuse epidermal hyperplasia | WO, WC-1, We-2. syncytia, 4–15 °C. | [39] |
| Virus name (abbreviation), Common name (abbreviation) | Host(s) | Disease | Ref. |
|---|---|---|---|
| Esocid HV 1 (EsHV1), Pike epidermal proliferative HV, pike HV | Northern pike Esox lucius and muskellunge E. masquinongy | blue spot disease-Flat, granular, bluish-white skin lesions caused by enlarged epidermal cells | [42] |
| Pleuronectid HV 1 (PlHV1), HV scopthalami | Turbot Scopthalamus maximus | Giant cells (polykaryocytes), greatly enlarged cells in skin and gill epithelium | [43,44] |
| Flounder HV (FHV) | Japanese flounder Paralichthys olivaceous | Epidermal hyperplasia, epidermal cells with virus particles, high losses of fry | 34 |
| Golden ide HV | Golden ide Leuciscus ide | Epidermal hyperplasia, papillomas- referred to as carp pox | [45] |
| Pacific cod HV | Pacific cod Gadus macrocephalus | Hypertrophy of epidermal cells | [46,47] |
| Sheatfish HV (SHV) | Wels catfish, Silurus glanis | Epidermal hyperplasia, papillomas | [48] |
| European Smelt HV, Smelt papillomatous virus, HV of Osmerus eperlanus | European smelt Osmerus eperlanus | Papillomas and Hyperplastic skin lesions on dorsal fin- virions are comet shaped | [49,50] |
| Rainbow smelt HV | Rainbow smelt Osmerus mordax | Papillomas and squamous cell carcinomas | [51] |
| Smooth dogfish HV | Smooth dogfish Mustelus canis | Epidermal depigmented lesions- epidermal cell necrosis with virus particles | [52] |
| Atlantic salmon HV | Atlantic salmon Salmo salar | Papillomas especially on smolts | [53] |
| Angelfish HV | Angelfish Pterophyllum altum | Skin hemorrhages, swollen spleen and liver, virus seen in splenic macrophages | [54] |
| Red striped rockfish HV | Red striped rockfish Sebastes proriger | Hepatomegally, Giant cells (polykaryocytes), hemorrhage, necrosis and inflammation in liver | [55] |
2.1. Gene Sequence Conservation
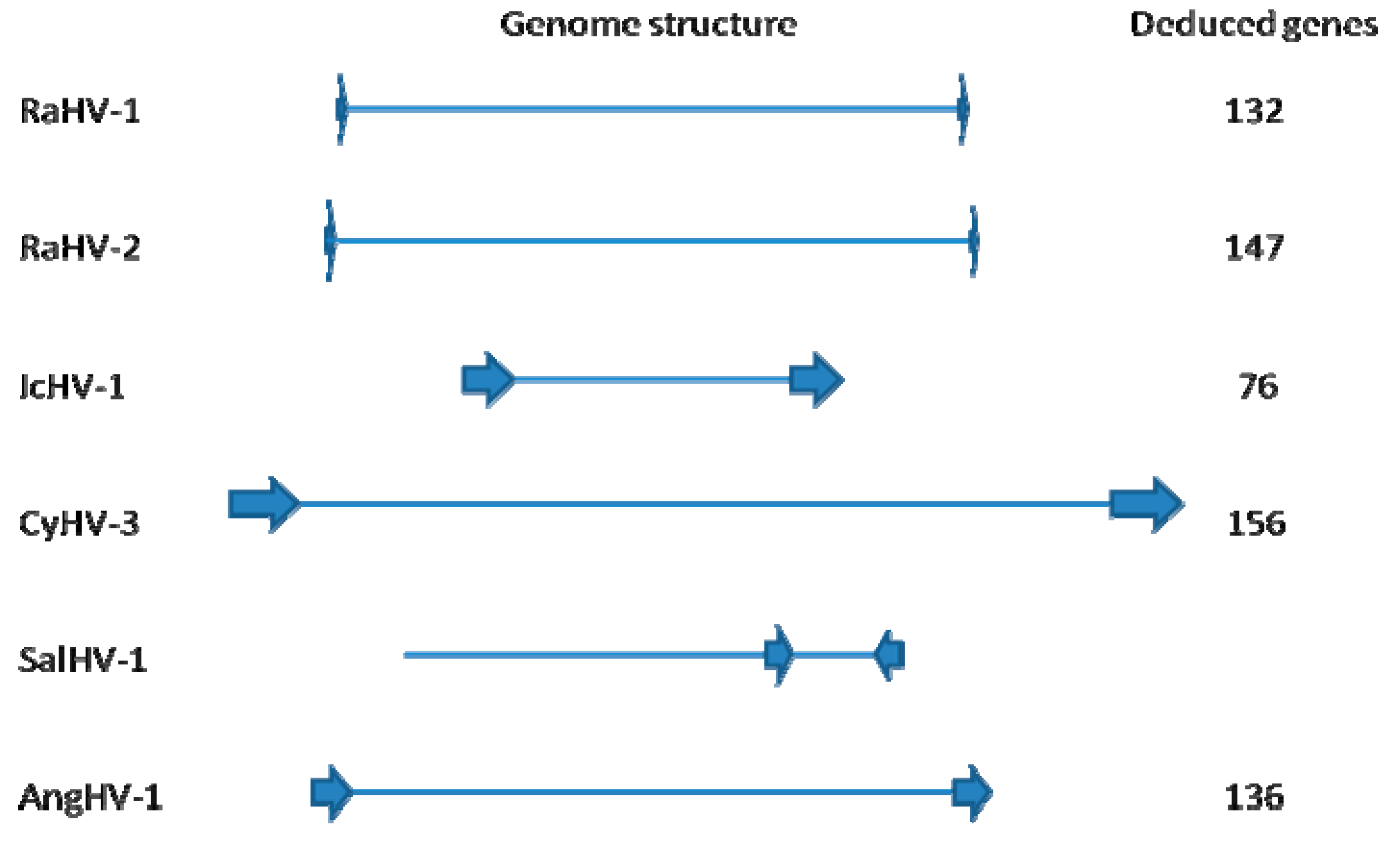
2.2. Genome Structure
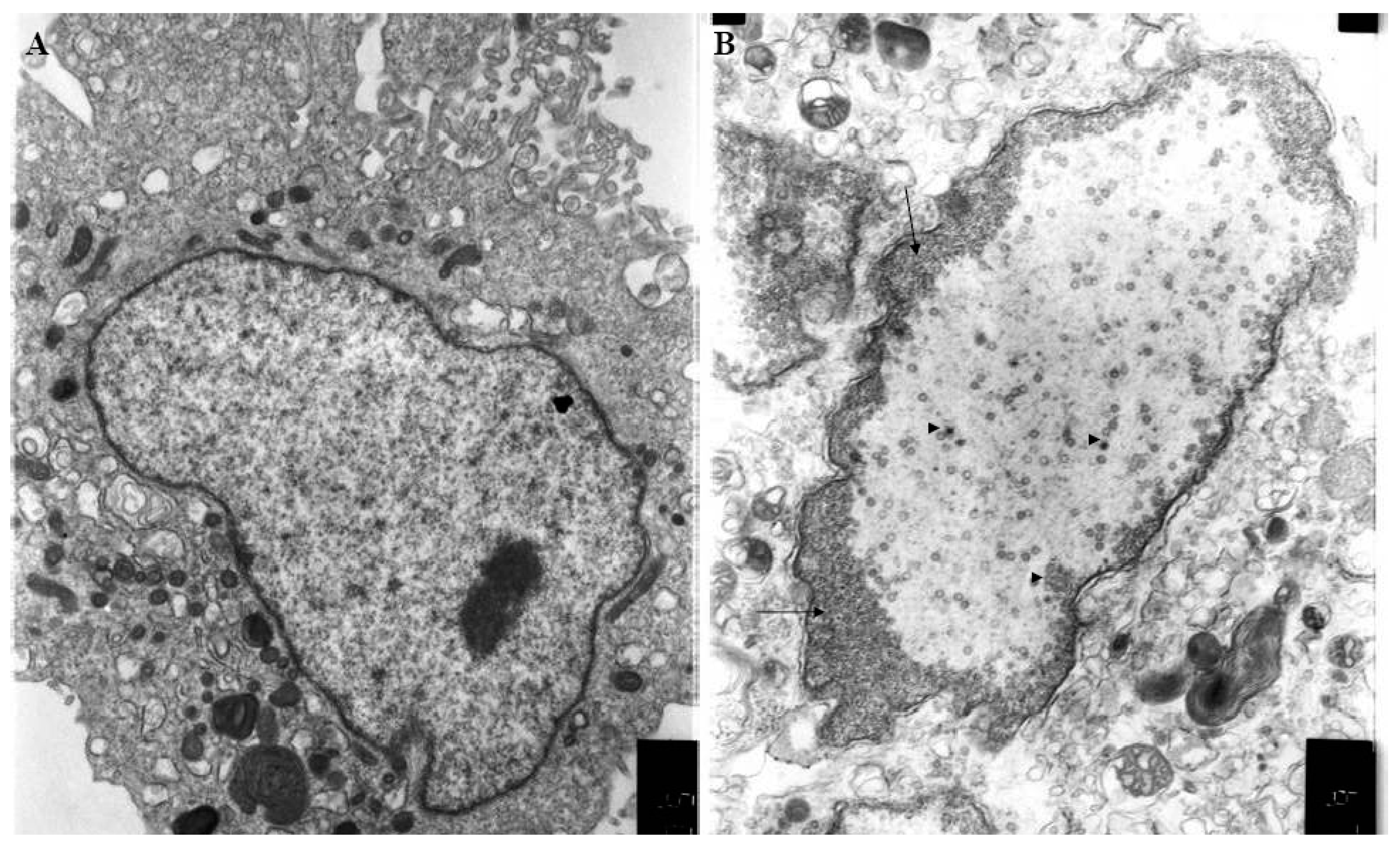
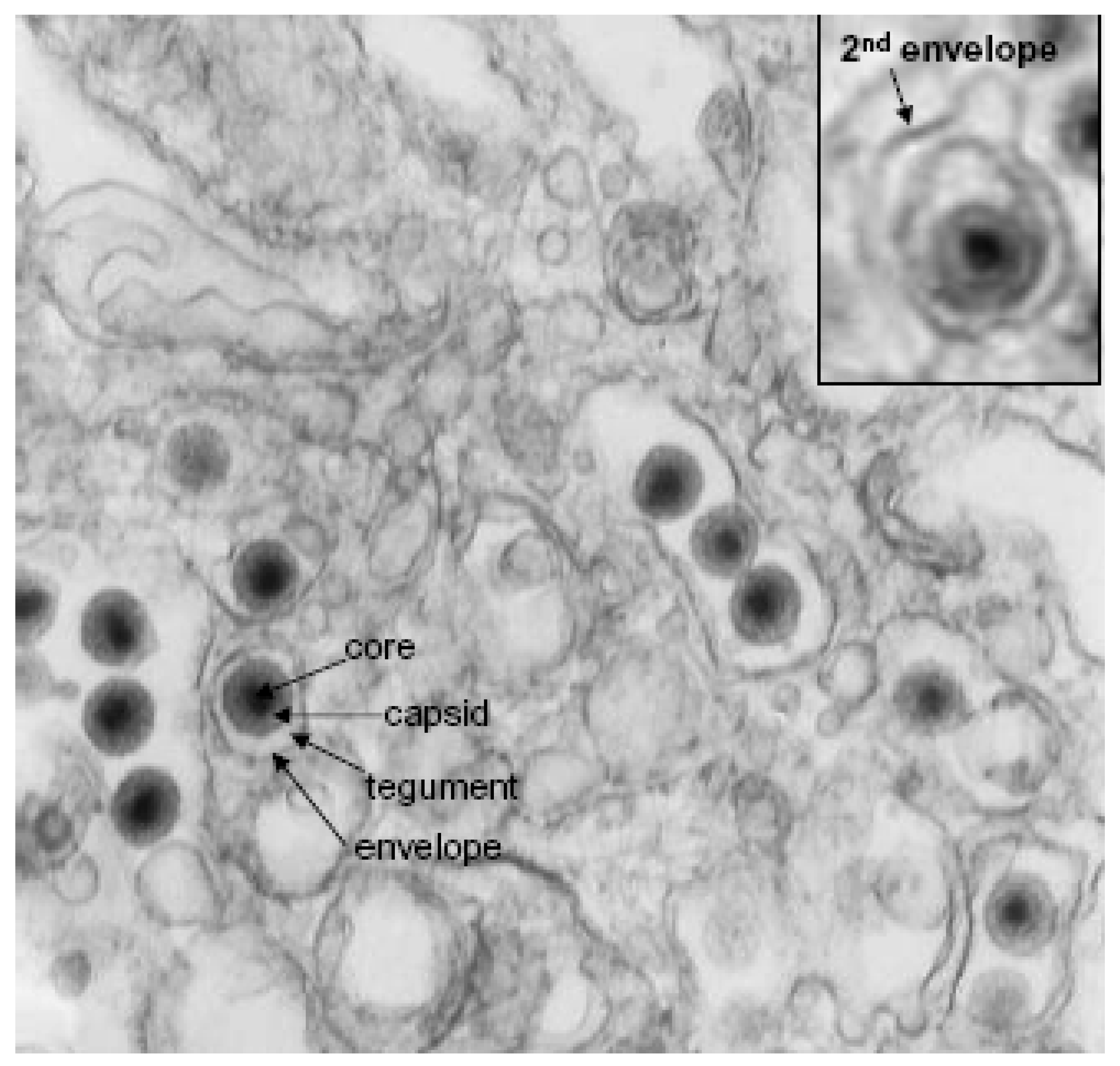
2.3. Virion Structure
3. Biological Characteristics of Fish Herpesviruses
3.1. Alloherpesviruses Display a High Level of Host Specificity
3.2. Alloherpesviruses Are Epitheliotrophic
3.3. Alloherpesviruses Establish Latent Infections
4. Cyprinid Herpesvirus 3 — A Model Clade 1 Alloherpesvirus
4.1. CyHV3 — “Disease Overview”
| Gene/ORF | Function | Class | Location |
|---|---|---|---|
| ORF 52 | Unknown | E | Unique large |
| ORF 105 | Unknown | E | Unique large |
| ORF 55 | TK gene | E | Unique large |
| ORF 139 | B22RH gene | E | Unique large |
| ORF1 | hypothetical protein | IE | Terminal repeat |
| ORF 3L | TF gene | IE | Terminal repeat |
| ORF6L | hypothetical protein | IE | Terminal repeat |
| ORF134 | IL-10 like gene | E | Unique large |
| ORF 4L | TNFR-1 like gene | E | Unique large |
| ORF 141* | RNR gene | E | Unique large |
| ORF 16 ** | GPCR gene | E | Unique large |
4.2. Molecular Characteristics of CyHV3
4.3. CyHV3 Disease Management
4.4. Latency
5. Ictalurid Herpesvirus 1 — A Model Clade 2 Herpesvirus
5.1. IcHV3 — Disease Overview
5.2. Molecular Characteristics of IcHV1
5.3. Molecular Analyses of Host Pathogen Interaction
5.4. Latency and Vertical Transmission
5.5. CCVD Management
6. Conclusions
Acknowledgments
Conflicts of Interest
References and Notes
- Plumb, J.A.; Hanson, L.A. Health Maintenance and Principal Microbial Diseases of Cultured Fishes, 3rd ed.; Wiley-Blackwell Publishing: Ames, IA, USA, 2011; p. 400. [Google Scholar]
- Davison, A.J. Channel catfish virus: A new type of herpesvirus. Virology 1992, 186, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Aoki, T.; Hirono, I.; Kurokawa, K.; Fukuda, H.; Nahary, R.; Eldar, A.; Davison, A.J.; Waltzek, T.B.; Bercovier, H.; Hedrick, R.P. Genome sequences of three koi herpesvirus isolates representing the expanding distribution of an emerging disease threatening koi and common carp worldwide. J. Virol. 2007, 81, 5058–5065. [Google Scholar] [CrossRef] [PubMed]
- van Beurden, S.J.; Bossers, A.; Voorbergen-Laarman, M.H.; Haenen, O.L.; Peters, S.; Abma-Henkens, M.H.; Peeters, B.P.; Rottier, P.J.; Engelsma, M.Y. Complete genome sequence and taxonomic position of anguillid herpesvirus 1. J. Gen. Virol. 2010, 91, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J.; Cunningham, C.; Sauerbier, W.; McKinnell, R.G. Genome sequences of two frog herpesviruses. J. Gen. Virol. 2006, 87, 3509–3514. [Google Scholar] [CrossRef]
- Davison, A.J.; Trus, B.L.; Cheng, N.; Steven, A.C.; Watson, M.S.; Cunningham, C.; Le Deuff, R.M.; Renault, T. A novel class of herpesvirus with bivalve hosts. J. Gen. Virol. 2005, 86, 41–53. [Google Scholar] [CrossRef]
- Davison, A.; Eberle, R.; Ehlers, B.; Hayward, G.; McGeoch, D.; Minson, A.; Pellett, P.; Roizman, B.; Studdert, M.; Thiry, E. The order herpesvirales. Arch. Virol. 2009, 154, 171–177. [Google Scholar] [CrossRef]
- McGeoch, D.J.; Rixon, F.J.; Davison, A.J. Topics in herpesvirus genomics and evolution. Virus Res. 2006, 117, 90–104. [Google Scholar] [CrossRef]
- Wu, G.A.; Jun, S.R.; Sims, G.E.; Kim, S.H. Whole-proteome phylogeny of large dsdna virus families by an alignment-free method. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 12826–12831. [Google Scholar] [CrossRef]
- Waltzek, T.B.; Kelley, G.O.; Alfaro, M.E.; Kurobe, T.; Davison, A.J.; Hedrick, R.P. Phylogenetic relationships in the family alloherpesviridae. Dis. Aquat. Org. 2009, 84, 179–194. [Google Scholar] [CrossRef]
- Sano, M.; Fukuda, H.; Sano, T. Isolation and characterization of a new herpesvirus from eel. In Pathology in Marine Sciences; Perkins, F.O., Cheng, T.C., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 15–31. [Google Scholar]
- Sano, T.; Fukuda, H.; Furukawa, M. Herpesvirus cyprini: Biological and oncogenic properties. Fish Pathol. 1985, 20, 381–388. [Google Scholar] [CrossRef]
- Sano, T.; Morita, N.; Sima, N.; Akimoto, M. Herpesvirus cyprini: Lethality and oncogenicity. J. Fish Dis. 1991, 14, 533–543. [Google Scholar] [CrossRef]
- Jung, S.J.; Miyazaki, T. Herpesviral haematopoietic necrosis of goldfish, carassius auratus (l.). J. Fish Dis. 1995, 18, 211–220. [Google Scholar] [CrossRef]
- Goodwin, A.E.; Khoo, L.; Lapatra, S.E.; Bonar, C.; Key, D.W.; Garner, M.; Lee, M.V.; Hanson, L. Goldfish hematopoietic necrosis herpesvirus (cyprinid herpesvirus 2) in the USA: Molecular confirmation of isolates from diseased fish. J. Aquat. Anim. Health 2006, 18, 11–18. [Google Scholar] [CrossRef]
- Goodwin, A.E.; Merry, G.E.; Sadler, J. Detection of the herpesviral hematopoietic necrosis disease agent (cyprinid herpesvirus 2) in moribund and healthy goldfish: Validation of a quantitative pcr diagnostic method. Dis. Aquat. Org. 2006, 69, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, R.P.; Gilad, O.; Yun, S.; Spangenberg, J.V.; Marty, G.D.; Nordhausen, R.W.; Kebus, M.J.; Bercovier, H.; Elder, A. A herpesvirus associated with mass mortality of juvenile and adult koi, a strain of common carp. J. Aquat. Anim. Health 2000, 12, 44–57. [Google Scholar] [CrossRef]
- Davidovich, M.; Dishon, A.; Ilouze, M.; Kotler, M. Susceptibility of cyprinid cultured cells to cyprinid herpesvirus 3. Arch. Virol. 2007, 152, 1541–1546. [Google Scholar] [CrossRef]
- Fijan, N.N.; Welborn, T.L.J.; Naftel, J.P. An Acute Viral Disease of Channel Catfish; Technical bulletin No. 43; U. S. Fish and Wildlife Service: Washington, D.C., USA, 1970. [Google Scholar]
- Wolf, K.; Darlington, R.W. Channel catfish virus: A new herpesvirus of ictalurid fish. J. Virol. 1971, 8, 525–533. [Google Scholar] [CrossRef]
- Alborali, L.; Bovo, G.; Lavazza, A.; Cappellaro, H.; Guadagnini, P.F. Isolation of an herpesvirus in breeding catfish (ictalurus melas). Bull. Eur. Assoc. Fish Pathol. 1996, 134–137. [Google Scholar]
- Hedrick, R.P.; McDowelll, T.S.; Gilad, O.; Adkison, M.; Bovo, G. Systemic herpes-like virus in catfish ictalurus melas (italy) differs from ictalurid herpesvirus 1 (north america). Dis. Aquat. Org. 2003, 55, 85–92. [Google Scholar] [CrossRef]
- Hedrick, R.P.; Groff, J.M.; McDowell, T.S. Isolation of an epitheliotropic herpesvirus from white sturgeon (acipenser transmontanus). Dis. Aquat. Org. 1991, 11, 49–56. [Google Scholar] [CrossRef]
- Watson, L.R.; Yun, S.C.; Groff, J.M.; Hedrick, R.P. Characteristics and pathogenicity of a novel herpesvirus isolated from adult and subadult white sturgeon acipenser transmontanus. Dis. Aquat. Org. 1995, 22, 199–210. [Google Scholar] [CrossRef]
- Wolf, K.; Darlington, R.W.; Taylor, W.G.; Quimby, M.C.; Nagabayashi, T. Herpesvirus salmonis: Characterization of a new pathogen of rainbow trout. J. Virol. 1978, 27, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Yoshimizu, M.; Tanaka, M.; Sannohe, H. Studies on a new virus (omv) from oncorhynchus masou–i.Characteristics and pathogenicity. Fish Pathol. 1981, 15, 143–147. [Google Scholar] [CrossRef]
- Sano, T. Viral diseases of cultured fishes in japan. Fish Pathol. 1976, 10, 221–226. [Google Scholar] [CrossRef]
- McAllister, P.E.; Herman, R.L. Epizootic mortality in hatchery-reared lake trout salvelinus namaycush caused by a putative virus possibly of the herpesvirus group. Dis. Aquat. Org. 1989, 6, 113–119. [Google Scholar] [CrossRef]
- Bradley, T.M.; Medina, D.J.; Chang, P.W.; McClain, J. Epizootic epitheliotropic disease of lake trout (salvelinus namaycush): History and viral etiology. Dis. Aquat. Org. 1989, 7, 195–201. [Google Scholar] [CrossRef]
- Marcos-Lopez, M.; Waltzek, T.B.; Hedrick, R.P.; Baxa, D.V.; Garber, A.F.; Liston, R.; Johnsen, E.; Forward, B.S.; Backman, S.; Ferguson, H.W. Characterization of a novel alloherpesvirus from atlantic cod (gadus morhua). J. Vet. Diagn. Invest. In press. 2011. [Google Scholar]
- Lunger, P.D.; Darlington, R.W.; Granoff, A. Cell-virus relationships in the lucké renal adenocarcinoma: An ultrastructure study. Ann. N. Y. Acad. Sci. 1965, 126, 289–314. [Google Scholar] [CrossRef] [PubMed]
- Rafferty, K.A. The cultivation of inclusion-associated viruses from lucké tumor frogs. Ann. N. Y. Acad. Sci. 1965, 126, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Gravell, M.; Granoff, A.; Darlington, R.W. Viruses and renal carcinoma of rana pipiens: Vii. Propagation of a herpes-type frog virus. Virology 1968, 36, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Doszpoly, A.; Benko, M.; Bovo, G.; Lapatra, S.E.; Harrach, B. Comparative analysis of a conserved gene block from the genome of the members of the genus ictalurivirus. Intervirology 2011, 54, 282–289. [Google Scholar] [CrossRef]
- Crockford, M.; Jones, J.B.; Crane, M.S.J.; Wilcox, G.E. Molecular detection of a virus, pilchard herpesvirus, associated with epizootics in australasian pilchards sardinops sagax neopilchardus. Dis. Aquat. Org. 2005, 68, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Whittington, R.J.; Jones, J.B.; Hine, P.M.; Hyatt, A.D. Epizootic mortality in the pilchard sardinops sagax neopilchardus in australia and new zealand in 1995. 1. Pathology and epizootiology. Dis. Aquat. Org. 1997, 28, 1–16. [Google Scholar] [CrossRef]
- Hyatt, A.D.; Hine, P.M.; Jones, J.B.; Whittington, R.J.; Kearns, C.; Wise, T.G.; Crane, M.S.; Williams, L.M. Epizootic mortality in the pilchard sardinops sagax neopilchardus in australia and new zealand in 1995. Ii. Identification of a herpesvirus within the gill epithelium. Dis. Aquat. Org. 1997, 28, 17–29. [Google Scholar] [CrossRef]
- Shlapobersky, M.; Sinyakov, M.S.; Katzenellenbogen, M.; Sarid, R.; Don, J.; Avtalion, R.R. Viral encephalitis of tilapia larvae: Primary characterization of a novel herpes-like virus. Virology 2010, 399, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.K.; Nielsen, O.; Mitchell, S.C.; Yamamoto, T. Characterization of herpes virus vitreum isolated from hyperplastic epidermal tissue of walleye, stizostedion vitreum vitreum (mitchill)*. J. Fish Dis. 1983, 6, 249–260. [Google Scholar] [CrossRef]
- Doszpoly, A.; Somogyi, V.; LaPatra, S.E.; Benkő, M. Partial genome characterization of acipenserid herpesvirus 2: Taxonomical proposal for the demarcation of three subfamilies in alloherpesviridae. Arch. Virol. 2011. [Google Scholar] [CrossRef]
- Winton, J.; Batts, W.; deKinkelin, P.; LeBerre, M.; Bremont, M.; Fijan, N. Current lineages of the epithelioma papulosum cyprini (epc) cell line are contaminated with fathead minnow, pimephales promelas, cells. J. Fish Dis. 2010, 33, 701–704. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kelly, R.K.; Nielsen, O. Epidermal hyperplasias of northern pike (esox lucius) associated with herpesvirus and c-type particles. Arch. Virol. 1984, 79, 255–272. [Google Scholar] [CrossRef]
- Buchanan, J.S.; Madeley, C.R. Studies on herpesvirus scophthalmi infection of turbot scophthalmus maximus (l.) ultrastructural observations. J. Fish Dis. 1978, 1, 283–295. [Google Scholar] [CrossRef]
- Hellberg, H.; Koppang, E.O.; Tørud, B.; Bjerkås, I. Subclinical herpesvirus infection in farmed turbot scophthalmus maximus. Dis. Aquat. Org. 2002, 49, 27–31. [Google Scholar] [CrossRef]
- McAllister, P.; Lidgerding, B.; Herman, R.; Hoyer, L.; Hankins, J. Viral diseases of fish: First report of carp pox in golden ide (leuciscus idus) in north america. J. Wildl. Dis. 1985, 21, 199–204. [Google Scholar] [CrossRef] [PubMed]
- McArn, G.E.; McCain, B.; Wellings, S.R. Skin lesions associated virus in pacific cod (gadus macrocephalus) in the bering sea. Fed. Proc. 1978, 37, 937. [Google Scholar]
- McCain, B.B.; Gronlund, W.D.; Myers, M.S.; Wellings, S.R. Tumours and microbial diseases of marine fishes in alaskan waters. J. Fish Dis. 1979, 2, 111–130. [Google Scholar] [CrossRef]
- Békési, L.; Kovacs-Gayer, E.; Ratz, F.; Turkovics, O. Skin infection of the sheatfish (Silurus Glanis l.) caused by a herpes virus. In Fish, Pathogens, and Environment in European Polyculture; Olah, J., Molnar, K., Jeney, Z., Eds.; Fisheries Research Institute: Szarvas, Hungary, 1981; pp. 58–69. [Google Scholar]
- Anders, K.; Moller, H. Spawning papillomatosis of smelt, osmerus eperlanus l, from the elbe estuary. J. Fish Dis. 1985, 8, 233–235. [Google Scholar] [CrossRef]
- Jakob, N.; Kehm, R.; Gelderblom, H. A novel fish herpesvirus osmerus eperlanus. Virus Genes 2010, 41, 81–85. [Google Scholar] [CrossRef]
- Morrison, C.M.; Leggiadro, C.T.; Martell, D.J. Visualization of viruses in tumors of rainbow smelt osmerus mordax. Dis. Aquat. Org. 1996, 26, 19–23. [Google Scholar] [CrossRef]
- Leibovitz, L.; Lebouitz, S.S. A viral dermatitis of the smooth dogfish, mustelus canis (mitchill). J. Fish Dis. 1985, 8, 273–279. [Google Scholar] [CrossRef]
- Shchelkunov, I.S.; Karaseva, T.A.; Kadoshnikov, Y.U.P. Atlantic salmon papillomatosis: Visualization of herpesvirus-like particles in skin growths of affected fish. Bull. Eur. Ass. Fish Pathol. 1992, 12, 28–31. [Google Scholar]
- Mellergard, S.; Bloch, B. Herpesvirus-like particles in angelfish pterophyllum altum. Dis. Aquat. Org. 1988, 5, 151–155. [Google Scholar] [CrossRef]
- Kent, M.L.; Myers, M.S. Hepatic lesions in a redstriped rockfish (sebastes proriger) suggestive of a herpesvirus infection. Dis. Aquat. Organ. 2000, 41, 237–239. [Google Scholar] [CrossRef]
- Davison, A.J. Evolution of the herpesviruses. Vet. Microbiol. 2002, 86, 69–88. [Google Scholar] [CrossRef] [PubMed]
- Hanson, L.A.; Rudis, M.R.; Vasquez-Lee, M.; Montgomery, R.D. A broadly applicable method to characterize large DNA viruses and adenoviruses based on the DNA polymerase gene. Virol. J. 2006, 3, 28. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J. The genome of salmonid herpesvirus 1. J. Virol. 1998, 72, 1974–1982. [Google Scholar] [CrossRef]
- Lu, Y.; Yu, Q.; Zamzow, J.P.; Wang, Y.; Losey, G.S.; Balazs, G.H.; Nerurkar, V.R.; Yanagihara, R. Detection of green turtle herpesviral sequence in saddleback wrasse thalassoma duperrey: A possible mode of transmission of green turtle fibropapilloma. J. Aquat. Anim. Health 2000, 12, 58–63. [Google Scholar] [CrossRef]
- Roizman, B.; Sears, A.E. Herpesviruses and their replication. In Fields Virology; Fields, B.N., Melnick, D.M., Monath, T.P., Roizman, B., Eds.; Raven Press: New York, NY, USA, 1990; pp. 1795–1829. [Google Scholar]
- Booy, F.P.; Trus, B.L.; Davison, A.J.; Steven, A.C. The capsid architecture of channel catfish virus, an evolutionarily distant herpesvirus, is largely conserved in the absence of discernible sequence homology with herpes simplex virus. Virology 1996, 215, 134–141. [Google Scholar] [CrossRef]
- Sano, N.; Moriwake, M.; Hondo, R.; Sano, T. Herpesvirus cyprini: A search for viral genome in infected fish by in situ hybridization. J. Fish Dis. 1993, 16, 495–499. [Google Scholar] [CrossRef]
- Eide, K.E.; Miller-Morgan, T.; Heidel, J.R.; Kent, M.L.; Bildfell, R.J.; Lapatra, S.; Watson, G.; Jin, L. Investigation of koi herpesvirus latency in koi. J. Virol. 2011, 85, 4954–4962. [Google Scholar] [CrossRef]
- Gou, D.F.; Kubota, H.; Onuma, M.; Kodama, H. Detection of salmonid herpesvirus (oncorhynchus masou virus) in fish by southern-blot technique. J. Vet. Med. Sci. 1991, 53, 43–48. [Google Scholar] [CrossRef]
- Boyle, J.; Blackwell, J. Use of polymerase chain reaction to detect latent channel catfish virus. Am. J. Vet. Res. 1991, 52, 1965–1968. [Google Scholar]
- Ariav, R.; Tinman, S.; Paperna, I.; Bejerano, I. First report of newly emerging viral disease of cyprinus carpio species in israel. In Proceedings of the EAFP 9th International Conference, Rhodes, Greece, 19–24 September 1999. [Google Scholar]
- Bretzinger, A.; Fischer-Scherl, T.; Oumouma, R.; Hoffmann, R.; Truyen, U. Mass mortalities in koi, cyprinus carpio, associated with gill and skin disease. Bull. Eur. Ass. Fish Pathol 1999, 19, 182–185. [Google Scholar]
- Crane, M.; Sano, M.; Komar, C. Infection with Koi Herpesvirus-Disease Card. Developed to Support the NACA/FAO/OIE Regional Quarterly Aquatic Animal Disease (QAAD) Reporting System in the Asia-Pacific; NACA: Bangkok, Thailand, 2004; p. 11. [Google Scholar]
- Haenen, O.L.M.; Way, K.; Bergmann, S.M.; Ariel, E. The emergenece of koi herpesvirus and its significance to european aquaculture. Bull. Eur. Ass. Fish Pathol 2004, 24, 293–307. [Google Scholar]
- Hoffmann, R. Koiseuche bedroht karpfenteichwirtschaft. Fischer und Teichwirt 2000, 11, 432. [Google Scholar]
- Michel, B.; Fournier, G.; Lieffrig, F.; Costes, B.; Vanderplasschen, A. Cyprinid herpesvirus 3. Emerg. Infect. Dis. 2010, 16, 1835–1843. [Google Scholar] [CrossRef] [PubMed]
- Pokorova, D.; Vesely, T.; Piackova, V.; Reschova, S.; Hulova, J. Current knowledge on koi herpesvirus (khv): A review. Vet Med. Czech 2005, 50, 139–147. [Google Scholar] [CrossRef]
- Ilouze, M.; Dishon, A.; Kotler, M. Characterization of a novel virus causing a lethal disease in carp and koi. Microbiol. Mol. Biol. Rev. 2006, 70, 147–156. [Google Scholar] [CrossRef]
- Michel, B.; Leroy, B.; Stalin Raj, V.; Lieffrig, F.; Mast, J.; Wattiez, R.; Vanderplasschen, A.F.; Costes, B. The genome of cyprinid herpesvirus 3 encodes 40 proteins incorporated in mature virions. J. Gen. Virol. 2010, 91, 452–462. [Google Scholar] [CrossRef]
- Gilad, O.; Yun, S.; Andree, K.B.; Adkison, M.A.; Zlotkin, A.; Bercovier, H.; Eldar, A.; Hedrick, R.P. Initial characteristics of koi herpesvirus and development of a polymerase chain reaction assay to detect the virus in koi, cyprinus carpio koi. Dis. Aquat. Org. 2002, 48, 101–108. [Google Scholar] [CrossRef]
- Perelberg, A.; Smirnov, M.; Hutoran, M.; Diamant, A.; Bejerano, I.; Kotler, M. Epidemiological description of a new viral disease afflicting cultured cyprinus carpio in israel. The Israel Journal of Aquaculture—Bamidgeh 2003, 55, 5–12. [Google Scholar] [CrossRef]
- Ronen, A.; Perelberg, A.; Abramowitz, J.; Hutoran, M.; Tinman, S.; Bejerano, I.; Steinitz, M.; Kotler, M. Efficient vaccine against the virus causing a lethal disease in cultured cyprinus carpio. Vaccine 2003, 21, 4677–4684. [Google Scholar] [CrossRef]
- Gray, W.L.; Mullis, L.; LaPatra, S.E.; Groff, J.M.; Goodwin, A. Detection of koi herpesvirus DNA in tissues of infected fish. J. Fish Dis. 2002, 25, 171–178. [Google Scholar] [CrossRef]
- Body, A.; Lieffrig, F.; Chalrlier, G.; Collard, A. Isolation of virus-like particles from koi (cyprinus carpio) suffering gill necrosis. Bull. Eur. Assoc. Fish Pathol. 2000, 20, 87–88. [Google Scholar]
- Minamoto, T.; Honjo, M.N.; Uchii, K.; Yamanaka, H.; Suzuki, A.A.; Kohmatsu, Y.; Iida, T.; Kawabata, Z. Detection of cyprinid herpesvirus 3 DNA in river water during and after an outbreak. Vet. Microbiol. 2009, 135, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Walster, C. Clinical observations of severe mortalities in koi carp, cyprinus carpio, with gill disease. Fish Vet. J. 1999, 3, 54–58. [Google Scholar]
- Gilad, O.; Yun, S.; Adkison, M.A.; Way, K.; Willits, N.H.; Bercovier, H.; Hedrick, R.P. Molecular comparison of isolates of an emerging fish pathogen, koi herpesvirus, and the effect of water temperature on mortality of experimentally infected koi. J. Gen. Virol. 2003, 84, 2661–2667. [Google Scholar] [CrossRef]
- Pikarsky, E.; Ronen, A.; Abramowitz, J.; Levavi-Sivan, B.; Hutoran, M.; Shapira, Y.; Steinitz, M.; Perelberg, A.; Soffer, D.; Kotler, M. Pathogenesis of acute viral disease induced in fish by carp interstitial nephritis and gill necrosis virus. J. Virol. 2004, 78, 9544–9551. [Google Scholar] [CrossRef]
- Neukirch, M.; Kunz, U. Isolation and preliminary characterization of several viruses from koi (cyprinus carpio) suffering gill necrosis and mortality. Bull. Eur. Assoc. Fish Pathol. 2001, 21, 125–135. [Google Scholar]
- Neukirch, M.; Bottcher, K.; Bunnajirakul, S. Isolation of a virus from koi with altered gills. Bull. Eur. Ass. Fish Pathol. 1999, 19, 221–224. [Google Scholar]
- El-Matbouli, M.; Rucker, U.; Soliman, H. Detection of cyprinid herpesvirus-3 (cyhv-3) DNA in infected fish tissues by nested polymerase chain reaction. Dis. Aquat. Org. 2007, 78, 23–28. [Google Scholar] [CrossRef]
- El-Matbouli, M.; Soliman, H. Transmission of cyprinid herpesvirus-3 (cyhv-3) from goldfish to naive common carp by cohabitation. Res. Vet. Sci. 2011, 90, 536–539. [Google Scholar] [CrossRef]
- Sadler, J.; Marecaux, E.; Goodwin, A.E. Detection of koi herpes virus (cyhv-3) in goldfish, carassius auratus (l.), exposed to infected koi. J. Fish Dis. 2008, 31, 71–72. [Google Scholar] [CrossRef]
- Bergmann, S.M.; Sadowski, J.; Kiełpiński, M.; Bartłomiejczyk, M.; Fichtner, D.; Riebe, R.; Lenk, M.; Kempter, J. Susceptibility of koi × crucian carp and koi × goldfish hybrids to koi herpesvirus (khv) and the development of khv disease (khvd). J. Fish Dis. 2010, 33, 267–272. [Google Scholar] [CrossRef]
- Sunarto, A.; McColl, K.A.; Crane, M.S.; Sumiati, T.; Hyatt, A.D.; Barnes, A.C.; Walker, P.J. Isolation and characterization of koi herpesvirus (khv) from indonesia: Identification of a new genetic lineage. J. Fish Dis. 2011, 34, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Kurita, J.; Yuasa, K.; Ito, T.; Sano, M.; Hedrick, R.P.; Engelsma, M.Y.; Haenen, O.L.M.; Sunarto, A.; Kholidin, E.B.; Chou, H.Y.; et al. Molecular epidemiology of koi herpesvirus. Fish Pathol. 2009, 44, 59–66. [Google Scholar] [CrossRef]
- Gilad, O.; Yun, S.; Zagmutt-Vergara, F.J.; Leutenegger, C.M.; Bercovier, H.; Hedrick, R.P. Concentrations of a koi herpesvirus (khv) in tissues of experimentally infected cyprinus carpio koi as assessed by real-time taqman pcr. Dis. Aquat. Organ. 2004, 60, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Costes, B.; Stalin Raj, V.; Michel, B.; Fournier, G.; Thirion, M.; Gillet, L.; Mast, J.; Lieffrig, F.; Bremont, M.; Vanderplasschen, A. The major portal of entry of koi herpesvirus in cyprinus carpio is the skin. J. Virol. 2009, 83, 2819–2830. [Google Scholar] [CrossRef] [PubMed]
- Hutoran, M.; Ronen, A.; Perelberg, A.; Ilouze, M.; Dishon, A.; Bejerano, I.; Chen, N.; Kotler, M. Description of an as yet unclassified DNA virus from diseased cyprinus carpio species. J. Virol. 2005, 79, 1983–1991. [Google Scholar] [CrossRef]
- Mettenleiter, T.C.; Klupp, B.G.; Granzow, H. Herpesvirus assembly: An update. Virus Res. 2009, 143, 222–234. [Google Scholar] [CrossRef]
- Miwa, S.; Ito, T.; Sano, M. Morphogenesis of koi herpesvirus observed by electron microscopy. J. Fish Dis. 2007, 30, 715–722. [Google Scholar] [CrossRef]
- Costes, B.; Fournier, G.; Michel, B.; Delforge, C.; Raj, V.S.; Dewals, B.; Gillet, L.; Drion, P.; Body, A.; Schynts, F.; et al. Cloning of koi herpesvirus genome as an infectious bacterial artificial chromosome: Demonstration that disruption of the TK locus induces a partial attenuation in Cyprinus carpio koi. J. Virol. 2008, 82, 4955–4964. [Google Scholar] [CrossRef]
- Dishon, A.; Davidovich, M.; Ilouze, M.; Kotler, M. Persistence of cyprinid herpesvirus 3 in infected cultured carp cells. J. Virol. 2007, 81, 4828–4836. [Google Scholar] [CrossRef]
- Waltzek, T.B.; Kelley, G.O.; Stone, D.M.; Way, K.; Hanson, L.; Fukuda, H.; Hirono, I.; Aoki, T.; Davison, A.J.; Hedrick, R.P. Koi herpesvirus represents a third cyprinid herpesvirus (cyhv-3) in the family herpesviridae. J. Gen. Virol. 2005, 86, 1659–1667. [Google Scholar] [CrossRef] [PubMed]
- Ilouze, M.; Dishon, A.; Kotler, M. Department of Pathology, Hadassah Medical School, The Hebrew University, Jerusalem, Israel. Coordinated and sequential transcription of the Cyprinid herpesvirus 3 annotated genes. To be submitted for publication, 2011.
- Rosenkranz, D.; Klupp, B.G.; Teifke, J.P.; Granzow, H.; Fichtner, D.; Mettenleiter, T.C.; Fuchs, W. Identification of envelope protein porf81 of koi herpesvirus. J. Gen. Virol. 2008, 89, 896–900. [Google Scholar] [CrossRef] [PubMed]
- Ilouze, M.; Dishon, A.; Kotler, M. Department of Pathology, Hadassah Medical School, The Hebrew University, Jerusalem, Israel. Down regulation of the Cyprinid herpesvirus-3 annotated genes in cultured cells maintain at restrictive high temperature. To be submitted for publication, 2011.
- Ronen, A.; Perelberg, A.; Hutoran, M.; Shapira, Y.; Steinitz, M.; Levavi-Sivan, B.; Pikarsky, E.; Kotler, M. Prevention of a mortal disease of carps induced by the carp nephritis and gill necrosis virus (cngv) in Israel. Bull. Fish Agen. Supplement 2005, 2, 9–11. [Google Scholar]
- Gracey, A.Y.; Fraser, E.J.; Li, W.; Fang, Y.; Taylor, R.R.; Rogers, J.; Brass, A.; Cossins, A.R. Coping with cold: An integrative, multitissue analysis of the transcriptome of a poikilothermic vertebrate. Proc. Natl. Acad. Sci. USA 2004, 101, 16970–16975. [Google Scholar] [CrossRef] [PubMed]
- Rakus, K.L.; Wiegertjes, G.F.; Adamek, M.; Siwicki, A.K.; Lepa, A.; Irnazarow, I. Resistance of common carp (cyprinus carpio l.) to cyprinid herpesvirus-3 is influenced by major histocompatibility (mh) class ii b gene polymorphism. Fish Shellfish Immunol 2009, 26, 737–743. [Google Scholar] [CrossRef]
- Kongchum, P.; Palti, Y.; Hallerman, E.M.; Hulata, G.; David, L. Snp discovery and development of genetic markers for mapping innate immune response genes in common carp (cyprinus carpio). Fish Shellfish Immunol. 2010, 29, 356–361. [Google Scholar] [CrossRef]
- Bejerano, I.; Perelberg, A.; Kolter, M. Department of Pathology, Central Fish Health Laboratory, Ministry of Agriculture and Rural Development, Israel; Hadassah Medical School, the Hebrew University, Jerusalem, Israel. Unpublished work, 2011.
- Benmansour, A.; de Kinkelin, P. Live fish vaccines: History and perspectives. In Fish Vaccinology; Gudding, R., Lillehuaug, A., Midtlyng, P.J., Brown, F., Eds.; Karger: Basel, Switzerland, 1997; pp. 279–289. [Google Scholar]
- Marsden, M.J.; Vaughan, L.M.; Foster, T.J.; Secombes, C.J. A live (delta aroa) aeromonas salmonicida vaccine for furunculosis preferentially stimulates t-cell responses relative to b-cell responses in rainbow trout (oncorhynchus mykiss). Infect. Immun. 1996, 64, 3863–3869. [Google Scholar] [CrossRef]
- Marsden, M.J.; Vaughan, L.M.; Fitzpatrick, R.M.; Foster, T.J.; Secombes, C.J. Potency testing of a live, genetically attenuated vaccine for salmonids. Vaccine 1998, 16, 1087–1094. [Google Scholar] [CrossRef]
- Perelberg, A.; Ronen, A.; Hutoran, M.; Smith, Y.; Kotler, M. Protection of cultured cyprinus carpio against a lethal viral disease by an attenuated virus vaccine. Vaccine 2005, 23, 3396–3403. [Google Scholar] [CrossRef]
- Dishon, A.; Ashoulin, O. Back passage/shed assay for Cyprinid Herpes Virus Type 3 Modified live virus. Kovax Ltd., Jerusalem, Israel; Madan Ltd. Maagan Michael, Israel. In vivo backpassage assay testing the potential reversion of the vaccine strain to a virulent phenotype. Unpublished work, 2009. 2009. [Google Scholar]
- Drake, J.W. Mutagenic mechanisms. Annu. Rev. Genetics 1969, 3, 247–268. [Google Scholar] [CrossRef]
- Drake, J.W. The biochemistry of mutagenesis. Annu. Rev. Biochem. 1976, 45, 11–37. [Google Scholar] [CrossRef] [PubMed]
- Freese, E. Molecular mechanisms of mutations. In Molecular Genetics; Taylor, J.H., Ed.; Academic Press: New York, NY, USA, 1963; p. 207. [Google Scholar]
- Perelberg, A.; Ilouze, M.; Kotler, M.; Steinitz, M. Antibody response and resistance of cyprinus carpio immunized with cyprinid herpes virus 3 (cyhv-3). Vaccine 2008, 26, 3750–3756. [Google Scholar] [CrossRef] [PubMed]
- Sano, N.; Moriwake, M.; Sano, T. Herpesvirus cyprini: Thermal effects on pathogenicity and oncogenicity. Fish Pathol. 1993, 28, 171–175. [Google Scholar] [CrossRef]
- van Regenmortel, M.H.V.; Fauquet, C.M.; Bishop, D.H.; Carstens, E.B.; Estes, M.K.; Lemon, S.M.; Maniloff, J.; Mayo, M.A.; McGeoch, D.J.; Pringle, C.R.; et al. Virus Taxonomy. Seventh Report of the International Committee on Taxonomy of Viruses; Academic Press: San Diego, CA, USA, 2000. [Google Scholar]
- St-Hilaire, S.; Beevers, N.; Way, K.; Le Deuff, R.M.; Martin, P.; Joiner, C. Reactivation of koi herpesvirus infections in common carp cyprinus carpio. Dis. Aquat. Org. 2005, 67, 15–23. [Google Scholar] [CrossRef]
- Eide, K.; Miller-Morgan, T.; Heidel, J.; Bildfell, R.; Jin, L. Results of total DNA measurement in koi tissue by koi herpes virus real-time pcr. J. Virol. Methods 2011, 172, 81–84. [Google Scholar] [CrossRef]
- Plumb, J.A. Epizootiology of channel catfish virus disease. Mar. Fish. Rev. 1978, 3, 26–29. [Google Scholar]
- Thompson, D.J.; Khoo, L.H.; Wise, D.J.; Hanson, L.A. Evaluation of channel catfish virus latency on fingerling production farms in mississippi. J. Aquat. Anim. Health 2005, 17, 211–215. [Google Scholar] [CrossRef]
- Hedrick, R.P.; Groff, J.M.; McDowell, T. Response of adult channel catfish to waterborne exposures of channel catfish virus. Prog. Fish Cult. 1987, 49, 181–187. [Google Scholar] [CrossRef]
- Plumb, J.A. Effects of temperature on mortality of fingerling channel catfish (ictalurus punctatus) experimentally infected with channel catfish virus. J. Fish. Res. Board Can. 1973, 30, 568–570. [Google Scholar] [CrossRef]
- Plumb, J.A.; Gaines, J.L.; Mora, E.C.; Bradley, G.G. Histopathology and electron microscopy of channel catfish virus in infected channel catfish, ictalurus punctatus (rafinesque). J. Fish. Biol. 1974, 6, 661–664. [Google Scholar] [CrossRef]
- Wolf, K.; Herman, R.L.; Carlson, C.P. Fish viruses: Histopathologic changes associated with experimental channel catfish virus disease. J. Fish. Res. Board Can. 1972, 29, 149–150. [Google Scholar] [CrossRef]
- Bowser, P.R.; Plumb, J.A. Growth rates of a new cell line from channel catfish ovary and channel catfish virus replication at different temperatures. Can. J. Fish. Aquat. Sci. 1980, 37, 871–873. [Google Scholar] [CrossRef]
- Plumb, J.A.; Bowser, P.R. Recent developments in channel catfish virus research. Mar. Fish. Rev. 1978, 40, 12–13. [Google Scholar]
- Plumb, J.A.; Hilge, V.; Quinlan, E.F. Resistance of the european catfish (silurus glanis) to channel catfish virus. J. Appl. Ichthyol. 1985, 1, 87–89. [Google Scholar] [CrossRef]
- Boon, J.H.; McDowell, T.; Hedrick, R.P. Resistance of the african (clarias gariepineus) and asian catfish (clarias batrachus) to channel catfish virus. Aqua (Oxf.) 1988, 74, 191–194. [Google Scholar]
- Silverstein, P.S.; Bosworth, B.G.; Gaunt, P.S. Differential susceptibility of blue catfish, ictalurus furcatus (valenciennes), channel catfish, i. Punctatus (rafinesque), and blue × channel catfish hybrids to channel catfish virus. J. Fish Dis. 2008, 31, 77–79. [Google Scholar] [CrossRef]
- Nusbaum, K.E.; Grizzle, J.M. Uptake of channel catfish virus from water by channel catfish and bluegills. Am. J. Vet. Res. 1987, 48, 375–377. [Google Scholar]
- Kancharla, S.R.; Hanson, L.A. Production and shedding of channel catfish virus (ccv) and thymidine kinase negative ccv in immersion exposed channel catfish fingerlings. Dis. Aquat. Org. 1996, 27, 25–34. [Google Scholar] [CrossRef]
- Dixon, R.A.F.; Farber, F.E. Channel catfish virus: Physicochemical properties of the viral genome and identification of viral polypeptides. Virology 1980, 103, 267–278. [Google Scholar] [CrossRef]
- Chousterman, S.; Lacasa, M.; Sheldrick, P. Physical map of the channel catfish virus genome: Location of sites for restriction endonucleases eco ri, hind iii, hpa i, and xba i. J. Virol. 1979, 31, 73–85. [Google Scholar] [CrossRef]
- Colyer, T.E.; Bowser, P.R.; Doyle, J.; Boyle, J.A. Channel catfish virus: Use of nucleic acids in studying viral relationships. Am. J. Vet. Res. 1986, 47, 2007–2011. [Google Scholar] [PubMed]
- Cebrian, J.; Bucchini, D.; Sheldrick, P. "Endless" viral DNA in cells infected with channel catfish virus. J. Virol. 1983, 46, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Hanson, L.A.; Kousoulas, K.G.; Thune, R.L. Channel catfish herpesvirus (ccv) encodes a functional thymidine kinase gene: Elucidation of a point mutation that confers resistance to ara-t. Virology 1994, 202, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Kunec, D.; Hanson, L.A.; van Haren, S.; Nieuwenhuizen, I.F.; Burgess, S.C. An over-lapping bacterial artificial chromosome system that generates vectorless progeny for channel catfish herpesvirus. J. Virol. 2008, 82, 3872–3881. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J.; Davison, M.D. Identification of structural proteins of channel catfish virus by mass spectrometry. Virology 1995, 206, 1035–1043. [Google Scholar] [CrossRef]
- Vanderheijden, N.; Alard, P.; Lecomte, C.; Martial, J.A. The attenuated v60 strain of channel catfish virus possesses a deletion in orf50 coding for a potentially secreted glycoprotein. Virology 1996, 218, 422–426. [Google Scholar] [CrossRef]
- Vanderheijden, N.; Hanson, L.A.; Thiry, E.; Martial, J.A. Channel catfish virus gene 50 encodes a secreted, mucin-like glycoprotein. Virology 1999, 257, 220–227. [Google Scholar] [CrossRef]
- Hanson, L.A.; Thune, R.L. Characterization of thymidine kinase encoded by channel catfish virus. J. Aquat. Anim. Health 1993, 5, 199–204. [Google Scholar] [CrossRef]
- Harrison, P.T.; Thompson, R.; Davison, A.J. Evolution of herpesvirus thymidine kinases from cellular deoxycytidine kinase. J. Gen. Virol. 1991, 72, 2583–2586. [Google Scholar] [CrossRef]
- Kunec, D.; Nanduri, B.; Burgess, S.C. Experimental annotation of channel catfish virus by probabilistic proteogenomic mapping. Proteomics 2009, 9, 2634–2647. [Google Scholar] [CrossRef]
- Huang, S.; Hanson, L.A. Temporal gene regulation of the channel catfish virus (ictalurid herpesvirus 1). J. Virol. 1998, 72, 1910–1917. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, P.S.; Bird, R.C.; Santen, V.L.v.; Nusbaum, K.E. Immediate-early transcription from the channel catfish virus genome: Characterisation of two immediate early transcripts. J. Virol. 1995, 69, 3161–3166. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, P.S.; van Santen, V.L.; Nusbaum, K.E.; Bird, R.C. Expression kinetics and mapping of the thymidine kinase transcript and an immediate-early transcript from channel catfish virus. J. Virol. 1998, 72, 3900–3906. [Google Scholar] [CrossRef] [PubMed]
- Stingley, R.L.; Gray, W.L. Transcriptional regulation of the channel catfish virus genome direct repeat region. J. Gen. Virol. 2000, 81, 2005–2010. [Google Scholar] [CrossRef] [PubMed]
- Pope, R.K.; Scheetz, R.W. Channel catfish virus: Interaction with the cytoskeleton. J. Fish Dis. 1998, 21, 433–442. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Rycyzyn, M.; Clem, L.W.; Miller, N.W. Productive infection of continuous lines of channel catfish leukocytes by channel catfish virus. Virology 1993, 193, 989–992. [Google Scholar] [CrossRef]
- Majji, S. Expression analysis of viral and host genes following in vitro infection. Ph.D. Dissertation, University of Mississippi Medical Center, Jackson, MS, USA, 2010. [Google Scholar]
- Plant, K.P.; Harbottle, H.; Thune, R.L. Poly i:C induces an antiviral state against ictalurid herpesvirus 1 and mx1 transcription in the channel catfish (ictalurus punctatus). Dev. Comp. Immunol. 2005, 29, 627–635. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Logue, O.; Antao, A.; Chinchar, G.D. Channel catfish reovirus (crv) inhibits replication of channel catfish herpesvirus (ccv) by two distinct mechanisms: Viral interference and induction of an anti-viral factor. Dis. Aquat. Organ. 1998, 33, 77–85. [Google Scholar] [CrossRef]
- Hogan, R.J.; Stuge, T.B.; Clem, L.W.; Miller, N.W.; Chinchar, V.G. Anti-viral cytotoxic cells in the channel catfish (ictalurus punctatus). Dev. Comp. Immunol. 1996, 20, 115–127. [Google Scholar] [CrossRef]
- Plumb, J.A. Neutralization of channel catfish virus by serum of channel catfish. J. Wildl. Dis. 1973, 9, 324–330. [Google Scholar] [CrossRef]
- Arnizaut, A.B.; Hanson, L.A. Antibody response of channel catfish after channel catfish virus infection and following dexamethasone treatment. Dis. Aquat. Org. 2011, 95, 189–201. [Google Scholar] [CrossRef]
- Crawford, S.A.; Gardner, I.A.; Hedrick, R.P. An enzyme linked immunosorbent assay (elisa) for detection of antibodies to channel catfish virus (ccv) in channel catfish. J. Aquat. Anim. Health 1999, 11, 148–153. [Google Scholar] [CrossRef]
- Hedrick, R.P.; McDowell, T. Passive transfer of sera with antivirus neutralizing activity from adult channel catfish protects juveniles from channel catfish virus. Trans. Am. Fish. Soc. 1987, 116, 277–281. [Google Scholar] [CrossRef]
- Bowser, P.R.; Munson, A.D. Seasonal variation in channel catfish virus antibody titers in adult channel catfish. Prog. Fish Cult. 1986, 48, 198–199. [Google Scholar] [CrossRef]
- Hanson, L.A.; Rudis, M.R.; Petrie-Hanson, L. Susceptibility of channel catfish fry to channel catfish virus (ccv) challenge increases with age. Dis. Aquat. Org. 2004, 62, 27–34. [Google Scholar] [CrossRef]
- Plumb, J.A.; Thune, R.L.; Klesius, P.H. Detection of channel catfish virus in adult fish. Develop. Biol. Standard 1981, 49, 29–34. [Google Scholar]
- Bowser, P.R.; Munson, A.D.; Jarboe, H.H.; Francis-Floyd, R.; Waterstrat, P.R. Isolation of channel catfish virus from channel catfish ictalurus punctatus (rafinesque), broodstock. J. Fish Dis. 1985, 8, 557–561. [Google Scholar] [CrossRef]
- Wise, J.A.; Bowser, P.R.; Boyle, J.A. Detection of channel catfish virus in asymptomatic adult channel catfish, ictalurus punctatus (rafinesque). J. Fish Dis. 1985, 8, 485–493. [Google Scholar] [CrossRef]
- Wise, J.A.; Harrell, S.F.; Busch, R.L.; Boyle, J.A. Vertical transmission of channel catfish virus. Am. J. Vet. Res. 1988, 49, 1506–1509. [Google Scholar]
- Baek, Y.-S.; Boyle, J.A. Detection of channel catfish virus in adult channel catfish by use of nested polymerase chain reaction. J. Aquat. Anim. Health 1996, 8, 97–103. [Google Scholar] [CrossRef]
- Gray, W.L.; Williams, R.J.; Jordan, R.L.; Griffin, B.R. Detection of channel catfish virus DNA in latently infected catfish. J. Gen. Virol. 1999, 80, 1817–1822. [Google Scholar] [CrossRef] [PubMed]
- Nusbaum, K.E.; Smith, B.F.; DeInnocentes, P.; Bird, R.C. Protective immunity induced by DNA vaccination of channel catfish with early and late transcripts of the channel catfish herpesvirus (ihv-1). Vet. Immunol. Immunopathol. 2002, 84, 151–168. [Google Scholar] [CrossRef] [PubMed]
- Noga, E.J.; Hartmann, J.X. Establishment of walking catfish (clarias batrachus) cell lines and development of a channel catfish (ictalurus punctatus) virus vaccine. Can. J. Fish. Aquat. Sci 1981, 38, 925–929. [Google Scholar] [CrossRef]
- Zhang, H.G.; Hanson, L.A. Deletion of thymidine kinase gene attenuates channel catfish herpesvirus while maintaining infectivity. Virology 1995, 209, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Vanderheijden, N.; Martial, J.A.; Hanson, L.A. Channel catfish virus vaccine. US 6,322,793, 2001.
- Harbottle, H.; Plant, K.P.; Thune, R.L. DNA vaccination against channel catfish virus results in minimal immune response and is not efficacious against challenge. J. Aquat. Anim. Health 2005, 17, 251–262. [Google Scholar] [CrossRef]
- Zhang, H.G.; Hanson, L.A. Recombinant channel catfish virus (ictalurid herpesvirus 1) can express foreign genes and induce antibody production against the gene product. J. Fish Dis. 1996, 19, 121–128. [Google Scholar] [CrossRef]
- Kunec, D.; van Haren, S.; Burgess, S.C.; Hanson, L.A. A gateway((r)) recombination herpesvirus cloning system with negative selection that produces vectorless progeny. J. Virol. Methods 2009, 155, 82–86. [Google Scholar] [CrossRef]

© 2011 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (https://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hanson, L.; Dishon, A.; Kotler, M. Herpesviruses that Infect Fish. Viruses 2011, 3, 2160-2191. https://doi.org/10.3390/v3112160
Hanson L, Dishon A, Kotler M. Herpesviruses that Infect Fish. Viruses. 2011; 3(11):2160-2191. https://doi.org/10.3390/v3112160
Chicago/Turabian StyleHanson, Larry, Arnon Dishon, and Moshe Kotler. 2011. "Herpesviruses that Infect Fish" Viruses 3, no. 11: 2160-2191. https://doi.org/10.3390/v3112160
APA StyleHanson, L., Dishon, A., & Kotler, M. (2011). Herpesviruses that Infect Fish. Viruses, 3(11), 2160-2191. https://doi.org/10.3390/v3112160





