The Molecular Biology of Frog Virus 3 and other Iridoviruses Infecting Cold-Blooded Vertebrates
Abstract
1. Taxonomy
2. Vital Statistics: Virion Structure and Genomic Organization
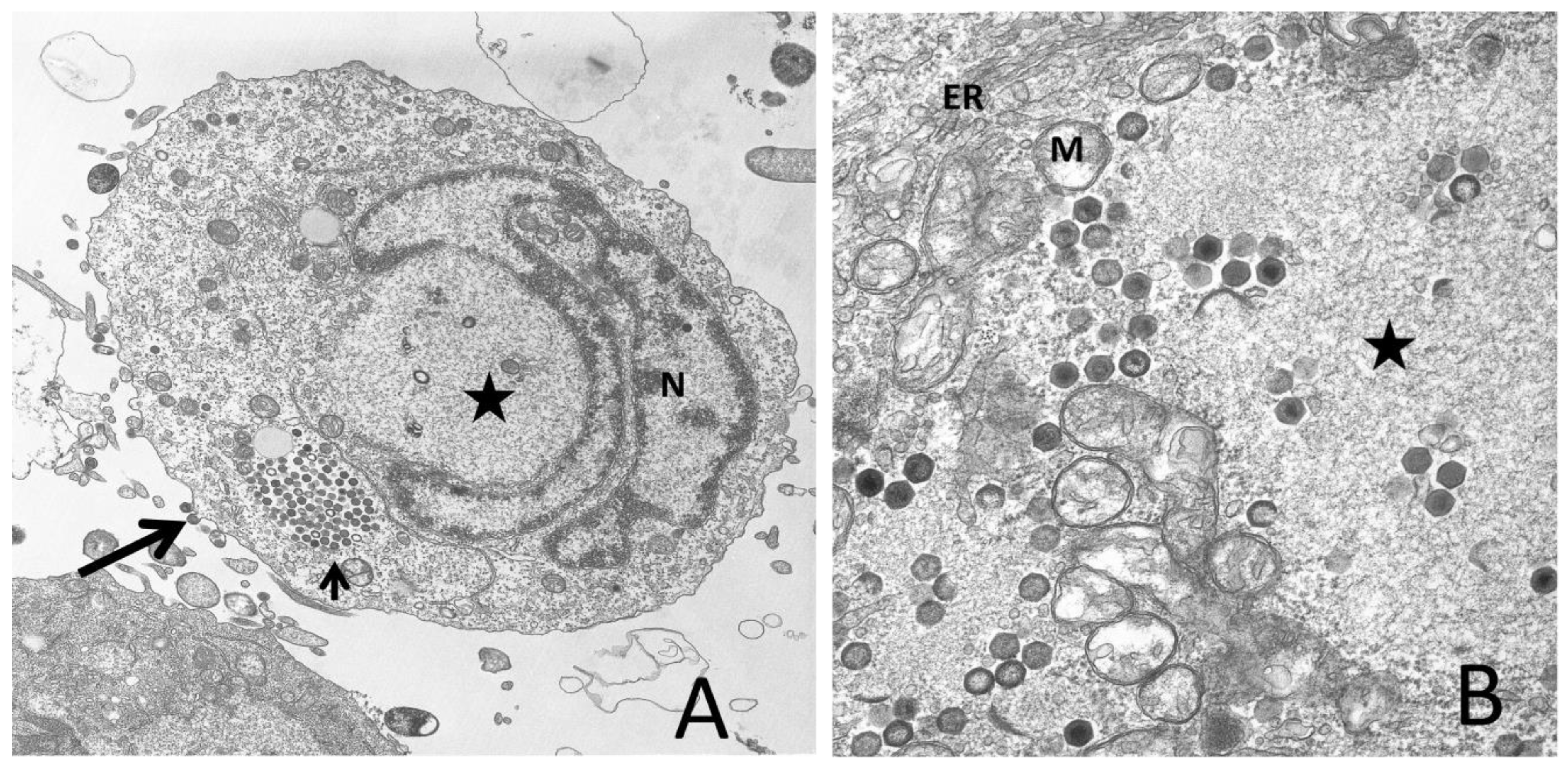
2.1. Virion Morphology and Composition
2.2. Genomic Organization
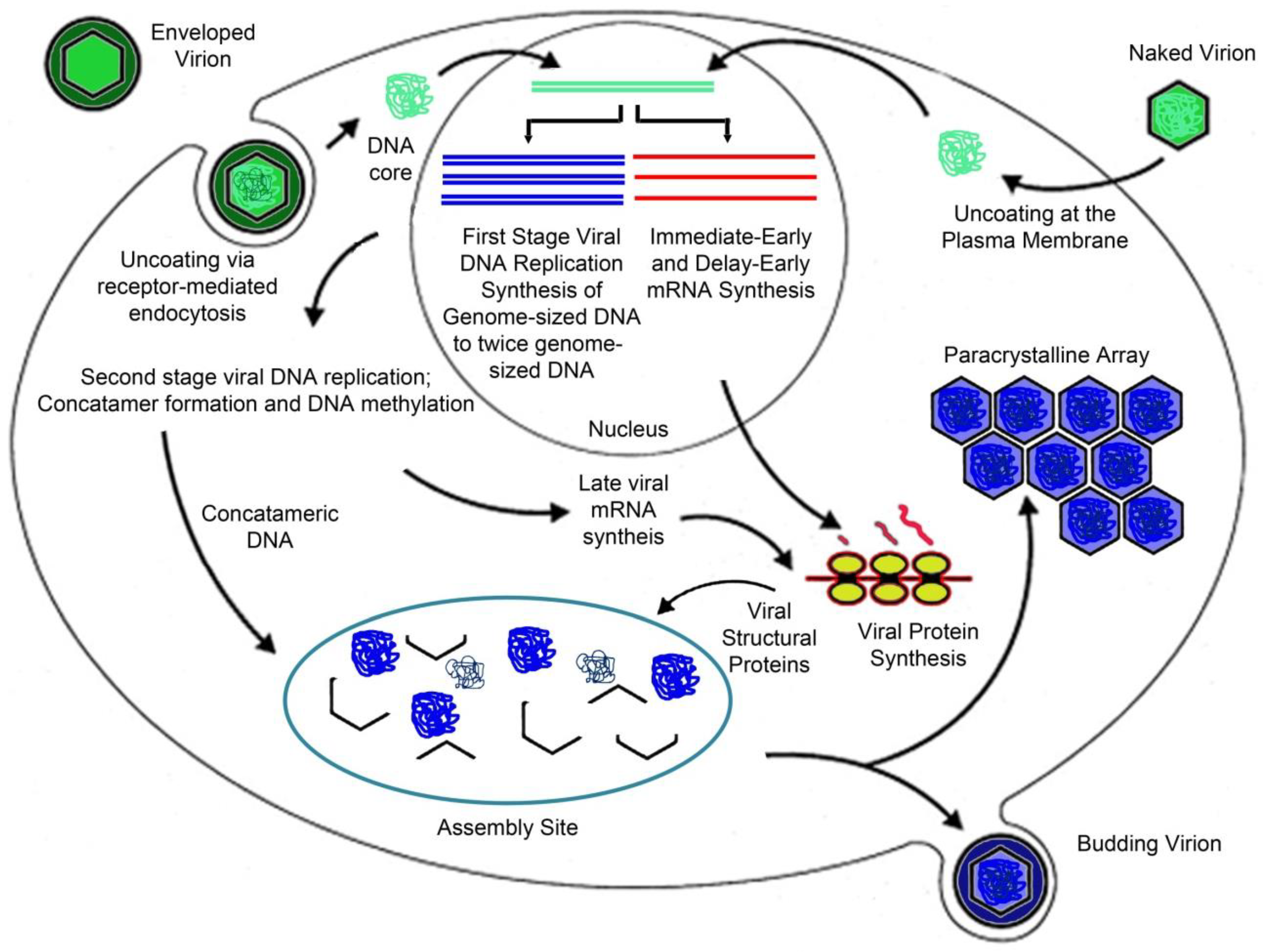
3. Viral Replication Strategy
3.1. Virion Entry
3.2. Early Events/Nuclear Phase
3.3. Late Events/Cytoplasmic Phase
3.4. Effect of Virus Infection on Host Functions
4. Viral Genomes and Evidence for Host Shifts
5. Pathological and Immunological Aspects of RV Infection
6. Ecology of Iridovirus Infections
7. Elucidation of Viral Gene Function
7.1. Early Studies: Temperature-Sensitive (ts) Mutants and Biochemical Studies
7.2. Knock Down of Viral Gene Expression Using asMOs
7.3. Knock Down of Viral Gene Expression Using siRNA
7.4. Knock out Approaches to Elucidate Viral Gene Function
7.5. Assessment of Viral Gene Function by Analysis of Recombinant Viral Proteins
7.5.1. ICP46
7.5.2. Viral Vascular Endothelial Growth Factor
7.5.3. Histone Binding Protein
7.5.4. SGIV 18K
7.5.5. vIF-2α
7.5.6. Additional Studies Using Recombinant Iridovirid Proteins
8. Future Directions
Acknowledgments
Conflict of Interest
References and Notes
- Chinchar, V.G. Ranaviruses (family Iridoviridae): emerging cold-blooded killers. Arch. Virol. 2002, 147, 447–470. [Google Scholar] [CrossRef] [PubMed]
- Chinchar, V.G.; Hyatt, A.; Miyazaki, T.; Williams, T. Family Iridoviridae: poor viral relations no longer. Curr. Top. Microbiol. Immunol. 2009, 328, 123–170. [Google Scholar]
- Jancovich, J.; Chinchar, V.G.; Hyatt, A.; Miyazaki, T.; Williams, T.; Zhang, Q.-Y. Family Iridoviridae. Virus Taxonomy: 9th Report of the International Committee on Taxonomy of Viruses; King, A.M.Q, Lefkowitz, E., Adams, M.J., Carstens, E.B., Eds.; Elsevier: San Diego, CA, USA, 2011. [Google Scholar]
- Iyer, L.M.; Balaji, S.; Koonin, E.V.; Aravind, L. Evolutionary genomics of nucleo-cytoplasmic large DNA viruses. Virus Res. 2006, 117, 156–184. [Google Scholar] [CrossRef] [PubMed]
- Iyer, L.M.; Aravind, L.; Koonin, E.V. Common origin of four diverse families of large eukaryotic DNA viruses. J. Virol. 2001, 75, 11720–11734. [Google Scholar] [CrossRef]
- Yutin, N.; Koonin, E.V. Evolution of DNA ligases of nucleo-cytoplasmic large DNA viruses of eukaryotes: a case of hidden complexity. Biol. Direct 2009, 4, 51. [Google Scholar] [CrossRef] [PubMed]
- Yutin, N.; Wolf, Y.I.; Raoult, D.; Koonin, E.V. Eukaryotic large nucleo-cytoplasmic DNA viruses: Clusters of orthologous genes and reconstruction of viral genome evolution. Virol. J. 2009, 6, 223. [Google Scholar] [CrossRef]
- Song, W.J.; Qin, Q.W.; Qiu, J.; Huang, C.H.; Wang, F.; Hew, C.L. Functional genomics analysis of Singapore grouper iridovirus: Complete sequence determination and proteomic analysis. J. Virol. 2004, 78, 12576–12590. [Google Scholar] [CrossRef]
- Just, F.; Essbauer, S.; Ahne, W.; Blahak, S. Occurrence of an invertebrate iridescent-like virus (Iridoviridae) in reptiles. J. Vet. Med. B Infect. Dis. Vet. Public Health 2001, 48, 685–694. [Google Scholar] [CrossRef]
- Weinmann, N.; Papp, T.; Alves de Matos, P.; Teifke, J.B.; Marschang, R.E. Experimental infection of crickets (Gryllus bimaculatus) with an invertebrate iridovirus isolated from the high casqued chameleon (Chamaeleo hoenelii). J. VET. Diagn. Invest. 2007, 19, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Eaton, H.E.; Metcalf, J.; Penny, E.; Tcherepanov, V.; Upton, C.; Brunetti, C.R. Comparative genomic analysis of the family Iridoviridae: Re-annotating and defining the core set of iridovirus genes. Virol. J. 2007, 4, 11. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Bremont, M.; Touchman, J.W.; Jacobs, B.L. Evidence for multiple recent host species shifts among the Ranaviruses (family Iridoviridae). J. Virol. 2010, 84, 2636–2647. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.K.; Young, V.L.; Kleffmann, T.; Ward, V.K. Genomic and proteomic analysis of invertebrate iridovirus type 9. J. Virol. 2011, 85, 7900–7911. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.Y.; Jia, K.T.; Yang, B.; Huang, J. Complete genome sequence of a megalocytivirus (family Iridoviridae) associated with turbot mortality in China. Virol. J. 2010, 7, 159. [Google Scholar] [CrossRef] [PubMed]
- Williams, T. The iridoviruses. Adv. Virus Res. 1996, 46, 345–412. [Google Scholar]
- Devauchelle, G.; Stoltz, D.B.; Darcy-Tripier, F. Comparative ultrastructure of iridoviridae. Curr. Top. Microbiol. Immunol. 1985, 116, 1–21. [Google Scholar]
- Yan, X.; Yu, Z.; Zhang, P.; Battisti, A.J.; Holdaway, H.A.; Chipman, P.R.; Bajaj, C.; Bergoin, M.; Rossmann, M.G.; Baker, T.S. The capsid proteins of a large, icosahedral dsDNA virus. J. Mol. Biol. 2009, 385, 1287–1299. [Google Scholar] [CrossRef]
- Mao, J.; Tham, T.N.; Gentry, G.A.; Aubertin, A.; Chinchar, V.G. Cloning, sequence analysis, and expression of the major capsid protein of the iridovirus frog virus 3. Virology 1996, 216, 431–436. [Google Scholar] [CrossRef]
- Mao, J.; Hedrick, R.P.; Chinchar, V.G. Molecular characterization, sequence analysis, and taxonomic position of newly isolated fish iridoviruses. Virology 1997, 229, 212–220. [Google Scholar] [CrossRef]
- Whitley, D.S.; Yu, K.; Sample, R.C.; Sinning, A.; Henegar, J.; Norcross, E.; Chinchar, V.G. Frog virus 3 ORF 53R, a putative myristoylated membrane protein, is essential for virus replication in vitro. Virology 2010, 405, 448–456. [Google Scholar] [CrossRef]
- Yan, Y.; Cui, H.; Jiang, S.; Huang, Y.; Huang, X.; Wei, S.; Xu, W.; Qin, Q. Identification of a novel marine fish virus, Singapore grouper iridovirus-encoded microRNAs expressed in grouper cells by Solexa sequencing. PLoS One 2011, 6, e19148. [Google Scholar] [CrossRef]
- Majji, S.; Thodima, V.; Sample, R.; Whitley, D.; Deng, Y.; Mao, J.; Chinchar, V.G. Transcriptome analysis of Frog virus 3, the type species of the genus Ranavirus, family Iridoviridae. Virology 2009, 391, 293–303. [Google Scholar] [CrossRef]
- Teng, Y.; Hou, Z.; Gong, J.; Liu, H.; Xie, X.; Zhang, L.; Chen, X.; Qin, Q.W. Whole-genome transcriptional profiles of a novel marine fish iridovirus, Singapore grouper iridovirus (SGIV) in virus-infected grouper spleen cell cultures and in orange-spotted grouper, Epinephulus coioides. Virology 2008, 377, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.M.; Wang, F.; Song, W.; Hew, C.L. Temporal and differential gene expression of Singapore grouper iridovirus. J. Gen. Virol. 2006, 87, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.; Barbosa-Solomieu, V.; Chinchar, V.G. A decade of advances in iridovirus research. Adv. Virus Res. 2005, 65, 173–248. [Google Scholar]
- Braunwald, J.; Nonnenmacher, H.; Tripier-Darcy, F. Ultrastructural and biochemical study of frog virus 3 uptake by BHK-21 cells. J. Gen. Virol. 1985, 66, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Braunwald, J.; Tripier, F.; Kirn, A. Comparison of the properties of enveloped and naked frog virus 3 (FV3) particles. J. Gen. Virol. 1979, 45, 673–682. [Google Scholar] [CrossRef]
- Gendrault, J.L.; Steffan, A.M.; Bingen, A.; Kirn, A. Penetration and uncoating of frog virus 3 (FV3) in cultured rat Kupffer cells. Virology 1981, 112, 375–384. [Google Scholar] [CrossRef]
- Guo, C.J.; Liu, D.; Wu, Y.Y.; Yang, X.B.; Yang, L.S.; Mi, S.; Huang, Y.X.; Luo, Y.W.; Jia, K.T.; Liu, Z.Y.; et al. Entry of Tiger Frog Virus (an Iridovirus) into HepG2 Cells via a pH-Dependent, Atypical, Caveola-Mediated Endocytosis Pathway. J. Virol. 2011, 85, 6416–6426. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.G.; Barkman, T.J.; Gregory Chinchar, V.; Essani, K. Comparative genomic analyses of frog virus 3, type species of the genus Ranavirus (family Iridoviridae). Virology 2004, 323, 70–84. [Google Scholar] [CrossRef] [PubMed]
- He, J.G.; Lu, L.; Deng, M.; He, H.H.; Weng, S.P.; Wang, X.H.; Zhou, S.Y.; Long, Q.X.; Wang, X.Z.; Chan, S.M. Sequence analysis of the complete genome of an iridovirus isolated from the tiger frog. Virology 2002, 292, 185–197. [Google Scholar] [CrossRef]
- Willis, D.B.; Goorha, R.; Granoff, A. Nongenetic reactivation of frog virus 3 DNA. Virology 1979, 98, 476–479. [Google Scholar] [CrossRef] [PubMed]
- Goorha, R.; Murti, G.; Granoff, A.; Tirey, R. Macromolecular synthesis in cells infected by frog virus 3. VIII. The nucleus is a site of frog virus 3 DNA and RNA synthesis. Virology 1978, 84, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Goorha, R. Frog virus 3 requires RNA polymerase II for its replication. J. Virol. 1981, 37, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Willis, D.B.; Granoff, A. Transactivation of an immediate-early frog virus 3 promoter by a virion protein. J. Virol. 1985, 56, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Goorha, R. Frog virus 3 DNA replication occurs in two stages. J. Virol. 1982, 43, 519–528. [Google Scholar] [CrossRef]
- Goorha, R.; Granoff, A.; Willis, D.B.; Murti, K.G. The role of DNA methylation in virus replication: inhibition of frog virus 3 replication by 5-azacytidine. Virology 1984, 138, 94–102. [Google Scholar] [CrossRef]
- Willis, D.B.; Goorha, R.; Granoff, A. DNA methyltransferase induced by frog virus 3. J. Virol. 1984, 49, 86–91. [Google Scholar] [CrossRef]
- Murti, K.G.; Porter, K.R.; Goorha, R.; Ulrich, M.; Wray, G. Interaction of frog virus 3 with the cytomatrix. II. Structure and composition of the virus assembly site. Exp. Cell Res. 1984, 154, 270–282. [Google Scholar] [CrossRef]
- Murti, K.G.; Goorha, R.; Granoff, A. An unusual replication strategy of an animal iridovirus. Adv. Virus Res. 1985, 30, 1–19. [Google Scholar]
- Suarez, C.; Salas, M.L.; Rodriguez, J.M. African swine fever virus polyprotein pp62 is essential for viral core development. J. Virol. 2010, 84, 176–187. [Google Scholar] [CrossRef]
- Andres, G.; Alejo, A.; Salas, J.; Salas, M.L. African swine fever virus polyproteins pp220 and pp62 assemble into the core shell. J. Virol. 2002, 76, 12473–12482. [Google Scholar] [CrossRef]
- Goorha, R.; Murti, K.G. The genome of frog virus 3, an animal DNA virus, is circularly permuted and terminally redundant. Proc. Natl. Acad. Sci. U. S. A. 1982, 79, 248–252. [Google Scholar] [CrossRef]
- Yang, K.; Baines, J.D. Tryptophan residues in the portal protein of herpes simplex virus 1 critical to the interaction with scafford protreins and incorporation of the portal into capsids. J. Virol. 2009, 83, 11726–11733. [Google Scholar] [CrossRef]
- Chang, J.T.; Schmid, M.F.; Rixon, F.J.; Chiu, W. Electron cryotomography reveals the portal in the herpesvirus capsid. J. Virol. 2007, 81, 2065–2068. [Google Scholar] [CrossRef]
- Zauberman, N.; Mutsafi, Y.; Ben Halevy, D.; Shimoni, E.; Klein, E.; Xiao, C.Y.; Sun, S.; Minsky, A. Distinct DNA exit and packaging portals in the virus Acanthamoeba polyphaga mimivirus. PLoS Biol. 2008, 6, e114. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Granoff, A. Temperature-sensitive mutants of frog virus 3: biochemical and genetic characterization. J. Virol. 1986, 58, 192–202. [Google Scholar] [CrossRef]
- Kim, Y.S.; Ke, F.; Lei, X.Y.; Zhu, R.; Zhang, Q.Y. Viral envelope protein 53R gene highly specific silencing and iridovirus resistance in fish cells by AmiRNA. PLoS One 2010, 5, e10308. [Google Scholar] [CrossRef]
- Zhao, Z.; Ke, F.; Huang, Y.H.; Zhao, J.G.; Gui, J.F.; Zhang, Q.Y. Identification and characterization of a novel envelope protein in Rana grylio virus. J. Gen. Virol. 2008, 89, 1866–1872. [Google Scholar] [CrossRef] [PubMed]
- Chinchar, V.G.; Granoff, A. Isolation and characterization of a frog virus 3 variant resistant to phosphonoacetate: genetic evidence for a virus-specific DNA polymerase. Virology 1984, 138, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Ke, F.; Gui, J.; Zhang, Q. Characterization of an early gene encoding for dUTPase in Rana grylio virus. Virus Res. 2007, 123, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Sample, R.; Bryan, L.; Long, S.; Majji, S.; Hoskins, G.; Sinning, A.; Olivier, J.; Chinchar, V.G. Inhibition of iridovirus protein synthesis and virus replication by antisense morpholino oligonucleotides targeted to the major capsid protein, the 18 kDa immediate-early protein, and a viral homolog of RNA polymerase II. Virology 2007, 358, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Su, H.-P.; Lin, D.Y.-W.; Garboczi, D.N. The structure of G4, the poxvirus disulfide oxidoreductase essential for virus maturation and infectivity. J. Virol. 2006, 80, 7706–7713. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Robert, J.; Storfer, A.T. Ecology of viruses infecting ectothermic vertebrates - the impact of ranavirus infections on amphibians. In Studies in Viral Ecology; Hurst, C.J., Ed.; Wiley-Blackwell: Hoboken, New Jersey, USA, 2011; Volume 2, pp. 231–260. [Google Scholar]
- Raghow, R.; Granoff, A. Macromolecular synthesis in cells infected by frog virus 3. X. Inhibition of cellular protein synthesis by heat-inactivated virus. Virology 1979, 98, 319–327. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Dholakia, J.N. Frog virus 3-induced translational shut-off: activation of an eIF-2 kinase in virus-infected cells. Virus Res. 1989, 14, 207–223. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Yu, W. Frog virus 3-mediated translational shut-off: frog virus 3 messages are translationally more efficient than host and heterologous viral messages under conditions of increased translational stress. Virus Res. 1990, 16, 163–174. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Bryan, L.; Wang, J.; Long, S.; Chinchar, G.D. Induction of apoptosis in frog virus 3-infected cells. Virology 2003, 306, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Chinchar, V.G.; Caughman, G.B. Heat-inactivated frog virus 3 selectively inhibits equine herpesvirus type 1 translation in a temporal class-dependent manner. Virology 1986, 152, 466–471. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Yu, W. Translational efficiency: iridovirus early mRNAs outcompete tobacco mosaic virus message in vitro. Biochem. Biophys. Res. Commun. 1990, 172, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Rothenburg, S.; Chinchar, V.G.; Dever, T.E. Characterization of a ranavirus inhibitor of the antiviral protein kinase PKR. BMC Microbiol. 2011, 11, 56. [Google Scholar] [CrossRef] [PubMed]
- Langland, J.O.; Jacobs, B.L. The role of the PKR-inhibitory genes, E3L and K3L, in determining vaccinia virus host range. Virology 2002, 299, 133–141. [Google Scholar] [CrossRef]
- Majji, S.; LaPatra, S.; Long, S.M.; Sample, R.; Bryan, L.; Sinning, A.; Chinchar, V.G. Rana catesbeiana virus Z (RCV-Z): a novel pathogenic ranavirus. Dis. Aquat. Org. 2006, 73, 1–11. [Google Scholar] [CrossRef]
- Essbauer, S.; Bremont, M.; Ahne, W. Comparison of the eIF-2alpha homologous proteins of seven ranaviruses (Iridoviridae). Virus Genes 2001, 23, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Reading, P.C.; Moore, J.B.; Smith, G.L. Steroid hormone synthesis by vaccinia virus suppresses the inflammatory response to infection. J. Exp. Med. 2003, 197, 1269–1278. [Google Scholar] [CrossRef]
- Moore, J.B.; Smith, G.L. Steroid hormone synthesis by a vaccinia enzyme: a new type of virus virulence factor. EMBO J. 1992, 11, 3490. [Google Scholar] [CrossRef] [PubMed]
- Besch, R.; Poeck, H.; Hohenauer, T.; Senft, D.; Hacker, G.; Berking, C.; Hornung, V.; Endres, S.; Ruzicka, T.; Rothenfusser, S.; et al. Proapoptotic signaling induced by RIG-I and MDA-5 results in type I interferon-independent apoptosis in human melanoma cells. J. Clin. Investig. 2009, 119, 2399–2411. [Google Scholar] [CrossRef] [PubMed]
- Bouchier-Hayes, L.; Hartin, S.J. CARD games in apoptosis and immunity. EMBO Rep. 2002, 3, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, K.; Bucher, P.; Tschopp, J. The CARD domain: a new apoptotic signalling motif. Trends Biochem. Sci. 1997, 22, 155–156. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Moore, C.B.; Liesman, R.M.; O'Connor, B.P.; Bergstralh, D.T.; Chen, Z.J.; Pickles, R.J.; Ting, J.P. MAVS-mediated apoptosis and its inhibition by viral proteins. PLoS One 2009, 4, e5466. [Google Scholar] [CrossRef]
- Yoneyama, M.; Kikuchi, M.; Natsukawa, T.; Shinobu, N.; Imaizumi, T.; Miyagishi, M.; Taira, K.; Akira, S.; Fujita, T. The RNA helicase RIG-I has an essential function in double-stranded RNA-induced innate antiviral responses. Nat. Immunol. 2004, 5, 730–737. [Google Scholar] [CrossRef]
- Hausmann, S.; Marq, J.B.; Tapparel, C.; Kolakofsky, D.; Garcin, D. RIG-I and dsRNA-induced IFNbeta activation. PLoS One 2008, 3, e3965. [Google Scholar] [CrossRef]
- Levy, D.E.; Marie, I.J. RIGging an antiviral defense―It's in the CARDs. Nat. Immunol. 2004, 5, 699–701. [Google Scholar] [CrossRef]
- Meylan, E.; Curran, J.; Hofmann, K.; Moradpour, D.; Binder, M.; Bartenschlager, R.; Tschopp, J. Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature 2005, 437, 1167–1172. [Google Scholar] [CrossRef]
- Zenke, K.; Kim, K.H. Functional characterization of the RNase III gene of rock bream iridovirus. Arch. Virol. 2008, 153, 1651–1656. [Google Scholar] [CrossRef]
- Johnston, J.B.; McFadden, G. Poxvirus immunomodulatory strategies: Current perspectives. J. Virol. 2003, 77, 6093–6100. [Google Scholar] [CrossRef]
- Seet, B.T.; Johnston, J.B.; Brunetti, C.R.; Barrett, J.W.; Everett, H.; Cameron, C.; Sypula, J.; Nazarian, S.H.; Lucas, A.; McFadden, G. Poxviruses and immune evasion. Annu. Rev. Immunol. 2003, 21, 377–423. [Google Scholar] [CrossRef]
- Barrett, J.W.; Alston, L.R.; Wang, F.; Stanford, M.M.; Gilbert, P.A.; Gao, X.; Jimenez, J.; Villeneuve, D.; Forsyth, P.; McFadden, G. Identification of host range mutants of myxoma virus with altered oncolytic potential in human glioma cells. J. Neurovirol. 2007, 13, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Daikoku, T.; Yamamoto, N.; Maeno, K.; Nishiyama, Y. Role of viral ribonucleotide reductase in the increase of dTTP pool size in herpes simplex virus-infected Vero cells. J. Gen. Virol. 1991, 72, 1441–1444. [Google Scholar] [CrossRef] [PubMed]
- de Wind, N.; Berns, A.; Gielkens, A.; Kimman, T. Ribonucleotide reductase-deficient mutants of pseudorabies virus are avirulent for pigs and induce partial protective immunity. J. Gen. Virol. 1993, 74, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Oliveros, M.; Garcia-Escudero, R.; Alejo, A.; Vinuela, E.; Salas, M.L.; Salas, J. African swine fever virus dUTPase is a highly specific enzyme required for efficient replication in swine macrophages. J. Virol. 1999, 73, 8934–8943. [Google Scholar] [CrossRef]
- Ting, J.W.; Wu, M.F.; Tsai, C.T.; Lin, C.C.; Guo, I.C.; Chang, C.Y. Identification and characterization of a novel gene of grouper iridovirus encoding a purine nucleoside phosphorylase. J. Gen. Virol. 2004, 85, 2883–2892. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Mao, J.; Chinchar, V.G.; Wyatt, C.; Case, S.T.; Kumar, S.; Valente, G.; Subramanian, S.; Davidson, E.W.; Collins, J.P.; et al. Genomic sequence of a ranavirus (family Iridoviridae) associated with salamander mortalities in North America. Virology 2003, 316, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Buller, R.M.; Arif, B.M.; Black, D.M.; Dumbell, K.R.; Esposito, J.J.; Lefkowitz, E.J.; McFadden, G.; Moss, B.; Mercer, A.A.; Moyer, R.M.; et al. Poxviridae. Virus Taxonomy: Eighth Report of the International Committee on Taxonomy of Viruses; Fauquet, C.M., Mayo, M.A., Maniloff, J., Desselberger, U., Ball, L.A., Eds.; Elsevier: San Diego, CA, USA, 2005; pp. 117–133. [Google Scholar]
- Lefkowitz, E.J.; Wang, C.; Upton, C. Poxviruses: past, present, and future. Virus Res. 2006, 117, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Mezencev, R.; Mereish, K. How similar are poxviruses? Science 2005, 308, 1259–1260. [Google Scholar] [CrossRef]
- Bandin, I.; Dopazo, C.P. Host range, host specificity and hypothesized host shift events among viruses of lower vertebrates. Vet. Res. 2011, 42, 67. [Google Scholar] [CrossRef] [PubMed]
- Schock, D.M.; Bollinger, T.K.; Chinchar, V.G.; Jancovich, J.K.; Collins, J.P. Experimental evidence that amphibian ranaviruses are multihost pathogens. Coepia 2008, 133–143. [Google Scholar] [CrossRef]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Robert, J. Antiviral Immunity in Amphibians. Viruses submitted for publication. 2011. [Google Scholar] [CrossRef]
- Miller, D.B.; Gray, M.; Storfer, A.T. Ecopathology of Ranaviruses Infecting Amphibians. Viruses submitted for publication. 2011. [Google Scholar] [CrossRef]
- Walker, P.J.; Winton, J.R. Emerging viral diseases of fish and shrimp. Vet. Res. 2010, 41, 51. [Google Scholar] [CrossRef]
- Whittington, R.J.; Becker, J.A.; Dennis, M.M. Iridovirus infections in finfish—Critical review with emphasis on ranaviruses. J. Fish Dis. 2010, 33, 95–122. [Google Scholar] [CrossRef]
- Mazzoni, R.; de Mesquita, A.J.; Fleury, L.F.; de Brito, W.M.; Nunes, I.A.; Robert, J.; Morales, H.; Coelho, A.S.; Barthasson, D.L.; Galli, L.; et al. Mass mortality associated with a frog virus 3-like Ranavirus infection in farmed tadpoles Rana catesbeiana from Brazil. Dis. Aquat. Org. 2009, 86, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Gray, M.J.; Miller, D.L.; Hoverman, J.T. Ecology and pathology of amphibian ranaviruses. Dis. Aquat. Org. 2009, 87, 243–266. [Google Scholar] [CrossRef]
- Gantress, J.; Maniero, G.D.; Cohen, N.; Robert, J. Development and characterization of a model system to study amphibian immune responses to iridoviruses. Virology 2003, 311, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Maniero, G.D.; Morales, H.; Gantress, J.; Robert, J. Generation of a long-lasting, protective, and neutralizing antibody response to the ranavirus FV3 by the frog Xenopus. Dev. Comp. Immunol. 2006, 30, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Morales, H.D.; Abramowitz, L.; Gertz, J.; Sowa, J.; Vogel, A.; Robert, J. Innate immune responses and permissiveness to ranavirus infection of peritoneal leukocytes in the frog Xenopus laevis. J. Virol. 2010, 84, 4912–4922. [Google Scholar] [CrossRef]
- Morales, H.D.; Robert, J. Characterization of primary and memory CD8 T-cell responses against ranavirus (FV3) in Xenopus laevis. J. Virol. 2007, 81, 2240–2248. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.; Abramowitz, L.; Gantress, J.; Morales, H.D. Xenopus laevis: A possible vector of Ranavirus infection? J. Wildl. Dis. 2007, 43, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.; George, E.; De Jesus Andino, F.; Chen, G. Waterborne infectivity of the Ranavirus frog virus 3 in Xenopus laevis. Virology 2011, 417, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.; Morales, H.; Buck, W.; Cohen, N.; Marr, S.; Gantress, J. Adaptive immunity and histopathology in frog virus 3-infected Xenopus. Virology 2005, 332, 667–675. [Google Scholar] [CrossRef]
- Caipang, C.M.; Hirono, I.; Aoki, T. Immunogenicity, retention and protective effects of the protein derivatives of formalin-inactivated red seabream iridovirus (RSIV) vaccine in red seabream, Pagrus major. Fish Shellfish Immunol. 2006, 20, 597–609. [Google Scholar] [CrossRef]
- Caipang, C.M.; Takano, T.; Hirono, I.; Aoki, T. Genetic vaccines protect red seabream, Pagrus major, upon challenge with red seabream iridovirus (RSIV). Fish Shellfish Immunol. 2006, 21, 130–138. [Google Scholar] [CrossRef]
- Green, D.E.; Converse, K.A.; Schrader, A.K. Epizootiology of sixty-four amphibian morbidity and mortality events in the USA 1996–2001. Ann. N. Y. Acad. Sci. 2002, 969, 323–339. [Google Scholar] [CrossRef] [PubMed]
- Cullen, B.R.; Owens, L. Experimental challenge and clinical cases of Bohle iridovirus (BIV) in native Australian anurans. Dis. Aquat. Org. 2002, 49, 83–92. [Google Scholar] [CrossRef]
- Mao, J.; Green, D.E.; Fellers, G.; Chinchar, V.G. Molecular characterization of iridoviruses isolated from sympatric amphibians and fish. Virus Res. 1999, 63, 45–52. [Google Scholar] [CrossRef]
- Jensen, B.B.; Ersboll, A.K.; Ariel, E. Susceptibility of pike Esox lucius to a panel of Ranavirus isolates. Dis. Aquat. Org. 2009, 83, 169–179. [Google Scholar] [CrossRef]
- Moody, N.J.G.; Owens, L. Experimental demonstration of pathogenicity of a frog virus, Bohle iridovirus, for a fish species, barramundi Lates Calcarifer. Dis. Aquat. Org. 1994, 18, 95–102. [Google Scholar] [CrossRef]
- Willis, D.B.; Goorha, R.; Chinchar, V.G. Macromolecular synthesis in cells infected by frog virus 3. Curr. Top. Microbiol. Immunol. 1985, 116, 77–106. [Google Scholar] [PubMed]
- Goorha, R.; Granoff, A. Icosahedral cytoplasmic deoxyriboviruses. Comprehensive Virology; Fraenkel-Conrat, H., Wagner, R.R., Eds.; Plenum Press: New York, NY, USA, 1979; Volume 14, pp. 347–399. [Google Scholar]
- Naegele, R.F.; Granoff, A. Viruses and renal carcinoma of Rana pipiens. XI. Isolation of frog virus 3 temperature-sensitive mutants; complementation and genetic recombination. Virology 1971, 44, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Purifoy, D.; Naegele, R.F.; Granoff, A. Viruses and renal carcinoma of Rana pipiens. XIV. Temperature-sensitive mutants of frog virus 3 with defective encapsidation. Virology 1973, 54, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Goorha, R.; Willis, D.B.; Granoff, A.; Naegele, R.F. Characterization of a temperature-sensitive mutant of frog virus 3 defective in DNA replication. Virology 1981, 112, 40–48. [Google Scholar] [CrossRef]
- Sample, R.C. Elucidation of Frog Virus 3 Gene Function and Pathways of Virion Formation. Ph.D. Thesis, University of Mississippi Medical Center, Jackson, MS, UA, 2010. [Google Scholar]
- Chinchar, V.G.; Goorha, R.; Granoff, A. Early proteins are required for the formation of frog virus 3 assembly sites. Virology 1984, 135, 148–156. [Google Scholar] [CrossRef]
- Dagle, J.M.; Weeks, D.L. Oligonucleotide-based strategies to reduce gene expression. Differentiation 2001, 69, 75–82. [Google Scholar] [CrossRef]
- Hardy, S.; Legagneux, V.; Audic, Y.; Paillard, L. Reverse genetics in eukaryotes. Biol. Cell. 2010, 102, 561–580. [Google Scholar] [CrossRef]
- Neuman, B.W.; Stein, D.A.; Kroeker, A.D.; Paulino, A.D.; Moulton, H.M.; Iversen, P.L.; Buchmeier, M.J. Antisense morpholino-oligomers directed against the 5' end of the genome inhibit coronavirus proliferation and growth. J. Virol. 2004, 78, 5891–5899. [Google Scholar] [CrossRef] [PubMed]
- Deas, T.S.; Binduga-Gajewska, I.; Tilgner, M.; Ren, P.; Stein, D.A.; Moulton, H.M.; Iversen, P.L.; Kauffman, E.B.; Kramer, L.D.; Shi, P.Y. Inhibition of flavivirus infections by antisense oligomers specifically suppressing viral translation and RNA replication. J. Virol. 2005, 79, 4599–4609. [Google Scholar] [CrossRef]
- Lai, S.H.; Stein, D.A.; Guerrero-Plata, A.; Liao, S.L.; Ivanciuc, T.; Hong, C.; Iversen, P.L.; Casola, A.; Garofalo, R.P. Inhibition of respiratory syncytial virus infections with morpholino oligomers in cell cultures and in mice. Mol. Ther. 2008, 16, 1120–1128. [Google Scholar] [CrossRef]
- Whitley, D.S.; Sample, R.C.; Sinning, A.R.; Henegar, J.; Chinchar, V.G. Antisense approaches for elucidating ranavirus gene function in an infected fish cell line. Dev. Comp. Immunol. 2011, 35, 937–948. [Google Scholar] [CrossRef]
- Yu, E.K.; Whitley, D.S.; Chinchar, V.G. Department of Microbiology, University of Mississippi Medical Center, Jackson, MS. Unpublished work, 2011.
- Dang, L.T.; Kondo, H.; Hirono, I.; Aoki, T. Inhibition of red seabream iridovirus (RSIV) replication by small interfering RNA (siRNA) in a cell culture system. Antivir. Res. 2008, 77, 142–149. [Google Scholar] [CrossRef]
- Xie, J.; Lu, L.; Deng, M.; Weng, S.; Zhu, J.; Wu, Y.; Gan, L.; Chan, S.M.; He, J. Inhibition of reporter gene and Iridovirus-tiger frog virus in fish cell by RNA interference. Virology 2005, 338, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Abrahim, A.M.; Asgari, S. An ascovirus-encoded RNase III autoregulates its expression and suppresses RNA interference-mediated gene silencing. J. Virol. 2010, 84, 3624–3630. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, S.; Brechling, K.; Moss, B. Vaccinia virus expression vector: Coexpression of beta-galactosidase provides visual screening of recombinant virus plaques. Mol. Cell. Biol. 1985, 5, 3403–3409. [Google Scholar] [CrossRef] [PubMed]
- Franke, C.A.; Rice, C.M.; Strauss, J.H.; Hruby, D.E. Neomycin resistance as a dominant selectable marker for selection and isolation of vaccinia virus recombinants. Mol. Cell. Biol. 1985, 5, 1918–1924. [Google Scholar]
- Ward, B.M.; Weisberg, A.S.; Moss, B. Mapping and functional analysis of internal deletions within the cytoplasmic domains of the vaccinia virus A33R and A36R entry proteins J. Virol. 2003, 77, 4113–4126. [Google Scholar] [CrossRef]
- Pallister, J.; Goldie, S.; Coupar, B.; Hyatt, A. Promoter activity in the 5' flanking regions of the Bohle iridovirus ICP 18, ICP 46 and major capsid protein genes. Arch. Virol. 2005, 150, 1911–1919. [Google Scholar] [CrossRef]
- Pallister, J.; Goldie, S.; Coupar, B.; Shiell, B.; Michalski, W.P.; Siddon, N.; Hyatt, A. Bohle iridovirus as a vector for heterologous gene expression. J. Virol. Methods 2007, 146, 419–423. [Google Scholar] [CrossRef]
- Jancovich, J.K.; Jacobs, B.L. Innate immune evasion mediated by the Ambystoma tigrinum virus eukaryotic translation initiation factor 2 alpha homologue. J. Virol. 2011, 85, 5061–5069. [Google Scholar] [CrossRef]
- Samuel, C.E. The eIF-2 alpha protein kinases, regulators of translation in eukaryotes from yeast to humans. J. Biol. Chem. 1993, 268, 7603–7606. [Google Scholar] [CrossRef] [PubMed]
- Langland, J.O.; Cameron, J.M.; Heck, M.C.; Jancovich, J.K.; Jacobs, B.L. Inhibition of PKR by RNA and DNA viruses. Virus Res. 2006, 119, 100–110. [Google Scholar] [CrossRef]
- Chen, G.; Ward, B.M.; Yu, E.K.; Chinchar, V.G.; Robert, J. Improved knockout methodology reveals that Frog virus 3 mutants lacking either the 18K immediate-early gene or the truncated vIF-2 alpha gene are defective for replication in vivo. J. Virol. 2011, 85, 11131–1138. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Huang, X.; Cai, J.; Ye, F.; Guan, L.; Liu, H.; Qin, Q. Construction of green fluorescent protein-tagged recombinant iridovirus to assess viral replication. Virus Res. 2011, 160, 221–229. [Google Scholar] [CrossRef]
- Xia, L.; Liang, H.; Huang, Y.; Ou-Yang, Z.; Qin, Q. Identification and characterization of Singapore grouper iridovirus (SGIV) ORF162L, an immediate-early gene involved in cell growth control and viral replication. Virus Res. 2010, 147, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Bi, X.; Chen, L.M.; Hew, C.L. ORF018R, a highly abundant virion protein from Singapore grouper iridovirus, is involved in serine/threonine phosphorylation and virion assembly. J. Gen. Virol. 2008, 89, 1169–1178. [Google Scholar] [CrossRef]
- Tran, B.N.; Chen, L.; Liu, Y.; Wu, J.; Velazquez Campoy, A.; Sivaraman, J.; Hew, C.L. A Novel Histone H3 Binding Protein ORF158L from the Singapore Grouper Iridovirus (SGIV). J. Virol. 2011, 85, 9195–9166. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Cao, J.; Huang, X.; Qin, Q. Characterization of Singapore grouper iridovirus (SGIV) ORF086R, a putative homolog of ICP18 involved in cell growth control and virus replication. Arch. Virol. 2009, 154, 1409–1416. [Google Scholar] [CrossRef] [PubMed]
- Ke, F.; Zhao, Z.; Zhang, Q. Cloning, expression and subcellular distribution of a Rana grylio virus late gene encoding ERV1 homologue. Mol. Biol. Rep. 2009, 36, 1651–1659. [Google Scholar] [CrossRef]
- Huang, X.; Huang, Y.; Gong, J.; Yan, Y.; Qin, Q. Identification and characterization of a putative lipopolysaccharide-induced TNF-alpha factor (LITAF) homolog from Singapore grouper iridovirus. Biochem. Biophys. Res. Commun. 2008, 373, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Shi, Y.; Ke, F.; Wei, S.; Gui, J.; Zhang, Q. Constitutive expression of thymidylate synthase from LCDV-C induces a transformed phenoptype in fish cells. Virology 2008, 372, 118–126. [Google Scholar] [CrossRef]
- Gray, M. Global Ranavirus Consortium. Available online: http://fwf.ag.utk.edu/mgray/ranavirus/Ranavirus.htm (accessed on 1 September 2011).

| Genus | Speciesa | Size (bp) | No. ORFsb | % G+C | GenBank Accession Number |
|---|---|---|---|---|---|
| Iridovirus | IIV-9 | 206,791 | 191 | 31 | GQ918152 |
| IIV-6 | 212,482 | 211 | 29 | AF303741 | |
| Chloriridovirus | IIV-3 | 191,132 | 126 | 48 | DQ643392 |
| Lymphocystivirus | LCDV-1 | 102,653 | 108 | 29 | L63545 |
| LCDV-C | 186,250 | 178 | 27 | AY380826 | |
| Ranavirus | TFV | 105,057 | 105 | 55 | AF389451 |
| ATV | 106,332 | 92 | 54 | AY150217 | |
| FV3 | 105,903 | 97 | 55 | AY548484 | |
| STIV | 105,890 | 103 | 55 | EU627010 | |
| EHNV | 127,011 | 100 | 54 | FJ433873 | |
| SGIV | 140,131 | 139 | 49 | AY521625 | |
| GIV | 139,793 | 139 | 49 | AY666015 | |
| Megalocytivirus | ISKNV | 111,362 | 117 | 55 | AF371960 |
| RBIV | 112,080 | 116 | 53 | AY532606 | |
| RSIV | 112,414 | 93 | 53 | BD143114 | |
| OSGIV | 112,636 | 116 | 54 | AY894343 | |
| TRBIV | 110,104 | 115 | 55 | GO273492 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2011 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chinchar, V.G.; Yu, K.H.; Jancovich, J.K. The Molecular Biology of Frog Virus 3 and other Iridoviruses Infecting Cold-Blooded Vertebrates. Viruses 2011, 3, 1959-1985. https://doi.org/10.3390/v3101959
Chinchar VG, Yu KH, Jancovich JK. The Molecular Biology of Frog Virus 3 and other Iridoviruses Infecting Cold-Blooded Vertebrates. Viruses. 2011; 3(10):1959-1985. https://doi.org/10.3390/v3101959
Chicago/Turabian StyleChinchar, V. Gregory, Kwang H. Yu, and James K. Jancovich. 2011. "The Molecular Biology of Frog Virus 3 and other Iridoviruses Infecting Cold-Blooded Vertebrates" Viruses 3, no. 10: 1959-1985. https://doi.org/10.3390/v3101959
APA StyleChinchar, V. G., Yu, K. H., & Jancovich, J. K. (2011). The Molecular Biology of Frog Virus 3 and other Iridoviruses Infecting Cold-Blooded Vertebrates. Viruses, 3(10), 1959-1985. https://doi.org/10.3390/v3101959




