Use of the Slow-Delivery Platform, VacSIM, Shapes the Host Immune Response to Increase Protection Against Influenza Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Protein Purification
2.3. Virus Growth and Titering
2.4. Hemagglutination Assays (HAs)
2.5. Influenza Challenge Readouts
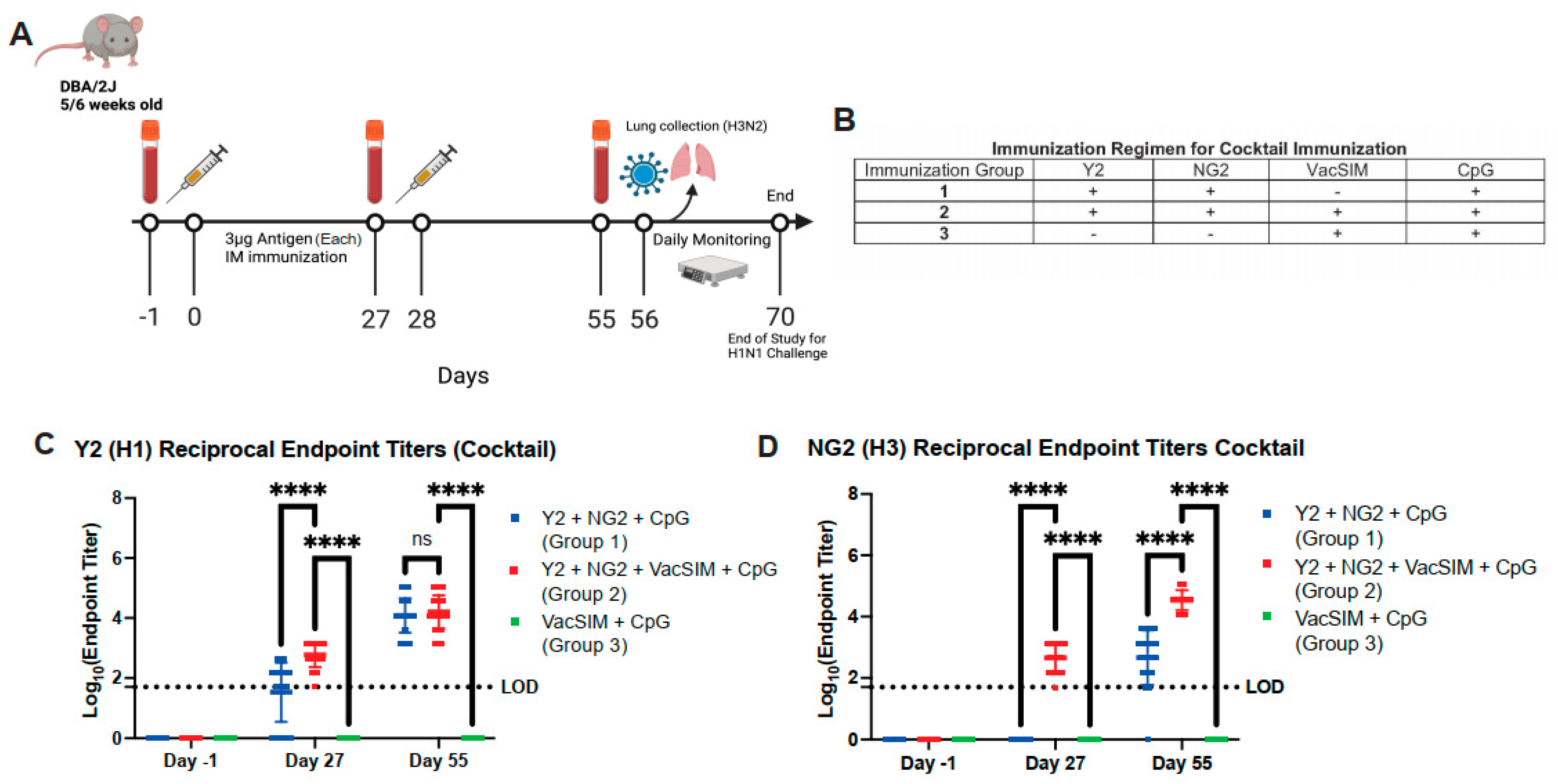
2.6. Mouse Experiments
2.7. Analysis of Lung Viral Titers
2.8. Endpoint Titer Experiments
2.9. Hemagglutination Inhibition Assays (HAIs)
2.10. Quantification and Statistical Analysis
3. Results
3.1. Vaccination with VacSIM and HA Increases Antigen-Specific IgG Antibody Titers
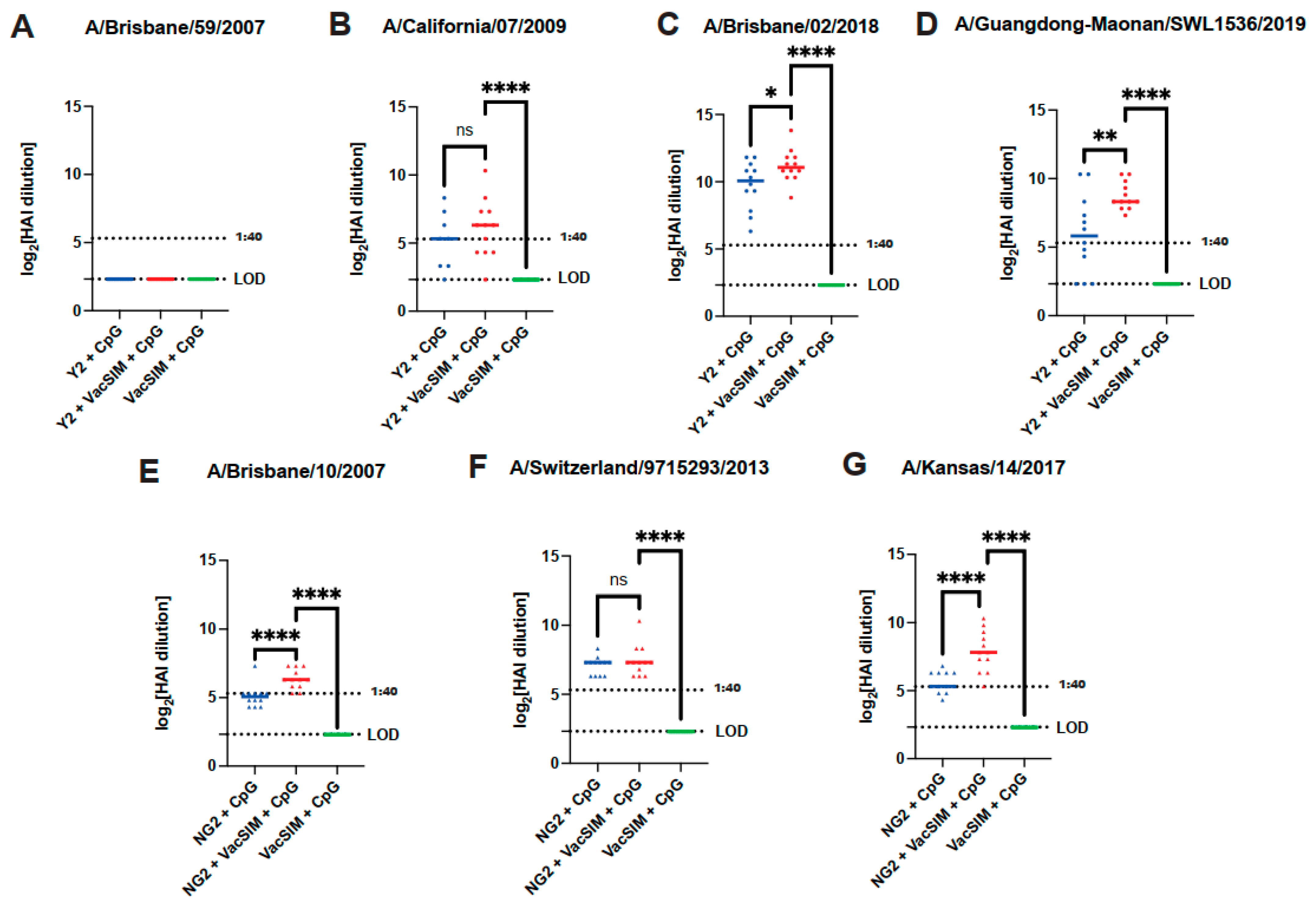
3.2. Vaccination with VacSIM and HA Increases HAI Titers Across Seasonal Influenza Viruses
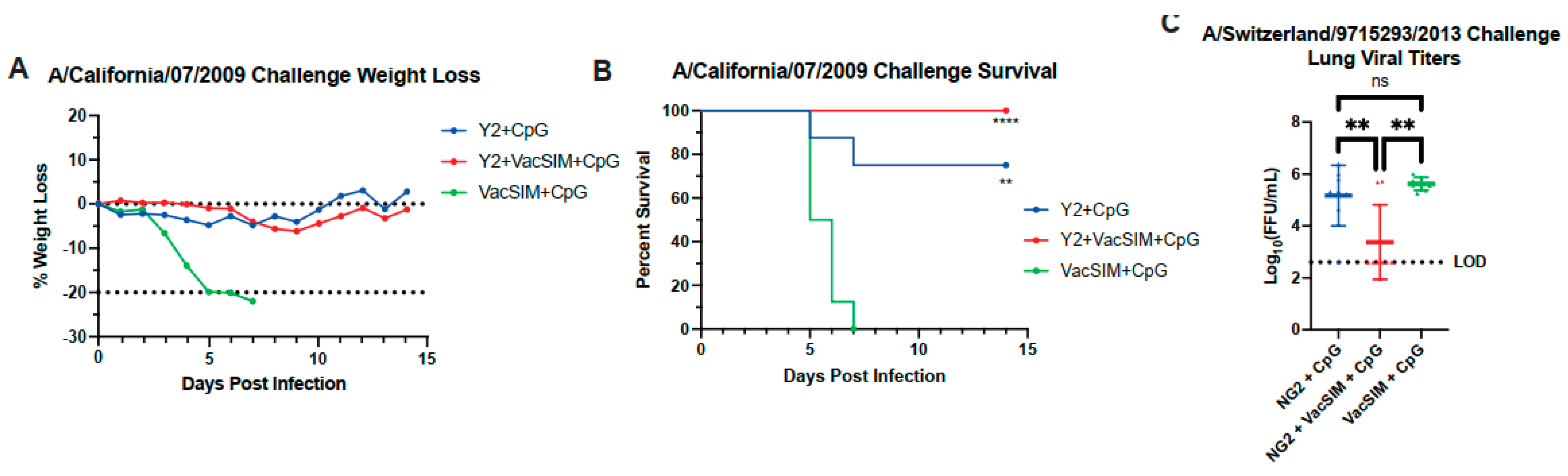
3.3. Vaccination with VacSIM and HA Improves Protection Against Influenza Infection
3.4. Vaccination with VacSIM and an HA Cocktail Increases Antigen-Specific IgG Antibody Titers Against Both Antigens
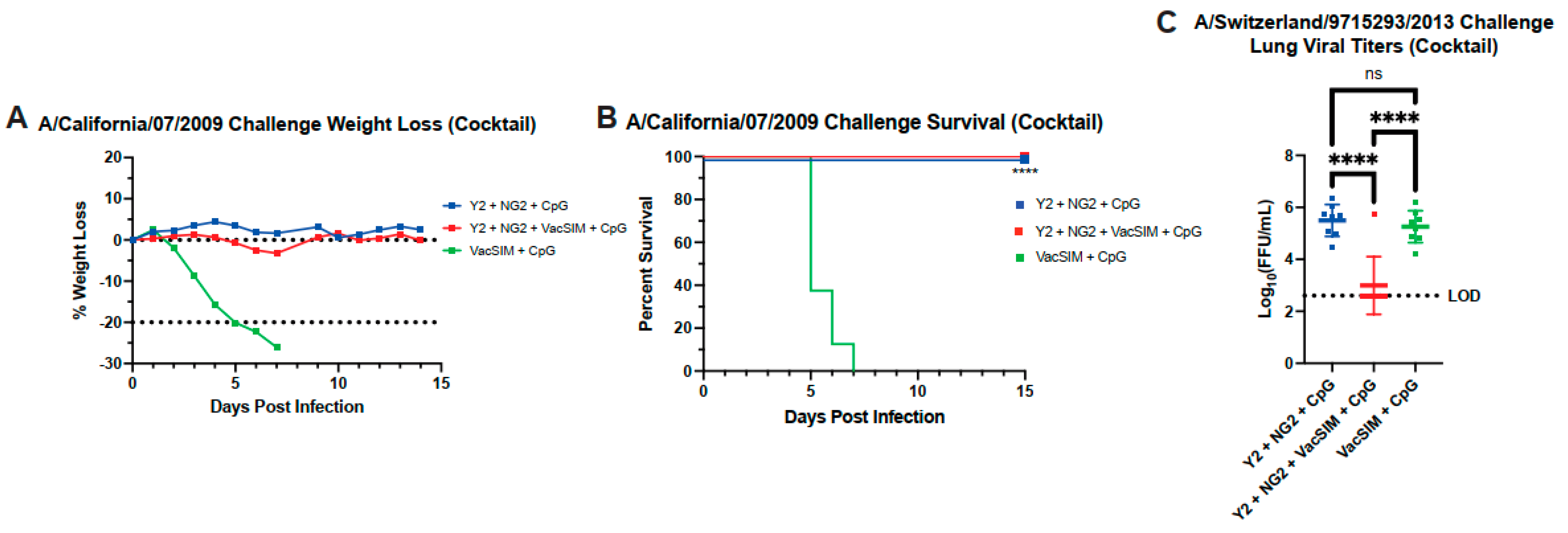
3.5. Vaccination with VacSIM and HA Cocktail Improves Protection Against Both H1N1 and H3N2 Influenza Challenge
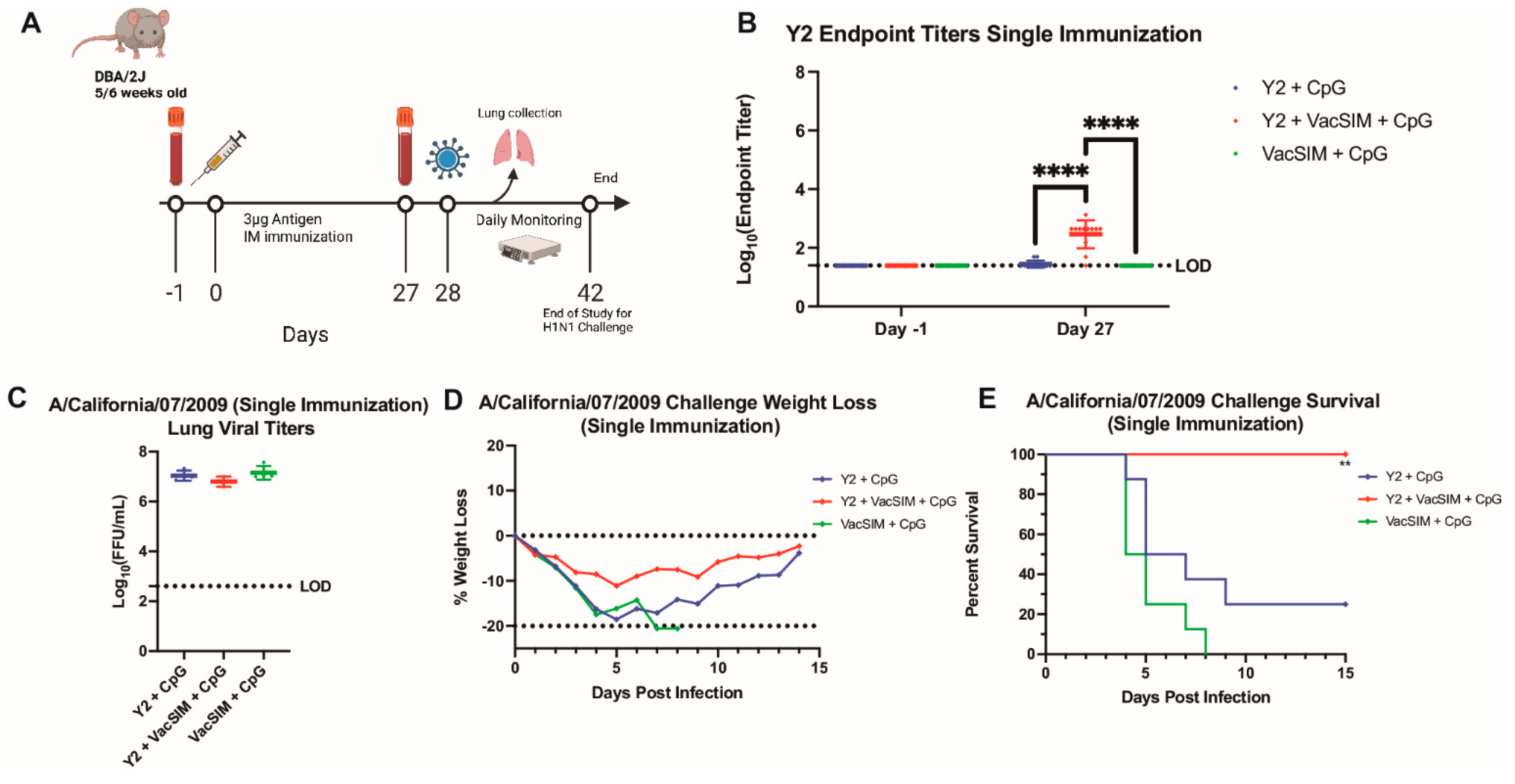
3.6. Single Vaccination with VacSIM and Y2 Increases Antigen-Specific Antibody Titers and Protects Against Lethal H1N1 Infection
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HA | hemagglutinin |
| NA | neuraminidase |
| VacSIM | Vaccine Self-Assembling Immune Matrix |
| COBRA | computationally optimized broadly reactive antigen |
| HAI | hemagglutination inhibition assay |
References
- Mathews, J.D.; Chesson, J.M.; Mccaw, J.M.; Mcvernon, J. Understanding Influenza Transmission, Immunity and Pandemic Threats. Influenza Other Respir Viruses 2009, 3, 143–149. [Google Scholar] [CrossRef]
- Moghadami, M. A Narrative Review of Influenza: A Seasonal and Pandemic Disease. Iran. J. Med. Sci. 2017, 42, 2–13. [Google Scholar]
- Treanor, J.J. CLINICAL PRACTICE. Influenza Vaccination. N. Engl. J. Med. 2016, 375, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Mohan, S. Influenza Vaccine: A Review on Current Scenario and Future Prospects. J. Genet. Eng. Biotechnol. 2023, 21, 154. [Google Scholar] [CrossRef] [PubMed]
- Houser, K.; Subbarao, K. Influenza Vaccines: Challenges and Solutions. Cell Host Microbe 2015, 17, 295–300. [Google Scholar] [CrossRef]
- Wong, S.S.; Webby, R.J. Traditional and New Influenza Vaccines. Clin. Microbiol. Rev. 2013, 26, 476–492. [Google Scholar] [CrossRef]
- Johansson, B.E.; Bucher, D.J.; Kilbourne, E.D. Purified Influenza Virus Hemagglutinin and Neuraminidase Are Equivalent in Stimulation of Antibody Response but Induce Contrasting Types of Immunity to Infection. J. Virol. 1989, 63, 1239–1246. [Google Scholar] [CrossRef]
- Byrd-Leotis, L.; Cummings, R.D.; Steinhauer, D.A. The Interplay between the Host Receptor and Influenza Virus Hemagglutinin and Neuraminidase. Int. J. Mol. Sci. 2017, 18, 1541. [Google Scholar] [CrossRef]
- Gamblin, S.J.; Skehel, J.J. Influenza Hemagglutinin and Neuraminidase Membrane Glycoproteins. J. Biol. Chem. 2010, 285, 28403–28409. [Google Scholar] [CrossRef]
- McMillan, C.L.D.; Young, P.R.; Watterson, D.; Chappell, K.J. The next Generation of Influenza Vaccines: Towards a Universal Solution. Vaccines 2021, 9, 26. [Google Scholar] [CrossRef]
- Abbadi, N.; Nagashima, K.; Pena-Briseno, A.; Ross, T.M.; Mousa, J.J. Differential Recognition of Computationally Optimized H3 Hemagglutinin Influenza Vaccine Candidates by Human Antibodies. J. Virol. 2022, 96, e0089622. [Google Scholar] [CrossRef]
- Shasha, D.; Valinsky, L.; Hershkowitz Sikron, F.; Glatman-Freedman, A.; Mandelboim, M.; Toledano, A.; Paran, Y.; Ben-Ami, R.; Goldman, D. Quadrivalent versus Trivalent Influenza Vaccine: Clinical Outcomes in Two Influenza Seasons, Historical Cohort Study. Clin. Microbiol. Infect. 2020, 26, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Carregaro, R.L.; Roscani, A.N.C.P.; Raimundo, A.C.S.; Ferreira, L.; Vanni, T.; da Graça Salomão, M.; Probst, L.F.; Viscondi, J.Y.K. Immunogenicity and Safety of Inactivated Quadrivalent Influenza Vaccine Compared with the Trivalent Vaccine for Influenza Infection: An Overview of Systematic Reviews. BMC Infect. Dis. 2023, 23, 561. [Google Scholar] [CrossRef] [PubMed]
- Ellebedy, A.H.; Webby, R.J. Influenza Vaccines. Vaccine 2009, 27, D65–D68. [Google Scholar] [CrossRef] [PubMed]
- Lowen, A.C. It’s in the Mix: Reassortment of Segmented Viral Genomes. PLoS Pathog. 2018, 14, e1007200. [Google Scholar] [CrossRef]
- Allen, J.D.; Ross, T.M. Bivalent H1 and H3 COBRA Recombinant Hemagglutinin Vaccines Elicit Seroprotective Antibodies against H1N1 and H3N2 Influenza Viruses from 2009 to 2019. J. Virol. 2022, 96, e0165221. [Google Scholar] [CrossRef]
- Huang, Y.; França, M.S.; Allen, J.D.; Shi, H.; Ross, T.M. Next Generation of Computationally Optimized Broadly Reactive Ha Vaccines Elicited Cross-Reactive Immune Responses and Provided Protection against H1n1 Virus Infection. Vaccines 2021, 9, 793. [Google Scholar] [CrossRef]
- Nagashima, K.; Abbadi, N.; Vyas, V.; Roegner, A.; Ross, T.M.; Mousa, J.J. Adjuvant-Mediated Differences in Antibody Responses to Computationally Optimized Hemagglutinin and Neuraminidase Vaccines. Viruses 2023, 15, 347. [Google Scholar] [CrossRef]
- Ou, B.S.; Saouaf, O.M.; Baillet, J.; Appel, E.A. Sustained Delivery Approaches to Improving Adaptive Immune Responses. Adv. Drug Deliv. Rev. 2022, 187, 114401. [Google Scholar] [CrossRef]
- Chen, J.; Wang, B.; Caserto, J.S.; Shariati, K.; Cao, P.; Pan, Y.; Xu, Q.; Ma, M. Sustained Delivery of SARS-CoV-2 RBD Subunit Vaccine Using a High Affinity Injectable Hydrogel Scaffold. Adv. Healthc. Mater. 2022, 11, e2101714. [Google Scholar] [CrossRef]
- Ho, W.; Gao, M.; Li, F.; Li, Z.; Zhang, X.Q.; Xu, X. Next-Generation Vaccines: Nanoparticle-Mediated DNA and MRNA Delivery. Adv. Healthc. Mater. 2021, 10, e2001812. [Google Scholar] [CrossRef]
- Roth, G.A.; Saouaf, O.M.; Smith, A.A.A.; Gale, E.C.; Hernandez, M.A.; Idoyaga, J.; Appel, E.A. Prolonged Codelivery of Hemagglutinin and a TLR7/8 Agonist in a Supramolecular Polymer−nanoparticle Hydrogel Enhances Potency and Breadth of Influenza Vaccination. ACS Biomater. Sci. Eng. 2021, 7, 1889–1899. [Google Scholar] [CrossRef]
- Cirelli, K.M.; Carnathan, D.G.; Nogal, B.; Martin, J.T.; Rodriguez, O.L.; Upadhyay, A.A.; Enemuo, C.A.; Gebru, E.H.; Choe, Y.; Viviano, F.; et al. Slow Delivery Immunization Enhances HIV Neutralizing Antibody and Germinal Center Responses via Modulation of Immunodominance. Cell 2019, 177, 1153–1171.e28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S. Fabrication of Novel Biomaterials through Molecular Self-Assembly. Nat. Biotechnol. 2003, 21, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Gil, E.S.; O’Neill, K.; Aleksi, E.; Budrewicz, J.; Melidone, R.; Spirio, L. Comparison of PuraStat Self-Assembling Peptide Hydrogel versus Mineral-Based Hemospray for Endoscopic Hemostasis of Upper and Lower Gastrointestinal Lesions in Pigs. Front. Gastroenterol. 2022, 1, 971353. [Google Scholar] [CrossRef]
- Branchi, F.; Klingenberg-Noftz, R.; Friedrich, K.; Bürgel, N.; Daum, S.; Buchkremer, J.; Sonnenberg, E.; Schumann, M.; Treese, C.; Tröger, H.; et al. PuraStat in Gastrointestinal Bleeding: Results of a Prospective Multicentre Observational Pilot Study. Surg. Endosc. 2022, 36, 2954–2961. [Google Scholar] [CrossRef]
- Jhala, D.; Vasita, R. A Review on Extracellular Matrix Mimicking Strategies for an Artificial Stem Cell Niche. Polym. Rev. 2015, 55, 561–595. [Google Scholar] [CrossRef]
- Kennedy, C. Enhancing Cobra H1 Efficacy Through the Vacsim® Delivery Platform. Master’s Thesis, The University of Georgia, Athens, GA, USA, 2022. [Google Scholar]
- Grenfell, R.F.Q.; Shollenberger, L.M.; Samli, E.F.; Harn, D.A. Vaccine Self-Assembling Immune Matrix Is a New Delivery Platform That Enhances Immune Responses to Recombinant HBsAg in Mice. Clin. Vaccine Immunol. 2015, 22, 336–343. [Google Scholar] [CrossRef]
- Ecker, J.W.; Kirchenbaum, G.A.; Pierce, S.R.; Skarlupka, A.L.; Abreu, R.B.; Cooper, R.E.; Taylor-Mulneix, D.; Ross, T.M.; Sautto, G.A. High-yield expression and purification of recombinant influenza virus proteins from stably-transfected mammalian cell lines. Vaccines 2020, 8, 462. [Google Scholar] [CrossRef]
- Elahi, S.M.; Nazemi-Moghaddam, N.; Gadoury, C.; Lippens, J.; Radinovic, S.; Venne, M.H.; Marcil, A.; Gilbert, R. A rapid Focus-Forming Assay for quantification of infectious adenoviral vectors. J. Virol. Methods 2021, 297, 114267. [Google Scholar] [CrossRef]
- Hoxie, I.; Vasilev, K.; Clark, J.J.; Bushfield, K.; Francis, B.; Loganathan, M.; Campbell, J.D.; Yu, D.; Guan, L.; Gu, C.; et al. A recombinant N2 neuraminidase-based CpG 1018® adjuvanted vaccine provides protection against challenge with heterologous influenza viruses in mice and hamsters. Vaccine 2024, 42, 126269. [Google Scholar] [CrossRef]
- Uno, N.; Ross, T.M. Multivalent next generation influenza virus vaccines protect against seasonal and pre-pandemic viruses. Sci. Rep. 2024, 14, 1440. [Google Scholar] [CrossRef] [PubMed]
- Marelli-Berg, F.M.; Peek, E.; Lidington, E.A.; Stauss, H.J.; Lechler, R.I. Isolation of endothelial cells from murine tissue. J. Immunol. Methods 2000, 244, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.M.; Allen, J.D.; Bebin-Blackwell, A.G.; Carter, D.M.; Alefantis, T.; DiNapoli, J.; Kleanthous, H.; Ross, T.M. Computationally Optimized Broadly Reactive Hemagglutinin Elicits Hemagglutination Inhibition Antibodies against a Panel of H3N2 Influenza Virus Cocirculating Variants. J. Virol. 2017, 91, e01581-17. [Google Scholar] [CrossRef]
- Committee for Medicinal Products for Human Use. Guideline on Influenza Vaccines—Non-Clinical and Clinical Module. European Medicines Agency EMA/CHMP/VWP/457259/2014 44. 2016. Available online: https://www.tga.gov.au/sites/default/files/2025-01/guideline_on_influenza-vaccines-non-clinical-and-clinical-module_en.pdf (accessed on 12 November 2024).
- Mousa, J.J.; Sauer, M.F.; Sevy, A.M.; Finn, J.A.; Bates, J.T.; Alvarado, G.; King, H.G.; Loerinc, L.B.; Fong, R.H.; Doranz, B.J.; et al. Structural basis for nonneutralizing antibody competition at antigenic site II of the respiratory syncytial virus fusion protein. Proc. Natl. Acad. Sci. USA 2016, 113, E6849–E6858. [Google Scholar] [CrossRef] [PubMed]
- McLellan, J.S.; Chen, M.; Joyce, M.G.; Sastry, M.; Stewart-Jones, G.B.; Yang, Y.; Zhang, B.; Chen, L.; Srivatsan, S.; Zheng, A.; et al. Structure-based design of a fusion glycoprotein vaccine for respiratory syncytial virus. Science 2013, 342, 592–598. [Google Scholar] [CrossRef]
- Waldock, J.; Remarque, E.J.; Zheng, L.; Ho, S.; Hoschler, K.; Neumann, B.; Sediri-Schön, H.; Trombetta, C.M.; Montomoli, E.; Marchi, S.; et al. Haemagglutination inhibition and virus microneutralisation serology assays: Use of harmonised protocols and biological standards in seasonal influenza serology testing and their impact on inter-laboratory variation and assay correlation: A FLUCOP collaborative study. Front. Immunol. 2023, 14, 1155552. [Google Scholar] [CrossRef]
- Irvine, D.J.; Aung, A.; Silva, M. Controlling timing and location in vaccines. Adv. Drug Deliv. Rev. 2020, 158, 91–115. [Google Scholar] [CrossRef]
- Beukema, M.; Gong, S.; Al-Jaawni, K.; de Vries-Idema, J.J.; Krammer, F.; Zhou, F.; Cox, R.J.; Huckriede, A. Prolonging the delivery of influenza virus vaccine improves the quantity and quality of the induced immune responses in mice. Front. Immunol. 2023, 14, 1249902. [Google Scholar] [CrossRef]
- García-Barreno, B.; Delgado, T.; Benito, S.; Casas, I.; Pozo, F.; Cuevas, M.T.; Mas, V.; Trento, A.; Rodriguez-Frandsen, A.; Falcón, A.; et al. Characterization of an enhanced antigenic change in the pandemic 2009 H1N1 influenza virus haemagglutinin. J. Gen. Virol. 2014, 95, 1033–1042. [Google Scholar] [CrossRef][Green Version]
- Batty, C.J.; Amouzougan, E.A.; Carlock, M.A.; Ross, T.M.; Bachelder, E.M.; Ainslie, K.M. Sustained delivery of CpG oligodeoxynucleotide by acetalated dextran microparticles augments effector response to Computationally Optimized Broadly Reactive Antigen (COBRA) influenza hemagglutinin. Int. J. Pharm. 2023, 630, 122429. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCormick, A.L.; Ross, T.M.; Harn, D.A.; Mousa, J.J. Use of the Slow-Delivery Platform, VacSIM, Shapes the Host Immune Response to Increase Protection Against Influenza Infection. Viruses 2025, 17, 1190. https://doi.org/10.3390/v17091190
McCormick AL, Ross TM, Harn DA, Mousa JJ. Use of the Slow-Delivery Platform, VacSIM, Shapes the Host Immune Response to Increase Protection Against Influenza Infection. Viruses. 2025; 17(9):1190. https://doi.org/10.3390/v17091190
Chicago/Turabian StyleMcCormick, Anna L., Ted M. Ross, Donald A. Harn, and Jarrod J. Mousa. 2025. "Use of the Slow-Delivery Platform, VacSIM, Shapes the Host Immune Response to Increase Protection Against Influenza Infection" Viruses 17, no. 9: 1190. https://doi.org/10.3390/v17091190
APA StyleMcCormick, A. L., Ross, T. M., Harn, D. A., & Mousa, J. J. (2025). Use of the Slow-Delivery Platform, VacSIM, Shapes the Host Immune Response to Increase Protection Against Influenza Infection. Viruses, 17(9), 1190. https://doi.org/10.3390/v17091190






