Unexpected Clinical and Laboratory Observations During and After 42-Day Versus 84-Day Treatment with Oral GS-441524 in Cats with Feline Infectious Peritonitis with Effusion
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Cohort
2.2. Monitoring of Cats
2.3. Unexpected Clinical and Laboratory Observations
2.4. Statistical Analysis
3. Results
3.1. Diarrhea
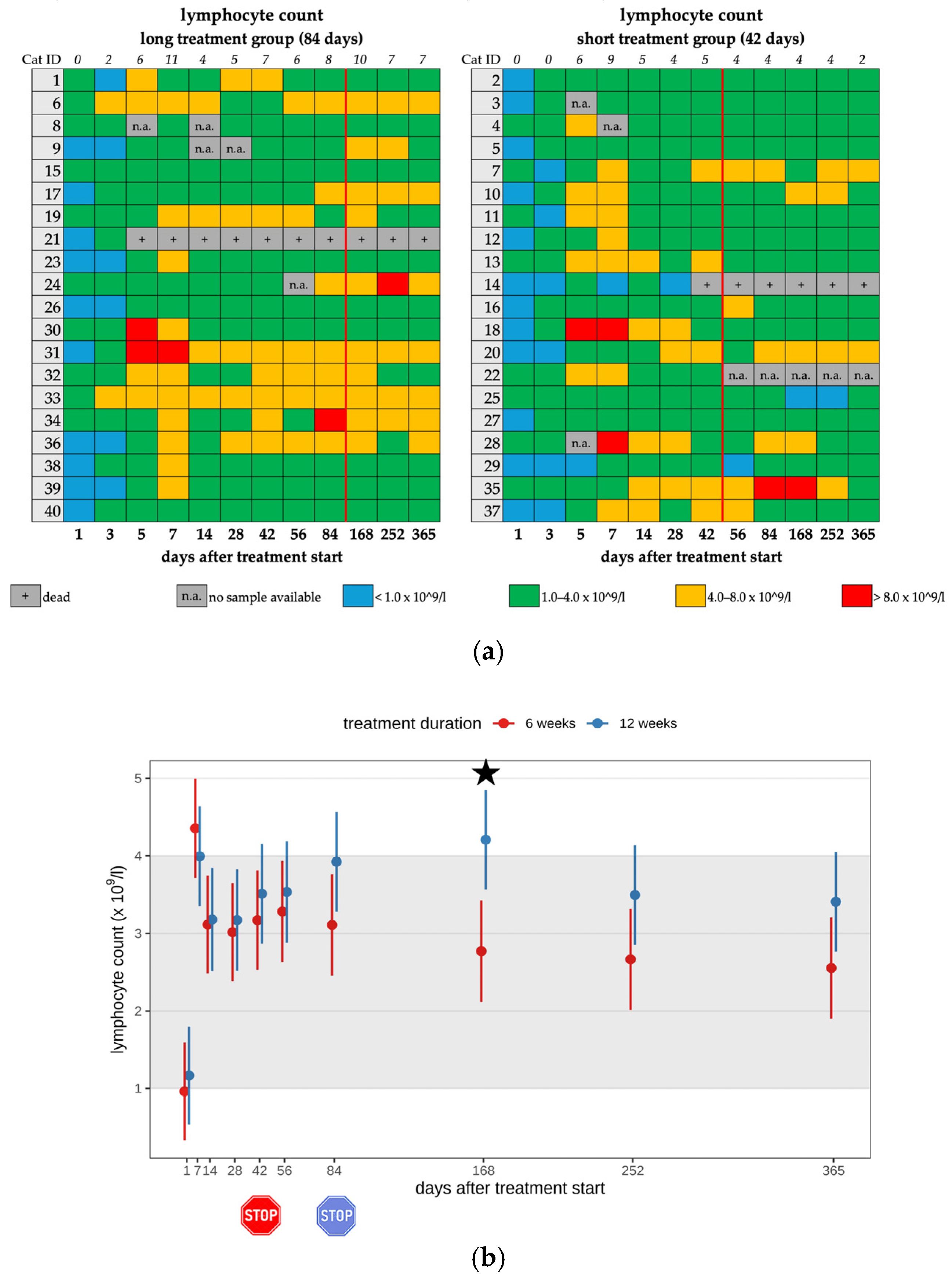
3.2. Lymphocytosis
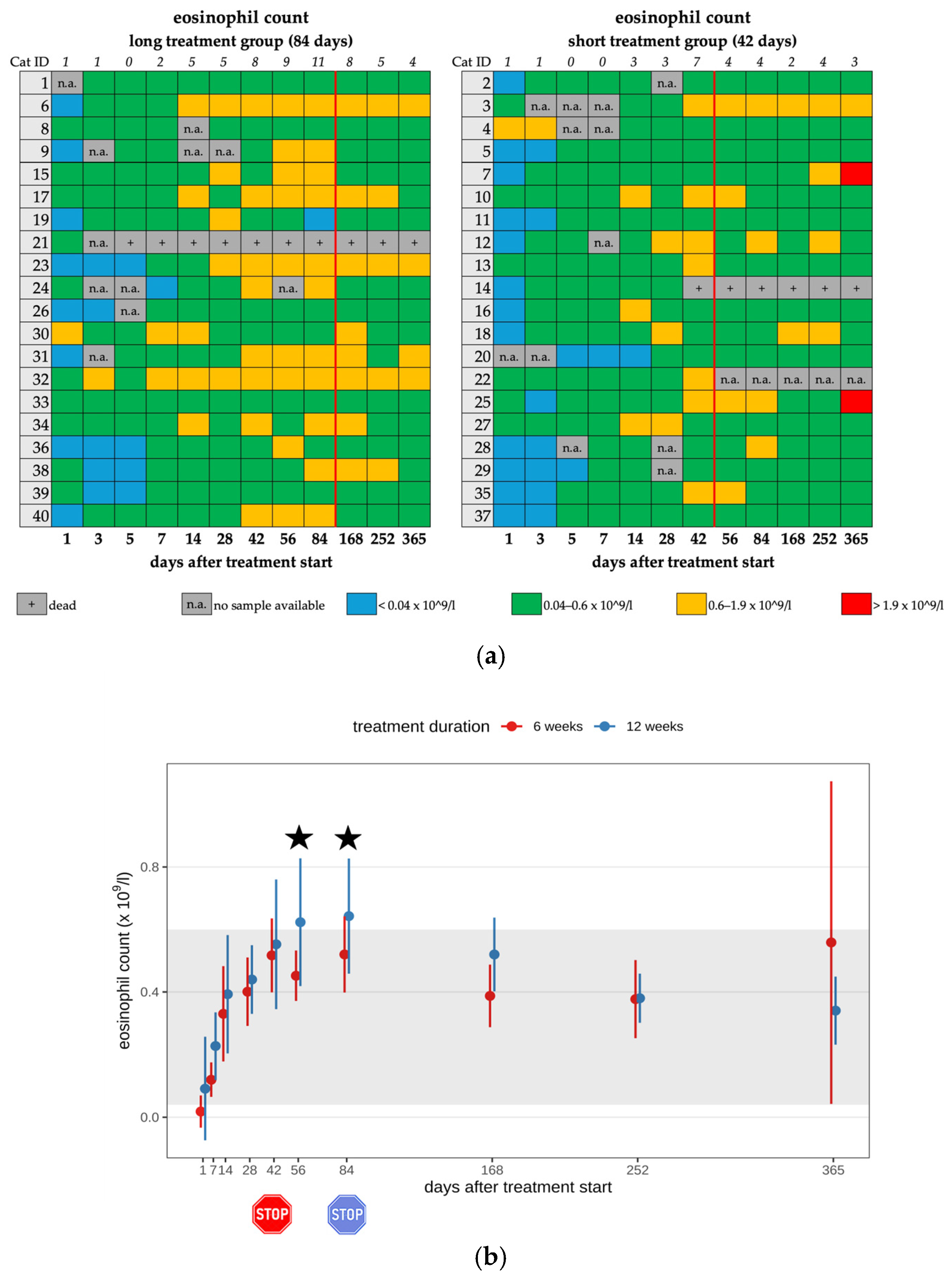
3.3. Eosinophilia
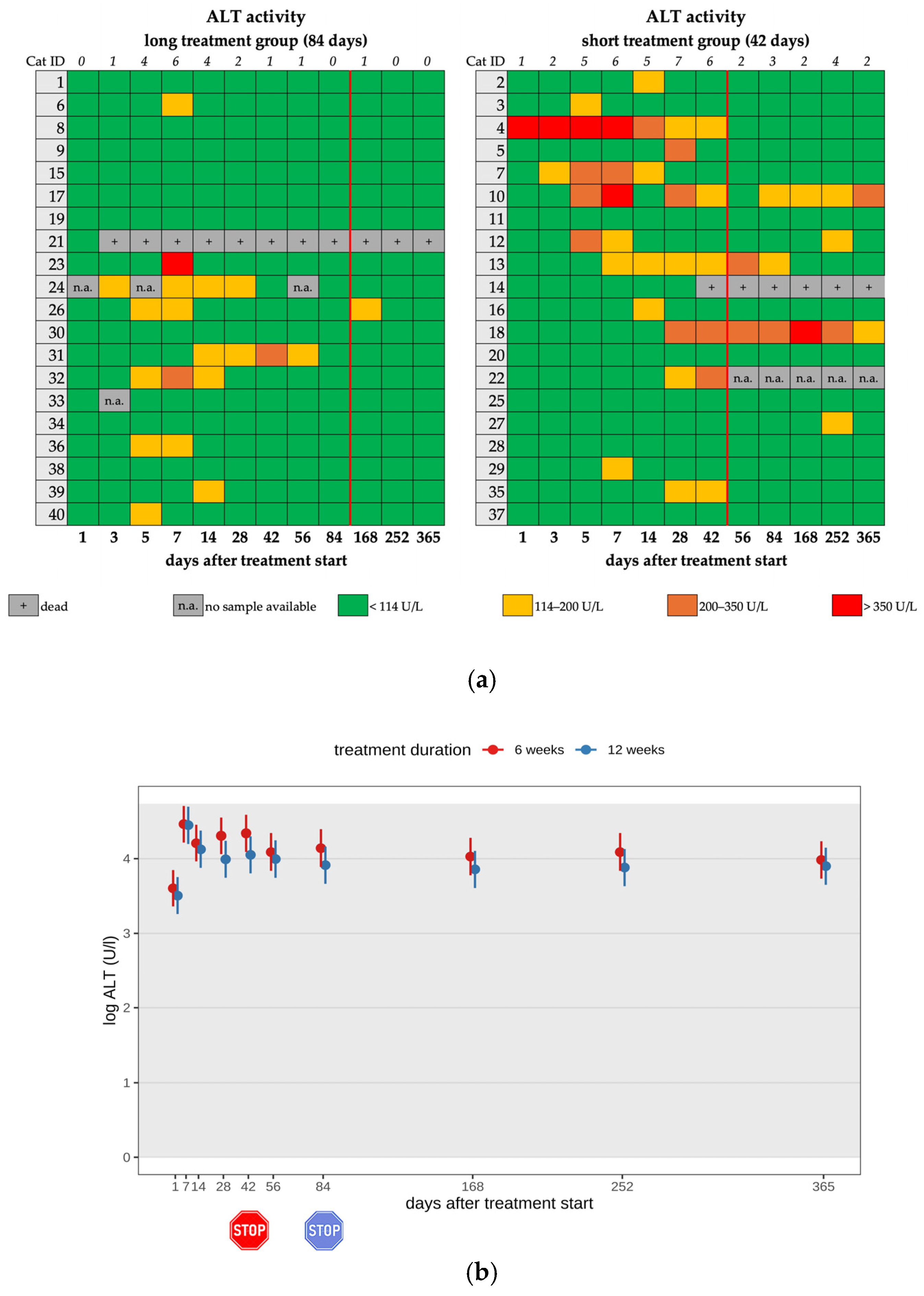
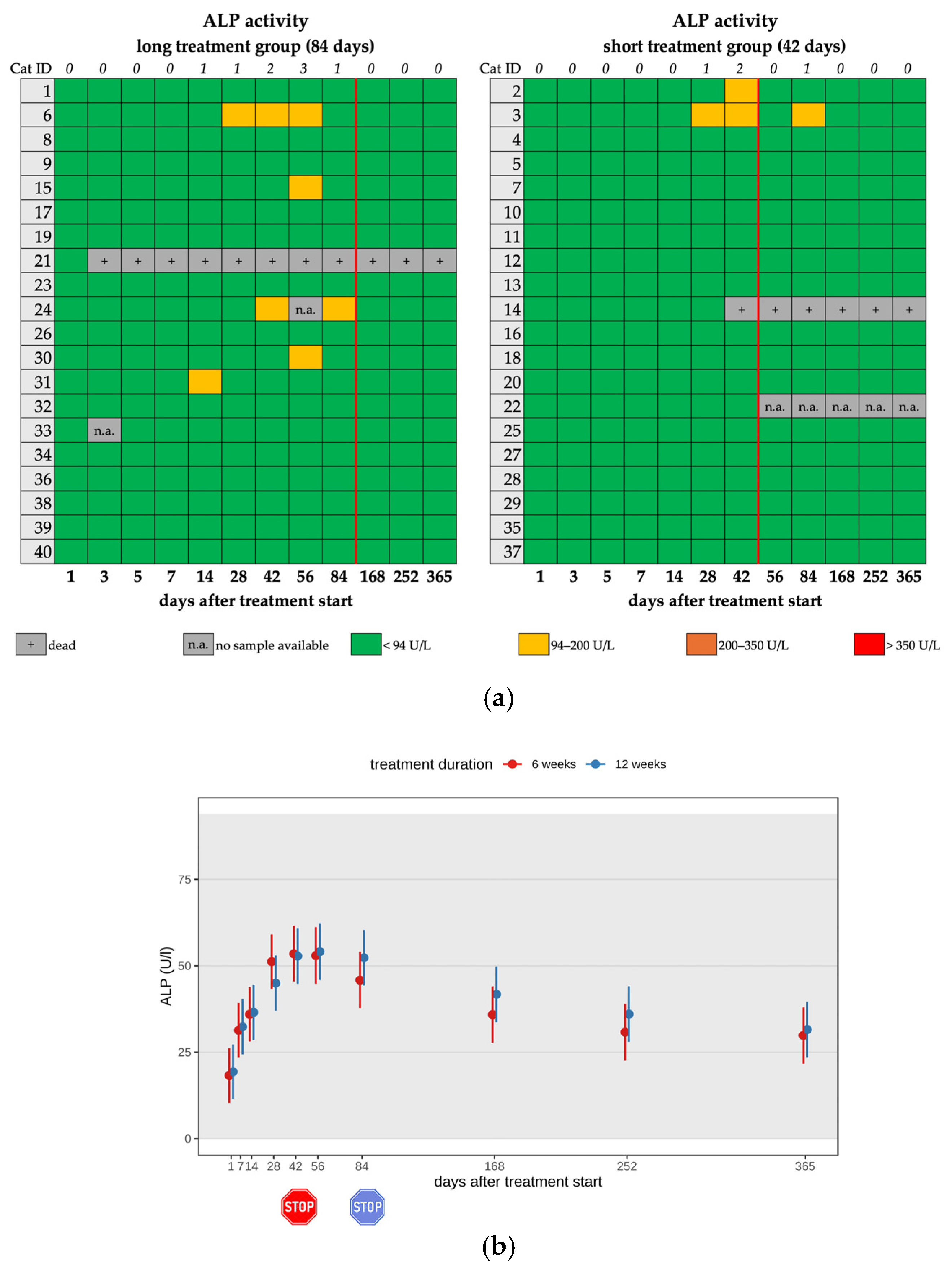
3.4. Changes in Liver Enzyme Activity
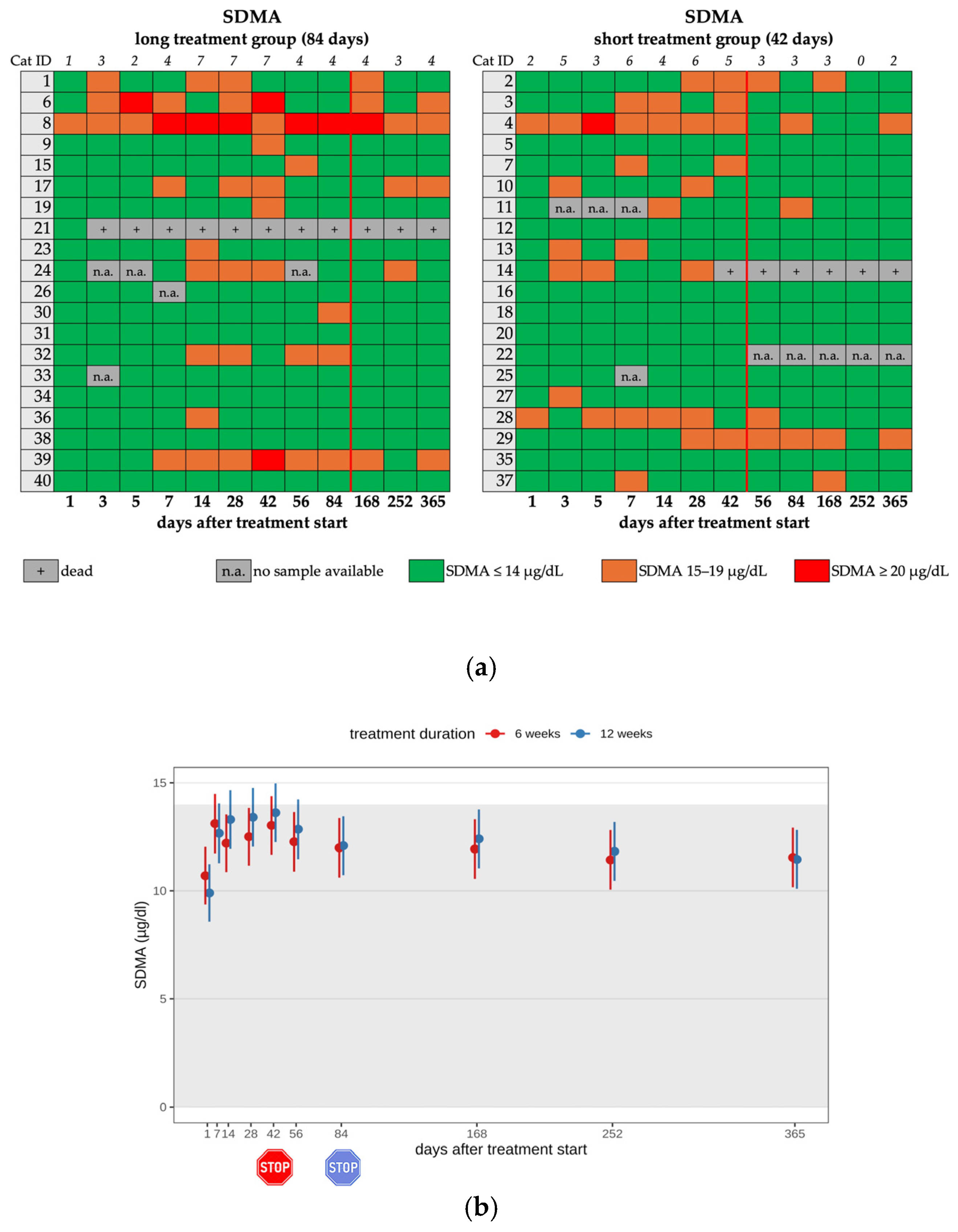
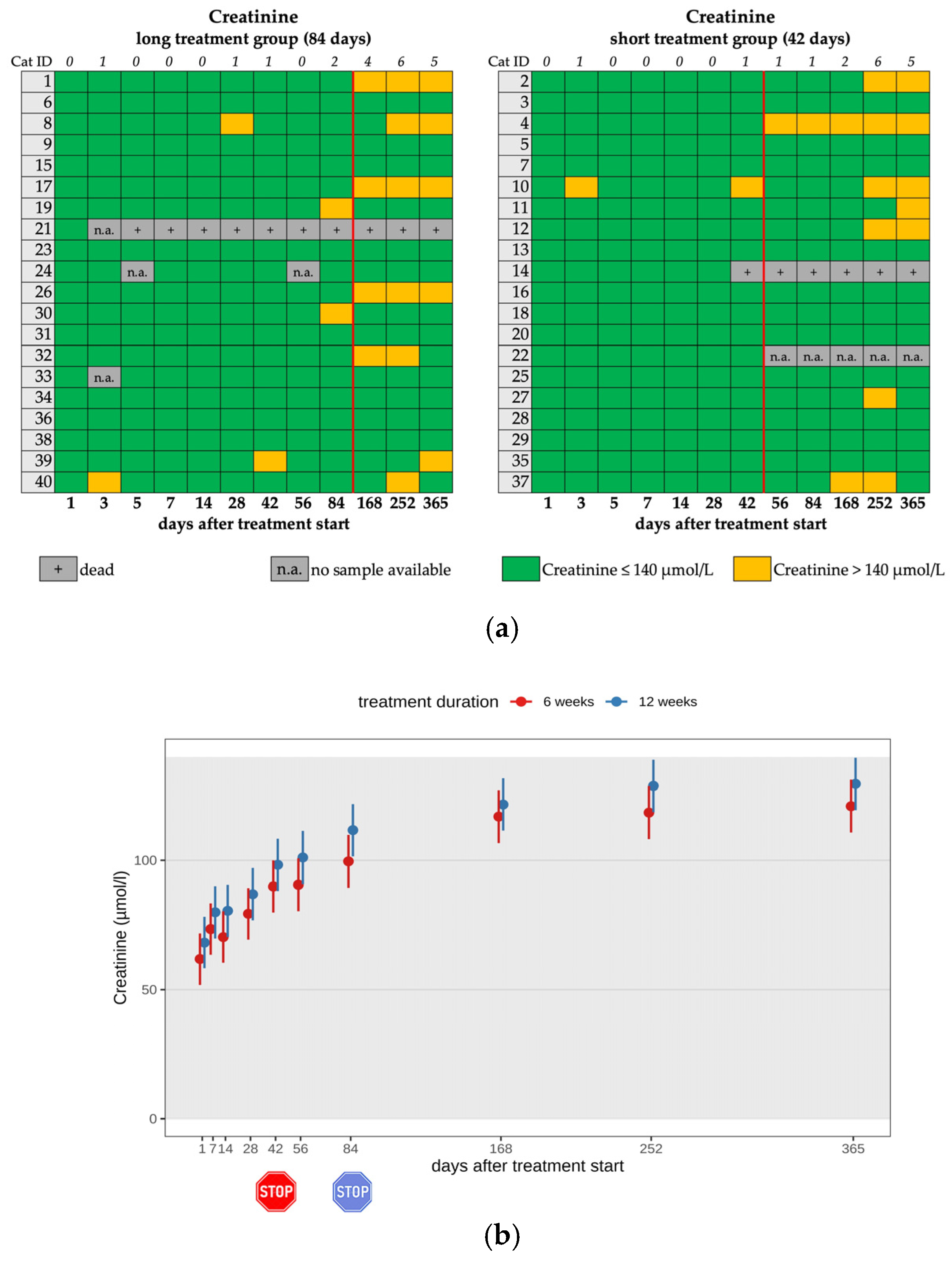
3.5. Changes in Renal Function Parameters
3.6. Hemolysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LMU | Ludwig-Maximilians University |
| FIP | feline infectious peritonitis |
| ALT | alanine aminotransferase |
| ALP | alkaline phosphatase |
| SDMA | symmetric dimethylarginine |
| RT-qPCR | reverse transcription quantitative polymerase chain reaction |
| FeLV | feline leukemia virus |
| FIV | feline immunodeficiency virus |
| UK | United Kingdom |
| PO | per os |
| q24h | every 24 h |
| FCoV | feline coronavirus |
| USG | urine specific gravity |
| UPC | urine protein creatinine ratio |
| RLMER | robust mixed-effects linear model |
| ICC | intraclass correlation coefficient |
| RMSE | root mean squared error |
| q12h | every 12 h |
| SAMe | S-adenosyl-methionine |
| RI | reference interval |
| SUB | subcutaneous ureteral bypass |
| IMHA | immune-mediated hemolytic anemia |
| COVID-19 | coronavirus disease 2019 |
| IL-5 | interleukin-5 |
| GFR | glomerular filtration rate |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus type 2 |
References
- Sparkes, A.H.; Gruffydd-Jones, T.J.; Harbour, D.A. Feline infectious peritonitis: A review of clinicopathological changes in 65 cases, and a critical assessment of their diagnostic value. Vet. Rec. 1991, 129, 209–212. [Google Scholar] [CrossRef]
- Pedersen, N.C.; Perron, M.; Bannasch, M.; Montgomery, E.; Murakami, E.; Liepnieks, M.; Liu, H. Efficacy and safety of the nucleoside analog GS-441524 for treatment of cats with naturally occurring feline infectious peritonitis. J. Feline Med. Surg. 2019, 21, 271–281. [Google Scholar] [CrossRef]
- Amirian, E.S.; Levy, J.K. Current knowledge about the antivirals remdesivir (GS-5734) and GS-441524 as therapeutic options for coronaviruses. One Health 2020, 9, 100128. [Google Scholar] [CrossRef] [PubMed]
- Krentz, D.; Zenger, K.; Alberer, M.; Felten, S.; Bergmann, M.; Dorsch, R.; Matiasek, K.; Kolberg, L.; Hofmann-Lehmann, R.; Meli, M.L.; et al. Curing cats with feline infectious peritonitis with an oral multi-component drug containing GS-441524. Viruses 2021, 13, 2228. [Google Scholar] [CrossRef] [PubMed]
- Zwicklbauer, K.; Krentz, D.; Bergmann, M.; Felten, S.; Dorsch, R.; Fischer, A.; Hofmann-Lehmann, R.; Meli, M.L.; Spiri, A.M.; Alberer, M.; et al. Long-term follow-up of cats in complete remission after treatment of feline infectious peritonitis with oral GS-441524. J. Feline Med. Surg. 2023, 25, 1098612X231183250. [Google Scholar] [CrossRef]
- Zuzzi-Krebitz, A.M.; Buchta, K.; Bergmann, M.; Krentz, D.; Zwicklbauer, K.; Dorsch, R.; Wess, G.; Fischer, A.; Matiasek, K.; Hönl, A.; et al. Short treatment of 42 days with oral GS-441524 results in equal efficacy as the recommended 84-day treatment in cats suffering from feline infectious peritonitis with effusion—A prospective randomized controlled study. Viruses 2024, 16, 1144. [Google Scholar] [CrossRef] [PubMed]
- Krentz, D.; Zwicklbauer, K.; Felten, S.; Bergmann, M.; Dorsch, R.; Hofmann-Lehmann, R.; Meli, M.L.; Spiri, A.M.; von Both, U.; Alberer, M.; et al. Clinical follow-up and postmortem findings in a cat that was cured of feline infectious peritonitis with an oral antiviral drug containing GS-441524. Viruses 2022, 14, 2040. [Google Scholar] [CrossRef]
- Murphy, B.G.; Perron, M.; Murakami, E.; Bauer, K.; Park, Y.; Eckstrand, C.; Liepnieks, M.; Pedersen, N.C. The nucleoside analog GS-441524 strongly inhibits feline infectious peritonitis (FIP) virus in tissue culture and experimental cat infection studies. Vet. Microbiol. 2018, 219, 226–233. [Google Scholar] [CrossRef]
- Addie, D.D.; Covell-Ritchie, J.; Jarrett, O.; Fosbery, M. Rapid resolution of non-effusive feline infectious peritonitis uveitis with an oral adenosine nucleoside analogue and feline interferon omega. Viruses 2020, 12, 1216. [Google Scholar] [CrossRef]
- Dickinson, P.J.; Bannasch, M.; Thomasy, S.M.; Murthy, V.D.; Vernau, K.M.; Liepnieks, M.; Montgomery, E.; Knickelbein, K.E.; Murphy, B.; Pedersen, N.C. Antiviral treatment using the adenosine nucleoside analogue GS-441524 in cats with clinically diagnosed neurological feline infectious peritonitis. J. Vet. Intern. Med. 2020, 34, 1587–1593. [Google Scholar] [CrossRef]
- Jones, S.; Novicoff, W.; Nadeau, J.; Evans, S. Unlicensed GS-441524-like antiviral therapy can be effective for at-home treatment of feline infectious peritonitis. Animals 2021, 11, 2257. [Google Scholar] [CrossRef] [PubMed]
- Katayama, M.; Uemura, Y. Therapeutic effects of mutian(®) Xraphconn on 141 client-owned cats with feline infectious peritonitis predicted by total bilirubin levels. Vet. Sci. 2021, 8, 328. [Google Scholar] [CrossRef]
- Lv, J.; Bai, Y.; Wang, Y.; Yang, L.; Jin, Y.; Dong, J. Effect of GS-441524 in combination with the 3C-like protease inhibitor GC376 on the treatment of naturally transmitted feline infectious peritonitis. Front. Vet. Sci. 2022, 9, 1002488. [Google Scholar] [CrossRef]
- Green, J.; Syme, H.; Tayler, S. Thirty-two cats with effusive or non-effusive feline infectious peritonitis treated with a combination of remdesivir and GS-441524. J. Vet. Intern. Med. 2023, 37, 1784–1793. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Li, T.; Wang, C.; Liu, X.; Ouyang, H.; Ji, W.; Liu, J.; Liao, X.; Li, J.; Hu, C. A retrospective study of clinical and laboratory features and treatment on cats highly suspected of feline infectious peritonitis in Wuhan, China. Sci. Rep. 2021, 11, 5208. [Google Scholar] [CrossRef]
- Taylor, S.S.; Coggins, S.; Barker, E.N.; Gunn-Moore, D.; Jeevaratnam, K.; Norris, J.M.; Hughes, D.; Stacey, E.; MacFarlane, L.; O’Brien, C.; et al. Retrospective study and outcome of 307 cats with feline infectious peritonitis treated with legally sourced veterinary compounded preparations of remdesivir and GS-441524 (2020–2022). J. Feline Med. Surg. 2023, 25, 1098612x231194460. [Google Scholar] [CrossRef]
- Coggins, S.J.; Norris, J.M.; Malik, R.; Govendir, M.; Hall, E.J.; Kimble, B.; Thompson, M.F. Outcomes of treatment of cats with feline infectious peritonitis using parenterally administered remdesivir, with or without transition to orally administered GS-441524. J. Vet. Intern. Med. 2023, 37, 1772–1783. [Google Scholar] [CrossRef]
- Cosaro, E.; Pires, J.; Castillo, D.; Murphy, B.G.; Reagan, K.L. Efficacy of oral remdesivir compared to gs-441524 for treatment of cats with naturally occurring effusive feline infectious peritonitis: A blinded, non-inferiority study. Viruses 2023, 15, 1680. [Google Scholar] [CrossRef] [PubMed]
- Addie, D.D.; Silveira, C.; Aston, C.; Brauckmann, P.; Covell-Ritchie, J.; Felstead, C.; Fosbery, M.; Gibbins, C.; Macaulay, K.; McMurrough, J.; et al. Alpha-1 acid glycoprotein reduction differentiated recovery from remission in a small cohort of cats treated for feline infectious peritonitis. Viruses 2022, 14, 744. [Google Scholar] [CrossRef]
- Allinder, M.; Tynan, B.; Martin, C.; Furbish, A.; Austin, G.; Bartges, J.; Lourenco, B.N. Uroliths composed of antiviral compound gs-441524 in 2 cats undergoing treatment for feline infectious peritonitis. J. Vet. Intern. Med. 2024, 38, 370–374. [Google Scholar] [CrossRef]
- Hartmann, K.; Kuffer, M. Karnofsky’s score modified for cats. Eur. J. Med. Res. 1998, 3, 95–98. [Google Scholar]
- Purina Fecal Scoring Chart. Available online: https://vetcentre.purina.co.uk/sites/default/files/2021-11/Faecal%20scoring%20chart_general%20use.pdf (accessed on 20 August 2025).
- Iris Staging of CKD. Available online: http://www.iris-kidney.com/pdf/2_IRIS_Staging_of_CKD_2023.pdf (accessed on 25 July 2024).
- Diagnosing, Staging, and Treating Chronic Kidney Disease in Dogs and Cats. Available online: http://www.iris-kidney.com/pdf/IRIS_Pocket_Guide_to_CKD_2023.pdf (accessed on 25 July 2024).
- Griffin, S. Feline abdominal ultrasonography: What’s normal? What’s abnormal? renal pelvis, ureters and urinary bladder. J. Feline Med. Surg. 2020, 22, 847–865. [Google Scholar] [CrossRef]
- Haagmans, B.L.; Egberink, H.F.; Horzinek, M.C. Apoptosis and t-cell depletion during feline infectious peritonitis. J. Virol. 1996, 70, 8977–8983. [Google Scholar] [CrossRef] [PubMed]
- Takano, T.; Hohdatsu, T.; Hashida, Y.; Kaneko, Y.; Tanabe, M.; Koyama, H. A “possible” involvement of TNF-alpha in apoptosis induction in peripheral blood lymphocytes of cats with feline infectious peritonitis. Vet. Microbiol. 2007, 119, 121–131. [Google Scholar] [CrossRef]
- Dean, G.A.; Olivry, T.; Stanton, C.; Pedersen, N.C. In vivo cytokine response to experimental feline infectious peritonitis virus infection. Vet. Microbiol. 2003, 97, 1–12. [Google Scholar] [CrossRef]
- Cianci, R.; Massaro, M.G.; De Santis, E.; Totti, B.; Gasbarrini, A.; Gambassi, G.; Giambra, V. Changes in lymphocyte subpopulations after remdesivir therapy for COVID-19: A brief report. Int. J. Mol. Sci. 2023, 24, 14973. [Google Scholar] [CrossRef]
- Yan, Y.; Li, J.; Jiao, Z.; Yang, M.; Li, L.; Wang, G.; Chen, Y.; Li, M.; Shen, Z.; Shi, Y.; et al. Better therapeutic effect of oral administration of GS441524 compared with GC376. Vet. Microbiol. 2023, 283, 109781. [Google Scholar] [CrossRef] [PubMed]
- Zwicklbauer, K.; von la Roche, D.; Krentz, D.; Kolberg, L.; Alberer, M.; Hartmann, K.; von Both, U.; Härtle, S. Characterization of the lymphocyte response in cats with feline infectious peritonitis during antiviral treatment using the smart tube technology for flow cytometry in feline full blood samples. J. Vet. Intern. Med. 2024, 38, 3596. [Google Scholar]
- Zwicklbauer, K.; von la Roche, D.; Krentz, D.; Kolberg, L.; Alberer, M.; Zablotski, Y.; Hartmann, K.; von Both, U.; Härtle, S. Adapting the smart tube technology for flow cytometry in feline full blood samples. Front. Vet. Sci. 2024, 11, 1377414. [Google Scholar] [CrossRef] [PubMed]
- Buchta, K.; Zuzzi-Krebitz, A.; Zwicklbauer, K.; Bergmann, M.; Dorsch, R.; Hofmann-Lehmann, R.; Meli, M.L.; Spiri, A.M.; Matiasek, K.; Zablotski, Y.; et al. Ein-Jahres-Follow-Up von Katzen nach Therapie der felinen infektiösen Peritonitis mit oralem GS-441524 für 42 versus 84 Tage. Tierärztliche Prax. Ausg. K Kleintiere/Heimtiere 2025, 53, V06. [Google Scholar] [CrossRef]
- Mateos González, M.; Sierra Gonzalo, E.; Casado Lopez, I.; Arnalich Fernández, F.; Beato Pérez, J.L.; Monge Monge, D.; Vargas Núñez, J.A.; García Fenoll, R.; Suárez Fernández, C.; Freire Castro, S.J.; et al. The prognostic value of eosinophil recovery in COVID-19: A multicentre, retrospective cohort study on patients hospitalised in spanish hospitals. J. Clin. Med. 2021, 10, 305. [Google Scholar] [CrossRef]
- Zimmermann, N.; Rothenberg, M.E. Mechanism of enhanced eosinophil survival in inflammation. Blood 2015, 125, 3831–3832. [Google Scholar] [CrossRef]
- Ian, M.; Williams, C. Drug-induced eosinophilia. Pharm. J. 2000, 264, 71–76. [Google Scholar]
- Tekes, G.; Thiel, H.J. Feline coronaviruses: Pathogenesis of feline infectious peritonitis. Adv. Virus. Res. 2016, 96, 193–218. [Google Scholar] [PubMed]
- Cony, F.G.; Pereira, V.C.; Slaviero, M.; Lima, R.P.; de Castro, L.T.; de Moraes, J.T.R.; Aliardi, J.M.G.; Driemeier, D.; Sonne, L.; Panziera, W.; et al. Anatomopathological characterization of hepatic lesions of feline infectious peritonitis in cats. J. Comp. Pathol. 2024, 215, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Malbon, A.J.; Fonfara, S.; Meli, M.L.; Hahn, S.; Egberink, H.; Kipar, A. Feline infectious peritonitis as a systemic inflammatory disease: Contribution of liver and heart to the pathogenesis. Viruses 2019, 11, 1144. [Google Scholar] [CrossRef]
- Xie, J.; Wang, Z. Can remdesivir and its parent nucleoside gs-441524 be potential oral drugs? an in vitro and in vivo dmpk assessment. Acta Pharm. Sin. B 2021, 11, 1607–1616. [Google Scholar] [CrossRef]
- Zampino, R.; Mele, F.; Florio, L.L.; Bertolino, L.; Andini, R.; Galdo, M.; De Rosa, R.; Corcione, A.; Durante-Mangoni, E. Liver injury in remdesivir-treated COVID-19 patients. Hepatol. Int. 2020, 14, 881–883. [Google Scholar] [CrossRef] [PubMed]
- Kramer, J.W.; Hoffman, W.E.; Kaneko, J.J.; Harvey, J.W.; Bruss, M.L. Clinical Enzymology; Academic Press: Cambridge, MA, USA, 1997. [Google Scholar]
- Levy, J.K.; Crawford, P.C.; Werner, L.L. Effect of age on reference intervals of serum biochemical values in kittens. J. Am. Vet. Med. Assoc. 2006, 228, 1033–1037. [Google Scholar] [CrossRef]
- Hall, J.A.; Yerramilli, M.; Obare, E.; Yerramilli, M.; Jewell, D.E. Comparison of serum concentrations of symmetric dimethylarginine and creatinine as kidney function biomarkers in cats with chronic kidney disease. J. Vet. Intern. Med. 2014, 28, 1676–1683. [Google Scholar] [CrossRef]
- Baral, R.M.; Freeman, K.P.; Flatland, B. Comparison of serum and plasma SDMA measured with point-of-care and reference laboratory analysers: Implications for interpretation of SDMA in cats. J. Feline Med. Surg. 2021, 23, 906–920. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sargent, H.J.; Elliott, J.; Jepson, R.E. The new age of renal biomarkers: Does sdma solve all of our problems? J. Small Anim. Pract. 2021, 62, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Harley, L.; Langston, C. Proteinuria in dogs and cats. Can. Vet. J. 2012, 53, 631–638. [Google Scholar] [PubMed]
- Furbish, A.; Allinder, M.; Austin, G.; Tynan, B.; Byrd, E.; Gomez, I.P. Peterson. First analytical confirmation of drug-induced crystal nephropathy in felines caused by GS-441524, the active metabolite of remdesivir. J. Pharm. Biomed. Anal. 2024, 247, 116248. [Google Scholar] [CrossRef] [PubMed]
- Kent, A.M.; Guan, S.; Jacque, N.; Novicoff, W.; Evans, S.J.M. Unlicensed antiviral products used for the at-home treatment of feline infectious peritonitis contain GS-441524 at significantly different amounts than advertised. J. Am. Vet. Med. Assoc. 2024, 262, 489–497. [Google Scholar] [CrossRef]
- Friedel, D.M.; Cappell, M.S. Diarrhea and coronavirus disease 2019 infection. Gastroenterol. Clin. N. Am. 2023, 52, 59–75. [Google Scholar] [CrossRef]
- Wu, Y.; Cheng, X.; Jiang, G.; Tang, H.; Ming, S.; Tang, L.; Lu, J.; Guo, C.; Shan, H.; Huang, X. Altered oral and gut microbiota and its association with SARS-CoV-2 viral load in COVID-19 patients during hospitalization. NPJ Biofilms Microbiomes 2021, 7, 61. [Google Scholar] [CrossRef]
 : start of treatment with GS-441524 (day 1),
: start of treatment with GS-441524 (day 1),  : end of treatment of 20/40 cats on day 42,
: end of treatment of 20/40 cats on day 42,  : end of treatment of 20/40 cats on day 84,
: end of treatment of 20/40 cats on day 84,  : blood tests (including hematology, serum biochemistry, and SDMA),
: blood tests (including hematology, serum biochemistry, and SDMA),  : physical examination,
: physical examination,  : abdominal ultrasonography,
: abdominal ultrasonography,  : detailed cardiologic examination (including electrocardiography and echocardiography),
: detailed cardiologic examination (including electrocardiography and echocardiography),  : neurologic examination.
: neurologic examination.
 : start of treatment with GS-441524 (day 1),
: start of treatment with GS-441524 (day 1),  : end of treatment of 20/40 cats on day 42,
: end of treatment of 20/40 cats on day 42,  : end of treatment of 20/40 cats on day 84,
: end of treatment of 20/40 cats on day 84,  : blood tests (including hematology, serum biochemistry, and SDMA),
: blood tests (including hematology, serum biochemistry, and SDMA),  : physical examination,
: physical examination,  : abdominal ultrasonography,
: abdominal ultrasonography,  : detailed cardiologic examination (including electrocardiography and echocardiography),
: detailed cardiologic examination (including electrocardiography and echocardiography),  : neurologic examination.
: neurologic examination.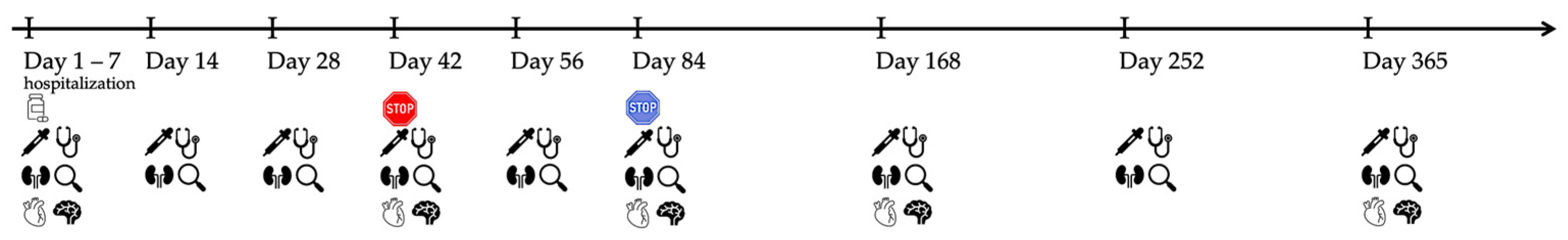

| Parameters | Short Treatment Group | Long Treatment Group |
|---|---|---|
| Age | ||
| Range of Age in Months | 6.4–98.6 | 5.1–116.3 |
| Median Age in Months | 12.3 | 14.95 |
| Breeds | ||
| Domestic Shorthair | 11 (55.0%) | 5 (25.0%) |
| British Shorthair | 3 (15.0%) | 5 (25.0%) |
| Maine Coon | 2 (10.0%) | 1 (5.0%) |
| Siamese | 1 (5.0%) | 0 (0.0%) |
| Holy Birman | 0 (0.0%) | 1 (5.0%) |
| Scottish Fold/Straight | 1 (5.0%) | 1 (5.0%) |
| Somali | 0 (0.0%) | 1 (5.0%) |
| Mixed Breed | 2 (10.0%) | 3 (15.0%) |
| Exotic Shorthair | 0 (0.0%) | 1 (5.0%) |
| Oriental Shorthair | 0 (0.0%) | 1 (5.0%) |
| Ragdoll | 0 (0.0%) | 1 (5.0%) |
| Sex | ||
| Male | 14 (70.0%) | 17 (85.0%) |
| (neutered) | 11/14 (78.6%) | 10/17 (58.8%) |
| Female | 6 (30.0%) | 3/20 (15.0%) |
| (neutered) | 1/6 (16.7%) | 1/3 (33.3%) |
| Partner Cats? | 17/20 (85.0%) | 13/20 (65.0%) |
| Unexpected Clinical and Laboratory Observations | Number of Cats | Grade | Symptomatic Treatment | |
|---|---|---|---|---|
| During Treatment (until Day 42) | ||||
| Diarrhea 1 | 14/20 | 11/14 | mild | |
| 3/14 | severe | probiotics, fluid therapy | ||
| Lymphocytosis 2 | 12/20 | 10/12 | mild | none |
| 2/12 | moderate | |||
| Eosinophilia 3 | 11/20 | 11/11 | mild | none |
| Increased ALT 4 | 13/20 | 6/13 | mild | |
| 5/13 | moderate | |||
| 2/13 | severe | silymarin 8, SAMe 9 | ||
| Increased ALP 5 | 2/20 | 2/2 | mild | none |
| Increased SDMA 6 | 12/20 | 11/12 | mild | none |
| 1/12 | moderate | |||
| After Treatment (Days 56, 84, 168, 252, 365) | ||||
| Lymphocytosis | 7/18 | 6/7 | mild | none |
| 1/7 | moderate | |||
| Eosinophilia | 8/18 | 6/8 | mild | none |
| 2/8 | moderate | |||
| Increased ALT | 5/18 | 1/5 | mild | |
| 3/5 | moderate | |||
| 1/5 | severe | silymarin | ||
| Increased ALP | 1/18 | 1/1 | mild | none |
| Increased SDMA | 6/18 | 6/6 | mild | none |
| Renal Azotemia 7 | 4/18 | 4/4 | mild | dietary changes |
| Unexpected Clinical and Laboratory Observations | Number of Cats | Grade | Symptomatic Treatment | |
|---|---|---|---|---|
| During Treatment (until Day 84) | ||||
| Diarrhea 1 | 11/20 | 9/11 | mild | |
| 2/11 | severe | probiotics, fluid therapy | ||
| Lymphocytosis 2 | 14/20 | 11/14 | mild | none |
| 3/14 | moderate | |||
| Eosinophilia 3 | 14/20 | 14/14 | mild | none |
| Increased ALT 4 | 9/20 | 6/9 | mild | |
| 2/9 | moderate | |||
| 1/9 | severe | silymarin 8, SAMe 9 | ||
| Increased ALP 5 | 5/20 | 5/5 | mild | none |
| Increased SDMA 6 | 13/20 | 10/13 | mild | none |
| 3/13 | moderate | |||
| After Treatment (Days 168, 252, 365) | ||||
| Lymphocytosis | 10/19 | 9/10 | mild | none |
| 1/10 | moderate | |||
| Eosinophilia | 8/19 | 8/8 | mild | none |
| Increased ALT | 1/19 | 1/1 | mild | |
| Increased SDMA | 6/19 | 5/6 | mild | none |
| 1/6 | moderate | |||
| Renal Azotemia 7 | 3/19 | 3/3 | mild | dietary changes |
| Unexpected Clinical and Laboratory Observations | Occurred During Treatment | Occurred After Treatment | Never Occurred | |||
|---|---|---|---|---|---|---|
| 42-Day Treatment | 84-Day Treatment | 42-Day Treatment | 84-Day Treatment | 42-Day Treatment | 84-Day Treatment | |
| Diarrhea 1 | 14/20 | 11/20 | 0/18 | 0/19 | 6/20 | 9/20 |
| Lymphocytosis | 12/20 | 14/20 | 7/18 | 10/19 | 7/20 | 5/20 |
| Eosinophilia | 11/20 | 14/20 | 8/18 | 8/19 | 7/20 | 6/20 |
| Increased ALT 2 | 13/20 | 9/20 | 5/18 | 1/19 | 6/20 | 11/20 |
| Increased ALP 3 | 2/20 | 5/20 | 1/18 | 0/19 | 18/20 | 15/20 |
| Increased SDMA 4 | 12/20 | 13/20 | 6/18 | 6/19 | 8/20 | 7/20 |
| Renal Azotemia 5 | 0/20 | 0/20 | 4/18 | 3/19 | 15/20 | 16/20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buchta, K.; Zuzzi-Krebitz, A.-M.; Bergmann, M.; Dorsch, R.; Zwicklbauer, K.; Matiasek, K.; Hofmann-Lehmann, R.; Meli, M.L.; Spiri, A.M.; Zablotski, Y.; et al. Unexpected Clinical and Laboratory Observations During and After 42-Day Versus 84-Day Treatment with Oral GS-441524 in Cats with Feline Infectious Peritonitis with Effusion. Viruses 2025, 17, 1181. https://doi.org/10.3390/v17091181
Buchta K, Zuzzi-Krebitz A-M, Bergmann M, Dorsch R, Zwicklbauer K, Matiasek K, Hofmann-Lehmann R, Meli ML, Spiri AM, Zablotski Y, et al. Unexpected Clinical and Laboratory Observations During and After 42-Day Versus 84-Day Treatment with Oral GS-441524 in Cats with Feline Infectious Peritonitis with Effusion. Viruses. 2025; 17(9):1181. https://doi.org/10.3390/v17091181
Chicago/Turabian StyleBuchta, Katharina, Anna-Maria Zuzzi-Krebitz, Michèle Bergmann, Roswitha Dorsch, Katharina Zwicklbauer, Kaspar Matiasek, Regina Hofmann-Lehmann, Marina L. Meli, Andrea M. Spiri, Yury Zablotski, and et al. 2025. "Unexpected Clinical and Laboratory Observations During and After 42-Day Versus 84-Day Treatment with Oral GS-441524 in Cats with Feline Infectious Peritonitis with Effusion" Viruses 17, no. 9: 1181. https://doi.org/10.3390/v17091181
APA StyleBuchta, K., Zuzzi-Krebitz, A.-M., Bergmann, M., Dorsch, R., Zwicklbauer, K., Matiasek, K., Hofmann-Lehmann, R., Meli, M. L., Spiri, A. M., Zablotski, Y., Alberer, M., von Both, U., & Hartmann, K. (2025). Unexpected Clinical and Laboratory Observations During and After 42-Day Versus 84-Day Treatment with Oral GS-441524 in Cats with Feline Infectious Peritonitis with Effusion. Viruses, 17(9), 1181. https://doi.org/10.3390/v17091181








