Molecular Characterization of a Novel Rubodvirus Infecting Raspberries
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant/Insect Samples and RNA Isolation
2.2. RT–PCR Screening and Sanger Sequencing of Plant and Insect Samples
2.3. High Throughput Sequencing and Analysis of the V2 Sample
2.4. Sanger Sequencing of V2 Isolate of Raspberry Rubodvirus 1 (RaRV1)
2.5. Transmission Experiments
2.6. Data Analyses
3. Results
3.1. HTS and Sanger Sequencing of the V2 Sample
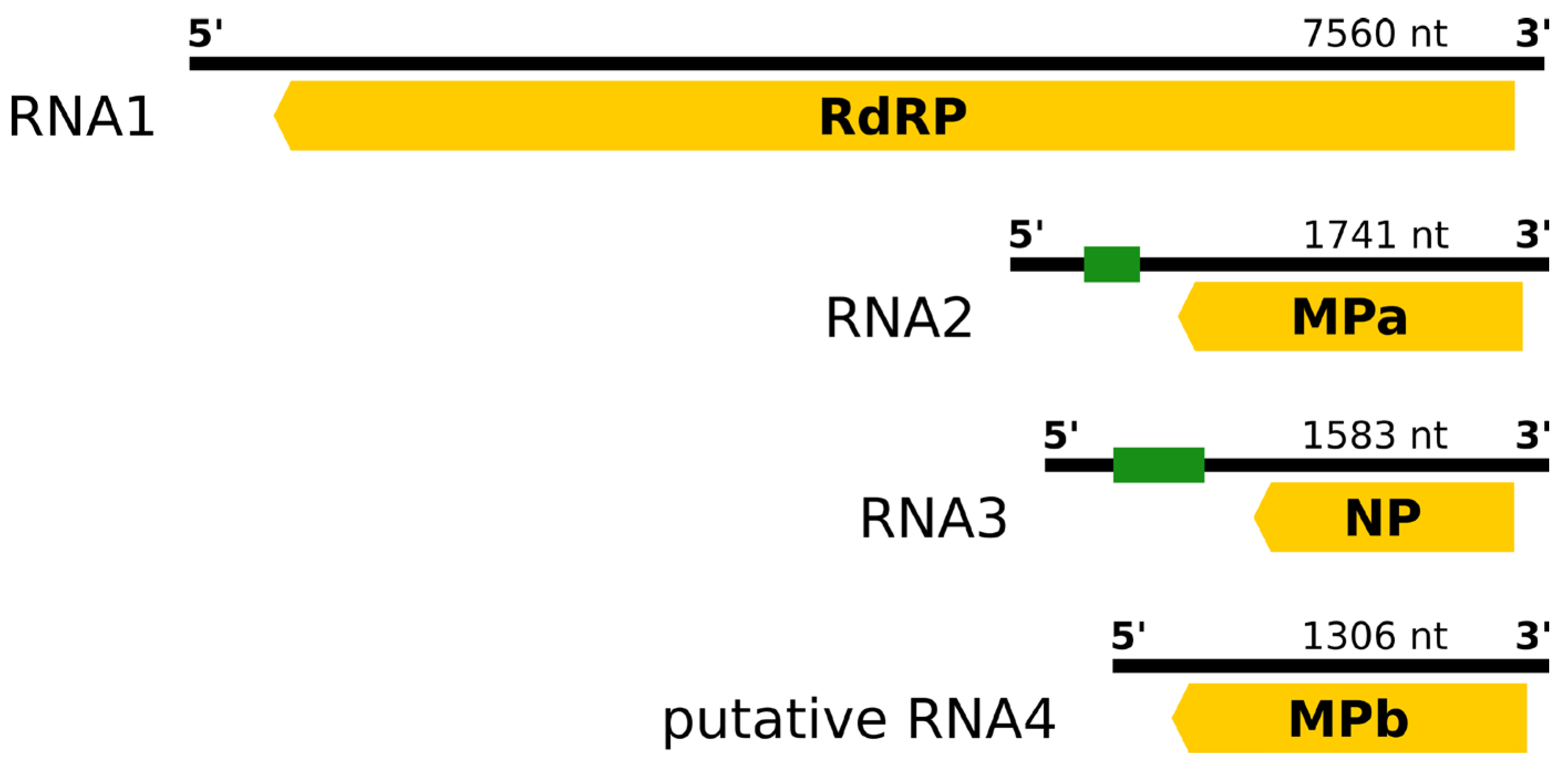
3.2. Genome of RaRV1—Isolate V2
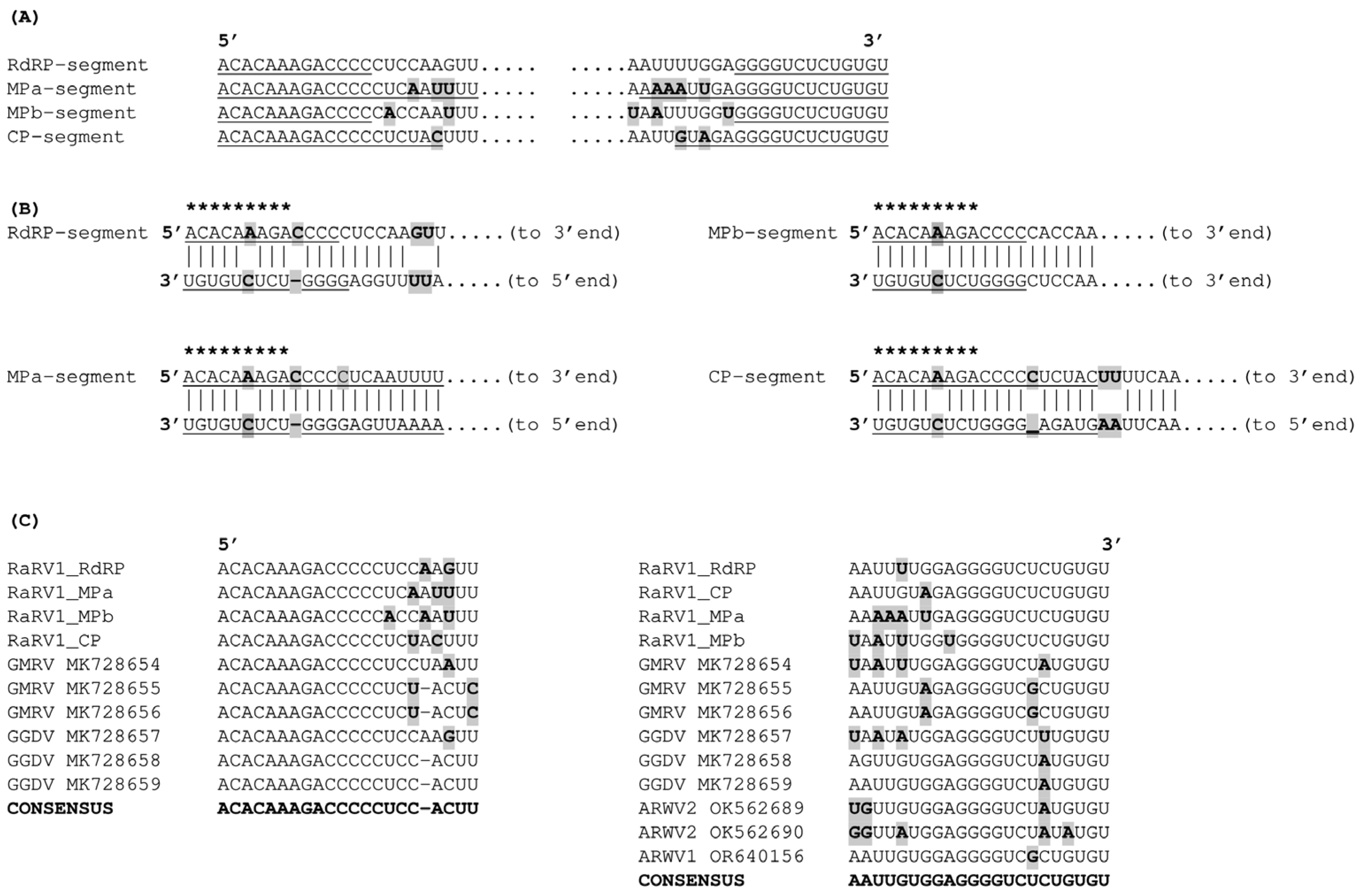
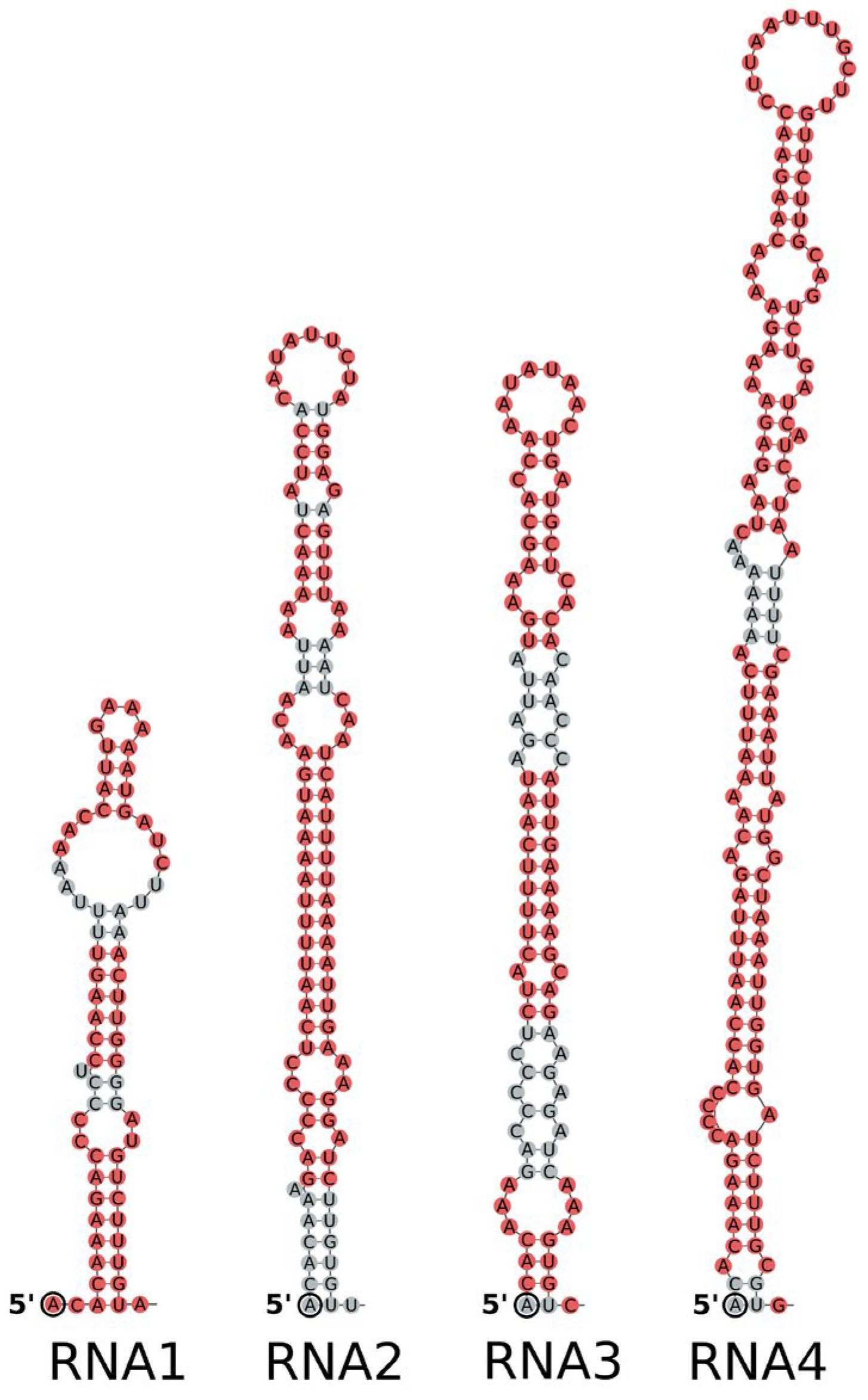
3.3. Genome Termini and Putative Secondary Structures
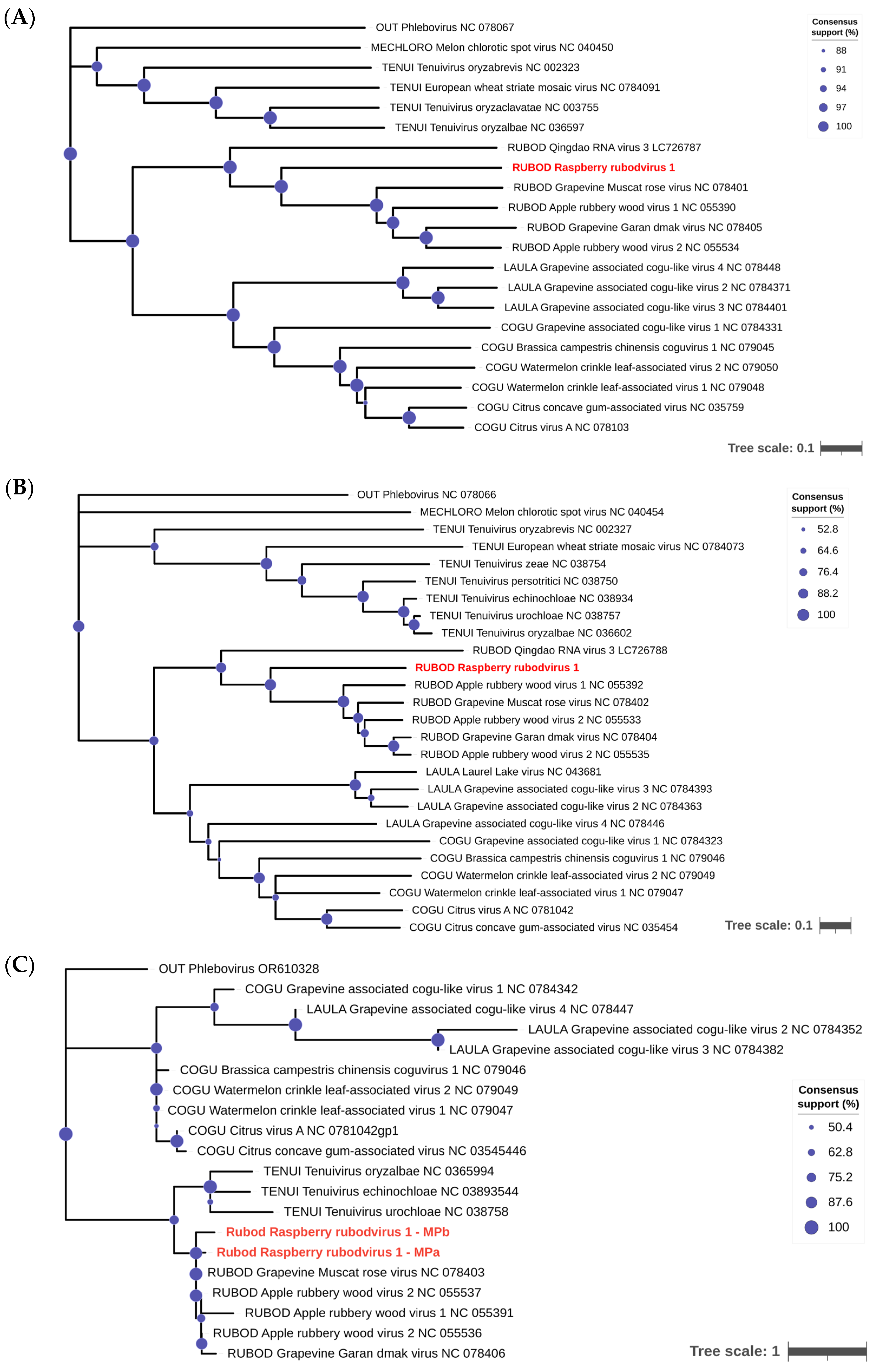
3.4. Phylogeny
3.5. Prevalence of RaRV1
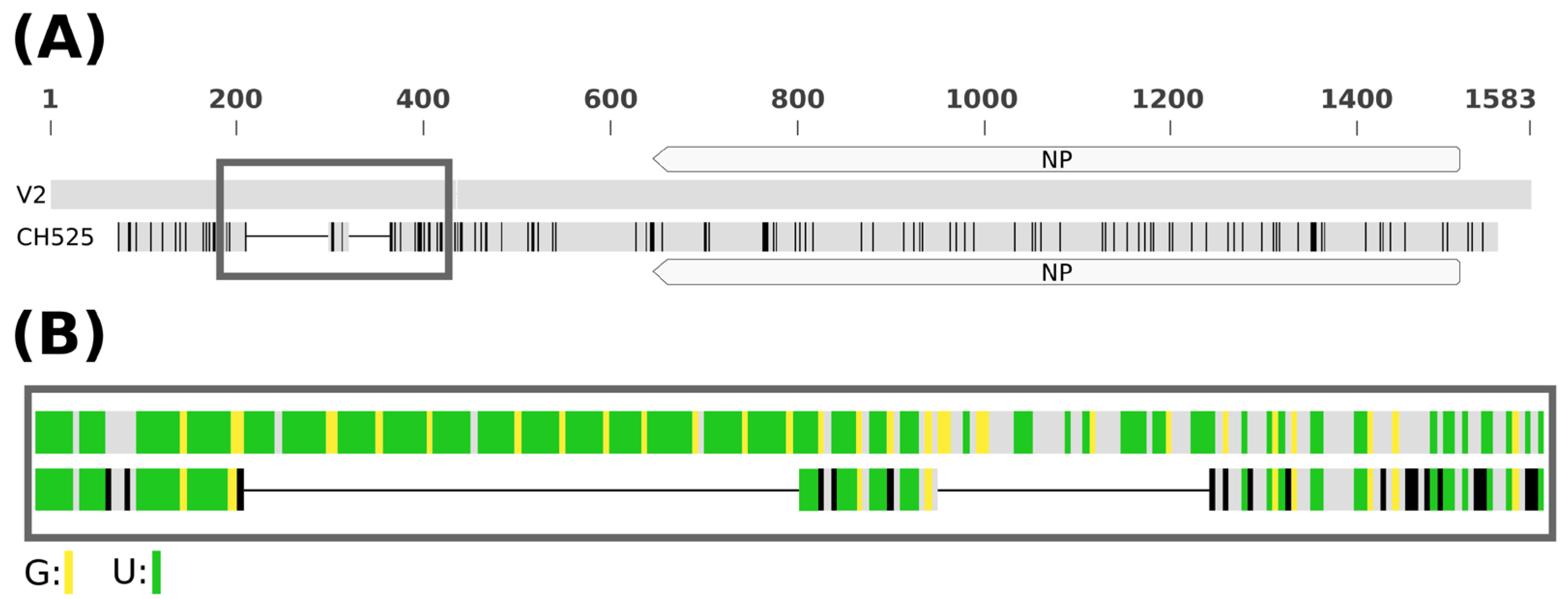
3.6. Variability of RaRV1 Isolates
3.7. Aphid Transmission Experiments
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rott, M.E.; Kesanakurti, P.; Berwarth, C.; Rast, H.; Boyes, I.; Phelan, J.; Jelkmann, W. Discovery of Negative-Sense RNA Viruses in Trees Infected with Apple Rubbery Wood Disease by Next-Generation Sequencing. Plant Dis. 2018, 102, 1254–1263. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Liu, J.; Pang, J.; Kondo, H.; Chi, S.; Zhang, J.; Sun, L.; Andika, I.B. Common but Nonpersistent Acquisitions of Plant Viruses by Plant-Associated Fungi. Viruses 2022, 14, 2279. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Wang, G.; Li, Q.; Zhang, Z.; Li, L.; Lv, Y.; Yang, Z.; Guo, J.; Hong, N. Molecular Characteristics and Incidence of Apple Rubbery Wood Virus 2 and Citrus Virus A Infecting Pear Trees in China. Viruses 2022, 14, 576. [Google Scholar] [CrossRef] [PubMed]
- Nickel, O.; Grynberg, P.; Fajardo, T.V.M. Detection of multiple viruses and viroid in apple trees in Brazil and their possible association with decline. Australasian Plant Dis. Notes 2023, 18, 10. [Google Scholar] [CrossRef]
- Sasaya, T.; Palacios, G.; Briese, T.; Di Serio, F.; Groschup, M.H.; Neriya, Y.; Song, J.W.; Tomitaka, Y. ICTV Virus Taxonomy Profile: Phenuiviridae 2023. J. Gen. Virol. 2023, 104, 001893. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Lara, A.; Navarro, B.; Di Serio, F.; Stevens, K.; Hwang, M.S.; Kohl, J.; Vu, S.T.; Falk, B.W.; Golino, D.; Al Rwahnih, M. Two Novel Negative-Sense RNA Viruses Infecting Grapevine Are Members of a Newly Proposed Genus within the Family Phenuiviridae. Viruses 2019, 11, 685. [Google Scholar] [CrossRef] [PubMed]
- Chiapello, M.; Rodríguez-Romero, J.; Nerva, L.; Forgia, M.; Chitarra, W.; Ayllón, M.; Turina, M. Putative new plant viruses associated with Plasmopara viticola-infected grapevine samples. Ann. Appl. Biol. 2020, 176, 180–191. [Google Scholar] [CrossRef]
- Navarro, B.; Minutolo, M.; De Stradis, A.; Palmisano, F.; Alioto, D.; Di Serio, F. The first phlebo-like virus infecting plants: A case study on the adaptation of negative-stranded RNA viruses to new hosts. Mol. Plant Pathol. 2018, 19, 1075–1089. [Google Scholar] [CrossRef]
- Xiong, R.; Wu, J.; Zhou, Y.; Zhou, X. Identification of a movement protein of the tenuivirus rice stripe virus. J. Virol. 2008, 82, 12304–12311. [Google Scholar] [CrossRef]
- Gaafar, Y.Z.A.; Richert-Pöggeler, K.R.; Sieg-Müller, A.; Lüddecke, P.; Herz, K.; Hartrick, J.; Seide, Y.; Vetten, H.J.; Ziebell, H. A divergent strain of melon chlorotic spot virus isolated from black medic (Medicago lupulina) in Austria. Virol. J. 2019, 16, 89. [Google Scholar] [CrossRef]
- Lim, S.; Baek, D.; Moon, J.S.; Cho, I.S.; Choi, G.S.; Do, Y.S.; Lee, D.H.; Lee, S.H. First report of Apple luteovirus 1 and Apple rubbery wood virus 1 on apple tree rootstocks in Korea. Plant Dis. 2019, 103, 591. [Google Scholar] [CrossRef]
- Bester, R.; Bougard, K.; Maree, H.J. First report of apple rubbery wood virus 2 infecting apples (Malus domestica) in South Africa. J. Plant Pathol. 2022, 104, 1199–1200. [Google Scholar] [CrossRef]
- Bougard, K.; Maree, H.J.; Pietersen, G.; Meitz-Hopkins, J.; Bester, R. First Report of apple rubbery wood virus 2 Infecting Pear (Pyrus communis) in South Africa. Plant Dis. 2022, 106, 1535. [Google Scholar] [CrossRef]
- Fontdevila Pareta, N.; Lateur, M.; Steyer, S.; Blouin, A.G.; Massart, S. First Reports of Apple Luteovirus 1, apple rubbery wood virus 1 and Apple Hammerhead Viroid Infecting Apples in Belgium. New Dis. Rep. 2022, 45, e12076. [Google Scholar] [CrossRef]
- Kaur, K.; Rinaldo, A.; Rodoni, B.R.; Constable, F.E. First report of Grapevine rubodvirus 2 infecting Vitis vinifera cv. Malbec in Australia. New Dis. Rep. 2023, 47, e12172. [Google Scholar] [CrossRef]
- Minutolo, M.; Cinque, M.; Di Serio, F.; Navarro, B.; Alioto, D. Occurrence of apple rubbery wood virus 1 and apple rubbery wood virus 2 in pear and apple in Campania (Southern Italy) and development of degenerate primers for the rapid detection of rubodviruses. J. Plant Pathol. 2023, 105, 567–572. [Google Scholar] [CrossRef]
- Khan, Z.A.; Diksha, D.; Thapa, P.; Mailem, Y.S.; Sharma, S.K.; Gupta, N.; Kishan, G.; Watpade, S.; Baranwal, V.K. Genome analysis of viruses of Phenuiviridae, Betaflexiviridae and Bromoviridae, and apple scar skin viroid in pear by high-throughput sequencing revealing host expansion of a rubodvirus and an ilarvirus. Physiol. Mol. Plant Pathol. 2024, 129, 102196. [Google Scholar] [CrossRef]
- Syller, J. Facilitative and antagonistic interactions between plant viruses in mixed infections. Mol. Plant Pathol. 2012, 13, 204–216. [Google Scholar] [CrossRef]
- Chávez-Calvillo, G.; Contreras-Paredes, C.A.; Mora-Macias, J.; Noa-Carrazana, J.C.; Serrano-Rubio, A.A.; Dinkova, T.D.; Carrillo-Tripp, M.; Silva-Rosales, L. Antagonism or synergism between papaya ringspot virus and papaya mosaic virus in Carica papaya is determined by their order of infection. Virology 2016, 489, 179–191. [Google Scholar] [CrossRef]
- Postman, J.D.; Tzanetakis, I.E.; Martin, R.R. First Report of Strawberry latent ringspot virus in a Mentha sp. from North America. Plant Dis. 2004, 88, 907. [Google Scholar] [CrossRef]
- Ochoa-Corona, F.M.; Lebas, B.S.M.; Tang, J.Z.; Bootten, T.J.; Stewart, F.J.; Harris, R.D.R.; Alexander, B.J.R. RT-PCR detection and strain typing of Raspberry ringspot virus. In Proceedings of the XXth International Symposium on Virus and Virus-like Diseases of Temperate Fruit Crops & XIth International Symposium on Small Fruit Virus Diseases, Antalya, Turkey, 22–26 May 2006. [Google Scholar]
- Jones, A.; Mcgavin, W.; Geering, A.; Lockhart, B. Identification of Rubus yellow net virus as a distinct badnavirus and its detection by PCR in Rubus species and in aphids. Ann. Appl. Biol. 2006, 141, 1–10. [Google Scholar] [CrossRef]
- Tzanetakis, I.E.; Halgren, A.; Mosier, N.; Martin, R.R. Identification and characterization of Raspberry mottle virus, a novel member of the Closteroviridae. Virus Res. 2007, 127, 26–33. [Google Scholar] [CrossRef] [PubMed]
- McGavin, W.J.; Cock, P.J.A.; MacFarlane, S.A. Partial sequence and RT-PCR diagnostic test for the plant rhabdovirus Raspberry vein chlorosis virus. Plant Pathol. 2011, 60, 462–467. [Google Scholar] [CrossRef]
- McGavin, W.J.; Mitchell, C.; Cock, P.J.A.; Wright, K.M.; MacFarlane, S.A. Raspberry leaf blotch virus, a putative new member of the genus Emaravirus, encodes a novel genomic RNA. J. Gen. Virol. 2012, 93 Pt 2, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Koloniuk, I.; Fránová, J.; Přibylová, J.; Sarkisova, T.; Špak, J.; Tan, J.L.; Zemek, R.; Čmejla, R.; Rejlová, M.; Valentová, L.; et al. Molecular Characterization of a Novel Enamovirus Infecting Raspberry. Viruses 2023, 15, 2281. [Google Scholar] [CrossRef]
- Thompson, J.R.; Wetzel, S.; Klerks, M.M.; Vasková, D.; Schoen, C.D.; Spak, J.; Jelkmann, W. Multiplex RT-PCR detection of four aphid-borne strawberry viruses in Fragaria spp. in combination with a plant mRNA specific internal control. J. Virol. Methods 2003, 111, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Schwartz, S.; Wagner, L.; Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol. 2000, 7, 203–214. [Google Scholar] [CrossRef]
- Hallgren, J.; Tsirigos, K.D.; Pedersen, M.D.; Armenteros, J.J.A.; Marcatili, P.; Nielsen, H.; Krogh, A.; Winther, O. DeepTMHMM predicts alpha and beta transmembrane proteins using deep neural networks. bioRxiv 2022. [Google Scholar] [CrossRef]
- Dobson, L.; Reményi, I.; Tusnády, G.E. CCTOP: A Consensus Constrained TOPology prediction web server. Nucleic Acids Res. 2015, 43, W408–W412. [Google Scholar] [CrossRef]
- Paysan-Lafosse, T.; Blum, M.; Chuguransky, S.; Grego, T.; Pinto, B.L.; Salazar, G.A.; Bileschi, M.L.; Bork, P.; Bridge, A.; Colwell, L.; et al. InterPro in 2022. Nucleic Acids Res. 2022, D1, D418–D427. [Google Scholar] [CrossRef]
- Lorenz, R.; Bernhart, S.H.; Höner Zu Siederdissen, C.; Tafer, H.; Flamm, C.; Stadler, P.F.; Hofacker, I.L. ViennaRNA Package 2.0. Algorithms. Mol. Biol. 2011, 6, 26. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v3: An Online Tool for the Display and Annotation of Phylogenetic and Other Trees. Nucleic Acids Res. 2016, 44, W242–W245. [Google Scholar] [CrossRef]
- Navarro, B.; Zicca, S.; Minutolo, M.; Saponari, M.; Alioto, D.; Di Serio, F. A Negative-Stranded RNA Virus Infecting Citrus Trees: The Second Member of a New Genus within the Order Bunyavirales. Front. Microbiol. 2018, 9, 2340. [Google Scholar] [CrossRef] [PubMed]
- Albariño, C.G.; Bird, B.H.; Nichol, S.T. A shared transcription termination signal on negative and ambisense RNA genome segments of Rift Valley fever, sandfly fever Sicilian, and Toscana viruses. J. Virol. 2007, 81, 5246–5256. [Google Scholar] [CrossRef] [PubMed]
- Malet, H.; Williams, H.M.; Cusack, S.; Rosenthal, M. The mechanism of genome replication and transcription in bunyaviruses. PLoS Pathog. 2023, 19, e1011060. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tian, X.; Navarro, B.; Di Serio, F.; Cao, M. Watermelon crinkle leaf-associated virus 1 and watermelon crinkle leaf-associated virus 2 have a bipartite genome with molecular signatures typical of the members of the genus Coguvirus (family Phenuiviridae). Arch. Virol. 2021, 166, 2829–2834. [Google Scholar] [CrossRef] [PubMed]
- Coll, M. Feeding and ovipositing on plants by an omnivorous insect predator. Oecologia 1996, 105, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Yano, E. Biological control using zoophytophagous bugs in Japan. J. Pest. Sci. 2022, 95, 1473–1484. [Google Scholar] [CrossRef]
- Martin, R.R.; MacFarlane, S.; Sabanadzovic, S.; Quito, D.; Poudel, B.; Tzanetakis, I.E. Viruses and virus diseases of Rubus. Plant Dis. 2013, 97, 168–182. [Google Scholar] [CrossRef]

| Segment | Contig | Maximal Blastx Hits | |||||
|---|---|---|---|---|---|---|---|
| Length (nt) | Number of Reads | Coverage (Reads/Base) | Virus | Accession | Identity (%) | Coverage (%) | |
| RNA1 | 7728 | 4612 | 89.4 | ARWV2 | AWC67514 | 37.6 | 83.3 |
| RNA2 | 1755 | 1758 | 150.3 | ARWV2 | UOA05155 | 50.9 | 45.3 |
| RNA3 | 1571 | 5314 | 504.3 | ARWV2 | UOA05158 | 45.8 | 52.7 |
| RNA4 | 1295 | 1434 | 165.3 | ARWV2 | WBB27576 | 23.9 | 65.8 |
| Virus | RdRP (2476 aa) | MPa (437 aa) | NP (287 aa) | MPb 1 (359 aa) |
|---|---|---|---|---|
| ARWV1 | 34.5 | 36.5 | 42.6 | 19.1 |
| ARWV2 | 34.2 | 36.6 | 45.3 | 19.7 |
| GGDV | 31.6 | 34.5 | 41.2 | 18.2 |
| GMRV | 31.1 | 35.2 | 42.5 | 18.4 |
| QRV3 | 24.8 | n.a. | 24.4 | n.a. |
| RaRV1-MPa 2 | n.a. | n.a. | n.a. | 19.0 |
| Sample | Location | GPS | Cultivar | Other Viruses Detected | Symptoms | Photo |
|---|---|---|---|---|---|---|
| V2 | Volanice | 50.3351314N 15.3996269E | Canby | RBDV, RVCV | Vein clearing, mosaic, leaf curl | B |
| V4 | Volanice | 50.3351314N 15.3996269E | Canby | RBDV, RVCV | Vein clearing, mosaic, leaf curl | C |
| V5 | Volanice | 50.3351314N 15.3996269E | Canby | RBDV, RVCV | Vein clearing, yellowing | D |
| MC1 | Míčov | 49.9036772N 15.6046294E | Unknown | RaEV1, RVCV | Vein clearing, leaf curl, necrosis | E |
| MC2 | Míčov | 49.9036772N 15.6046294E | Unknown | RBDV, RVCV | Vein clearing, yellowing/reddening, necrosis | F |
| CH525 | Chlum | 50.3792903N 15.6024481E | Unknown | BRNV, RVCV | Vein clearing, mosaic | G |
| NB1 | Nový Bydžov | 50.2345183N 15.4883225E | Bulharský rubín | RaEV1, RBDV, RLBV, RVCV | Yellow blotches, leaf malformation | H |
| D23 | Doudleby | 50.1014906N, 16.2515422E | Canby | BRNV, RBDV, RLMV | No symptoms * | I |
| H1 | Holovousy | Unknown 1 | Malling Jewel 4 | BRNV, RBDV, RLMV | Leaf curling | J |
| O4M | Ostroměř | 50.3722167N, 15.5470811E | Unknown | RaEV1 | Vein clearing, yellowing | K |
| Isolate | Locality | MPa | MPb | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Detected | acc.no. | Length (nt) | nt/aa Changes | Detected | acc.no. | Length (nt) | nt/aa Changes | ||
| V2 | Volanice | yes | PP732067 | 1315 | 0/0 | yes | PP732068 | 1080 | 0/0 |
| V4 | Volanice | yes | PP934000 | 705 | 1/0 | yes | PP942705 | 417 | 1/0 |
| V5 | Volanice | yes | PP934001 | 705 | 1/0 | yes | PP934002 | 1054 | 3/2 |
| MC1 | Míčov | yes | PP934003 | 705 | 1/0 | yes | PP934004 | 416 | 3/2 |
| MC2 | Míčov | yes | PP934005 | 688 | 1/0 | yes | PP934006 | 419 | 2/1 |
| O4M | Ostroměř | yes | PP977435 1 | 688 | 0/0 | yes | PP977434 2 | 1038 | 4/3 |
| NB1 | Nový Bydžov | yes | PP942707 | 705 | 1/STOP 3 | no 2 | --- | --- | --- |
| H1 | Holovousy | yes | PP942706 | 705 | 1/0 | no 2 | --- | --- | --- |
| CH525 | Chlum | no 1 | --- | --- | --- | no 2 | --- | --- | --- |
| D23 | Doudleby | no 1 | --- | --- | --- | no 2 | --- | --- | --- |
| O. minutus | Volanice | no 1 | --- | --- | --- | yes | PP933998 | 434 | 1/0 |
| A. idaei | Volanice | no 1 | --- | --- | --- | yes | PP933999 | 422 | 1/0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenz, O.; Koloniuk, I.; Sarkisová, T.; Čmejla, R.; Valentová, L.; Rejlová, M.; Sedlák, J.; Blystad, D.-R.; Sapkota, B.; Hamborg, Z.; et al. Molecular Characterization of a Novel Rubodvirus Infecting Raspberries. Viruses 2024, 16, 1074. https://doi.org/10.3390/v16071074
Lenz O, Koloniuk I, Sarkisová T, Čmejla R, Valentová L, Rejlová M, Sedlák J, Blystad D-R, Sapkota B, Hamborg Z, et al. Molecular Characterization of a Novel Rubodvirus Infecting Raspberries. Viruses. 2024; 16(7):1074. https://doi.org/10.3390/v16071074
Chicago/Turabian StyleLenz, Ondřej, Igor Koloniuk, Tatiana Sarkisová, Radek Čmejla, Lucie Valentová, Martina Rejlová, Jiří Sedlák, Dag-Ragnar Blystad, Bijaya Sapkota, Zhibo Hamborg, and et al. 2024. "Molecular Characterization of a Novel Rubodvirus Infecting Raspberries" Viruses 16, no. 7: 1074. https://doi.org/10.3390/v16071074
APA StyleLenz, O., Koloniuk, I., Sarkisová, T., Čmejla, R., Valentová, L., Rejlová, M., Sedlák, J., Blystad, D.-R., Sapkota, B., Hamborg, Z., Tan, J. L., Zemek, R., Jaroslava, P., & Fránová, J. (2024). Molecular Characterization of a Novel Rubodvirus Infecting Raspberries. Viruses, 16(7), 1074. https://doi.org/10.3390/v16071074








