Transcriptomic Analysis of mRNA Expression Profiles in the Microglia of Mouse Brains Infected with Rabies Viruses of Varying Virulence
Abstract
1. Introduction
2. Materials and Methods
2.1. Viruses
2.2. Virus Titration
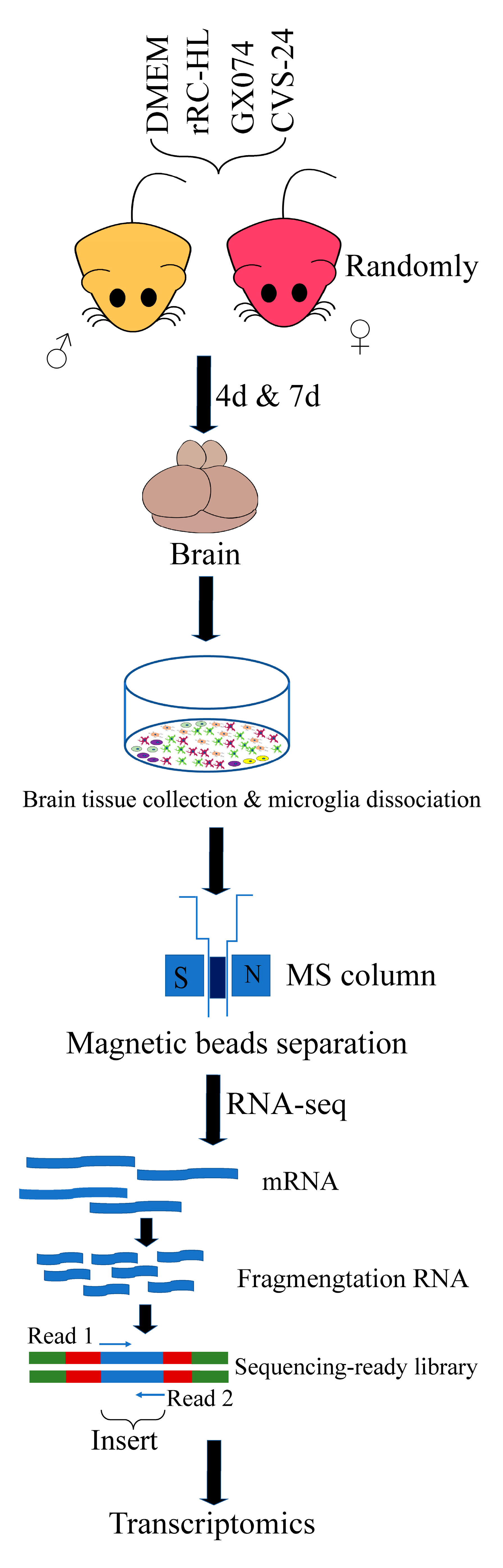
2.3. Mouse Infection Test
2.4. Brain Sample Collection and Microglia Dissociation
2.5. Microglia Purity and Morphological Analysis
2.6. RNA Extraction, cDNA Library Construction, Sequencing and Data Processing
2.7. RT-qPCR for mRNA Sequencing Validation
2.8. Bioinformatics Analysis
3. Results
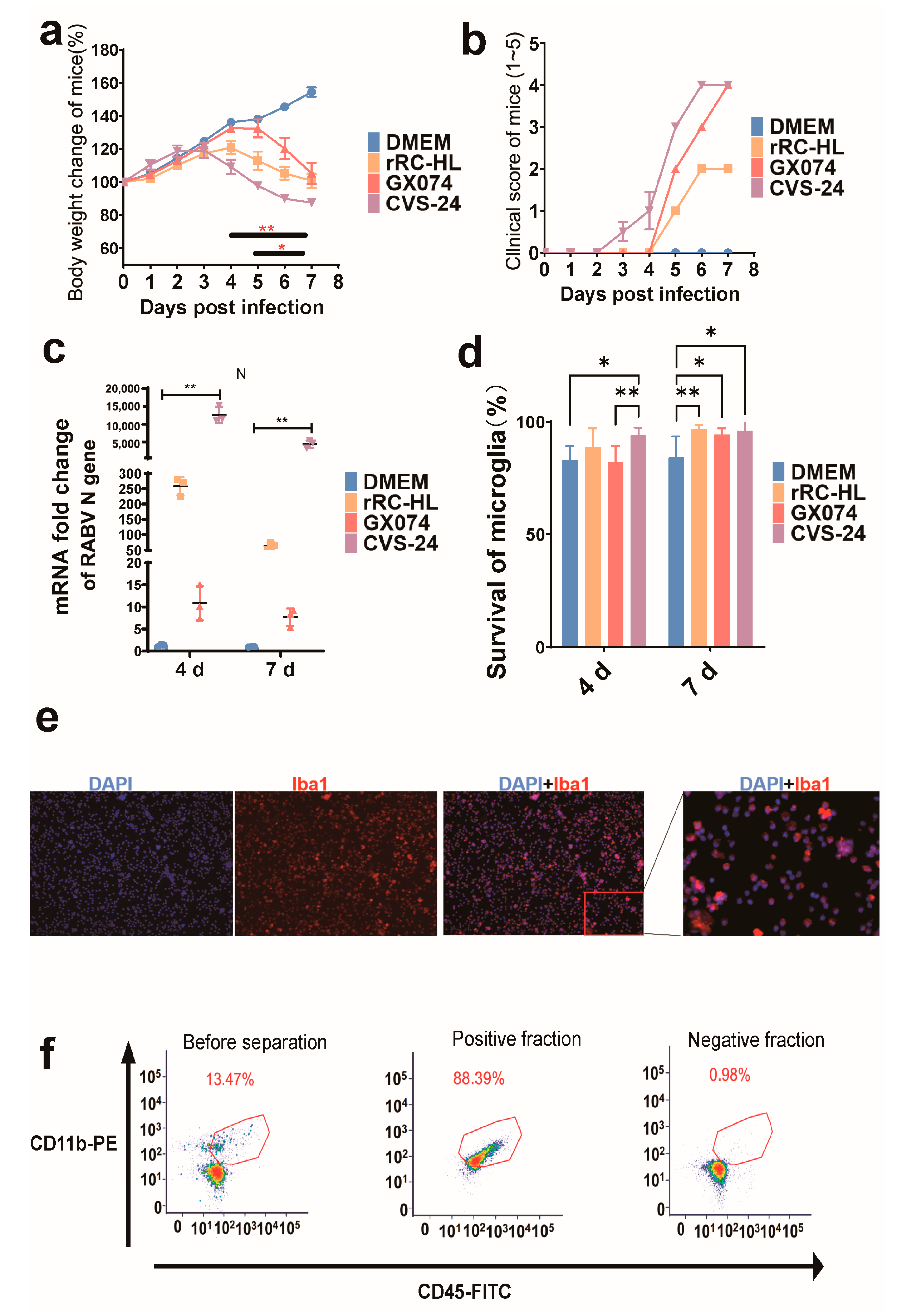
3.1. Mouse Infection Test
3.2. Separation and Identification of Microglia
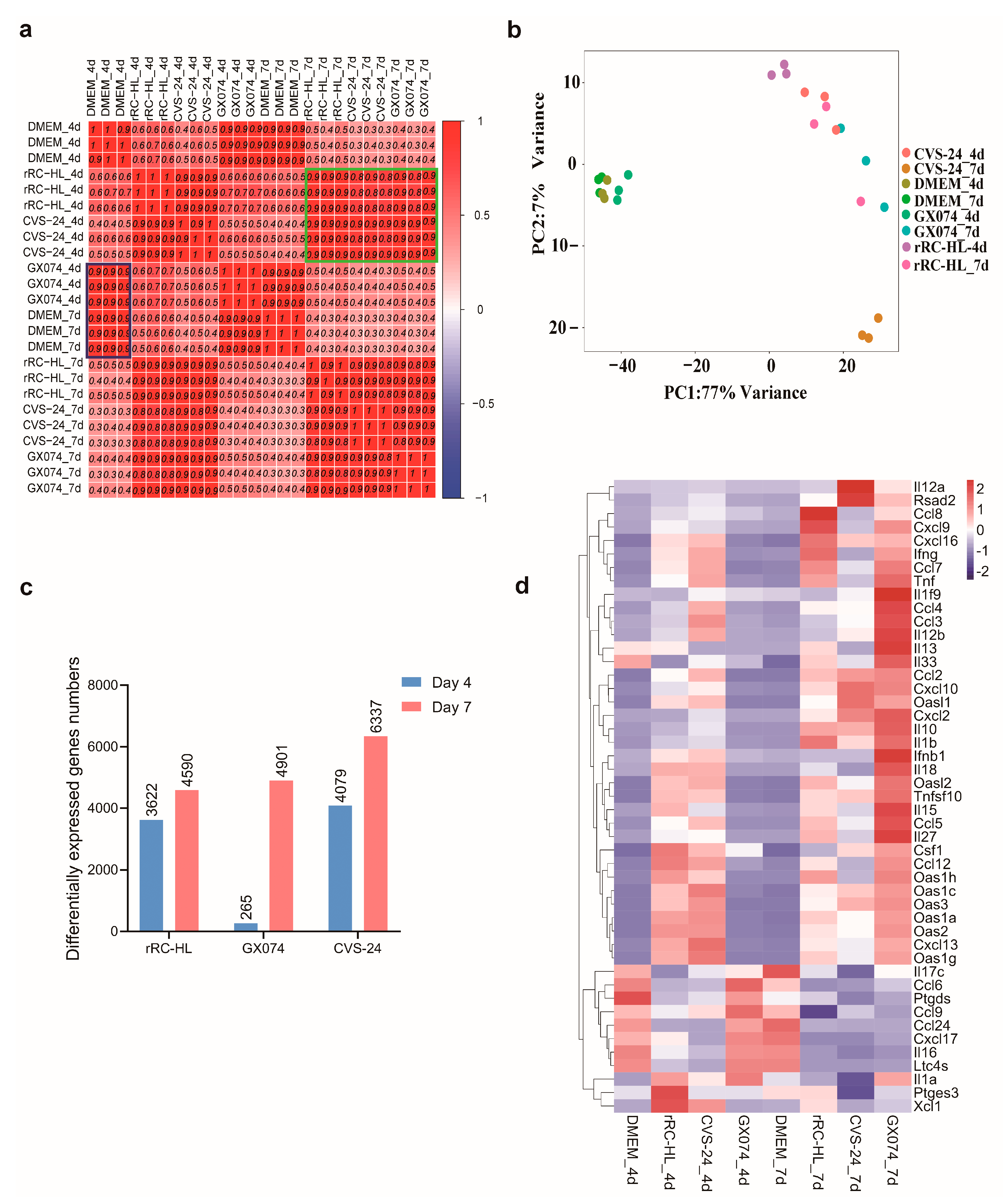
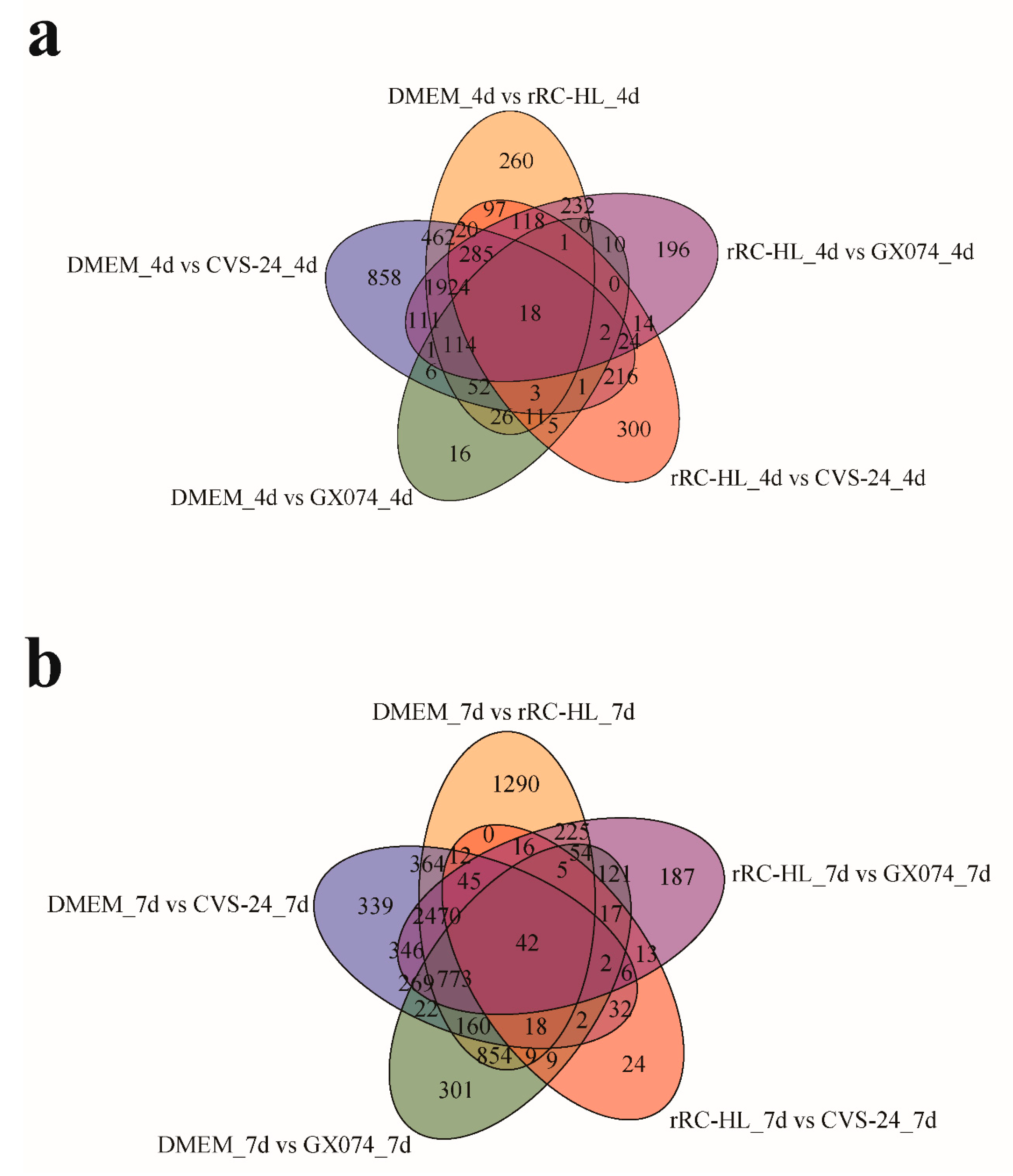
3.3. Determination of Differentially Expressed mRNAs in Microglia from RABV-Infected Brains
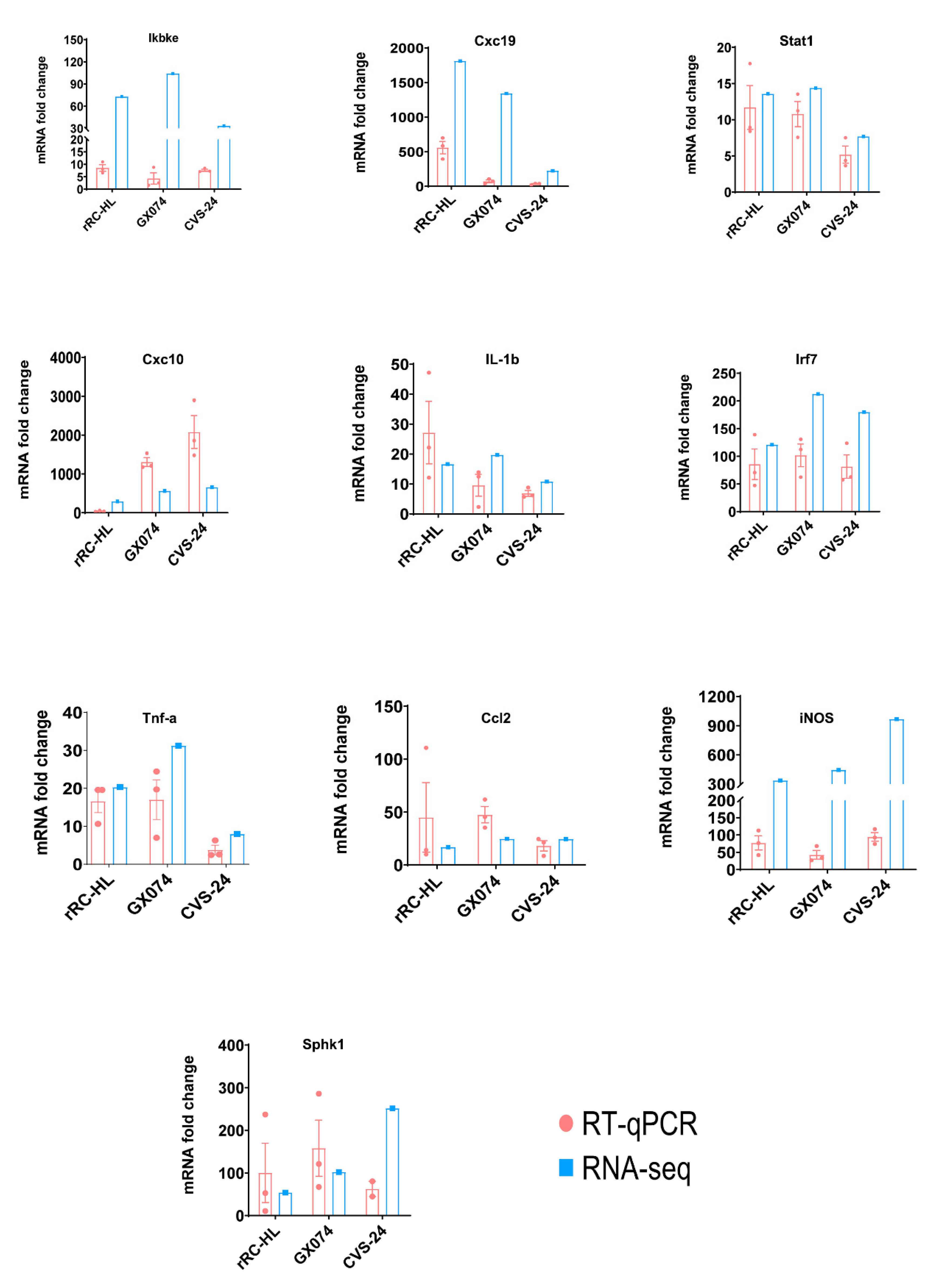
3.4. RT-qPCR Validation of mRNA Expression in Microglia
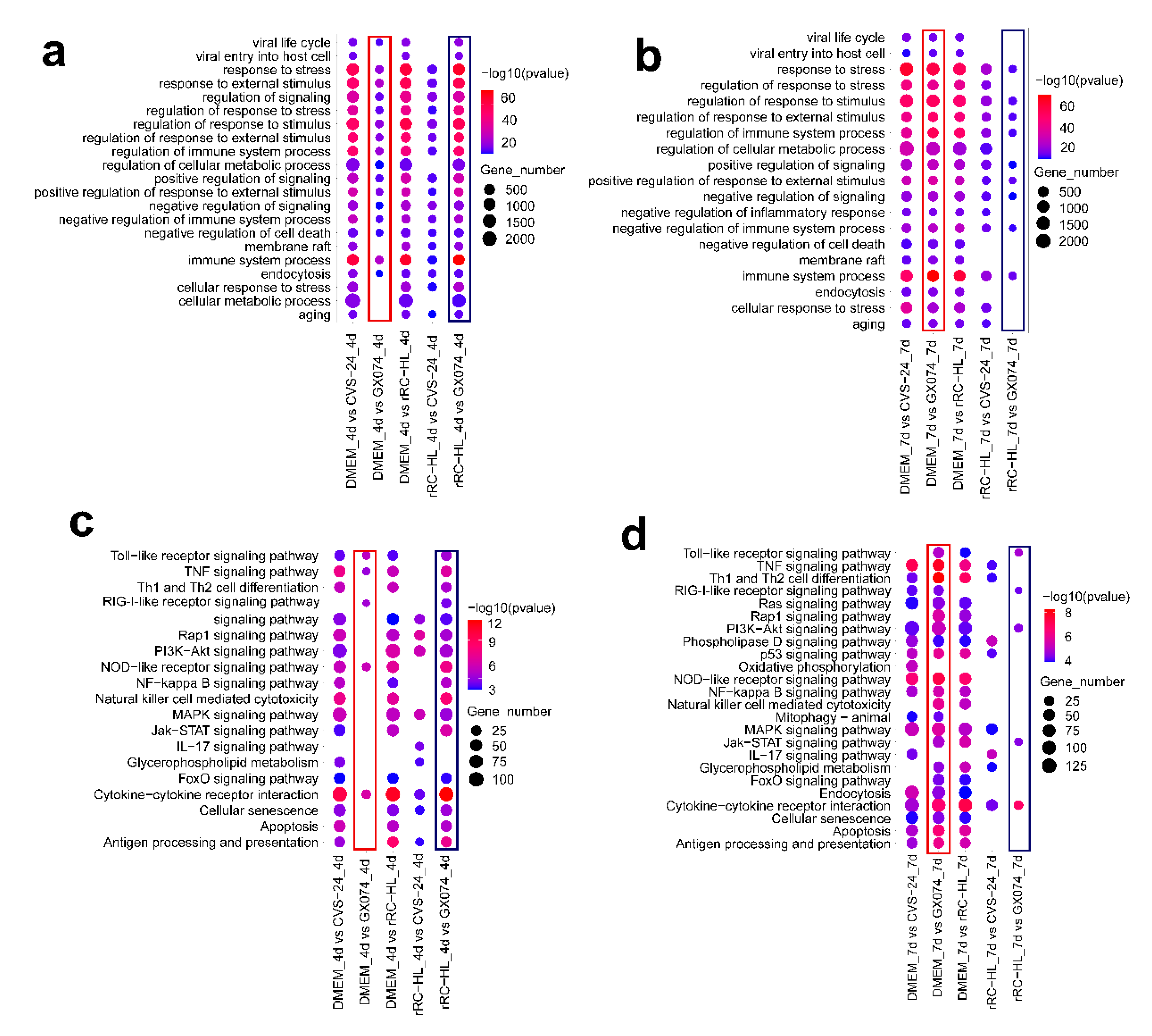
3.5. Functional Prediction and Signaling Pathway Analysis via GO and KEGG Enrichment Analysis
3.6. PPI Network Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fooks, A.R.; Cliquet, F.; Finke, S.; Freuling, C.; Hemachudha, T.; Mani, R.S.; Müller, T.; Nadin-Davis, S.; Picard-Meyer, E.; Wilde, H.; et al. Rabies. Nat. Rev. Dis. Primers 2017, 3, 17091. [Google Scholar] [CrossRef] [PubMed]
- Fooks, A.R.; Banyard, A.C.; Horton, D.L.; Johnson, N.; McElhinney, L.M.; Jackson, A.C. Current Status of Rabies and Prospects for Elimination. Lancet 2014, 384, 1389–1399. [Google Scholar] [CrossRef] [PubMed]
- Finke, S.; Conzelmann, K.-K. Replication Strategies of Rabies Virus. Virus Res. 2005, 111, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Saitgareeva, A.R.; Bulygin, K.V.; Gareev, I.F.; Beylerli, O.A.; Akhmadeeva, L.R. The Role of Microglia in the Development of Neurodegeneration. Neurol. Sci. 2020, 41, 3609–3615. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Nicola, D.; Perry, V.H. Microglial Dynamics and Role in the Healthy and Diseased Brain: A Paradigm of Functional Plasticity. Neuroscientist 2015, 21, 169–184. [Google Scholar] [CrossRef]
- Nakamichi, K.; Saiki, M.; Sawada, M.; Takayama-Ito, M.; Yamamuro, Y.; Morimoto, K.; Kurane, I. Rabies Virus-Induced Activation of Mitogen-Activated Protein Kinase and NF-KB Signaling Pathways Regulates Expression of CXC and CC Chemokine Ligands in Microglia. J. Virol. 2005, 79, 11801–11812. [Google Scholar] [CrossRef]
- Zhao, P.; Liu, S.; Zhong, Z.; Jiang, T.; Weng, R.; Xie, M.; Yang, S.; Xia, X. Analysis of Expression Profiles of Long Noncoding RNAs and MRNAs in Brains of Mice Infected by Rabies Virus by RNA Sequencing. Sci. Rep. 2018, 8, 11858. [Google Scholar] [CrossRef]
- Sui, B.; Chen, D.; Liu, W.; Tian, B.; Lv, L.; Pei, J.; Wu, Q.; Zhou, M.; Fu, Z.F.; Zhang, Y.; et al. Comparison of LncRNA and MRNA Expression in Mouse Brains Infected by a Wild-Type and a Lab-Attenuated Rabies Lyssavirus. J. Gen. Virol. 2021, 102, 001538. [Google Scholar] [CrossRef]
- Zhao, P.; Zhao, L.; Zhang, T.; Wang, H.; Qin, C.; Yang, S.; Xia, X. Changes in MicroRNA Expression Induced by Rabies Virus Infection in Mouse Brains. Microb. Pathog. 2012, 52, 47–54. [Google Scholar] [CrossRef]
- Ji, S.; Zhu, M.; Zhang, J.; Cai, Y.; Zhai, X.; Wang, D.; Li, G.; Su, S.; Zhou, J. Microarray Analysis of LncRNA Expression in Rabies Virus Infected Human Neuroblastoma Cells. Infect. Genet. Evol. 2019, 67, 88–100. [Google Scholar] [CrossRef]
- Kim, S.; Larrous, F.; Varet, H.; Legendre, R.; Feige, L.; Dumas, G.; Matsas, R.; Kouroupi, G.; Grailhe, R.; Bourhy, H. Early Transcriptional Changes in Rabies Virus-Infected Neurons and Their Impact on Neuronal Functions. Front. Microbiol. 2021, 12, 730892. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Sankowski, R.; Staszewski, O.; Böttcher, C.; Amann, L.; Sagar; Scheiwe, C.; Nessler, S.; Kunz, P.; van Loo, G.; et al. Spatial and Temporal Heterogeneity of Mouse and Human Microglia at Single-Cell Resolution. Nature 2019, 566, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Jordão, M.J.C.; Sankowski, R.; Brendecke, S.M.; Sagar; Locatelli, G.; Tai, Y.-H.; Tay, T.L.; Schramm, E.; Armbruster, S.; Hagemeyer, N.; et al. Single-Cell Profiling Identifies Myeloid Cell Subsets with Distinct Fates during Neuroinflammation. Science 2019, 363, eaat7554. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xing, X.; Long, B.; Cao, Y.; Hu, S.; Li, X.; Yu, Y.; Tian, D.; Sui, B.; Luo, Z.; et al. A Spatial and Cellular Distribution of Rabies Virus Infection in the Mouse Brain Revealed by FMOST and Single-Cell RNA Sequencing. Clin. Transl. Med. 2022, 12, e700. [Google Scholar] [CrossRef]
- Ito, N.; Kakemizu, M.; Ito, K.A.; Yamamoto, A.; Yoshida, Y.; Sugiyama, M.; Minamoto, N. A Comparison of Complete Genome Sequences of the Attenuated RC-HL Strain of Rabies Virus Used for Production of Animal Vaccine in Japan, and the Parental Nishigahara Strain. Microbiol. Immunol. 2001, 45, 51–58. [Google Scholar] [CrossRef]
- Tang, H.-B.; Pan, Y.; Wei, X.-K.; Lu, Z.-L.; Lu, W.; Yang, J.; He, X.-X.; Xie, L.-J.; Zeng, L.; Zheng, L.-F.; et al. Re-Emergence of Rabies in the Guangxi Province of Southern China. PLoS Negl. Trop. Dis. 2014, 8, e3114. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, Y.; Li, M.; Zhu, J.; Li, X.; Luo, T.R.; Liang, J. Host Desmin Interacts with RABV Matrix Protein and Facilitates Virus Propagation. Viruses 2023, 15, 434. [Google Scholar] [CrossRef]
- Liu, W.; Yang, Y.; Zeng, Z.; Tian, Y.; Wu, Q.; Zhou, M.; Fu, Z.F.; Zhao, L. G Protein-Coupled Receptor 17 Restricts Rabies Virus Replication via BAK-Mediated Apoptosis. Vet. Microbiol. 2022, 265, 109326. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A Simple Method of Estimating Fifty per Cent Endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Chopy, D.; Pothlichet, J.; Lafage, M.; Mégret, F.; Fiette, L.; Si-Tahar, M.; Lafon, M. Ambivalent Role of the Innate Immune Response in Rabies Virus Pathogenesis. J. Virol. 2011, 85, 6657–6668. [Google Scholar] [CrossRef]
- Lee, J.-K.; Tansey, M.G. Microglia Isolation from Adult Mouse Brain. In Microglia; Methods in Molecular Biology; Joseph, B., Venero, J.L., Eds.; Humana Press: Totowa, NJ, USA, 2013; Volume 1041, pp. 17–23. ISBN 978-1-62703-519-4. [Google Scholar]
- Yousef, H.; Czupalla, C.; Lee, D.; Butcher, E.; Wyss-Coray, T. Papain-Based Single Cell Isolation of Primary Murine Brain Endothelial Cells Using Flow Cytometry. Bio-Protocol 2018, 8, e3091. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yu, Q.; Ding, X.; Hu, X.; Hou, K.; Liu, X.; Nie, S.; Xie, M. RNA-Seq Based Elucidation of Mechanism Underlying Ganoderma Atrum Polysaccharide Induced Immune Activation of Murine Myeloid-Derived Dendritic Cells. J. Funct. Foods 2019, 55, 104–116. [Google Scholar] [CrossRef]
- Wen, N.; Guo, B.; Zheng, H.; Xu, L.; Liang, H.; Wang, Q.; Wang, D.; Chen, X.; Zhang, S.; Li, Y.; et al. Bromodomain Inhibitor Jq1 Induces Cell Cycle Arrest and Apoptosis of Glioma Stem Cells through the VEGF/PI3K/AKT Signaling Pathway. Int. J. Oncol. 2019, 55, 879–895. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing Real-Time PCR Data by the Comparative CT Method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Pan, J.; Wan, J. Methodological Comparison of FACS and MACS Isolation of Enriched Microglia and Astrocytes from Mouse Brain. J. Immunol. Methods 2020, 486, 112834. [Google Scholar] [CrossRef]
- Martin, E.; El-Behi, M.; Fontaine, B.; Delarasse, C. Analysis of Microglia and Monocyte-Derived Macrophages from the Central Nervous System by Flow Cytometry. J. Vis. Exp. 2017, 124, 55781. [Google Scholar] [CrossRef]
- Erny, D.; Dokalis, N.; Mezö, C.; Mossad, O.; Blank, T.; Prinz, M. Flow-Cytometry-Based Protocol to Analyze Respiratory Chain Function in Mouse Microglia. STAR Protoc. 2022, 3, 101186. [Google Scholar] [CrossRef]
- Qian, C.; Cao, X. Regulation of Toll-like Receptor Signaling Pathways in Innate Immune Responses: Qian & Cao. Ann. N. Y. Acad. Sci. 2013, 1283, 67–74. [Google Scholar] [CrossRef]
- Newton, K.; Dixit, V.M. Signaling in Innate Immunity and Inflammation. Cold Spring Harb. Perspect. Biol. 2012, 4, a006049. [Google Scholar] [CrossRef]
- Tian, B.; Zhou, M.; Yang, Y.; Yu, L.; Luo, Z.; Tian, D.; Wang, K.; Cui, M.; Chen, H.; Fu, Z.F.; et al. Lab-Attenuated Rabies Virus Causes Abortive Infection and Induces Cytokine Expression in Astrocytes by Activating Mitochondrial Antiviral-Signaling Protein Signaling Pathway. Front. Immunol. 2018, 8, 2011. [Google Scholar] [CrossRef]
- Liu, S.Q.; Xie, Y.; Gao, X.; Wang, Q.; Zhu, W.Y. Inflammatory Response and MAPK and NF-ΚB Pathway Activation Induced by Natural Street Rabies Virus Infection in the Brain Tissues of Dogs and Humans. Virol. J. 2020, 17, 157. [Google Scholar] [CrossRef] [PubMed]
- Feige, L.; Kozaki, T.; Dias de Melo, G.; Guillemot, V.; Larrous, F.; Ginhoux, F.; Bourhy, H. Susceptibilities of CNS Cells towards Rabies Virus Infection Is Linked to Cellular Innate Immune Responses. Viruses 2022, 15, 88. [Google Scholar] [CrossRef] [PubMed]
- Rossiter, J.P.; Jackson, A.C. 9 Pathology. In Rabies, 3rd ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 351–386. [Google Scholar] [CrossRef]
- Li, C.; Zhang, H.; Ji, L.; Wang, X.; Wen, Y.; Li, G.; Fu, Z.F.; Yang, Y. Deficient Incorporation of Rabies Virus Glycoprotein into Virions Enhances Virus-Induced Immune Evasion and Viral Pathogenicity. Viruses 2019, 11, 218. [Google Scholar] [CrossRef] [PubMed]
- Wiltzer, L.; Okada, K.; Yamaoka, S.; Larrous, F.; Kuusisto, H.V.; Sugiyama, M.; Blondel, D.; Bourhy, H.; Jans, D.A.; Ito, N.; et al. Interaction of Rabies Virus P-Protein with STAT Proteins Is Critical to Lethal Rabies Disease. J. Infect. Dis. 2014, 209, 1744–1753. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; He, F.; Bi, S.; Guo, H.; Zhang, B.; Wu, F.; Liang, J.; Yang, Y.; Tian, Q.; Ju, C.; et al. Genome-Wide Transcriptional Profiling Reveals Two Distinct Outcomes in Central Nervous System Infections of Rabies Virus. Front. Microbiol. 2016, 7, 751. [Google Scholar] [CrossRef]
- Kojima, D.; Park, C.-H.; Satoh, Y.; Inoue, S.; Noguchi, A.; Oyamad, T. Pathology of the Spinal Cord of C57BL/6J Mice Infected with Rabies Virus (CVS-11 Strain). J. Vet. Med. Sci. 2009, 71, 319–324. [Google Scholar] [CrossRef]
- Kojima, D.; Park, C.-H.; Tsujikawa, S.; Kohara, K.; Hatai, H.; Oyamad, T.; Noguchi, A.; Inoue, S. Lesions of the Central Nervous System Induced by Intracerebral Inoculation of BALB/c Mice with Rabies Virus (CVS-11). J. Vet. Med. Sci. 2010, 72, 1011–1016. [Google Scholar] [CrossRef]
- Miyamoto, K.; Matsumoto, S. Comparative studies between pathogenesis of street and fixed rabies infection. J. Exp. Med. 1967, 125, 447–456. [Google Scholar] [CrossRef]
- Yoshizumi, T.; Imamura, H.; Taku, T.; Kuroki, T.; Kawaguchi, A.; Ishikawa, K.; Nakada, K.; Koshiba, T. RLR-Mediated Antiviral Innate Immunity Requires Oxidative Phosphorylation Activity. Sci. Rep. 2017, 7, 5379. [Google Scholar] [CrossRef]

| Gene Name | Primer Sequences (5′ to 3′) |
|---|---|
| N-F | GGCATTGGCAGATGATGGAACT |
| N-R | GGCTTGATGATTGGAACTGACTGA |
| Iknke-F | ACCACTAACTACCTGTGGCAT |
| Iknke -R | ACTGCGAATAGCTTCACGATG |
| CXCL9-F | TCCTTTTGGGCATCATCTTCC |
| CXCL9-R | TTTGTAGTGGATCGTGCCTCG |
| CXCL10-F | CCAAGTGCTGCCGTCATTTTC |
| CXCL10-R | GGCTCGCAGGGATGATTTCAA |
| IL-1β-F | ATCTCGCAGCAGCACATC |
| IL-1β-R | TCATCTCGGAGCCTGTAGTG |
| IRF7-F | GAGACTGGCTATTGGGGGAG |
| IRF7-R | GACCGAAATGCTTCCAGGG |
| TNF-α-F | GACGTGGAACTGGCAGAAG |
| TNF-α-R | GTAGACAGAAGAGCGTGGT |
| MCP-1-F | TACAAGAGGATCACCAGCAG |
| MCP-1-R | CTCATTTGGTTCCGATCCAG |
| iNOS-F | TGGAGCGAGTTGTGGATTG |
| iNOS-R | TGAGGGCTTGGCTGAGTGA |
| Stat1-F | TCACAGTGGTTCGAGCTTCAG |
| Stat1-R | GCAAACGAGACATCATAGGCA |
| Sphk1-F | GGCTCTGCAGCTCTTCCAGAG |
| Sphk1-R | CTCCTCTGCACACACCAGCTC |
| β-actin-F | AGACCTCTATGCCAACACAGT |
| β-actin-R | CATCGTACTCCTGCTTGCTGAT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Li, W.; Yu, D.; Jin, R.; Hou, H.; Ling, X.; Kiflu, A.B.; Wei, X.; Yang, X.; Li, X.; et al. Transcriptomic Analysis of mRNA Expression Profiles in the Microglia of Mouse Brains Infected with Rabies Viruses of Varying Virulence. Viruses 2023, 15, 1223. https://doi.org/10.3390/v15061223
Liu J, Li W, Yu D, Jin R, Hou H, Ling X, Kiflu AB, Wei X, Yang X, Li X, et al. Transcriptomic Analysis of mRNA Expression Profiles in the Microglia of Mouse Brains Infected with Rabies Viruses of Varying Virulence. Viruses. 2023; 15(6):1223. https://doi.org/10.3390/v15061223
Chicago/Turabian StyleLiu, Jundan, Wangchang Li, Dongling Yu, Rong Jin, Hualin Hou, Xiaoqing Ling, Abraha Bahlbi Kiflu, Xiankai Wei, Xiaogan Yang, Xiaoning Li, and et al. 2023. "Transcriptomic Analysis of mRNA Expression Profiles in the Microglia of Mouse Brains Infected with Rabies Viruses of Varying Virulence" Viruses 15, no. 6: 1223. https://doi.org/10.3390/v15061223
APA StyleLiu, J., Li, W., Yu, D., Jin, R., Hou, H., Ling, X., Kiflu, A. B., Wei, X., Yang, X., Li, X., He, Y., & Luo, T. R. (2023). Transcriptomic Analysis of mRNA Expression Profiles in the Microglia of Mouse Brains Infected with Rabies Viruses of Varying Virulence. Viruses, 15(6), 1223. https://doi.org/10.3390/v15061223






