Detection of SARS-CoV-2 in Terrestrial Animals in Southern Nigeria: Potential Cases of Reverse Zoonosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
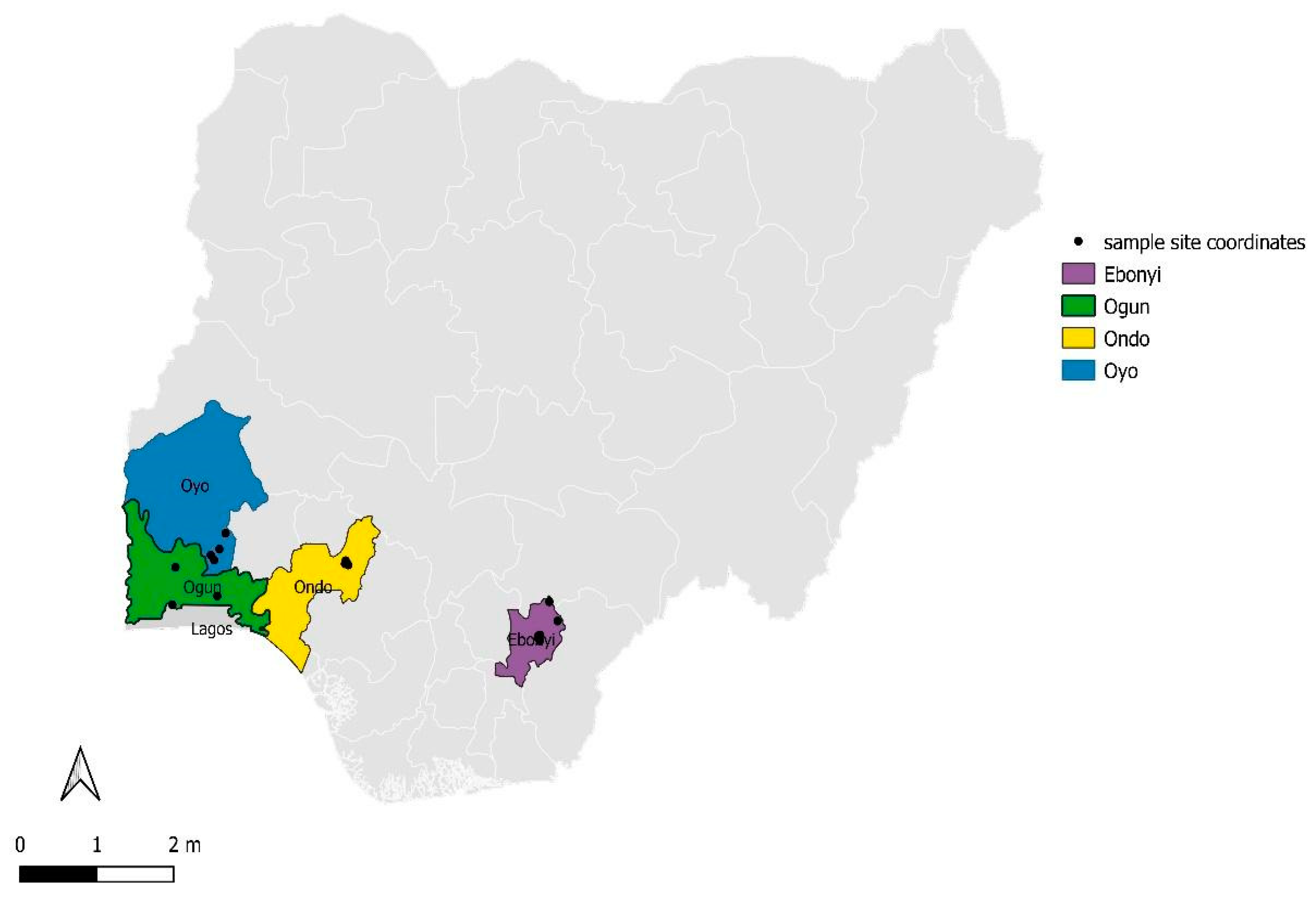
2.2. Study Setting
2.3. Study Design
2.4. RNA Extraction and Real-Time RT-qPCR Detection for SARS-CoV-2
2.5. ELISA for Detection of Antibodies to SARS-CoV-2
2.6. Statistical Analysis
3. Results
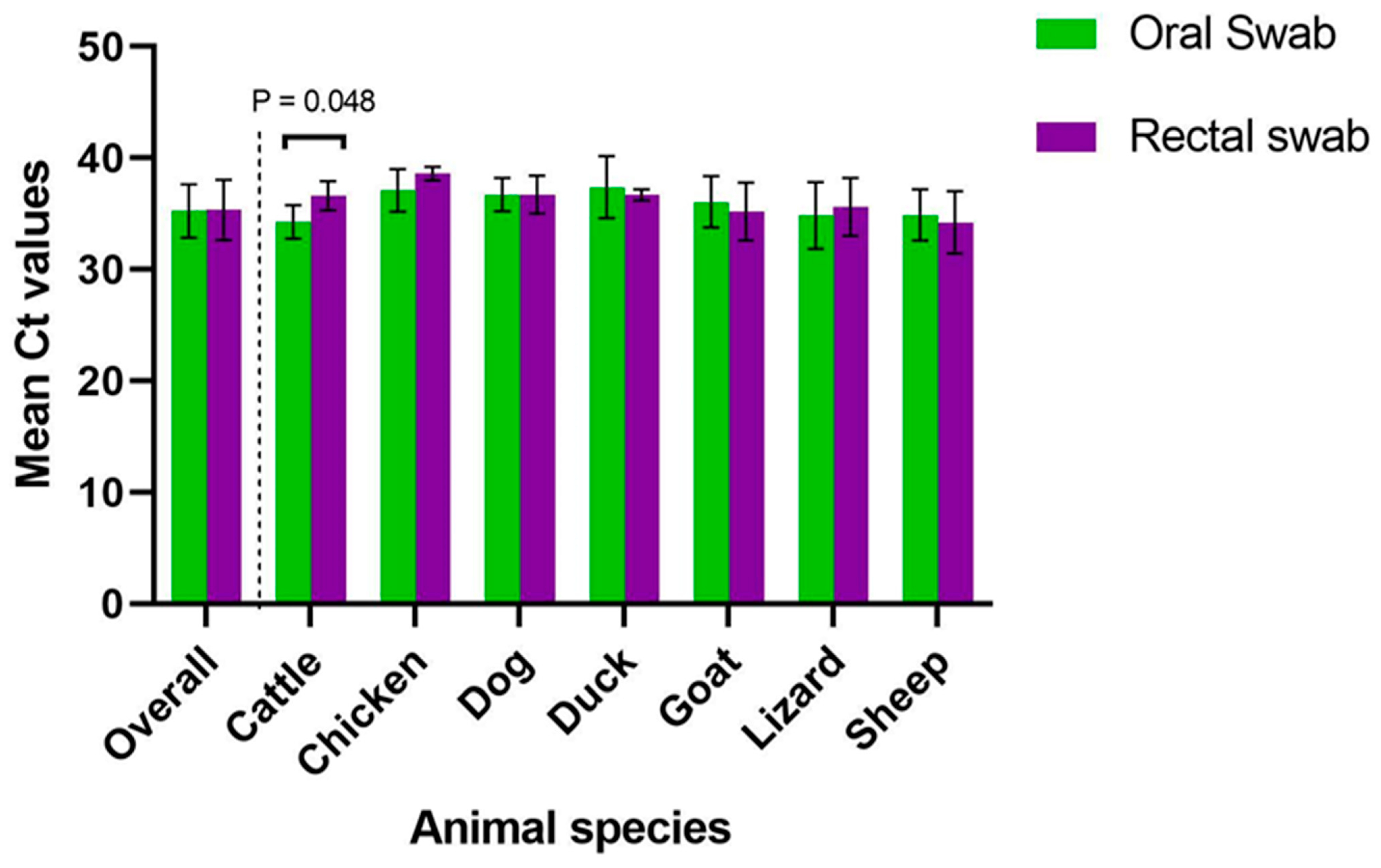
3.1. RT-qPCR Results for SARS-CoV-2 Detection in Animals
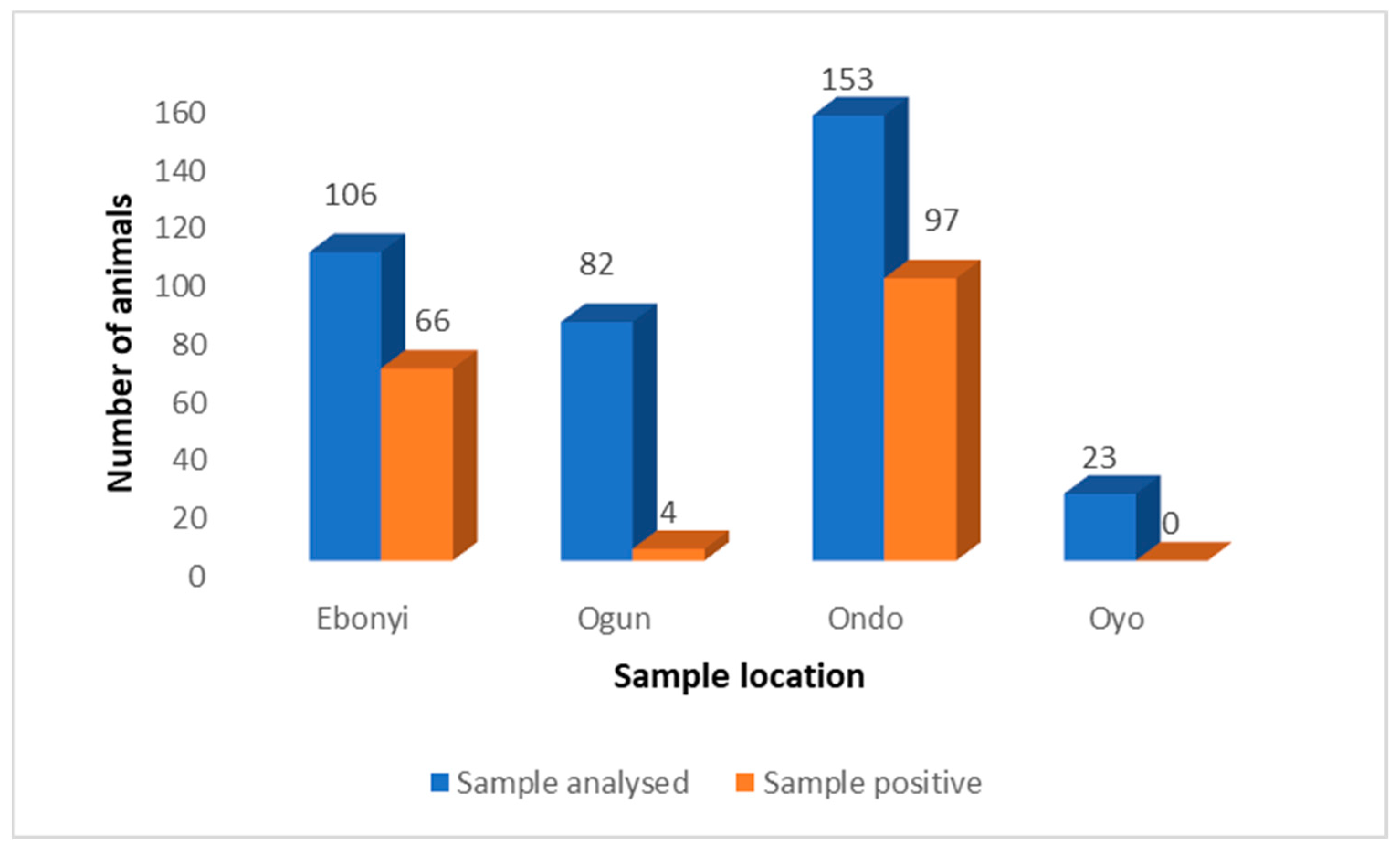
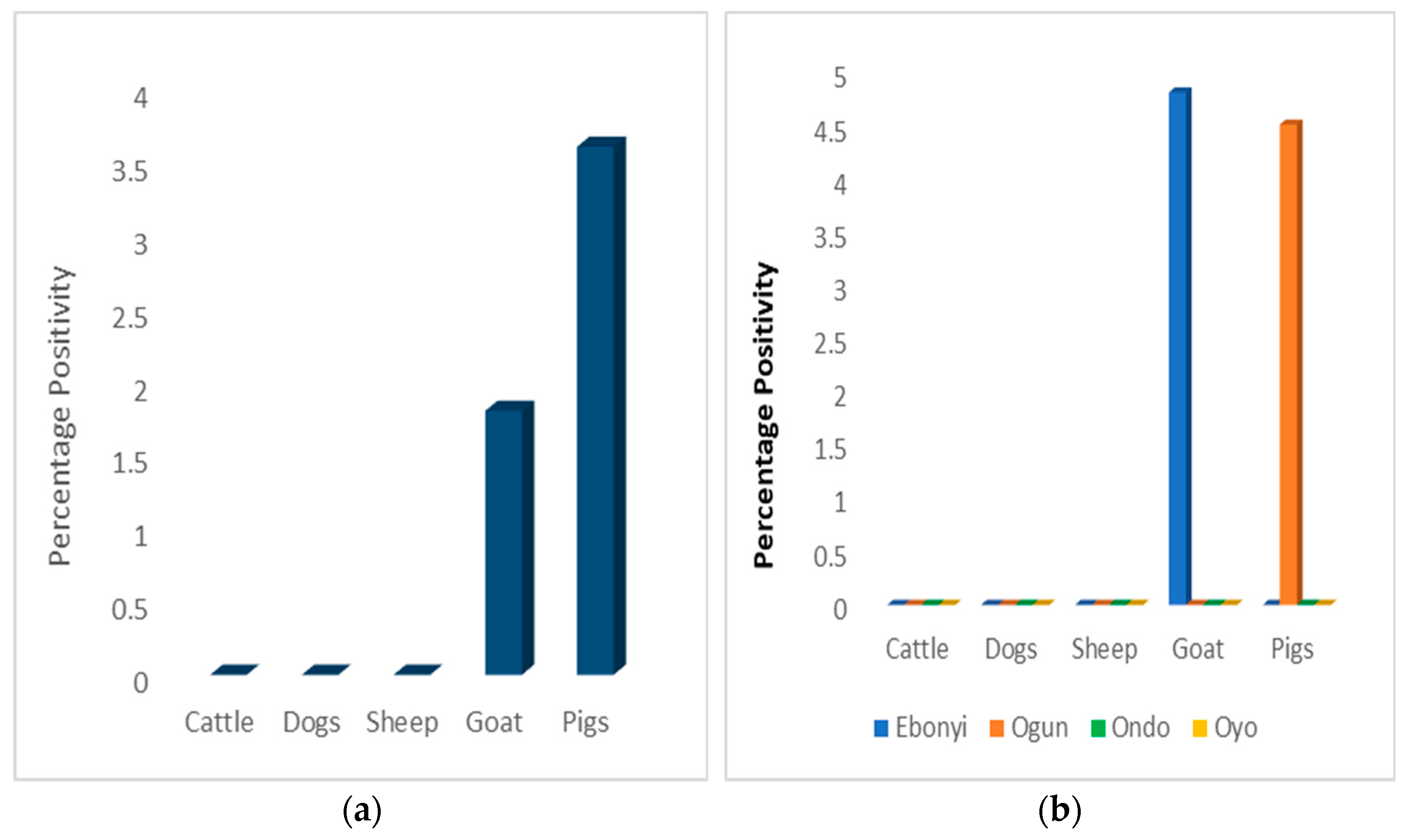
3.1.1. Comparison of SARS-CoV-2 RT-qPCR Positivity Rates by Location
3.1.2. Comparison of SARS-CoV-2 Positivity Rates by Animal Species per Location Using RT-qPCR
3.1.3. Comparison of SARS-CoV-2 RT-qPCR Positivity Rates by Health Status
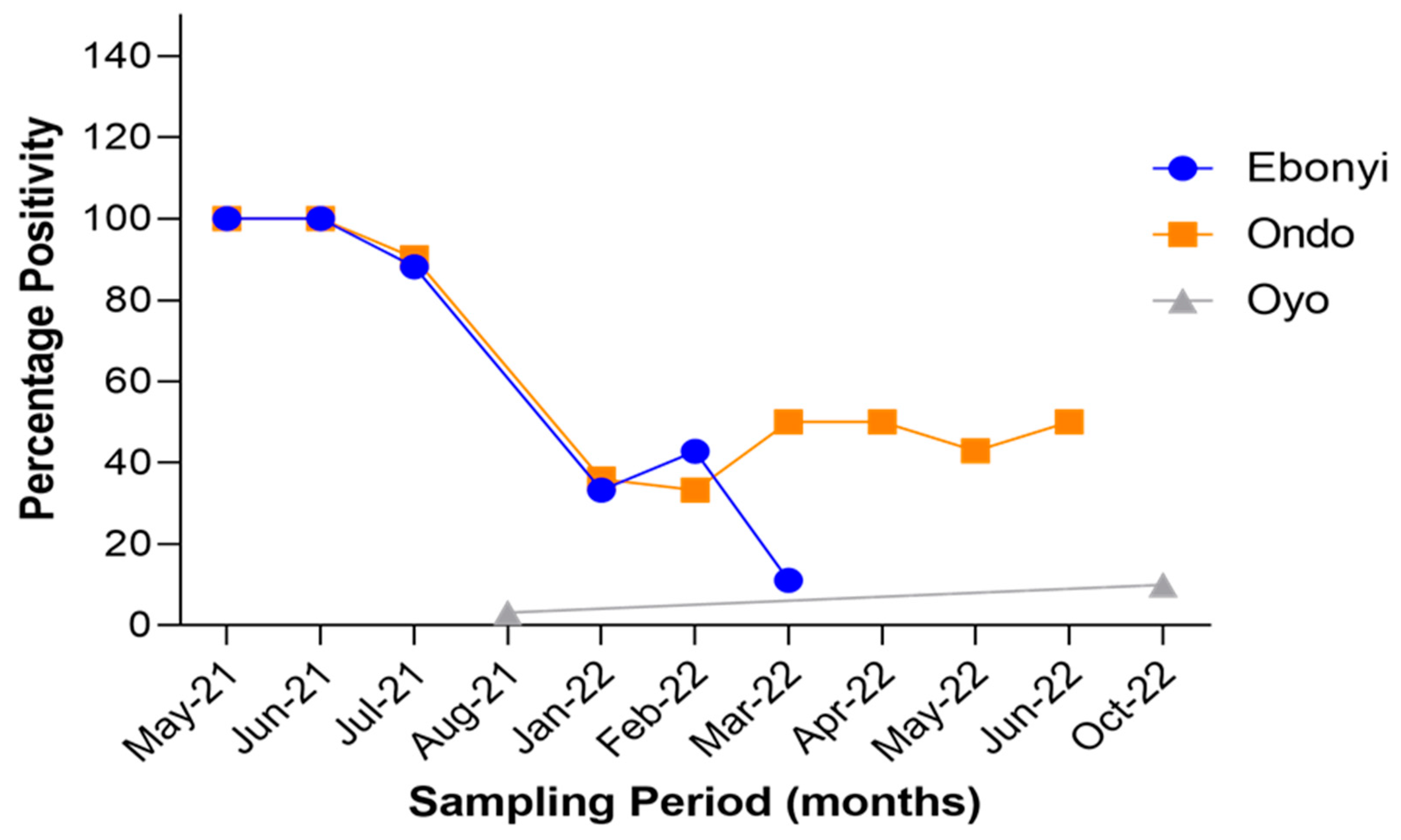
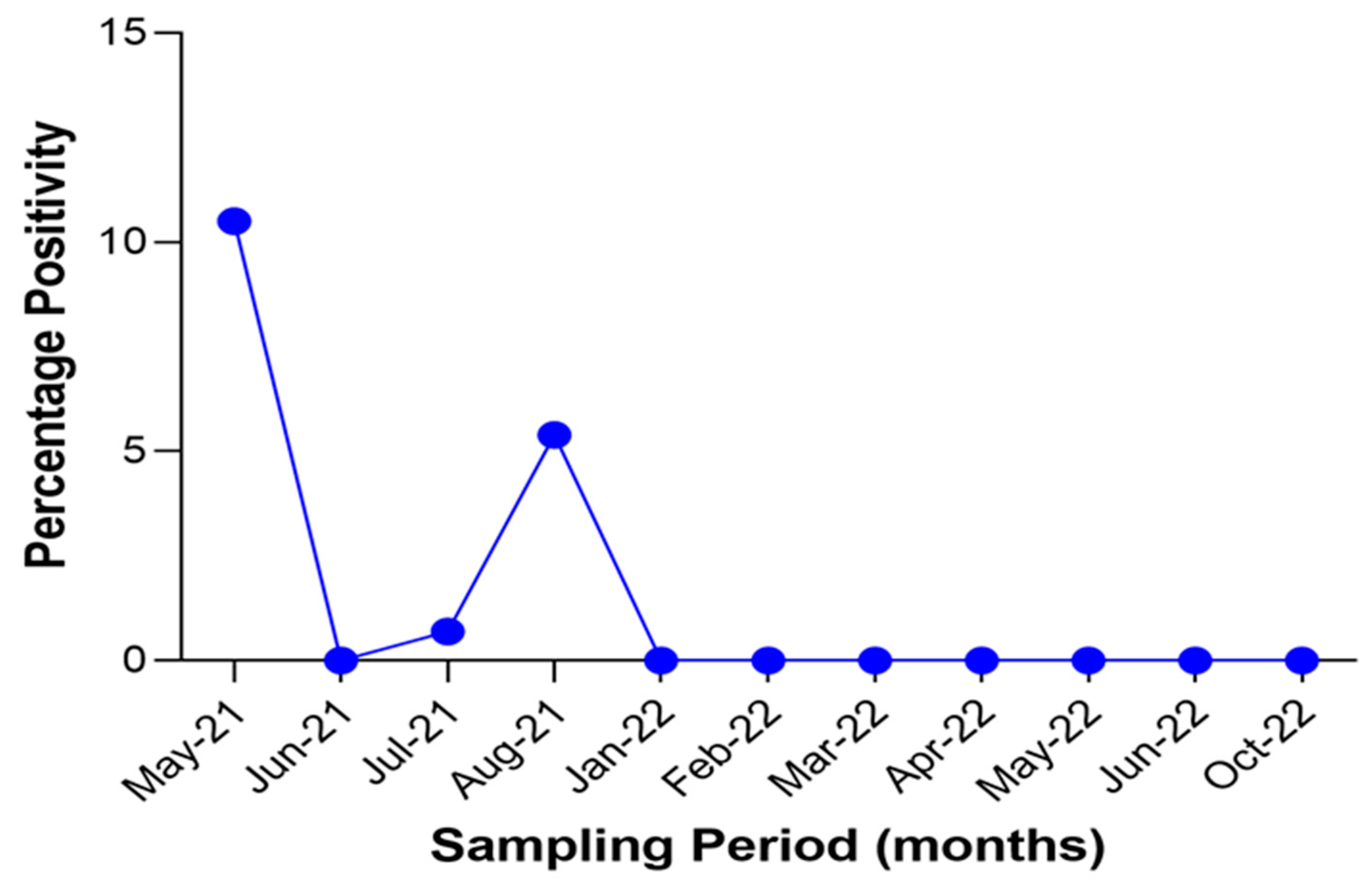
3.1.4. Comparison of SARS-CoV-2 RT-qPCR Positivity Rates by Sampling Period and Location
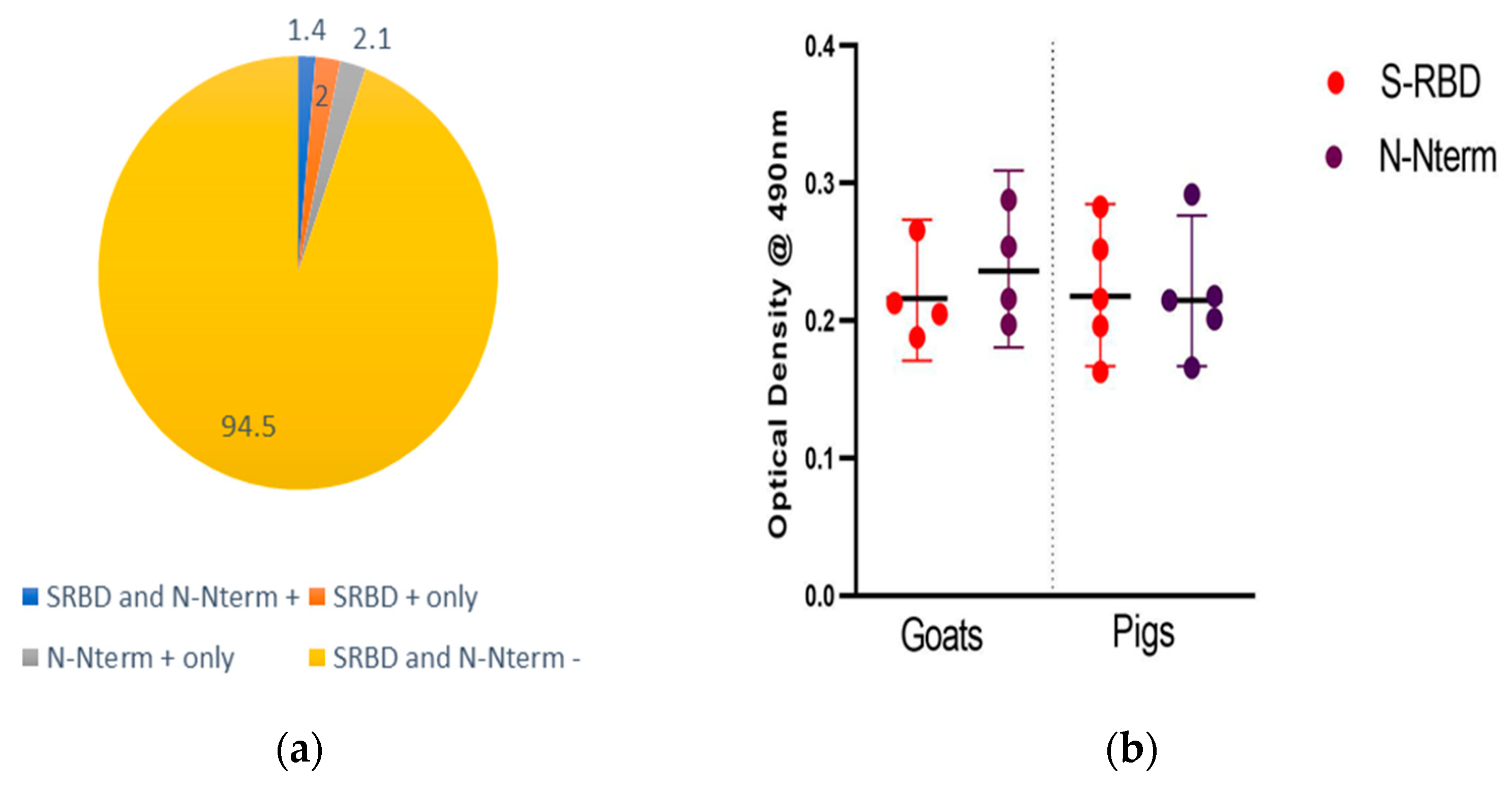
3.2. ELISA Results of Domestic Animals Naturally Infected with SARS-CoV-2
3.2.1. Comparison of SARS-CoV-2 ELISA Positivity Rates by Location
3.2.2. Comparison of SARS-CoV-2 ELISA Positivity Rates by Animal Species per State
3.2.3. Comparison of SARS-CoV-2 ELISA Positivity Rates by Health Status
3.2.4. Comparison of SARS-CoV-2 ELISA Positivity Rates by Sampling Period and Location
3.3. Juxtaposition of RT-qPCR and ELISA Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Han, Q.; Lin, Q.; Jin, S.; You, L. Coronavirus 2019-nCoV: A brief perspective from the front line. J. Infect. 2020, 80, 373. [Google Scholar] [CrossRef] [PubMed]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; di Napoli, R. Features, Evaluation, and Treatment of Coronavirus (COVID-19). StatPearls 2022. [Google Scholar]
- Gralinski, L.E.; Menachery, V.D. Return of the Coronavirus: 2019-nCoV. Viruses 2020, 12, 135. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. Brief Report: A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565. [Google Scholar] [CrossRef]
- Wang, M.; Yan, M.; Xu, H.; Liang, W.; Kan, B.; Zheng, B.; Chen, H.; Zheng, H.; Xu, Y.; Zhang, E.; et al. SARS-CoV Infection in a Restaurant from Palm Civet. Emerg. Infect. Dis. 2005, 11, 1860. [Google Scholar] [CrossRef] [PubMed]
- Letko, M.; Marzi, A.; Munster, V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat. Microbiol. 2020, 5, 562–569. [Google Scholar] [CrossRef]
- WHO Coronavirus (COVID-19) Dashboard with Vaccination Data n.d. Available online: https://covid19.who.int/ (accessed on 11 April 2023).
- NCDC Coronavirus COVID-19 Microsite n.d. Available online: https://covid19.ncdc.gov.ng/ (accessed on 11 April 2023).
- Elimian, K.O.; Ochu, C.L.; Ilori, E.; Oladejo, J.; Igumbor, E.; Steinhardt, L.; Wagai, J.; Arinze, C.; Ukponu, W.; Obiekea, C.; et al. Descriptive epidemiology of coronavirus disease 2019 in Nigeria, 27 February–6 June 2020. Epidemiol. Infect. 2020, 148, e208. [Google Scholar] [CrossRef]
- Zhao, S.; Musa, S.S.; Lin, Q.; Ran, J.; Yang, G.; Wang, W.; Lou, Y.; Yang, L.; Gao, D.; He, D.; et al. Estimating the Unreported Number of Novel Coronavirus (2019-nCoV) Cases in China in the First Half of January 2020: A Data-Driven Modelling Analysis of the Early Outbreak. J. Clin. Med. 2020, 9, 388. [Google Scholar] [CrossRef]
- Aronna, M.S.; Guglielmi, R.; Moschen, L.M. Estimate of the rate of unreported COVID-19 cases during the first outbreak in Rio de Janeiro. Infect. Dis. Model. 2022, 7, 317–332. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Itoh, N.; Yufika, A.; Winardi, W.; Keam, S.; Te, H.; Megawati, D.; Hayati, Z.; Wagner, A.L.; Mudatsir, M. Coronavirus disease 2019 (COVID-19): A literature review. J. Infect. Public Health 2020, 13, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Ameh Yaro, C.; Udama Eneche, P.S.; Abu Anyebe, D. Risk analysis and hot spots detection of SARS-CoV-2 in Nigeria using demographic and environmental variables: An early assessment of transmission dynamics. Int. J. Environ. Health Res. 2022, 32, 1111–1122. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.Q.; Peng, H.J. Characteristics of and Public Health Responses to the Coronavirus Disease 2019 Outbreak in China. J. Clin. Med. 2020, 9, 575. [Google Scholar] [CrossRef]
- Jiang, S.; Du, L.; Shi, Z. An emerging coronavirus causing pneumonia outbreak in Wuhan, China: Calling for developing therapeutic and prophylactic strategies. Emerg. Microbes Infect. 2020, 9, 275–277. [Google Scholar] [CrossRef]
- Murakami, S.; Kitamura, T.; Suzuki, J.; Sato, R.; Aoi, T.; Fujii, M.; Matsugo, H.; Kamiki, H.; Ishida, H.; Takenaka-Uema, A.; et al. Detection and Characterization of Bat Sarbecovirus Phylogenetically Related to SARS-CoV-2, Japan. Emerg. Infect. Dis. 2020, 26, 3025–3029. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, X.; Hu, T.; Li, J.; Song, H.; Liu, Y.; Wang, P.; Liu, D.; Yang, J.; Holmes, E.C.; et al. A Novel Bat Coronavirus Closely Related to SARS-CoV-2 Contains Natural Insertions at the S1/S2 Cleavage Site of the Spike Protein. Curr. Biol. 2020, 30, 2196–2203.e3. [Google Scholar] [CrossRef]
- Andersen, K.G.; Rambaut, A.; Lipkin, W.I.; Holmes, E.C.; Garry, R.F. The proximal origin of SARS-CoV-2. Nat. Med. 2020, 26, 450–452. [Google Scholar] [CrossRef]
- Goraichuk, I.V.; Arefiev, V.; Stegniy, B.T.; Gerilovych, A.P. Zoonotic and Reverse Zoonotic Transmissibility of SARS-CoV-2. Virus Res. 2021, 302, 198473. [Google Scholar] [CrossRef]
- Sit, T.H.C.; Brackman, C.J.; Ip, S.M.; Tam, K.W.S.; Law, P.Y.T.; To, E.M.W.; Yu, V.Y.T.; Sims, L.D.; Tsang, D.N.C.; Chu, D.K.W.; et al. Infection of dogs with SARS-CoV-2. Nature 2020, 586, 776–778. [Google Scholar] [CrossRef] [PubMed]
- Calvet, G.A.; Pereira, S.A.; Ogrzewalska, M.; Pauvolid-Corrêa, A.; Resende, P.C.; de Tassinari, W.S.; Costa, A.D.P.; Keidel, L.O.; da Rocha, A.S.B.; da Silva, M.F.B.; et al. Investigation of SARS-CoV-2 infection in dogs and cats of humans diagnosed with COVID-19 in Rio de Janeiro, Brazil. PLoS ONE 2021, 16, e0250853. [Google Scholar] [CrossRef] [PubMed]
- Fuentealba, N.A.; Moré, G.; Bravi, M.E.; Unzaga, J.M.; de Felice, L.; Salina, M.; Viegas, M.; Jodar, M.S.N.; Valinotto, L.E.; Rivero, F.D.; et al. First detection and molecular analysis of SARS-CoV-2 from a naturally infected cat from Argentina. Vet. Microbiol. 2021, 260, 109179. [Google Scholar] [CrossRef] [PubMed]
- Meisner, J.; Baszler, T.V.; Kuehl, K.E.; Ramirez, V.; Baines, A.; Frisbie, L.A.; Lofgren, E.T.; de Avila, D.M.; Wolking, R.M.; Bradway, D.S.; et al. Household Transmission of SARS-CoV-2 from Humans to Pets, Washington and Idaho, USA. Emerg. Infect. Dis. 2022, 28, 2425–2434. [Google Scholar] [CrossRef]
- Diezma-Díaz, C.; Álvarez-García, G.; Regidor-Cerrillo, J.; Miró, G.; Villanueva-Saz, S.; Dolores Pérez, M.; Lofgren, E.T.; de Avila, D.M.; Wolking, R.M.; Bradway, D.S.; et al. A comparative study of eight serological methods shows that spike protein-based ELISAs are the most accurate tests for serodiagnosing SARS-CoV-2 infections in cats and dogs. Front. Vet. Sci. 2023, 10, 1121935. [Google Scholar] [CrossRef] [PubMed]
- Larsen, H.D.; Fonager, J.; Lomholt, F.K.; Dalby, T.; Benedetti, G.; Kristensen, B.; Urth, T.R.; Rasmussen, M.; Lassaunière, R.; Rasmussen, T.B.; et al. Preliminary report of an outbreak of SARS-CoV-2 in mink and mink farmers associated with community spread, Denmark, June to November 2020. Eurosurveillance 2021, 26, 2100009. [Google Scholar] [CrossRef]
- Oude Munnink, B.B.; Sikkema, R.S.; Nieuwenhuijse, D.F.; Molenaar, R.J.; Munger, E.; Molenkamp, R.; van der Spek, A.; Tolsma, P.; Rietveld, A.; Brouwer, M.; et al. Transmission of SARS-CoV-2 on mink farms between humans and mink and back to humans. Science 2021, 371, 172–177. [Google Scholar] [CrossRef]
- Eckstrand, C.D.; Baldwin, T.J.; Rood, K.A.; Clayton, M.J.; Lott, J.K.; Wolking, R.M.; Bradway, D.S.; Baszler, T. An outbreak of SARS-CoV-2 with high mortality in mink (Neovison vison) on multiple Utah farms. PLoS Pathog. 2021, 17, e1009952. [Google Scholar] [CrossRef]
- Pomorska-Mól, M.; Włodarek, J.; Gogulski, M.; Rybska, M. Review: SARS-CoV-2 infection in farmed minks—An overview of current knowledge on occurrence, disease and epidemiology. Animal 2021, 15, 100272. [Google Scholar] [CrossRef]
- Chandler, J.C.; Bevins, S.N.; Ellis, J.W.; Linder, T.J.; Tell, R.M.; Jenkins-Moore, M.; Root, J.J.; Lenoch, J.B.; Robbe-Austerman, S.; DeLiberto, T.G.; et al. SARS-CoV-2 exposure in wild white-tailed deer (Odocoileus virginianus). Proc. Natl. Acad. Sci. USA 2021, 118, e2114828118. [Google Scholar] [CrossRef]
- Koeppel, K.N.; Mendes, A.; Strydom, A.; Rotherham, L.; Mulumba, M.; Venter, M. SARS-CoV-2 Reverse Zoonoses to Pumas and Lions, South Africa. Viruses 2022, 14, 120. [Google Scholar] [CrossRef] [PubMed]
- Phan, I.Q.; Subramanian, S.; Kim, D.; Murphy, M.; Pettie, D.; Carter, L.; Anishchenko, I.; Barrett, L.K.; Craig, J.; Tillery, L.; et al. In silico detection of SARS-CoV-2 specific B-cell epitopes and validation in ELISA for serological diagnosis of COVID-19. Sci. Rep. 2021, 11, 4290. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wu, Q.; Zhang, Z. Probable Pangolin Origin of SARS-CoV-2 Associated with the COVID-19 Outbreak. Curr. Biol. 2020, 30, 1346. [Google Scholar] [CrossRef] [PubMed]
- Dhama, K.; Patel, S.K.; Sharun, K.; Pathak, M.; Tiwari, R.; Yatoo, M.I.; Malik, Y.S.; Sah, R.; Rabaan, A.A.; Panwar, P.K.; et al. SARS-CoV-2 jumping the species barrier: Zoonotic lessons from SARS, MERS and recent advances to combat this pandemic virus. Travel Med. Infect. Dis. 2020, 37, 101830. [Google Scholar] [CrossRef]
- Ulrich, L.; Wernike, K.; Hoffmann, D.; Mettenleiter, T.C.; Beer, M. Experimental Infection of Cattle with SARS-CoV-2. Emerg. Infect. Dis. 2020, 26, 2979. [Google Scholar] [CrossRef]
- Bosco-Lauth, A.M.; Walker, A.; Guilbert, L.; Porter, S.; Hartwig, A.; McVicker, E.; Bielefeldt-Ohmann, H.; Bowen, R.A. Susceptibility of livestock to SARS-CoV-2 infection. Emerg. Microbes Infect. 2021, 10, 2199–2201. [Google Scholar] [CrossRef]
- Fernández-Bastit, L.; Roca, N.; Romero-Durana, M.; Rodon, J.; Cantero, G.; García, Ó.; López, C.; Pérez, M.; López, R.; Carrillo, J.; et al. Susceptibility of Domestic Goat (Capra aegagrus hircus) to Experimental Infection with Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) B.1.351/Beta Variant. Viruses 2022, 14, 2002. [Google Scholar] [CrossRef]
- Gaudreault, N.N.; Cool, K.; Trujillo, J.D.; Morozov, I.; Meekins, D.A.; McDowell, C.; Bold, D.; Carossino, M.; Balaraman, V.; Mitzel, D.; et al. Susceptibility of sheep to experimental co-infection with the ancestral lineage of SARS-CoV-2 and its alpha variant. Emerg. Microbes Infect. 2022, 11, 662. [Google Scholar] [CrossRef]
- Schlottau, K.; Rissmann, M.; Graaf, A.; Schön, J.; Sehl, J.; Wylezich, C.; Höper, D.; Mettenleiter, T.C.; Balkema-Buschmann, A.; Harder, T.; et al. SARS-CoV-2 in fruit bats, ferrets, pigs, and chickens: An experimental transmission study. Lancet Microbe 2020, 1, e218–e225. [Google Scholar] [CrossRef]
- Suarez, D.L.; Pantin-Jackwood, M.J.; Swayne, D.E.; Lee, S.A.; DeBlois, S.M.; Spackman, E. Lack of Susceptibility to SARS-CoV-2 and MERS-CoV in Poultry. Emerg. Infect. Dis. 2020, 26, 3074. [Google Scholar] [CrossRef]
- Buckley, A.; Falkenberg, S.; Martins, M.; Laverack, M.; Palmer, M.V.; Lager, K.; Diel, D.G. Intravenous, Intratracheal, and Intranasal Inoculation of Swine with SARS-CoV-2. Viruses 2021, 13, 1506. [Google Scholar] [CrossRef] [PubMed]
- Vergara-Alert, J.; Rodon, J.; Carrillo, J.; Te, N.; Izquierdo-Useros, N.; Rodríguez de la Concepción, M.L.; ÁvilaNieto, M.C.; Guallar, V.; Valencia, A.; Cantero, G.; et al. Pigs are not susceptible to SARS-CoV-2 infection but are a model for viral immunogenicity studies. Transbound. Emerg. Dis. 2021, 68, 1721. [Google Scholar] [CrossRef] [PubMed]
- Bamgboye, E.L.; Omiye, J.A.; Afolaranmi, O.J.; Davids, M.R.; Tannor, E.K.; Wadee, S.; Niang, A.; Were, A.; Naicker, S. COVID-19 Pandemic: Is Africa Different? J. Natl. Med. Assoc. 2021, 113, 324. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Liu, J.; Liu, Q.; Kang, L.; Liu, R.; Jig, W.; Wu, Y.; Liu, M. Global Percentage of Asymptomatic SARS-CoV-2 Infections Among the Tested Population and Individuals with Confirmed COVID-19 Diagnosis: A Systematic Review and Meta-analysis. JAMA Netw. Open 2021, 4, e2137257. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Wong, G.; Shi, W.; Liu, J.; Lai, A.C.K.; Zhou, J.; Liu, W.; Bi, Y.; Gao, G.F. Epidemiology, Genetic Recombination, and Pathogenesis of Coronaviruses. Trends Microbiol. 2016, 24, 490. [Google Scholar] [CrossRef]
- Vincent, A.L.; Anderson, T.K.; Lager, K.M. A Brief Introduction to Influenza A Virus in Swine. Methods Mol. Biol. 2020, 2123, 249–271. [Google Scholar] [CrossRef]
- Wang, Q.; Vlasova, A.N.; Kenney, S.P.; Saif, L.J. Emerging and re-emerging coronaviruses in pigs. Curr. Opin. Virol. 2019, 34, 39–49. [Google Scholar] [CrossRef]
- Kim, Y.I.; Kim, S.G.; Kim, S.M.; Kim, E.H.; Park, S.J.; Yu, K.M.; Chang, J.-H.; Kim, E.J.; Lee, S.; Casel, M.A.B.; et al. Infection and Rapid Transmission of SARS-CoV-2 in Ferrets. Cell Host. Microbe 2020, 27, 704. [Google Scholar] [CrossRef]
| State | Animal Type | Total Number Analyzed (%) | Number of RT-qPCR Analyzed Samples | Number of ELISA Analyzed Samples |
|---|---|---|---|---|
| Overall | Cat | 2 (0.3) | 2 | 0 |
| Cattle | 24 (3) | 24 | 3 | |
| Chicken | 38 (4.8) | 38 | 0 | |
| Dog | 172 (21.7) | 23 | 161 | |
| Duck | 3 (0.4) | 3 | 0 | |
| Goat | 234 (29.6) | 77 | 223 | |
| Lizard | 31 (3.9) | 31 | 0 | |
| Pig | 147 (18.6) | 92 | 137 | |
| Pigeon | 1 (0.1) | 1 | 0 | |
| Sheep | 132 (16.7) | 56 | 130 | |
| Turkey | 7 (0.9) | 7 | 0 | |
| Total | 791 (100) | 364 | 654 | |
| Ebonyi | Cat | 1 (0.4) | 1 | 0 |
| Cattle | 17 (7) | 17 | 0 | |
| Chicken | 9 (3.7) | 9 | 0 | |
| Dog | 56 (23) | 11 | 50 | |
| Goat | 94 (38.7) | 34 | 83 | |
| Lizard | 12 (4.9) | 12 | 0 | |
| Pig | 12 (4.9) | 3 | 10 | |
| Sheep | 42 (17.3) | 19 | 41 | |
| Total | 243 | 106 | 184 | |
| Ogun | Dog | 5 (3.5) | 5 | 5 |
| Goat | 17 (11.8) | 5 | 17 | |
| Pig | 116 (80.6) | 72 | 110 | |
| Sheep | 6 (4.2) | 0 | 6 | |
| Total | 144 | 82 | 138 | |
| Ondo | Cat | 1 (0.3) | 1 | 0 |
| Cattle | 4 (1) | 4 | 0 | |
| Chicken | 29 (7.6) | 29 | 0 | |
| Dog | 111 (29.1) | 17 | 106 | |
| Duck | 3 (0.8) | 3 | 0 | |
| Goat | 120 (31.5) | 35 | 120 | |
| Lizard | 19 (5) | 19 | 0 | |
| Pig | 4 (1) | 2 | 2 | |
| Pigeon | 1 (0.3) | 1 | 0 | |
| Sheep | 82 (21.5) | 35 | 81 | |
| Turkey | 7 (1.8) | 7 | 0 | |
| Total | 381 | 153 | 309 | |
| Oyo | Cattle | 3 (13) | 3 | 3 |
| Goat | 3 (13) | 3 | 3 | |
| Pig | 15 (65.2) | 15 | 15 | |
| Sheep | 2 (8.7) | 2 | 2 | |
| Total | 23 | 23 | 23 |
| State | Animal Species | No. Analyzed | Positive (%) | p Value |
|---|---|---|---|---|
| Overall | Cat | 2 | 1 (50) | 1.0 |
| Cattle | 24 | 17 (70.8) | 0.01 | |
| Chicken | 38 | 10 (26.3) | 0.01 | |
| Dog | 33 | 17 (51.5) | 0.496 | |
| Duck | 3 | 2 (66.7) | 0.596 | |
| Goat | 77 | 46 (59.7) | 0.01 | |
| Lizard | 31 | 19 (61.3) | 0.07 | |
| Pig | 92 | 4 (4.3) | <0.0001 | |
| Pigeon | 1 | 0 (0) | - | |
| Sheep | 56 | 50 (89.3) | <0.0001 | |
| Turkey | 7 | 1 (14.3) | 0.13 | |
| Total | 364 | 167 (45.9) | ||
| Ebonyi | Cat | 1 | 1 (100) | - |
| Cattle | 17 | 15 (88.2) | 0.02 | |
| Chicken | 9 | 4 (44.4) | 0.29 | |
| Dog | 11 | 5 (45.5) | 0.33 | |
| Goat | 34 | 20 (58.8) | 0.62 | |
| Lizard | 12 | 2 (16.7) | 0.001 | |
| Sheep | 19 | 18 (94.7) | 0.001 | |
| Pig | 3 | 1 (33.3) | 0.56 | |
| Ogun | Dog | 5 | 1 (20) | 0.23 |
| Goat | 5 | 1 (20) | 0.23 | |
| Pig | 72 | 2 (2.8) | 0.07 | |
| Ondo | Cat | 1 | 0 (0) | - |
| Cattle | 4 | 2 (50) | 0.62 | |
| Chicken | 29 | 6 (20.7) | <0.0001 | |
| Dog | 17 | 11 (64.7) | 0.91 | |
| Duck | 3 | 2 (66.7) | 1.0 | |
| Goat | 35 | 25 (71.4) | 0.26 | |
| Lizard | 19 | 17 (89.5) | 0.01 | |
| Pig | 2 | 1 (50) | 1.0 | |
| Pigeon | 1 | 0 (0) | - | |
| Sheep | 35 | 32 (91.4) | <0.0001 | |
| Turkey | 7 | 1 (14.3) | 0.01 | |
| Oyo | Cattle | 3 | 0 (0) | - |
| Goat | 3 | 0 (0) | - | |
| Pig | 15 | 0 (0) | - | |
| Sheep | 2 | 0 (0) | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Happi, A.N.; Ayinla, A.O.; Ogunsanya, O.A.; Sijuwola, A.E.; Saibu, F.M.; Akano, K.; George, U.E.; Sopeju, A.E.; Rabinowitz, P.M.; Ojo, K.K.; et al. Detection of SARS-CoV-2 in Terrestrial Animals in Southern Nigeria: Potential Cases of Reverse Zoonosis. Viruses 2023, 15, 1187. https://doi.org/10.3390/v15051187
Happi AN, Ayinla AO, Ogunsanya OA, Sijuwola AE, Saibu FM, Akano K, George UE, Sopeju AE, Rabinowitz PM, Ojo KK, et al. Detection of SARS-CoV-2 in Terrestrial Animals in Southern Nigeria: Potential Cases of Reverse Zoonosis. Viruses. 2023; 15(5):1187. https://doi.org/10.3390/v15051187
Chicago/Turabian StyleHappi, Anise N., Akeemat O. Ayinla, Olusola A. Ogunsanya, Ayotunde E. Sijuwola, Femi M. Saibu, Kazeem Akano, Uwem E. George, Adebayo E. Sopeju, Peter M. Rabinowitz, Kayode K. Ojo, and et al. 2023. "Detection of SARS-CoV-2 in Terrestrial Animals in Southern Nigeria: Potential Cases of Reverse Zoonosis" Viruses 15, no. 5: 1187. https://doi.org/10.3390/v15051187
APA StyleHappi, A. N., Ayinla, A. O., Ogunsanya, O. A., Sijuwola, A. E., Saibu, F. M., Akano, K., George, U. E., Sopeju, A. E., Rabinowitz, P. M., Ojo, K. K., Barrett, L. K., Van Voorhis, W. C., & Happi, C. T. (2023). Detection of SARS-CoV-2 in Terrestrial Animals in Southern Nigeria: Potential Cases of Reverse Zoonosis. Viruses, 15(5), 1187. https://doi.org/10.3390/v15051187






