Efficient Purification of Cowpea Chlorotic Mottle Virus by a Novel Peptide Aptamer
Abstract
1. Introduction
2. Materials and Methods
2.1. Biochemicals and Other Reagents
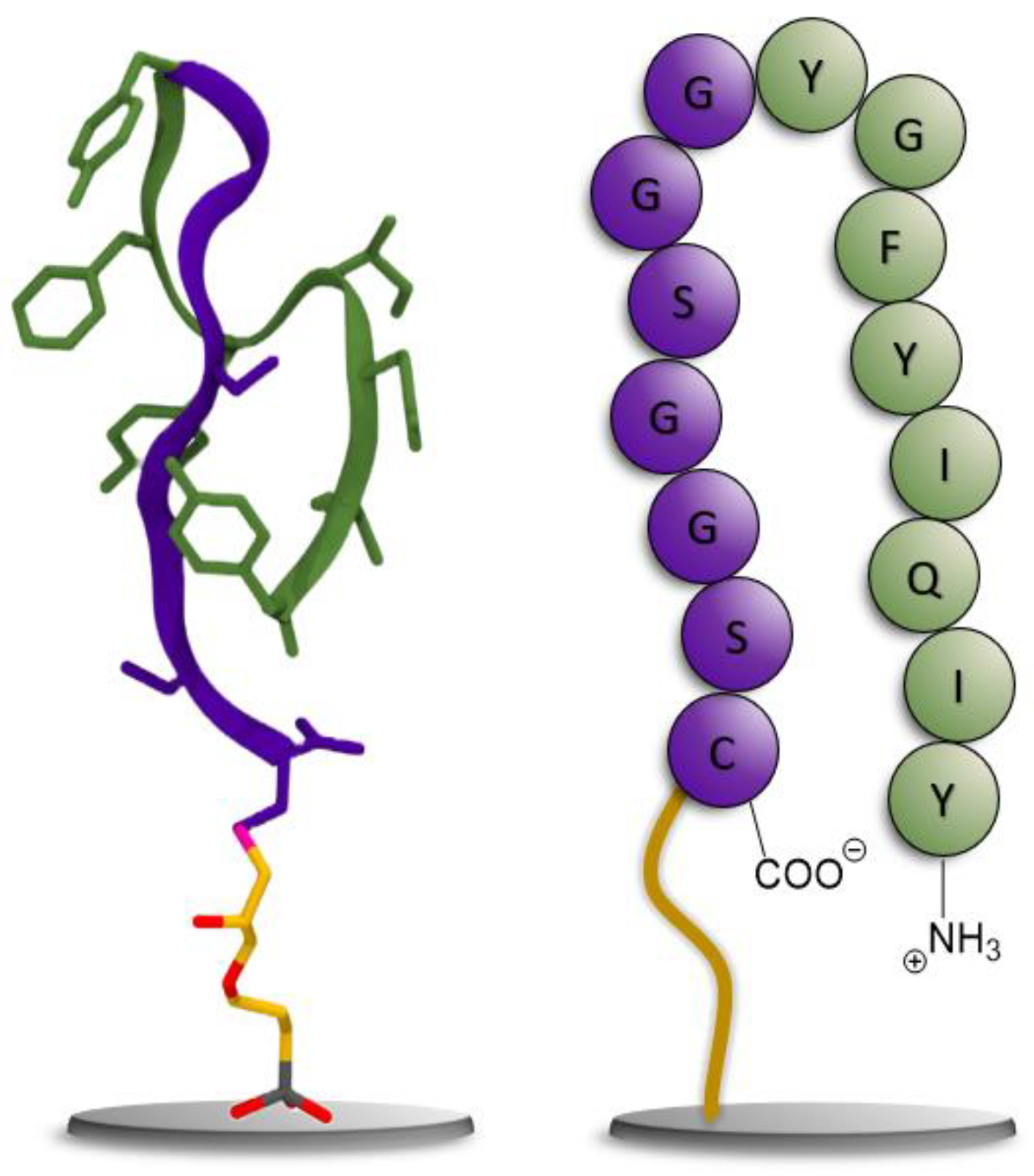
2.2. Generation of a Peptide Binder for CCMV
2.2.1. Experimental Peptide Binder Generation
2.2.2. Molecular Dynamics Simulations of the Peptide with GROMACS
2.2.3. Markov Modeling to Obtain the Dominant Equilibrium Peptide Structure in Solution
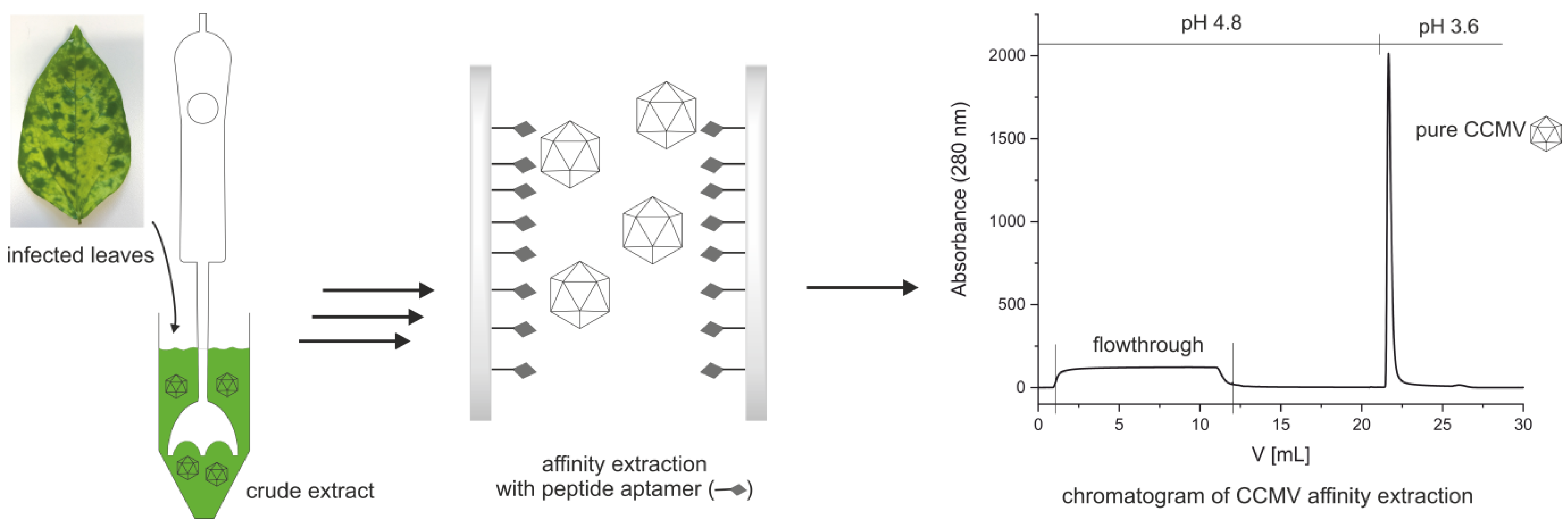
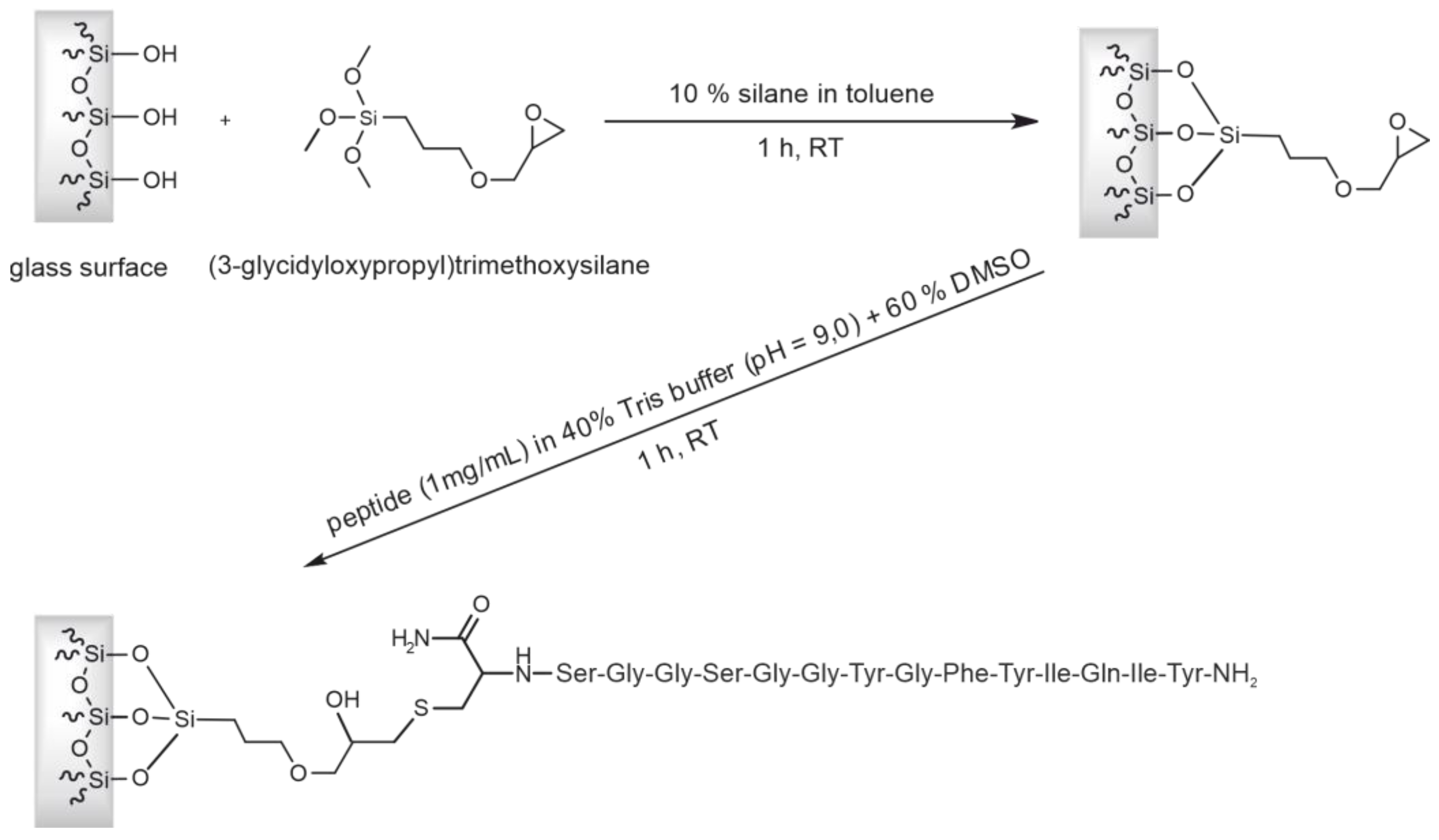
2.3. Preparation of the Affinity Column with CCMV-Binding Peptide
2.4. Optimized Protocol for the Purification of CCMV
2.5. Characterization of Purification Steps with Silver-Stained SDS-PAGE
2.6. Characterization of Purification Steps with Size Exclusion Chromatography (SEC)
2.7. Characterization of Purification Steps with MALDI-TOF MS
2.8. Purity Determination by Reversed-Phase HPLC
2.9. Determination of Integrity and Monodispersity
2.10. Quantification of CCMV by ELISA
3. Results
3.1. Generation of a Peptide Aptamer for CCMV
3.2. Preparation of an Affinity Column with the CCMV-Binding Peptide Aptamer
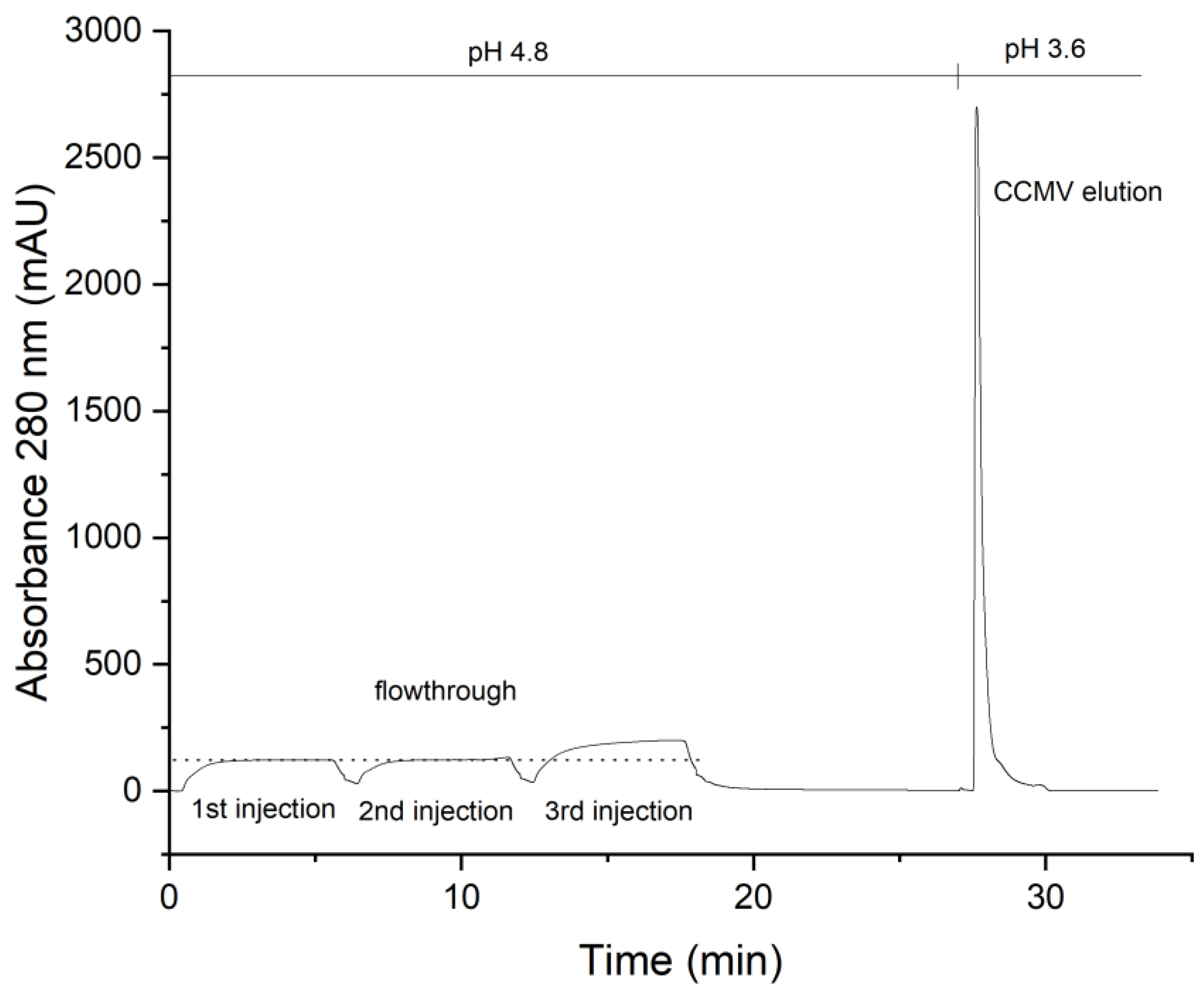
3.3. Optimized Protocol for the Purification of CCMV
3.4. Characterization of Purification Steps with Silver-Stained SDS-PAGE
3.5. Characterization of Purification Steps with Size Exclusion Chromatography
3.6. Characterization of Purification Steps with MALDI-TOF MS
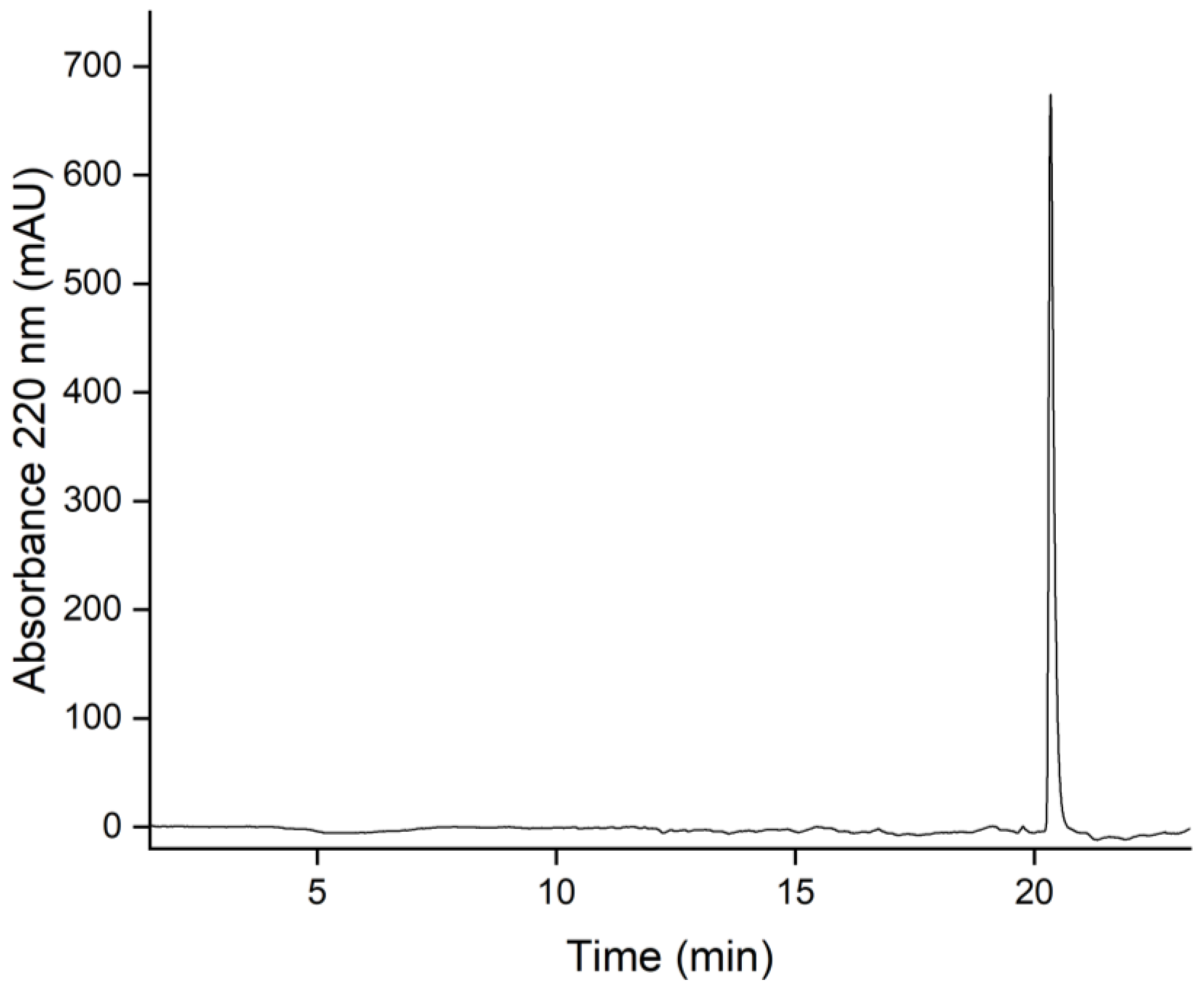
3.7. Purity Determination by Reversed-Phase Liquid Chromatography (HPLC)
3.8. Determination of Particle Integrity and Size Distribution
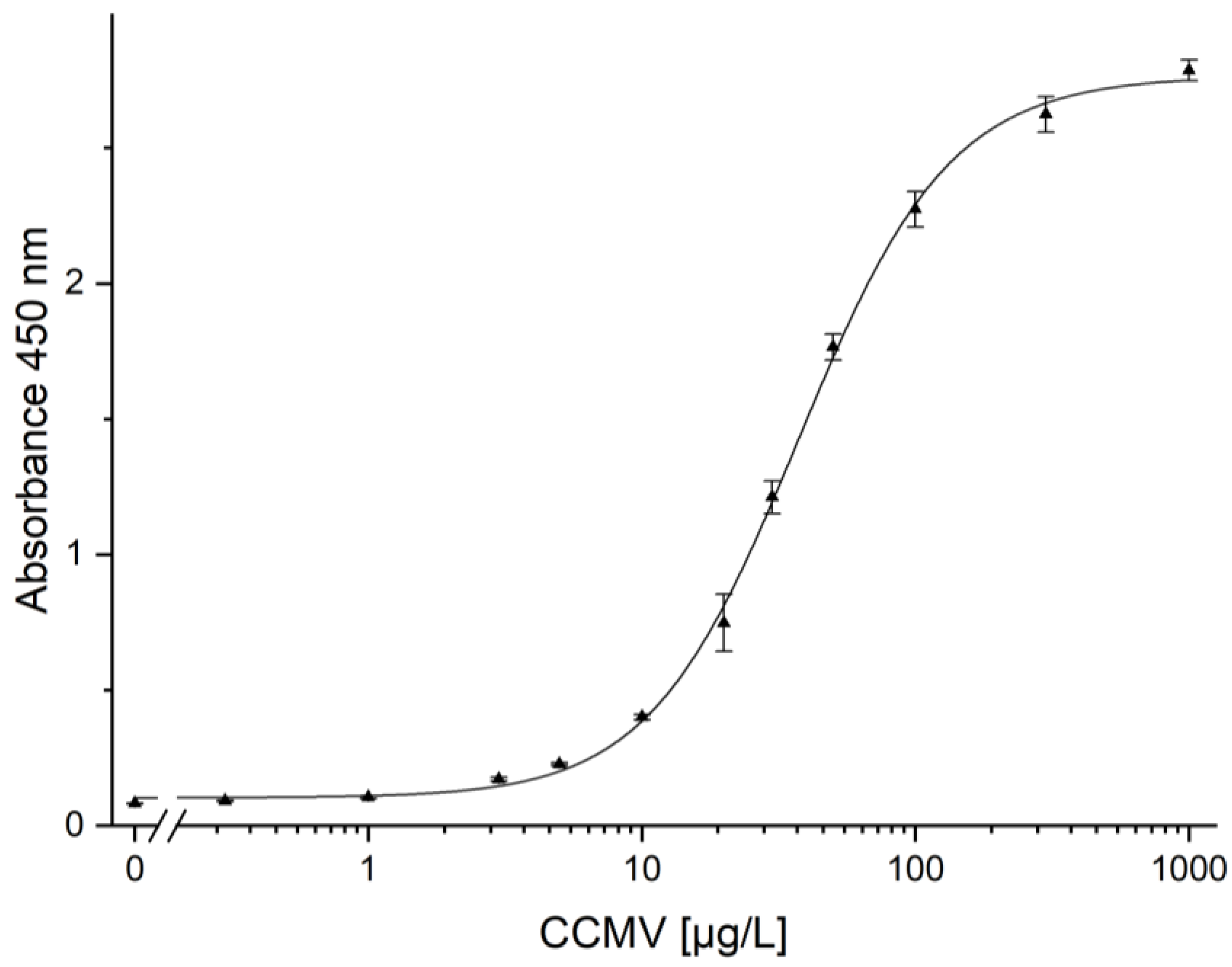
3.9. Quantification of CCMV by ELISA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hespenheide, B.M.; Jacobs, D.J.; Thorpe, M.F. Structural rigidity in the capsid assembly of cowpea chlorotic mottle virus. J. Phys.-Condens. Matter 2004, 16, S5055–S5064. [Google Scholar] [CrossRef]
- Kaiser, C.R.; Flenniken, M.L.; Gillitzer, E.; Harmsen, A.L.; Harmsen, A.G.; Jutila, M.A.; Douglas, T.; Young, M.J. Biodistribution studies of protein cage nanoparticles demonstrate broad tissue distribution and rapid clearance in vivo. Int. J. Nanomed. 2007, 2, 715–733. [Google Scholar]
- Azizgolshani, O.; Garmann, R.F.; Cadena-Nava, R.; Knobler, C.M.; Gelbart, W.M. Reconstituted plant viral capsids can release genes to mammalian cells. Virology 2013, 441, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Sanchez, L.; Cadena-Nava, R.D.; Palomares, L.A.; Ruiz-Garcia, J.; Koay, M.S.; Cornelissen, J.J.; Vazquez-Duhalt, R. Chemotherapy pro-drug activation by biocatalytic virus-like nanoparticles containing cytochrome P450. Enzym. Microb. Technol. 2014, 60, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Pretto, C.; Tang, M.; Chen, M.; Xu, H.P.; Subrizi, A.; Urtti, A.; van Hest, J.C.M. Cowpea chlorotic mottle virus-like particles as potential platform for antisense oligonucleotide delivery in posterior segment ocular diseases. Macromol. Biosci. 2021, 21, 2100095. [Google Scholar] [CrossRef]
- Villagrana-Escareño, M.V.; Reynaga-Hernández, E.; Galicia-Cruz, O.G.; Durán-Meza, A.L.; la Cruz-González, D.; Hernández-Carballo, C.Y.; Ruíz-García, J. VLPs derived from the CCMV plant virus can directly transfect and deliver heterologous genes for translation into mammalian cells. BioMed Res. Int. 2019, 2019, 4630891. [Google Scholar] [CrossRef]
- Timmermans, S.; Mesman, R.; Blezer, K.J.R.; van Niftrik, L.; van Hest, J.C.M. Cargo-loading of hybrid cowpea chlorotic mottle virus capsids via a co-expression approach. Virology 2022, 577, 99–104. [Google Scholar] [CrossRef]
- Tresset, G.; Chen, J.Z.; Chevreuil, M.; Nhiri, N.; Jacquet, E.; Lansac, Y. Two-dimensional phase transition of viral capsid gives insights into subunit interactions. Phys. Rev. Appl. 2017, 7, 014005. [Google Scholar] [CrossRef]
- Chen, J.Z.; Lansac, Y.; Tresset, G. Interactions between the molecular components of the cowpea chlorotic mottle virus investigated by molecular dynamics simulations. J. Phys. Chem. B 2018, 122, 9490–9498. [Google Scholar] [CrossRef]
- Lu, X.Y.; Thompson, J.R.; Perry, K.L. Encapsidation of DNA, a protein and a fluorophore into virus-like particles by the capsid protein of cucumber mosaic virus. J. Gen. Virol. 2012, 93, 1120–1126. [Google Scholar] [CrossRef]
- Gillitzer, E.; Willits, D.; Young, M.; Douglas, T. Chemical modification of a viral cage for multivalent presentation. Chem. Commun. 2002, 20, 2390–2391. [Google Scholar] [CrossRef]
- Hassani-Mehraban, A.; Creutzburg, S.; van Heereveld, L.; Kormelink, R. Feasibility of Cowpea chlorotic mottle virus-like particles as scaffold for epitope presentations. BMC Biotechnol. 2015, 15, 80. [Google Scholar] [CrossRef] [PubMed]
- Barwal, I.; Kumar, R.; Kateriya, S.; Dinda, A.K.; Yadav, S.C. Targeted delivery system for cancer cells consist of multiple ligands conjugated genetically modified CCMV capsid on doxorubicin GNPs complex. Sci. Rep. 2016, 6, 37096. [Google Scholar] [CrossRef] [PubMed]
- Vervoort, D.F.; Heiringhoff, R.; Timmermans, S.B.; Van Stevendaal, M.H.; Van Hest, J.C. Dual site-selective presentation of functional handles on protein-engineered cowpea chlorotic mottle virus-like particles. Bioconjugate Chem. 2021, 32, 958–963. [Google Scholar] [CrossRef]
- Hommersom, C.A.; Matt, B.; van der Ham, A.; Cornelissen, J.J.; Katsonis, N. Versatile post-functionalization of the external shell of cowpea chlorotic mottle virus by using click chemistry. Org. Biomol. Chem. 2014, 12, 4065–4069. [Google Scholar] [CrossRef]
- Pomwised, R.; Intamaso, U.; Teintze, M.; Young, M.; Pincus, S.H. Coupling Peptide Antigens to Virus-Like Particles or to Protein Carriers Influences the Th1/Th2 Polarity of the Resulting Immune Response. Vaccines 2016, 4, 15. [Google Scholar] [CrossRef]
- Loeb, T.; Zinder, N.D. A bacteriophage containing RNA. Proc. Natl. Acad. Sci. USA 1961, 47, 282–289. [Google Scholar] [CrossRef]
- Eiben, S.; Koch, C.; Altintoprak, K.; Southan, A.; Tovar, G.; Laschat, S.; Weiss, I.M.; Wege, C. Plant virus-based materials for biomedical applications: Trends and prospects. Adv. Drug Deliv. Rev. 2019, 145, 96–118. [Google Scholar] [CrossRef]
- Smith, T.J.; Chase, E.; Schmidt, T.; Perry, K.L. The structure of cucumber mosaic virus and comparison to cowpea chlorotic mottle virus. J. Virol. 2000, 74, 7578–7586. [Google Scholar] [CrossRef] [PubMed]
- Tama, F.; Brooks, C.L., 3rd. The mechanism and pathway of pH induced swelling in cowpea chlorotic mottle virus. J. Mol. Biol. 2002, 318, 733–747. [Google Scholar] [CrossRef]
- Speir, J.A.; Munshi, S.; Wang, G.; Baker, T.S.; Johnson, J.E. Structures of the native and swollen forms of cowpea chlorotic mottle virus determined by X-ray crystallography and cryo-electron microscopy. Structure 1995, 3, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Sehnal, D.; Bittrich, S.; Deshpande, M.; Svobodova, R.; Berka, K.; Bazgier, V.; Velankar, S.; Burley, S.K.; Koca, J.; Rose, A.S. Mol* Viewer: Modern web app for 3D visualization and analysis of large biomolecular structures. Nucleic Acids Res. 2021, 49, W431–W437. [Google Scholar] [CrossRef] [PubMed]
- Bancroft, J.B.; Hills, G.J.; Markham, R. A study of the self-assembly process in a small spherical virus. Formation of organized structures from protein subunits in vitro. Virology 1967, 31, 354–379. [Google Scholar] [CrossRef] [PubMed]
- Lam, P.; Steinmetz, N.F. Delivery of siRNA therapeutics using cowpea chlorotic mottle virus-like particles. Biomater. Sci. 2019, 7, 3138–3142. [Google Scholar] [CrossRef]
- Hu, Y.F.; Zandi, R.; Anavitarte, A.; Knobler, C.M.; Gelbart, W.M. Packaging of a polymer by a viral capsid: The interplay between polymer length and capsid size. Biophys. J. 2008, 94, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Douglas, T.; Young, M. Host-guest encapsulation of materials by assembled virus protein cages. Nature 1998, 393, 152–155. [Google Scholar] [CrossRef]
- Aniagyei, S.E.; Kennedy, C.J.; Stein, B.; Willits, D.A.; Douglas, T.; Young, M.J.; De, M.; Rotello, V.M.; Srisathiyanarayanan, D.; Kao, C.C.; et al. Synergistic effects of mutations and nanoparticle templating in the self-assembly of cowpea chlorotic mottle virus capsids. Nano Lett. 2009, 9, 393–398. [Google Scholar] [CrossRef]
- Rurup, W.F.; Verbij, F.; Koay, M.S.; Blum, C.; Subramaniam, V.; Cornelissen, J.J. Predicting the loading of virus-like particles with fluorescent proteins. Biomacromolecules 2014, 15, 558–563. [Google Scholar] [CrossRef]
- Shukla, S.; Wang, C.; Beiss, V.; Cai, H.; Washington, T., 2nd; Murray, A.A.; Gong, X.; Zhao, Z.; Masarapu, H.; Zlotnick, A.; et al. The unique potency of Cowpea mosaic virus (CPMV) in situ cancer vaccine. Biomater. Sci. 2020, 8, 5489–5503. [Google Scholar] [CrossRef]
- Almendarez-Rodriguez, C.; Solis-Andrade, K.I.; Govea-Alonso, D.O.; Comas-Garcia, M.; Rosales-Mendoza, S. Production and characterization of chimeric SARS-CoV-2 antigens based on the capsid protein of cowpea chlorotic mottle virus. Int. J. Biol. Macromol. 2022, 213, 1007–1017. [Google Scholar] [CrossRef]
- Speir, J.A.; Bothner, B.; Qu, C.X.; Willits, D.A.; Young, M.J.; Johnson, J.E. Enhanced local symmetry interactions globally stabilize a mutant virus capsid that maintains infectivity and capsid dynamics. J. Virol. 2006, 80, 3582–3591. [Google Scholar] [CrossRef]
- Vajda, B.P. Concentration and purification of viruses and bacteriophages with polyethylene glycol. Folia Microbiol. 1978, 23, 88–96. [Google Scholar] [CrossRef]
- Bhat, A.I.; Rao, G.P. Characterization of Plant Viruses; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Ali, A.; Roossinck, M.J. Rapid and efficient purification of Cowpea chlorotic mottle virus by sucrose cushion ultracentrifugation. J. Virol. Methods 2007, 141, 84–86. [Google Scholar] [CrossRef]
- Michel, J.P.; Gingery, M.; Lavelle, L. Efficient purification of bromoviruses by ultrafiltration. J. Virol. Methods 2004, 122, 195–198. [Google Scholar] [CrossRef]
- Berkeley Office of Environment, Health & Safety. Ultracentrifuges: Hazards and Precautions. Available online: https://ehs.berkeley.edu/publications/ultracentrifuges-hazards-and-precautions (accessed on 24 January 2023).
- Zhao, X.; Fox, J.M.; Olson, N.H.; Baker, T.S.; Young, M.J. In vitro assembly of cowpea chlorotic mottle virus from coat protein expressed in Escherichia coli and in vitro-transcribed viral cDNA. Virology 1995, 207, 486–494. [Google Scholar] [CrossRef]
- Cantin, G.T.; Resnick, S.; Jin, H.F.; O’Hanlon, R.; Espinosa, O.; Stevens, A.; Payne, J.; Glenn, N.R.; Rasochova, L.; Allen, J.R. Comparison of methods for chemical conjugation of an influenza peptide to wild-type and cysteine-mutant virus-like particles expressed in pseudomonas fluorescens. Int. J. Pept. Res. Ther. 2011, 17, 217–224. [Google Scholar] [CrossRef]
- Wehbe, M.; Labib, M.; Muharemagic, D.; Zamay, A.S.; Berezovski, M.V. Switchable aptamers for biosensing and bioseparation of viruses (SwAps-V). Biosens. Bioelectron. 2015, 67, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Yang, J.; Ye, W.; Wang, Y.; Ye, C.; Weng, D.; Gao, H.; Zhang, F.; Xu, Z.; Lei, Y. Isolation of endogenously assembled RNA-protein complexes using affinity purification based on streptavidin aptamer S1. Int. J. Mol. Sci. 2015, 16, 22456–22472. [Google Scholar] [CrossRef] [PubMed]
- Bunka, D.H.; Stockley, P.G. Aptamers come of age—At last. Nat. Rev. Microbiol. 2006, 4, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Wilke, M.; Roder, B.; Paul, M.; Weller, M.G. Sintered glass monoliths as supports for affinity columns. Separations 2021, 8, 56. [Google Scholar] [CrossRef]
- Schwaar, T.; Lettow, M.; Remmler, D.; Borner, H.G.; Weller, M.G. Efficient screening of combinatorial peptide libraries by spatially ordered beads immobilized on conventional glass slides. High Throughput 2019, 8, 11. [Google Scholar] [CrossRef]
- Furka, A. Forty years of combinatorial technology. Drug Discov. Today 2022, 27, 103308. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, J.O.; Brangsch, J.; Kader, A.; Saatz, J.; Mangarova, D.B.; Zacharias, M.; Kempf, W.E.; Schwaar, T.; Ponader, M.; Adams, L.C.; et al. ADAMTS4-specific MR probe to assess aortic aneurysms in vivo using synthetic peptide libraries. Nat. Commun. 2022, 13, 2867. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 2015, 1, 19–25. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Zidek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Hornak, V.; Abel, R.; Okur, A.; Strockbine, B.; Roitberg, A.; Simmerling, C. Comparison of multiple Amber force fields and development of improved protein backbone parameters. Proteins 2006, 65, 712–725. [Google Scholar] [CrossRef]
- Hockney, R.W.; Goel, S.P.; Eastwood, J.W. Quiet high-resolution computer models of a plasma. J. Comput. Phys. 1974, 14, 148–158. [Google Scholar] [CrossRef]
- Bussi, G.; Donadio, D.; Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 2007, 126, 014101. [Google Scholar] [CrossRef]
- Parrinello, M.; Rahman, A. Polymorphic transitions in single-crystals—A new molecular-dynamics method. J. Appl. Phys. 1981, 52, 7182–7190. [Google Scholar] [CrossRef]
- Essmann, U.; Perera, L.; Berkowitz, M.L.; Darden, T.; Lee, H.; Pedersen, L.G. A smooth particle mesh Ewald method. J. Chem. Phys. 1995, 103, 8577–8593. [Google Scholar] [CrossRef]
- Scherer, M.K.; Trendelkamp-Schroer, B.; Paul, F.; Perez-Hernandez, G.; Hoffmann, M.; Plattner, N.; Wehmeyer, C.; Prinz, J.H.; Noe, F. PyEMMA 2: A software package for estimation, validation, and analysis of Markov models. J. Chem. Theory Comput. 2015, 11, 5525–5542. [Google Scholar] [CrossRef]
- Wehmeyer, C.; Scherer, M.K.; Hempel, T.; Husic, B.E.; Olsson, S.; Noé, F. Introduction to Markov state modeling with the PyEMMA software—v1. 0. Living J. Comput. Mol. Sci. 2018, 1, 10.33011. [Google Scholar] [CrossRef]
- Vitalini, F.; Mey, A.S.; Noe, F.; Keller, B.G. Dynamic properties of force fields. J. Chem. Phys. 2015, 142, 084101. [Google Scholar] [CrossRef]
- Schor, M.; Mey, A.; MacPhee, C.E. Analytical methods for structural ensembles and dynamics of intrinsically disordered proteins. Biophys. Rev. 2016, 8, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Perez-Hernandez, G.; Paul, F.; Giorgino, T.; De Fabritiis, G.; Noe, F. Identification of slow molecular order parameters for Markov model construction. J. Chem. Phys. 2013, 139, 015102. [Google Scholar] [CrossRef]
- Röblitz, S.; Weber, M. Fuzzy spectral clustering by PCCA+: Application to Markov state models and data classification. Adv. Data Anal. Classif. 2013, 7, 147–179. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Bancroft, J.B.; Hiebert, E.; Rees, M.W.; Markham, R. Properties of cowpea chlorotic mottle virus, its protein and nucleic acid. Virology 1968, 34, 224–239. [Google Scholar] [CrossRef] [PubMed]
- De, S.; Khan, A. Efficient synthesis of multifunctional polymers via thiol-epoxy “click” chemistry. Chem. Commun. 2012, 48, 3130–3132. [Google Scholar] [CrossRef] [PubMed]
- Reinmuth-Selzle, K.; Tchipilov, T.; Backes, A.T.; Tscheuschner, G.; Tang, K.; Ziegler, K.; Lucas, K.; Pöschl, U.; Fröhlich-Nowoisky, J.; Weller, M.G. Determination of the protein content of complex samples by aromatic amino acid analysis, liquid chromatography-UV absorbance, and colorimetry. Anal. Bioanal. Chem. 2022, 414, 4457–4470. [Google Scholar] [CrossRef]
- Tscheuschner, G.; Schwaar, T.; Weller, M.G. Fast confirmation of antibody identity by MALDI-TOF MS fingerprints. Antibodies 2020, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Tscheuschner, G.; Kaiser, M.N.; Lisec, J.; Beslic, D.; Muth, T.; Kruger, M.; Mages, H.W.; Dorner, B.G.; Knospe, J.; Schenk, J.A.; et al. MALDI-TOF-MS-based identification of monoclonal murine anti-SARS-CoV-2 antibodies within one hour. Antibodies 2022, 11, 27. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.K.; Steinmetz, N.F. Isolation of cowpea mosaic virus-binding peptides. Biomacromolecules 2021, 22, 3613–3623. [Google Scholar] [CrossRef] [PubMed]


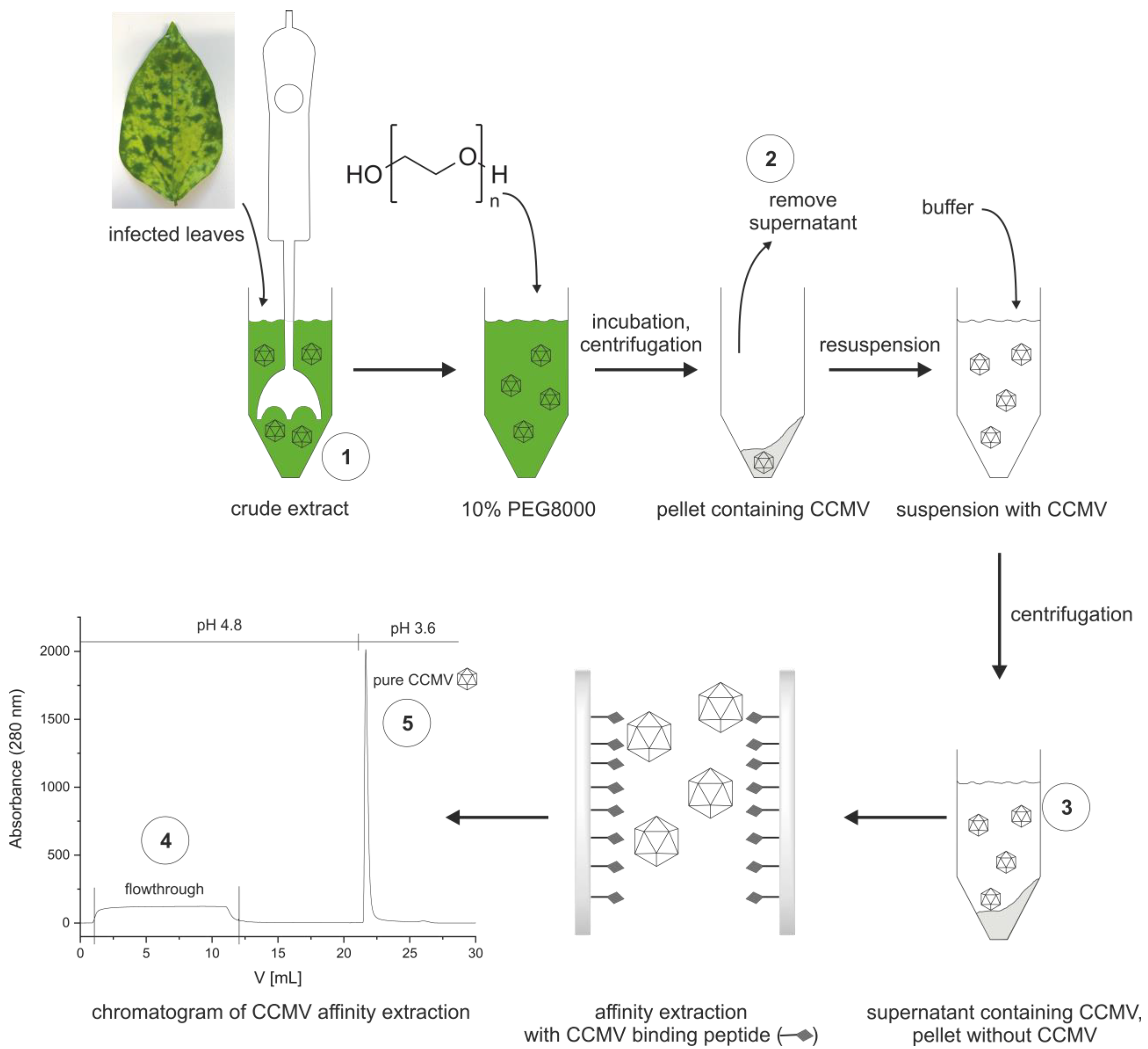

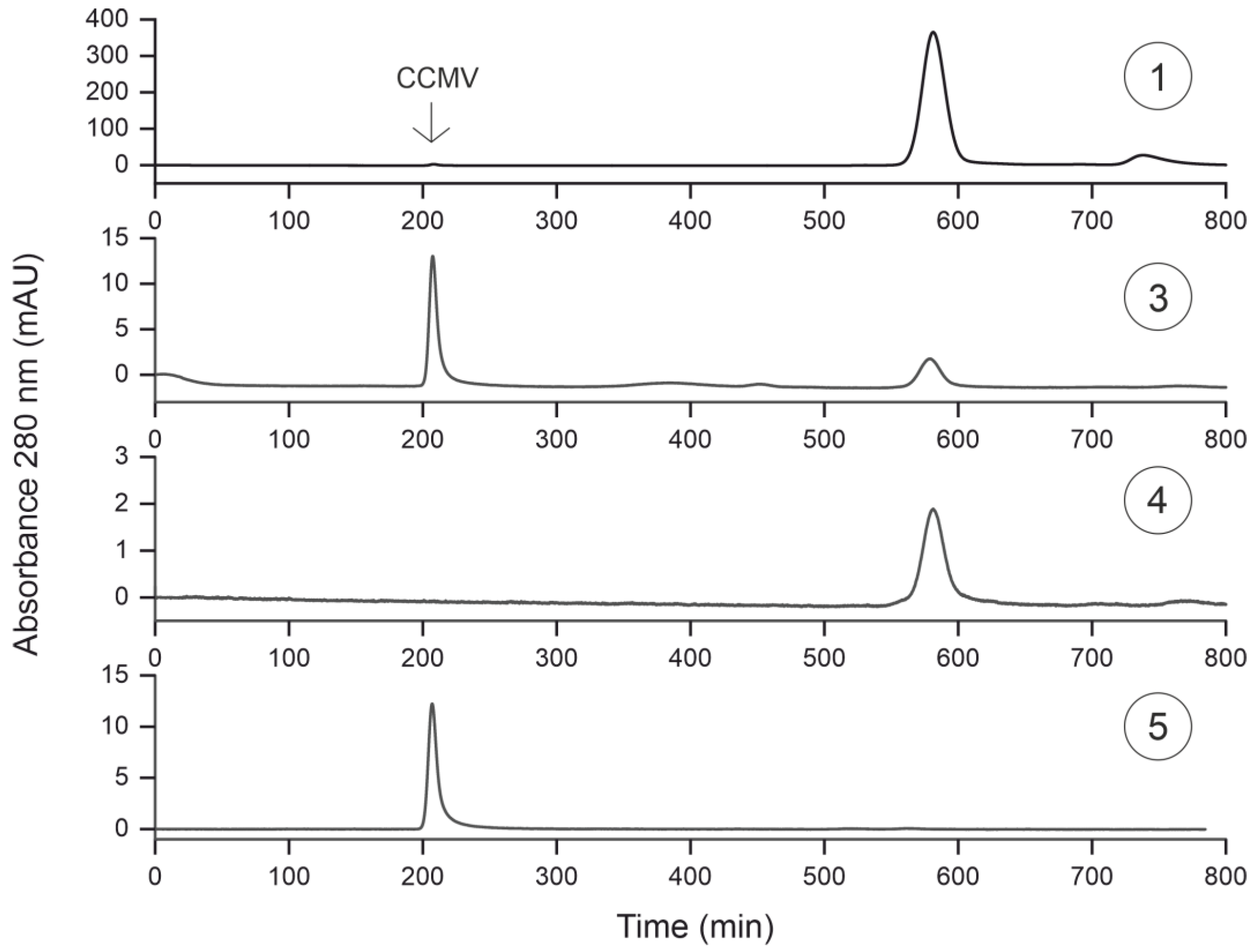
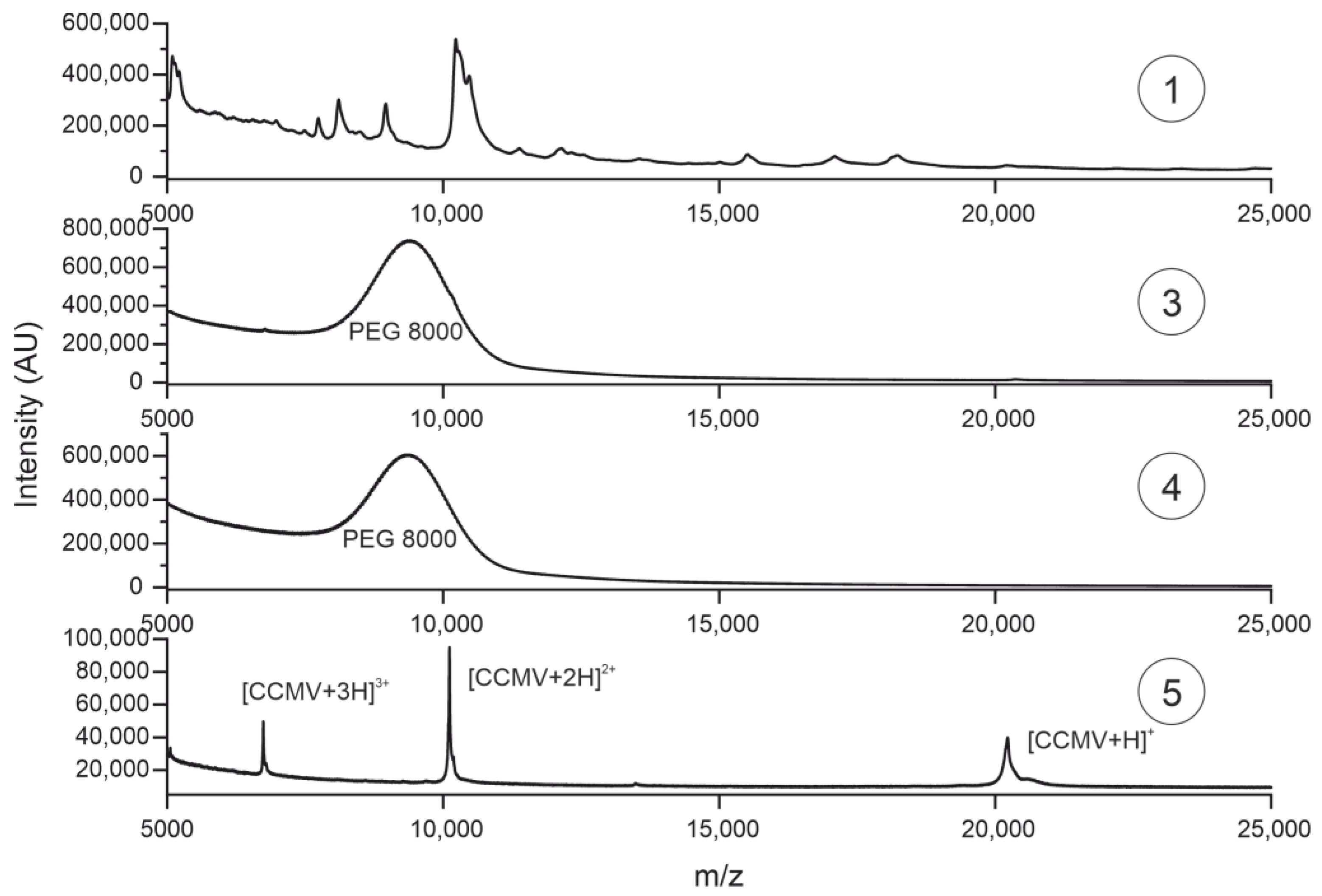

| Purification Step | Absolute Yield [mg] | Relative Yield [%] |
|---|---|---|
| ①—Crude extract | 3.52 ± 0.27 | 100 (def.) |
| ②—Supernatant of 1 | 0.040 ± 0.001 | (1.1) |
| ③—Resuspended pellet | 2.91 ± 0.04 | 83 |
| ④—Flowthrough of affinity column | <0.001 | (0) |
| ⑤—Eluate of affinity step | 1.57 ± 0.02 | 45 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tscheuschner, G.; Ponader, M.; Raab, C.; Weider, P.S.; Hartfiel, R.; Kaufmann, J.O.; Völzke, J.L.; Bosc-Bierne, G.; Prinz, C.; Schwaar, T.; et al. Efficient Purification of Cowpea Chlorotic Mottle Virus by a Novel Peptide Aptamer. Viruses 2023, 15, 697. https://doi.org/10.3390/v15030697
Tscheuschner G, Ponader M, Raab C, Weider PS, Hartfiel R, Kaufmann JO, Völzke JL, Bosc-Bierne G, Prinz C, Schwaar T, et al. Efficient Purification of Cowpea Chlorotic Mottle Virus by a Novel Peptide Aptamer. Viruses. 2023; 15(3):697. https://doi.org/10.3390/v15030697
Chicago/Turabian StyleTscheuschner, Georg, Marco Ponader, Christopher Raab, Prisca S. Weider, Reni Hartfiel, Jan Ole Kaufmann, Jule L. Völzke, Gaby Bosc-Bierne, Carsten Prinz, Timm Schwaar, and et al. 2023. "Efficient Purification of Cowpea Chlorotic Mottle Virus by a Novel Peptide Aptamer" Viruses 15, no. 3: 697. https://doi.org/10.3390/v15030697
APA StyleTscheuschner, G., Ponader, M., Raab, C., Weider, P. S., Hartfiel, R., Kaufmann, J. O., Völzke, J. L., Bosc-Bierne, G., Prinz, C., Schwaar, T., Andrle, P., Bäßler, H., Nguyen, K., Zhu, Y., Mey, A. S. J. S., Mostafa, A., Bald, I., & Weller, M. G. (2023). Efficient Purification of Cowpea Chlorotic Mottle Virus by a Novel Peptide Aptamer. Viruses, 15(3), 697. https://doi.org/10.3390/v15030697






