Rescue Therapy after Failure of HCV Antiviral Treatment with Interferon-Free Regimens
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Ethical Considerations
2.3. Statistical Analysis
3. Results
3.1. Characteristics of the Study Group
3.2. Treatment Regimens
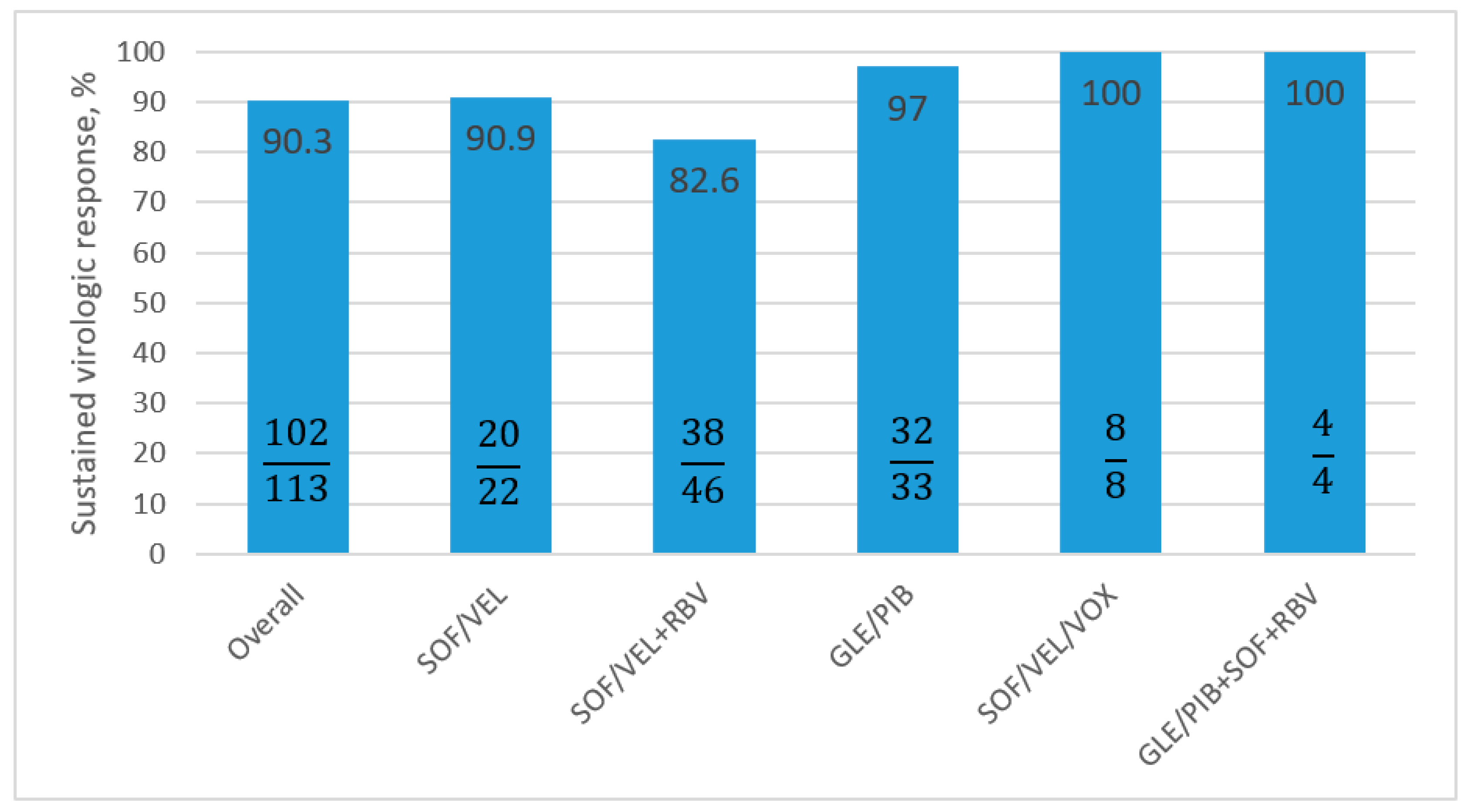
3.3. Antiviral Treatment Effectiveness
3.4. Safety
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Solitano, V.; Plaz Torres, M.C.; Pugliese, N.; Aghemo, A. Management and Treatment of Hepatitis C: Are There Still Unsolved Problems and Unique Populations? Viruses 2021, 13, 1048. [Google Scholar] [CrossRef]
- Stanaway, J.D.; Flaxman, A.D.; Naghavi, M.; Fitzmaurice, C.; Vos, T.; Abubakar, I.; Abu-Raddad, L.J.; Assadi, R.; Bhala, N.; Cowie, B.; et al. The Global Burden of Viral Hepatitis from 1990 to 2013: Findings from the Global Burden of Disease Study 2013. Lancet 2016, 388, 1081–1088. [Google Scholar] [CrossRef]
- Carrat, F.; Fontaine, H.; Dorival, C.; Simony, M.; Diallo, A.; Hezode, C.; De Ledinghen, V.; Larrey, D.; Haour, G.; Bronowicki, J.-P.; et al. Clinical Outcomes in Patients with Chronic Hepatitis C after Direct-Acting Antiviral Treatment: A Prospective Cohort Study. Lancet 2019, 393, 1453–1464. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Recommendations on Treatment of Hepatitis C: Final Update of the Series☆. J. Hepatol. 2020, 73, 1170–1218. [Google Scholar] [CrossRef]
- Wedemeyer, H.; Duberg, A.S.; Buti, M.; Rosenberg, W.M.; Frankova, S.; Esmat, G.; Örmeci, N.; Van Vlierberghe, H.; Gschwantler, M.; Akarca, U.; et al. Strategies to Manage Hepatitis C Virus (HCV) Disease Burden. J. Viral Hepat. 2014, 21 (Suppl. 1), 60–89. [Google Scholar] [CrossRef]
- AASLD-IDSA HCV Guidance Panel. Hepatitis C Guidance 2018 Update: AASLD-IDSA Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection. Clin. Infect. Dis. 2018, 67, 1477–1492. [Google Scholar] [CrossRef]
- World Health Organization. Global Health Sector Strategy on Viral Hepatitis 2016–2021. Towards Ending Viral Hepatitis; World Health Organization: Geneva, Switzerland, 2016; Available online: https://apps.who.int/iris/bitstream/handle/10665/246177/WHO-HIV-2016.06-eng.pdf (accessed on 9 January 2023).
- Parigi, T.L.; Torres, M.C.P.; Aghemo, A. Upcoming Direct Acting Antivirals for Hepatitis C Patients with a Prior Treatment Failure. Clin. Mol. Hepatol. 2019, 25, 360–365. [Google Scholar] [CrossRef]
- Lu, M.; Li, J.; Rupp, L.B.; Holmberg, S.D.; Moorman, A.C.; Spradling, P.R.; Teshale, E.H.; Zhou, Y.; Boscarino, J.A.; Schmidt, M.A.; et al. Hepatitis C Treatment Failure Is Associated with Increased Risk of Hepatocellular Carcinoma. J. Viral Hepat. 2016, 23, 718–729. [Google Scholar] [CrossRef]
- Sarrazin, C. Treatment Failure with DAA Therapy: Importance of Resistance. J. Hepatol. 2021, 74, 1472–1482. [Google Scholar] [CrossRef]
- Zarębska-Michaluk, D.; Buczyńska, I.; Simon, K.; Tudrujek-Zdunek, M.; Janczewska, E.; Dybowska, D.; Sitko, M.; Dobracka, B.; Jaroszewicz, J.; Pabjan, P.; et al. Real World Experience of Chronic Hepatitis C Retreatment with Genotype Specific Regimens in Nonresponders to Previous Interferon-Free Therapy. Can. J. Gastroenterol. Hepatol. 2019, 2019, 4029541. [Google Scholar] [CrossRef]
- Halota, W.; Flisiak, R.; Juszczyk, J.; Małkowski, P.; Pawłowska, M.; Simon, K.; Tomasiewicz, K. Recommendations of the Polish Group of Experts for HCV for the Treatment of Hepatitis C in 2020. Clin. Exp. Hepatol. 2020, 6, 163–169. [Google Scholar] [CrossRef]
- Reddy, K.R.; Bourlière, M.; Sulkowski, M.; Omata, M.; Zeuzem, S.; Feld, J.J.; Lawitz, E.; Marcellin, P.; Welzel, T.M.; Hyland, R.; et al. Ledipasvir and Sofosbuvir in Patients with Genotype 1 Hepatitis C Virus Infection and Compensated Cirrhosis: An Integrated Safety and Efficacy Analysis. Hepatology 2015, 62, 79–86. [Google Scholar] [CrossRef]
- Flisiak, R.; Zarębska-Michaluk, D.; Janczewska, E.; Łapiński, T.; Rogalska, M.; Karpińska, E.; Mikuła, T.; Bolewska, B.; Białkowska, J.; Flejscher-Stępniewska, K.; et al. Five-Year Follow-up of Cured HCV Patients under Real-World Interferon-Free Therapy. Cancers 2021, 13, 3694. [Google Scholar] [CrossRef]
- Butt, A.A.; Yan, P.; Shaikh, O.S.; Lo Re, V., 3rd; Abou-Samra, A.-B.; Sherman, K.E. Treatment of HCV Reduces Viral Hepatitis-Associated Liver-Related Mortality in Patients: An ERCHIVES Study. J. Hepatol. 2020, 73, 277–284. [Google Scholar] [CrossRef]
- Curry, M.P.; O’Leary, J.G.; Bzowej, N.; Muir, A.J.; Korenblat, K.M.; Fenkel, J.M.; Reddy, K.R.; Lawitz, E.; Flamm, S.L.; Schiano, T.; et al. Sofosbuvir and Velpatasvir for HCV in Patients with Decompensated Cirrhosis. N. Engl. J. Med. 2015, 373, 2618–2628. [Google Scholar] [CrossRef]
- Naggie, S.; Muir, A.J. Oral Combination Therapies for Hepatitis C Virus Infection: Successes, Challenges, and Unmet Needs. Annu. Rev. Med. 2017, 68, 345–358. [Google Scholar] [CrossRef]
- Konjeti, V.R.; John, B.V. Interaction between Hepatocellular Carcinoma and Hepatitis C Eradication with Direct-Acting Antiviral Therapy. Curr. Treat. Options Gastroenterol. 2018, 16, 203–214. [Google Scholar] [CrossRef]
- Zarębska-Michaluk, D.; Jaroszewicz, J.; Parfieniuk-Kowerda, A.; Janczewska, E.; Dybowska, D.; Pawłowska, M.; Halota, W.; Mazur, W.; Lorenc, B.; Janocha-Litwin, J.; et al. Effectiveness and Safety of Pangenotypic Regimens in the Most Difficult to Treat Population of Genotype 3 HCV Infected Cirrhotics. J. Clin. Med. 2021, 10, 3280. [Google Scholar] [CrossRef]
- Degasperi, E.; Spinetti, A.; Lombardi, A.; Landonio, S.; Rossi, M.C.; Pasulo, L.; Pozzoni, P.; Giorgini, A.; Fabris, P.; Romano, A.; et al. Real-Life Effectiveness and Safety of Sofosbuvir/Velpatasvir/Voxilaprevir in Hepatitis C Patients with Previous DAA Failure. J. Hepatol. 2019, 71, 1106–1115. [Google Scholar] [CrossRef]
- Bourlière, M.; Gordon, S.C.; Flamm, S.L.; Cooper, C.L.; Ramji, A.; Tong, M.; Ravendhran, N.; Vierling, J.M.; Tran, T.T.; Pianko, S.; et al. Sofosbuvir, Velpatasvir, and Voxilaprevir for Previously Treated HCV Infection. N. Engl. J. Med. 2017, 376, 2134–2146. [Google Scholar] [CrossRef]
- Belperio, P.S.; Shahoumian, T.A.; Loomis, T.P.; Backus, L.I. Real-World Effectiveness of Sofosbuvir/Velpatasvir/Voxilaprevir in 573 Direct-Acting Antiviral Experienced Hepatitis C Patients. J. Viral Hepat. 2019, 26, 980–990. [Google Scholar] [CrossRef]
- Xie, J.; Xu, B.; Wei, L.; Huang, C.; Liu, W. Effectiveness and Safety of Sofosbuvir/Velpatasvir/Voxilaprevir as a Hepatitis C Virus Infection Salvage Therapy in the Real World: A Systematic Review and Meta-Analysis. Infect. Dis. Ther. 2022, 11, 1661–1682. [Google Scholar] [CrossRef]
- Pearlman, B.; Perrys, M.; Hinds, A. Sofosbuvir/Velpatasvir/Voxilaprevir for Previous Treatment Failures with Glecaprevir/Pibrentasvir in Chronic Hepatitis C Infection. Am. J. Gastroenterol. 2019, 114, 1550–1552. [Google Scholar] [CrossRef]
- Mangia, A.; Milligan, S.; Khalili, M.; Fagiuoli, S.; Shafran, S.D.; Carrat, F.; Ouzan, D.; Papatheodoridis, G.; Ramji, A.; Borgia, S.M.; et al. Global Real-World Evidence of Sofosbuvir/Velpatasvir as Simple, Effective HCV Treatment: Analysis of 5552 Patients from 12 Cohorts. Liver Int. 2020, 40, 1841–1852. [Google Scholar] [CrossRef]
- Gane, E.J.; Shiffman, M.L.; Etzkorn, K.; Morelli, G.; Stedman, C.A.M.; Davis, M.N.; Hinestrosa, F.; Dvory-Sobol, H.; Huang, K.C.; Osinusi, A.; et al. Sofosbuvir-Velpatasvir with Ribavirin for 24 Weeks in Hepatitis C Virus Patients Previously Treated with a Direct-Acting Antiviral Regimen. Hepatology 2017, 66, 1083–1089. [Google Scholar] [CrossRef]
- Shen, C.; Fan, H.; Ge, Z.; Cai, W.; Shao, J.; Dong, C.; Xue, H.; Fu, Z.; Li, J.; Zhang, Y.; et al. Efficacy and Safety of Glecaprevir/Pibrentasvir in HCV Patients with Previous Direct-Acting Antiviral Therapy Failures: A Meta-Analysis. Front. Med. 2020, 7, 592472. [Google Scholar] [CrossRef]
- Poordad, F.; Pol, S.; Asatryan, A.; Buti, M.; Shaw, D.; Hézode, C.; Felizarta, F.; Reindollar, R.W.; Gordon, S.C.; Pianko, S.; et al. Glecaprevir/Pibrentasvir in Patients with Hepatitis C Virus Genotype 1 or 4 and Past Direct-Acting Antiviral Treatment Failure. Hepatology 2018, 67, 1253–1260. [Google Scholar] [CrossRef]
- Berkan-Kawińska, A.; Piekarska, A.; Janczewska, E.; Lorenc, B.; Tudrujek-Zdunek, M.; Tomasiewicz, K.; Berak, H.; Horban, A.; Zarębska-Michaluk, D.; Pabjan, P.; et al. Real-World Effectiveness and Safety of Direct-Acting Antivirals in Patients with Cirrhosis and History of Hepatic Decompensation: Epi-Ter2 Study. Liver Int. 2021, 41, 1789–1801. [Google Scholar] [CrossRef]
- Ridruejo, E.; Piñero, F.; Mendizabal, M.; Cheinquer, H.; Wolff, F.H.; Anders, M.; Reggiardo, V.; Ameigeiras, B.; Palazzo, A.; Alonso, C.; et al. Decompensated Cirrhosis and Liver Transplantation Negatively Impact in DAA Treatment Response: Real-World Experience from HCV-LALREAN Cohort. J. Med. Virol. 2020, 92, 3545–3555. [Google Scholar] [CrossRef]
- Llaneras, J.; Riveiro-Barciela, M.; Lens, S.; Diago, M.; Cachero, A.; García-Samaniego, J.; Conde, I.; Arencibia, A.; Arenas, J.; Gea, F.; et al. Effectiveness and Safety of Sofosbuvir/Velpatasvir/Voxilaprevir in Patients with Chronic Hepatitis C Previously Treated with DAAs. J. Hepatol. 2019, 71, 666–672. [Google Scholar] [CrossRef]
- Lok, A.S.; Sulkowski, M.S.; Kort, J.J.; Willner, I.; Reddy, K.R.; Shiffman, M.L.; Hassan, M.A.; Pearlman, B.L.; Hinestrosa, F.; Jacobson, I.M.; et al. Efficacy of Glecaprevir and Pibrentasvir in Patients with Genotype 1 Hepatitis C Virus Infection with Treatment Failure after NS5A Inhibitor plus Sofosbuvir Therapy. Gastroenterology 2019, 157, 1506–1517.e1. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wu, K.-H.; Li, J.; Moorman, A.C.; Spradling, P.R.; Teshale, E.H.; Boscarino, J.A.; Daida, Y.G.; Schmidt, M.A.; Rupp, L.B.; et al. Adjuvant Ribavirin and Longer Direct-Acting Antiviral Treatment Duration Improve Sustained Virological Response among Hepatitis C Patients at Risk of Treatment Failure. J. Viral Hepat. 2019, 26, 1210–1217. [Google Scholar] [CrossRef] [PubMed]

| Regimen | NS3 Inhibitor | NS5A Inhibitor | NS5B Inhibitor |
|---|---|---|---|
| Genotype-specific | |||
| ASV+DCV | asunaprevir | daclatasvir | |
| LDV/SOF±RBV | ledipasvir | sofosbuvir | |
| OBV/PTV/r±DSV±RBV | paritaprevir | ombitasvir | dasabuvir |
| GZR/EBR±RBV | grazoprevir | elbasvir | |
| Pangenotypic | |||
| GLE/PIB | glecaprevir | pibrentasvir | |
| GLE/PIB+SOF+RBV | glecaprevir | pibrentasvir | sofosbuvir |
| SOF/VEL±RBV | velpatasvir | sofosbuvir | |
| SOF/VEL/VOX | voxilaprevir | velpataswir | sofosbuvir |
| Parameter | All Patients, n = 120 |
|---|---|
| Gender, females/males, n(%) | 33 (27.5)/87 (72.5) |
| Age [years], median (IQR) | 53 (40–63.5) |
| Females | 62 (58–69) |
| Males | 49 (38–61) |
| BMI, median (IQR) | 26.7 (23.8–29.7) |
| Comorbidities, n(%) | |
| Any comorbidity | 72 (60) |
| Hypertension | 39 (32.5) |
| Diabetes | 11 (9.2) |
| Renal disease | 6 (5) |
| Autoimmune diseases | 2 (1.7) |
| Non-HCC tumours | 3 (2.5) |
| Other | 47 (39.2) |
| Concomitant medications, n(%) | 79 (65.8) |
| ALT IU/L, median (IQR) | 59 (40.4–97.5) |
| Bilirubin mg/dL, median (IQR) | 0.6 (0.5–1) |
| Albumin g/dL, median (IQR) | 4.1 (3.8–4.5) |
| Creatinine mg/dL/GFR (ml/min)/1.73 m2, median (IQR) | 0.9 (0.7–1)/98.6 (88.1–111) |
| Haemoglobin g/dL, median (IQR) | 14.8 (14–16) |
| Platelets, ×1000/µL, median (IQR) | 173 (120–233.5) |
| HCV RNA ×106 IU/mL, median (IQR) | 1.5 (0.6–3.7) |
| Parameter | All Patients, n = 120 |
|---|---|
| GT, n(%) | |
| 1 | 0 |
| 1a | 9 (7.5) |
| 1b | 103 (85.8) |
| 2 | 0 |
| 3 | 2 (1.7) |
| 4 | 5 (4.2) |
| 5 | 0 |
| 6 | 0 |
| Mixed genotype | 1 (0.8) |
| Liver fibrosis, n(%) | |
| F0 | 1 (0.8) |
| F1 | 32 (26.6) |
| F2 | 26 (21.7) |
| F3 | 17 (14.2) |
| F4 | 42 (35) |
| No data | 2 (1.7) |
| Child-Pugh, n(%) | |
| B | 6 (5) |
| C | 1 (0.8) |
| History of hepatic decompensation, n(%) | |
| Ascites | 7 (5.8) |
| Encephalopathy | 2 (1.7) |
| Documented oesophageal varices, n(%) | 11 (9.2) |
| Hepatic decompensation at baseline, n(%) | |
| Moderate ascites—responded to diuretics | 5 (4.2) |
| Tense ascites—not responded to diuretics | 0 |
| Encephalopathy | 1 (0.8) |
| HCC history, n(%) | 0 |
| OLTx history, n(%) | 1 (0.8) |
| HBV coinfection (HBsAg+), n(%) | 2 (1.7) |
| HIV coinfection, n(%) | 13 (10.8) |
| Parameter | All Patients, n = 120 |
|---|---|
| History of previous therapy, n(%) | |
| Nonresponder | 31 (25.8) |
| Relapser | 77 (64.2) |
| Discontinuation due to safety reason | 5 (4.2) |
| Unknown type of response | 7 (5.8) |
| Number of patients with previous treatment failure, n(%) | |
| ASV+DCV | 12 (10) |
| LDV/SOF±RBV | 36 (30) |
| OBV/PTV/r±DSV±RBV | 39 (32.5) |
| GZR/EBR±RBV | 33 (27.5) |
| Current treatment regimen, n(%)1 July 2015–30 June 2022 | |
| GLE/PIB | 34 (28.3) |
| GLE/PIB+SOF+RBV | 4 (3.3) |
| SOF/VEL±RBV | 74 (61.7) |
| SOF/VEL+RBV | 49 (40.9) |
| SOF/VELMay 2021–30 June 2022 | 25 (20.8) |
| SOF/VEL/VOX | 8 (6.7) |
| Patient | Age | GT | F, CP | Current Regimen | History of Previous Therapy | Baseline HCV RNA × 106 IU/mL | Treatment Course | EOT |
|---|---|---|---|---|---|---|---|---|
| Male 1 | 64 | 1b | 4, A | SOF/VEL+RBV | Relapser, LDV/SOF±RBV | 0.276 | according to schedule | TD |
| Male 2 | 43 | 1b | 2, A | SOF/VEL+RBV | Relapser, LDV/SOF±RBV | 0.878 | therapy discontinuation | TND |
| Male 3 | 62 | 1b | 2, A | SOF/VEL | Relapser, GZR/EBR | 3.88 | according to schedule | TD |
| Male 4 | 58 | 1b | 4, A | SOF/VEL+RBV | Relapser, LDV/SOF±RBV | 6.7 | according to schedule | TD |
| Male 5 | 40 | 1b | 4, A | SOF/VEL+RBV | Relapser, LDV/SOF±RBV | 0.907 | according to schedule | TD |
| Male 6 | 61 | 1b | 4, A | SOF/VEL | Relapser, OBV/PTV/r+DSV±RBV | 0.495 | according to schedule | TND |
| Woman 1 | 67 | 1b | 3, A | SOF/VEL+RBV | Discontinuation due to safety reason, LDV/SOF±RBV | 2.63 | therapy discontinuation | TND |
| Woman 2 | 61 | 1b | 4, A | SOF/VEL+RBV | Relapser, LDV/SOF±RBV | 0.513 | according to schedule | TD |
| Woman 3 | 63 | 1b | 4, A | SOF/VEL+RBV | Relapser, ASV+DCV | 1.3 | according to schedule | TD |
| Woman 4 | 71 | 1b | 4, A | GLE/PIB | Nonrespoder, GZR/EBR | 7.32 | according to schedule | TND |
| Woman 5 | 69 | 1b | 3, A | SOF/VEL+RBV | Nonrespoder, GZR/EBR | 6.04 | according to schedule | TND |
| Parameter | Responders, n = 102 | Non-Responders, n = 11 | P1 |
|---|---|---|---|
| Gender, females/males, n(%) | 26 (25.5)/76 (74.5) | 5 (45.5)/6 (54.5) | 0.1585 2 |
| Age [years], median (IQR) | 51.5 (40–63) | 62 (58–67) | 0.0533 3 |
| Females | 61 (43–70) | 67 (63–69) | 0.2400 3 |
| Males | 48.5 (37.5–58) | 59.5 (43–62) | 0.3920 3 |
| BMI, median (IQR) | 26.7 (23.9–29.4) | 29.3 (25.4–33.5) | 0.2279 3 |
| Current treatment regimen, n(%) | |||
| GLE/PIB | 32 (31.4) | 1 (9.1) | 0.1115 |
| GLE/PIB+SOF+RBV | 4 (3.9) | 0 | 0.6600 |
| SOF/VEL±RBV | 58 (56.9) | 10 (90.9) | 0.0251 |
| SOF/VEL | 20 (19.6) | 2 (18.2) | 0.6360 |
| SOF/VEL+RBV | 38 (37.3) | 8 (72.7) | 0.0262 |
| SOF/VEL/VOX | 8 (7.8) | 0 | 0.4286 |
| GT, n(%) | |||
| GT1b | 87 (85.3) | 11 (100) | 0.1720 2 |
| non-GT1b | 15 (14.7) | 0 | |
| Comorbidities, n(%) | |||
| Any comorbidity | 61 (59.8) | 7 (63.6) | 0.8051 2 |
| Hypertension | 36 (35.3) | 3 (27.3) | 0.4332 |
| Diabetes | 9 (8.8) | 1 (9.1) | 0.6569 |
| Renal disease | 6 (5.9) | 0 | 0.5330 |
| Autoimmune diseases | 2 (2) | 0 | 0.8140 |
| Non-HCC tumours | 1 (1) | 1 (9.1) | 0.1860 |
| Other | 41 (40.2) | 3 (27.3) | 0.3116 |
| Concomitant medications, n(%) | 64 (62.7) | 9 (81.8) | 0.2088 2 |
| History of previous therapy, n(%) | |||
| Nonresponder | 26 (25.5) | 2 (18.2) | 0.4551 |
| Relapser | 69 (67.6) | 8 (72.7) | 0.5127 |
| Discontinuation due to safety reason | 2 (2) | 1 (9.1) | 0.2667 |
| Unknown type of response | 5 (4.9) | 0 | 0.5934 |
| Number of patients with treatment failure, n(%) | |||
| ASV+DCV | 11 (10.8) | 1 (9.1) | 0.6699 |
| LDV/SOF±RBV | 26 (25.5) | 6 (54.5) | 0.0513 |
| OBV/PTV/r±DSV±RBV | 35 (34.3) | 1 (9.1) | 0.0794 |
| GZR/EBR±RBV | 30 (29.4) | 3 (27.3) | 0.6212 |
| History of hepatic decompensation, n(%) | |||
| Ascites | 4 (3.9) | 2 (18.2) | 0.1044 |
| Encephalopathy | 1 (1) | 0 | 0.9026 |
| Documented oesophageal varices, n(%) | 10 (9.8) | 1 (9.1) | 0.7095 |
| Hepatic decompensation at baseline, n(%) | |||
| Moderate ascites—responded to diuretics | 3 (2.9) | 0 | 0.7333 |
| Tense ascites—not responded to diuretics | 0 | 0 | NA |
| Encephalopathy | 0 | 0 | NA |
| HCC history, n(%) | 0 | 0 | NA |
| OLTx history, n(%) | 1 (1) | 0 | 0.9026 |
| Liver fibrosis, n(%) | |||
| F0-F3 | 68 (68.6) | 4 (36.4) | 0.0424 |
| F4 | 32 (31.4) | 7 (63.6) | |
| No data | 2 (2) | 0 | |
| Child-Pugh, n(%) | |||
| B | 5 (4.9) | 0 | 0.4526 2 |
| C | 0 | 0 | NA |
| HBV coinfection (HBsAg+), n(%) | 2 (2) | 0 | 0.8140 |
| HIV coinfection, n(%) | 10 (9.8) | 1 (9.1) | 0.7095 |
| ALT IU/L, median (IQR) | 59 (40.7–99) | 62 (35–76) | 0.6913 3 |
| Bilirubin mg/dL, median (IQR) | 0.6 (0.5–1) | 0.8 (0.5–1) | 0.6877 3 |
| Albumin g/dL, median (IQR) | 4.1 (3.9–4.5) | 3.9 (3.6–4.2) | 0.1969 3 |
| Creatinine mg/dL, median (IQR) | 0.9 (0.7–1) | 0.8 (0.7–1) | 0.3232 3 |
| Haemoglobin g/dL, median (IQR) | 15.2 (14–16) | 14 (13.2–15.9) | 0.1782 3 |
| Platelets, ×1000/µL, median (IQR) | 173 (127–234) | 188 (84–237) | 0.6247 3 |
| HCV RNA ×106 IU/ml, median (IQR) | 1.6 (0.6–3.6) | 1.3 (0.5–6) | 0.7751 3 |
| Parameter | All Patients, n = 120 |
|---|---|
| Treatment course, n(%) | |
| according to schedule | 109 (90.8) |
| therapy modification (including RBV dose) | 5 (4.2) |
| therapy discontinuation | 3 (2.5) |
| No data | 3 (2.5) |
| Patients with at least one AE, n(%) | 28 (23.3) |
| Serious adverse events, n(%) | 4 (3.3) |
| AEs leading to treatment discontinuation, n(%) | 3 (2.5) |
| Most common AEs (≥2%), n(%) | |
| weakness/fatigue | 12 (10) |
| Anaemia | 3 (2.5) |
| AEs of particular interest (cirrhotic), n(%) | |
| Ascites | 1 (0.8) |
| hepatic encephalopathy | 1 (0.8) |
| gastrointestinal bleeding | 0 |
| Liver decompensation—OLTx, n(%) | 0 |
| Liver acute rejection in patients after liver transplantation, n(%) | 0 |
| HCC de novo during or after therapy, n(%) | 1 (0.8) |
| Death, n(%) | 1 (0.8) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tronina, O.; Brzdęk, M.; Zarębska-Michaluk, D.; Dybowska, D.; Lorenc, B.; Janczewska, E.; Mazur, W.; Parfieniuk-Kowerda, A.; Piekarska, A.; Krygier, R.; et al. Rescue Therapy after Failure of HCV Antiviral Treatment with Interferon-Free Regimens. Viruses 2023, 15, 677. https://doi.org/10.3390/v15030677
Tronina O, Brzdęk M, Zarębska-Michaluk D, Dybowska D, Lorenc B, Janczewska E, Mazur W, Parfieniuk-Kowerda A, Piekarska A, Krygier R, et al. Rescue Therapy after Failure of HCV Antiviral Treatment with Interferon-Free Regimens. Viruses. 2023; 15(3):677. https://doi.org/10.3390/v15030677
Chicago/Turabian StyleTronina, Olga, Michał Brzdęk, Dorota Zarębska-Michaluk, Dorota Dybowska, Beata Lorenc, Ewa Janczewska, Włodzimierz Mazur, Anna Parfieniuk-Kowerda, Anna Piekarska, Rafał Krygier, and et al. 2023. "Rescue Therapy after Failure of HCV Antiviral Treatment with Interferon-Free Regimens" Viruses 15, no. 3: 677. https://doi.org/10.3390/v15030677
APA StyleTronina, O., Brzdęk, M., Zarębska-Michaluk, D., Dybowska, D., Lorenc, B., Janczewska, E., Mazur, W., Parfieniuk-Kowerda, A., Piekarska, A., Krygier, R., Klapaczyński, J., Berak, H., Jaroszewicz, J., Garlicki, A., Tomasiewicz, K., Citko, J., & Flisiak, R. (2023). Rescue Therapy after Failure of HCV Antiviral Treatment with Interferon-Free Regimens. Viruses, 15(3), 677. https://doi.org/10.3390/v15030677








