HIV Infection, Chromosome Instability, and Micronucleus Formation
Abstract
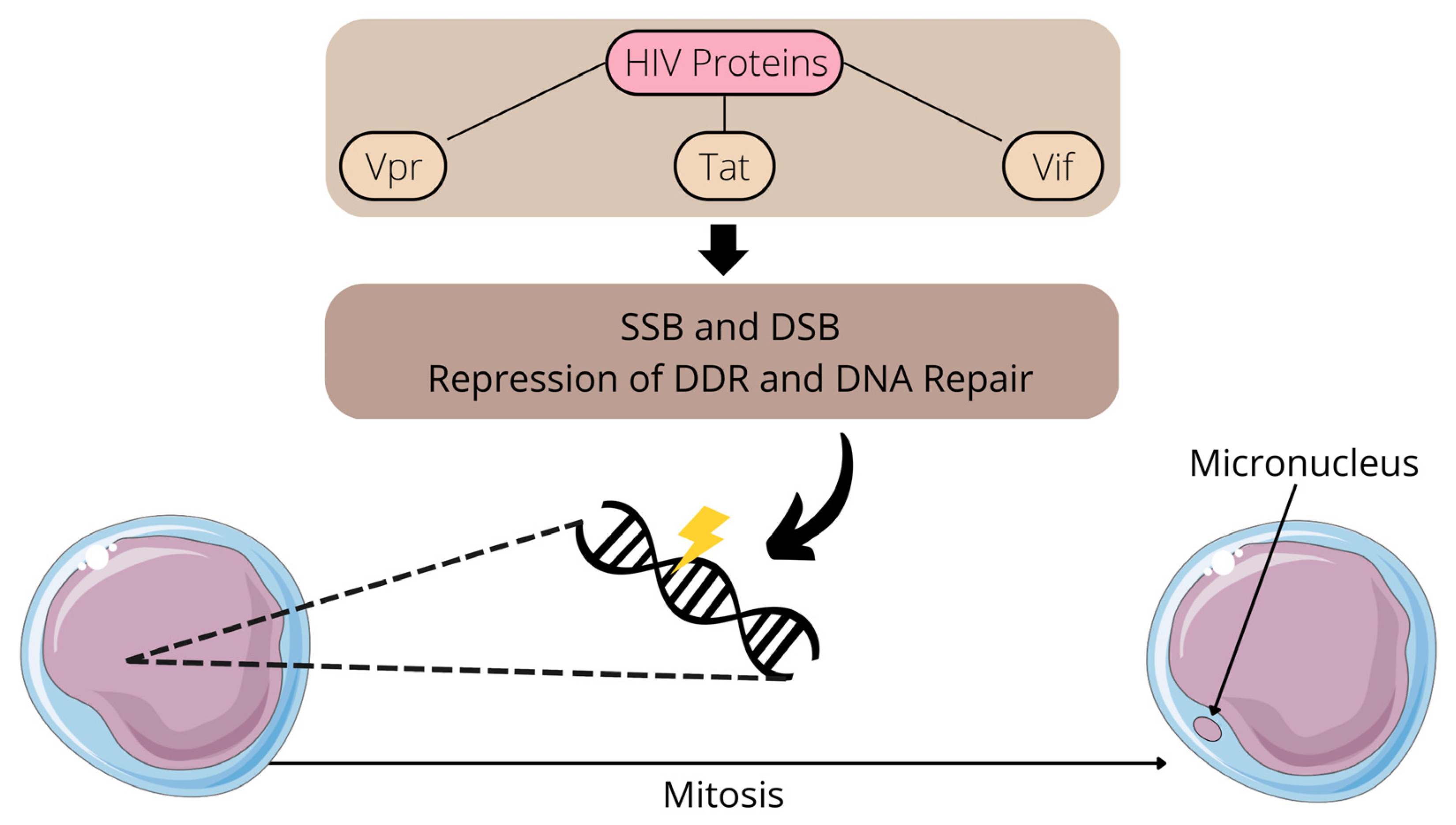
1. Introduction
2. Impacts of HIV Infection and Treatment on Chromosomal Integrity: A Focus on Human Studies
Potential Lifestyle and Nutritional Interventions to Be Used in Association with ART
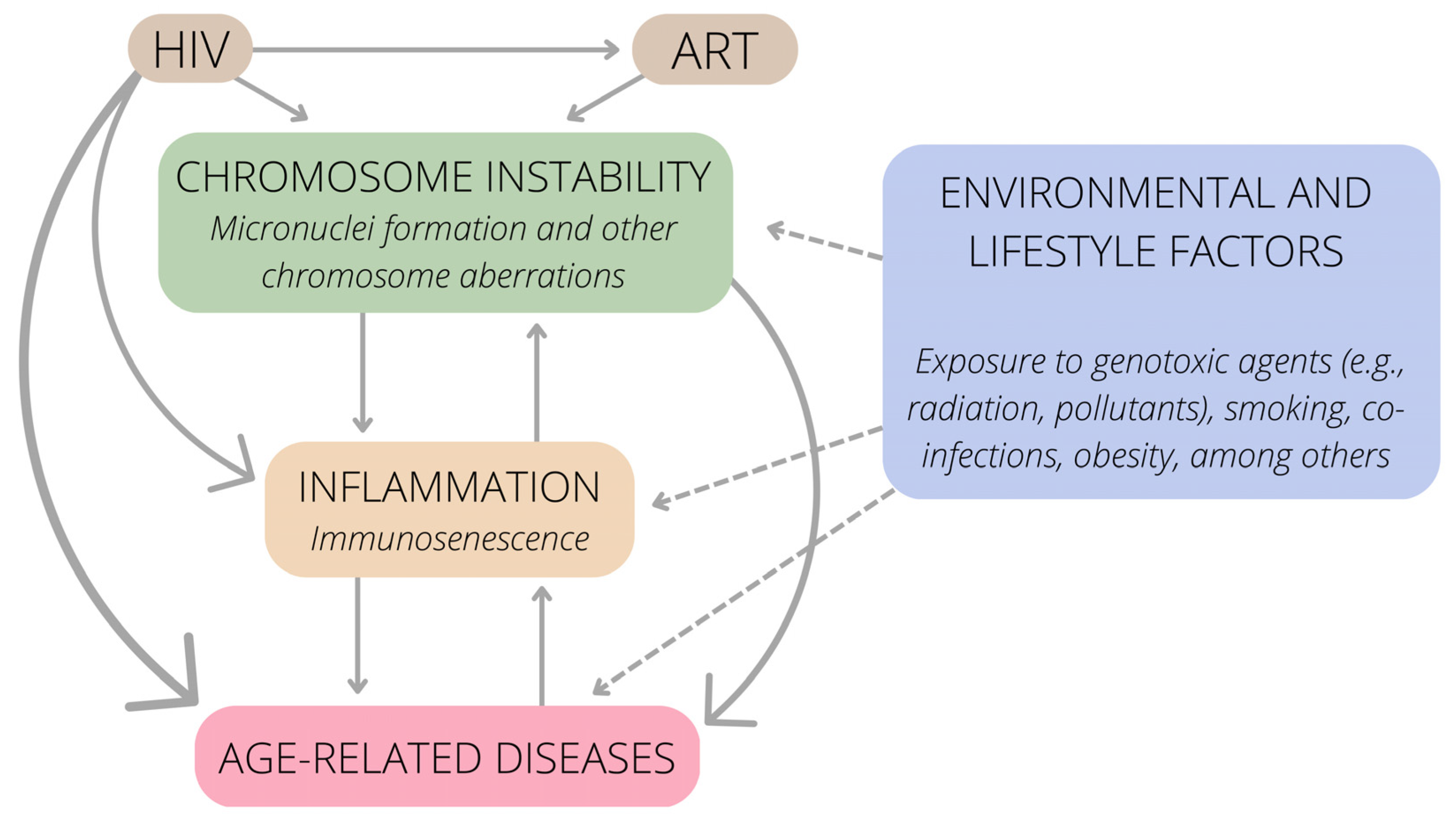
3. Immunosenescence, HIV Infection and Chromosome Instability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schumacher, B.; Pothof, J.; Vijg, J.; Hoeijmakers, J.H.J. The central role of DNA damage in the ageing process. Nature 2021, 592, 695–703. [Google Scholar] [CrossRef]
- Yousefzadeh, M.; Henpita, C.; Vyas, R.; Soto-Palma, C.; Robbins, P.; Niedernhofer, L. DNA damage-how and why we age? Elife 2021, 10, e62852. [Google Scholar] [CrossRef] [PubMed]
- Valverde, M.; Rojas, E. Environmental and occupational biomonitoring using the Comet assay. Mutat. Res. 2009, 681, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Fenech, M.; Bonassi, S. The effect of age, gender, diet and lifestyle on DNA damage measured using micronucleus frequency in human peripheral blood lymphocytes. Mutagenesis 2011, 26, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Drews, R.M.; Hernando, B.; Tarabichi, M.; Haase, K.; Lesluyes, T.; Smith, P.S.; Morrill Gavarró, L.; Couturier, D.L.; Liu, L.; Schneider, M.; et al. A pan-cancer compendium of chromosomal instability. Nature 2022, 606, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.; Leibowitz, M.L.; Lee, J.H. Small but mighty: The causes and consequences of micronucleus rupture. Exp. Mol. Med. 2020, 52, 1777–1786. [Google Scholar] [CrossRef]
- Bonassi, S.; Znaor, A.; Ceppi, M.; Lando, C.; Chang, W.P.; Holland, N.; Kirsch-Volders, M.; Zeiger, E.; Ban, S.; Barale, R.; et al. An increased micronucleus frequency in peripheral blood lymphocytes predicts the risk of cancer in humans. Carcinogenesis 2007, 28, 625–631. [Google Scholar] [CrossRef]
- Bonassi, S.; Coskun, E.; Ceppi, M.; Lando, C.; Bolognesi, C.; Burgaz, S.; Holland, N.; Kirsh-Volders, M.; Knasmueller, S.; Zeiger, E.; et al. The HUman MicroNucleus project on eXfoLiated buccal cells (HUMNXL): The role of life-style, host factors, occupational exposures, health status, and assay protocol. Mutat. Res. 2011, 728, 88–97. [Google Scholar] [CrossRef]
- Fenech, M.; Knasmueller, S.; Bolognesi, C.; Holland, N.; Bonassi, S.; Kirsch-Volders, M. Micronuclei as biomarkers of DNA damage, aneuploidy, inducers of chromosomal hypermutation and as sources of pro-inflammatory DNA in humans. Mutat. Res. Rev. Mutat. Res. 2020, 786, 108342. [Google Scholar] [CrossRef]
- Tang, S.; Stokasimov, E.; Cui, Y.; Pellman, D. Breakage of cytoplasmic chromosomes by pathological DNA base excision repair. Nature 2022, 606, 930–936. [Google Scholar] [CrossRef]
- Weitzman, M.D.; Weitzman, J.B. What’s the damage? The impact of pathogens on pathways that maintain host genome integrity. Cell Host Microbe 2014, 15, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Katerji, M.; Duerksen-Hughes, P.J. DNA damage in cancer development: Special implications in viral oncogenesis. Am. J. Cancer Res. 2021, 11, 3956–3979. [Google Scholar] [PubMed]
- Lopez, A.; Nichols Doyle, R.; Sandoval, C.; Nisson, K.; Yang, V.; Fregoso, O.I. Viral modulation of the DNA damage response and innate immunity: Two sides of the same coin. J. Mol. Biol. 2022, 434, 167327. [Google Scholar] [CrossRef] [PubMed]
- Shimura, M.; Tanaka, Y.; Nakamura, S.; Minemoto, Y.; Yamashita, K.; Hatake, K.; Takaku, F.; Ishizaka, Y. Micronuclei formation and aneuploidy induced by Vpr, an accessory gene of human immunodeficiency virus type 1. FASEB J. 1999, 13, 621–637. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.; Re, F.; Sebastian, S.; Sazer, S.; Luban, J. HIV-1 Vpr induces defects in mitosis, cytokinesis, nuclear structure, and centrosomes. Mol. Biol. Cell 2004, 15, 1793–1801. [Google Scholar] [CrossRef][Green Version]
- Moraes Filho, A.V.; Carvalho, C.J.S.; Carneiro, C.C.; Vale, C.R.; Lima, D.C.S.; Carvalho, W.F.; Vieira, T.B.; Silva, D.M.; Cunha, K.S.; Chen-Chen, L. Genotoxic and cytotoxic effects of antiretroviral combinations in mice bone marrow. PLoS ONE 2016, 11, e0165706. [Google Scholar] [CrossRef]
- Montano, M.; Oursler, K.K.; Xu, K.; Sun, Y.V.; Marconi, C.V. Biological ageing with HIV infection: Evaluating the geroscience hypothesis. Lancet Healthy Longev. 2022, 3, e194–e205. [Google Scholar] [CrossRef]
- Deeks, S.G.; Lewin, S.R.; Havlir, D.V. The end of AIDS: HIV infection as a chronic disease. Lancet 2013, 382, 1525–1533. [Google Scholar] [CrossRef]
- Valverde-Villegas, J.M.; de Medeiros, R.M.; Ellwanger, J.H.; Santos, B.R.; Melo, M.G.; Almeida, S.E.M.; Chies, J.A.B. High CXCL10/IP-10 levels are a hallmark in the clinical evolution of the HIV infection. Infect. Genet. Evol. 2018, 57, 51–58. [Google Scholar] [CrossRef]
- Zicari, S.; Sessa, L.; Cotugno, N.; Ruggiero, A.; Morrocchi, E.; Concato, C.; Rocca, S.; Zangari, P.; Manno, E.C.; Palma, P. Immune activation, inflammation, and non-AIDS co-morbidities in HIV-infected patients under long-term ART. Viruses 2019, 11, 200. [Google Scholar] [CrossRef]
- Ellwanger, J.H.; Valverde-Villegas, J.M.; Kaminski, V.L.; de Medeiros, R.M.; Almeida, S.E.M.; Santos, B.R.; de Melo, M.G.; Hackenhaar, F.S.; Chies, J.A.B. Increased IL-8 levels in HIV-infected individuals who initiated ART with CD4+ T cell counts <350 cells/mm3-A potential hallmark of chronic inflammation. Microbes Infect. 2020, 22, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Kay, J.; Thadhani, E.; Samson, L.; Engelward, B. Inflammation-induced DNA damage, mutations and cancer. DNA Repair 2019, 83, 102673. [Google Scholar] [CrossRef] [PubMed]
- Nardacci, R.; Ciccosanti, F.; Marsella, C.; Ippolito, G.; Piacentini, M.; Fimia, G.M. Role of autophagy in HIV infection and pathogenesis. J. Intern. Med. 2017, 281, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.; Jena, K.K.; Mehto, S.; Chauhan, N.R.; Sahu, R.; Dhar, K.; Yadav, R.; Krishna, S.; Jaiswal, P.; Chauhan, S. Innate immunity and inflammophagy: Balancing the defence and immune homeostasis. FEBS J. 2022, 289, 4112–4131. [Google Scholar] [CrossRef]
- Talens, F.; Van Vugt, M.A.T.M. Inflammatory signaling in genomically instable cancers. Cell Cycle 2019, 18, 1830–1848. [Google Scholar] [CrossRef]
- Krupina, K.; Goginashvili, A.; Cleveland, D.W. Causes and consequences of micronuclei. Curr. Opin. Cell Biol. 2021, 70, 91–99. [Google Scholar] [CrossRef]
- Rello-Varona, S.; Lissa, D.; Shen, S.; Niso-Santano, M.; Senovilla, L.; Mariño, G.; Vitale, I.; Jemaá, M.; Harper, F.; Pierron, G.; et al. Autophagic removal of micronuclei. Cell Cycle 2012, 11, 170–176. [Google Scholar] [CrossRef]
- Van Grol, J.; Subauste, C.; Andrade, R.M.; Fujinaga, K.; Nelson, J.; Subauste, C.S. HIV-1 inhibits autophagy in bystander macrophage/monocytic cells through Src-Akt and STAT3. PLoS ONE. 2010, 5, e11733. [Google Scholar] [CrossRef]
- Dinkins, C.; Pilli, M.; Kehrl, J.H. Roles of autophagy in HIV infection. Immunol. Cell Biol. 2015, 93, 11–17. [Google Scholar] [CrossRef]
- Sardo, L.; Iordanskiy, S.; Klase, Z.; Kashanchi, F. HIV-1 Nef blocks autophagy in human astrocytes. Cell Cycle 2015, 14, 3781–3782. [Google Scholar] [CrossRef]
- Thomas, P.; Holland, N.; Bolognesi, C.; Kirsch-Volders, M.; Bonassi, S.; Zeiger, E.; Knasmueller, S.; Fenech, M. Buccal micronucleus cytome assay. Nat. Protoc. 2009, 4, 825–837. [Google Scholar] [CrossRef]
- Lima, C.F.; Alves, M.G.O.; Furtado, J.J.D.; Marcucci, M.; Balducci, I.; Almeida, J.D. Effect of HIV infection in the micronuclei frequency on the oral mucosa. J. Oral Pathol. Med. 2017, 46, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Baeyens, A.; Slabbert, J.P.; Willem, P.; Jozela, S.; Van Der Merwe, D.; Vral, A. Chromosomal radiosensitivity of HIV positive individuals. Int. J. Radiat. Biol. 2010, 86, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Herd, O.; Francies, F.; Slabbert, J.; Baeyens, A. The effect of HIV and antiretroviral therapy on chromosomal radiosensitivity. J. AIDS Clin. Res. 2014, 5, 12. [Google Scholar] [CrossRef]
- Herd, O.; Francies, F.; Kotzen, J.; Smith, T.; Nxumalo, Z.; Muller, X.; Slabbert, J.; Vral, A.; Baeyens, A. Chromosomal radiosensitivity of human immunodeficiency virus positive/negative cervical cancer patients in South Africa. Mol. Med. Rep. 2016, 13, 130–136. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zizza, A.; Grima, P.; Andreassi, M.G.; Tumolo, M.R.; Borghini, A.; De Donno, A.; Negro, P.; Guido, M. HIV infection and frequency of micronucleus in human peripheral blood cells. J. Prev. Med. Hyg. 2019, 60, E191–E196. [Google Scholar] [CrossRef]
- Stern, M.; Cid, M.G.; Larripa, I.; Slavutsky, I. AZT-induction of micronuclei in human lymphocyte subpopulations. Toxicol. Lett. 1994, 70, 235–242. [Google Scholar] [CrossRef]
- Ayers, K.M.; Clive, D.; Tucker, W.E., Jr.; Hajian, G.; de Miranda, P. Nonclinical toxicology studies with zidovudine: Genetic toxicity tests and carcinogenicity bioassays in mice and rats. Fundam. Appl. Toxicol. 1996, 32, 148–158. [Google Scholar] [CrossRef]
- Bayram, S.; Topaktaş, M. Confirmation of the chromosome damaging effects of lamivudine in in vitro human peripheral blood lymphocytes. Environ. Mol. Mutagen. 2008, 49, 328–333. [Google Scholar] [CrossRef]
- Olivero, O.A.; Vazquez, I.L.; Cooch, C.C.; Ming, J.; Keller, E.; Yu, M.; Borojerdi, J.P.; Braun, H.M.; McKee, E.; Poirier, M.C. Long-term AZT exposure alters the metabolic capacity of cultured human lymphoblastoid cells. Toxicol. Sci. 2010, 115, 109–117. [Google Scholar] [CrossRef]
- Grando, A.C.; Guimarães, N.N.; de Souza, A.P.; Lehmann, M.; Cunha, K.S.; Dihl, R.R. Assessment of complex genomic alterations induced by AZT, 3TC, and the combination AZT +3TC. Drug Chem. Toxicol. 2020, 43, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Witt, K.L.; Cunningham, C.K.; Patterson, K.B.; Kissling, G.E.; Dertinger, S.D.; Livingston, E.; Bishop, J.B. Elevated frequencies of micronucleated erythrocytes in infants exposed to zidovudine in utero and postpartum to prevent mother-to-child transmission of HIV. Environ. Mol. Mutagen. 2007, 48, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Singaraju, S.; Bertin, E.T.; Singaraju, M.; Sharma, A. Quantification of micronuclei in exfoliated cells of human immunodeficiency virus/AIDS-infected female patients. J. Oral Maxillofac. Pathol. 2019, 23, 301. [Google Scholar] [CrossRef]
- Gutiérrez-Sevilla, J.E.; Cárdenas-Bedoya, J.; Escoto-Delgadillo, M.; Zúñiga-González, G.M.; Pérez-Ríos, A.M.; Gómez-Meda, B.C.; González-Enríquez, G.V.; Figarola-Centurión, I.; Chavarría-Avila, E.; Torres-Mendoza, B.M. Genomic instability in people living with HIV. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2021, 865, 503336. [Google Scholar] [CrossRef]
- Zhang, X. Anti-retroviral drugs: Current state and development in the next decade. Acta Pharm. Sin. B 2018, 8, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Ellwanger, J.H.; Kulmann-Leal, B.; Kaminski, V.L.; Rodrigues, A.G.; Bragatte, M.A.S.; Chies, J.A.B. Beyond HIV infection: Neglected and varied impacts of CCR5 and CCR5Δ32 on viral diseases. Virus Res. 2020, 286, 198040. [Google Scholar] [CrossRef] [PubMed]
- Jiao, X.; Velasco-Velázquez, M.A.; Wang, M.; Li, Z.; Rui, H.; Peck, A.R.; Korkola, J.E.; Chen, X.; Xu, S.; DuHadaway, J.B.; et al. CCR5 governs DNA damage repair and breast cancer stem cell expansion. Cancer Res. 2018, 78, 1657–1671. [Google Scholar] [CrossRef] [PubMed]
- Jiao, X.; Wang, M.; Zhang, Z.; Li, Z.; Ni, D.; Ashton, A.W.; Tang, H.Y.; Speicher, D.W.; Pestell, R.G. Leronlimab, a humanized monoclonal antibody to CCR5, blocks breast cancer cellular metastasis and enhances cell death induced by DNA damaging chemotherapy. Breast Cancer Res. 2021, 23, 11. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, N.; Borghese, C.; Aldinucci, D. In classical Hodgkin lymphoma the combination of the CCR5 antagonist maraviroc with trabectedin synergizes, enhances DNA damage and decreases three-dimensional tumor-stroma heterospheroid viability. Haematologica 2022, 107, 287–291. [Google Scholar] [CrossRef]
- Wang, W.J.; Mao, L.F.; Lai, H.L.; Wang, Y.W.; Jiang, Z.B.; Li, W.; Huang, J.M.; Xie, Y.J.; Xu, C.; Liu, P.; et al. Dolutegravir derivative inhibits proliferation and induces apoptosis of non-small cell lung cancer cells via calcium signaling pathway. Pharmacol. Res. 2020, 161, 105129. [Google Scholar] [CrossRef]
- Schank, M.; Zhao, J.; Moorman, J.P.; Yao, Z.Q. The impact of HIV- and ART-induced mitochondrial dysfunction in cellular senescence and aging. Cells 2021, 10, 174. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.; Tu-Sekine, B.; Jin, S.; Anokye-Danso, F.; Ahima, R.S.; Brown, T.T.; Kim, S.F. Dolutegravir suppresses thermogenesis via disrupting uncoupling protein 1 expression and mitochondrial function in brown/beige adipocytes in preclinical models. J. Infect. Dis. 2022, 226, 1626–1636. [Google Scholar] [CrossRef] [PubMed]
- Ajaykumar, A.; Caloren, L.C.; Povshedna, T.; Hsieh, A.Y.Y.; Zakaria, A.; Cai, R.; Smith, M.R.; Thompson, C.A.H.; Becquart, P.; Uday, P.; et al. Dolutegravir-containing HIV therapy reversibly alters mitochondrial health and morphology in cultured human fibroblasts and peripheral blood mononuclear cells. AIDS 2023, 37, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Lazalde-Ramos, B.P.; Zamora-Perez, A.L.; Ortega-Guerrero, A.I.; Quintero-Fraire, S.Z.; Palacios-Lara, O.; Quirarte-Báez, S.M.; Galaviz-Hernández, C.; Sosa-Macías, M.; Ortiz-García, Y.M.; Morales-Velazquez, G. Genomic instability decreases in HIV patient by complementary therapy with Rosmarinus officinalis extracts. J. Med. Food 2020, 23, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Borges, R.S.; Ortiz, B.L.S.; Pereira, A.C.M.; Keita, H.; Carvalho, J.C.T. Rosmarinus officinalis essential oil: A review of its phytochemistry, anti-inflammatory activity, and mechanisms of action involved. J. Ethnopharmacol. 2019, 229, 29–45. [Google Scholar] [CrossRef]
- Jaruga, P.; Jaruga, B.; Gackowski, D.; Olczak, A.; Halota, W.; Pawlowska, M.; Olinski, R. Supplementation with antioxidant vitamins prevents oxidative modification of DNA in lymphocytes of HIV-infected patients. Free Radic. Biol. Med. 2002, 32, 414–420. [Google Scholar] [CrossRef]
- Ellwanger, J.H.; Prá, D.; Rieger, A.; Franke, S.I.R. Influência do estado nutricional de selênio sobre a progressão da infecção pelo HIV. Nutrire 2011, 36, 109–122. [Google Scholar]
- Ellwanger, J.H.; Franke, S.I.R.; Bordin, D.L.; Prá, D.; Henriques, J.A.P. Biological functions of selenium and its potential influence on Parkinson’s disease. An. Acad. Bras. Cienc. 2016, 88, 1655–1674. [Google Scholar] [CrossRef]
- Zhang, L.; Zeng, H.; Cheng, W.H. Beneficial and paradoxical roles of selenium at nutritional levels of intake in healthspan and longevity. Free Radic. Biol. Med. 2018, 127, 3–13. [Google Scholar] [CrossRef]
- Gangcuangco, L.M.A.; Kohorn, L.B.; Chow, D.C.; Keating, S.M.; Norris, P.J.; Nagamine, L.S.; Ndhlovu, L.C.; Souza, S.A.; Kallianpur, K.J.; Shikuma, C.M. High 25-hydroxyvitamin D is associated with unexpectedly high plasma inflammatory markers in HIV patients on antiretroviral therapy. Medicine 2016, 95, e5270. [Google Scholar] [CrossRef]
- Bouillon, R.; Manousaki, D.; Rosen, C.; Trajanoska, K.; Rivadeneira, F.; Richards, J.B. The health effects of vitamin D supplementation: Evidence from human studies. Nat. Rev. Endocrinol. 2022, 18, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef]
- Ricker, M.A.; Haas, W.C. Anti-inflammatory diet in clinical practice: A review. Nutr. Clin. Pract. 2017, 32, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Tsiodras, S.; Poulia, K.A.; Yannakoulia, M.; Chimienti, S.N.; Wadhwa, S.; Karchmer, A.W.; Mantzoros, C.S. Adherence to Mediterranean diet is favorably associated with metabolic parameters in HIV-positive patients with the highly active antiretroviral therapy-induced metabolic syndrome and lipodystrophy. Metabolism 2009, 58, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Dokmanović, S.K.; Kolovrat, K.; Laškaj, R.; Jukić, V.; Vrkić, N.; Begovac, J. Effect of extra virgin olive oil on biomarkers of inflammation in HIV-infected patients: A randomized, crossover, controlled clinical trial. Med. Sci. Monit. 2015, 21, 2406–2413. [Google Scholar] [CrossRef]
- Manzano, M.; Talavera-Rodríguez, A.; Moreno, E.; Madrid, N.; Gosalbes, M.J.; Ron, R.; Dronda, F.; Pérez-Molina, J.A.; Lanza, V.F.; Díaz, J.; et al. Relationship of diet to gut microbiota and inflammatory biomarkers in people with HIV. Nutrients 2022, 14, 1221. [Google Scholar] [CrossRef]
- Pastor-Ibáñez, R.; Blanco-Heredia, J.; Etcheverry, F.; Sánchez-Palomino, S.; Díez-Fuertes, F.; Casas, R.; Navarrete-Muñoz, M.Á.; Castro-Barquero, S.; Lucero, C.; Fernández, I.; et al. Adherence to a supplemented mediterranean diet drives changes in the gut microbiota of HIV-1-infected individuals. Nutrients 2021, 13, 1141. [Google Scholar] [CrossRef]
- Sansoni, P.; Vescovini, R.; Fagnoni, F.; Biasini, C.; Zanni, F.; Zanlari, L.; Telera, A.; Lucchini, G.; Passeri, G.; Monti, D.; et al. The immune system in extreme longevity. Exp. Gerontol. 2008, 43, 61–65. [Google Scholar] [CrossRef]
- Deeks, S.G.; Verdin, E.; McCune, J.M. Immunosenescence and HIV. Curr. Opin. Immunol. 2012, 24, 501–506. [Google Scholar] [CrossRef]
- Pawelec, G.; Akbar, A.; Caruso, C.; Solana, R.; Grubeck-Loebenstein, B.; Wikby, A. Human immunosenescence: Is it infectious? Immunol. Rev. 2005, 205, 257–268. [Google Scholar] [CrossRef]
- Agarwal, S.; Busse, P.J. Innate and adaptive immunosenescence. Ann. Allergy Asthma Immunol. 2010, 104, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Wang, W.; Su, D.M. Contributions of age-related thymic involution to immunosenescence and inflammaging. Immun. Ageing 2020, 17, 2. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, C.; Joo, V.; Jacquier, P.; Noto, A.; Banga, R.; Perreau, M.; Pantaleo, G. T-cell exhaustion in HIV infection. Immunol. Rev. 2019, 292, 149–163. [Google Scholar] [CrossRef]
- Day, C.L.; Kaufmann, D.E.; Kiepiela, P.; Brown, J.A.; Moodley, E.S.; Reddy, S.; Mackey, E.W.; Miller, J.D.; Leslie, A.J.; DePierres, C.; et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 2006, 443, 350–354. [Google Scholar] [CrossRef]
- Chew, G.M.; Fujita, T.; Webb, G.M.; Burwitz, B.J.; Wu, H.L.; Reed, J.S.; Hammond, K.B.; Clayton, K.L.; Ishii, N.; Abdel-Mohsen, M.; et al. TIGIT marks exhausted t cells, correlates with disease progression, and serves as a target for immune restoration in HIV and SIV infection. PLoS Pathog. 2016, 12, e1005349. [Google Scholar] [CrossRef]
- Fromentin, R.; Bakeman, W.; Lawani, M.B.; Khoury, G.; Hartogensis, W.; DaFonseca, S.; Killian, M.; Epling, L.; Hoh, R.; Sinclair, E.; et al. CD4+ T cells expressing PD-1, TIGIT and LAG-3 contribute to HIV persistence during ART. PLoS Pathog. 2016, 12, e1005761. [Google Scholar] [CrossRef] [PubMed]
- Martínez de Toda, I.; Maté, I.; Vida, C.; Cruces, J.; De la Fuente, M. Immune function parameters as markers of biological age and predictors of longevity. Aging 2016, 8, 3110–3119. [Google Scholar] [CrossRef]
- Horvath, S.; Levine, A.J. HIV-1 infection accelerates age according to the epigenetic clock. J. Infect. Dis. 2015, 212, 1563–1573. [Google Scholar] [CrossRef]
- Sehl, M.E.; Breen, E.C.; Shih, R.; Chen, L.; Wang, R.; Horvath, S.; Bream, J.H.; Duggal, P.; Martinson, J.; Wolinsky, S.M.; et al. Increased rate of epigenetic aging in men living with HIV prior to treatment. Front. Genet. 2022, 12, 796547. [Google Scholar] [CrossRef]
- Appay, V.; Sauce, D. Immune activation and inflammation in HIV-1 infection: Causes and consequences. J. Pathol. 2008, 214, 231–241. [Google Scholar] [CrossRef]
- Dock, J.N.; Effros, R.B. Role of CD8 T cell replicative senescence in human aging and in HIV-mediated immunosenescence. Aging Dis. 2011, 2, 382–397. [Google Scholar]
- Paul, L. Diet, nutrition and telomere length. J. Nutr. Biochem. 2011, 22, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Malan-Müller, S.; Hemmings, S.M.; Spies, G.; Kidd, M.; Fennema-Notestine, C.; Seedat, S. Shorter telomere length-A potential susceptibility factor for HIV-associated neurocognitive impairments in South African women. PLoS ONE 2013, 8, e58351. [Google Scholar] [CrossRef]
- Auld, E.; Lin, J.; Chang, E.; Byanyima, P.; Ayakaka, I.; Musisi, E.; Worodria, W.; Davis, J.L.; Segal, M.; Blackburn, E.; et al. HIV infection is associated with shortened telomere length in Ugandans with suspected tuberculosis. PLoS ONE 2016, 11, e0163153. [Google Scholar] [CrossRef] [PubMed]
- Minami, R.; Takahama, S.; Yamamoto, M. Correlates of telomere length shortening in peripheral leukocytes of HIV-infected individuals and association with leukoaraiosis. PLoS ONE 2019, 14, e0218996. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, S.; Cȏté, H.C.F.; Fitch, K.V.; Torriani, M.; Feldpausch, M.; Srinivasa, S. Relationship of telomere length to fat redistribution in HIV. Open Forum Infect. Dis. 2020, 7, ofaa523. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.R.; Iudicello, J.E.; Lin, J.; Ellis, R.J.; Morgan, E.; Okwuegbuna, O.; Cookson, D.; Karris, M.; Saloner, R.; Heaton, R.; et al. Telomere length is associated with HIV infection, methamphetamine use, inflammation, and comorbid disease risk. Drug Alcohol Depend. 2021, 221, 108639. [Google Scholar] [CrossRef]
- Chauvin, M.; Sauce, D. Mechanisms of immune aging in HIV. Clin. Sci. 2022, 136, 61–80. [Google Scholar] [CrossRef]
- Martínez de Toda, I.; Ceprián, N.; Díaz-Del Cerro, E.; De la Fuente, M. The role of immune cells in oxi-inflamm-aging. Cells 2021, 10, 2974. [Google Scholar] [CrossRef]
- Fulop, T.; Larbi, A.; Dupuis, G.; Le Page, A.; Frost, E.H.; Cohen, A.A.; Witkowski, J.M.; Franceschi, C. Immunosenescence and inflamm-aging as two sides of the same coin: Friends or foes? Front. Immunol. 2018, 8, 1960. [Google Scholar] [CrossRef]
- Ginaldi, L.; De Martinis, M.; D’Ostilio, A.; Marini, L.; Loreto, M.F.; Corsi, M.P.; Quaglino, D. Cell proliferation and apoptosis in the immune system in the elderly. Immunol. Res. 2000, 21, 31–38. [Google Scholar] [CrossRef] [PubMed]
- George, A.; Dey, R.; Bhuria, V.; Banerjee, S.; Ethirajan, S.; Siluvaimuthu, A.; Saraswathy, R. Nuclear anomalies, chromosomal aberrations and proliferation rates in cultured lymphocytes of head and neck cancer patients. Asian Pac. J. Cancer Prev. 2014, 15, 1119–1123. [Google Scholar] [CrossRef] [PubMed][Green Version]
- González-Sánchez, M.; García-Martínez, V.; Bravo, S.; Kobayashi, H.; Martínez de Toda, I.; González-Bermúdez, B.; Plaza, G.R.; De la Fuente, M. Mitochondrial DNA insertions into nuclear DNA affecting chromosome segregation: Insights for a novel mechanism of immunosenescence in mice. Mech. Ageing Dev. 2022, 207, 111722. [Google Scholar] [CrossRef] [PubMed]
- Bolognesi, C.; Lando, C.; Forni, A.; Landini, E.; Scarpato, R.; Migliore, L.; Bonassi, S. Chromosomal damage and ageing: Effect on micronuclei frequency in peripheral blood lymphocytes. Age Ageing 1999, 28, 393–397. [Google Scholar] [CrossRef]
- Richter, C. Do mitochondrial DNA fragments promote cancer and aging? FEBS Lett. 1988, 241, 1–5. [Google Scholar] [CrossRef]
- Sikora, E. Activation-induced and damage-induced cell death in aging human T cells. Mech. Ageing Dev. 2015, 151, 85–92. [Google Scholar] [CrossRef]
- Fiala, M.; Murphy, T.; MacDougall, J.; Yang, W.; Luque, A.; Iruela-Arispe, L.; Cashman, J.; Buga, G.; Byrns, R.E.; Barbaro, G.; et al. HAART drugs induce mitochondrial damage and intercellular gaps and gp120 causes apoptosis. Cardiovasc. Toxicol. 2004, 4, 327–337. [Google Scholar] [CrossRef]
- Foster, C.; Lyall, H. HIV and mitochondrial toxicity in children. J. Antimicrob. Chemother. 2008, 61, 8–12. [Google Scholar] [CrossRef]
- Garrabou, G.; López, S.; Morén, C.; Martínez, E.; Fontdevila, J.; Cardellach, F.; Gatell, J.M.; Miró, O. Mitochondrial damage in adipose tissue of untreated HIV-infected patients. AIDS 2011, 25, 165–170. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, M.; Li, H.; Zhang, H.; Shi, Y.; Wei, F.; Liu, D.; Liu, K.; Chen, D. Accumulation of nuclear and mitochondrial DNA damage in the frontal cortex cells of patients with HIV-associated neurocognitive disorders. Brain. Res. 2012, 1458, 1–11. [Google Scholar] [CrossRef]
- Kallianpur, K.J.; Gerschenson, M.; Mitchell, B.I.; LiButti, D.E.; Umaki, T.M.; Ndhlovu, L.C.; Nakamoto, B.K.; Chow, D.C.; Shikuma, C.M. Oxidative mitochondrial DNA damage in peripheral blood mononuclear cells is associated with reduced volumes of hippocampus and subcortical gray matter in chronically HIV-infected patients. Mitochondrion 2016, 28, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Darbinian, N.; Darbinyan, A.; Merabova, N.; Selzer, M.E.; Amini, S. HIV-1 and HIV-1-Tat induce mitochondrial DNA damage in human neurons. J. HIV AIDS 2020, 6, 176. [Google Scholar] [CrossRef] [PubMed]
- Roca-Bayerri, C.; Robertson, F.; Pyle, A.; Hudson, G.; Payne, B.A.I. Mitochondrial DNA damage and brain aging in human immunodeficiency virus. Clin. Infect. Dis. 2021, 73, e466–e473. [Google Scholar] [CrossRef] [PubMed]
- Siegel, K.; Lekas, H.M. AIDS as a chronic illness: Psychosocial implications. AIDS 2002, 4, S69–S76. [Google Scholar] [CrossRef]
- Deeks, S.G. HIV infection, inflammation, immunosenescence, and aging. Annu. Rev. Med. 2011, 62, 141–155. [Google Scholar] [CrossRef]
- Nasi, M.; Pinti, M.; De Biasi, S.; Gibellini, L.; Ferraro, D.; Mussini, C.; Cossarizza, A. Aging with HIV infection: A journey to the center of inflammAIDS, immunosenescence and neuroHIV. Immunol. Lett. 2014, 162, 329–333. [Google Scholar] [CrossRef]
- Langie, S.A.S.; Koppen, G.; Desaulniers, D.; Al-Mulla, F.; Al-Temaimi, R.; Amedei, A.; Azqueta, A.; Bisson, W.H.; Brown, D.G.; Brunborg, G.; et al. Causes of genome instability: The effect of low dose chemical exposures in modern society. Carcinogenesis 2015, 36, S61–S88. [Google Scholar] [CrossRef]
- Kirsch-Volders, M.; Bolognesi, C.; Ceppi, M.; Bruzzone, M.; Fenech, M. Micronuclei, inflammation and auto-immune disease. Mutat. Res. Rev. Mutat. Res. 2020, 786, 108335. [Google Scholar] [CrossRef]
- Ye, C.J.; Sharpe, Z.; Heng, H.H. Origins and consequences of chromosomal instability: From cellular adaptation to genome chaos-mediated system survival. Genes 2020, 11, 1162. [Google Scholar] [CrossRef]
- Canva. Available online: https://www.canva.com/ (accessed on 26 December 2022).
- Servier Medical Art. Available online: https://smart.servier.com/) (accessed on 25 November 2022).
| Country | Cell Type Investigated | Main Findings | References |
|---|---|---|---|
| Brazil | Exfoliated oral cells | Increased mean of single MN in cells of controls compared to those of HIV-infected individuals; non-significant increase in the occurrence of multiple micronuclei in cells of HIV group compared to controls | Lima et al. [32] |
| South Africa | Blood cells | MN frequencies were significantly higher in irradiated lymphocytes from HIV-infected individuals compared to controls | Baeyens et al. [33]; Herd et al. [34]; Herd et al. [35] |
| Italy | Blood cells | Increased MN frequency in the HIV-infected group (HCV co-infection and HIV-RNA load being risk factors for increased MN frequency); HIV-infected individuals with undetectable viremia showed reduced MN frequency compared to those with uncontrolled viremia | Zizza et al. [36] |
| USA | Reticulocytes | A 10-fold increase in micronucleated reticulocyte frequencies was observed in mothers and children pre-natal with zidovudine-based ART compared to controls | Witt et al. [42] |
| India | Exfoliated oral cells | HIV-infected individuals showed significantly increased in MN rate (~two-fold) compared to controls | Shah et al. [43] |
| Mexico | Exfoliated oral cells | Higher frequencies of binucleated cells and nuclear buds in both HIV-infected ART-naive individuals and HIV-infected individuals undergoing ART compared to HIV-negative controls; karyorrhexis, binucleated cells, and nuclear buds were found to be increased in a subgroup of HIV-infected individuals receiving RTIs as ART compared to controls | Gutiérrez-Sevilla et al. [44] |
| Mexico | Exfoliated oral cells | Increased frequency of MN and other nuclear abnormalities in HIV-infected individuals undergoing ART compared to controls | Lazarde-Ramos et al. [54] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ellwanger, J.H.; Kulmann-Leal, B.; Ziliotto, M.; Chies, J.A.B. HIV Infection, Chromosome Instability, and Micronucleus Formation. Viruses 2023, 15, 155. https://doi.org/10.3390/v15010155
Ellwanger JH, Kulmann-Leal B, Ziliotto M, Chies JAB. HIV Infection, Chromosome Instability, and Micronucleus Formation. Viruses. 2023; 15(1):155. https://doi.org/10.3390/v15010155
Chicago/Turabian StyleEllwanger, Joel Henrique, Bruna Kulmann-Leal, Marina Ziliotto, and José Artur Bogo Chies. 2023. "HIV Infection, Chromosome Instability, and Micronucleus Formation" Viruses 15, no. 1: 155. https://doi.org/10.3390/v15010155
APA StyleEllwanger, J. H., Kulmann-Leal, B., Ziliotto, M., & Chies, J. A. B. (2023). HIV Infection, Chromosome Instability, and Micronucleus Formation. Viruses, 15(1), 155. https://doi.org/10.3390/v15010155








