Development of a Genetically Engineered Bivalent Vaccine against Porcine Epidemic Diarrhea Virus and Porcine Rotavirus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells, Viruses, and Antibodies
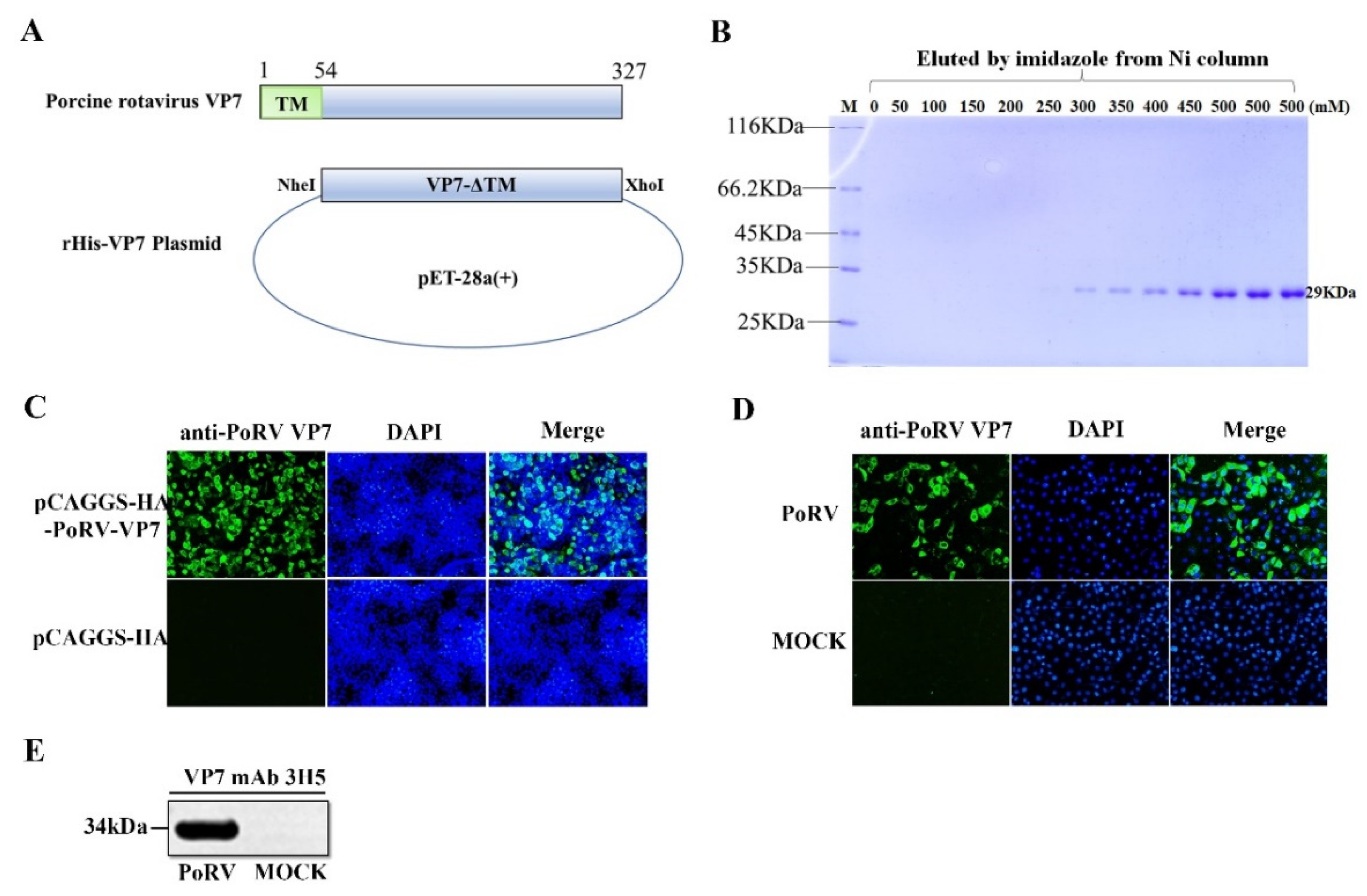
2.2. Preparation of the Monoclonal Antibody against VP7
2.3. Indirect Immunofluorescence Assay (IFA)
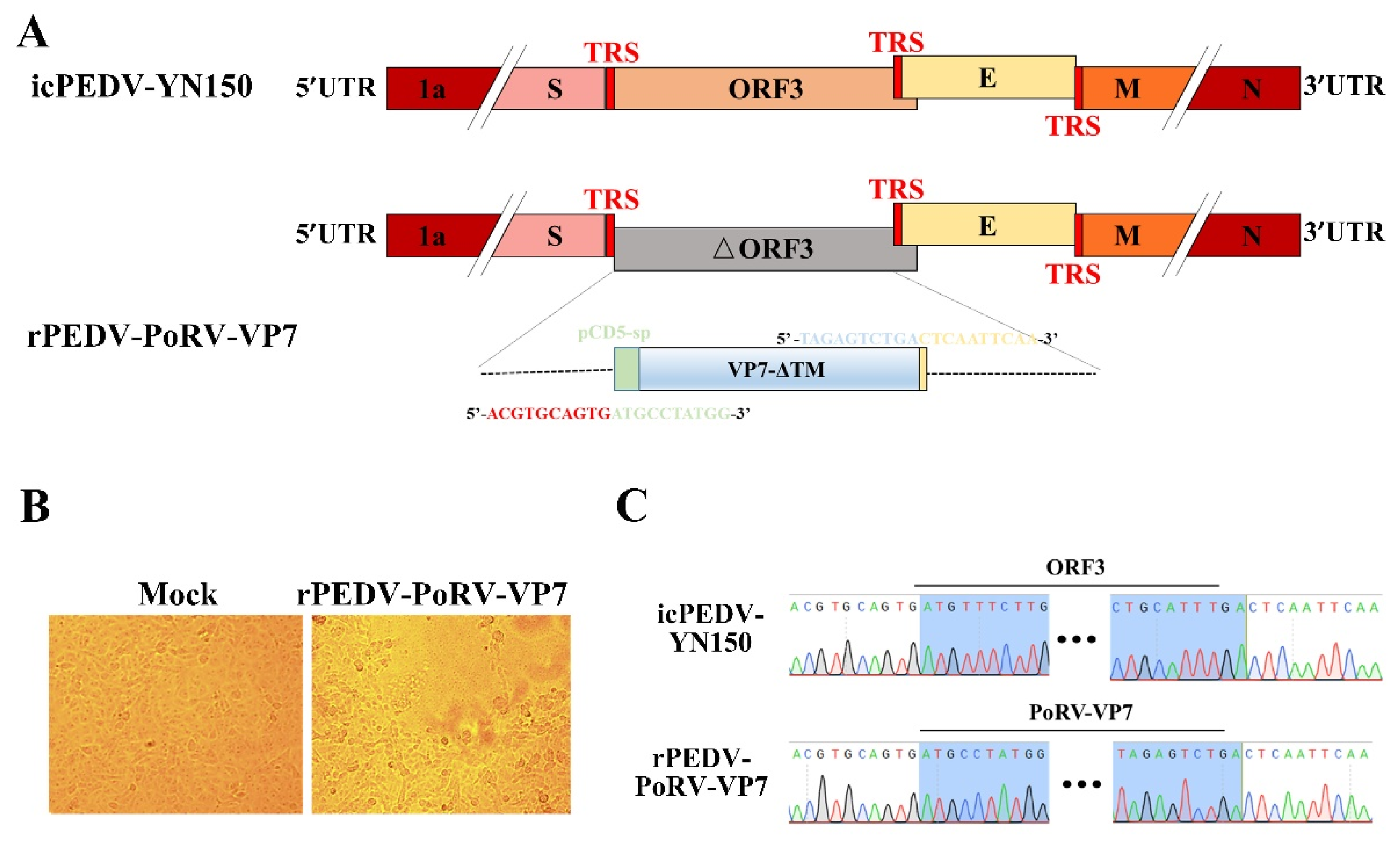
2.4. The Construction of a Recombinant PEDV Expressing PoRV VP7 Gene
2.5. Virus Genome Sequencing
2.6. Immunization
2.7. Sample Collection
2.8. Expression of PEDV-S1
2.9. Enzyme-Linked Immunosorbent Assay (ELISA) Analysis
2.10. Neutralizing Antibody Titration
2.11. Statistical Analysis
3. Results
3.1. The Preparation of Monoclonal Antibody (mAb) against PoRV VP7
3.2. Rescue of rPEDV-PoRV-VP7
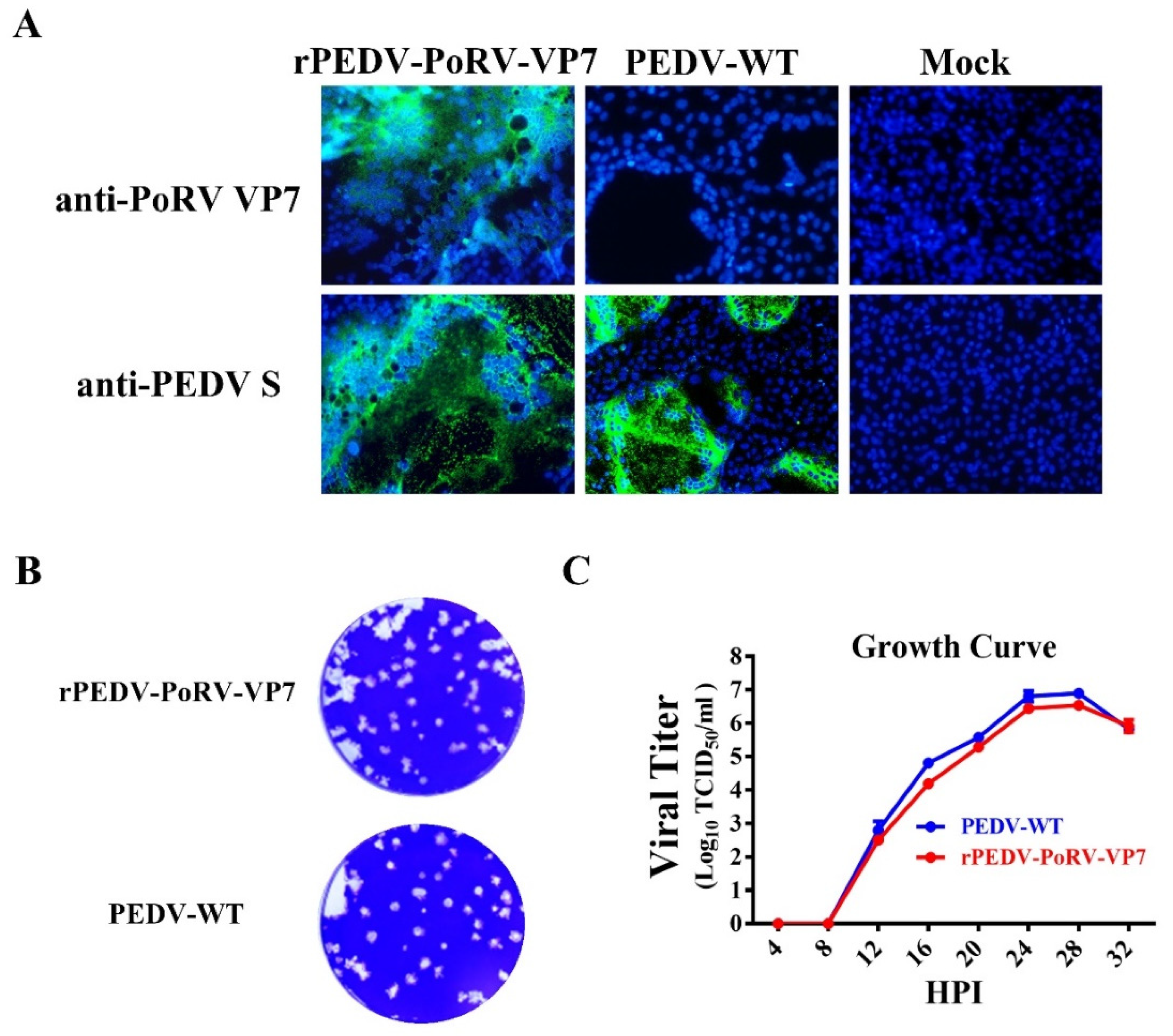
3.3. Characterization of rPEDV-PoRV-VP7
3.4. Clinical Manifestations of the rYN150-PoRV-VP7-Inoculated Piglets
3.5. IgA Levels in rPEDV-PoRV-VP7-Vaccinated Piglets
3.6. IgG Levels and Neutralizing Antibodies Induced by rPEDV-PoRV-VP7
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chasey, D.; Cartwright, S. Virus-like particles associated with porcine epidemic diarrhoea. Res. Veter. Sci. 1978, 25, 255–256. [Google Scholar] [CrossRef]
- Li, W.T.; Li, H.; Liu, Y.B.; Pan, Y.F.; Deng, F.; Song, Y.H.; Tang, X.B.; He, Q.G. New Variants of Porcine Epidemic Diarrhea Virus, China, 2011. Emerg. Infect. Dis. 2012, 18, 1350–1353. [Google Scholar] [CrossRef]
- Bi, J.; Zeng, S.; Xiao, S.; Chen, H.; Fang, L. Complete Genome Sequence of Porcine Epidemic Diarrhea Virus Strain AJ1102 Isolated from a Suckling Piglet with Acute Diarrhea in China. J. Virol. 2012, 86, 10910–10911. [Google Scholar] [CrossRef] [Green Version]
- Cima, G. Fighting a deadly pig disease. Industry, veterinarians trying to contain PED virus, new to the US. J. Am. Veter. Med. Assoc. 2013, 243, 469–470. [Google Scholar]
- Lee, S.; Lee, C. Outbreak-Related Porcine Epidemic Diarrhea Virus Strains Similar to US Strains, South Korea, 2013. Emerg. Infect. Dis. 2014, 20, 1223–1226. [Google Scholar] [CrossRef]
- Crawford, K.; Lager, K.M.; Kulshreshtha, V.; Miller, L.; Faaberg, K.S. Status of vaccines for porcine epidemic diarrhea virus in the United States and Canada. Virus Res. 2016, 226, 108–116. [Google Scholar] [CrossRef] [Green Version]
- Stadler, J.; Zoels, S.; Fux, R.; Hanke, D.; Pohlmann, A.; Blome, S.; Weissenböck, H.; Weissenbacher-Lang, C.; Ritzmann, M.; Ladinig, A. Emergence of porcine epidemic diarrhea virus in southern Germany. BMC Veter. Res. 2015, 11, 142. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Fang, L.; Xiao, S. Porcine epidemic diarrhea in China. Virus Res. 2016, 226, 7–13. [Google Scholar] [CrossRef]
- Jung, K.; Saif, L.J.; Wang, Q. Porcine epidemic diarrhea virus (PEDV): An update on etiology, transmission, pathogenesis, and prevention and control. Virus Res. 2020, 286, 198045. [Google Scholar] [CrossRef]
- Lee, C. Porcine epidemic diarrhea virus: An emerging and re-emerging epizootic swine virus. Virol. J. 2015, 12, 193. [Google Scholar] [CrossRef] [Green Version]
- Have, P.; Moving, V.; Svansson, V.; Uttenthal, A.; Bloch, B. Coronavirus infection in mink (Mustela vision). Serological evidence of infection with a coronavirus related to transmissible gastroenteritis virus and porcine epidemic diarrhea virus. Veter. Microbiol. 1992, 31, 1–10. [Google Scholar] [CrossRef]
- Kocherhans, R.; Bridgen, A.; Ackermann, M.; Tobler, K. Completion of the Porcine Epidemic Diarrhoea Coronavirus (PEDV) Genome Sequence. Virus Genes 2001, 23, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Grunewald, M.; Perlman, S. Coronaviruses: An Updated Overview of Their Replication and Pathogenesis. Methods Mol. Biol. 2020, 2203, 1–29. [Google Scholar] [CrossRef]
- Kaewborisuth, C.; He, Q.; Jongkaewwattana, A. The Accessory Protein ORF3 Contributes to Porcine Epidemic Diarrhea Virus Replication by Direct Binding to the Spike Protein. Viruses 2018, 10, 399. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Zhang, M.; Zheng, H.; Zhou, P.; Liu, Z.; Jongkaewwattana, A.; Luo, R.; He, Q. Construction of a Recombinant Porcine Epidemic Diarrhea Virus Encoding Nanoluciferase for High-Throughput Screening of Natural Antiviral Products. Viruses 2021, 13, 1866. [Google Scholar] [CrossRef]
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Rotavirus infection. Nat. Rev. Dis. Primers. 2017, 3, 17083. [Google Scholar] [CrossRef] [Green Version]
- Vlasova, A.N.; Amimo, J.O.; Saif, L.J. Porcine Rotaviruses: Epidemiology, Immune Responses and Control Strategies. Viruses 2017, 9, 48. [Google Scholar] [CrossRef]
- Wang, Q.; Vlasova, A.N.; Kenney, S.P.; Saif, L.J. Emerging and re-emerging coronaviruses in pigs. Curr. Opin. Virol. 2019, 34, 39–49. [Google Scholar] [CrossRef]
- Zhang, F.; Luo, S.; Gu, J.; Li, Z.; Li, K.; Yuan, W.; Ye, Y.; Li, H.; Ding, Z.; Song, D.; et al. Prevalence and phylogenetic analysis of porcine diarrhea associated viruses in southern China from 2012 to 2018. BMC Veter. Res. 2019, 15, 470. [Google Scholar] [CrossRef] [Green Version]
- Duan, Q.; Li, X.; Zhao, S.; Qin, Y.; Lu, B.; Li, B.; Su, Q.; Zhao, W.; He, Y.; Liang, J.; et al. A Epidemiological Investigation on Diarrhea Viral Disease of Piglets in Some Pig Farms in Guangxi During 2016 to 2019. China Anim. Husb. Vet. Med. 2020, 47, 564–574. [Google Scholar]
- Li, C.; Lu, H.; Geng, C.; Yang, K.; Liu, W.; Liu, Z.; Yuan, F.; Gao, T.; Wang, S.; Wen, P.; et al. Epidemic and Evolutionary Characteristics of Swine Enteric Viruses in South-Central China from 2018 to 2021. Viruses 2022, 14, 1420. [Google Scholar] [CrossRef]
- Zhu, Z.; Yan, L.; Ju, Y.; Tang, Z.; Yang, J.; Zhu, Q. The epidemiological investigation on the viral diarrhea in pigs in the Northeast region of China during 2015–2016. Zhongguo Yufang Shouyi Xuebao/Chin. J. Prev. Vet. Med. 2017, 39, 356–360. [Google Scholar]
- Bishop, R.; Davidson, G. Virus Particles in Epithelial Cells of Duodenal Mucosa from Children with Acute Non-Bacterial Gastroenteritis. Lancet 1973, 2, 1281–1283. [Google Scholar] [CrossRef]
- Rodger, S.M.; Craven, J.A.; Williams, I. Letter: Demonstration of reovirus-like particles in intestinal contents of piglets with diarrhoea. Aust. Vet. J. 1975, 51, 536. [Google Scholar] [CrossRef]
- Kasari, T.R.; Naylor, J.M. Further studies on the clinical features and clinicopathological findings of a syndrome of metabolic acidosis with minimal dehydration in neonatal calves. Can. J. Veter. Res. 1986, 50, 502–508. [Google Scholar]
- He, B.; Yang, F.; Yang, W.; Zhang, Y.; Feng, Y.; Zhou, J.; Xie, J.; Feng, Y.; Bao, X.; Guo, H.; et al. Characterization of a Novel G3P[3] Rotavirus Isolated from a Lesser Horseshoe Bat: A Distant Relative of Feline/Canine Rotaviruses. J. Virol. 2013, 87, 12357–12366. [Google Scholar] [CrossRef] [Green Version]
- Greenberg, H.B.; Estes, M.K. Rotaviruses: From Pathogenesis to Vaccination. Gastroenterology 2009, 136, 1939–1951. [Google Scholar] [CrossRef] [Green Version]
- Banyai, K.; Kemenesi, G.; Budinski, I.; Foldes, F.; Zana, B.; Marton, S.; Varga-Kugler, R.; Oldal, M.; Kurucz, K.; Jakab, F. Candidate new rotavirus species in Schreiber’s bats, Serbia. Infect. Genet. Evol. 2017, 48, 19–26. [Google Scholar] [CrossRef]
- Matthijnssens, J.; Otto, P.H.; Ciarlet, M.; Desselberger, U.; Van Ranst, M.; Johne, R. VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Arch. Virol. 2012, 157, 1177–1182. [Google Scholar] [CrossRef]
- Matthijnssens, J.; Ciarlet, M.; Rahman, M.; Attoui, H.; Bányai, K.; Estes, M.K.; Gentsch, J.R.; Iturriza-Gómara, M.; Kirkwood, C.D.; Martella, V.; et al. Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Arch. Virol. 2008, 153, 1621–1629. [Google Scholar] [CrossRef] [Green Version]
- Okitsu, S.; Khamrin, P.; Thongprachum, A.; Maneekarn, N.; Mizuguchi, M.; Ushijima, H. Predominance of Porcine P[23] Genotype Rotaviruses in Piglets with Diarrhea in Northern Thailand. J. Clin. Microbiol. 2011, 49, 442–445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.-H.; Kobayashi, N.; Nagashima, S.; Zhou, X.; Ghosh, S.; Peng, J.-S.; Hu, Q.; Zhou, D.-J.; Yang, Z.-Q. Full genomic analysis of a porcine-bovine reassortant G4P[6] rotavirus strain R479 isolated from an infant in China. J. Med. Virol. 2010, 82, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zhu, Y.; Wu, M.; Ku, X.; Ye, S.; Li, Z.; Guo, X.; He, Q. Comparative Genomic Analysis of Classical and Variant Virulent Parental/Attenuated Strains of Porcine Epidemic Diarrhea Virus. Viruses 2015, 7, 5525–5538. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chen, F.; Ye, S.; Guo, X.; Memon, A.M.; Wu, M.; He, Q. Comparative Proteome Analysis of Porcine Jejunum Tissues in Response to a Virulent Strain of Porcine Epidemic Diarrhea Virus and Its Attenuated Strain. Viruses 2016, 8, 323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Won, H.; Lim, J.; Noh, Y.; Yoon, I.; Yoo, H. Efficacy of Porcine Epidemic Diarrhea Vaccines: A Systematic Review and Meta-Analysis. Vaccines 2020, 8, 642. [Google Scholar] [CrossRef]
- Feng, Y.; Xu, Z.; Zhu, L. Prevalence and phylogenetic analysis of porcine deltacoronavirus in Sichuan province, China. Arch Virol. 2020, 165, 2883–2889. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Li, B.; Tao, J.; Cheng, J.; Liu, H. The Complex Co-infections of Multiple Porcine Diarrhea Viruses in Local Area Based on the Luminex xTAG Multiplex Detection Method. Front. Veter. Sci. 2021, 8, 602866. [Google Scholar] [CrossRef]
- Yin, Y.; Zhu, L.; Liu, P.; Zhao, J.; Fan, Y.; Sun, X.; Xu, Z. Evaluation on the efficacy and immunogenicity of recombinant DNA plasmids expressing S gene from porcine epidemic diarrhea virus and VP7 gene from porcine rotavirus. Braz. J. Microbiol. 2019, 50, 279–286. [Google Scholar] [CrossRef] [Green Version]
- Guo, T.; Gao, C.; Hao, J.; Lu, X.; Xie, K.; Wang, X.; Li, J.; Zhou, H.; Cui, W.; Shan, Z.; et al. Strategy of Developing Oral Vaccine Candidates Against Co-infection of Porcine Diarrhea Viruses Based on a Lactobacillus Delivery System. Front. Microbiol. 2022, 13, 872550. [Google Scholar] [CrossRef]
- Ribes, J.M.; Ortego, J.; Ceriani, J.; Montava, R.; Enjuanes, L.; Buesa, J. Transmissible gastroenteritis virus (TGEV)-based vectors with engineered murine tropism express the rotavirus VP7 protein and immunize mice against rotavirus. Virology 2011, 410, 107–118. [Google Scholar] [CrossRef] [Green Version]
- Clements-Mann, M.L.; Makhene, M.K.; Mrukowicz, J.; Wright, P.F.; Hoshino, Y.; Midthun, K.; Sperber, E.; Karron, R.; Kapikian, A.Z. Safety and immunogenicity of live attenuated human-bovine (UK) reassortant rotavirus vaccines with VP7-specificity for serotypes 1, 2, 3 or 4 in adults, children and infants. Vaccine 1999, 17, 2715–2725. [Google Scholar] [CrossRef]
- Li, C.H.; Li, Z.; Zou, Y.; Wicht, O.; van Kuppeveld, F.J.M.; Rottier, P.J.M.; Bosch, B.J. Manipulation of the Porcine Epidemic Diarrhea Virus Genome Using Targeted RNA Recombination. PLoS ONE 2013, 8, e69997. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovjazin, R.; Carmon, L. The use of signal peptide domains as vaccine candidates. Hum Vacc Immunother 2014, 10, 2733–2740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yolitz, J.; Schwing, C.; Chang, J.; Van Ryk, D.; Nawaz, F.; Wei, D.; Cicala, C.; Arthos, J.; Fauci, A.S. Signal peptide of HIV envelope protein impacts glycosylation and antigenicity of gp120. Proc. Natl. Acad. Sci. USA 2018, 115, 2443–2448. [Google Scholar] [CrossRef] [Green Version]
- Fonseca, J.A.; McCaffery, J.N.; Caceres, J.; Kashentseva, E.; Singh, B.; Dmitriev, I.P.; Curiel, D.T.; Moreno, A. Inclusion of the murine IgG kappa signal peptide increases the cellular immunogenicity of a simian adenoviral vectored Plasmodium vivax multistage vaccine. Vaccine 2018, 36, 2799–2808. [Google Scholar] [CrossRef]
- Langel, S.N.; Wang, Q.; Vlasova, A.N.; Saif, L.J. Host Factors Affecting Generation of Immunity Against Porcine Epidemic Diarrhea Virus in Pregnant and Lactating Swine and Passive Protection of Neonates. Pathogens 2020, 9, 130. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.; Lei, M.; Li, Z.; Li, H.; Liu, Z.; He, Q.; Luo, R. Development of a Genetically Engineered Bivalent Vaccine against Porcine Epidemic Diarrhea Virus and Porcine Rotavirus. Viruses 2022, 14, 1746. https://doi.org/10.3390/v14081746
Li W, Lei M, Li Z, Li H, Liu Z, He Q, Luo R. Development of a Genetically Engineered Bivalent Vaccine against Porcine Epidemic Diarrhea Virus and Porcine Rotavirus. Viruses. 2022; 14(8):1746. https://doi.org/10.3390/v14081746
Chicago/Turabian StyleLi, Wan, Mingkai Lei, Zhuofei Li, Huimin Li, Zheng Liu, Qigai He, and Rui Luo. 2022. "Development of a Genetically Engineered Bivalent Vaccine against Porcine Epidemic Diarrhea Virus and Porcine Rotavirus" Viruses 14, no. 8: 1746. https://doi.org/10.3390/v14081746
APA StyleLi, W., Lei, M., Li, Z., Li, H., Liu, Z., He, Q., & Luo, R. (2022). Development of a Genetically Engineered Bivalent Vaccine against Porcine Epidemic Diarrhea Virus and Porcine Rotavirus. Viruses, 14(8), 1746. https://doi.org/10.3390/v14081746





