Largemouth Bass Virus Infection Induced Non-Apoptotic Cell Death in MsF Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Primary Cell Culture and Subculture
2.2. Viral Replication Dynamics
2.3. RNA Extraction and Quantitative Polymerase Chain Reaction (qPCR)
2.4. Western Blotting
2.5. Immunofluorescence Assay (IFA)
2.6. Virus Titer Assay
2.7. TUNEL Assay
2.8. Flow Cytometric Analysis
2.9. Detection of Caspase Activities
2.10. Evaluation of the Mitochondrial Membrane Potential (ΔΨm)
2.11. Reactive Oxygen Species (ROS) Activity
2.12. Statistical Analysis
3. Results
3.1. Primary Culture and Subculture
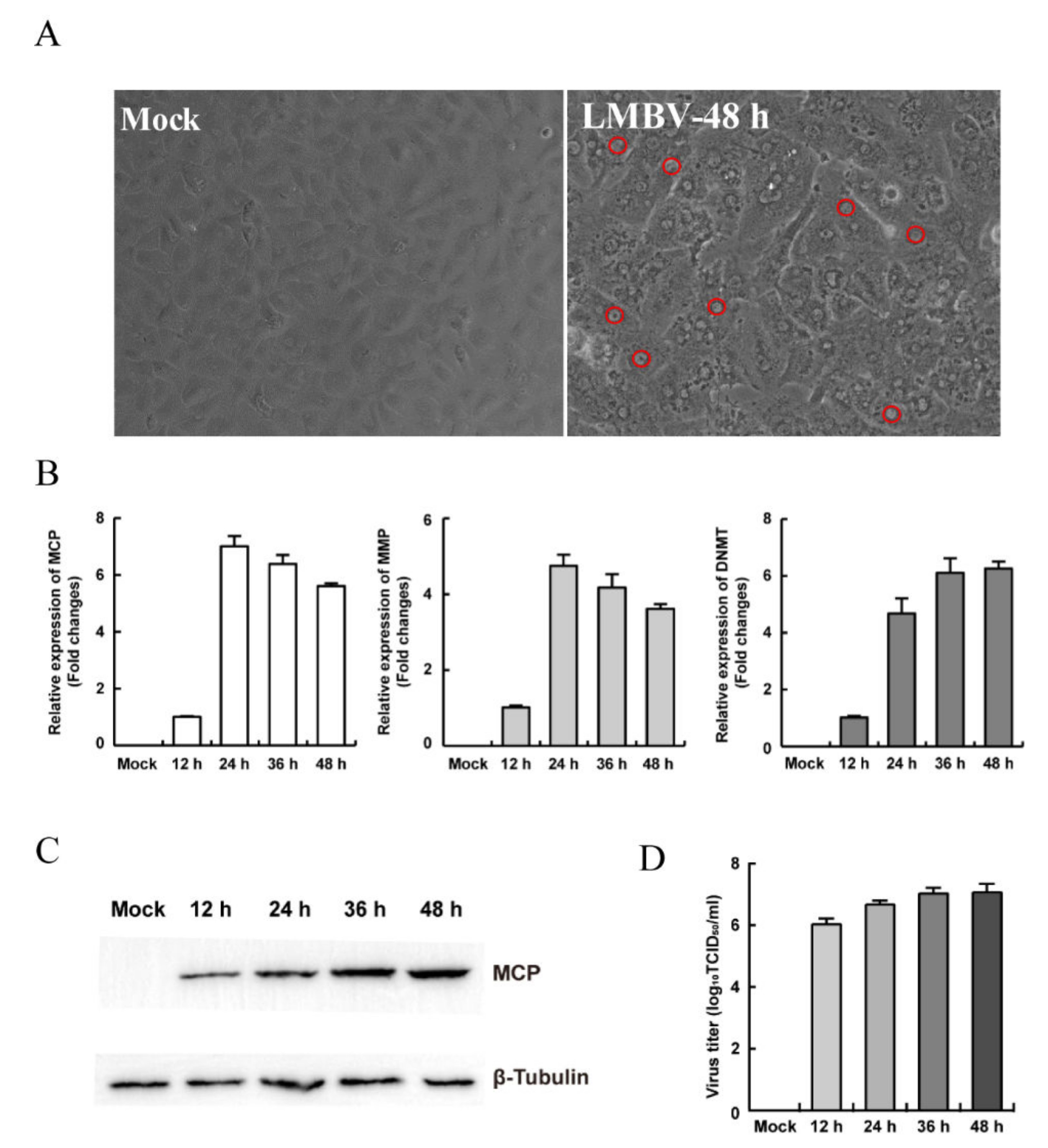
3.2. Establishment of the In Vitro LMBV Infection Model
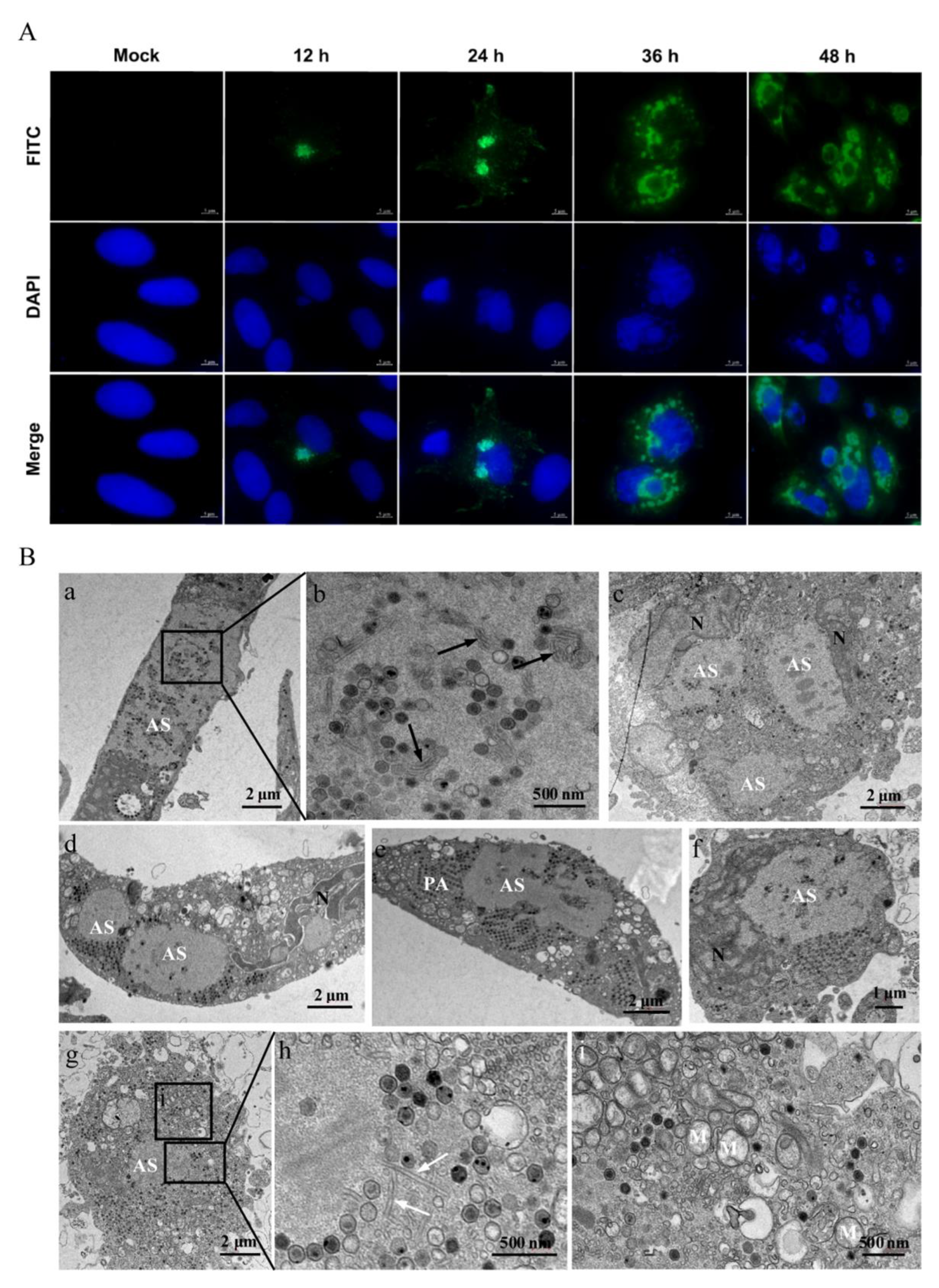
3.3. The Characteristics of Replication and Assembly of LMBV in MsF Cells
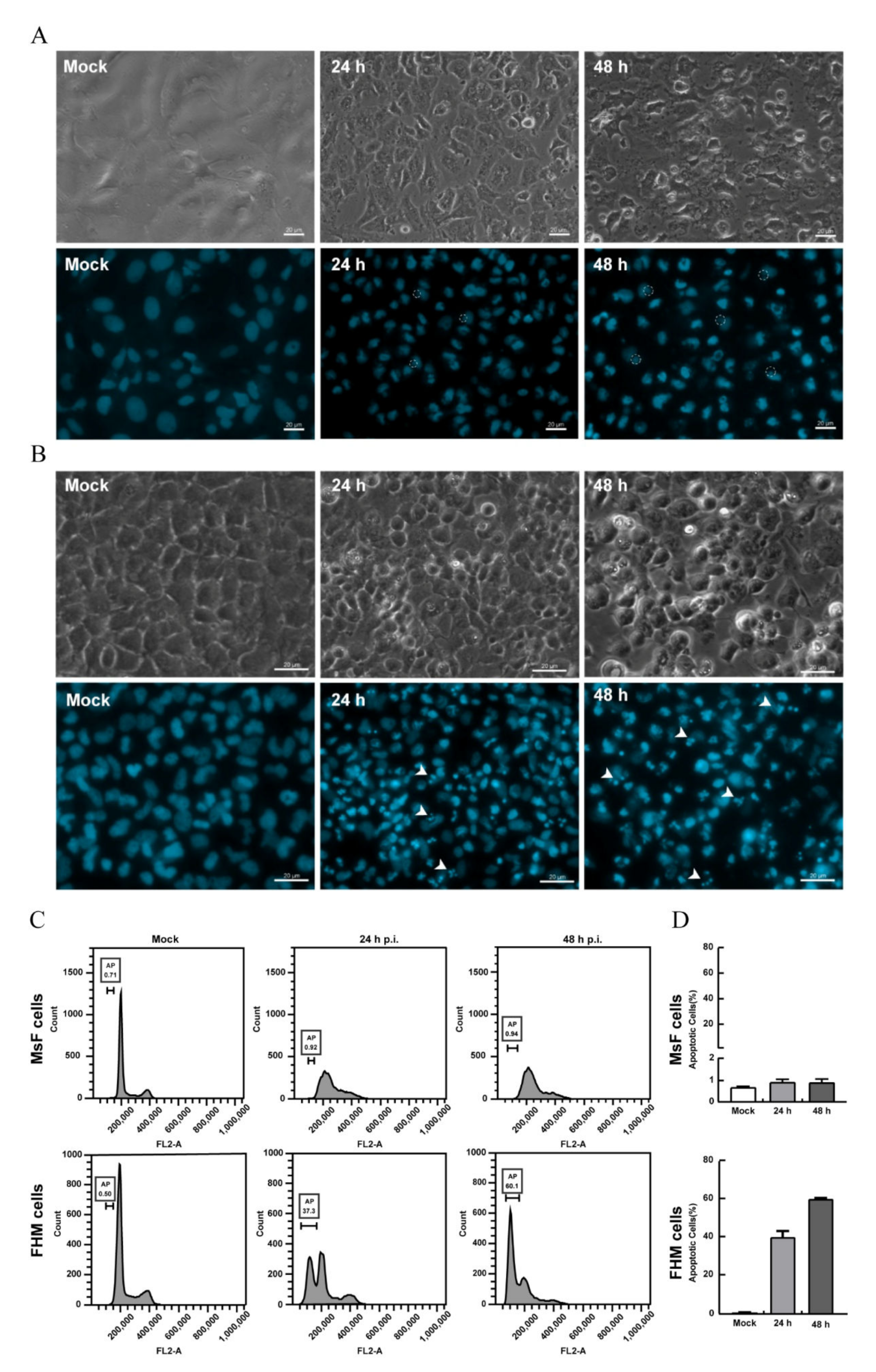
3.4. LMBV Infection Induced Nonapoptotic Cell Death in MsF Cells
3.5. Caspase-3 and Caspase-8 Were Activated in LMBV-Infected MsF Cells
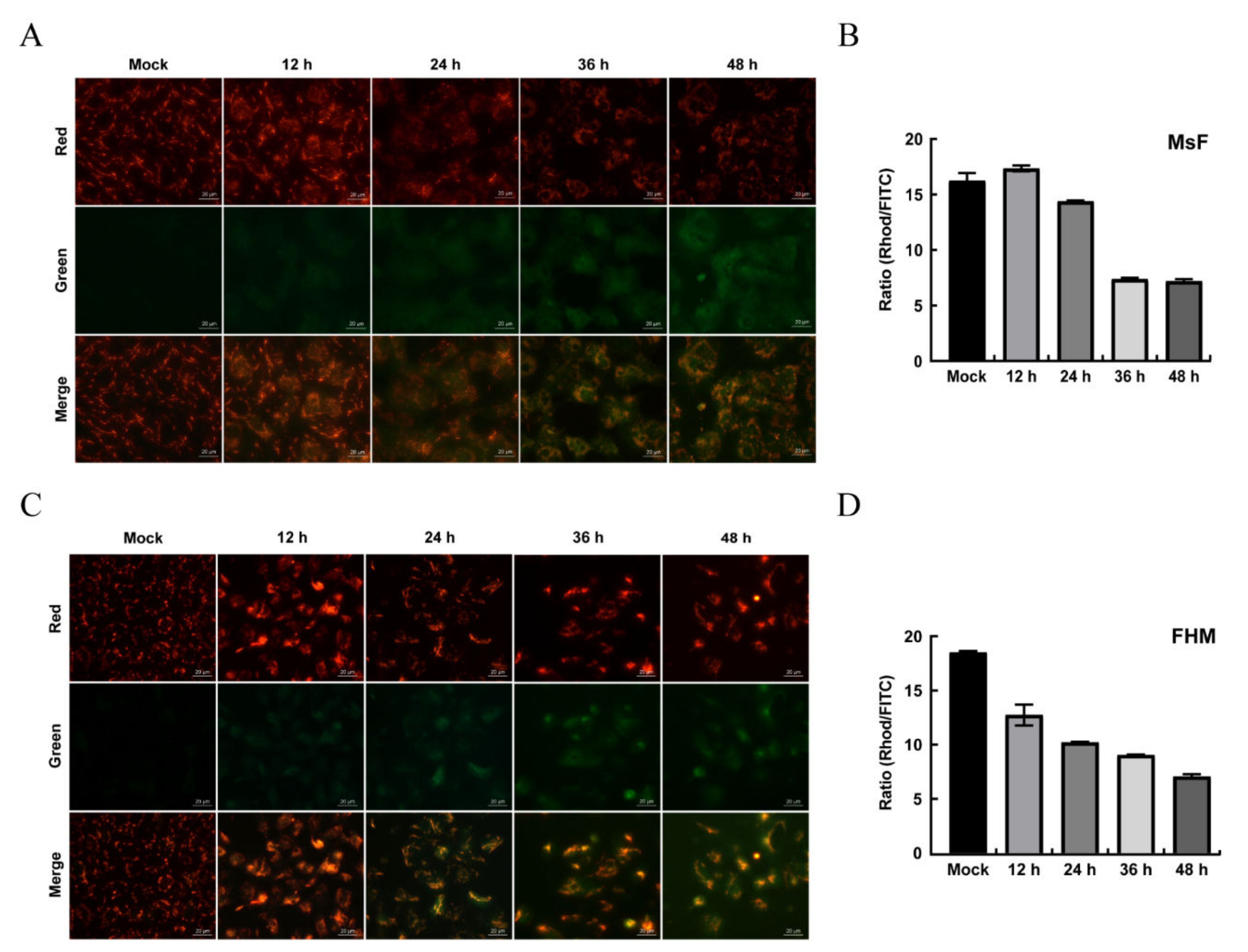
3.6. LMBV Induced ΔΨm Depolarization and ROS Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nguyen, L.N.; Kanneganti, T.D. PANoptosis in Viral Infection: The Missing Puzzle Piece in the Cell Death Field. J. Mol. Biol. 2022, 434, 167249. [Google Scholar] [CrossRef] [PubMed]
- Danthi, P. Viruses and the Diversity of Cell Death. Annu. Rev. Virol. 2016, 3, 533–553. [Google Scholar] [CrossRef] [PubMed]
- Mehrbod, P.; Ande, S.R.; Alizadeh, J.; Rahimizadeh, S.; Shariati, A.; Malek, H.; Hashemi, M.; Glover, K.K.M.; Sher, A.A.; Coombs, K.M.; et al. The roles of apoptosis, autophagy and unfolded protein response in arbovirus, influenza virus, and HIV infections. Virulence 2019, 10, 376–413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, T.Y.; Chu, J.J.H. Dengue virus-infected human monocytes trigger late activation of caspase-1, which mediates pro-inflammatory IL-1β secretion and pyroptosis. J. Gen. Virol. 2013, 94 Pt 10, 2215–2220. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.; Loh, Z.; Ullah, M.A.; Lynch, J.P.; Werder, R.B.; Collinson, N.; Zhang, V.; Dondelinger, Y.; Bertrand, M.J.M.; Everard, M.L.; et al. Respiratory Syncytial Virus Infection Promotes Necroptosis and HMGB1 Release by Airway Epithelial Cells. Am. J. Respir. Crit. Care Med. 2020, 201, 1358–1371. [Google Scholar] [CrossRef] [PubMed]
- Mbita, Z.; Hull, R.; Dlamini, Z. Human immunodeficiency virus-1 (HIV-1)-mediated apoptosis: New therapeutic targets. Viruses 2014, 6, 3181–3227. [Google Scholar] [CrossRef] [Green Version]
- Silberstein, E.; Ulitzky, L.; Lima, L.A.; Cehan, N.; Teixeira-Carvalho, A.; Roingeard, P.; Taylor, D.R. HCV-Mediated Apoptosis of Hepatocytes in Culture and Viral Pathogenesis. PLoS ONE 2016, 11, e0155708. [Google Scholar]
- James, S.F.; Mahalingam, R.; Gilden, D. Does apoptosis play a role in varicella zoster virus latency and reactivation? Viruses 2012, 4, 1509–1514. [Google Scholar] [CrossRef]
- Lowy, R.J. Influenza virus induction of apoptosis by intrinsic and extrinsic mechanisms. Int. Rev. Immunol. 2003, 22, 425–449. [Google Scholar] [CrossRef]
- Parquet, M.C.; Kumatori, A.; Hasebe, F.; Morita, K.; Igarashi, A. West Nile virus-induced bax-dependent apoptosis. FEBS Lett. 2001, 500, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Jiang, W.; Liu, Z.; Liu, S.; Liang, X. Virus Infection and Death Receptor-Mediated Apoptosis. Viruses 2017, 9, 316. [Google Scholar] [CrossRef] [Green Version]
- Place, D.E.; Lee, S.; Kanneganti, T.D. PANoptosis in microbial infection. Curr. Opin. Microbiol. 2021, 59, 42–49. [Google Scholar] [CrossRef]
- Wang, Y.; Kanneganti, T.D. From pyroptosis, apoptosis and necroptosis to PANoptosis: A mechanistic compendium of programmed cell death pathways. Comput. Struct. Biotechnol. J. 2021, 19, 4641–4657. [Google Scholar] [CrossRef]
- Jiang, M.; Qi, L.; Li, L.; Wu, Y.; Song, D.; Li, Y. Caspase-8: A key protein of cross-talk signal way in “PANoptosis” in cancer. Int. J. Cancer 2021, 149, 1408–1420. [Google Scholar] [CrossRef]
- Whittington, R.J.; Becker, J.A.; Dennis, M.M. Iridovirus infections in finfish—Critical review with emphasis on ranaviruses. J. Fish Dis. 2010, 33, 95–122. [Google Scholar] [CrossRef]
- Johan, C.A.C.; Zainathan, S.C. Megalocytiviruses in ornamental fish: A review. Vet. World 2020, 13, 2565–2577. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Waltzek, T.B.; Subramaniam, K. Ranaviruses and other members of the family Iridoviridae: Their place in the virosphere. Virology 2017, 511, 259–271. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Hick, P.; Ince, I.A.; Jancovich, J.K.; Marschang, R.; Qin, Q.; Subramaniam, K.; Waltzek, T.B.; Whittington, R.; Williams, T.; et al. ICTV Virus Taxonomy Profile: Iridoviridae. J. Gen. Virol. 2017, 98, 890–891. [Google Scholar] [CrossRef]
- Huang, Y.-H.; Huang, X.-H.; Gui, J.-F.; Zhang, Q.-Y. Mitochondrion-mediated apoptosis induced by Rana grylio virus infection in fish cells. Apoptosis 2007, 12, 1569–1577. [Google Scholar] [CrossRef]
- Chinchar, V.G.; Bryan, L.; Wang, J.; Long, S.; Chinchar, G.D. Induction of apoptosis in frog virus 3-infected cells. Virology 2003, 306, 303–312. [Google Scholar] [CrossRef] [Green Version]
- Pham, P.H.; Lai, Y.S.; Lee, F.F.; Bols, N.C.; Chiou, P.P. Differential viral propagation and induction of apoptosis by grouper iridovirus (GIV) in cell lines from three nonhost species. Virus Res. 2012, 167, 16–25. [Google Scholar] [CrossRef]
- Essbauer, S.; Ahne, W. The epizootic haematopoietic necrosis virus (Iridoviridae) induces apoptosis in vitro. J. Vet. Med. B Infect. Dis. Vet. Public Health 2002, 49, 25–30. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, N.; Fan, Y.; Zhou, Y.; Liu, W.; Xue, M.; Meng, Y.; Zeng, L. Chinese Giant Salamander (Andrias davidianus) Iridovirus Infection Leads to Apoptotic Cell Death through Mitochondrial Damage, Caspases Activation, and Expression of Apoptotic-Related Genes. Int. J. Mol. Sci. 2019, 20, 6149. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Huang, Y.; Ouyang, Z.; Xu, L.; Yan, Y.; Cui, H.; Han, X.; Qin, Q. Singapore grouper iridovirus, a large DNA virus, induces nonapoptotic cell death by a cell type dependent fashion and evokes ERK signaling. Apoptosis 2011, 16, 831–845. [Google Scholar] [CrossRef]
- Lai, Y.S.; Chiou, P.P.; Chen, W.J.; Chen, Y.C.; Chen, C.W.; Chiu, I.S.; Chen, S.D.; Cheng, Y.H.; Chang, C.Y. Characterization of apoptosis induced by grouper iridovirus in two newly established cell lines from barramundi, Lates calcarifer (Bloch). J. Fish Dis. 2008, 31, 825–834. [Google Scholar] [CrossRef]
- Bai, J.J.; Li, S.J. Current status and development trend on China largemouth bass industry. Chin. Fish. Econ. 2013, 31, 104–108. (In Chinese) [Google Scholar]
- Li, S.J.; Wang, G.J. Culture techniques of largemouth bass. Ocean Fish. 2016, 4, 72–74. (In Chinese) [Google Scholar]
- Mao, J.; Wang, J.; Chinchar, G.D.; Chinchar, V.G. Molecular characterization of a ranavirus isolated from largemouth bass Micropterus salmoides. Dis. Aquat. Org. 1999, 37, 107–114. [Google Scholar] [CrossRef] [Green Version]
- Deng, G.C.; Li, S.J.; Xie, J.; Bai, J.J.; Chen, K.C.; Ma, D.M.; Jiang, X.Y.; Lao, H.H.; Yu, L.Y. Characterization of a ranavirus isolated from cultured largemouth bass (Micropterus salmoides) in China. Aquaculture 2011, 312, 198–204. [Google Scholar] [CrossRef]
- Zhao, R.X.; Geng, Y.; Qin, Z.Y.; Wang, K.Y.; Yang, P.O.; Chen, D.F.; Huang, X.L.; Zuo, Z.C. A new ranavirus of the Santee-Cooper group invades largemouth bass (Micropterus salmoides) culture in southwest China. Aquaculture 2020, 526, 735363. [Google Scholar] [CrossRef]
- Huang, X.; Wang, W.; Huang, Y.; Xu, L.; Qin, Q. Involvement of the PI3K and ERK signaling pathways in largemouth bass virus-induced apoptosis and viral replication. Fish Shellfish Immunol. 2014, 41, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Feng, Y.; OuYang, P.; Chen, D.; Huang, X.; Guo, H.; Deng, H.; Fang, J.; Lai, W.; Geng, Y. Autophagy induced by largemouth bass virus inhibits virus replication and apoptosis in epithelioma papulosum cyprini cells. Fish Shellfish Immunol. 2022, 123, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Huang, X.H.; Ouyang, Z.L.; Wei, S.N.; Guo, C.Y.; Qin, Q.W. Development of a new cell line from the snout of giant grouper, Epinephelus lanceolatus (Bloch), and its application in iridovirus and nodavirus pathogenesis. Aquaculture 2014, 432, 265–272. [Google Scholar] [CrossRef]
- Van Opdenbosch, N.; Lamkanfi, M. Caspases in Cell Death, Inflammation, and Disease. Immunity 2019, 50, 1352–1364. [Google Scholar] [CrossRef] [PubMed]
- Gao, E.B.; Chen, G. Micropterus salmoides rhabdovirus (MSRV) infection induced apoptosis and activated interferon signaling pathway in largemouth bass skin cells. Fish Shellfish Immunol. 2018, 76, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.H.; Huang, Y.H.; Yuan, X.P.; Zhang, Q.Y. Electron microscopic examination of the viromatrix of Rana grylio virus in a fish cell line. J. Virol. Methods 2006, 133, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Brookes, S.M.; Dixon, L.K.; Parkhouse, R.M. Assembly of African Swine fever virus: Quantitative ultrastructural analysis in vitro and in vivo. Virology 1996, 224, 84–92. [Google Scholar] [CrossRef]
- Kesavardhana, S.; Malireddi, R.K.S.; Kanneganti, T.D. Caspases in Cell Death, Inflammation, and Pyroptosis. Annu. Rev. Immunol. 2020, 38, 567–595. [Google Scholar] [CrossRef] [Green Version]
- Xia, S.; Hollingsworth, L.R., IV; Wu, H. Mechanism and Regulation of Gasdermin-Mediated Cell Death. Cold Spring Harb. Perspect. Biol. 2020, 12, a036400. [Google Scholar] [CrossRef] [Green Version]
- Fritsch, M.; Günther, S.D.; Schwarzer, R.; Albert, M.C.; Schorn, F.; Werthenbach, J.P.; Schiffmann, L.M.; Stair, N.; Stocks, H.; Seeger, J.M.; et al. Caspase-8 is the molecular switch for apoptosis, necroptosis and pyroptosis. Nature 2019, 575, 683–687. [Google Scholar] [CrossRef]
- Radogna, F.; Cerella, C.; Gaigneaux, A.; Christov, C.; Dicato, M.; Diederich, M. Cell type-dependent ROS and mitophagy response leads to apoptosis or necroptosis in neuroblastoma. Oncogene 2016, 35, 3839–3853. [Google Scholar] [CrossRef]
- Holze, C.; Michaudel, C.; Mackowiak, C.; Haas, D.A.; Benda, C.; Hubel, P.; Pennemann, F.L.; Schnepf, D.; Wettmarshausen, J.; Braun, M.; et al. Oxeiptosis, a ROS-induced caspase-independent apoptosis-like cell-death pathway. Nat. Immunol. 2018, 19, 130–140. [Google Scholar] [CrossRef]




| Primer Names | Sequence (5’-3’) |
|---|---|
| LMBV-MCP-F | CTCGCCACTTATGACAGCCTTGAC |
| LMBV-MCP-R | AACCCACGGGATAATGCTCTTTGAC |
| LMBV-MMP-F | GCGTATTTCGCACCCTCTG |
| LMBV-MMP-R | TAAGCGTCGCCCTTGTCTG |
| LMBV-DNMT-F | AATGTTTGGGTTTGAGGTAG |
| LMBV-DNMT-R | TCTTTAGCAGGCTGAGGG |
| MsF-β-actin-F | CCACCACAGCCGAGAGGGAA |
| MsF-β-actin-R | TCATGGTGGATGGGGCCAGG |
| FHM-β-actin-F | TACGAGCTGCCTGACGGACA |
| FHM-β-actin-R | GGCTGTGATCTCCTTCTGCA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Xu, W.; Wang, W.; Pan, Z.; Qin, Q.; Huang, X.; Huang, Y. Largemouth Bass Virus Infection Induced Non-Apoptotic Cell Death in MsF Cells. Viruses 2022, 14, 1568. https://doi.org/10.3390/v14071568
Yang J, Xu W, Wang W, Pan Z, Qin Q, Huang X, Huang Y. Largemouth Bass Virus Infection Induced Non-Apoptotic Cell Death in MsF Cells. Viruses. 2022; 14(7):1568. https://doi.org/10.3390/v14071568
Chicago/Turabian StyleYang, Jiahui, Weihua Xu, Wenji Wang, Zanbin Pan, Qiwei Qin, Xiaohong Huang, and Youhua Huang. 2022. "Largemouth Bass Virus Infection Induced Non-Apoptotic Cell Death in MsF Cells" Viruses 14, no. 7: 1568. https://doi.org/10.3390/v14071568
APA StyleYang, J., Xu, W., Wang, W., Pan, Z., Qin, Q., Huang, X., & Huang, Y. (2022). Largemouth Bass Virus Infection Induced Non-Apoptotic Cell Death in MsF Cells. Viruses, 14(7), 1568. https://doi.org/10.3390/v14071568







