Palmitoylation Is Indispensable for Remorin to Restrict Tobacco Mosaic Virus Cell-to-Cell Movement in Nicotiana benthamiana
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Growth Conditions
2.2. RNA Extraction and Plasmid Construction
2.3. Agroinfiltration in N. benthamiana
2.4. Multiple Sequence Alignment and Phylogenetic Analysis
2.5. Reverse Transcription-Quantitative PCR (RT-qPCR)
2.6. Virus Inoculation and Detection
2.7. Acyl-Resin Assisted Capture (Acyl-RAC)
2.8. Immunoblotting and Antibodies
2.9. Bimolecular Fluorescence Complementation (BIFC) and Subcellular Localization Assays
2.10. Tobacco Rattle Virus (TRV)-Induced Gene Silencing
2.11. Callose Deposition Assay
2.12. Quantification and Statistical Analysis
3. Results
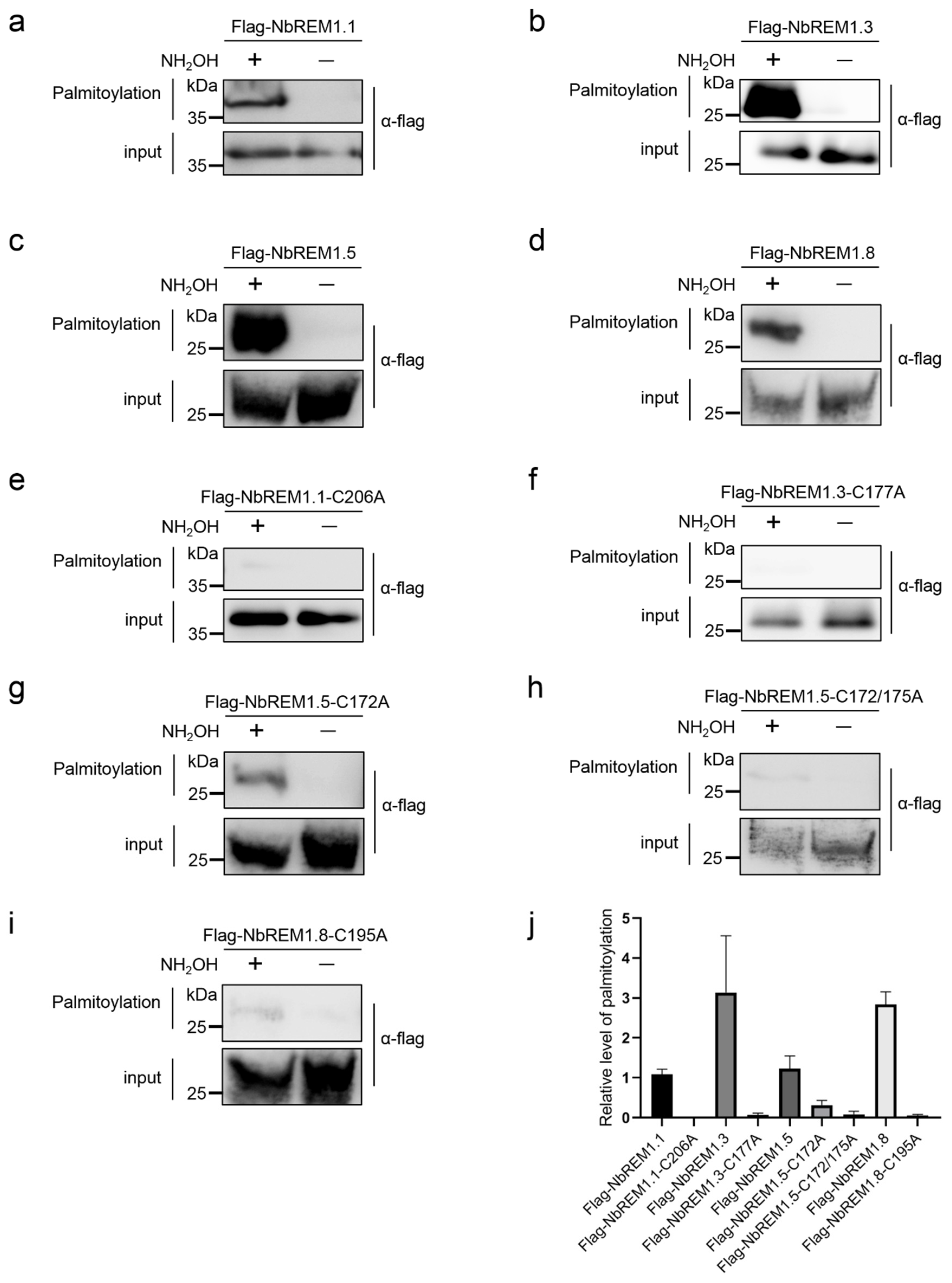
3.1. Group 1 Remorin Proteins Are Palmitoylated in N. benthamiana
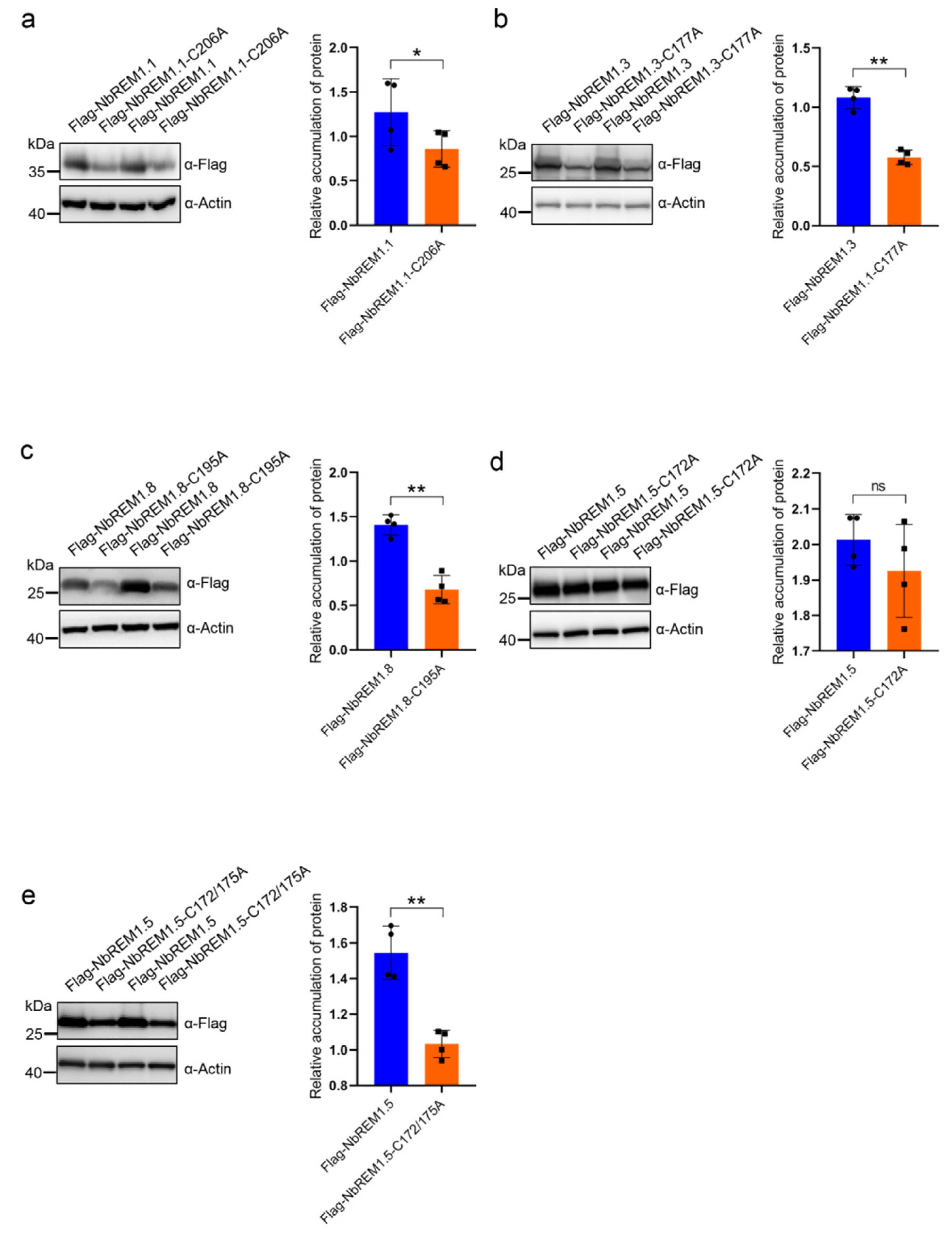
3.2. Palmitoylation Contributes to Remorin Protein Accumulation
3.3. Palmitoylation Contributes to Plasma Membrane Localization for Remorin Proteins
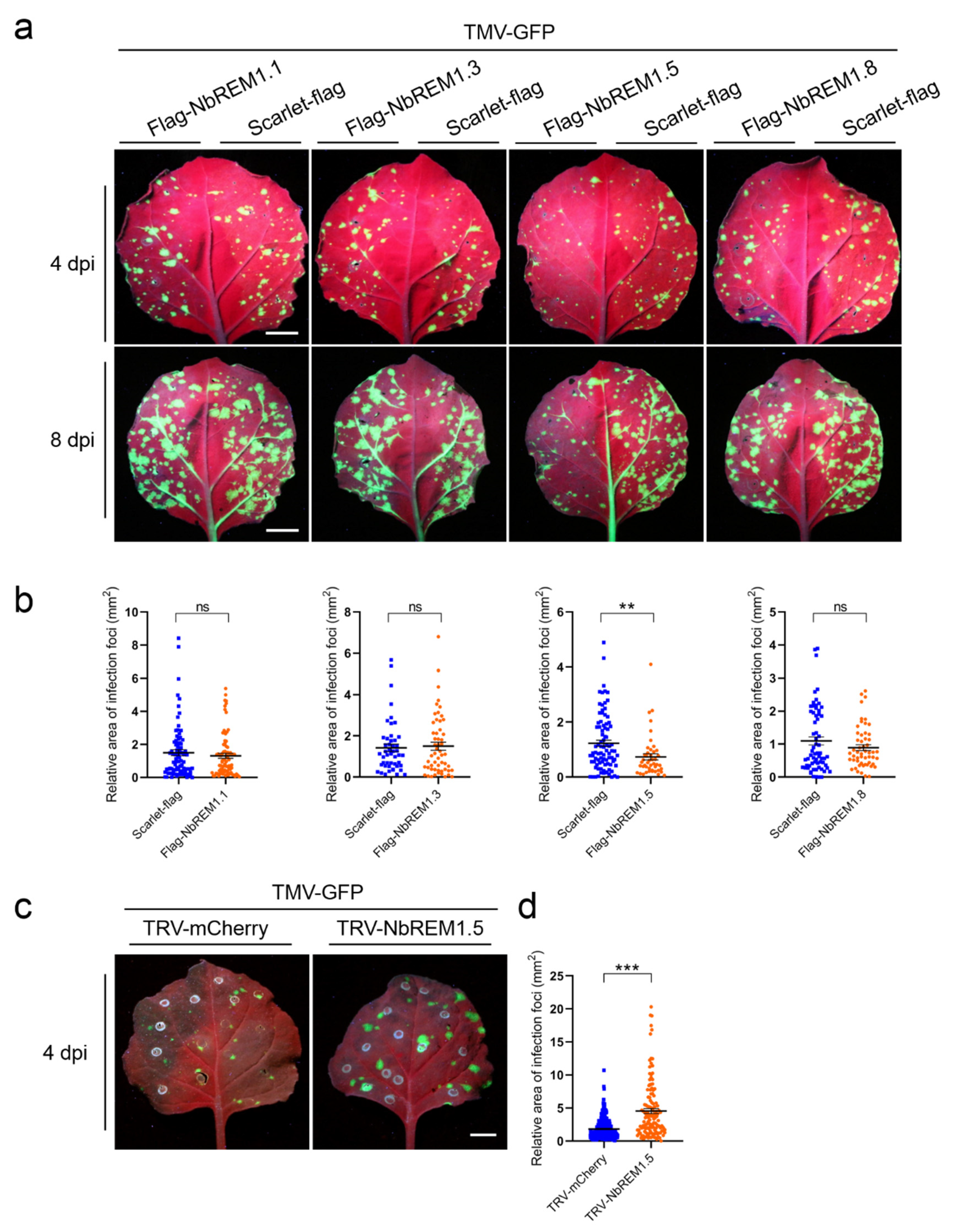
3.4. NbREM1.5 Negatively Regulates TMV Cell-to-Cell Movement
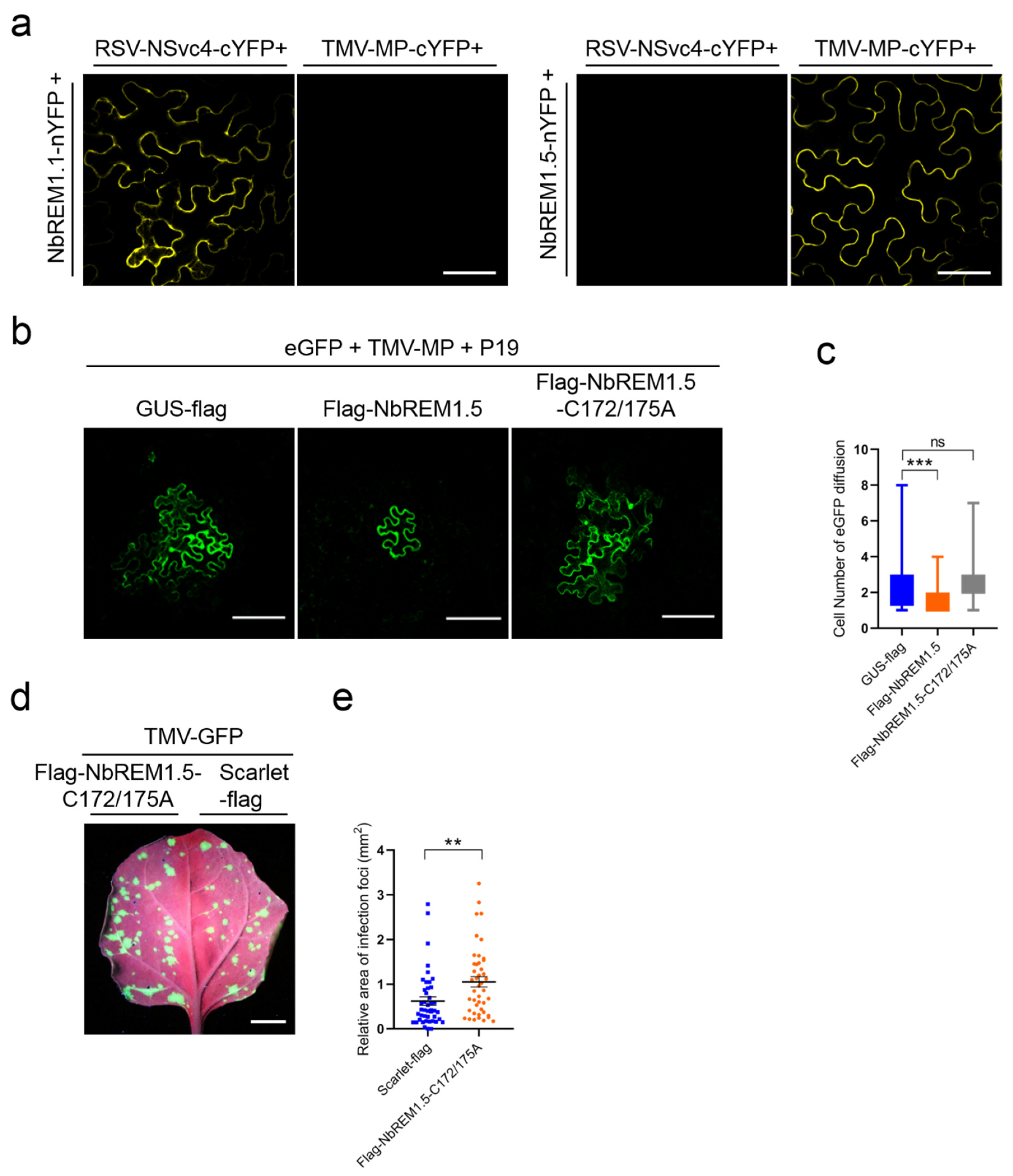
3.5. NbREM1.5 Interacts with TMV-MP and Interferes with Its Ability for Expanding Plasmodesmata Size Exclusion Limit
3.6. Palmitoylation Is Required for NbREM1.5 to Inhibit TMV Cell-to-Cell Movement
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petit, J.D.; Li, Z.P.; Nicolas, W.J.; Grison, M.S.; Bayer, E.M. Dare to change, the dynamics behind plasmodesmata-mediated cell-to-cell communication. Curr. Opin. Plant Biol. 2020, 53, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, W.J.; Grison, M.S.; Bayer, E.M. Shaping intercellular channels of plasmodesmata: The structure-to-function missing link. J. Exp. Bot. 2017, 69, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, K.; Mitra, R.; Payton, M.E.; Verchot-Lubicz, J. Cell-to-cell movement of the PVX 12K, 8K, or coat proteins may depend on the host, leaf developmental stage, and the PVX 25K protein. Virology 2002, 300, 269–281. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wright, K.M.; Chapman, S.; Roberts, A.G. Plasmodesmal targeting and accumulation of TMV movement protein. Plant Signal. Behav. 2007, 2, 180–181. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, S.W.; Kumar, R.; Iswanto, A.B.B.; Kim, J.Y. Callose balancing at plasmodesmata. J. Exp. Bot. 2018, 69, 5325–5339. [Google Scholar] [CrossRef] [PubMed]
- Heinlein, M. Plasmodesmata: Channels for viruses on the move. Methods Mol. Biol. 2015, 1217, 25–52. [Google Scholar]
- Gui, J.; Liu, C.; Shen, J.; Li, L. Grain setting defect1, encoding a remorin protein, affects the grain setting in rice through regulating plasmodesmatal conductance. Plant Physiol. 2014, 166, 1463–1478. [Google Scholar] [CrossRef]
- Fu, S.; Xu, Y.; Li, C.; Li, Y.; Wu, J.; Zhou, X. Rice stripe virus interferes with S-acylation of remorin and induces its autophagic degradation to facilitate virus infection. Mol. Plant 2018, 11, 269–287. [Google Scholar] [CrossRef]
- Huang, D.; Sun, Y.; Ma, Z.; Ke, M.; Cui, Y.; Chen, Z.; Chen, C.; Ji, C.; Tran, T.M.; Yang, L.; et al. Salicylic acid-mediated plasmodesmal closure via remorin-dependent lipid organization. Proc. Natl. Acad. Sci. USA 2019, 116, 21274–21284. [Google Scholar] [CrossRef]
- Gouguet, P.; Gronnier, J.; Legrand, A.; Perraki, A.; Jolivet, M.D.; Deroubaix, A.F.; German-Retana, S.; Boudsocq, M.; Habenstein, B.; Mongrand, S.; et al. Connecting the dots: From nanodomains to physiological functions of REMORINs. Plant Physiol. 2021, 185, 632–649. [Google Scholar] [CrossRef]
- Perraki, A.; Cacas, J.L.; Crowet, J.M.; Lins, L.; Castroviejo, M.; German-Retana, S.; Mongrand, S.; Raffaele, S. Plasma membrane localization of Solanum tuberosum remorin from group 1, homolog 3 is mediated by conformational changes in a novel C-terminal anchor and required for the restriction of potato virus X movement. Plant Physiol. 2012, 160, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Legrand, A.; Martinez, D.; Grelard, A.; Berbon, M.; Morvan, E.; Tawani, A.; Loquet, A.; Mongrand, S.; Habenstein, B. Nanodomain clustering of the plant protein remorin by solid-state NMR. Front. Mol. Biosci. 2019, 6, 107. [Google Scholar] [CrossRef] [PubMed]
- Gronnier, J.; Crowet, J.M.; Habenstein, B.; Nasir, M.N.; Bayle, V.; Hosy, E.; Platre, M.P.; Gouguet, P.; Raffaele, S.; Martinez, D.; et al. Structural basis for plant plasma membrane protein dynamics and organization into functional nanodomains. Elife 2017, 6, e26404. [Google Scholar] [CrossRef] [PubMed]
- Konrad, S.S.; Popp, C.; Stratil, T.F.; Jarsch, I.K.; Thallmair, V.; Folgmann, J.; Marin, M.; Ott, T. S-acylation anchors remorin proteins to the plasma membrane but does not primarily determine their localization in membrane microdomains. New Phytol. 2014, 203, 758–769. [Google Scholar] [CrossRef]
- Kordyukova, L.; Krabben, L.; Serebryakova, M.; Veit, M. S-acylation of proteins. Methods Mol. Biol. 2019, 1934, 265–291. [Google Scholar]
- Bijlmakers, M.J.; Marsh, M. The on-off story of protein palmitoylation. Trends Cell Biol. 2003, 13, 32–42. [Google Scholar] [CrossRef]
- Das, T.; Yount, J.S.; Hang, H.C. Protein S-palmitoylation in immunity. Open Biol. 2021, 11, 200411. [Google Scholar] [CrossRef]
- Gui, J.; Zheng, S.; Shen, J.; Li, L. Grain setting defect1 (GSD1) function in rice depends on S-acylation and interacts with actin 1 (OsACT1) at its C-terminal. Front. Plant Sci. 2015, 6, 804. [Google Scholar] [CrossRef]
- Bindels, D.S.; Haarbosch, L.; van Weeren, L.; Postma, M.; Wiese, K.E.; Mastop, M.; Aumonier, S.; Gotthard, G.; Royant, A.; Hink, M.A.; et al. mScarlet: A bright monomeric red fluorescent protein for cellular imaging. Nat. Methods 2017, 14, 53–56. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.J.; Higgins, D.G. Multiple sequence alignment using ClustalW and ClustalX. In Current Protocols in Bioinformatics; John Wiley and Sons: Hoboken, NJ, USA, 2002; Chapter 2, Unit 2.3. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Shivprasad, S.; Pogue, G.P.; Lewandowski, D.J.; Hidalgo, J.; Donson, J.; Grill, L.K.; Dawson, W.O. Heterologous sequences greatly affect foreign gene expression in tobacco mosaic virus-based vectors. Virology 1999, 255, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Forrester, M.T.; Hess, D.T.; Thompson, J.W.; Hultman, R.; Moseley, M.A.; Stamler, J.S.; Casey, P.J. Site-specific analysis of protein S-acylation by resin-assisted capture. J. Lipid Res. 2011, 52, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Wang, K.; Ma, T.; Liang, Y.; Ma, Z.; Wu, J.; Xu, Y.; Zhou, X. An evolutionarily conserved C4HC3-type E3 ligase regulates plant broad-spectrum resistance against pathogens. Plant Cell 2022, 34, 1822–1843. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Kopperud, K.; Chakrabarty, R.; Banerjee, R.; Brooks, R.; Goodin, M.M. Transient expression in Nicotiana benthamiana fluorescent marker lines provides enhanced definition of protein localization, movement and interactions in planta. Plant J. 2009, 59, 150–162. [Google Scholar] [CrossRef]
- Ning, W.; Jiang, P.; Guo, Y.; Wang, C.; Tan, X.; Zhang, W.; Peng, D.; Xue, Y. GPS-Palm: A deep learning-based graphic presentation system for the prediction of S-palmitoylation sites in proteins. Brief. Bioinform. 2021, 22, 1836–1847. [Google Scholar] [CrossRef]
- Hurst, C.H.; Turnbull, D.; Plain, F.; Fuller, W.; Hemsley, P.A. Maleimide scavenging enhances determination of protein S-palmitoylation state in acyl-exchange methods. Biotechniques 2017, 62, 69–75. [Google Scholar] [CrossRef]
- Perraki, A.; Binaghi, M.; Mecchia, M.A.; Gronnier, J.; German-Retana, S.; Mongrand, S.; Bayer, E.; Zelada, A.M.; Germain, V. StRemorin1.3 hampers Potato virus X TGBp1 ability to increase plasmodesmata permeability, but does not interfere with its silencing suppressor activity. FEBS Lett. 2014, 588, 1699–1705. [Google Scholar] [CrossRef]
- Voinnet, O.; Rivas, S.; Mestre, P.; Baulcombe, D. An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein of tomato bushy stunt virus. Plant J. 2003, 33, 949–956. [Google Scholar] [CrossRef]
- Daniotti, J.L.; Pedro, M.P.; Taubas, J.V. The role of S-acylation in protein trafficking. Traffic 2017, 18, 699–710. [Google Scholar] [CrossRef]
- Hemsley, P.A. S-acylation in plants: An expanding field. Biochem. Soc. Trans. 2020, 48, 529–536. [Google Scholar] [CrossRef]
- Cheng, G.; Yang, Z.; Zhang, H.; Zhang, J.; Xu, J. Remorin interacting with PCaP1 impairs turnip mosaic virus intercellular movement but is antagonised by VPg. New Phytol. 2020, 225, 2122–2139. [Google Scholar] [CrossRef] [PubMed]
- Perraki, A.; Gronnier, J.; Gouguet, P.; Boudsocq, M.; Deroubaix, A.F.; Simon, V.; German-Retana, S.; Legrand, A.; Habenstein, B.; Zipfel, C.; et al. REM1.3′s phospho-status defines its plasma membrane nanodomain organization and activity in restricting PVX cell-to-cell movement. PLoS Pathog. 2018, 14, e1007378. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, N.; Takashima, E.; Nyunoya, H. Altered subcellular localization of a tobacco membrane raft-associated remorin protein by tobamovirus infection and transient expression of viral replication and movement proteins. Front. Plant Sci. 2018, 9, 619. [Google Scholar] [CrossRef]
- Raffaele, S.; Bayer, E.; Lafarge, D.; Cluzet, S.; German Retana, S.; Boubekeur, T.; Leborgne-Castel, N.; Carde, J.P.; Lherminier, J.; Noirot, E.; et al. Remorin, a solanaceae protein resident in membrane rafts and plasmodesmata, impairs potato virus X movement. Plant Cell 2009, 21, 1541–1555. [Google Scholar] [CrossRef]
- Jarsch, I.K.; Konrad, S.S.; Stratil, T.F.; Urbanus, S.L.; Szymanski, W.; Braun, P.; Braun, K.H.; Ott, T. Plasma membranes are subcompartmentalized into a plethora of coexisting and diverse microdomains in Arabidopsis and Nicotiana benthamiana. Plant Cell 2014, 26, 1698–1711. [Google Scholar] [CrossRef] [PubMed]
- Bucherl, C.A.; Jarsch, I.K.; Schudoma, C.; Segonzac, C.; Mbengue, M.; Robatzek, S.; MacLean, D.; Ott, T.; Zipfel, C. Plant immune and growth receptors share common signalling components but localise to distinct plasma membrane nanodomains. Elife 2017, 6, e25114. [Google Scholar] [CrossRef]
- Marin, M.; Thallmair, V.; Ott, T. The intrinsically disordered N-terminal region of AtREM1.3 remorin protein mediates protein-protein interactions. J. Biol. Chem. 2012, 287, 39982–39991. [Google Scholar] [CrossRef]
- Marin, M.; Ott, T. Phosphorylation of intrinsically disordered regions in remorin proteins. Front. Plant Sci. 2012, 3, 86. [Google Scholar] [CrossRef]
- Albers, P.; Ustun, S.; Witzel, K.; Kraner, M.; Bornke, F. A remorin from Nicotiana benthamiana interacts with the Pseudomonas Type-III effector protein HopZ1a and is phosphorylated by the immune-related kinase PBS1. Mol. Plant Microbe Interact. 2019, 32, 1229–1242. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, T.; Fu, S.; Wang, K.; Wang, Y.; Wu, J.; Zhou, X. Palmitoylation Is Indispensable for Remorin to Restrict Tobacco Mosaic Virus Cell-to-Cell Movement in Nicotiana benthamiana. Viruses 2022, 14, 1324. https://doi.org/10.3390/v14061324
Ma T, Fu S, Wang K, Wang Y, Wu J, Zhou X. Palmitoylation Is Indispensable for Remorin to Restrict Tobacco Mosaic Virus Cell-to-Cell Movement in Nicotiana benthamiana. Viruses. 2022; 14(6):1324. https://doi.org/10.3390/v14061324
Chicago/Turabian StyleMa, Tingting, Shuai Fu, Kun Wang, Yaqin Wang, Jianxiang Wu, and Xueping Zhou. 2022. "Palmitoylation Is Indispensable for Remorin to Restrict Tobacco Mosaic Virus Cell-to-Cell Movement in Nicotiana benthamiana" Viruses 14, no. 6: 1324. https://doi.org/10.3390/v14061324
APA StyleMa, T., Fu, S., Wang, K., Wang, Y., Wu, J., & Zhou, X. (2022). Palmitoylation Is Indispensable for Remorin to Restrict Tobacco Mosaic Virus Cell-to-Cell Movement in Nicotiana benthamiana. Viruses, 14(6), 1324. https://doi.org/10.3390/v14061324







