Associations and Disease–Disease Interactions of COVID-19 with Congenital and Genetic Disorders: A Comprehensive Review
Abstract
1. Introduction
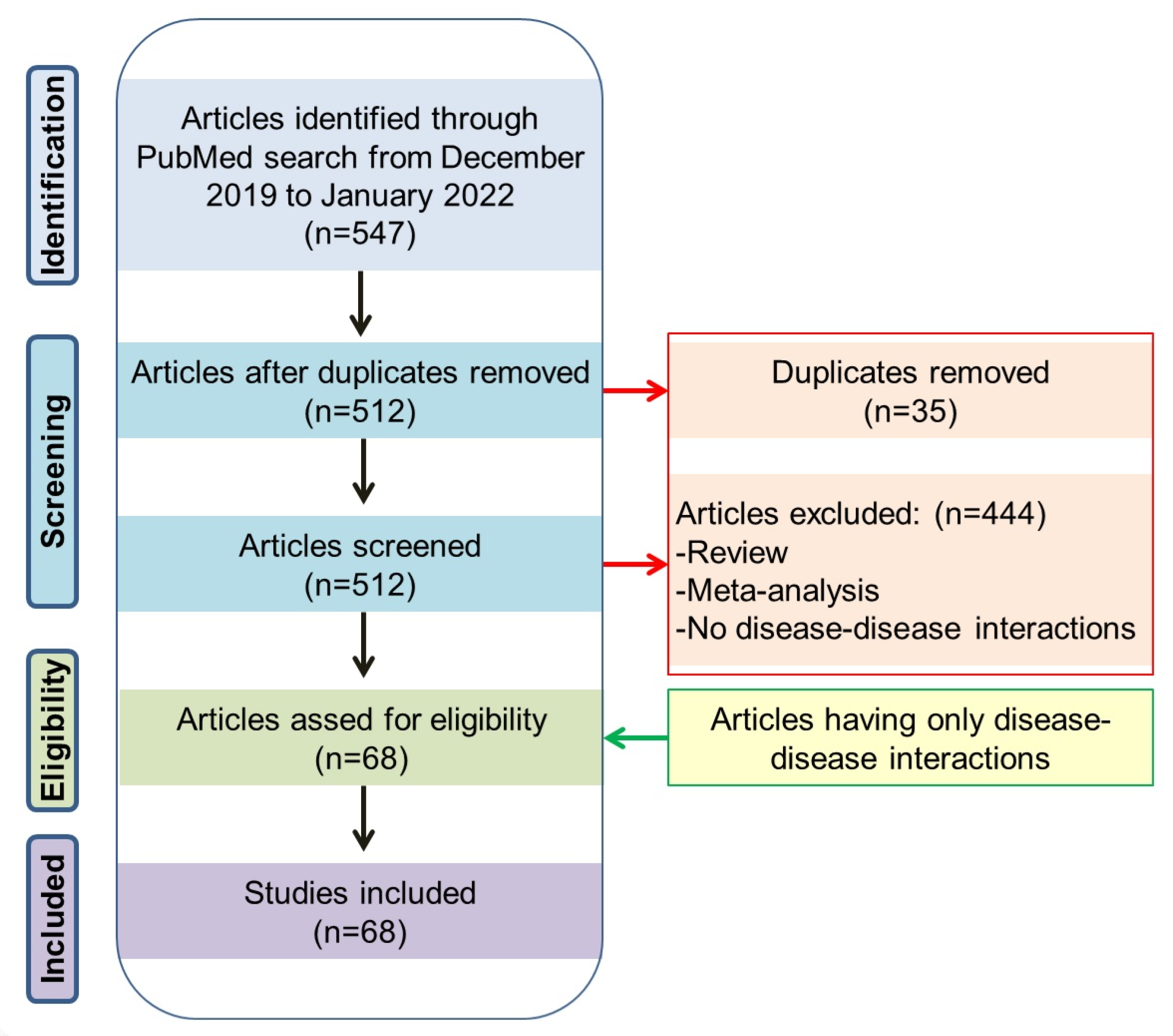
2. Methods
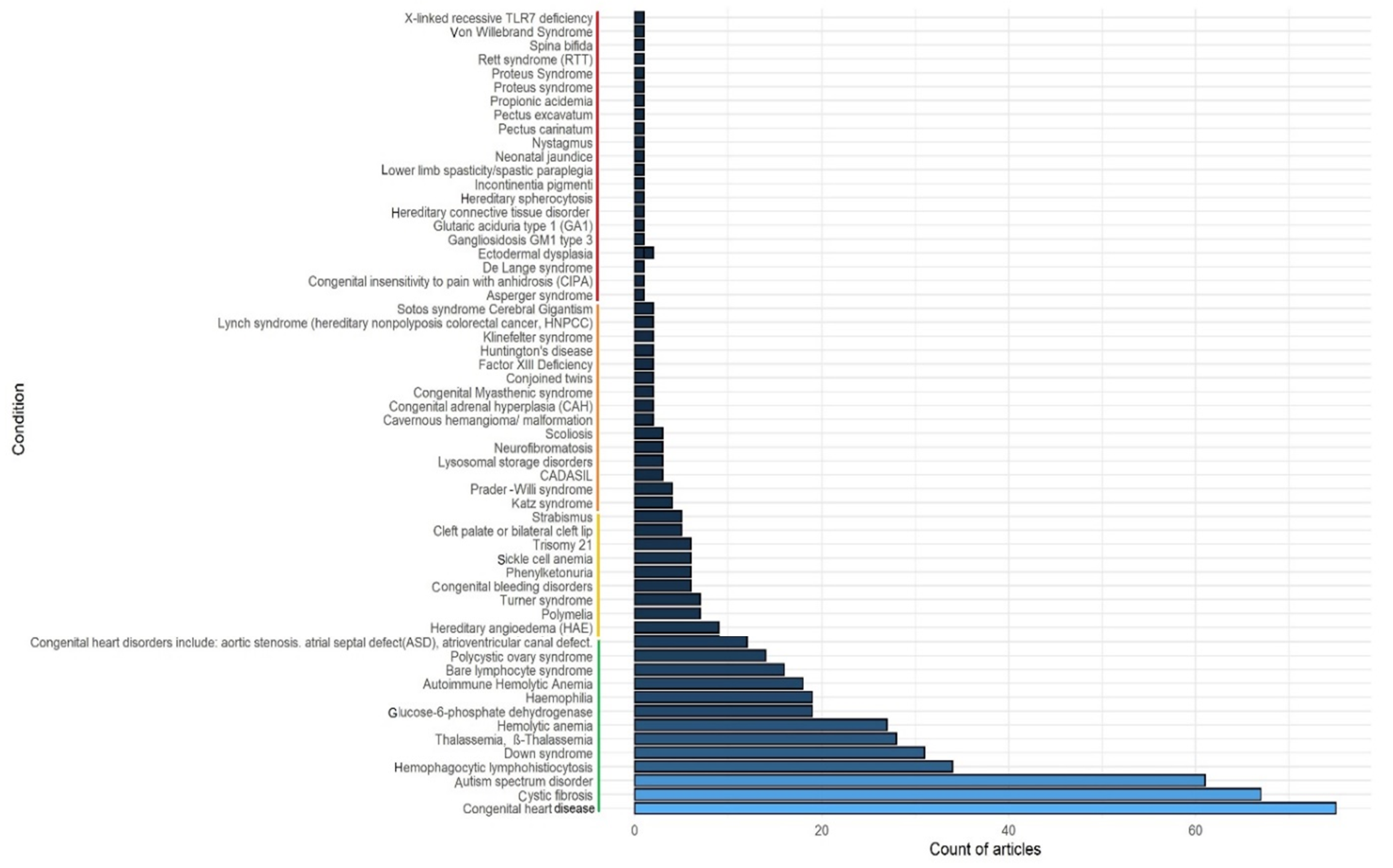
3. Literature Search Results
4. Most Frequently (>10 PubMed Hits) Associated Congenital and Genetic Disorders with COVID-19
4.1. COVID-19 and Congenital Heart Disease (CHD)
4.2. COVID-19 and Cystic Fibrosis (CF)
4.3. COVID-19 and Autism Spectrum Disorder (ASD)
4.4. COVID-19 and Autoimmune Hemolytic Anemia (AIHA)
4.5. COVID-19 and Hematophagocytic Lymphohistocytosis (HLH)
4.6. COVID-19 in Adult Patients and Children with Down Syndrome (DS)
4.7. COVID-19 and Thalassemia
4.8. COVID-19 and G6P Dehydrogenase Insufficiency
4.9. COVID-19 and Agammaglobulinemia
4.10. COVID-19 and Polycystic Ovary Syndrome (PCOS)
5. Congenital and Genetic Conditions with Five to Ten PubMed Hits
5.1. COVID-19 and Hereditary Angioedema (HAE)
5.2. COVID-19 and Parsonage-Turner Syndrome (PTS)
6. Congenital and Genetic Conditions Less Frequently (<5 PubMed Hits) Associated with COVID-19
6.1. COVID-19 and Prader–Willi Syndrome (PWS)
6.2. COVID-19 and Lysosomal Storage Disorders
6.3. COVID-19 and Hereditary Spherocytosis (HS)
6.4. COVID-19 in Patients with Spina Bifida (SB)
6.5. COVID-19 and Hypothyroidism
6.6. COVID-19 and Fragile X-Syndrome (FXS)
6.7. COVID-19 and Duchenne/Becker Muscular Dystrophy
6.8. COVID-19 and Neuromyelitis Optica Spectrum Disorder
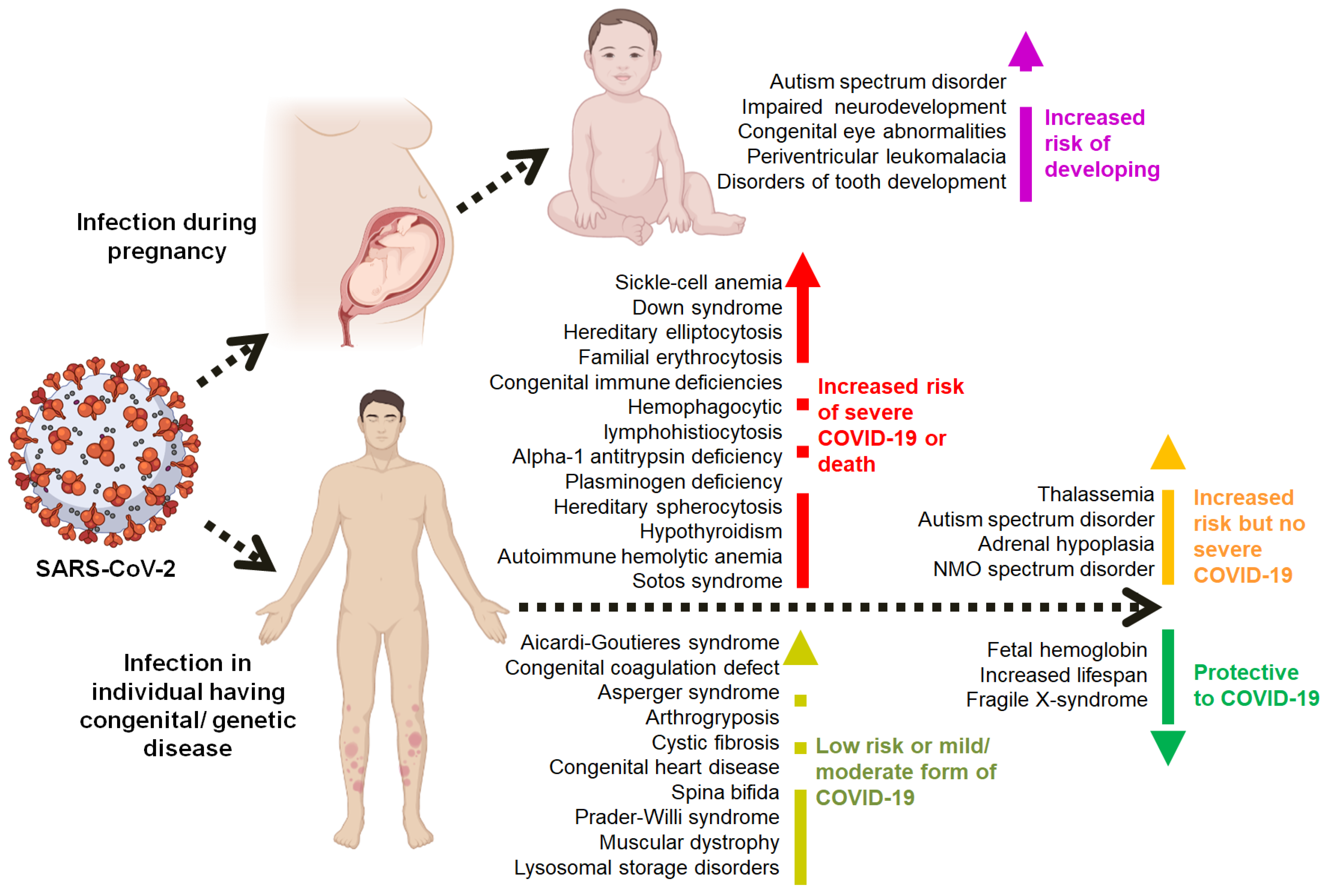
7. Multi-Omics-Based Predicted Congenital Anomalies and Genetic Diseases and Their Associations with COVID-19
8. COVID-19 during Early Pregnancy
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.-L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2020, 19, 141–154. [Google Scholar] [CrossRef]
- Khan, M.S.I.; Nabeka, H.; Akbar, S.M.F.; Al Mahtab, M.; Shimokawa, T.; Islam, F.; Matsuda, S. Risk of Congenital Birth Defects during COVID-19 Pandemic: Draw Attention to the Physicians and Policymakers. J. Glob. Health 2020, 10, 020378. [Google Scholar] [CrossRef]
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. More than 50 Long-Term Effects of COVID-19: A Systematic Review and Meta-Analysis. Sci. Rep. 2021, 11, 16144. [Google Scholar] [CrossRef]
- Schwerzmann, M.; Ruperti-Repilado, F.J.; Baumgartner, H.; Bouma, B.; Bouchardy, J.; Budts, W.; Campens, L.; Chessa, M.; del Cerro Marin, M.J.; Gabriel, H.; et al. Clinical Outcome of COVID-19 in Patients with Adult Congenital Heart Disease. Heart 2021, 107, 1226–1232. [Google Scholar] [CrossRef]
- Barh, D.; Tiwari, S.; Andrade, B.S.; Weener, M.E.; Góes-Neto, A.; Azevedo, V.; Ghosh, P.; Blum, K.; Ganguly, N.K. A Novel Multi-Omics-Based Highly Accurate Prediction of Symptoms, Comorbid Conditions, and Possible Long-Term Complications of COVID-19. Mol. Omics 2021, 17, 317–337. [Google Scholar] [CrossRef]
- Galloway, J.B.; Norton, S.; Barker, R.D.; Brookes, A.; Carey, I.; Clarke, B.D.; Jina, R.; Reid, C.; Russell, M.D.; Sneep, R.; et al. A Clinical Risk Score to Identify Patients with COVID-19 at High Risk of Critical Care Admission or Death: An Observational Cohort Study. J. Infect. 2020, 81, 282–288. [Google Scholar] [CrossRef]
- Lewis, M.J.; Anderson, B.R.; Fremed, M.; Argenio, M.; Krishnan, U.; Weller, R.; Levasseur, S.; Sommer, R.; Lytrivi, I.D.; Bacha, E.A.; et al. Impact of Coronavirus Disease 2019 (COVID-19) on Patients with Congenital Heart Disease Across the Lifespan: The Experience of an Academic Congenital Heart Disease Center in New York City. J. Am. Heart Assoc. 2020, 9, e017580. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, H.; Budts, W.; Chessa, M.; Deanfield, J.; Eicken, A.; Holm, J.; Iserin, L.; Meijboom, F.; Stein, J.; Szatmari, A.; et al. Recommendations for Organization of Care for Adults with Congenital Heart Disease and for Training in the Subspecialty of “Grown-up Congenital Heart Disease” in Europe: A Position Paper of the Working Group on Grown-up Congenital Heart Disease of the European Society of Cardiology. Eur. Heart J. 2014, 35, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Guzik, T.J.; Mohiddin, S.A.; Dimarco, A.; Patel, V.; Savvatis, K.; Marelli-Berg, F.M.; Madhur, M.S.; Tomaszewski, M.; Maffia, P.; D’Acquisto, F.; et al. COVID-19 and the Cardiovascular System: Implications for Risk Assessment, Diagnosis, and Treatment Options. Cardiovasc. Res. 2020, 116, 1666–1687. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Aboulhosn, J. The Cardiovascular Burden of Coronavirus Disease 2019 (COVID-19) with a Focus on Congenital Heart Disease. Int. J. Cardiol. 2020, 309, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, J.; Ferrero, P.; Chessa, M.; Bianco, F.; Ciliberti, P.; Secinaro, A.; Oreto, L.; Avesani, M.; Bucciarelli, V.; Calcaterra, G.; et al. COVID-19 and Congenital Heart Disease: Results from a Nationwide Survey. J. Clin. Med. 2020, 9, 1774. [Google Scholar] [CrossRef] [PubMed]
- Nepogodiev, D.; Bhangu, A.; Glasbey, J.C.; Li, E.; Omar, O.M.; Simoes, J.F.; Abbott, T.E.; Alser, O.; Arnaud, A.P.; Bankhead-Kendall, B.K.; et al. Mortality and Pulmonary Complications in Patients Undergoing Surgery with Perioperative SARS-CoV-2 Infection: An International Cohort Study. Lancet 2020, 396, 27–38. [Google Scholar] [CrossRef]
- Atalay, A.; Soran Türkcan, B.; Taşoğluİ, İ.; Külahçıoğlu, E.; Yilmaz, M.; Ecevit, A.N.; Aydin, N.H. Management of Congenital Cardiac Surgery during COVID-19 Pandemic. Cardiol. Young 2020, 30, 1797–1805. [Google Scholar] [CrossRef]
- Elborn, J.S. Cystic Fibrosis. Lancet 2016, 388, 2519–2531. [Google Scholar] [CrossRef]
- Wark, P.A.B.; Tooze, M.; Cheese, L.; Whitehead, B.; Gibson, P.G.; Wark, K.F.; McDonald, V.M. Viral Infections Trigger Exacerbations of Cystic Fibrosis in Adults and Children. Eur. Respir. J. 2012, 40, 510–512. [Google Scholar] [CrossRef]
- Mathew, H.R.; Choi, M.Y.; Parkins, M.D.; Fritzler, M.J. Systematic Review: Cystic Fibrosis in the SARS-CoV-2/COVID-19 Pandemic. BMC Pulm. Med. 2021, 21, 173. [Google Scholar] [CrossRef]
- Cosgriff, R.; Ahern, S.; Bell, S.C.; Brownlee, K.; Burgel, P.-R.; Byrnes, C.; Corvol, H.; Cheng, S.Y.; Elbert, A.; Faro, A.; et al. A Multinational Report to Characterise SARS-CoV-2 Infection in People with Cystic Fibrosis. J. Cyst. Fibros. 2020, 19, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Kanner, L. Autistic Disturbances of Affective Contact. Nerv. Child 1943, 2, 217–250. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013; ISBN 978-0-89042-554-1. [Google Scholar]
- Khan, S.; Siddique, R.; Li, H.; Ali, A.; Shereen, M.A.; Bashir, N.; Xue, M. Impact of Coronavirus Outbreak on Psychological Health. J. Glob. Health 2020, 10, 010331. [Google Scholar] [CrossRef] [PubMed]
- Sanchack, K.E.; Thomas, C.A. Autism Spectrum Disorder: Primary Care Principles. Am. Fam. Physician 2016, 94, 972–979. [Google Scholar]
- Amorim, R.; Catarino, S.; Miragaia, P.; Ferreras, C.; Viana, V.; Guardiano, M. The Impact of COVID-19 on Children with Autism Spectrum Disorder. Rev. Neurol. 2020, 71, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Arunpriyandan, V.; Kumanan, S.; Pakkiyaretnam, M. First Case of Autoimmune Hemolytic Anemia Associated With COVID-19 Infection in Sri Lanka: A Case Report. Cureus 2021, 13, e19118. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.; Kim, J.; Pandey, A.; Huang, T.; DeLoughery, T.G. Simultaneous Onset of COVID-19 and Autoimmune Haemolytic Anaemia. Br. J. Haematol. 2020, 190, 31–32. [Google Scholar] [CrossRef]
- Lazarian, G.; Quinquenel, A.; Bellal, M.; Siavellis, J.; Jacquy, C.; Re, D.; Merabet, F.; Mekinian, A.; Braun, T.; Damaj, G.; et al. Autoimmune Haemolytic Anaemia Associated with COVID-19 Infection. Br. J. Haematol. 2020, 190, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, G.; de Mast, Q.; Markou, N.; Theodorakopoulou, M.; Komnos, A.; Mouktaroudi, M.; Netea, M.G.; Spyridopoulos, T.; Verheggen, R.J.; Hoogerwerf, J.; et al. Favorable Anakinra Responses in Severe Covid-19 Patients with Secondary Hemophagocytic Lymphohistiocytosis. Cell Host Microbe 2020, 28, 117–123.e1. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider Cytokine Storm Syndromes and Immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Schnaubelt, S.; Tihanyi, D.; Strassl, R.; Schmidt, R.; Anders, S.; Laggner, A.N.; Agis, H.; Domanovits, H. Hemophagocytic Lymphohistiocytosis in COVID-19: Case Reports of a Stepwise Approach. Medicine 2021, 100, e25170. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.B.; June, C.H. Cytokine Release Syndrome in Severe COVID-19. Science 2020, 368, 473–474. [Google Scholar] [CrossRef]
- Nisihara, R.M.; Skare, T.L.; Silva, M.B.G.; Messias-Reason, I.T.; Oliveira, N.P.; Fiedler, P.T.; Utiyama, S.R.R. High Positivity of Anti-CCP Antibodies in Patients with Down Syndrome. Clin. Rheumatol. 2007, 26, 2031–2035. [Google Scholar] [CrossRef]
- Colvin, K.L.; Yeager, M.E. What People with Down Syndrome Can Teach Us about Cardiopulmonary Disease. Eur. Respir. Rev. 2017, 26, 160098. [Google Scholar] [CrossRef]
- Englund, A.; Jonsson, B.; Zander, C.S.; Gustafsson, J.; Annerén, G. Changes in Mortality and Causes of Death in the Swedish Down Syndrome Population. Am. J. Med. Genet. Part A 2013, 161, 642–649. [Google Scholar] [CrossRef]
- Vita, S.; Di Bari, V.; Corpolongo, A.; Goletti, D.; Espinosa, J.; Petracca, S.; Palmieri, F.; Nicastri, E.; Abbonizio; Agrati, C.; et al. Down Syndrome Patients with COVID-19 Pneumonia: A High-Risk Category for Unfavourable Outcome. Int. J. Infect. Dis. 2021, 103, 607–610. [Google Scholar] [CrossRef]
- Epstein, C.J.; Korenberg, J.R.; Annerén, G.; Antonarakis, S.E.; Aymé, S.; Courchesne, E.; Epstein, L.B.; Fowler, A.; Groner, Y.; Huret, J.L. Protocols to Establish Genotype-Phenotype Correlations in Down Syndrome. Am. J. Hum. Genet. 1991, 49, 207–235. [Google Scholar] [PubMed]
- Picchianti Diamanti, A.; Rosado, M.M.; Pioli, C.; Sesti, G.; Laganà, B. Cytokine Release Syndrome in COVID-19 Patients, a New Scenario for an Old Concern: The Fragile Balance between Infections and Autoimmunity. Int. J. Mol. Sci. 2020, 21, 3330. [Google Scholar] [CrossRef] [PubMed]
- Tisoncik, J.R.; Korth, M.J.; Simmons, C.P.; Farrar, J.; Martin, T.R.; Katze, M.G. Into the Eye of the Cytokine Storm. Microbiol. Mol. Biol. Rev. 2012, 76, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Araya, P.; Waugh, K.A.; Sullivan, K.D.; Núñez, N.G.; Roselli, E.; Smith, K.P.; Granrath, R.E.; Rachubinski, A.L.; Enriquez Estrada, B.; Butcher, E.T.; et al. Trisomy 21 Dysregulates T Cell Lineages toward an Autoimmunity-Prone State Associated with Interferon Hyperactivity. Proc. Natl. Acad. Sci. USA 2019, 116, 24231–24241. [Google Scholar] [CrossRef]
- Zhang, Q.; Bastard, P.; Liu, Z.; Le Pen, J.; Moncada-Velez, M.; Chen, J.; Ogishi, M.; Sabli, I.K.D.; Hodeib, S.; Korol, C.; et al. Inborn Errors of Type I IFN Immunity in Patients with Life-Threatening COVID-19. Science 2020, 370, eabd4570. [Google Scholar] [CrossRef]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical Predictors of Mortality Due to COVID-19 Based on an Analysis of Data of 150 Patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef]
- Kantar, A.; Mazza, A.; Bonanomi, E.; Odoni, M.; Seminara, M.; Verde, I.D.; Lovati, C.; Bolognini, S.; D′Antiga, L. COVID-19 and Children with Down Syndrome: Is There Any Real Reason to Worry? Two Case Reports with Severe Course. BMC Pediatr. 2020, 20, 561. [Google Scholar] [CrossRef]
- Stockman, L.J.; Massoudi, M.S.; Helfand, R.; Erdman, D.; Siwek, A.M.; Anderson, L.J.; Parashar, U.D. Severe Acute Respiratory Syndrome in Children. Pediatr. Infect. Dis. J. 2007, 26, 68–74. [Google Scholar] [CrossRef]
- Assiri, A.; Al-Tawfiq, J.A.; Al-Rabeeah, A.A.; Al-Rabiah, F.A.; Al-Hajjar, S.; Al-Barrak, A.; Flemban, H.; Al-Nassir, W.N.; Balkhy, H.H.; Al-Hakeem, R.F.; et al. Epidemiological, Demographic, and Clinical Characteristics of 47 Cases of Middle East Respiratory Syndrome Coronavirus Disease from Saudi Arabia: A Descriptive Study. Lancet Infect. Dis. 2013, 13, 752–761. [Google Scholar] [CrossRef]
- Riphagen, S.; Gomez, X.; Gonzalez-Martinez, C.; Wilkinson, N.; Theocharis, P. Hyperinflammatory Shock in Children during COVID-19 Pandemic. Lancet 2020, 395, 1607–1608. [Google Scholar] [CrossRef]
- Verdoni, L.; Mazza, A.; Gervasoni, A.; Martelli, L.; Ruggeri, M.; Ciuffreda, M.; Bonanomi, E.; D′Antiga, L. An Outbreak of Severe Kawasaki-like Disease at the Italian Epicentre of the SARS-CoV-2 Epidemic: An Observational Cohort Study. Lancet 2020, 395, 1771–1778. [Google Scholar] [CrossRef]
- Cammarata-Scalisi, F.; Cárdenas Tadich, A.; Medina, M.; Callea, M. Trisomy 21 and the Coronavirus Disease 2019 (COVID-19). Arch. Argent. Pediatr. 2020, 118, 230–231. [Google Scholar] [CrossRef]
- Modell, B.; Darlison, M. Global Epidemiology of Haemoglobin Disorders and Derived Service Indicators. Bull. World Health Organ. 2008, 86, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Taher, A.T.; Weatherall, D.J.; Cappellini, M.D. Thalassaemia. Lancet 2018, 391, 155–167. [Google Scholar] [CrossRef]
- Motta, I.; Migone De Amicis, M.; Pinto, V.M.; Balocco, M.; Longo, F.; Bonetti, F.; Gianesin, B.; Graziadei, G.; Cappellini, M.D.; De Franceschi, L.; et al. SARS-CoV-2 Infection in Beta Thalassemia: Preliminary Data from the Italian Experience. Am. J. Hematol. 2020, 95, E198–E199. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.; Kim, L.; Whitaker, M.; O’Halloran, A.; Cummings, C.; Holstein, R.; Prill, M.; Chai, S.J.; Kirley, P.D.; Alden, N.B.; et al. Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019—COVID-NET, 14 States, March 1–30, 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 458–464. [Google Scholar] [CrossRef]
- Cappellini, M.D.; Fiorelli, G. Glucose-6-Phosphate Dehydrogenase Deficiency. Lancet 2008, 371, 64–74. [Google Scholar] [CrossRef]
- Wu, Y.-H.; Tseng, C.-P.; Cheng, M.-L.; Ho, H.-Y.; Shih, S.-R.; Chiu, D.T.-Y. Glucose-6-Phosphate Dehydrogenase Deficiency Enhances Human Coronavirus 229E Infection. J. Infect. Dis. 2008, 197, 812–816. [Google Scholar] [CrossRef]
- Lupescu, A.; Bissinger, R.; Goebel, T.; Salker, M.S.; Alzoubi, K.; Liu, G.; Chirigiu, L.; Mack, A.F.; Qadri, S.M.; Lang, F. Enhanced Suicidal Erythrocyte Death Contributing to Anemia in the Elderly. Cell. Physiol. Biochem. 2015, 36, 773–783. [Google Scholar] [CrossRef]
- De Franceschi, L.; Costa, E.; Dima, F.; Morandi, M.; Olivieri, O. Acute Hemolysis by Hydroxycloroquine Was Observed in G6PD-Deficient Patient with Severe COVD-19 Related Lung Injury. Eur. J. Intern. Med. 2020, 77, 136–137. [Google Scholar] [CrossRef] [PubMed]
- Iaboni, A.; Wong, N.; Betschel, S.D. A Patient with X-Linked Agammaglobulinemia and COVID-19 Infection Treated with Remdesivir and Convalescent Plasma. J. Clin. Immunol. 2021, 41, 923–925. [Google Scholar] [CrossRef]
- Quinti, I.; Lougaris, V.; Milito, C.; Cinetto, F.; Pecoraro, A.; Mezzaroma, I.; Mastroianni, C.M.; Turriziani, O.; Bondioni, M.P.; Filippini, M.; et al. A Possible Role for B Cells in COVID-19? Lesson from Patients with Agammaglobulinemia. J. Allergy Clin. Immunol. 2020, 146, 211–213.e4. [Google Scholar] [CrossRef]
- Devassikutty, F.M.; Jain, A.; Edavazhippurath, A.; Joseph, M.C.; Peedikayil, M.M.T.; Scaria, V.; Sandhya, P.; Govindaraj, G.M. X-Linked Agammaglobulinemia and COVID-19: Two Case Reports and Review of Literature. Pediatr. Allergy Immunol. Pulmonol. 2021, 34, 115–118. [Google Scholar] [CrossRef]
- Kyrou, I.; Karteris, E.; Robbins, T.; Chatha, K.; Drenos, F.; Randeva, H.S. Polycystic Ovary Syndrome (PCOS) and COVID-19: An Overlooked Female Patient Population at Potentially Higher Risk during the COVID-19 Pandemic. BMC Med. 2020, 18, 220. [Google Scholar] [CrossRef]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J. International PCOS Network Recommendations from the International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. Hum. Reprod. 2018, 33, 1602–1618. [Google Scholar] [CrossRef] [PubMed]
- Asunción, M.; Calvo, R.M.; San Millán, J.L.; Sancho, J.; Avila, S.; Escobar-Morreale, H.F. A Prospective Study of the Prevalence of the Polycystic Ovary Syndrome in Unselected Caucasian Women from Spain. J. Clin. Endocrinol. Metab. 2000, 85, 2434–2438. [Google Scholar] [CrossRef]
- Joshi, S.R. Polycystic Ovary Syndrome and COVID-19: An Emerging Risk Factor. J. Hum. Reprod. Sci. 2021, 14, 211–212. [Google Scholar] [CrossRef]
- Subramanian, A.; Anand, A.; Adderley, N.J.; Okoth, K.; Toulis, K.A.; Gokhale, K.; Sainsbury, C.; O’Reilly, M.W.; Arlt, W.; Nirantharakumar, K. Increased COVID-19 Infections in Women with Polycystic Ovary Syndrome: A Population-Based Study. Eur. J. Endocrinol. 2021, 184, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Busse, P.J.; Christiansen, S.C. Hereditary Angioedema. N. Engl. J. Med. 2020, 382, 1136–1148. [Google Scholar] [CrossRef]
- Roche, J.A.; Roche, R. A Hypothesized Role for Dysregulated Bradykinin Signaling in COVID-19 Respiratory Complications. FASEB J. 2020, 34, 7265–7269. [Google Scholar] [CrossRef] [PubMed]
- Veronez, C.L.; Christiansen, S.C.; Smith, T.D.; Riedl, M.A.; Zuraw, B.L. COVID-19 and Hereditary Angioedema: Incidence, Outcomes, and Mechanistic Implications. Allergy Asthma Proc. 2021, 42, 506–514. [Google Scholar] [CrossRef]
- Feinberg, J.H.; Radecki, J. Parsonage-Turner Syndrome. HSS J. 2010, 6, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Fibuch, E.E.; Mertz, J.; Geller, B. Postoperative Onset of Idiopathic Brachial Neuritis. Anesthesiology 1996, 84, 455–458. [Google Scholar] [CrossRef]
- Queler, S.C.; Towbin, A.J.; Milani, C.; Whang, J.; Sneag, D.B. Parsonage-Turner Syndrome Following COVID-19 Vaccination: MR Neurography. Radiology 2022, 302, 84–87. [Google Scholar] [CrossRef]
- Mitry, M.A.; Collins, L.K.; Kazam, J.J.; Kaicker, S.; Kovanlikaya, A. Parsonage-Turner Syndrome Associated with SARS-CoV2 (COVID-19) Infection. Clin. Imaging 2021, 72, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Voss, T.G.; Stewart, C.M. Case Report: Parsonage-Turner Syndrome after COVID-19 Infection. JSES Rev. Rep. Tech. 2022. [Google Scholar] [CrossRef]
- Cassidy, S.B.; Schwartz, S.; Miller, J.L.; Driscoll, D.J. Prader-Willi Syndrome. Genet. Med. 2012, 14, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ashcroft, T.; Chung, A.; Dighero, I.; Dozier, M.; Horne, M.; McSwiggan, E.; Shamsuddin, A.; Nair, H. Risk Factors for Poor Outcomes in Hospitalised COVID-19 Patients: A Systematic Review and Meta-Analysis. J. Glob. Health 2021, 11, 10001. [Google Scholar] [CrossRef] [PubMed]
- Bar, C.; Diene, G.; Molinas, C.; Bieth, E.; Casper, C.; Tauber, M. Early Diagnosis and Care Is Achieved but Should Be Improved in Infants with Prader-Willi Syndrome. Orphanet J. Rare Dis. 2017, 12, 118. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Formoso, G.; Pugliese, G.; Ruggeri, R.M.; Scarano, E.; Colao, A.; on behalf of RESTARE. Prader- Willi Syndrome: An Uptodate on Endocrine and Metabolic Complications. Rev. Endocr. Metab. Disord. 2019, 20, 239–250. [Google Scholar] [CrossRef]
- Coupaye, M.; Laurier, V.; Benvegnu, G.; Poitou, C.; Faucher, P.; Mosbah, H.; Diene, G.; Pinto, G.; Briceño, L.G.; Merrien, C.; et al. Paradoxical Low Severity of COVID-19 in Prader-Willi Syndrome: Data from a French Survey on 647 Patients. Orphanet J. Rare Dis. 2021, 16, 325. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Wang, Y.-F. Cardiovascular Protective Properties of Oxytocin against COVID-19. Life Sci. 2021, 270, 119130. [Google Scholar] [CrossRef]
- Charoenngam, N.; Shirvani, A.; Reddy, N.; Vodopivec, D.M.; Apovian, C.M.; Holick, M.F. Association of Vitamin D Status With Hospital Morbidity and Mortality in Adult Hospitalized Patients With COVID-19. Endocr. Pract. 2021, 27, 271–278. [Google Scholar] [CrossRef]
- Liu, N.; Sun, J.; Wang, X.; Zhang, T.; Zhao, M.; Li, H. Low Vitamin D Status Is Associated with Coronavirus Disease 2019 Outcomes: A Systematic Review and Meta-Analysis. Int. J. Infect. Dis 2021, 104, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Hoertel, N.; Sánchez-Rico, M.; Vernet, R.; Beeker, N.; Jannot, A.-S.; Neuraz, A.; Salamanca, E.; Paris, N.; Daniel, C.; Gramfort, A.; et al. Association between Antidepressant Use and Reduced Risk of Intubation or Death in Hospitalized Patients with COVID-19: Results from an Observational Study. Mol. Psychiatry 2021, 26, 5199–5212. [Google Scholar] [CrossRef]
- Lukito, A.A.; Pranata, R.; Henrina, J.; Lim, M.A.; Lawrensia, S.; Suastika, K. The Effect of Metformin Consumption on Mortality in Hospitalized COVID-19 Patients: A Systematic Review and Meta-Analysis. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 2177–2183. [Google Scholar] [CrossRef]
- Hariyanto, T.I.; Kurniawan, A. Metformin Use Is Associated with Reduced Mortality Rate from Coronavirus Disease 2019 (COVID-19) Infection. Obes. Med. 2020, 19, 100290. [Google Scholar] [CrossRef]
- Kow, C.S.; Hasan, S.S. Mortality Risk with Preadmission Metformin Use in Patients with COVID-19 and Diabetes: A Meta-Analysis. J. Med. Virol. 2021, 93, 695–697. [Google Scholar] [CrossRef]
- Platt, F.M.; d′Azzo, A.; Davidson, B.L.; Neufeld, E.F.; Tifft, C.J. Lysosomal Storage Diseases. Nat. Rev. Dis. Primers 2018, 4, 27. [Google Scholar] [CrossRef]
- Kristal, E.; Pode-Shakked, B.; Hazan, G.; Banne, E.; Ling, G.; David, O.; Shany, E.; Raas-Rothschild, A.; Anikster, Y.; Kneller, K.; et al. The Effects of the COVID-19 Pandemic on Patients with Lysosomal Storage Disorders in Israel. Orphanet J. Rare Dis. 2021, 16, 379. [Google Scholar] [CrossRef]
- Buhrman, D.; Thakkar, K.; Poe, M.; Escolar, M.L. Natural History of Sanfilippo Syndrome Type A. J. Inherit. Metab. Dis. 2014, 37, 431–437. [Google Scholar] [CrossRef]
- Delgadillo, V.; O′Callaghan, M.; del, M.; Gort, L.; Coll, M.J.; Pineda, M. Natural History of Sanfilippo Syndrome in Spain. Orphanet J. Rare Dis. 2013, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Ciepela, C. Old and New Insights into the Diagnosis of Hereditary Spherocytosis. Ann. Transl. Med. 2018, 6, 339. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lu, L.; Li, X.; Buffet, P.A.; Dao, M.; Karniadakis, G.E.; Suresh, S. Mechanics of Diseased Red Blood Cells in Human Spleen and Consequences for Hereditary Blood Disorders. Proc. Natl. Acad. Sci. USA 2018, 115, 9574–9579. [Google Scholar] [CrossRef]
- Severance, T.S.; Rahim, M.Q.; French II, J.; Baker, R.M.; Shriner, A.; Khaitan, A.; Overholt, K.M. COVID-19 and Hereditary Spherocytosis: A Recipe for Hemolysis. Pediatr. Blood Cancer 2021, 68, e28548. [Google Scholar] [CrossRef]
- Tateno, Y.; Suzuki, R.; Kitamura, Y. Previously Undiagnosed Hereditary Spherocytosis in a Patient with Jaundice and Pyelonephritis: A Case Report. J. Med. Case Rep. 2016, 10, 337. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marchetti, D.; Fontanesi, L.; Mazza, C.; Di Giandomenico, S.; Roma, P.; Verrocchio, M.C. Parenting-Related Exhaustion During the Italian COVID-19 Lockdown. J. Pediatr. Psychol. 2020, 45, 1114–1123. [Google Scholar] [CrossRef] [PubMed]
- Copp, A.J.; Adzick, N.S.; Chitty, L.S.; Fletcher, J.M.; Holmbeck, G.N.; Shaw, G.M. Spina Bifida. Nat. Rev. Dis. Primers 2015, 1, 15007. [Google Scholar] [CrossRef]
- Plevinsky, J.M.; Young, M.A.; Carmody, J.K.; Durkin, L.K.; Gamwell, K.L.; Klages, K.L.; Ghosh, S.; Hommel, K.A. The Impact of COVID-19 on Pediatric Adherence and Self-Management. J. Pediatr. Psychol. 2020, 45, 977–982. [Google Scholar] [CrossRef]
- Psihogios, A.M.; Stiles-Shields, C.; Neary, M. The Needle in the Haystack: Identifying Credible Mobile Health Apps for Pediatric Populations during a Pandemic and Beyond. J. Pediatr. Psychol. 2020, 45, 1106–1113. [Google Scholar] [CrossRef]
- Hiraoka, D.; Tomoda, A. Relationship between Parenting Stress and School Closures Due to the COVID-19 Pandemic. Psychiatry Clin. Neurosci. 2020, 74, 497–498. [Google Scholar] [CrossRef]
- Kazak, A.E.; Alderfer, M.; Enlow, P.T.; Lewis, A.M.; Vega, G.; Barakat, L.; Kassam-Adams, N.; Pai, A.; Canter, K.S.; Hildenbrand, A.K.; et al. COVID-19 Exposure and Family Impact Scales: Factor Structure and Initial Psychometrics. J. Pediatr. Psychol. 2021, 46, 504–513. [Google Scholar] [CrossRef]
- Stiles-Shields, C.; Kritikos, T.K.; Ridosh, M.M.; Starnes, M.; Holmbeck, G.N. “We Are Anxious Every Day”: COVID-19 Impacts on Youth with Spina Bifida. J. Pediatr. Psychol. 2021, 46, 1040–1050. [Google Scholar] [CrossRef]
- Puelles, V.G.; Lütgehetmann, M.; Lindenmeyer, M.T.; Sperhake, J.P.; Wong, M.N.; Allweiss, L.; Chilla, S.; Heinemann, A.; Wanner, N.; Liu, S.; et al. Multiorgan and Renal Tropism of SARS-CoV-2. N. Engl. J. Med. 2020, 383, 590–592. [Google Scholar] [CrossRef] [PubMed]
- Lui, D.T.W.; Lee, C.H.; Chow, W.S.; Lee, A.C.H.; Tam, A.R.; Fong, C.H.Y.; Law, C.Y.; Leung, E.K.H.; To, K.K.W.; Tan, K.C.B.; et al. Thyroid Dysfunction in Relation to Immune Profile, Disease Status and Outcome in 191 Patients with COVID-19. J. Clin. Endocrinol. Metab. 2020, 106, e926–e935. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, R.J.; Berry-Kravis, E.; Hazlett, H.C.; Bailey, D.B.; Moine, H.; Kooy, R.F.; Tassone, F.; Gantois, I.; Sonenberg, N.; Mandel, J.L.; et al. Fragile X Syndrome. Nat. Rev. Dis. Primers 2017, 3, 17065. [Google Scholar] [CrossRef]
- Niu, M.; Han, Y.; Dy, A.B.C.; Du, J.; Jin, H.; Qin, J.; Zhang, J.; Li, Q.; Hagerman, R.J. Fragile X Syndrome: Prevalence, Treatment, and Prevention in China. Front. Neurol. 2017, 8, 254. [Google Scholar] [CrossRef] [PubMed]
- Careaga, M.; Rose, D.; Tassone, F.; Berman, R.F.; Hagerman, R.; Ashwood, P. Immune Dysregulation as a Cause of Autoinflammation in Fragile X Premutation Carriers: Link between FMRI CGG Repeat Number and Decreased Cytokine Responses. PLoS ONE 2014, 9, e94475. [Google Scholar] [CrossRef]
- Zhang, N.; Zhao, Y.D.; Wang, X.M. CXCL10 an Important Chemokine Associated with Cytokine Storm in COVID-19 Infected Patients. Eur. Rev. 2020, 24, 7497–7505. [Google Scholar]
- Kleiman, J.D.; Veerapaneni, K.; Escovar, J.; Orsini, J. COVID-19 Infection in a Patient With Fragile-X Syndrome. Cureus 2020, 12, e11266. [Google Scholar] [CrossRef]
- Van Dijck, A.; Barbosa, S.; Bermudez-Martin, P.; Khalfallah, O.; Gilet, C.; Martinuzzi, E.; Elinck, E.; Kooy, R.F.; Glaichenhaus, N.; Davidovic, L. Reduced Serum Levels of Pro-Inflammatory Chemokines in Fragile X Syndrome. BMC Neurol. 2020, 20, 138. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.-S.; Shu, T.; Kang, L.; Wu, D.; Zhou, X.; Liao, B.-W.; Sun, X.-L.; Zhou, X.; Wang, Y.-Y. Temporal Profiling of Plasma Cytokines, Chemokines and Growth Factors from Mild, Severe and Fatal COVID-19 Patients. Signal Transduct. Target. Ther. 2020, 5, 100. [Google Scholar] [CrossRef]
- Channappanavar, R.; Perlman, S. Pathogenic Human Coronavirus Infections: Causes and Consequences of Cytokine Storm and Immunopathology. Semin. Immunopathol. 2017, 39, 529–539. [Google Scholar] [CrossRef]
- Ruscitti, P.; Berardicurti, O.; Iagnocco, A.; Giacomelli, R. Cytokine Storm Syndrome in Severe COVID-19. Autoimmun. Rev. 2020, 19, 102562. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical Features of Patients Infected with 2019 Novel Coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Mah, J.K.; Korngut, L.; Dykeman, J.; Day, L.; Pringsheim, T.; Jette, N. A Systematic Review and Meta-Analysis on the Epidemiology of Duchenne and Becker Muscular Dystrophy. Neuromuscul. Disord. 2014, 24, 482–491. [Google Scholar] [CrossRef]
- Hoffman, E.P.; Fischbeck, K.H.; Brown, R.H.; Johnson, M.; Medori, R.; Loike, J.D.; Harris, J.B.; Waterston, R.; Brooke, M.; Specht, L. Characterization of Dystrophin in Muscle-Biopsy Specimens from Patients with Duchenne’s or Becker’s Muscular Dystrophy. N. Engl. J. Med. 1988, 318, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.M.; Abresch, R.T.; Carter, G.T.; Fowler, W.M.; Johnson, E.R.; Kilmer, D.D.; Sigford, B.J. Profiles of Neuromuscular Diseases. Duchenne Muscular Dystrophy. Am. J. Phys. Med. Rehabil. 1995, 74, S70–S92. [Google Scholar] [CrossRef]
- Finder, J.D.; Birnkrant, D.; Carl, J.; Farber, H.J.; Gozal, D.; Iannaccone, S.T.; Kovesi, T.; Kravitz, R.M.; Panitch, H.; Schramm, C.; et al. Respiratory Care of the Patient with Duchenne Muscular Dystrophy: ATS Consensus Statement. Am. J. Respir. Crit. Care Med. 2004, 170, 456–465. [Google Scholar] [CrossRef]
- Bello, L.; Gordish-Dressman, H.; Morgenroth, L.P.; Henricson, E.K.; Duong, T.; Hoffman, E.P.; Cnaan, A.; McDonald, C.M. Prednisone/Prednisolone and Deflazacort Regimens in the CINRG Duchenne Natural History Study. Neurology 2015, 85, 1048–1055. [Google Scholar] [CrossRef]
- McDonald, C.M.; Gordish-Dressman, H.; Henricson, E.K.; Duong, T.; Joyce, N.C.; Jhawar, S.; Leinonen, M.; Hsu, F.; Connolly, A.M.; Cnaan, A.; et al. Longitudinal Pulmonary Function Testing Outcome Measures in Duchenne Muscular Dystrophy: Long-Term Natural History with and without Glucocorticoids. Neuromuscul. Disord. 2018, 28, 897–909. [Google Scholar] [CrossRef]
- McDonald, C.M.; Henricson, E.K.; Abresch, R.T.; Han, J.J.; Escolar, D.M.; Florence, J.M.; Duong, T.; Arrieta, A.; Clemens, P.R.; Hoffman, E.P.; et al. The Cooperative International Neuromuscular Research Group Duchenne Natural History Study—A Longitudinal Investigation In The Era Of Glucocorticoid Therapy: Design Of Protocol And The Methods Used. Muscle Nerve 2013, 48, 32–54. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Prais, D.; Aharoni, S.; Nevo, Y.; Katz, J.; Rahmani, E.; Goldberg, L.; Scheuerman, O. COVID-19 in Advanced Duchenne/Becker Muscular Dystrophy Patients. Neuromuscul. Disord. 2021, 31, 607–611. [Google Scholar] [CrossRef]
- Huda, S.; Whittam, D.; Bhojak, M.; Chamberlain, J.; Noonan, C.; Jacob, A.; Kneen, R. Neuromyelitis Optica Spectrum Disorders. Clin. Med. 2019, 19, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Lennon, V.A.; Wingerchuk, D.M.; Kryzer, T.J.; Pittock, S.J.; Lucchinetti, C.F.; Fujihara, K.; Nakashima, I.; Weinshenker, B.G. A Serum Autoantibody Marker of Neuromyelitis Optica: Distinction from Multiple Sclerosis. Lancet 2004, 364, 2106–2112. [Google Scholar] [CrossRef]
- Lennon, V.A.; Kryzer, T.J.; Pittock, S.J.; Verkman, A.S.; Hinson, S.R. IgG Marker of Optic-Spinal Multiple Sclerosis Binds to the Aquaporin-4 Water Channel. J. Exp. Med. 2005, 202, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Matiello, M.; Kim, H.J.; Kim, W.; Brum, D.G.; Barreira, A.A.; Kingsbury, D.J.; Plant, G.T.; Adoni, T.; Weinshenker, B.G. Familial Neuromyelitis Optica. Neurology 2010, 75, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Estrada, K.; Whelan, C.W.; Zhao, F.; Bronson, P.; Handsaker, R.E.; Sun, C.; Carulli, J.P.; Harris, T.; Ransohoff, R.M.; McCarroll, S.A.; et al. A Whole-Genome Sequence Study Identifies Genetic Risk Factors for Neuromyelitis Optica. Nat. Commun. 2018, 9, 1929. [Google Scholar] [CrossRef] [PubMed]
- Pittock, S.J.; Lennon, V.A.; de Seze, J.; Vermersch, P.; Homburger, H.A.; Wingerchuk, D.M.; Lucchinetti, C.F.; Zéphir, H.; Moder, K.; Weinshenker, B.G. Neuromyelitis Optica and Non Organ-Specific Autoimmunity. Arch. Neurol. 2008, 65, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.; Zarei, M.; Kenton, A.; Allroggen, H. Gluten Sensitivity and Neuromyelitis Optica: Two Case Reports. J. Neurol. Neurosurg. Psychiatry 2005, 76, 1028–1030. [Google Scholar] [CrossRef]
- Leite, M.I.; Coutinho, E.; Lana-Peixoto, M.; Apostolos, S.; Waters, P.; Sato, D.; Melamud, L.; Marta, M.; Graham, A.; Spillane, J.; et al. Myasthenia Gravis and Neuromyelitis Optica Spectrum Disorder. Neurology 2012, 78, 1601–1607. [Google Scholar] [CrossRef] [PubMed]
- Paybast, S.; Shahrab, F.; Hejazi, S.A. Recurrence of COVID-19 in a Patient With NMO Spectrum Disorder While Treating With Rituximab A Case Report and Review of the Literature. Neurologist 2021, 26, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, E.P. Neuromyelitis Optica Spectrum Disorder and Other Non-Multiple Sclerosis Central Nervous System Inflammatory Diseases. Continuum 2019, 25, 815–844. [Google Scholar] [CrossRef] [PubMed]
- Carnero Contentti, E.; Correa, J. Immunosuppression during the COVID-19 Pandemic in Neuromyelitis Optica Spectrum Disorders Patients: A New Challenge. Mult. Scler. Relat. Disord. 2020, 41, 102097. [Google Scholar] [CrossRef]
- Brownlee, W.; Bourdette, D.; Broadley, S.; Killestein, J.; Ciccarelli, O. Treating Multiple Sclerosis and Neuromyelitis Optica Spectrum Disorder during the COVID-19 Pandemic. Neurology 2020, 94, 949–952. [Google Scholar] [CrossRef] [PubMed]
- Thakolwiboon, S.; Zhao-Fleming, H.; Pan, J.; Scott, J.K.; Shoji, E.; Sohn, G.; Avila, M. Disease-Modifying Therapies During the COVID-19 Outbreak. Int. J. MS Care 2020, 22, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Niemi, M.E.K.; Karjalainen, J.; Liao, R.G.; Neale, B.M.; Daly, M.; Ganna, A.; Pathak, G.A.; Andrews, S.J.; Kanai, M.; Veerapen, K.; et al. Mapping the Human Genetic Architecture of COVID-19. Nature 2021, 600, 472–477. [Google Scholar] [CrossRef]
- Mousa, M.; Vurivi, H.; Kannout, H.; Uddin, M.; Alkaabi, N.; Mahboub, B.; Tay, G.K.; Alsafar, H.S.; UAE COVID-19 Collaborative Partnership. Genome-Wide Association Study of Hospitalized COVID-19 Patients in the United Arab Emirates. EBioMedicine 2021, 74, 103695. [Google Scholar] [CrossRef] [PubMed]
- Fallerini, C.; Picchiotti, N.; Baldassarri, M.; Zguro, K.; Daga, S.; Fava, F.; Benetti, E.; Amitrano, S.; Bruttini, M.; Palmieri, M.; et al. Common, Low-Frequency, Rare, and Ultra-Rare Coding Variants Contribute to COVID-19 Severity. Hum. Genet. 2021, 141, 147–173. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Richards, A.; Barrasa, M.I.; Hughes, S.H.; Young, R.A.; Jaenisch, R. Reverse-Transcribed SARS-CoV-2 RNA Can Integrate into the Genome of Cultured Human Cells and Can Be Expressed in Patient-Derived Tissues. Proc. Natl. Acad. Sci. USA 2021, 118, e2105968118. [Google Scholar] [CrossRef]
- He, Y.; Wang, J.; Ren, J.; Zhao, Y.; Chen, J.; Chen, X. Effect of COVID-19 on Male Reproductive System—A Systematic Review. Front. Endocrinol. 2021, 12, 541. [Google Scholar] [CrossRef]
- Ding, T.; Wang, T.; Zhang, J.; Cui, P.; Chen, Z.; Zhou, S.; Yuan, S.; Ma, W.; Zhang, M.; Rong, Y.; et al. Analysis of Ovarian Injury Associated With COVID-19 Disease in Reproductive-Aged Women in Wuhan, China: An Observational Study. Front. Med. 2021, 8, 286. [Google Scholar] [CrossRef] [PubMed]
- Mali, A.S.; Magdum, M.; Novotny, J. COVID-19 Impact on Reproduction and Fertility. JBRA Assist. Reprod. 2021, 25, 310–313. [Google Scholar] [CrossRef]
- Freitas, D.A.; Souza-Santos, R.; Carvalho, L.M.A.; Barros, W.B.; Neves, L.M.; Brasil, P.; Wakimoto, M.D. Congenital Zika Syndrome: A Systematic Review. PLoS ONE 2020, 15, e0242367. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Guida, J.P.; Costa, M.L.; Mysorekar, I.U. Host and Viral Mechanisms of Congenital Zika Syndrome. Virulence 2019, 10, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Justino, C.C.; Campanharo, F.F.; Augusto, M.N.; de Morais, S.C.; Figueiredo, M.S. COVID-19 as a Trigger of Acute Chest Syndrome in a Pregnant Woman with Sickle Cell Anemia. Hematol. Transfus. Cell Ther. 2020, 42, 212–214. [Google Scholar] [CrossRef] [PubMed]
- Kehinde, T.A.; Osundiji, M.A. Sickle Cell Trait and the Potential Risk of Severe Coronavirus Disease 2019-A Mini-Review. Eur. J. Haematol. 2020, 105, 519–523. [Google Scholar] [CrossRef]
- Balanchivadze, N.; Kudirka, A.A.; Askar, S.; Almadhoun, K.; Kuriakose, P.; Fadel, R.; Dabak, V. Impact of COVID-19 Infection on 24 Patients with Sickle Cell Disease. One Center Urban Experience, Detroit, MI, USA. Hemoglobin 2020, 44, 284–289. [Google Scholar] [CrossRef]
- Waghmare, R.; Chaaithanya, I.K.; Zala, S.; Deshmukh, J.; Uikey, P.; Wankhede, S.; Palve, T.; Sirsam, S.; Chavan, R.; Waikar, M.; et al. Outcomes of COVID-19 in Pregnant Women with Sickle Cell Disease in India: A Case Series. Indian J. Hematol. Blood Transfus. 2021, 38, 191–193. [Google Scholar] [CrossRef]
- Marhaeni, W.; Wijaya, A.B.; Khairiyadi; Munawaroh; Hendriyono. Coagulation Abnormalities Due to COVID-19 in a Child with Thalassemia. Indian J. Pediatr. 2021, 88, 396–397. [Google Scholar] [CrossRef]
- Pinto, V.M.; Derchi, G.E.; Bacigalupo, L.; Pontali, E.; Forni, G.L. COVID-19 in a Patient with β-Thalassemia Major and Severe Pulmonary Arterial Hypertension. Hemoglobin 2020, 44, 218–220. [Google Scholar] [CrossRef]
- Marziali, M.; Ribersani, M.; Losardo, A.A.; Taglietti, F.; Pugliese, P.; Micozzi, A.; Gentile, G.; Angeloni, A. COVID-19 Pneumonia and Pulmonary Microembolism in a Patient with B-Thalassemia Major. Clin. Case Rep. 2020, 8, 3138–3141. [Google Scholar] [CrossRef]
- Frontiers|SAMHD1 as the Potential Link between SARS-CoV-2 Infection and Neurological Complications|Neurology. Available online: https://www.frontiersin.org/articles/10.3389/fneur.2020.562913/full (accessed on 3 March 2022).
- Pararajasingam, A.; Goodwin, R. Generalised Panniculitis as a Post-COVID-19 Presentation in a Child with Aicardi-Goutières Syndrome. Pediatr. Dermatol. 2020, 37, 14. [Google Scholar]
- Emes, D.; Hüls, A.; Baumer, N.; Dierssen, M.; Puri, S.; Russell, L.; Sherman, S.L.; Strydom, A.; Bargagna, S.; Brandão, A.C.; et al. COVID-19 in Children with Down Syndrome: Data from the Trisomy 21 Research Society Survey. J. Clin. Med. 2021, 10, 5125. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Haghpanah, S.; Shahsavani, A. Prevalence and Clinical Features of COVID-19 in Iranian Patients with Congenital Coagulation Disorders. Blood Transfus. 2020, 18, 413–414. [Google Scholar] [CrossRef] [PubMed]
- Dorgalaleh, A.; Dabbagh, A.; Tabibian, S.; Baghaeipour, M.R.; Jazebi, M.; Bahraini, M.; Fazeli, S.; Rad, F.; Baghaeipour, N. Patients with Congenital Bleeding Disorders Appear to Be Less Severely Affected by SARS-CoV-2: Is Inherited Hypocoagulability Overcoming Acquired Hypercoagulability of Coronavirus Disease 2019 (COVID-19)? Semin. Thromb. Hemost. 2020, 46, 853–855. [Google Scholar] [CrossRef] [PubMed]
- Lovisetto, F.; Guala, A.; Facciotto, G.; Zonta, S. COVID-19 and Psychiatric Illness: Rectal Foreign Bodies (30 Stylus Batteries) in a Young Male Patient Affected by Asperger Syndrome. J. Surg. Case Rep. 2020, 2020, rjaa345. [Google Scholar] [CrossRef]
- Valenza, M.; Steardo, L.; Steardo, L.; Verkhratsky, A.; Scuderi, C. Systemic Inflammation and Astrocyte Reactivity in the Neuropsychiatric Sequelae of COVID-19: Focus on Autism Spectrum Disorders. Front. Cell. Neurosci. 2021, 15, 748136. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, T.R.; Prasad, S.; Munzer, T.; Laventhal, N. The Impact of the COVID-19 Pandemic on Children with Autism Spectrum Disorders. J. Pediatr. Rehabil. Med. 2020, 13, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Citoni, B.; Digilio, M.C.; Capolino, R.; Gagliardi, M.G.; Campana, A.; Drago, F.; Calcagni, G. SARS-CoV-2 and Pre-Tamponade Pericardial Effusion. Could Sotos Syndrome Be a Major Risk Factor? Genes 2021, 12, 1782. [Google Scholar] [CrossRef] [PubMed]
- Natera-de Benito, D.; Aguilera-Albesa, S.; Costa-Comellas, L.; García-Romero, M.; Miranda-Herrero, M.C.; Rúbies Olives, J.; García-Campos, Ó.; Martínez del Val, E.; Martinez Garcia, M.J.; Medina Martínez, I.; et al. COVID-19 in Children with Neuromuscular Disorders. J. Neurol. 2021, 268, 3081–3085. [Google Scholar] [CrossRef] [PubMed]
- Burgel, P.-R.; Goss, C. COVID-19 Outcomes in People with Cystic Fibrosis. Curr. Opin. Pulm. Med. 2021, 27, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Abdelzaher, M.A.; Ibrahim, A.E.S.; Negm, E.M. Increasing Fetal Hemoglobin as a Possible Key for Improvement of Hypoxia and Saving Last Breath in COVID-19 Patient: “Postulating a Hypothesis”. Egypt. J. Bronchol. 2021, 15, 33. [Google Scholar] [CrossRef]
- Vervaat, F.E.; Houthuizen, P. Case Report of SARS Co-V2 Infection, Acute Pulmonary Embolism, and Right Ventricular Thrombus. Eur. Heart J. Case Rep. 2020, 4, 1–5. [Google Scholar] [CrossRef]
- Focosi, D.; Franchini, M. COVID-19 Neutralizing Antibody-Based Therapies in Humoral Immune Deficiencies: A Narrative Review. Transfus. Apher. Sci. 2021, 60, 103071. [Google Scholar] [CrossRef]
- Tholin, B.; Hauge, M.T.; Aukrust, P.; Fehrle, L.; Tvedt, T.H. Hemophagocytic Lymphohistiocytosis in a Patient with COVID-19 Treated with Tocilizumab: A Case Report. J. Med. Case Rep. 2020, 14, 187. [Google Scholar] [CrossRef] [PubMed]
- von der Thüsen, J.H.; van Bommel, J.; Kros, J.M.; Verdijk, R.M.; Lopuhaä, B.; Lam, K.H.; Dik, W.A.; Miedema, J.R. Case Report: A Fatal Combination of Hemophagocytic Lymphohistiocytosis with Extensive Pulmonary Microvascular Damage in COVID-19 Pneumonia. J. Hematop. 2020, 14, 79–83. [Google Scholar] [CrossRef]
- Shuffrey, L.C.; Firestein, M.R.; Kyle, M.H.; Fields, A.; Alcántara, C.; Amso, D.; Austin, J.; Bain, J.M.; Barbosa, J.; Bence, M.; et al. Association of Birth During the COVID-19 Pandemic with Neurodevelopmental Status at 6 Months in Infants with and without In Utero Exposure to Maternal SARS-CoV-2 Infection. JAMA Pediatr. 2022, e215563. [Google Scholar] [CrossRef]
- Leyser, M.; Marques, F.J.P.; do Nascimento, O.J.M. Potential Risk Of Brain Damage And Poor Developmental Outcomes In Children Prenatally Exposed To Sars-Cov-2: A Systematic Review. Rev. Paul. Pediatr. 2020, 40, e2020415. [Google Scholar] [CrossRef]
- Engert, V.; Siauw, C.; Stock, A.; Rehn, M.; Wöckel, A.; Härtel, C.; Wirbelauer, J. Severe Brain Damage in a Moderate Preterm Infant as Complication of Post-COVID-19 Response during Pregnancy. NEO 2021, 118, 505–508. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Chapman, K.R.; Wong, A.; Liu, M. A1-Antitrypsin Deficiency and the Risk of COVID-19: An Urgent Call to Action. Lancet Respir. Med. 2021, 9, 337–339. [Google Scholar] [CrossRef]
- Della-Morte, D.; Pacifici, F.; Ricordi, C.; Massoud, R.; Rovella, V.; Proietti, S.; Iozzo, M.; Lauro, D.; Bernardini, S.; Bonassi, S.; et al. Low Level of Plasminogen Increases Risk for Mortality in COVID-19 Patients. Cell Death Dis. 2021, 12, 773. [Google Scholar] [CrossRef] [PubMed]
- Morhart, P.; Mardin, C.; Rauh, M.; Jüngert, J.; Hammersen, J.; Kehl, S.; Schuh, W.; Maier-Wohlfart, S.; Hermes, K.; Neubert, A.; et al. Maternal SARS-CoV-2 Infection during Pregnancy: Possible Impact on the Infant. Eur. J. Pediatr. 2022, 181, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.Q.; Mendonca, B.B. Adrenal Insufficiency and Glucocorticoid Use during the COVID-19 Pandemic. Clinics 2020, 75, e2022. [Google Scholar] [CrossRef] [PubMed]
- dos Santos Junior, V.E.; da Silva, J.V.F.; de Lima, F.J.C.; Borges, C.D.A.; Vieira, A.E.; Silva, L.C. Clinical and Molecular Disorders Caused by COVID-19 During Pregnancy as a Potential Risk for Enamel Defects. Pesqui. Bras. Odontopediatr. Clín. Integr. 2021, 21. [Google Scholar] [CrossRef]
- Donders, H.C.M.; van der Sleen, J.M.; Kleinbergen, Y.J.; Su, N.; de Lange, J.; Loos, B.G. Alveolar Bone Loss and Tooth Loss Are Associated with COVID-19 Severity but Are Not Independent Risk Factors. An Explorative Study. Adv. Oral Maxillofac. Surg. 2022, 5, 100223. [Google Scholar] [CrossRef]
- Barberá-Pérez, P.M.; Baquedano Lobera, I.; Paúl-Vidaller, P.J. Haemolytic Anemia Triggered by SARS-CoV-2 in Patient with Hereditary Spherocytosis. Med. Clin. 2021, 157, e304–e305. [Google Scholar] [CrossRef]
- Ying, K.; Zhai, R.; Pyrkov, T.V.; Shindyapina, A.V.; Mariotti, M.; Fedichev, P.O.; Shen, X.; Gladyshev, V.N. Genetic and Phenotypic Analysis of the Causal Relationship between Aging and COVID-19. Commun. Med. 2021, 1, 35. [Google Scholar] [CrossRef]
- Araujo, J.J. Impact of the SARS-Cov-2 Virus Pandemic on Children and Adults with Congenital Heart Disease: Its Burden and Risk Factors. Int. J. Clin. Cardiol. 2020, 7, 190. [Google Scholar]
- Radke, R.M.; Frenzel, T.; Baumgartner, H.; Diller, G.-P. Adult Congenital Heart Disease and the COVID-19 Pandemic. Heart 2020, 106, 1302–1309. [Google Scholar] [CrossRef]
- Broberg, C.S.; Kovacs, A.H.; Sadeghi, S.; Rosenbaum, M.S.; Lewis, M.J.; Carazo, M.R.; Rodriguez, F.H.; Halpern, D.G.; Feinberg, J.; Galilea, F.A.; et al. COVID-19 in Adults With Congenital Heart Disease. J. Am. Coll. Cardiol. 2021, 77, 1644–1655. [Google Scholar] [CrossRef] [PubMed]
- Di Mascio, D.; Khalil, A.; Saccone, G.; Rizzo, G.; Buca, D.; Liberati, M.; Vecchiet, J.; Nappi, L.; Scambia, G.; Berghella, V.; et al. Outcome of Coronavirus Spectrum Infections (SARS, MERS, COVID-19) during Pregnancy: A Systematic Review and Meta-Analysis. Am. J. Obstet. Gynecol. MFM 2020, 2, 100107. [Google Scholar] [CrossRef] [PubMed]
- Triunfol, M. A New Mosquito-Borne Threat to Pregnant Women in Brazil. Lancet Infect. Dis. 2016, 16, 156–157. [Google Scholar] [CrossRef]
- Luteijn, J.M.; Brown, M.J.; Dolk, H. Influenza and congenital anomalies: A systematic review and meta-analysis. Hum. Reprod. 2014, 29, 809–823. [Google Scholar] [CrossRef] [PubMed]
- Blom, H.J.; Shaw, G.M.; Heijer, M.D.; Finnell, R. Neural tube defects and folate: Case far from closed. Nat. Rev. Neurosci. 2006, 7, 724–731. [Google Scholar] [CrossRef]
- Mednick, S.A.; Machon, R.A.; Huttunen, M.O.; Bonett, D. Adult Schizophrenia Following Prenatal Exposure to an Influenza Epidemic. Arch. Gen. Psychiatry 1988, 45, 189–192. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, E.; Sham, P.; Takei, N.; Murray, R.; Glover, G. Schizophrenia after prenatal exposure to 1957 A2 influenza epidemic. Lancet 1991, 337, 1248–1250. [Google Scholar] [CrossRef]
- Brown, A.S.; Schaefer, C.A.; Wyatt, R.J.; Goetz, R.; Begg, M.D.; Gorman, J.M.; Susser, E.S. Maternal Exposure to Respiratory Infections and Adult Schizophrenia Spectrum Disorders: A Prospective Birth Cohort Study. Schizophr. Bull. 2000, 26, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Hornig, M.; Bresnahan, M.A.; Che, X.; Schultz, A.F.; Ukaigwe, J.E.; Eddy, M.L.; Hirtz, D.; Gunnes, N.; Lie, K.K.; Magnus, P.; et al. Prenatal fever and autism risk. Mol. Psychiatry 2018, 23, 759–766. [Google Scholar] [CrossRef]
- Dreier, J.W.; Andersen, A.-M.N.; Hvolby, A.; Garne, E.; Andersen, P.K.; Berg-Beckhoff, G. Fever and infections in pregnancy and risk of attention deficit/hyperactivity disorder in the offspring. J. Child Psychol. Psychiatry 2016, 57, 540–548. [Google Scholar] [CrossRef]
- Dong, L.; Tian, J.; He, S.; Zhu, C.; Wang, J.; Liu, C.; Yang, J. Possible Vertical Transmission of SARS-CoV-2 From an Infected Mother to Her Newborn. J. Am. Med. Assoc. 2020, 323, 1846–1848. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, L.; Wu, T.; Shi, H.; Li, Q.; Jiang, H.; Zheng, D.; Wang, X.; Wei, Y.; Zhao, Y.; et al. Impact of Covid-19 in pregnancy on mother’s psychological status and infant’s neurobehavioral development: A longitudinal cohort study in China. BMC Med. 2020, 18, 347. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, T.; Harii, N.; Goto, J.; Harada, D.; Sugawara, H.; Takamino, J.; Ueno, M.; Sakata, H.; Kondo, K.; Myose, N.; et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020, 94, 55–58. [Google Scholar] [CrossRef]
- Colaco, S.; Chhabria, K.; Singh, D.; Bhide, A.; Singh, N.; Singh, A.; Husein, A.; Mishra, A.; Sharma, R.; Ashary, N.; et al. A Single-Cell RNA Expression Map of Coronavirus Receptors and Associated Factors in Developing Human Embryos. arXiv 2020, arXiv:2004.04935. [Google Scholar] [CrossRef]
- Meyyazhagan, A.; Pushparaj, K.; Balasubramanian, B.; Bhotla, H.K.; Pappusamy, M.; Arumugam, V.A.; Easwaran, M.; Pottail, L.; Mani, P.; Tsibizova, V.; et al. COVID-19 in pregnant women and children: Insights on clinical manifestations, complexities, and pathogenesis. Int. J. Gynecol. Obstet. 2021, 156, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Dogra, N.; Ledesma-Feliciano, C.; Sen, R. Developmental Aspects of SARS-CoV-2, Potential Role of Exosomes and Their Impact on the Human Transcriptome. J. Dev. Biol. 2021, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Qian, H.; Cao, S.; Dong, B.; Yan, X.; Luo, S.; Zhou, M.; Zhou, S.; Ning, B.; Zhao, L. Is there possibility of vertical transmission of COVID-19: A systematic review. Transl. Pediatr. 2021, 10, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Robaina-Castellanos, G.R.; Riesgo-Rodríguez, S.; de la, C. Congenital and Intrapartum SARS-CoV-2 Infection in Neonates: Hypotheses, Evidence and Perspectives. MEDICC Rev. 2021, 23, 72–83. [Google Scholar] [CrossRef]
- Tolu, L.B.; Ezeh, A.; Feyissa, G.T. Vertical transmission of Severe Acute Respiratory Syndrome Coronavirus 2: A scoping review. PLoS ONE 2021, 16, e0250196. [Google Scholar] [CrossRef]
- Fenizia, C.; Biasin, M.; Cetin, I.; Vergani, P.; Mileto, D.; Spinillo, A.; Gismondo, M.R.; Perotti, F.; Callegari, C.; Mancon, A.; et al. Analysis of SARS-CoV-2 vertical transmission during pregnancy. Nat. Commun. 2020, 11, 5128. [Google Scholar] [CrossRef] [PubMed]
- Sinaci, S.; Ocal, D.F.; Seven, B.; Anuk, A.T.; Besimoglu, B.; Keven, M.C.; Ayhan, S.G.; Akin, M.S.; Tayman, C.; Keskin, H.L.; et al. Vertical transmission of SARS-CoV-2: A prospective cross-sectional study from a tertiary center. J. Med. Virol. 2021, 93, 5864–5872. [Google Scholar] [CrossRef]
- Raschetti, R.; Vivanti, A.J.; Vauloup-Fellous, C.; Loi, B.; Benachi, A.; De Luca, D. Synthesis and Systematic Review of Reported Neonatal SARS-CoV-2 Infections. Nat. Commun. 2020, 11, 5164. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Gornet, M.; Sims, H.; Kisanga, E.; Knight, Z.; Segars, J. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) and Its Effect on Gametogenesis and Early Pregnancy. Am. J. Reprod. Immunol. 2020, 84, e13351. [Google Scholar] [CrossRef]
- Hayden, F.G.; Shindo, N. Influenza Virus Polymerase Inhibitors in Clinical Development. Curr. Opin. Infect. Dis. 2019, 32, 176–186. [Google Scholar] [CrossRef]
- Zash, R.; Holmes, L.; Diseko, M.; Jacobson, D.L.; Brummel, S.; Mayondi, G.; Isaacson, A.; Davey, S.; Mabuta, J.; Mmalane, M.; et al. Neural-Tube Defects and Antiretroviral Treatment Regimens in Botswana. N. Engl. J. Med. 2019, 381, 827–840. [Google Scholar] [CrossRef] [PubMed]
| Disease | Incidence | Association | References (DOI) |
|---|---|---|---|
| Sickle-cell anemia | Moderate | Increased risk of developing severe COVID-19 symptoms including acute chest syndrome, vasoocclusive crises, and death. | [139,140,141,142] |
| Thalassemia | Low | In risk population but does not increase the severity of COVID-19. However, pulmonary microembolism is reported and death occurred due to another comorbidity. | [48,143,144,145] |
| Aicardi-Goutieres Syndrome (AGS) | Low | Mostly asymptomatic or mild or shows rash on cheeks and arms. Post-COVID-19 generalized panniculitis is observed and the SAMDH1 gene could be a potential link between COVID-19 and AGS | [146,147] |
| Down Syndrome (DS) | Moderate | DS exhibits a higher risk of COVID-19 severity and 10 times higher mortality of SARS-CoV-2 infections. | [33,148] |
| Congenital coagulation defect | Low | May not be a risk factor for increased severity from COVID-19. Hypercoagulability may have a protective role against SARS-CoV-2 infection. | [149,150] |
| Genital organ defects | Moderate | Although the genetic or congenital condition of genital organ defects is not reported, testicular spermatogenesis dysfunction and reduced sperm count are frequent after SARS-CoV-2 infection. | https://www.europeanreview.org/article/24682 (accessed on 14 February 2022). |
| Asperger’s syndrome (AS) | Low | AS shows mental health and behavioral issues in COVID-19 patients. | [151] |
| Autism Spectrum Disorder (ASD) | Low | SARS-CoV-2 may impair brain development via cytokine storm during pregnancy increasing the risk of ASD. ASD increased vulnerability to COVID-19 in children and affected their behavior. | [22,152,153] |
| Sotos syndrome | Low | Pericardial effusion after infection with SARS-CoV-2 is reported. | [154] |
| Arthrogryposis | Low | Shows mild or no symptoms of COVID-19. | [155] |
| Cystic fibrosis (CF) | Low | CF is not a high-risk, however, CF patients with low lung function or transplants may show severe symptoms of COVID-19. | [16,156] |
| Fetal hemoglobin quantitative trait locus | High | Increased level of fetal hemoglobin may prevent hypoxia and cure respiratory distress syndrome in COVID-19. | [157] |
| Hereditary elliptocytosis | Low | May show varying severity and risk of hemolysis in COVID-19. | [88] |
| Familial erythrocytosis | Low | The presence of erythrocytosis increases the risk of thrombosis in COVID-19. | [158] |
| Bare lymphocyte syndrome (BLS)/Congenital immune deficiencies (CID) | Low | Convalescent plasma therapy may be effective in CID patients suffering from COVID-19. | [159] |
| Hemophagocytic lymphohistiocytosis (HL) | Low | HL may be a secondary event or a risk factor of severe COVID-19 | [160,161] |
| Aicardi syndrome/Malformations in brain | Low | Differences in neurodevelopment are observed in infants of six months infected by SARS-CoV-2 at the fetal stage. Long-term observation is required. | [162,163,164] |
| Alpha-1 antitrypsin deficiency (AAD) | High | AAD patients show worse outcome due to TMPRSS2 being activated more easily. They also have an increased risk of coagulation disorder and severe acute lung injury from COVID-19. | [165] |
| Plasminogen deficiency | Moderate | Low plasminogen level exhibits 12-fold higher mortality from COVID-19. | [166] |
| Congenital anomalies of the eye/congenital euryblepharon | Low | Eye abnormalities are observed in newborns infected by SARS-CoV-2 at the fetal stage. | [167] |
| Leukomalacia | Low | Periventricular leukomalacia is reported in newborns infected by SARS-CoV-2 at the fetal stage. | [164] |
| Adrenal hypoplasia (AH) | Low | AH patients undergoing glucocorticoid replacement therapy for adrenal insufficiency are vulnerable to developing severe complications from COVID-19. | [168] |
| Disorders of tooth development | Low | A fetus may be at high risk for enamel defects due to the stress of COVID-19 during pregnancy. Tooth loss is observed in severe COVID-19. | [169,170] |
| Hereditary spherocytosis (HS) | Moderate | HS patients show an increased risk of hemolysis and splenomegaly due to COVID-19. | [88,171] |
| Lifespan | Not Known Available | Genetic polymorphisms that are linked to longer lifespans are significantly associated with a low risk of SARS-CoV-2 infection and hospitalization. | [172] |
| Congenital heart disease (CHD) | Low | CHD are of low or moderate risk, which may develop hemodynamic abnormalities upon SARS-CoV-2 infection but has no impact on mortality. | [173,174,175] |
| Spina Bifida (SB) | Low | Impact on mental health | [90,91,92,93,94,95,96] |
| Fragile X-Syndrome (FXS) | Low | Decreased CXCL-10 as protection against Cytokine Storm Syndrome | [99,100,101,102,103,104,105,106,107,108] |
| Prader-Willi Syndrome (PWS) | Low | Youthful age of the cohort had a positive impact, so far | [70,71,72,73,74,75,76,77,78,79,80,81] |
| Hypothyroidism | Low to Moderate | Low fT3 level can lead to poor outcomes | [97,98] |
| Duchenne/Becker Muscular Dystrophy (DMD/BMD) | Low | All symptomatic individuals recovered without any long-term consequences | [109,110,111,112,113,114,115,116] |
| Lysosomal Storage Disorders (LSD) | Low | Impact on mental health | [82,83,84,85] |
| Autoimmune Hemolytic Anemia (AIHA) | Moderate | Hemolysis | [23,24,25] |
| NMO Spectrum Disorder | Low | Readmission | [117,118,119,120,121,122,123,124,125,126,127,128,129] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hromić-Jahjefendić, A.; Barh, D.; Ramalho Pinto, C.H.; Gabriel Rodrigues Gomes, L.; Picanço Machado, J.L.; Afolabi, O.O.; Tiwari, S.; Aljabali, A.A.A.; Tambuwala, M.M.; Serrano-Aroca, Á.; et al. Associations and Disease–Disease Interactions of COVID-19 with Congenital and Genetic Disorders: A Comprehensive Review. Viruses 2022, 14, 910. https://doi.org/10.3390/v14050910
Hromić-Jahjefendić A, Barh D, Ramalho Pinto CH, Gabriel Rodrigues Gomes L, Picanço Machado JL, Afolabi OO, Tiwari S, Aljabali AAA, Tambuwala MM, Serrano-Aroca Á, et al. Associations and Disease–Disease Interactions of COVID-19 with Congenital and Genetic Disorders: A Comprehensive Review. Viruses. 2022; 14(5):910. https://doi.org/10.3390/v14050910
Chicago/Turabian StyleHromić-Jahjefendić, Altijana, Debmalya Barh, Cecília Horta Ramalho Pinto, Lucas Gabriel Rodrigues Gomes, Jéssica Lígia Picanço Machado, Oladapo Olawale Afolabi, Sandeep Tiwari, Alaa A. A. Aljabali, Murtaza M. Tambuwala, Ángel Serrano-Aroca, and et al. 2022. "Associations and Disease–Disease Interactions of COVID-19 with Congenital and Genetic Disorders: A Comprehensive Review" Viruses 14, no. 5: 910. https://doi.org/10.3390/v14050910
APA StyleHromić-Jahjefendić, A., Barh, D., Ramalho Pinto, C. H., Gabriel Rodrigues Gomes, L., Picanço Machado, J. L., Afolabi, O. O., Tiwari, S., Aljabali, A. A. A., Tambuwala, M. M., Serrano-Aroca, Á., Redwan, E. M., Uversky, V. N., & Lundstrom, K. (2022). Associations and Disease–Disease Interactions of COVID-19 with Congenital and Genetic Disorders: A Comprehensive Review. Viruses, 14(5), 910. https://doi.org/10.3390/v14050910













