Updates on the Epidemiology of the Human T-Cell Leukemia Virus Type 1 Infection in the Countries of the Eastern Mediterranean Regional Office of the World Health Organization with Special Emphasis on the Situation in Iran
Abstract
1. Introduction
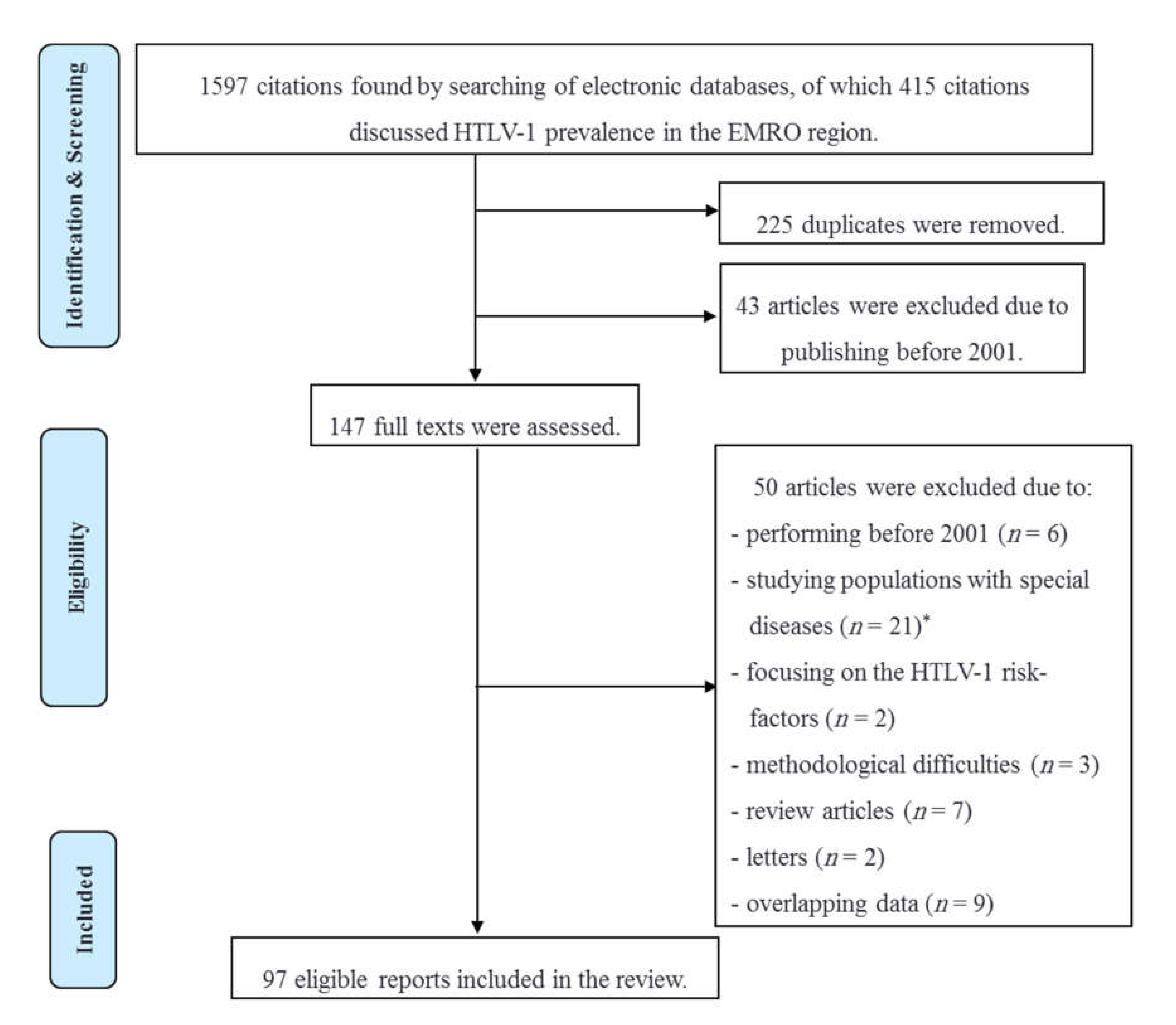
2. Material and Methods
3. Results and Discussion
3.1. HTLV-1 Infection in Iran
3.1.1. HTLV-1 Infection in Iranian Low-Risk Population
3.1.2. HTLV-1 Infection among High-Risk Iranian Population
3.2. HTLV-2 Infection in Iran
3.3. HTLV-1 Infection in EMRO Countries except for Iran
3.3.1. HTLV-1 Infection in Asian Countries of EMRO
3.3.2. HTLV-1 Infection in African Countries of EMRO
3.4. Study Limitations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Poiesz, B.J.; Ruscetti, F.W.; Gazdar, A.F.; Bunn, P.A.; Minna, J.D.; Gallo, R.C. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc. Natl. Acad. Sci. USA 1980, 77, 7415–7419. [Google Scholar] [CrossRef] [PubMed]
- Kalyanaraman, V.S.; Sarngadharan, M.G.; Robert-Guroff, M.; Miyoshi, I.; Blayney, D.; Golde, D.; Gallo, R.C. A new subtype of human t-cell leukemia virus (HTLV-II) associated with a t-cell variant of hairy cell leukemia. Science 1982, 218, 571–573. [Google Scholar] [CrossRef] [PubMed]
- Willems, L.; Hasegawa, H.; Accolla, R.; Bangham, C.; Bazarbachi, A.; Bertazzoni, U.; Carneiro-Proietti, A.B.D.F.; Cheng, H.; Chieco-Bianchi, L.; Ciminale, V.; et al. Reducing the global burden of HTLV-1 infection: An agenda for research and action. Antivir. Res. 2016, 137, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Gessain, A.; Cassar, O. Epidemiological Aspects and World Distribution of HTLV-1 Infection. Front. Microbiol. 2012, 3, 388. [Google Scholar] [CrossRef] [PubMed]
- Catovsky, D.; Greaves, M.F.; Rose, M.; Galton, D.A.; Goolden, A.W.; McCluskey, D.R.; White, J.M.; Lampert, I.; Bourikas, G.; Ireland, R.; et al. Adult T-cell lymphoma-leukaemia in Blacks from the West Indies. Lancet 1982, 1, 639–643. [Google Scholar] [CrossRef]
- Harrington, W.J., Jr.; Ucar, A.; Gill, P.; Snodgrass, S.; Sheremata, W.; Cabral, L.; Rabin, M.; Byrne, G.E., Jr.; Berger, J.; Voight, W.; et al. Clinical spectrum of HTLV-I in south Florida. J. Acquir. Immune Defic. Syndr. Hum. Retrovirology 1995, 8, 466–473. [Google Scholar] [CrossRef]
- Afonso, P.V.; Cassar, O.; Gessain, A. Molecular epidemiology, genetic variability and evolution of HTLV-1 with special emphasis on African genotypes. Retrovirology 2019, 16, 39. [Google Scholar] [CrossRef]
- Cassar, O.; Einsiedel, L.; Afonso, P.V.; Gessain, A. Human T-cell lymphotropic virus type 1 subtype c molecular variants among indigenous Australians: New insights into the molecular epidemiology of HTLV-1 in Australo-melanesia. PLoS Neglected Trop. Dis. 2013, 7, e2418. [Google Scholar] [CrossRef]
- Percher, F.; Jeannin, P.; Martin-Latil, S.; Gessain, A.; Afonso, P.V.; Vidy-Roche, A.; Ceccaldi, P.-E. Mother-to-Child Transmission of HTLV-1 Epidemiological Aspects, Mechanisms and Determinants of Mother-to-Child Transmission. Viruses 2016, 8, 40. [Google Scholar] [CrossRef]
- Roucoux, D.F.; Wang, B.; Smith, D.; Nass, C.C.; Smith, J.; Hutching, S.T.; Newman, B.; Lee, T.; Chafets, D.M.; Murphy, E.L.; et al. A prospective study of sexual transmission of human T lymphotropic virus (HTLV)–I and HTLV-II. J. Infect. Dis. 2005, 191, 1490–1497. [Google Scholar] [CrossRef]
- Murphy, E. Infection with human T-lymphotropic virus types-1 and -2 (HTLV-1 and -2): Implications for blood transfusion safety. Transfus. Clin. Biol. 2016, 23, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Rowhani-Rahbar, A.; Tabatabee-Yazdi, A.; Panahi, M. Prevalence of common blood-borne infections among imprisoned injection drug users in Mashhad, North-East of Iran. Arch. Iran. Med. 2004, 7, 190–194. [Google Scholar]
- Proietti, F.A.; Carneiro-Proietti AB, F.; Catalan-Soares, B.C.; Murphy, E.L. Global epidemiology of HTLV-I infection and associated diseases. Oncogene 2005, 24, 6058–6068. [Google Scholar] [CrossRef] [PubMed]
- Schierhout, G.; McGregor, S.; Gessain, A.; Einsiedel, L.; Martinello, M.; Kaldor, J. Association between HTLV-1 infection and adverse health outcomes: A systematic review and meta-analysis of epidemiological studies. Lancet Infect. Dis. 2019, 20, 133–143. [Google Scholar] [CrossRef]
- Keikha, M.; Karbalaei, M. Overview on coinfection of HTLV-1 and tuberculosis: Mini-review. J. Clin. Tuberc. Other Mycobact. Dis. 2021, 23, 100224. [Google Scholar] [CrossRef] [PubMed]
- Page, J.B.; Lai, S.H.; Chitwood, D.D.; Klimas, N.G.; Smith, P.C.; Fletcher, M.A. HTLV-I/II seropositivity and death from AIDS among HIV-1 seropositive intravenous drug users. Lancet 1990, 335, 1439–1441. [Google Scholar] [CrossRef]
- World Health Organization. Human T-Lymphotropic Virus Type 1: Technical Report; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- World Health Organization Regional Office for the Eastern Mediterranean. Available online: http://www.emro.who.int/countries.html (accessed on 15 May 2021).
- Azarpazhooh, M.R.; Hasanpour, K.; Ghanbari, M.; Rezaee, S.R.; Mashkani, B.; Hedayati-Moghaddam, M.R.; Valizadeh, N.; Hosseini, R.F.; Foroghipoor, M.; Soltanifar, A.; et al. Human T-lymphotropic virus type 1 prevalence in northeastern Iran, Sabzevar: An epidemiologic-based study and phylogenetic analysis. AIDS Res. Hum. Retrovir. 2012, 28, 1095–1101. [Google Scholar] [CrossRef]
- Kalavi, K.; Moradi, A.; Tabarraei, A. Population-based Seroprevalence of HTLV-I Infection in Golestan Province, South East of Caspian Sea, Iran. Iran J. Basic Med. Sci. 2013, 16, 225–228. [Google Scholar]
- Rafatpanah, H.; Hedayati-Moghaddam, M.R.; Fathimoghadam, F.; Bidkhori, H.R.; Shamsian, S.K.; Ahmadi, S.; Sohgandi, L.; Azarpazhooh, M.R.; Rezaee, S.A.; Farid, R.; et al. High prevalence of HTLV-I infection in Mashhad, Northeast Iran: A population-based seroepidemiology survey. J. Clin. Virol. 2011, 52, 172–176. [Google Scholar] [CrossRef]
- Rafatpanah, H.; Torkamani, M.; Valizadeh, N.; Vakili, R.; Meshkani, B.; Khademi, H.; Gerayli, S.; Mozhgani, S.H.R.; Rezaee, S.A. Prevalence and phylogenetic analysis of HTLV-1 in a segregated population in Iran. J. Med. Virol. 2016, 88, 1247–1253. [Google Scholar] [CrossRef]
- Yousefi, M.; Sharifzadeh, G.R.; Ebrahimzadeh, A.; Azarkar, Z.; Namaei, M.H.; Azarkar, G.; Ghezeldasht, S.A.; Rezaee, R.; Zare, N.V.; Mosavat, A.; et al. Prevalence and Associated Risk Factors of HTLV-1 and Co-infections of Blood-Borne Viruses in Birjand, Iran’s Eastern Border. Arch. Iran. Med. 2020, 23, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Farid Hoseini, R.; Parizadeh, M.; Ghafari, J.; Miri, S.; Nasirian, A.; Rafatpanah, H. Seroepidemiology of HTLV-1 infection in Neyshabour. Med. J. Mashad Univ. Med. Sci. 2005, 47, 417–424. (In Persian) [Google Scholar]
- Rezvan, H.; Ahmadi, J.; Farhadi, M. A cluster of HTLV1 infection in northeastern of Iran. Transfus. Today 1996, 27, 8–9. [Google Scholar]
- Safai, B.; Huang, J.-L.; Boeri, E.; Farid, R.; Raafat, J.; Schutzer, P.; Ahkami, R.; Franchini, G. Prevalence of HTLV Type I Infection in Iran: A Serological and Genetic Study. AIDS Res. Hum. Retrovir. 1996, 12, 1185–1190. [Google Scholar] [CrossRef]
- Hedayati-Moghaddam, M.R.; Fathimoghadam, F.; Eftekharzadeh Mashhadi, I.; Soghandi, L.; Bidkhori, H.R. Epidemiology of HTLV-1 in Neyshabour, Northeast of Iran. Iran. Red Crescent Med. J. 2011, 13, 424–427. [Google Scholar]
- Keramati, M.R.; Sadeghian, M.H.; Ayatollahi, H. Clinical and laboratory features in adult T-cell leukemia/lymphoma in Khorasan, Iran. Leuk. Lymphoma 2010, 51, 727–729. [Google Scholar] [CrossRef]
- Shoeibi, A.; Rafatpanah, H.; Azarpazhooh, A.; Mokhber, N.; Hedayati-Moghaddam, M.R.; Amiri, A.; Hashemi, P.; Foroghipour, M.; Hoseini, R.F.; Bazarbachi, A.; et al. Clinical features of HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) in northeast Iran. Acta Neurol. Belg. 2013, 113, 427–433. [Google Scholar] [CrossRef]
- Darroudi, S.; Abolbashari, S.; Ahangari, N.; Tayefi, M.; Khashyarmanesh, Z.; Zamani, P.; Haghighi, H.M.; Mohammadpour, A.H.; Tavalaei, S.; Esmaily, H.; et al. Association Between Trace Element Status and Depression in HTLV-1-Infected Patients: A Retrospective Cohort Study. Biol. Trace Element Res. 2019, 191, 75–80. [Google Scholar] [CrossRef]
- Karimi, G.; Zadsar, M.; Pourfathollah, A.A. Seroprevalence and geographical distribution of human T-lymphotropic virus type 1 among volunteer blood donors in endemic areas of Iran. Virol. J. 2017, 14, 14. [Google Scholar] [CrossRef]
- Omidkhoda, A.; Razi, B.; Arabkhazaeli, A.; Kafi-Abad, S.A. Trends and epidemiological analysis of hepatitis B virus, hepatitis C virus, human immunodeficiency virus, and human T-cell lymphotropic virus among Iranian blood donors: Strategies for improving blood safety. BMC Infect. Dis. 2020, 20, 736. [Google Scholar] [CrossRef]
- Poukarim, M.R.; Khamisipour, G.; Zandi, K.; Roustaee, M.H. Prevalence of anti-HTLV-I & anti- HTLV-II antibodies in blood donors in Bushehr province. Iran South Med. J. 2004, 6, 161–164. (In Persian) [Google Scholar]
- Karimi, A.; Nafici, M.; Imani, R. Comparison of human T-cell leukemia virus type-1 [HTLV-1]seroprevalence in high risk patients [thalassemia and hemodialysis]and healthy individuals from charmahal-Bakhtiari province, Iran. KMJ-Kuwait Med. J. 2007, 39, 259–261. [Google Scholar]
- Arjmandi, F.; Shahriari, M.; Sadeghi Hasanabadi, A. A comparitive study of the prevalence of HTLV–I infection in luekemia/ non–hodgkins lymphoma patients, thalassemic patients and blood donors. J. Shahid Sadoughi Univ. Med. Sci. 2003, 10, 25–29. [Google Scholar]
- Habibabadi, H.M.; Parsania, M.; Pourfathollah, A.A.; Bahrami, A.; Sharifi, Z. Prevalence and phylogenetic analysis of HTLV-1 in blood donors in Golestan Province, in the Northeast of Iran. J. Virol. Methods 2021, 290, 114073. [Google Scholar] [CrossRef]
- Abedi, F.; Yavarian, M.; Shakibzadeh, A.; Khalvati, B.; Asadi, A. A pilot Seroepidemiologic study of HTLV in thalassemia, hemophilia, and hemodialysed patients in Hormozgan. Hormozgan Med. J. 2009, 13, e88635. [Google Scholar]
- Zalei, B.; Pourmand, D.; Rahmani, Y.; Bilvaye, S. Investigating the prevalence of HTLV and HCV infection in blood donors (for the first time) in blood transfusion organization of Kermanshah, Iran. Ann. Clin. Anal. Med. 2017, 8, 509–512. [Google Scholar] [CrossRef]
- Ghadiri, K.; Hashemian, A.; Rezaei, M.; Payandeh, M.; Zare, M.E. Serologic prevalence of human T-lymphotropic virus (HTLV) among major thalassemic patients in Kermanshah 2010. Int. J. Hematol.-Oncol. Stem Cell Res. 2011, 5, 14–17. [Google Scholar]
- Yahyapour, Y.; Aghajanipour, K.; Mir, S.M.; Khademian, A.; Sadeghi, F. Human T- Lymphotropic Virus Type 1 in Blood Donors from Babol County Blood Transfusion Center: A Pilot Study From Northern Iran. Jundishapur J. Microbiol. 2017, 10, e13757. [Google Scholar] [CrossRef]
- Tarhini, M.; Kchour, G.; Zanjani, D.S.; Rafatpanah, H.; Otrock, Z.K.; Bazarbachi, A.; Farid, R. Declining tendency of human T-cell leukaemia virus type I carrier rates among blood donors in Mashhad, Iran. Pathology 2009, 41, 498–499. [Google Scholar] [CrossRef]
- Hatami, H.; Karimi, G.; Safabakhsh, H.R. Seroepidemiologic prevalence of HTLV in voluntary blood donors in Mashhad. Sci. J. Iran. Blood Transfus. Organ. 2012, 9, 149–159. [Google Scholar]
- Sayadpour Zanjani, D.; Shahabi, M.; Talaei, N.; Afzalaghaee, M.; Tehranian, F.; Bazargani, R. Molecular Analysis of Human T Cell Lymphotropic Virus Type 1 and 2 (HTLV-1/2) Seroindeterminate Blood Donors from Northeast Iran: Evidence of Proviral tax, env, and gag Sequences. AIDS Res. Hum. Retrovir. 2011, 27, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Safabakhsh, H.r.; Karimi, G.; Hatami, H. The prevalence of HTLV-1 infection in blood donation volunteers in Mashhad. J. Sch. Public Health Inst. Public Health Res. 2014, 11, 85–94. [Google Scholar]
- Tabriznia Tabrizi, S.; Gholampour, M.; Zarmehri, A.M.; Sadeghian, M.H.; Keramati, M.R. The frequency and risk factors of human T-cell lymphotropic virus type I among blood donors in Khorasan-e Razavi, Iran, from 2002 to 2013. Niger. Med. J. Niger. Med. Assoc. 2020, 61, 151–155. [Google Scholar] [CrossRef]
- Safabakhsh, H.; Jalalian, M.; Karimi, G. Seroepidemiology of human T-Cell lymphotropic virus Type-1 (HTLV1) in mashhad. Glob. J. Health Sci. 2014, 6, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Maghsudlu, M.; Safabakhsh, H.; Jamili, P. Seroepidemiology of human T-cell lymphotropic virus type-I in blood donors of Northeastern Iran, Sabzevar. Asian J. Transfus. Sci. 2015, 9, 203–206. [Google Scholar] [CrossRef]
- Ferdowsi, S.; Najafzadeh, M.; Ameli, M.R.; Sajjadi, S.M. Evaluating the Frequency of Human T-Lymphotropic Virus Type 1 Infection in Iranian Population. Iran. J. Blood Cancer 2019, 11, 47–50. [Google Scholar]
- Pirayeshfard, L.; Sharifi, Z.; Amini-Kafiabad, S.; Sadaghiani, N.H. Phylogenetic analysis of HTLV-1 in Iranian blood donors, HIV-1 positive patients and patients with beta thalassemia. J. Med. Virol. 2018, 90, 1398–1405. [Google Scholar] [CrossRef]
- Rostamzadeh Khameneh, Z.; Baradaran, M.; Sepehrvand, N. Survey of the seroprovalence of HTLV I/II in hemodialysis patients and blood donors in Urmia. Saudi J. Kidney Dis. Transplant. Off. Publ. Saudi Cent. Organ Transplant. Saudi Arab. 2008, 19, 838–841. [Google Scholar]
- Emadi, B.; Rezaieh, M.G.; Sedighi, M. Seroprevalence and trends of transfusion transmissible infections among retrospective blood donors in Western Azerbaijan Regional Blood Transfusion Center, Iran: A ten-years evaluation. Transfus. Apher. Sci. Off. J. World Apher. Assoc. Off. J. Eur. Soc. Haemapheresis 2021, 60, 103132. [Google Scholar] [CrossRef]
- Rezaei Kanavi, M.; Javadi, M.; Nazparvar, B.; Bayat Makoo, K. Seropositivity for human T-cell lymphotropic virus [HTL V] in donors at the eye bank of I.R. Iran. Bina J. Ohthalmol. 2008, 14, 40–43. [Google Scholar]
- Arjmand, B.; Aghayan, S.H.; Goodarzi, P.; Farzanehkhah, M.; Mortazavi, S.M.; Nicknam, M.H.; Jafarian, A.; Arjmand, F.; Far, S.J. Seroprevalence of human T lymphtropic virus (HTLV) among tissue donors in Iranian tissue bank. Cell Tissue Bank. 2008, 10, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Hamedi, A.; Akhlaghi, F.; Meshkat, Z.; Sezavar, M.; Nomani, H. The Prevalence of Human T-Cell lymphotropic Virus Type 1 in Pregnant Women and Their Newborns. ISRN Obstet. Gynecol. 2012, 2012, 975135. [Google Scholar] [CrossRef] [PubMed]
- Salehi, M.; Mostafavi, S.K.S.; Ghasemian, A.; Gholami, M.; Kazemi-Vardanjani, A.; Rahimi, M.K. Seroepidemiology of HTLV-1 and HTLV-2 Infection in Neyshabur City, North-Eastern Iran, during 2010–2014. Iran. Biomed. J. 2017, 21, 57–60. [Google Scholar] [CrossRef]
- Salehi, M.; Mobini, M. The seroepidemiological of Human T-lymphotropic virus on children, teenage and juveniles in Nyshabor city. Iran. J. Med. Microbiol. 2015, 9, 31–35. [Google Scholar]
- Mobini, M.; Salehi, M. Prevalence of Human T-cell Leukemia Virus Antibodies among Young Females Attending a Large Medical Diagnostic Laboratory in Neyshabour, Northeast of Iran, During 2011 to 2015. Arch. Clin. Infect. Dis. 2018, 13, e62305. [Google Scholar] [CrossRef]
- Ghaffari, J.; Naghshvar, F.; Nazari, Z.; Farid, R. Seroprevalence of human T-cell lymphotropic virus type 1 infection (HTLV1) in different patients in the north of Iran. Afr. J. Biotechnol. 2011, 10, 10752–10755. [Google Scholar] [CrossRef]
- Ghodsi, A.; Jamehdar, S.A.; Hamedi, A.K. Frequency of HTLV-1 Antibody in Children of 6 Months to 14 Years old Hospitalized in the Pediatric Ward of Imam Reza Hospital, Mashhad, Iran. Int. J. Pediatrics-Mashhad 2021, 9, 12975–12980. [Google Scholar] [CrossRef]
- Tahaei, S.M.E.; Mohebbi, S.R.; Fatemi, S.R.; Azimzadeh, P.; Mirsattari, D.; Sanati, A.; Sharifian, A. Evaluation of antibody frequency against HBV, HCV and HTLV-1. Gastroenterol. Hepatol. Bed Bench 2012, 5, 161–165. [Google Scholar]
- Razavi Pashabayg, C.; Momenifar, N.; Malekpour, S.A.; Sadeghi, M.; Foroushani, A.R.; Rafatpanah, H.; Valizadeh, N.; Sabet, F.; Jazayeri, S.M.; Keyvani, H.; et al. Phylogenetic and phylodynamic study of Human T-cell lymphotropic virus Type 1 (HTLV-1) in Iran. Infect. Genet. Evol. 2020, 85, 104426. [Google Scholar] [CrossRef]
- Hedayati-Moghaddam, M.R. A Systematic Review for Estimation of HTLV-I Infection in the Blood Donors of Iran. Iran. J. Basic Med. Sci. 2013, 16, 196–201. [Google Scholar] [CrossRef]
- Azami, M.; Badfar, G.; Esmaeli, E.; Moslemirad, M.; Rahmati, S. Epidemiology of human T-lymphotropic virus type 1 among blood donors and general population in Iran: A meta-analysis. Futur. Virol. 2018, 13, 585–599. [Google Scholar] [CrossRef]
- Rezvan, H.; Abolghassemi, H.; Kafiabad, S.A. Transfusion-transmitted infections among multitransfused patients in Iran: A review. Transfus. Med. 2007, 17, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Hedayati-Moghaddam, M.R.; Tehranian, F.; Bayati, M. Human T-Lymphotropic Virus Type I (HTLV-1) Infection among Iranian Blood Donors: First Case-Control Study on the Risk Factors. Viruses 2015, 7, 5736–5745. [Google Scholar] [CrossRef] [PubMed]
- Dayyani, M.; Hedayati-Moghaddam, M.R.; Amini, A.R. HTLV-1 infection as a serious health issue among iranian multi-transfused patients: Evidence from a systematic review and meta-analysis. Iran. J. Blood Cancer 2015, 7, 85–94. [Google Scholar]
- Ahmadi Ghezeldasht, S.; Hassannia, T.; Rafatpanah, H.; Hekmat, R.; Valizadeh, N.; Mobarhan, M.G.; Rezaee, S.A. Oncogenic Virus Infections in the General Population and End-stage Renal Disease Patients with Special Emphasis on Kaposi’s Sarcoma Associated Herpes Virus (KSHV) in Northeast of Iran. Jundishapur J. Microbiol. 2015, 8, e14920. [Google Scholar] [CrossRef] [PubMed]
- Hedayati-Moghaddam, M.R.; Fathimoghadam, F.; Soghandi, L.; Darrudi, A. High Prevalence of HTLV-1 Infection Among Hemodialysis Patients in Neyshabur, Northeast of Iran. Int. J. Infect. 2019, 6, e85164. [Google Scholar] [CrossRef]
- Karimi, A.; Nafisi, M. Seroprevalence of Human T-Cell Leukemia Virus Type-1 (HTLV-1) in High Risk Patients. J. Res. Health Sci. 2006, 6, 44–47. [Google Scholar]
- Anaraki Mohammadi, G.; Sadeghipour, A.R.; Vossough, P.; Nour Mohammadi, I.; Mirnateghi, A.M. Assessment of the Prevalence of Human T-Lymphotropic Virus type 1 among Thalassemic Patients with Frequent Blood Transfusion in Tehran in 2003. Razi J. Med. Sci. 2005, 12, 19–24. [Google Scholar]
- Keshvari, M.; Hajibeigi, B.; Azarkeivan, A.; Keyvani, H.; Behnava, B.; Hosseini, S.Y.S.; Sharafi, H.; Alavian, S.M. Seroepidemiology of human T-cell lymphotropic virus among Iranian adult thalassemic patients. Transfus. Med. 2014, 24, 227–232. [Google Scholar] [CrossRef]
- Abbasnia, S.; Hassannia, T.; Ariaee, N.; Sabet, F.; Pashabayg, K.R.; Keyvani, H.; Sarv-Ghad, M.R.; Ziaeemehr, A.; Fakor, F.; Valizadeh, N. Immunovirological Assessment of HIV-Infected Patients and Phylogenetic Analysis of the Virus in Northeast of Iran. Jundishapur J. Microbiol. 2021, 14, e112123. [Google Scholar] [CrossRef]
- Alavi, S.M.; Etemadi, A. HIV/HBV, HIV/HCV and HIV/HTLV-1 Co infection among injecting drug user patients hospitalized at the infectious disease ward of a training hospital in Iran. Pak. J. Med. Sci. 2007, 23, 510–513. [Google Scholar]
- Khajedaluee, M.; Babaei, A.; Vakili, R.; Valizade, N.; Shandiz, F.H.; Alavian, S.M.; Nozadi, M.S.; Jazayeri, S.M.; Hassannia, T. Sero-Prevalence of Bloodborne Tumor Viruses (HCV, HBV, HTLV-I and KSHV Infections) and Related Risk Factors among Prisoners in Razavi Khorasan Province, Iran, in 2008. Hepat. Mon. 2016, 16, e31541. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention and the U.S.P.H.S. Working Group. Guidelines for counseling persons infected with human T-lymphotropic virus type I (HTLV-I) and type II (HTLV-II). Ann. Intern. Med. 1993, 118, 448–454. [Google Scholar] [CrossRef]
- Pourkarim, M.; Khamisipour, G.; Hajiani, G.; Tahmasebi, R.; Ardeshirdavani, N. Seroepidemiological investigation of HTLV I, II infection among Busherian multi-transfused patients in 2003. Sci. J. Iran. Blood Transfus. Organ. 2005, 2, 99–104. [Google Scholar]
- Ardalan, N.; Abdi, M.; Zarif, B.R.; Amini, A.; Meamari, F.; Haydari, E.; Ahmadi, A. Prevalence of human T-lymphotropic virus types I & II among high risk groups in Sanandaj in 2010. Sci. J. Kurd. Univ. Med. Sci. 2013, 18, 51–57. [Google Scholar]
- Ghaffari, J.; Ebrahimi, M.; Makhlough, A.; Mohammadjafari, H.; Nazari, Z. Seroepidemiology of human T-cell lymphotropic virus 1 infection in hemodialysis patients: Should we be concerned about it? Iran. J. Kidney Dis. 2013, 7, 187–190. [Google Scholar]
- Ziaee, M.; Azizee, R.; Namaei, M.H.; Masood, Z.; Roghiya, A.; Hasan, N. Prevalence of HCV infection in hemodialysis patients of South Khorasan in comparison with HBV, HDV, HTLV I/II, And HIV infection. Bangladesh J. Med. Sci. 2013, 13, 36–39. [Google Scholar] [CrossRef]
- Yazdani, R.; Dadmanesh, M.; Ghorban, K. First Report of the Prevalence of Human T-Lymphotropic Virus Type 1 (HTLV-1) for Hemodialysis Patients in Tehran. Arch. Clin. Infect. Dis. 2018, 13, e65512. [Google Scholar] [CrossRef]
- Hatatian, F.; Babakhani, F.; Gudarzi, H.; Momenifar, N.; Norouzi, M.; Shafieifar, M.; Kakavandi, E.; Sadeghi, A.; Sharbatdar-Alaei, H.; Farahmand, M.; et al. Relative Frequency of Blood-Borne Viruses in Hemodialysis-Dependent and Kidney Transplant Recipients in Iran. Iran. J. Public Health 2020, 49, 2136–2143. [Google Scholar] [CrossRef]
- Moradi, A.; Mansurian, A.; Ahmadi, A.; Ghaemi, E.A.; Kalavi, K.; Marjani, A.; Moghaddam, E.S. Prevalence of HTLV-1 Antibody among Major Thalassemic Patients in Gorgan (South East of Caspian Sea). J. Appl. Sci. 2008, 8, 391–393. [Google Scholar] [CrossRef][Green Version]
- Mortezaie, Z.; Bouzari, M.; Roghanian, R. Evaluating the frequency of HTLV-I/Π infection among blood donors, major thalassemic patients and individuals infected with hepatitis B and C viruses in Isfahan, Iran. Iran. J. Blood Cancer 2012, 4, 75–80. [Google Scholar]
- Mahzounieh, M.; Ghorani, M.; Karimi, A.; Pourgheysari, B.; Nikoozad, R. Prevalence of Human T-Lymphotropic Virus Types I and II in Patients with Hematological Disorders in Isfahan, Iran. Jundishapur J. Microbiol. 2015, 8, e17201. [Google Scholar] [CrossRef]
- Ghaffari, J.; Kowsarian, M.; Mahdavi, M.; Shahi, K.V.; Rafatpanah, H.; Tafreshian, A. Prevalence of HTLV-I Infection in Patients with Thalassemia Major in Mazandaran, North of Iran. Jundishapur J. Microbiol. 2012, 6, 57–60. [Google Scholar] [CrossRef]
- Hamidi, S.; Bashizadeh-Fakhar, H.; Nazemi, A. Identification of Human T-Cell Lymphotropic Virus Type 1 Pro-Invasion in Patients with β-Thalassemia Major Using TaqMan Real-Time PCR in Tonekabon, Iran. Zahedan J. Res. Med. Sci. 2018, 20, e59961. [Google Scholar] [CrossRef]
- Mansouritorghabeh, H.; Badiei, Z. Transfusion-Transmitted Viruses in Individuals with β Thalassemia Major at Northeastern Iran, a Retrospective Sero-Epidemiological Survey. Iran. J. Blood Cancer 2008, 1, 1–4. [Google Scholar]
- Abedi, F.; Mozhgani, S.-H.; Rahimzadegan, M.; Gudarzi, H.; Valizadeh, N.; Rezaee, S.A. Prevalence and phylogenic study of human T-lymphotropic virus 1 in patients with thalassemia in the northeast of Iran. Futur. Virol. 2017, 12, 253–258. [Google Scholar] [CrossRef]
- Ziaee, M.; Zarban, A.; Malekinejad, P.; Akhbary, H. Evaluation of HGV viremia prevalence and its co-infection with HBV, HCV, HIV and HTLV-1 in hemophilic patients of southern Khorassan, Iran. Hepat. Mon. 2007, 7, 11–14. [Google Scholar]
- Ziaee, M.; Namaei, M.H.; Azarkar, G. The prevalence of HTLV-1 and its Co-Infection with HCV, HBV and HIV in Hemophilic patients. Pak. J. Med. Sci. 2015, 31, 1246–1249. [Google Scholar] [CrossRef]
- Rostamzadeh, Z.; Valizadeh, N.; Mohammadian, M. Prevalence of Seropositivity for Human T lymphocytes Virus in Patients with Hereditary Bleeding Diseases in Population of West Azerbaijan. Int. J. Med. Lab. 2016, 3, 159–162. [Google Scholar]
- Mansouritorghabeh, H.; Farid Hosseini, R.; Pourfath Elah, A.A.; Behjati, A.A. Serological markers of blood borne viruses among persons with combined factor V & VIII deficiency. Hepat. Mon. 2008, 8, 221–224. [Google Scholar]
- Meidani, M.; Ahmadi, N.; Jamali, B.; Askarian, Z. Relative frequency of Human T-cell Lymphotropic Virus I/II in HIV/AIDS patients. Adv. Biomed. Res. 2014, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Meidani, M.; Farzaneh, S.; Baferani, A.; Zade, A. [Seroprevalence of HTLV1, 2 virus among injection drug addicts in Isfahan, 2007–2008]. J. Shaheed Sadoughi Univ. Med. Sci. 2009, 17, 286–290. [Google Scholar]
- Tavanaee Sani, A.; Khaleghinia, M. Epidemiologic evaluation and some species in injection drug users that admitted in infectious department of Imam Reza hospital [2007–2009]. J. Med. Counc. Iran 2012, 30, 155–161. [Google Scholar]
- Ramezani, A.; Aghakhani, A.; Banifazl, M.; Boland-Ghamat, Z.; Foroughi, M.; Gachkar, L.; Khadem-Sadegh, A.; Mohraz, M. Frequency of human T-Cell lymphotropic virus (HTLV) type 1 and 2 infection in HIV infected patients. Iran. J. Pathol. 2012, 7, 9–13. [Google Scholar]
- Abedi, F.; Saeedi, E.; Sharifzadeh, G.R.; Ziaee, M. Comparing the Prevalence of HTLV-1 and Its Risk Factors in Prisoners with Intravenous and Non-Intravenous Drug Use in Birjand. Mod. Care J. 2019, 16, e95601. [Google Scholar] [CrossRef]
- Rafatpanah, H.; Hedayati-Moghaddam, M.R.; Fathimoghadam, F.; Bidkhori, H.R.; Shamsian, S.K.; Ahmadi, S.; Sohgandi, L.; Azarpazhooh, M.R.; Rezaee, S.A.; Farid, R.; et al. Prevalence of human T-cell lymphotropic virus type 2 infection in general population of Mashhad, Iran. J. Isfahan Med. Sch. 2015, 32, 2260–2267. [Google Scholar]
- Rafatpanah, H.; Fathimoghadam, F.; Shahabi, M.; Eftekharzadeh, I.; Hedayati-Moghaddam, M.; Valizadeh, N.; Tadayon, M.; Shamsian, S.A.; Bidkhori, H.; Miri, R.; et al. No Evidence of HTLV-II Infection Among Immonoblot Indeterminate Samples Using Nested PCR in Mashhad, Northeast of Iran. Iran J. Basic Med. Sci. 2013, 16, 229–234. [Google Scholar]
- Lowis, G. Epidemiologic Features of HTLV-II Serologic and Molecular Evidence. Ann. Epidemiol. 2002, 12, 46–66. [Google Scholar] [CrossRef]
- Husseini, A.A.; Saeed, K.M.I.; Yurdcu, E.; Sertoz, R.; Bozdayi, A.M. Epidemiology of blood-borne viral infections in Afghanistan. Arch. Virol. 2019, 164, 2083–2090. [Google Scholar] [CrossRef]
- Mousavi, S.H.; Khairkhah, N.; Bahri, T.D.; Anvar, A.; Saraji, A.A.; Behnava, B.; Alavian, S.M.; Namvar, A. First Report of Prevalence of Blood-Borne Viruses (HBV, HCV, HIV, HTLV-1 and Parvovirus B19) Among Hemophilia Patients in Afghanistan. Sci. Rep. 2019, 9, 7259. [Google Scholar] [CrossRef]
- Zaki, S.M.; Darwish, M.M.; Mahmoud, M.H. Sporadic carriers of human T-cell lymphotropic virus type 1 among blood donors in Egypt. Egypt. J. Med. Lab. Sci. 2007, 16, 77–87. [Google Scholar]
- Ibrahim, A.I.; Al-Musawi, Y.A.; Abdullah, A.I. Seroprevalence of HTLV-type-1 and type-2 among Blood Donors in Some Iraqi Provinces. Indian J. Forensic Med. Toxicol. 2020, 14, 2179–2184. [Google Scholar] [CrossRef]
- Souan, L.; Tout, F.; Siag, M.; Sughayer, M.A. Seroprevalence rates of transfusion-transmitted infections among blood donors in Jordan. J. Infect. Dev. Ctries. 2016, 10, 377–383. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ameen, R.; Sanad, N.; Al-Shemmari, S.; Siddique, I.; Chowdhury, R.I.; Al-Hamdan, S.; Al-Bashir, A. Prevalence of viral markers among first-time Arab blood donors in Kuwait. Transfusion 2005, 45, 1973–1980. [Google Scholar] [CrossRef] [PubMed]
- Ramia, S.; Klayme, S.; Naman, R. Infection with hepatitis B and C viruses and human retroviruses (HTLV-I and HIV) among high-risk Lebanese patients. Ann. Trop. Med. Parasitol. 2003, 97, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Tamim, H.; Musharrafieh, U.; Ramia, S.; Almawi, W.Y.; Al-Jisr, T.; Ayoub, T.; Nabulsi-Majzoub, M.; Kazma, H.; Baz, E.K. Is seroprevalence of HTLV-I/II among blood donors in Lebanon relevant? Am. J. Infect. Control 2004, 32, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, Z.O.; Al Malki, A.; Al Mutawa, H. Prevalence of Human T-Lymphotropic Virus (HTLV) Antibodies Among the Donor Population in the State of Qatar. Qatar Med. J. 2006, 2006, 9. [Google Scholar] [CrossRef]
- Aabdien, M.; Selim, N.; Himatt, S.; Hmissi, S.; Merenkov, Z.; AlKubaisi, N.; Abdel-Rahman, M.; Abdelmola, A.; Khelfa, S.; Farag, E.; et al. Prevalence and trends of transfusion transmissible infections among blood donors in the State of Qatar, 2013–2017. BMC Infect. Dis. 2020, 20, 617. [Google Scholar] [CrossRef]
- Niazi, S.K.; Bhatti, F.A.; Salamat, N. Seroprevalence of Human T-Cell Lymphotropic Virus-1/2 in Blood Donors in Northern Pakistan: Implications for Blood Donor Screening. J. Coll. Physicians Surg. Pak. JCPSP 2015, 25, 874–877. [Google Scholar]
- Taha, M.A.; Bashawri, L.A.; Ahmed, M.S.; Ahmed, M.A. Prevalence of antibodies to human T-lymphotropic viruses types I and II among healthy blood donors. Saudi Med. J. 2003, 24, 637–640. [Google Scholar]
- Fawaz, N.; Tamim, H.; Almawi, W. Low prevalence of antibodies to human T-lymphotropic virus-I/II among blood donors in Eastern Saudi Arabia. Am. J. Infect. Control 2005, 33, 189–191. [Google Scholar] [CrossRef]
- Balkhy, H.H.; Memish, Z.A.; Abed, E.; Qasem, L.; Bin Amer, A.; Masoud, S.; Hajeer, A.H. Saudi national guard donor screening for human T cell lymphotropic virus I/II: Time to use molecular biology techniques. Mil. Med. 2004, 169, 251–253. [Google Scholar] [CrossRef] [PubMed][Green Version]
- El-Hazmi, M. Prevalence of HBV, HCV, HIV-1, 2 and HTLV-I/II infections among blood donors in a teaching hospital in the Central region of Saudi Arabia. Saudi Med. J. 2004, 25, 26–33. [Google Scholar] [PubMed]
- Ul-Hassan, Z.; Al-Bahrani, A.T.; Panhotra, B.R. Prevalence of human T-lymphotropic virus type I and type II antibody among blood donors in Eastern Saudi Arabia. Saudi Med. J. 2004, 25, 1419–1422. [Google Scholar] [PubMed]
- Kawashti, M.I.; Hindawi, S.I.; Damanhouri, G.A.; Rowehy, N.G.; Bawazeer, M.M.; Alshawa, M. Serologial screening of human T cell lymphotropic virus I and II (HTLV I/II) in blood banks by immunoblotting and enzyme-immuno assays: To demand or to defeat? Egypt. J. Immunol. 2005, 12, 137–142. [Google Scholar]
- Hindawi, S.; Badawi, M.; Fouda, F.; Mallah, B.; Rajab, H.; Madani, T.A. Testing for HTLV 1 and HTLV 2 among blood donors in Western Saudi Arabia: Prevalence and cost considerations. Transfus. Med. 2018, 28, 60–64. [Google Scholar] [CrossRef]
- Almutairi, H.H.; AlAhmari, M.M.; Al-Zahran, B.H.; Abbas, I.S.; Al Ghamdi, J.S.; Raja’a, Y.A.; Sallam, T.A. Prevalence of serological markers and nucleic acid for blood-borne viral infections in blood donors in Al-Baha, Saudi Arabia. J. Infect. Dev. Ctries. 2016, 10, 619–625. [Google Scholar] [CrossRef]
- AlShehri, A. Human T Lymphotropic Virus-I (HTLV-I), the Causative Agent of Acute T-Cell Leukaemia/Lymphoma, Is Absent among Blood Donors in Aseer Region, Saudi Arabia. J. King Abdulaziz Univ. Sci. 2014, 26, 35–52. [Google Scholar] [CrossRef]
- Kilany, M.; Bin Dajem, S.M.; Ibrahim, Y.M.; Alshehri, A.; Aljeamelani, A.A.; Ibrahim, E.H. Seroprevalence of Anti-Treponemapallidum Antibodies (Syphilis) In Blood Donors in the Southern Area of Saudi Arabia. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 549–556. [Google Scholar]
- Almaiman, A.A.; Almaiman, S.H. Evaluation of Blood Donors and Transfusion Transmitted Infections and their Association with ABO and Rh Blood Groups in Unaizah, Saudi Arabia: A Retrospective Study. Int. J. Med. Res. Health Sci. 2018, 7, 143–150. [Google Scholar]
- Sarah, Y.A.E.G.A.; Sabry, A.E.G.A.E.H.E.S.; Maryam, A.A.-S. Seropositivity of TTIs among blood donors in Hail, Saudi Arabia, from 2014 to 2015. Asian Pac. J. Trop. Dis. 2016, 6, 141–146. [Google Scholar] [CrossRef]
- Alaidarous, M.; Choudhary, R.K.; Waly, M.I.; Mir, S.; Bin Dukhyil, A.; Banawas, S.S.; Alshehri, B.M. The prevalence of transfusion-transmitted infections and nucleic acid testing among blood donors in Majmaah, Saudi Arabia. J. Infect. Public Health 2018, 11, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Alabdulmonem, W.; Shariq, A.; Alqossayir, F.; AbaAlkhail, F.M.; Al-Musallam, A.Y.; Alzaaqi, F.O.; Aloqla, A.A.; Alodhaylah, S.A.; Alsugayyir, A.H.; Aldoubiab, R.K.; et al. Sero-prevalence ABO and Rh blood groups and their associated Transfusion-Transmissible Infections among Blood Donors in the Central Region of Saudi Arabia. J. Infect. Public Health 2020, 13, 299–305. [Google Scholar] [CrossRef]
- Alshehri, O.M.; Nahari, M.H.; Hassan, E.E.; Alqahtani, M.F.; Awaji, T.H. Prevalence of ABO, Rh and KELL Blood Group Types and Transfusion- Transmissible Infections (TTI) among Blood Donors in Najran City, Saudi Arabia. Biomed. Pharmacol. J. 2021, 14, 1065–1076. [Google Scholar] [CrossRef]
- Bitar, N.; El Hajj, H.; Houmani, Z.; Sabbah, A.; Otrock, Z.K.; Mahfouz, R.; Zaatari, G.; Bazarbachi, A. Adult T-cell leukemia/lymphoma in the Middle East: First report of two cases from Lebanon. Transfusion 2009, 49, 1859–1864. [Google Scholar] [CrossRef] [PubMed]
- Cassar, O.; Desrames, A.; Marçais, A.; Gout, O.; Taylor, G.P.; Hermine, O.; Soriano, V.; De Mendoza, C.; Dehan, O.; Le Mener, M.; et al. Multiple recombinant events in human T-cell Leukemia virus Type 1: Complete sequences of recombinant African strains. Emerg. Microbes Infect. 2020, 9, 913–923. [Google Scholar] [CrossRef]

| Population | Province (City, Direction in Country) | Study Year | Sample Size | Participants’ Age; Ranges (Mean ± SD) | Male to Female Ratio | Lab Techniques | Total Prevalence (%) | Prevalence by Sex (%) | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Males | Females | p-Value | |||||||||
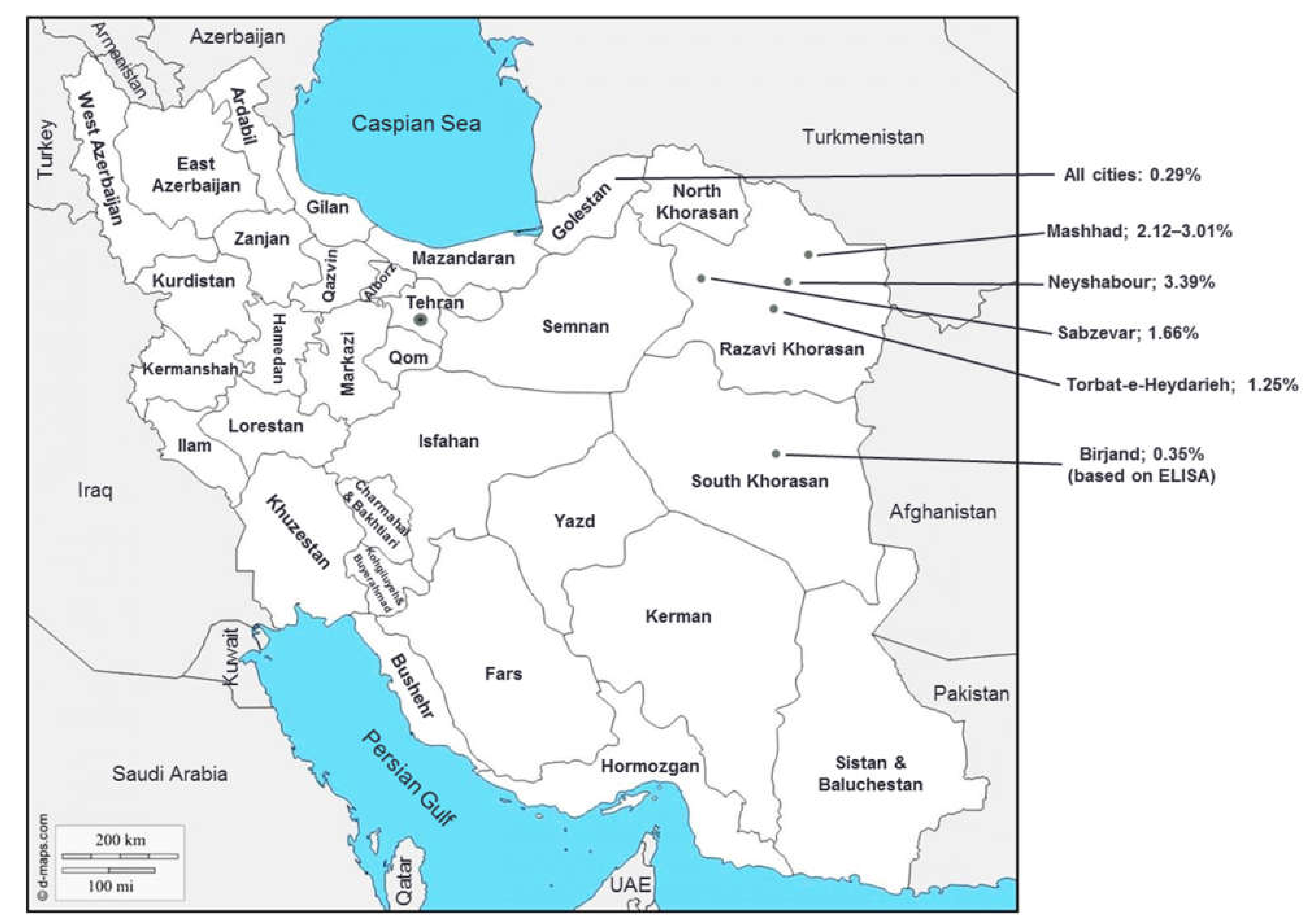
| General population | Golestan (North) | 2007 | 2034 | (38.7 ± 16.50) | 0.72 | ELISA, WB | 0.29 | 0.35 | 0.25 | 0.49 | [20] |
| Razavi Khorasan (Mashhad, Northeast) | ? | 9274 | ? | 0.67 | ELISA, PCR | 3.01 | 2.34 | 3.46 | 0.002 | [30] | |
| 2009 | 1654 | 1–90 | 0.83 | ELISA, WB, PCR | 2.12 | 1.46 | 2.66 | 0.093 | [21] | ||
| Razavi Khorasan (Neyshabour, Northeast) | 2002 | 1003 | 10–80 (32 ± ?) | 0.68 | ELISA, WB | 3.39 | 2.96 | 3.68 | 0.539 | [24] | |
| Razavi Khorasan (Sabzevar, Northeast) | 2008 | 1445 | 5–88 (36.6 ± 15.6) | 0.46 | ELISA, PCR | 1.66 | 2.42 | 1.31 | 0.128 | [19] | |
| Razavi Khorasan (Torbat-e-Heydarieh, Northeast) | 2011 | 400 | 14–89 (45.0 ± 16.8) | 1.05 | ELISA, PCR | 1.25 | 1.46 | 1.03 | 0.09 | [22] | |
| South Khorasan (East) | 2013–2014 | 3441 | 14–70 (38.7 ± 14.50) | 0.88 | ELISA | 0.35 | 0.25 | 0.44 | 0.34 | [23] | |
| Blood donors | Seven provinces | 2009–2013 | 1,864,489 | 18–65 (34.8 ± 10.4) | 10.32 | ELISA, WB | 0.10 | 0.09 | 0.18 | <0.001 | [31] |
| Razavi Khorasan (Northeast) | 628,667 | ? | ? | ELISA, WB | 0.21 | ? | ? | ? | |||
| West Azerbaijan (Northwest) | 307,422 | ? | ? | ELISA, WB | 0.07 | ? | ? | ? | |||
| North Khorasan (Northeast) | 79,035 | ? | ? | ELISA, WB | 0.06 | ? | ? | ? | |||
| Alborz (Center) | 264,340 | ? | ? | ELISA, WB | 0.06 | ? | ? | ? | |||
| South Khorasan (East) | 72,185 | ? | ? | ELISA, WB | 0.03 | ? | ? | ? | |||
| Gilan (North) | 373,227 | ? | ? | ELISA, WB | 0.02 | ? | ? | ? | |||
| Ardabil (Northwest) | 139,613 | ? | ? | ELISA, WB | 0.01 | ? | ? | ? | |||
| Seven provinces (East, North, Northeast, Northwest, Center)1 | 2010–2018 | 3,622,860 | 18–65 | 18.48 | ELISA, WB | 0.07 | 0.06 | 0.23 | <0.001 | [32] | |
| Bushehr (Southwest) | 2002–2003 | 22,740 | ? | 3.79 | ELISA, WB | 0.01 | 0.02 | 0 | - | [33] | |
| Charmahal and Bakhtiari (Southwest) | 2005–2006 | 800 | ? | ? | ELISA, WB | 0.50 | ? | ? | ? | [34] | |
| Fars (Shiraz) | ? | 500 | ? | 15.97 | ELISA | 0.20 | ? | ? | ? | [35] | |
| Golestan (North) | 2017 | 4226 | males: (37.6 ± 7.9), females: (38.3 ± 8.9) | ELISA, WB, PCR | 0.09 | ? | ? | ? | [36] | ||
| Hormozgan (South) | 2007–2008 | 1100 | ? | ? | ELISA, WB, PCR | 0.18 | ? | ? | ? | [37] | |
| Kermanshah (West) | 2015 | 470 | 10–59 | 9.0 | ELISA, WB | 0 | 0 | 0 | - | [38] | |
| 2011 | 1000 | (43.1 ± 19.8) | 0.71 | ELISA, WB | 0.50 | 0 | 0.86 | 0.079 | [39] | ||
| Mazandaran (Babol, North) | 2015–2016 | 503 | 19–61, males: (40.8 ± 9.8), females: (41.2 ± 11.4) | 24.15 | ELISA, PCR | 0.20 | 0.21 | 0 | - | [40] | |
| Razavi Khorasan (Mashhad, Northeast) | 2001–2002 | 60,892 | ? | ? | ELISA, WB | 0.66 | ? | ? | ? | [12] | |
| 2004–2006 | 232,648 | 18–65 (30 ± ?) | 9.88 | ELISA, WB | 0.45 | 0.42 | 0.76 | <0.001 | [41] | ||
| 2006–2008 | 250,582 | 17–63 (38.3 ± 10.8) | 11.74 | ELISA, WB | 0.40 | 0.36 | 0.88 | <0.001 | [42] | ||
| 2008–2009 | 79,687 | 17–65 | ? | ELISA, WB | 0.38 | ? | ? | ? | [43] | ||
| 2009–2010 | 165,860 | 17–59 (39.9 ± 10.5) | 13.71 | ELISA, WB | 0.26 | 0.23 | 0.70 | <0.001 | [44] | ||
| 2002–2013 | 983,000 | (35 ± 10 to 41 ± 11) | 14.63 | ELISA, WB | 0.30 | 0.28 | 0.48 | <0.001 | [45] | ||
| 2011–2013 | 174,662 | ? | 12.90 | ELISA, WB | 0.19 | 0.16 | 0.56 | <0.001 | [46] | ||
| Razavi Khorasan (Sabzevar, Northeast) | 2009–2012 | 35,067 | 17–59 (38.1 ± 11.8) | 9.06 | ELISA, WB | 0.14 | 0.13 | 0.26 | 0.095 | [47] | |
| South Khorasan (East) | 2005–2015 | 165,267 | males: (29.5 ± ?), females: (32.8 ± ?) | 11.55 | ELISA, WB | 0.03 | 0.03 | 0.02 | 0.038 | [48] | |
| Tehran (North) | ? | 2000 | ? | ? | ELISA, PCR | 0.05 | ? | ? | ? | [49] | |
| West Azerbaijan (Northwest) | 2005 | 2046 | (31.9 ± 10.2) | 14.04 | ELISA, WB | 0.34 | 0.26 | 1.47 | 0.074 | [50] | |
| 2009–2019 | 682,171 | 18–65 | 30.13 | ELISA, WB | 0.05 | 0.05 | 0.14 | <0.001 | [51] | ||
| Corneal donors | Nationwide | 2005–2007 | 5533 | 2–84 | 4.19 | ELISA, WB | 0.67 | 0.58 | 1.03 | 0.105 | [52] |
| Tissue donors | Nationwide | 2002–2007 | 1548 | 0–66 (29.1 ± 11.6) | 3.11 | ELISA | 1.61 | 1.45 | 2.12 | 0.369 | [53] |
| Pregnant women | Razavi Khorasan (Mashhad, Northeast) | 2010–2011 | 407 | (26 ± ?) | ? | ELISA, PCR | 1.47 | ? | ? | ? | [54] |
| People who referred to a medical lab | Razavi Khorasan (Neyshabour, Northeast) | 2009 | 483 | 5–84 (37.4 ± 15.4) | 0.26 | ELISA, WB | 7.25 | 10.31 | 6.49 | 0.196 | [27] |
| 2010–2014 | 8054 | males: (46 ± 3), females: (51 ± 3) | 0.24 | ELISA | 6.56 | 8.31 | 6.13 | 0.002 | [55] | ||
| 2013–2014 | 1169 2 | 2–35, (26.3 ± 5.4) | 0.14 | ELISA | 3.34 | 6.38 | 2.92 | 0.058 | [56] | ||
| 2011–2015 | 5724 3 | 15–40 (28.2 ± 6.0) | ? | ELISA | 2.04 | - | 2.04 | - | [57] | ||
| People who admitted to a referral hospital | Mazandaran (Sari, North) | 2009–2010 | 1200 | 1–76 | 0.55 | ELISA, WB | 0.08 | 0.24 | 0 | - | [58] |
| Razavi Khorasan (Northeast) | 2016–2017 | 758 4 | 0–14 (6.4 ± 1.8) | 1.14 | ELISA, PCR | 1.45 | ? | ? | ? | [59] | |
| Tehran (North) | 2009–2011 | 219 | 13–84 (39.9 ± 16.5) | 0.72 | ELISA | 1.83 | 1.09 | 2.36 | 0.641 | [60] | |
| Population | Province (City, Direction in Country) | Study Year | Sample Size | Participants’ Age; Ranges (Mean ± SD) | Male to Female Ratio | Lab Techniques | Prevalence (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Hemodialysis patients | Busher (Southwest) | 2003 | 101 | ? | ? | ELISA, WB | 0 | [76] |
| Charmahal and Bakhtiari (Southwest) | 2005 | 107 | 18–90, (65 ± ?) | ? | ELISA, WB | 6.54 | [69] | |
| Hormozgan (South) | 2007–2008 | 40 | ? | ? | ELISA, WB | 0 | [37] | |
| Kurdistan (Sanandaj, West) | 2010 | 65 | (45.1 ± ?) | ? | ELISA, WB | 0 | [77] | |
| Mazandaran (Sari and Ghaemshahr, North) | 2011 | 160 | (59.1 ± 14.7) | 1.0 | ELISA, WB | 0.63 | [78] | |
| Razavi Khorasan (Mashhad, Northeast) | 2009–2010 | 135 | males: (43.5 ± 12.5), females: (50.5 ± 13.2) | 0.99 | ELISA, PCR | 5.93 | [67] | |
| Razavi Khorasan (Neyshabour, Northeast) | 2012 | 138 | 12–84, (53.3 ± 17.9) | 1.23 | ELISA, WB | 14.49 | [68] | |
| South Khorasan (Birjand, East) | 2010 | 41 | (54.9 ± 16.5) | 2.15 | ELISA, WB | 2.44 | [79] | |
| Tehran (North) | ? | 150 | 24–88, (63.6 ± 13.4) | 1.34 | ELISA, WB | 0.67 | [80] | |
| 2016–2017 | 174 | 17–86, (56 ± ?) | 1.32 | ELISA, PCR | 1.15 | [81] | ||
| West Azerbaijan (Urmia, Northwest) | 2006 | 95 | (31.9 ± 10.2) | ? | ELISA, WB | 1.05 | [50] | |
| Thalassemia patients | Busher (Southwest) | 2003 | 455 | ? | ? | ELISA, WB | 3.08 | [76] |
| Charmahal and Bakhtiari (Southwest) | 2005 | 250 | 1–45, (25 ± ?) | ? | ELISA, WB | 6.80 | [69] | |
| Fars (Shiraz, South) | ? | 200 | ? | ? | ELISA | 3.00 | [35] | |
| Golestan (Gorgan, North) | 2004–2005 | 181 | 1–25, (14.1 ± 6.5) | 1.06 | ELISA, WB | 4.42 | [82] | |
| Hormozgan (South) | 2007–2008 | 163 | ? | ? | ELISA, WB, PCR | 3.07 | [37] | |
| Isfahan (Center) | 2007 | 150 | 1–49, (17.7 ± ?) | 1.42 | ELISA, WB | 3.33 | [83] | |
| 2012 | 67 | ? | ? | ELISA, PCR | 1.49 | [84] | ||
| Kermanshah (West) | 2011 | 116 | (16.8 ± 6.6) | 1.23 | ELISA, WB | 3.45 | [39] | |
| Kurdistan (Sanandaj, West) | 2010 | 46 | (13.3 ± ?) | ? | ELISA, WB | 2.17 | [77] | |
| Mazandaran (North) | 2009 | 288 | (21.5 ± 6.6) | 0.91 | ELISA, PCR | 1.39 | [85] | |
| Mazandaran (Tonekabon, North) | 2015 | 80 | ? | ? | ELISA, PCR | 2.50 | [86] | |
| Razavi Khorasan (Mashhad, Northeast) | 2007 | 360 | 1–52, (11.6 ± 0.5) | 1.38 | ELISA | 6.11 | [87] | |
| 2006–2013 | 100 | 5–46, (22.7 ± ?) | 1.38 | ELISA, PCR | 4.00 | [88] | ||
| Tehran (North) | 2003 | 175 | (18.1 ± 1.0) | 1.36 | ELISA, WB | 6.29 | [70] | |
| 2008–2010 | 257 | ? | 0.89 | ELISA, WB | 11.28 | [71] | ||
| ? | 100 | ? | ? | ELISA, PCR | 8.00 | [49] | ||
| Hemophilia patients | Busher (Southwest) | 2003 | 86 | ? | ? | ELISA, WB | 0 | [76] |
| South Khorasan (East) | ? | 80 | (21.3 ± 12.1) | 25.67 | ELISA, WB | 1.25 | [89] | |
| 2010–2012 | 108 | 14–85, (27.7 ± 16.4) | 14.43 | ELISA | 2.78 | [90] | ||
| West Azerbaijan (Northwest) | ? | 50 | (10.3 ± ?) | 6.14 | ELISA, WB | 0 | [91] | |
| Patients with combined factor 5 and 8 deficiency | Razavi Khorasan (Mashhad, Northeast) | 2007 | 24 | 6–61, (26.9 ± 15.1) | 2.0 | ELISA, WB | 0 | [92] |
| HIV-positive patients | Isfahan (Center) | 2010–2011 | 56 | (37.0 ± 8.7) | 5.22 | ELISA | 1.79 | [93] |
| Khuzestan (Ahwaz, Southwest) | 2001–2003 | 104 1 | ? | * | ELISA, WB | 16.35 | [73] | |
| Kurdistan (Sanandaj, West) | 2010 | 130 1 | (27.2 ± ?) | 239.0 | ELISA, WB | 0.77 | [77] | |
| Razavi Khorasan (Mashhad, Northeast) | ? | 20 | 22–50, (36.4 ± 8.6) | 4.0 | ELISA, WB | 15.0 | [72] | |
| Tehran (North) | ? | 100 | ? | ? | ELISA, PCR | 5.00 | [49] | |
| People who inject drugs | Isfahan (Center) | 2007–2008 | 150 2 | (30.7 ± 7.1) | 74.0 | ELISA | 2.67 | [94] |
| Khuzestan (Ahwaz, Southwest) | 2001–2003 | 104 1 | ? | * | ELISA, WB | 16.35 | [73] | |
| Kurdistan (Sanandaj, West) | 2010 | 130 1 | (27.2 ± ?) | 239.0 | ELISA, WB | 0.77 | [77] | |
| 2010 | 110 3 | ? | ? | ELISA, WB | 0.91 | [77] | ||
| Razavi Khorasan (Mashhad, Northeast) | 2001 | 101 4 | (32.8 ± 8.9) | ? | ELISA, WB | 51.49 | [12] | |
| 2007–2008 | 62 5 | (34.3 ± ?) | 30.0 | ELISA | 8.06 | [95] | ||
| Tehran (North) | ? | 180 | 9–67, (36.9 ± 9.2) | ? | ELISA, WB | 0 | [96] | |
| Prisoners | Razavi Khorasan (Northeast) | 2001 | 101 4 | (32.8 ± 8.9) | ? | ELISA, WB | 51.49 | [12] |
| 2008 | 1114 6 | males: (34.4 ± 10.9), females: (40.7 ± 14.2) | 8.13 | ELISA, PCR | 1.97 | [74] | ||
| South Khorasan (Birjand, East) | 2014–2015 | 300 7 | 20–78, (37.4 ± 9.4) | * | ELISA, WB | 0 | [97] |
| Population | Country (Province, City) | Study Year | Sample Size | Participants’ Age; Ranges (Mean ± SD) | Male to Female Ratio | Lab Techniques | Prevalence (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| General population | Afghanistan (5 provinces) 1 | 2015 | 466 | 25–70, (38.9 ± 12.0) | 0.98 | CMIA | 0.64 | [101] |
| Blood donors | Egypt (Cairo) | 2005–2006 | 90 | 18–47, (29.3 ± 6.5) | 6.50 | ELISA, PCR | 1.11 | [103] |
| Iraq (7 provinces) 2 | 2015 | 15,239 | 20–57, (36 ± ?) | ? | ELISA, CMIA | 0.26 | [104] | |
| Jordan (Amman) | 2009–2013 | 62,933 | 18–60 | ? | ELISA | 0 | [105] | |
| Kuwait (Nationwide) | 2002 | 12,798 | males: 20–56, (35 ± ?), females: 24–50, (30 ± ?) | ? | ELISA, CMIA | 0.01 | [106] | |
| Lebanon (Beirut) | 2000–2002 | 500 | (38.7 ± ?) | 3.17 | ELISA | 0 | [107] | |
| Lebanon (Nationwide) | 2001–2002 | 3529 | (30.0 ± 8.9) | 22.80 | ELISA, WB, PCR | 0.06 (WB), 0.03 (PCR) | [108] | |
| Qatar (Nationwide) | 1991–2003 | 124,266 | ? | ? | ELISA, WB | 0.0002 | [109] | |
| Qatar (Doha) | 2013–2017 | 190,509 | ? | 10.03 | CMIA, LIA | 0.18 | [110] | |
| Pakistan (Rawalpindi) | 2013 | 2100 | 18–60, (29.0 ± 9.3) | 62.64 | CMIA, LIA, PCR | 0.19 | [111] | |
| Saudi Arabia (Al-Khobar) | 1995–2001 | 23,493 | (33.8 ± ?) | ? | ELISA, WB | 0.05 | [112] | |
| Saudi Arabia (Dammam) | 1998–2001 | 13,443 | ? | ? | ELISA, WB | 0.06 | [113] | |
| Saudi Arabia (Riyadh) | 1999–2001 | 24,654 | ? | ? | ELISA, WB | 0.004 | [114] | |
| Saudi Arabia (Riyadh) | 2000–2002 | 24,173 | (33.8 ± ?) | 108.38 | ELISA, LIA | 0 | [115] | |
| Saudi Arabia (Al-Hasa) | 1997–2003 | 47,426 | 18–55 | ? | ELISA, WB | 0.01 | [116] | |
| Saudi Arabia (Jeddah) | ? | 30,000 | ? | ? | ELISA, WB | 0 | [117] | |
| 2006–2015 | 107,419 | ? | ? | CMIA, WB | 0 | [118] | ||
| Saudi Arabia (Al-Baha) | 2009–2011 | 2807 | 16–66 | * | ELISA | 0.04 | [119] | |
| Saudi Arabia (Aseer) | 2012 | 4432 | 18–60, (30 ± ?) | * | ELISA | 0 | [120] | |
| Saudi Arabia (Aseer) | 2012–2013 | 7267 | 18–60 | 278.50 | ELISA | 0 | [121] | |
| Saudi Arabia (Qassim/Unaizah) | 2013–2016 | 9460 | 18–48, (31.4 ± 8.7) | 26.82 | ELISA, PCR | 0.10 | [122] | |
| Saudi Arabia (Hail) | 2016 | 361 | <20 to >50 | ? | ELISA | 2.22 | [123] | |
| Saudi Arabia (Majmaah) | 2015–2017 | 3028 | 18–61 | 44.6 | ELISA | 0.20 | [124] | |
| Saudi Arabia (Buraidah) | 2017–2018 | 4590 | 16–65 | 4.88 | ELISA | 0 | [125] | |
| Saudi Arabia (Najran) | ? | 953 | ? | 237.25 | ELISA | 0 | [126] | |
| Hemophilia patients | Afghanistan (four cities) 3 | 2017 | 80 | 2–38, (13.7 ± 9.8) | ? | ELISA | 0 | [102] |
| Lebanon (Beirut) | 2000–2002 | 30 | (37.4 ± ?) | * | ELISA | 0 | [107] | |
| Malignancy patients with multiply transfusions | Lebanon (Beirut) | 2000–2002 | 65 | (30.5 ± ?) | 1.32 | ELISA | 0 | [107] |
| People who inject drugs | Lebanon (Beirut) | 2000–2002 | 40 | (50.3 ± ?) | 3.0 | ELISA | 0 | [107] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hedayati-Moghaddam, M.R.; Jafarzadeh Esfehani, R.; El Hajj, H.; Bazarbachi, A. Updates on the Epidemiology of the Human T-Cell Leukemia Virus Type 1 Infection in the Countries of the Eastern Mediterranean Regional Office of the World Health Organization with Special Emphasis on the Situation in Iran. Viruses 2022, 14, 664. https://doi.org/10.3390/v14040664
Hedayati-Moghaddam MR, Jafarzadeh Esfehani R, El Hajj H, Bazarbachi A. Updates on the Epidemiology of the Human T-Cell Leukemia Virus Type 1 Infection in the Countries of the Eastern Mediterranean Regional Office of the World Health Organization with Special Emphasis on the Situation in Iran. Viruses. 2022; 14(4):664. https://doi.org/10.3390/v14040664
Chicago/Turabian StyleHedayati-Moghaddam, Mohammad Reza, Reza Jafarzadeh Esfehani, Hiba El Hajj, and Ali Bazarbachi. 2022. "Updates on the Epidemiology of the Human T-Cell Leukemia Virus Type 1 Infection in the Countries of the Eastern Mediterranean Regional Office of the World Health Organization with Special Emphasis on the Situation in Iran" Viruses 14, no. 4: 664. https://doi.org/10.3390/v14040664
APA StyleHedayati-Moghaddam, M. R., Jafarzadeh Esfehani, R., El Hajj, H., & Bazarbachi, A. (2022). Updates on the Epidemiology of the Human T-Cell Leukemia Virus Type 1 Infection in the Countries of the Eastern Mediterranean Regional Office of the World Health Organization with Special Emphasis on the Situation in Iran. Viruses, 14(4), 664. https://doi.org/10.3390/v14040664







