Immunoinformatics-Aided Design of a Peptide Based Multiepitope Vaccine Targeting Glycoproteins and Membrane Proteins against Monkeypox Virus
Abstract
1. Introduction
2. Methodology
2.1. Retrieval and Analysis of Protein Sequence
2.2. Prediction of T-Cell and B-Cell Epitopes and to Determine Their Antigenicity, Ctoxicity, Allergenicity and Interferon-γ Activation Potential
2.3. Anlysing Epitope Conservancy
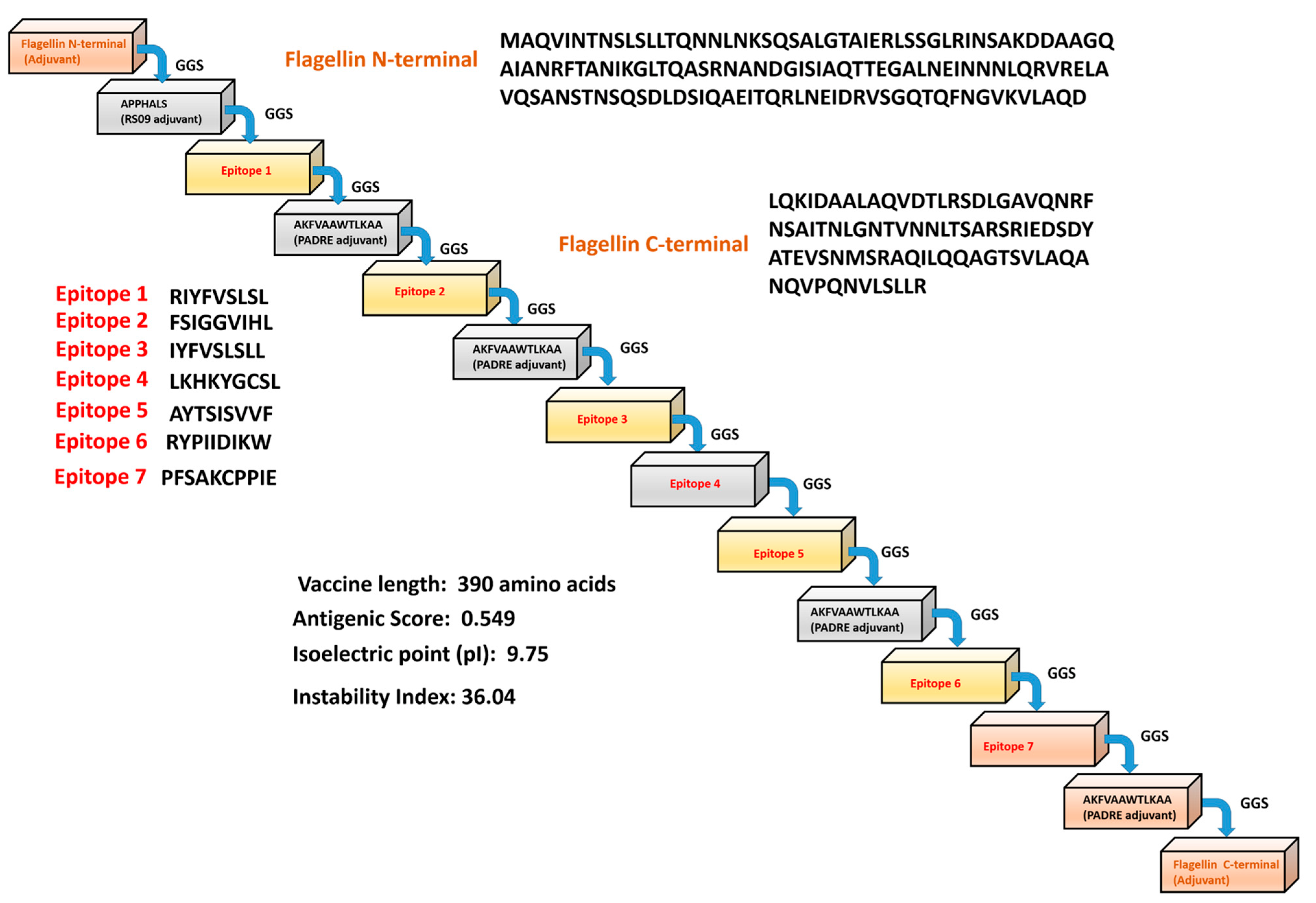
2.4. Designing a Vaccine Construct and to Determine Its Physiochemical Properties
2.5. Prediction of Immune Response Profile of MEV
2.6. Prediction of Binding Affinity of MPXV-MEV with TLR5 Using Molecular Modeling and Docking
2.7. Molecular Dynamic Simulations of MPXV-MEV Complexed with TLR5
3. Results
3.1. Protein Sequence Retrieval and Analysis
3.2. Prediction of T Cell and B Cell Epitopes and Analysis of Their Antigenic, Allergic, Toxic and Interferon-γ Activation Potential
3.3. Engineering Vaccine and to Determine Its Physiochemical Properties
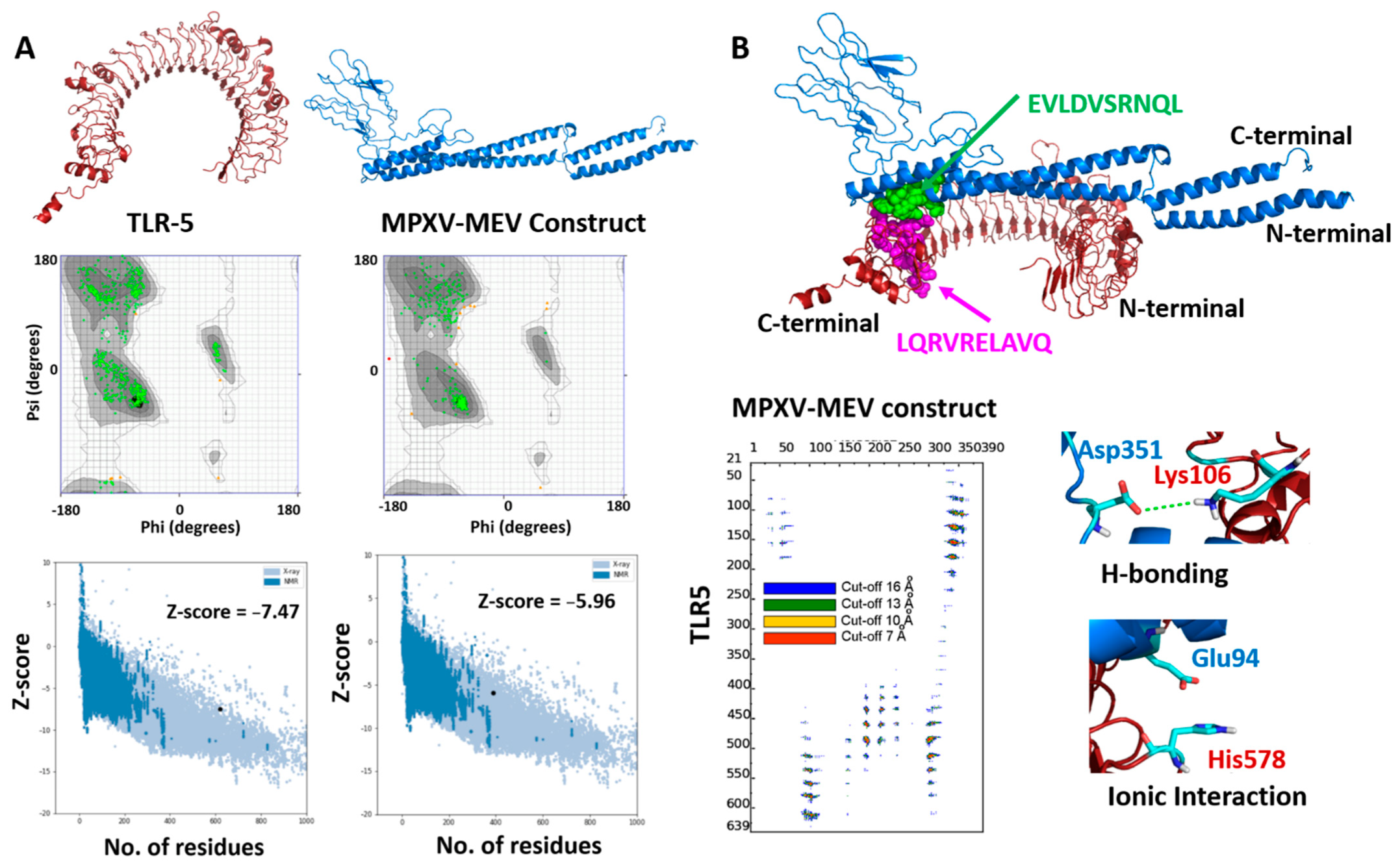
3.4. Performing Modeling and Docking of TLR5 MPXV-MEV Construct
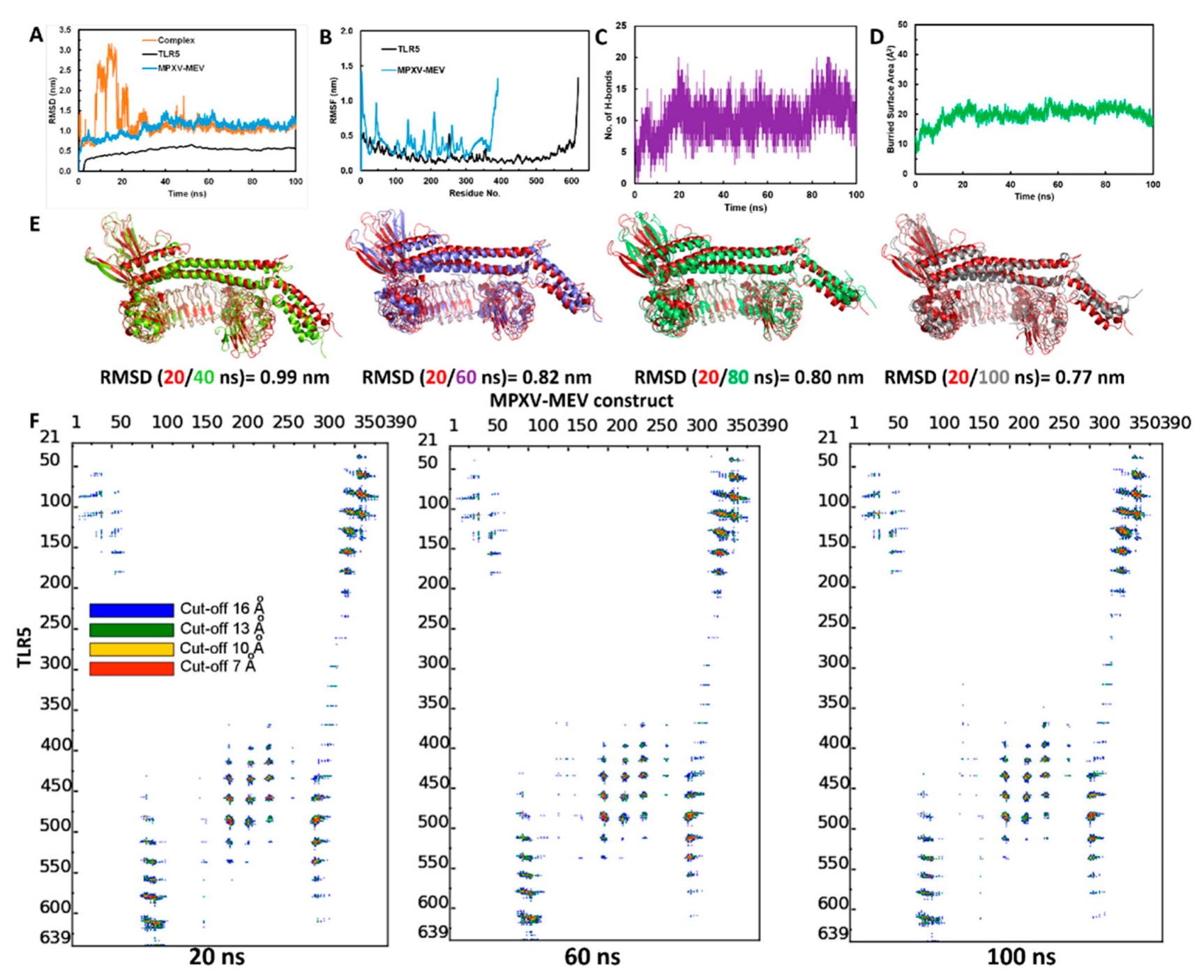
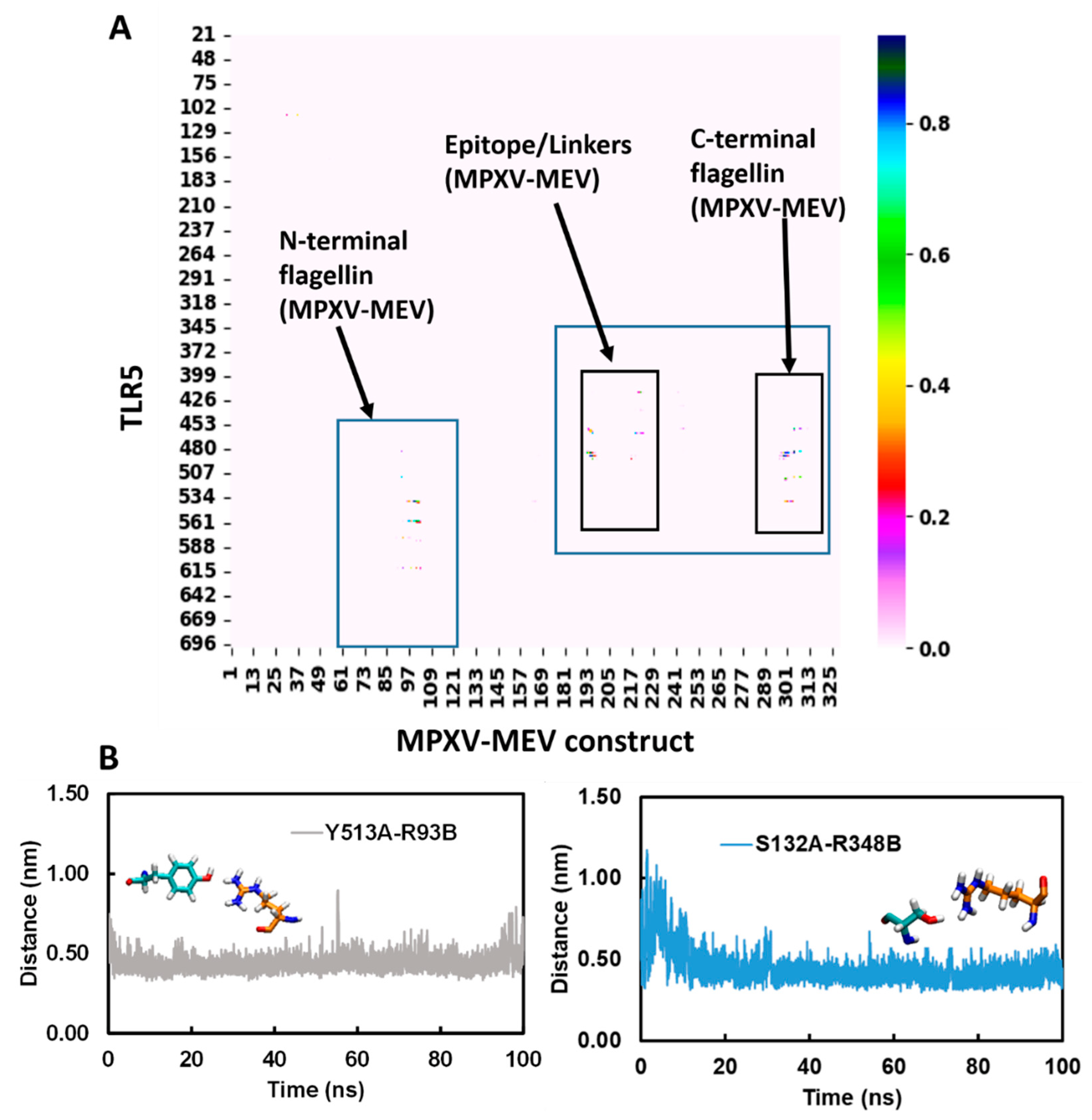
3.5. Structural Stability of the MPXV-MEV Complexed with TLR5
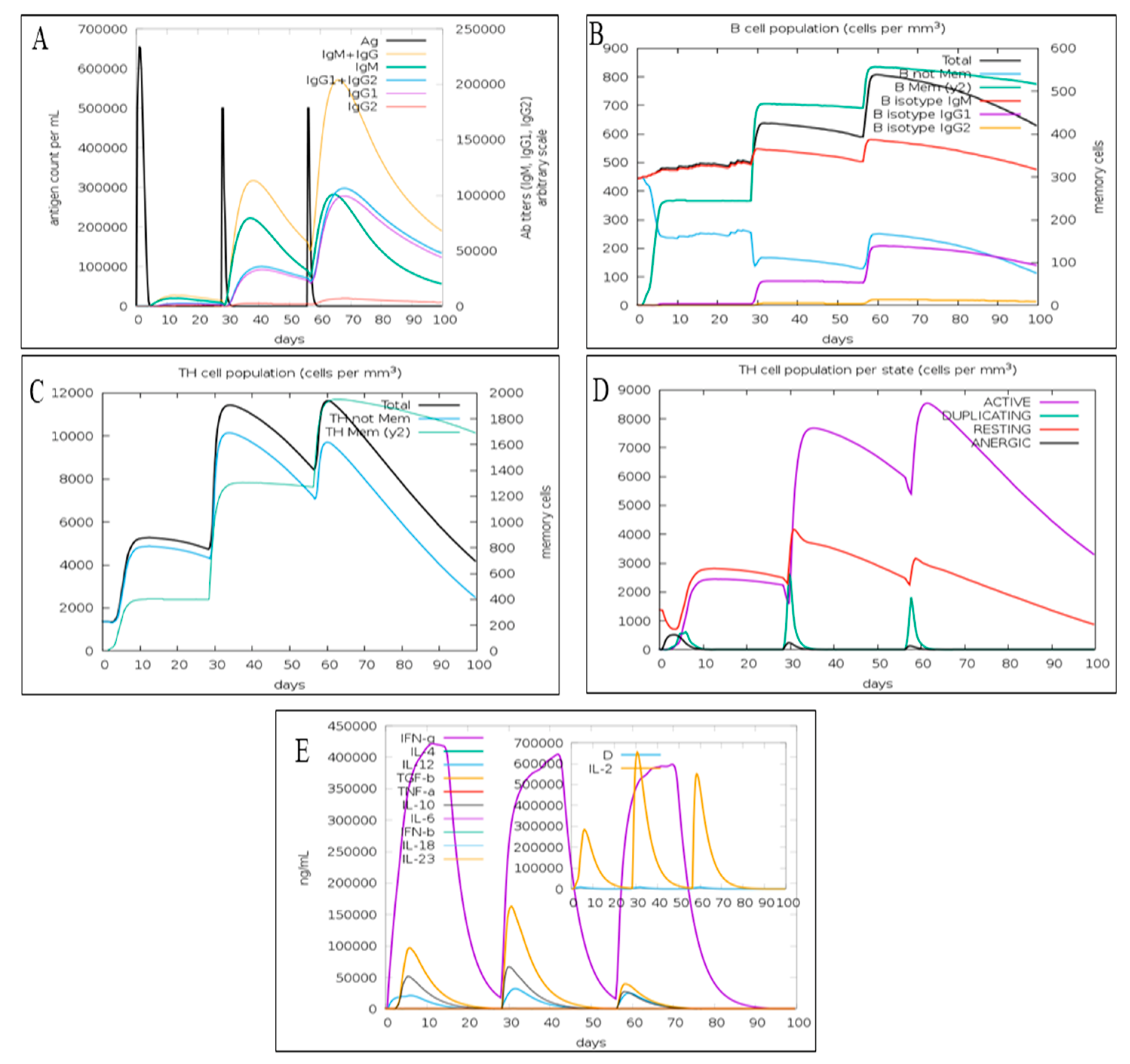
3.6. Immune Simulation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thornhill, J.P.; Barkati, S.; Walmsley, S.; Rockstroh, J.; Antinori, A.; Harrison, L.B.; Palich, R.; Nori, A.; Reeves, I.; Habibi, M.S.; et al. Monkeypox Virus Infection in Humans across 16 Countries—April-June 2022. N. Engl. J. Med. 2022, 8, 679–691. [Google Scholar] [CrossRef]
- Bunge, E.M.; Hoet, B.; Chen, L.; Lienert, F.; Weidenthaler, H.; Baer, L.R.; Steffen, R. The changing epidemiology of human monkeypox—A potential threat? A systematic review. PLoS Negl. Trop. Dis. 2022, 16, e0010141. [Google Scholar] [CrossRef]
- Ladnyj, I.D.; Ziegler, P.; Kima, E. A human infection caused by monkeypox virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593–597. [Google Scholar]
- Saxena, S.K.; Ansari, S.; Maurya, V.K.; Kumar, S.; Jain, A.; Paweska, J.T.; Tripathi, A.K.; Abdel-Moneim, A.S. Re-emerging human monkeypox: A major public-health debacle. J. Med. Virol. 2022. [Google Scholar] [CrossRef]
- Parker, S.; Buller, R.M. A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Futur. Virol. 2013, 8, 129–157. [Google Scholar] [CrossRef] [PubMed]
- Besombes, C.; Gonofio, E.; Konamna, X.; Selekon, B.; Grant, R.; Gessain, A.; Berthet, N.; Manuguerra, J.C.; Fontanet, A.; Nakoune, E. Intrafamily Transmission of Monkeypox Virus, Central African Republic, 2018. Emerg. Infect. Dis. 2019, 25, 1602–1604. [Google Scholar] [CrossRef] [PubMed]
- Mauldin, M.R.; McCollum, A.M.; Nakazawa, Y.J.; Mandra, A.; Whitehouse, E.R.; Davidson, W.; Zhao, H.; Gao, J.; Li, Y.; Doty, J.; et al. Exportation of Monkeypox Virus From the African Continent. J. Infect. Dis. 2020, 225, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.; Kantele, A.; Koopmans, M.; Asogun, D.; Yinka-Ogunleye, A.; Ihekweazu, C.; Zumla, A. Human Monkeypox: Epidemiologic and Clinical Characteristics, Diagnosis, and Prevention. Infect. Dis. Clin. N. Am. 2019, 33, 1027–1043. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Subramaniam, G.; Karuppanan, K. Human monkeypox outbreak in 2022. J. Med. Virol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.W.; Kabamba, J.; McCollum, A.M.; Lushima, R.S.; Wemakoy, E.O.; Tamfum, J.-J.M.; Nguete, B.; Hughes, C.M.; Monroe, B.P.; Reynolds, M.G. Vaccinating against monkeypox in the Democratic Republic of the Congo. Antivir. Res. 2018, 162, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.W.; Harms, T.J.; Reynolds, M.G.; Harrison, L.H. Use of Vaccinia Virus Smallpox Vaccine in Laboratory and Health Care Personnel at Risk for Occupational Exposure to Orthopoxviruses—Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2015. Morb. Mortal. Wkly. Rep. 2016, 65, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Sbrana, E.; Jordan, R.; Hruby, D.E.; Mateo, R.I.; Xiao, S.Y.; Siirin, M.; Newman, P.C.; Da Rosa, A.P.A.T.; Tesh, R.B. Efficacy of the Antipoxvirus Compound St-246 for Treatment of Severe Orthopoxvirus Infection. Am. J. Trop. Med. Hyg. 2007, 76, 768–773. [Google Scholar] [CrossRef] [PubMed]
- Jordan, R.; Goff, A.; Frimm, A.; Corrado, M.L.; Hensley, L.; Byrd, C.M.; Mucker, E.; Shamblin, J.; Bolken, T.C.; Wlazlowski, C.; et al. ST-246 Antiviral Efficacy in a Nonhuman Primate Monkeypox Model: Determination of the Minimal Effective Dose and Human Dose Justification. Antimicrob. Agents Chemother. 2009, 53, 1817–1822. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, J.; Filardo, T.D.; Morris, S.B.; Weiser, J.; Petersen, B.; Brooks, J.T. Interim Guidance for Prevention and Treatment of Monkeypox in Persons with HIV Infection—United States, August 2022. Morb. Mortal. Wkly. Rep. 2022, 71, 1023–1028. [Google Scholar] [CrossRef]
- Rizk, J.G.; Lippi, G.; Henry, B.M.; Forthal, D.N.; Rizk, Y. Prevention and Treatment of Monkeypox. Drugs 2022, 82, 957–963. [Google Scholar] [CrossRef]
- Johnston, S.C.; Lin, K.L.; Connor, J.H.; Ruthel, G.; Goff, A.; E Hensley, L. In vitro inhibition of monkeypox virus production and spread by Interferon-β. Virol. J. 2012, 9, 5. [Google Scholar] [CrossRef]
- Iizuka, I.; Ami, Y.; Suzaki, Y.; Nagata, N.; Fukushi, S.; Ogata, M.; Morikawa, S.; Hasegawa, H.; Mizuguchi, M.; Kurane, I.; et al. A Single Vaccination of Nonhuman Primates with Highly Attenuated Smallpox Vaccine, LC16m8, Provides Long-term Protection against Monkeypox. Jpn. J. Infect. Dis. 2017, 70, 408–415. [Google Scholar] [CrossRef]
- Denzler, K.L.; Babas, T.; Rippeon, A.; Huynh, T.; Fukushima, N.; Rhodes, L.; Silvera, P.M.; Jacobs, B.L. Attenuated NYCBH vaccinia virus deleted for the E3L gene confers partial protection against lethal monkeypox virus disease in cyno-molgus macaques. Vaccine 2011, 29, 9684–9690. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Sharma, R.; Grover, A. Immunoinformatics guided designing of a multi-epitope vaccine against Dengue and Zika. J. Biomol. Struct. Dyn. 2021, 1–15. [Google Scholar] [CrossRef]
- Dixit, N.K. Design of Monovalent and Chimeric Tetravalent Dengue Vaccine Using an Immunoinformatics Approach. Int. J. Pept. Res. Ther. 2021, 27, 2607–2624. [Google Scholar] [CrossRef]
- Ali, M.; Pandey, R.K.; Khatoon, N.; Narula, A.; Mishra, A.; Prajapati, V.K. Exploring dengue genome to construct a multi-epitope based subunit vaccine by utilizing immunoinformatics approach to battle against dengue infection. Sci. Rep. 2017, 7, 9232. [Google Scholar] [CrossRef] [PubMed]
- Cuspoca, A.F.; Diaz, L.L.; Acosta, A.F.; Penaloza, M.K.; Mendez, Y.R.; Clavijo, D.C.; Reyes, J.Y. An Immunoinformatics Approach for SARS-CoV-2 in Latam Populations and Multi-Epitope Vaccine Candidate Directed towards the World’s Population. Vaccines 2021, 9, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Joshi, A.; Akhtar, N.; Kaushik, V. Immunoinformatics designed T cell multi epitope dengue peptide vaccine derived from non structural proteome. Microb. Pathog. 2021, 150, 104728. [Google Scholar] [CrossRef]
- Kaushik, V.; Krishnan, S.; Gupta, L.R.; Kalra, U.; Shaikh, A.R.; Cavallo, L.; Chawla, M. Immunoinformatics Aided Design and In-Vivo Validation of a Cross-Reactive Peptide Based Multi-Epitope Vaccine Targeting Multiple Serotypes of Dengue Virus. Front. Immunol. 2022, 13, 865180. [Google Scholar] [CrossRef]
- Kaushik, V.; Jain, P.; Akhtar, N.; Joshi, A.; Gupta, L.R.; Grewal, R.K.; Oliva, R.; Shaikh, A.R.; Cavallo, L.; Chawla, M. Immunoinformatics-Aided Design and In Vivo Validation of a Peptide-Based Multiepitope Vaccine Targeting Canine Circovirus. ACS Pharmacol. Transl. Sci. 2022, 5, 679–691. [Google Scholar] [CrossRef]
- Doytchinova, I.A.; Flower, D.R. VaxiJen: A server for prediction of protective antigens, tumour antigens and subunit vaccines. BMC Bioinform. 2007, 8, 4. [Google Scholar] [CrossRef]
- Dimitrov, I.; Naneva, L.; Doytchinova, I.; Bangov, I. AllergenFP: Allergenicity prediction by descriptor fingerprints. Bioinformatics 2013, 30, 846–851. [Google Scholar] [CrossRef]
- Reynisson, B.; Alvarez, B.; Paul, S.; Peters, B.; Nielsen, M. NetMHCpan-4.1 and NetMHCIIpan-4.0: Improved predictions of MHC antigen presentation by concurrent motif deconvolution and integration of MS MHC eluted ligand data. Nucleic Acids Res. 2020, 48, W449–W454. [Google Scholar] [CrossRef]
- Zhang, L.M.; Udaka, K.; Mamitsuka, H.; Zhu, S.F. Toward more accurate pan-specific MHC-peptide binding prediction: A review of current methods and tools. Brief. Bioinform. 2012, 13, 350–364. [Google Scholar] [CrossRef]
- Lin, H.H.; Zhang, G.L.; Tongchusak, S.; Reinherz, E.L.; Brusic, V. Evaluation of MHC-II peptide binding predic-tion servers: Applications for vaccine research. BMC Bioinform. 2008, 9, S22. [Google Scholar] [CrossRef]
- Larsen, J.E.P.; Lund, O.; Nielsen, M. Improved method for predicting linear B-cell epitopes. Immunome Res. 2006, 2, 2. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dhanda, S.K.; Vir, P.; Raghava, G.P.S. Designing of interferon-gamma inducing MHC class-II binders. Biol. Direct 2013, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kapoor, P.; Chaudhary, K.; Gautam, A.; Kumar, R.; Raghava, G.P.S.; Open Source Drug Discovery Consortium. In Silico Approach for Predicting Toxicity of Peptides and Proteins. PLoS ONE 2013, 8, e73957. [Google Scholar] [CrossRef] [PubMed]
- Bui, H.H.; Sidney, J.; Li, W.; Fusseder, N.; Sette, A. Development of an epitope conservancy analysis tool to facilitate the design of epitope-based diagnostics and vaccines. BMC Bioinform. 2007, 8, 361. [Google Scholar] [CrossRef]
- Gupta, S.K.; Bajwa, P.; Deb, R.; Chellappa, M.M.; Dey, S. Flagellin A Toll-Like Receptor 5 Agonist as an Adjuvant in Chicken Vaccines. Clin. Vaccine Immunol. 2014, 21, 261–270. [Google Scholar] [CrossRef]
- Forstneric, V.; Ivicak-Kocjan, K.; Plaper, T.; Jerala, R.; Bencina, M. The role of the C-terminal D0 domain of flagellin in activation of Toll like receptor 5. PLoS Pathog. 2017, 13, e1006574. [Google Scholar] [CrossRef]
- La Rosa, C.; Longmate, J.; Lacey, S.F.; Kaltcheva, T.; Sharan, R.; Marsano, D.; Kwon, P.; Drake, J.; Williams, B.; Denison, S.; et al. Clinical Evaluation of Safety and Immunogenicity of PADRE-Cytomegalovirus (CMV) and Tetanus-CMV Fusion Peptide Vaccines With or Without PF03512676 Adjuvant. J. Infect. Dis. 2012, 205, 1294–1304. [Google Scholar] [CrossRef]
- Gasteiger, E.; Hoogland, C.; Gattiker, A.; Duvaud, S.; Wilkins, M.; Appel, R.; Bairoch, A. The Proteomics Protocols Handbook; Humana Press Inc.: Totowa, NJ, USA, 2005. [Google Scholar]
- Rapin, N.; Lund, O.; Bernaschi, M.; Castiglione, F. Computational Immunology Meets Bioinformatics: The Use of Prediction Tools for Molecular Binding in the Simulation of the Immune System. PLoS ONE 2010, 5, e9862. [Google Scholar] [CrossRef]
- Castiglione, F.; Mantile, F.; De Berardinis, P.; Prisco, A. How the Interval between Prime and Boost Injection Af-fects the Immune Response in a Computational Model of the Immune System. Comput. Math. Methods Med. 2012, 2012, 842329. [Google Scholar] [CrossRef]
- Robinson, C.L.; Romero, J.R.; Kempe, A.; Pellegrini, C. Advisory Committee on Immunization Practices (ACIP) Child/Adolescent Immunization Work Group Advisory Committee on Immunization Practices Recommended Immunization Schedule for Children and Adolescents Aged 18 Years or Younger—United States, 2017. MMWR. Morb. Mortal. Wkly. Rep. 2017, 66, 134–135. [Google Scholar] [CrossRef]
- Safavi, A.; Kefayat, A.; Abiri, A.; Mahdevar, E.; Behnia, A.H.; Ghahremani, F. In silico analysis of transmembrane protein 31 (TMEM31) antigen to design novel multiepitope peptide and DNA cancer vaccines against melanoma. Mol. Immunol. 2019, 112, 93–102. [Google Scholar] [CrossRef]
- Safavi, A.; Kefayat, A.; Mahdevar, E.; Abiri, A.; Ghahremani, F. Exploring the out of sight antigens of SARS-CoV-2 to design a candidate multi-epitope vaccine by utilizing immunoinformatics approaches. Vaccine 2020, 38, 7612–7628. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Islam, S.; Tankhilevich, E.; Sternberg, M.J.E. The AlphaFold Database of Protein Structures: A Biologist’s Guide. J. Mol. Biol. 2022, 434, 167336. [Google Scholar] [CrossRef] [PubMed]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Wiederstein, M.; Sippl, M.J. ProSA-web: Interactive web service for the recognition of errors in three-dimensional structures of proteins. Nucleic Acids Res. 2007, 35, W407–W410. [Google Scholar] [CrossRef] [PubMed]
- van Zundert, G.C.P.; Rodrigues, J.; Trellet, M.; Schmitz, C.; Kastritis, P.L.; Karaca, E.; Melquiond, A.S.J.; van Dijk, M.; de Vries, S.J.; Bonvin, A. The HADDOCK2.2 Web Server: User-Friendly Integrative Modeling of Biomolecular Com-plexes. J. Mol. Biol. 2016, 428, 720–725. [Google Scholar]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 2015, 1–2, 19–25. [Google Scholar] [CrossRef]
- Lindorff-Larsen, K.; Piana, S.; Palmo, K.; Maragakis, P.; Klepeis, J.L.; Dror, R.O.; Shaw, D.E. Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins Struct. Funct. Bioinform. 2010, 78, 1950–1958. [Google Scholar] [CrossRef]
- Joung, I.S.; Cheatham, T.E. Determination of alkali and halide monovalent ion parameters for use in explicitly solvated biomolecular simulations. J. Phys. Chem. B 2008, 112, 9020–9041. [Google Scholar] [CrossRef]
- Parrinello, M.; Rahman, A. Polymorphic transitions in single crystals: A new molecular dynamics method. J. Appl. Phys. 1981, 52, 7182–7190. [Google Scholar] [CrossRef]
- Darden, T.; York, D.; Pedersen, L. Particle Mesh Ewald—An N.log(N) Method for Ewald Sums in Large Systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef]
- Hess, B.; Bekker, H.; Berendsen, H.J.C.; Fraaije, J.G.E.M. LINCS: A linear constraint solver for molecular simula-tions. J. Comput. Chem. 1997, 18, 1463–1472. [Google Scholar] [CrossRef]
- Taheriniya, S.; Behboodi, Z. Comparing green chemical methods and chemical methods for the synthesis of titani-um dioxide naoparticles. Int. J. Pharm. Sci. Res. 2016, 7, 4927–4932. [Google Scholar]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 27–38. [Google Scholar] [CrossRef]
- Oliva, R.; Shaikh, A.R.; Petta, A.; Vangone, A.; Cavallo, L. D936Y and Other Mutations in the Fusion Core of the SARS-CoV-2 Spike Protein Heptad Repeat 1: Frequency, Geographical Distribution, and Structural Effect. Molecules 2021, 26, 2622. [Google Scholar] [PubMed]
- Grewal, R.K.; Shaikh, A.R.; Gorle, S.; Kaur, M.; Videira, P.A.; Cavallo, L.; Chawla, M. Structural Insights in Mammalian Sialyltransferases and Fucosyltransferases: We Have Come a Long Way, but It Is Still a Long Way Down. Molecules 2021, 26, 5203. [Google Scholar] [CrossRef]
- Chawla, M.; Credendino, R.; Poater, A.; Oliva, R.; Cavallo, L. Structural Stability, Acidity, and Halide Selectivity of the Fluoride Riboswitch Recognition Site. J. Am. Chem. Soc. 2014, 137, 299–306. [Google Scholar] [CrossRef]
- Chawla, M.; Gorle, S.; Shaikh, A.R.; Oliva, R.; Cavallo, L. Replacing thymine with a strongly pairing fifth Base: A combined quantum mechanics and molecular dynamics study. Comput. Struct. Biotechnol. J. 2021, 19, 1312–1324. [Google Scholar] [CrossRef]
- Kalra, K.; Gorle, S.; Cavallo, L.; Oliva, R.; Chawla, M. Occurrence and stability of lone pair-pi and OH-pi interac-tions between water and nucleobases in functional RNAs. Nucleic Acids. Res. 2020, 48, 5825–5838. [Google Scholar]
- Jacchieri, S.G.; Torquato, R.; Brentani, R.R. Structural Study of Binding of Flagellin by Toll-Like Receptor 5. J. Bacteriol. 2003, 185, 4243–4247. [Google Scholar] [CrossRef] [PubMed]
- Vangone, A.; Oliva, R.; Cavallo, L. CONS-COCOMAPS: A novel tool to measure and visualize the conservation of inter-residue contacts in multiple docking solutions. BMC Bioinform. 2012, 13, S19. [Google Scholar] [CrossRef] [PubMed]
- Vangone, A.; Spinelli, R.; Scarano, V.; Cavallo, L.; Oliva, R. COCOMAPS: A web application to analyze and visu-alize contacts at the interface of biomolecular complexes. Bioinformatics 2011, 27, 2915–2916. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Azeim, S.; Chermak, E.; Vangone, A.; Oliva, R.; Cavallo, L. MDcons: Intermolecular contact maps as a tool to analyze the interface of protein complexes from molecular dynamics trajectories. BMC Bioinform. 2014, 15, S1. [Google Scholar] [CrossRef] [PubMed]
- Barradas-Bautista, D.; Cao, Z.; Cavallo, L.; Oliva, R. The CASP13-CAPRI targets as case studies to illustrate a novel scoring pipeline integrating CONSRANK with clustering and interface analyses. BMC Bioinform. 2020, 21, 262. [Google Scholar] [CrossRef]
- Chermak, E.; Donato, R.; Lensink, M.F.; Petta, A.; Serra, L.; Scarano, V.; Cavallo, L.; Oliva, R. Introducing a Clus-tering Step in a Consensus Approach for the Scoring of Protein-Protein Docking Models. PLoS ONE 2016, 11, e0166460. [Google Scholar] [CrossRef] [PubMed]
- Oliva, R.; Chermak, E.; Cavallo, L. Analysis and Ranking of Protein-Protein Docking Models Using Inter-Residue Contacts and Inter-Molecular Contact Maps. Molecules 2015, 20, 12045–12060. [Google Scholar] [CrossRef]
- Vangone, A.; Cavallo, L.; Oliva, R. Using a consensus approach based on the conservation of inter-residue con-tacts to rank CAPRI models. Proteins-Struct. Funct. Bioinform. 2013, 81, 2210–2220. [Google Scholar] [CrossRef] [PubMed]
- Oliva, R.; Vangone, A.; Cavallo, L. Ranking multiple docking solutions based on the conservation of inter-residue contacts. Proteins Struct. Funct. Bioinform. 2013, 81, 1571–1584. [Google Scholar] [CrossRef]
- Silva, N.; de Oliveira, J.; Kroon, E.; Trindade, G.; Drumond, B. Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses. Viruses 2020, 13, 43. [Google Scholar] [CrossRef] [PubMed]
- Kugelman, J.R.; Johnston, S.C.; Mulembakani, P.M.; Kisalu, N.; Lee, M.S.; Koroleva, G.; McCarthy, S.E.; Gestole, M.C.; Wolfe, N.D.; Fair, J.N.; et al. Genomic Variability of Monkeypox Virus among Humans, Democratic Republic of the Congo. Emerg. Infect. Dis. 2014, 20, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Guarner, J.; Johnson, B.J.; Paddock, C.D.; Shieh, W.-J.; Goldsmith, C.S.; Reynolds, M.; Damon, I.K.; Regnery, R.L.; Zaki, S.R. The Veterinary Monkeypox Virus Working Group Monkeypox Transmission and Pathogenesis in Prairie Dogs. Emerg. Infect. Dis. 2004, 10, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef]
- Hutson, C.L.; Nakazawa, Y.J.; Self, J.; Olson, V.A.; Regnery, R.L.; Braden, Z.; Weiss, S.; Malekani, J.; Jackson, E.; Tate, M.; et al. Laboratory Investigations of African Pouched Rats (Cricetomysgambianus) as a Potential Reservoir Host Species for Monkeypox Virus. PLoS Negl. Trop. Dis. 2015, 9, e0004013. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; Leggat, P.A. Human Monkeypox: Current State of Knowledge and Implications for the Future. Trop. Med. Infect. Dis. 2016, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Yokote, H.; Shinmura, Y.; Kanehara, T.; Maruno, S.; Kuranaga, M.; Matsui, H.; Hashizume, S. Safety of Attenuat-ed Smallpox Vaccine LC16m8 in Immunodeficient Mice. Clin. Vaccine Immunol. 2014, 21, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.S.; Gurwith, M.; Dekker, C.L.; Frey, S.E.; Edwards, K.M.; Kenner, J.; Lock, M.; Empig, C.; Morikawa, S.; Saijo, M.; et al. Safety and Immunogenicity of LC16m8, an Attenuated Smallpox Vaccine in Vaccinia-Naive Adults. J. Infect. Dis. 2011, 204, 1395–1402. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.N.; Deng, Y.; Huang, B.Y.; Han, D.; Wang, W.; Huang, M.J.; Zhai, C.C.; Zhao, Z.M.; Yang, R.; Zhao, Y.; et al. DNA Vaccines Expressing the Envelope and Membrane Proteins Provide Partial Protection Against SARS-CoV-2 in Mice. Front. Immunol. 2022, 13, 827605. [Google Scholar] [CrossRef] [PubMed]
- Sluder, A.E.; Paul, S.R.; Moise, L.; Dold, C.; Richard, G.; Silva-Reyes, L.; Baeten, L.A.; Scholzen, A.; Reeves, P.M.; Pollard, A.J.; et al. Evaluation of a Human T Cell-Targeted Multi-Epitope Vaccine for Q Fever in Animal Models of Coxiella burnetii Immunity. Front. Immunol. 2022, 13, 901372. [Google Scholar] [CrossRef] [PubMed]
- Safavi, A.; Kefayat, A.; Sotoodehnejadnematalahi, F.; Salehi, M.; Modarressi, M.H. In Silico Analysis of Synap-tonemal Complex Protein 1 (SYCP1) and Acrosin Binding Protein (ACRBP) Antigens to Design Novel Multiepitope Peptide Cancer Vaccine Against Breast Cancer. Int. J. Pept. Res. Ther. 2019, 25, 1343–1359. [Google Scholar] [CrossRef]
- Sanami, S.; Azadegan-Dehkordi, F.; Rafieian-Kopaei, M.; Salehi, M.; Ghasemi-Dehnoo, M.; Mahooti, M.; Alizadeh, M.; Bagheri, N. Design of a multi-epitope vaccine against cervical cancer using immunoinformatics approaches. Sci. Rep. 2021, 11, 12397. [Google Scholar] [CrossRef] [PubMed]
- Shey, R.A.; Ghogomu, S.M.; Esoh, K.K.; Nebangwa, N.D.; Shintouo, C.M.; Nongley, N.F.; Asa, B.F.; Ngale, F.N.; Vanhamme, L.; Souopgui, J. In-silico design of a multi-epitope vaccine candidate against onchocerciasis and related filarial dis-eases. Sci. Rep. 2019, 9, 4409. [Google Scholar] [CrossRef]
- Tarang, S.; Kesherwani, V.; LaTendresse, B.; Lindgren, L.; Rocha-Sanchez, S.M.; Weston, M.D. In silico Design of a Multivalent Vaccine against Candida albicans. Sci. Rep. 2020, 10, 1066. [Google Scholar] [CrossRef] [PubMed]
- Soltan, M.A.; Eldeen, M.A.; Elbassiouny, N.; Kamel, H.L.; Abdelraheem, K.M.; Abd El-Gayyed, H.; Gouda, A.M.; Sheha, M.F.; Fayad, E.; Abu Ali, O.A.; et al. In Silico Designing of a Multitope Vaccine against Rhizopus microsporus with Potential Activity against Other Mucormycosis Causing Fungi. Cells 2021, 10, 3014. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, N.; Joshi, A.; Singh, J.; Kaushik, V. Design of a novel and potent multivalent epitope based human cyto-megalovirus peptide vaccine: An immunoinformatics approach. J. Mol. Liq. 2021, 335, 116586. [Google Scholar] [CrossRef]
- Dar, H.A.; Ismail, S.; Waheed, Y.; Ahmad, S.; Jamil, Z.; Aziz, H.; Hetta, H.F.; Muhammad, K. Designing a multi-epitope vaccine against Mycobacteroidesabscessus by pangenome-reverse vaccinology. Sci. Rep. 2021, 11, 11197. [Google Scholar] [CrossRef]
- Kumar, P.; Lata, S.; Shankar, U.N.; Akif, M. Immunoinformatics-Based Designing of a Multi-Epitope Chimeric Vaccine From Multi-Domain Outer Surface Antigens of Leptospira. Front. Immunol. 2021, 12, 735373. [Google Scholar] [CrossRef] [PubMed]
- Qamar, M.T.U.; Saleem, S.; Ashfaq, U.A.; Bari, A.; Anwar, F.; Alqahtani, S. Epitope-based peptide vaccine design and target site depiction against Middle East Respiratory Syndrome Coronavirus: An immune-informatics study. J. Transl. Med. 2019, 17, 362. [Google Scholar] [CrossRef]
- Abdi, S.A.H.; Ali, A.; Sayed, S.F.; Abutahir Ali, A.; Alam, P. Multi-Epitope-Based Vaccine Candidate for Mon-keypox: An In Silico Approach. Vaccines 2022, 10, 1564. [Google Scholar] [CrossRef] [PubMed]
- Shantier, S.W.; Mustafa, M.I.; Abdelmoneim, A.H.; Fadl, H.A.; Elbager, S.G.; Makhawi, A.M. Novel multi epitope-based vaccine against monkeypox virus: Vaccinomic approach. Sci. Rep. 2022, 12, 15983. [Google Scholar] [CrossRef] [PubMed]
- Aiman, S.; Alhamhoom, Y.; Ali, F.; Rahman, N.; Rastrelli, L.; Khan, A.; Farooq, Q.U.A.; Ahmed, A.; Khan, A.; Li, C. Multi-epitope chimeric vaccine design against emerging Monkeypox virus via reverse vaccinology techniques- a bioinformatics and immunoinformatics approach. Front. Immunol. 2022, 13, 985450. [Google Scholar] [CrossRef]
- Ullah, A.; Shahid, F.A.; Ul Haq, M.; ul Qamar, M.T.; Irfan, M.; Shaker, B.; Ahmad, S.; Alrumaihi, F.; Allemailem, K.S.; Alma-troudi, A. An integrative reverse vaccinology, immunoinformatic, docking and simulation approaches towards design-ing of multi-epitopes based vaccine against monkeypox virus. J. Biomol. Struct. Dyn. 2022, 1–14. [Google Scholar] [CrossRef]
- Song, H.; Sidney, J.; Wiseman, R.W.; Josleyn, N.; Cohen, M.; E Blaney, J.; Jahrling, P.B.; Sette, A. Characterizing monkeypox virus specific CD8+ T cell epitopes in rhesus macaques. Virology 2013, 447, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Moise, L.; Buller, R.M.; Schriewer, J.; Lee, J.; Frey, S.E.; Weiner, D.B.; Martin, W.; De Groot, A.S. VennVax, a DNA-prime, peptide-boost multi-T-cell epitope poxvirus vaccine, induces protective immunity against vaccinia infection by T cell response alone. Vaccine 2011, 29, 501–511. [Google Scholar] [CrossRef]
- Harrison, C. Monkeypox response relies on three vaccine suppliers. Nat. Biotechnol. 2022, 40, 1306–1307. [Google Scholar] [CrossRef]
- Banerjee, N.; Mukhopadhyay, S. Viral glycoproteins: Biological role and application in diagnosis. Virus Dis. 2016, 27, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Brown, H.M.; Hwang, S. Direct Antiviral Mechanisms of Interferon-Gamma. Immune Netw. 2018, 18, e33. [Google Scholar] [CrossRef]
| GenBank Protein ID | Protein Name | Length (Amino Acids) | Vaxijen Score | Allergen (AllergenFP) |
|---|---|---|---|---|
| AIE40790.1 | putative membrane-associated glycoprotein | 1880 | 0.5262 (Antigenic) | Allergen |
| AIE40786.1 | IFN-alpha/beta-receptor-like secreted glycoprotein | 352 | 0.5453 (Antigenic) | Non-allergen |
| AIE40780.1 | bifunctional 21 kDa precursor protein of 18 kDa membrane protein | 182 | 0.4395 (Antigenic) | Allergen |
| AIE40778.1 | EEV type-I membrane glycoprotein | 317 | 0.5786 (Antigenic) | Non-allergen |
| AIE40774.1 | bifunctional hemagglutinin/type-I membrane glycoprotein | 313 | 0.4638 (Antigenic) | Allergen |
| AIE40766.1 | putative type-I membrane glycoprotein | 196 | 0.5230 (Antigenic) | Non-allergen |
| AIE40764.1 | bifunctional secreted glycoprotein | 221 | 0.3864 (Non-antigenic) | Allergen |
| AIE40763.1 | CD47-like putative membrane protein | 277 | 0.4324 (Antigenic) | Non-allergen |
| AIE40759.1 | EEV glycoprotein | 168 | 0.3728 (Non-antigenic) | Non-allergen |
| AIE40758.1 | bifunctional EEV membrane phosphoglycoprotein | 181 | 0.4998 (Antigenic) | Allergen |
| AIE40739.1 | IV and IMV membrane protein | 53 | 0.7480 (Antigenic) | Non-allergen |
| AIE40738.1 | phosphorylated IMV membrane protein | 90 | 0.4759 (Antigenic) | Non-allergen |
| AIE40737.1 | IMV membrane protein | 70 | 0.5019 (Antigenic) | Non-allergen |
| AIE40733.1 | IMV membrane protein | 100 | 0.3923 (Non-antigenic) | Non-allergen |
| AIE40718.1 | IMV membrane protein | 304 | 0.5316 (Antigenic) | Non-allergen |
| AIE40702.1 | late 16 kDa putative membrane protein | 133 | 0.7559 (Antigenic) | Non-allergen |
| AIE40669.1 | membrane protein | 273 | 0.4199 (Antigenic) | Non-allergen |
| AIE40657.1 | palmytilated EEV membrane protein | 372 | 0.4754 (Antigenic) | Allergen |
| Epitope | Peptide or Protein | Vaxijen | Antigenicity | Allergenicity | Toxicity | Interferon Activation |
|---|---|---|---|---|---|---|
| CD4+ T cell | RIYFVSLSL/AIE40786.1 | 1.6615 | Yes | No | No | Yes |
| CD4+ T cell | FSIGGVIHL/AIE40778.1 | 1.2283 | Yes | No | No | Yes |
| CD8+ T cell | IYFVSLSLL/AIE40786.1 | 1.4551 | Yes | No | No | Yes |
| CD8+ T cell | LKHKYGCSL/AIE40766.1 | 1.2793 | Yes | No | No | Yes |
| CD8+ T cell | AYTSISVVF/AIE40763.1 | 1.1050 | Yes | No | No | Yes |
| CD8+ T cell | RYPIIDIKW/AIE40702.1 | 2.5629 | Yes | No | No | Yes |
| B cell | PFSAKCPPIE/AIE40786.1 | 1.1314 | Yes | No | No | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akhtar, N.; Kaushik, V.; Grewal, R.K.; Wani, A.K.; Suwattanasophon, C.; Choowongkomon, K.; Oliva, R.; Shaikh, A.R.; Cavallo, L.; Chawla, M. Immunoinformatics-Aided Design of a Peptide Based Multiepitope Vaccine Targeting Glycoproteins and Membrane Proteins against Monkeypox Virus. Viruses 2022, 14, 2374. https://doi.org/10.3390/v14112374
Akhtar N, Kaushik V, Grewal RK, Wani AK, Suwattanasophon C, Choowongkomon K, Oliva R, Shaikh AR, Cavallo L, Chawla M. Immunoinformatics-Aided Design of a Peptide Based Multiepitope Vaccine Targeting Glycoproteins and Membrane Proteins against Monkeypox Virus. Viruses. 2022; 14(11):2374. https://doi.org/10.3390/v14112374
Chicago/Turabian StyleAkhtar, Nahid, Vikas Kaushik, Ravneet Kaur Grewal, Atif Khurshid Wani, Chonticha Suwattanasophon, Kiattawee Choowongkomon, Romina Oliva, Abdul Rajjak Shaikh, Luigi Cavallo, and Mohit Chawla. 2022. "Immunoinformatics-Aided Design of a Peptide Based Multiepitope Vaccine Targeting Glycoproteins and Membrane Proteins against Monkeypox Virus" Viruses 14, no. 11: 2374. https://doi.org/10.3390/v14112374
APA StyleAkhtar, N., Kaushik, V., Grewal, R. K., Wani, A. K., Suwattanasophon, C., Choowongkomon, K., Oliva, R., Shaikh, A. R., Cavallo, L., & Chawla, M. (2022). Immunoinformatics-Aided Design of a Peptide Based Multiepitope Vaccine Targeting Glycoproteins and Membrane Proteins against Monkeypox Virus. Viruses, 14(11), 2374. https://doi.org/10.3390/v14112374







