The Petasites hybridus CO2 Extract (Ze 339) Blocks SARS-CoV-2 Replication In Vitro
Abstract
:1. Introduction
2. Materials and Methods
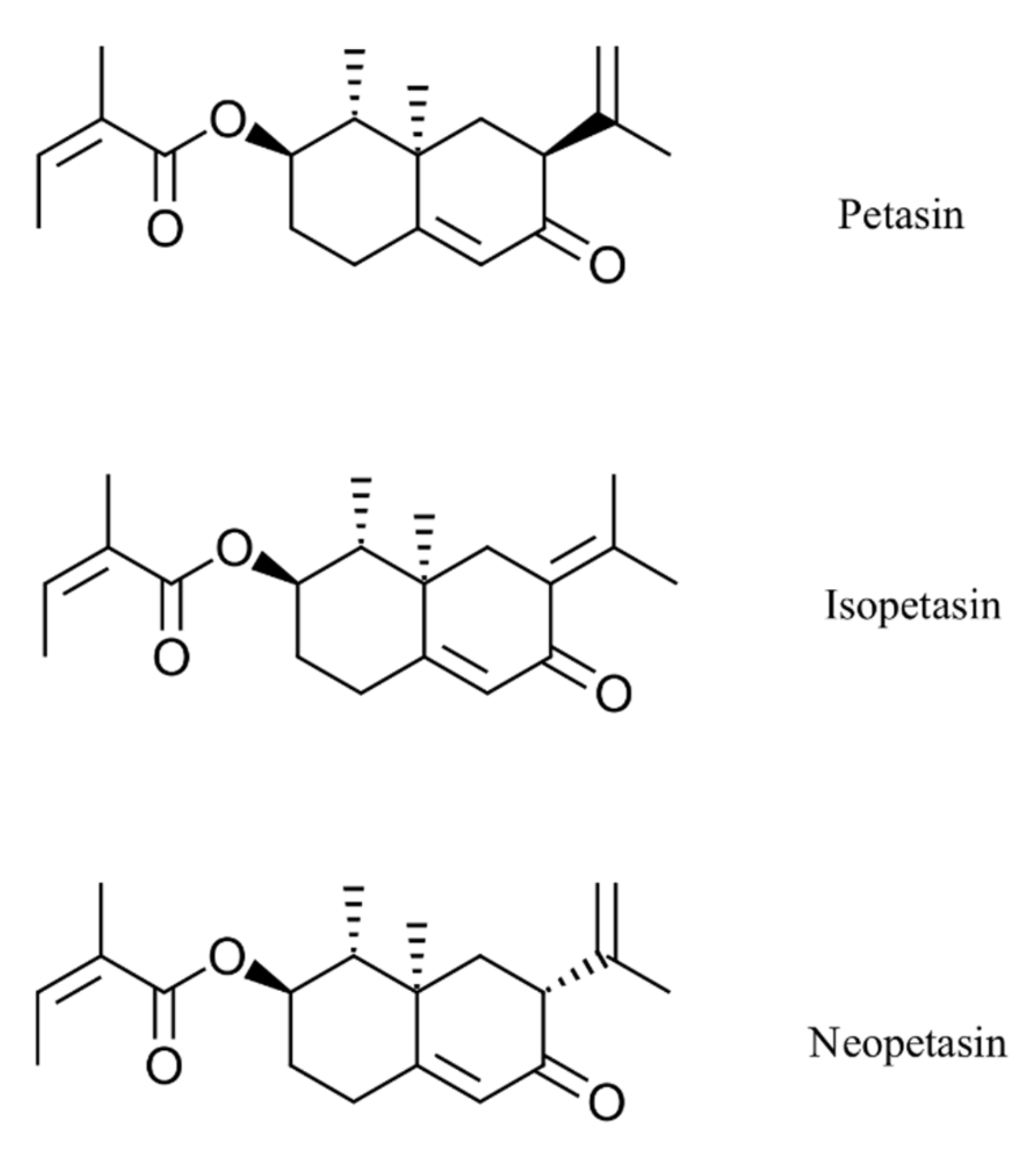
2.1. Compounds and Extract
2.2. Cells
2.3. Viral Reconstitution
2.4. Plaque Assay Protocol
2.5. Cytotoxicity Testing
2.6. Statistics
3. Results
3.1. Inhibition Assay of Remdesivir, Ze 339, Petasin, Isopetasin and Neopetasin against the Original SARS-CoV-2 Wuhan Variant
3.2. Inhibition Assay of Remdesivir, Ze 339 and Isopetasin against the SARS-CoV-2 Delta Variant
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Coronavirus Resource Center. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). Available online: https://coronavirus.jhu.edu/map.html (accessed on 1 December 2021).
- Tizaoui, K.; Zidi, I.; Lee, K.H.; Ghayda, R.A.; Hong, S.H.; Li, H.; Smith, L.; Koyanagi, A.; Jacob, L.; Kronbichler, A.; et al. Update of the current knowledge on genetics, evolution, immunopathogenesis, and transmission for coronavirus disease 19 (COVID-19). Int. J. Biol. Sci. 2020, 16, 2906–2923. [Google Scholar] [CrossRef] [PubMed]
- England, J.T.; Abdulla, A.; Biggs, C.M.; Lee, A.Y.Y.; Hay, K.A.; Hoiland, R.L.; Wellington, C.L.; Sekhon, M.; Jamal, S.; Shojania, K.; et al. Weathering the COVID-19 storm: Lessons from hematologic cytokine syndromes. Blood Rev. 2021, 45, 100707. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis 2020, 71, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Clark, I.A. The advent of the cytokine storm. Immunol. Cell Biol. 2007, 85, 271–273. [Google Scholar] [CrossRef]
- Jose, R.J.; Manuel, A. COVID-19 cytokine storm: The interplay between inflammation and coagulation. Lancet Respir. Med. 2020, 8, e46–e47. [Google Scholar] [CrossRef]
- Carsana, L.; Sonzogni, A.; Nasr, A.; Rossi, R.S.; Pellegrinelli, A.; Zerbi, P.; Rech, R.; Colombo, R.; Antinori, S.; Corbellino, M.; et al. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study. Lancet Infect. Dis. 2020, 20, 1135–1140. [Google Scholar] [CrossRef]
- Grasselli, G.; Tonetti, T.; Protti, A.; Langer, T.; Girardis, M.; Bellani, G.; Laffey, J.; Carrafiello, G.; Carsana, L.; Rizzuto, C.; et al. Pathophysiology of COVID-19-associated acute respiratory distress syndrome: A multicentre prospective observational study. Lancet Respir. Med. 2020, 8, 1201–1208. [Google Scholar] [CrossRef]
- Sonzogni, A.; Previtali, G.; Seghezzi, M.; Grazia Alessio, M.; Gianatti, A.; Licini, L.; Morotti, D.; Zerbi, P.; Carsana, L.; Rossi, R.; et al. Liver histopathology in severe COVID 19 respiratory failure is suggestive of vascular alterations. Liver Int. 2020, 40, 2110–2116. [Google Scholar] [CrossRef]
- Maiuolo, J.; Mollace, R.; Gliozzi, M.; Musolino, V.; Carresi, C.; Paone, S.; Scicchitano, M.; Macri, R.; Nucera, S.; Bosco, F.; et al. The Contribution of Endothelial Dysfunction in Systemic Injury Subsequent to SARS-Cov-2 Infection. Int. J. Mol. Sci. 2020, 21, 9309. [Google Scholar] [CrossRef]
- Combes, A.J.; Courau, T.; Kuhn, N.F.; Hu, K.H.; Ray, A.; Chen, W.S.; Chew, N.W.; Cleary, S.J.; Kushnoor, D.; Reeder, G.C.; et al. Global absence and targeting of protective immune states in severe COVID-19. Nature 2021, 591, 124–130. [Google Scholar] [CrossRef]
- Rodriguez, C.; Luque, N.; Blanco, I.; Sebastian, L.; Barbera, J.A.; Peinado, V.I.; Tura-Ceide, O. Pulmonary Endothelial Dysfunction and Thrombotic Complications in Patients with COVID-19. Am. J. Respir. Cell Mol. Biol. 2021, 64, 407–415. [Google Scholar] [CrossRef]
- Yang, L.; Xie, X.; Tu, Z.; Fu, J.; Xu, D.; Zhou, Y. The signal pathways and treatment of cytokine storm in COVID-19. Signal. Transduct Target. Ther. 2021, 6, 255. [Google Scholar] [CrossRef]
- Couzin-Frankel, J. Antiviral pills could change pandemic’s course. Science 2021, 374, 799–800. [Google Scholar] [CrossRef]
- Xiong, M.; Su, H.; Zhao, W.; Xie, H.; Shao, Q.; Xu, Y. What coronavirus 3C-like protease tells us: From structure, substrate selectivity, to inhibitor design. Med. Res. Rev. 2021, 41, 1965–1998. [Google Scholar] [CrossRef]
- Pfizer Pfizer Press Release: Pfizer Initiates Phase 1 Study of Novel Oral Antiviral Therapeutic Agent Against SARS-COV-2. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-initiates-phase-1-study-novel-oral-antiviral (accessed on 30 November 2021).
- Group, R.C.; Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; et al. Dexamethasone in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef]
- Mahase, E. Covid-19: Demand for dexamethasone surges as RECOVERY trial publishes preprint. BMJ 2020, 369, m2512. [Google Scholar] [CrossRef] [PubMed]
- Olender, S.A.; Perez, K.K.; Go, A.S.; Balani, B.; Price-Haywood, E.G.; Shah, N.S.; Wang, S.; Walunas, T.L.; Swaminathan, S.; Slim, J.; et al. Remdesivir for Severe COVID-19 versus a Cohort Receiving Standard of Care. Clin. Infect. Dis. 2020, 73, e4166–e4174. [Google Scholar] [CrossRef]
- Olender, S.A.; Walunas, T.L.; Martinez, E.; Perez, K.K.; Castagna, A.; Wang, S.; Kurbegov, D.; Goyal, P.; Ripamonti, D.; Balani, B.; et al. Remdesivir Versus Standard-of-Care for Severe Coronavirus Disease 2019 Infection: An Analysis of 28-Day Mortality. Open Forum Infect. Dis. 2021, 8, ofab278. [Google Scholar] [CrossRef]
- Spinner, C.D.; Gottlieb, R.L.; Criner, G.J.; Arribas Lopez, J.R.; Cattelan, A.M.; Soriano Viladomiu, A.; Ogbuagu, O.; Malhotra, P.; Mullane, K.M.; Castagna, A.; et al. Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19: A Randomized Clinical Trial. JAMA 2020, 324, 1048–1057. [Google Scholar] [CrossRef]
- Wang, L.Y.; Cui, J.J.; Ouyang, Q.Y.; Zhan, Y.; Guo, C.X.; Yin, J.Y. Remdesivir and COVID-19. Lancet 2020, 396, 953–954. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, D.; Du, G.; Du, R.; Zhao, J.; Jin, Y.; Fu, S.; Gao, L.; Cheng, Z.; Lu, Q.; et al. Remdesivir in adults with severe COVID-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2020, 395, 1569–1578. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, F.; Zhang, D.; Zhao, J.; Du, R.; Hu, Y.; Cheng, Z.; Gao, L.; Jin, Y.; Luo, G.; et al. Evaluation of the efficacy and safety of intravenous remdesivir in adult patients with severe COVID-19: Study protocol for a phase 3 randomized, double-blind, placebo-controlled, multicentre trial. Trials 2020, 21, 422. [Google Scholar] [CrossRef] [PubMed]
- Almerie, M.Q.; Kerrigan, D.D. The association between obesity and poor outcome after COVID-19 indicates a potential therapeutic role for montelukast. Med. Hypotheses 2020, 143, 109883. [Google Scholar] [CrossRef]
- Bozek, A.; Winterstein, J. Montelukast’s ability to fight COVID-19 infection. J. Asthma 2021, 58, 1348–1349. [Google Scholar] [CrossRef]
- Funk, C.D.; Ardakani, A. A Novel Strategy to Mitigate the Hyperinflammatory Response to COVID-19 by Targeting Leukotrienes. Front. Pharmacol. 2020, 11, 1214. [Google Scholar] [CrossRef]
- Fidan, C.; Aydogdu, A. As a potential treatment of COVID-19: Montelukast. Med. Hypotheses 2020, 142, 109828. [Google Scholar] [CrossRef]
- Citron, F.; Perelli, L.; Deem, A.K.; Genovese, G.; Viale, A. Leukotrienes, a potential target for Covid-19. Prostaglandins Leukot. Essent. Fat. Acids 2020, 161, 102174. [Google Scholar] [CrossRef]
- Archambault, A.S.; Zaid, Y.; Rakotoarivelo, V.; Turcotte, C.; Dore, E.; Dubuc, I.; Martin, C.; Flamand, O.; Amar, Y.; Cheikh, A.; et al. High levels of eicosanoids and docosanoids in the lungs of intubated COVID-19 patients. FASEB J. 2021, 35, e21666. [Google Scholar] [CrossRef]
- Zaid, Y.; Dore, E.; Dubuc, I.; Archambault, A.S.; Flamand, O.; Laviolette, M.; Flamand, N.; Boilard, E.; Flamand, L. Chemokines and eicosanoids fuel the hyperinflammation within the lungs of patients with severe COVID-19. J. Allergy Clin. Immunol. 2021, 148, 368–380.e3. [Google Scholar] [CrossRef]
- Funk, C.D. Prostaglandins and leukotrienes: Advances in eicosanoid biology. Science 2001, 294, 1871–1875. [Google Scholar] [CrossRef] [Green Version]
- Kanaoka, Y.; Boyce, J.A. Cysteinyl leukotrienes and their receptors: Cellular distribution and function in immune and inflammatory responses. J. Immunol. 2004, 173, 1503–1510. [Google Scholar] [CrossRef] [Green Version]
- Werz, O.; Steinhilber, D. Therapeutic options for 5-lipoxygenase inhibitors. Pharmacol. Ther. 2006, 112, 701–718. [Google Scholar] [CrossRef]
- Sharma, J.N.; Mohammed, L.A. The role of leukotrienes in the pathophysiology of inflammatory disorders: Is there a case for revisiting leukotrienes as therapeutic targets? Inflammopharmacology 2006, 14, 10–16. [Google Scholar] [CrossRef]
- Singh, R.K.; Tandon, R.; Dastidar, S.G.; Ray, A. A review on leukotrienes and their receptors with reference to asthma. J. Asthma 2013, 50, 922–931. [Google Scholar] [CrossRef]
- Brattström, A. A newly developed extract (Ze 339) from butterbur (Petasites hybridus L.) is clinically efficient in allergic rhinitis (hay fever). Phytomedicine 2003, 10, 50–52. [Google Scholar] [CrossRef]
- Kodjadjiku, U.; Nagele, B.; Halbsguth, C.; Butterweck, V. Extract matrix composition does not affect in vitro leukotriene inhibitory effects of the Petasites hybridus extract Ze 339. Fitoterapia 2021, 153, 104986. [Google Scholar] [CrossRef]
- Thomet, O.A.; Schapowal, A.; Heinisch, I.V.; Wiesmann, U.N.; Simon, H.U. Anti-inflammatory activity of an extract of Petasites hybridus in allergic rhinitis. Int. Immunopharmacol. 2002, 2, 997–1006. [Google Scholar] [CrossRef]
- Thomet, O.A.; Wiesmann, U.N.; Blaser, K.; Simon, H.U. Differential inhibition of inflammatory effector functions by petasin, isopetasin and neopetasin in human eosinophils. Clin. Exp. Allergy 2001, 31, 1310–1320. [Google Scholar] [CrossRef]
- Thomet, O.A.R.; Wiesmann, U.N.; Schapowal, A.; Bizer, C.; Simon, H.-U. Role of petasin in the potential anti-inflammatory activity of a plant extract of petasites hybridus. Biochem. Pharmacol. 2001, 61, 1041–1047. [Google Scholar] [CrossRef]
- Steiert, S.A.; Zissler, U.M.; Chaker, A.M.; Esser-von-Bieren, J.; Dittlein, D.; Guerth, F.; Jakwerth, C.A.; Piontek, G.; Zahner, C.; Drewe, J.; et al. Anti-inflammatory effects of the petasin phyto drug Ze339 are mediated by inhibition of the STAT pathway. Biofactors 2017, 43, 388–399. [Google Scholar] [CrossRef]
- Koch, V.; Rittinghausen, R. Composition Containing Pyrrolizidine-Alkaloid-Free Petasites. US Patent US6551626B1, 22 April 2003. [Google Scholar]
- Steiner, R.; Hauk, A.; Tratz, W. Method for Producing Medicinal Plant Extracts. International Patent WO/1999/018984, 29 April 1999. [Google Scholar]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brunink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020, 25, 2000045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honko, A.N.; Storm, N.; Bean, D.J.; Henao Vasquez, J.; Downs, S.N.; Griffiths, A. Rapid Quantification and Neutralization Assays for Novel Coronavirus SARS-CoV-2 Using Avicel RC-591 Semi-Solid Overlay. Preprints 2020, 2020050264. [Google Scholar] [CrossRef]
- Rasband, W.S. Image J. Available online: https://imagej.nih.gov/ij/ (accessed on 2 October 2021).
- Baer, A.; Kehn-Hall, K. Viral concentration determination through plaque assays: Using traditional and novel overlay systems. J. Vis. Exp. 2014, e52065. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Y.; Jan, J.T.; Ma, S.H.; Kuo, C.J.; Juan, H.F.; Cheng, Y.S.; Hsu, H.H.; Huang, H.C.; Wu, D.; Brik, A.; et al. Small molecules targeting severe acute respiratory syndrome human coronavirus. Proc. Natl. Acad. Sci. USA 2004, 101, 10012–10017. [Google Scholar] [CrossRef] [Green Version]
- Ao, Z.; Chan, M.; Ouyang, M.J.; Olukitibi, T.A.; Mahmoudi, M.; Kobasa, D.; Yao, X. Identification and evaluation of the inhibitory effect of Prunella vulgaris extract on SARS-coronavirus 2 virus entry. PLoS ONE 2021, 16, e0251649. [Google Scholar] [CrossRef]
- Jeon, S.; Ko, M.; Lee, J.; Choi, I.; Byun, S.Y.; Park, S.; Shum, D.; Kim, S. Identification of Antiviral Drug Candidates against SARS-CoV-2 from FDA-Approved Drugs. Antimicrob. Agents Chemother. 2020, 64, e00819-20. [Google Scholar] [CrossRef]
- Ku, K.B.; Shin, H.J.; Kim, H.S.; Kim, B.T.; Kim, S.J.; Kim, C. Repurposing Screens of FDA-Approved Drugs Identify 29 Inhibitors of SARS-CoV-2. J. Microbiol. Biotechnol. 2020, 30, 1843–1853. [Google Scholar] [CrossRef]
- Matsuyama, S.; Nao, N.; Shirato, K.; Kawase, M.; Saito, S.; Takayama, I.; Nagata, N.; Sekizuka, T.; Katoh, H.; Kato, F.; et al. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. USA 2020, 117, 7001–7003. [Google Scholar] [CrossRef] [Green Version]
- Tietjen, I.; Cassel, J.; Register, E.T.; Zhou, X.Y.; Messick, T.E.; Keeney, F.; Lu, L.D.; Beattie, K.D.; Rali, T.; Tebas, P.; et al. The natural stilbenoid (-)-hopeaphenol inhibits cellular entry of SARS-CoV-2 USA-WA1/2020, B.1.1.7 and B.1.351 variants. Antimicrob. Agents Chemother. 2021, 65, AAC0077221. [Google Scholar] [CrossRef]
- Ellinger, B.; Bojkova, D.; Zaliani, A.; Cinatl, J.; Claussen, C.; Westhaus, S.; Keminer, O.; Reinshagen, J.; Kuzikov, M.; Wolf, M.; et al. A SARS-CoV-2 cytopathicity dataset generated by high-content screening of a large drug repurposing collection. Sci. Data 2021, 8, 70. [Google Scholar] [CrossRef]
- Indrayanto, G.; Putra, G.S.; Suhud, F. Validation of in-vitro bioassay methods: Application in herbal drug research. Profiles Drug Subst. Excip. Relat. Methodol. 2021, 46, 273–307. [Google Scholar] [CrossRef]
- GHDDI Info Sharing Portal. COVID-19 Preclinical Studies. Available online: https://ghddi-ailab.github.io/Targeting2019-nCoV/preclinical/ (accessed on 20 November 2021).
- Dittmar, M.; Lee, J.S.; Whig, K.; Segrist, E.; Li, M.; Kamalia, B.; Castellana, L.; Ayyanathan, K.; Cardenas-Diaz, F.L.; Morrisey, E.E.; et al. Drug repurposing screens reveal cell-type-specific entry pathways and FDA-approved drugs active against SARS-Cov-2. Cell Rep. 2021, 35, 108959. [Google Scholar] [CrossRef]
- Blosa, M.; Uricher, J.; Nebel, S.; Zahner, C.; Butterweck, V.; Drewe, J. Treatment of Early Allergic and Late Inflammatory Symptoms of Allergic Rhinitis with Petasites hybridus Leaf Extract (Ze 339): Results of a Noninterventional Observational Study in Switzerland. Pharmaceuticals 2021, 14, 180. [Google Scholar] [CrossRef]
- Dumitru, A.F.; Shamji, M.; Wagenmann, M.; Hindersin, S.; Scheckenbach, K.; Greve, J.; Klenzner, T.; Hess, L.; Nebel, S.; Zimmermann, C.; et al. Petasol butenoate complex (Ze 339) relieves allergic rhinitis-induced nasal obstruction more effectively than desloratadine. J. Allergy Clin. Immunol. 2011, 127, 1515–1521. [Google Scholar] [CrossRef]
- Schapowal, A.; Petasites Study, G. Randomised controlled trial of butterbur and cetirizine for treating seasonal allergic rhinitis. BMJ 2002, 324, 144–146. [Google Scholar] [CrossRef] [Green Version]
- Schapowal, A.; Petasites Study, G. Butterbur Ze339 for the treatment of intermittent allergic rhinitis: Dose-dependent efficacy in a prospective, randomized, double-blind, placebo-controlled study. Arch. Otolaryngol. Head Neck Surg. 2004, 130, 1381–1386. [Google Scholar] [CrossRef] [Green Version]
- Schapowal, A.; Study Group. Treating intermittent allergic rhinitis: A prospective, randomized, placebo and antihistamine-controlled study of Butterbur extract Ze 339. Phytother. Res. 2005, 19, 530–537. [Google Scholar] [CrossRef]
- Product Information. Available online: https://www.swissmedicinfo.ch/?Lang=EN (accessed on 1 December 2021).
- Thomet, O.A.; Simon, H.U. Petasins in the treatment of allergic diseases: Results of preclinical and clinical studies. Int. Arch. Allergy Immunol. 2002, 129, 108–112. [Google Scholar] [CrossRef]
- Adachi, Y.; Kanbayashi, Y.; Harata, I.; Ubagai, R.; Takimoto, T.; Suzuki, K.; Miwa, T.; Noguchi, Y. Petasin activates AMP-activated protein kinase and modulates glucose metabolism. J. Nat. Prod. 2014, 77, 1262–1269. [Google Scholar] [CrossRef]
- Sharma, S.; Ray, A.; Sadasivam, B. Metformin in COVID-19: A possible role beyond diabetes. Diabetes Res. Clin. Pract. 2020, 164, 108183. [Google Scholar] [CrossRef]
- Karam, B.S.; Morris, R.S.; Bramante, C.T.; Puskarich, M.; Zolfaghari, E.J.; Lotfi-Emran, S.; Ingraham, N.E.; Charles, A.; Odde, D.J.; Tignanelli, C.J. mTOR inhibition in COVID-19: A commentary and review of efficacy in RNA viruses. J. Med. Virol. 2021, 93, 1843–1846. [Google Scholar] [CrossRef]
- Kow, C.S.; Hasan, S.S. Mortality risk with preadmission metformin use in patients with COVID-19 and diabetes: A meta-analysis. J. Med. Virol. 2021, 93, 695–697. [Google Scholar] [CrossRef]
- Lally, M.A.; Tsoukas, P.; Halladay, C.W.; O’Neill, E.; Gravenstein, S.; Rudolph, J.L. Metformin is Associated with Decreased 30-Day Mortality Among Nursing Home Residents Infected with SARS-CoV2. J. Am. Med. Dir. Assoc. 2021, 22, 193–198. [Google Scholar] [CrossRef]
- Nafakhi, H.; Alareedh, M.; Al-Buthabhak, K.; Shaghee, F.; Nafakhi, A.; Kasim, S. Predictors of adverse in-hospital outcome and recovery in patients with diabetes mellitus and COVID-19 pneumonia in Iraq. Diabetes Metab Syndr. 2021, 15, 33–38. [Google Scholar] [CrossRef]
- Chen, X.; Guo, H.; Qiu, L.; Zhang, C.; Deng, Q.; Leng, Q. Immunomodulatory and Antiviral Activity of Metformin and Its Potential Implications in Treating Coronavirus Disease 2019 and Lung Injury. Front. Immunol. 2020, 11, 2056. [Google Scholar] [CrossRef]
- Durdagi, S.; Avsar, T.; Orhan, M.D.; Serhatli, M.; Balcioglu, B.K.; Ozturk, H.U.; Kayabolen, A.; Cetin, Y.; Aydinlik, S.; Bagci-Onder, T.; et al. The neutralization effect of montelukaston SARS-CoV-2 is shown by multiscale in silicosimulations and combined in vitro studies. Mol. Ther. 2021, 30, 1–12. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, B.; Kumari, P.; Kumar, P.V.; Agnihotri, G.; Khan, S.; Kant Beuria, T.; Syed, G.H.; Dixit, A. Identification of multipotent drugs for COVID-19 therapeutics with the evaluation of their SARS-CoV2 inhibitory activity. Comput. Struct. Biotechnol. J. 2021, 19, 1998–2017. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Ghose, A.K.; Viswanadhan, V.N.; Wendoloski, J.J. A knowledge-based approach in designing combinatorial or medicinal chemistry libraries for drug discovery. 1. A qualitative and quantitative characterization of known drug databases. J. Comb. Chem. 1999, 1, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Veber, D.F.; Johnson, S.R.; Cheng, H.Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- KC, G.B.; Bocci, G.; Verma, S.; Hassan, M.; Holmes, J.; Yang, J.J.; Sirimulla, S.; Oprea, T.I. A machine learning platform to estimate anti-SARS-CoV-2 activities. Nat. Mach. Intell. 2021, 3, 527–535. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urda, L.; Kreuter, M.H.; Drewe, J.; Boonen, G.; Butterweck, V.; Klimkait, T. The Petasites hybridus CO2 Extract (Ze 339) Blocks SARS-CoV-2 Replication In Vitro. Viruses 2022, 14, 106. https://doi.org/10.3390/v14010106
Urda L, Kreuter MH, Drewe J, Boonen G, Butterweck V, Klimkait T. The Petasites hybridus CO2 Extract (Ze 339) Blocks SARS-CoV-2 Replication In Vitro. Viruses. 2022; 14(1):106. https://doi.org/10.3390/v14010106
Chicago/Turabian StyleUrda, Lorena, Matthias Heinrich Kreuter, Jürgen Drewe, Georg Boonen, Veronika Butterweck, and Thomas Klimkait. 2022. "The Petasites hybridus CO2 Extract (Ze 339) Blocks SARS-CoV-2 Replication In Vitro" Viruses 14, no. 1: 106. https://doi.org/10.3390/v14010106
APA StyleUrda, L., Kreuter, M. H., Drewe, J., Boonen, G., Butterweck, V., & Klimkait, T. (2022). The Petasites hybridus CO2 Extract (Ze 339) Blocks SARS-CoV-2 Replication In Vitro. Viruses, 14(1), 106. https://doi.org/10.3390/v14010106






