Unique Severe COVID-19 Placental Signature Independent of Severity of Clinical Maternal Symptoms
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Virology
2.3. Placenta Histopathology
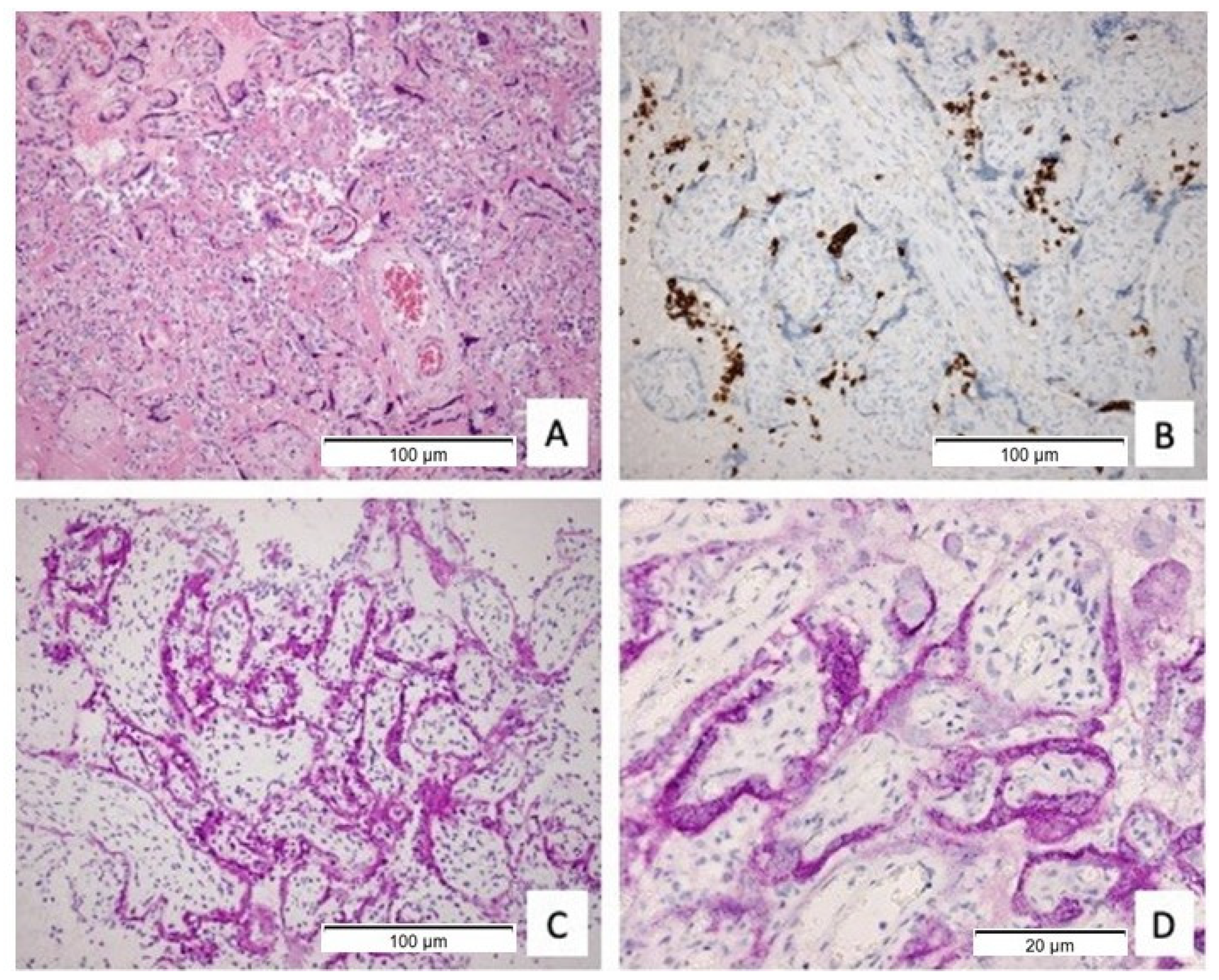
2.4. Immunohistochemical Stainings for Immune Subsets (CD3, CD20, CD68)
2.5. Immunohistochemistry of SARS-CoV-2 NP and Spike
2.6. Statistics
3. Results
3.1. Maternal Characteristics, Obstetric and Neonatal Outcomes
3.2. Placental Histopathological Examination
3.3. Clinical Presentation of COVID-19 in Cases of Confirmed Placental SARS-CoV-2
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Villar, J.; Ariff, S.; Gunier, R.B.; Thiruvengadam, R.; Rauch, S.; Kholin, A.; Roggero, P.; Prefumo, F.; Do Vale, M.S.; Cardona-Perez, J.A.; et al. Maternal and Neonatal Morbidity and Mortality Among Pregnant Women With and Without COVID-19 Infection: The INTERCOVID Multinational Cohort Study. JAMA Pediatr. 2021, 175, 817–826. [Google Scholar] [CrossRef]
- Zambrano, L.D.; Ellington, S.; Strid, P.; Galang, R.R.; Oduyebo, T.; Tong, V.T.; Woodworth, K.R.; Nahabedian, J.F., III; Azziz-Baumgartner, E.; Gilboa, S.M.; et al. Update: Characteristics of Symptomatic Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status—United States, 22 January–3 October 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 1641. [Google Scholar] [CrossRef]
- Allotey, J.; Stallings, E.; Bonet, M.; Yap, M.; Chatterjee, S.; Kew, T.; Debenham, L.; Llavall, A.C.; Dixit, A.; Zhou, D.; et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: Living systematic review and meta-analysis. BMJ 2020, 370, m3320. [Google Scholar] [CrossRef]
- Adhikari, E.H.; Moreno, W.; Zofkie, A.C.; MacDonald, L.; McIntire, D.D.; Collins, R.R.; Spong, C.Y. Pregnancy Outcomes Among Women with and Without Severe Acute Respiratory Syndrome Corona-virus 2 Infection. JAMA Netw. Open 2020, 3, e2029256. [Google Scholar] [CrossRef]
- Chmielewska, B.; Barratt, I.; Townsend, R.; Kalafat, E.; van der Meulen, J.; Gurol-Urganci, I.; O’Brien, P.; Morris, E.; Draycott, T.; Thangaratinam, S.; et al. Effects of the COVID-19 pandemic on maternal and perinatal outcomes: A systematic review and meta-analysis. Lancet Glob. Health 2021, 9, e759–e772. [Google Scholar] [CrossRef]
- Kotlyar, A.M.; Grechukhina, O.; Chen, A.; Popkhadze, S.; Grimshaw, A.; Tal, O.; Taylor, H.S.; Tal, R. Vertical transmission of coronavirus disease 2019: A systematic review and meta-analysis. Am. J. Obstet. Gynecol. 2021, 224, 35–53.e3. [Google Scholar] [CrossRef]
- Sharps, M.C.; Hayes, D.J.; Lee, S.; Zou, Z.; Brady, C.A.; Almoghrabi, Y.; Kerby, A.; Tamber, K.K.; Jones, C.J.; Waldorf, K.M.A.; et al. A structured review of placental morphology and histopathological lesions associated with SARS-CoV-2 infection. Placenta 2020, 101, 13–29. [Google Scholar] [CrossRef]
- Mor, G.; Aldo, P.; Alvero, A.B. The unique immunological and microbial aspects of pregnancy. Nat. Rev. Immunol. 2017, 17, 469–482. [Google Scholar] [CrossRef]
- Sykes, L.; MacIntyre, D.A.; Yap, X.J.; Teoh, T.G.; Bennett, P.R. The Th1:Th2 Dichotomy of Pregnancy and Preterm Labour. Mediat. Inflamm. 2012, 2012, 967629. [Google Scholar] [CrossRef]
- LoMauro, A.; Aliverti, A. Respiratory physiology of pregnancy: Physiology masterclass. Breathe 2015, 11, 297–301. [Google Scholar] [CrossRef]
- Alzamora, M.C.; Paredes, T.; Caceres, D.; Webb, C.; Valdez, L.M.; La Rosa, M. Severe COVID-19 during Pregnancy and Possible Vertical Transmission. Am. J. Perinatol. 2020, 37, 861–865. [Google Scholar] [CrossRef]
- Hoo, R.; Nakimuli, A.; Vento-Tormo, R. Innate Immune Mechanisms to Protect Against Infection at the Human Decidual-Placental Interface. Front. Immunol. 2020, 11, 2070. [Google Scholar] [CrossRef]
- Silasi, M.; Cardenas, I.; Kwon, J.-Y.; Racicot, K.; Aldo, P.; Mor, G. Viral Infections During Pregnancy. Am. J. Reprod. Immunol. 2015, 73, 199–213. [Google Scholar] [CrossRef]
- Pereira, L. Congenital Viral Infection: Traversing the Uterine-Placental Interface. Annu. Rev. Virol. 2018, 5, 273–299. [Google Scholar] [CrossRef]
- Van der Eijk, A.A.; van Genderen, P.J.; Verdijk, R.M.; Reusken, C.B.; Mögling, R.; van Kampen, J.J.; Widagdo, W.; Aron, G.I.; GeurtsvanKessel, C.H.; Pas, S.D.; et al. Miscarriage Associated with Zika Virus Infection. N. Engl. J. Med. 2016, 375, 1002–1004. [Google Scholar] [CrossRef]
- Heerema-McKenney, A. Defense and infection of the human placenta. APMIS 2018, 126, 570–588. [Google Scholar] [CrossRef]
- Neerukonda, S.N.; Katneni, U. A review on SARS-CoV-2 virology, pathophysiology, animal models, and anti-viral. Pathogens 2020, 9, 426. [Google Scholar] [CrossRef]
- Park, S.E. Epidemiology, virology, and clinical features of severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2; Coronavirus Disease-19). Clin. Exp. Pediatrics 2020, 63, 119. [Google Scholar] [CrossRef]
- Fajnzylber, J.; Regan, J.; Coxen, K.; Corry, H.; Wong, C.; Rosenthal, A.; Worrall, D.; Giguel, F.; Piechocka-Trocha, A.; Atyeo, C.; et al. SARS-CoV-2 viral load is associated with increased disease severity and mortality. Nat. Commun. 2020, 11, 5493. [Google Scholar] [CrossRef]
- Edlow, A.G.; Li, J.Z.; Collier, A.-R.Y.; Atyeo, C.; James, K.E.; Boatin, A.A.; Gray, K.J.; Bordt, E.A.; Shook, L.L.; Yonker, L.M.; et al. Assessment of Maternal and Neonatal SARS-CoV-2 Viral Load, Transplacental Antibody Transfer, and Placental Pathology in Pregnancies During the COVID-19 Pandemic. JAMA Netw. Open 2020, 3, e2030455. [Google Scholar] [CrossRef]
- De Fátima Yukie Maeda, M.; de Lourdes Brizot, M.; Gibelli, M.A.B.; Ibidi, S.M.; Brunow de Carvalho, W.; Hoshida, M.S.; Machado, C.M.; Sabino, E.C.; Campos de Oliveira da Silva, L.; Jaenisch, T.; et al. Vertical transmission of SARS-CoV2 during pregnancy: A high-risk cohort. Prenat. Diagn. 2021, 41, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Ashary, N.; Bhide, A.; Chakraborty, P.; Colaco, S.; Mishra, A.; Chhabria, K.; Jolly, M.K.; Modi, D. Single-Cell RNA-seq Identifies Cell Subsets in Human Placenta That Highly Expresses Factors Driving Path-ogenesis of SARS-CoV-2. Front. Cell Dev. Biol. 2020, 8, 783. [Google Scholar] [CrossRef]
- Patberg, E.T.; Adams, T.; Rekawek, P.; Vahanian, S.A.; Akerman, M.; Hernandez, A.; Rapkiewicz, A.V.; Ragolia, L.; Sicuranza, G.; Chavez, M.R.; et al. Coronavirus disease 2019 infection and placental histopathology in women delivering at term. Am. J. Obstet. Gynecol. 2021, 224, 382.e1–382.e18. [Google Scholar] [CrossRef]
- Schoenmakers, S.; Snijder, P.; Verdijk, R.M.; Kuiken, T.; Kamphuis, S.S.M.; Koopman, L.P.; Krasemann, T.B.; Rousian, M.; Broekhuizen, M.; Steegers, E.A.P.; et al. Severe Acute Respiratory Syndrome Coronavirus 2 Placental Infection and Inflammation Leading to Fetal Distress and Neonatal Multi-Organ Failure in an Asymptomatic Woman. J. Pediatr. Infect. Dis. Soc. 2020, 10, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Vivanti, A.J.; Vauloup-Fellous, C.; Prevot, S.; Zupan, V.; Suffee, C.; Do Cao, J.; Benachi, A.; De Luca, D. Transplacental transmission of SARS-CoV-2 infection. Nat. Commun. 2020, 11, 3572. [Google Scholar] [CrossRef] [PubMed]
- Shanes, E.D.; Mithal, L.B.; Otero, S.; Azad, H.A.; Miller, E.S.; Goldstein, J.A. Placental Pathology in COVID-19. Am. J. Clin. Pathol. 2020, 154, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.A.; Morotti, D. Placental Pathology of COVID-19 with and without Fetal and Neonatal Infection: Trophoblast Necrosis and Chronic Histiocytic Intervillositis as Risk Factors for Transplacental Transmission of SARS-CoV-2. Viruses 2020, 12, 1308. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.A.; Baldewijns, M.; Benachi, A.; Bugatti, M.; Collins, R.R.; De Luca, D.; Facchetti, F.; Linn, R.L.; Marcelis, L.; Morotti, D.; et al. Chronic Histiocytic Intervillositis with Trophoblast Necrosis are Risk Factors Associated with Placental Infection from Coronavirus Disease 2019 (COVID-19) and Intrauterine Maternal-Fetal Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Transmission in Liveborn and Stillborn Infants. Arch. Pathol. Lab. Med. 2020, 145, 517–528. [Google Scholar]
- Linehan, L.; O’Donoghue, K.; Dineen, S.; White, J.; Higgins, J.R.; Fitzgerald, B. SARS-CoV-2 placentitis: An uncommon complication of maternal COVID-19. Placenta 2021, 104, 261–266. [Google Scholar] [CrossRef]
- Nikkels, P.G.; Evers, A.C.; Schuit, E.; Brouwers, H.A.; Bruinse, H.W.; Bont, L.; Houben, M.L.; Kwee, A. Placenta Pathology from Term Born Neonates with Normal or Adverse Outcome. Pediatr. Dev. Pathol. 2021, 24, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.C.; Murthy, S.; Diaz, J.; Adhikari, N.K.; Angus, D.C.; Arabi, Y.M.; Baillie, K.; Bauer, M.; Berry, S.; Blackwood, B. A minimal common outcome measure set for COVID-19 clinical re-search. Lancet Infect. Dis. 2020, 20, e192–e197. [Google Scholar] [CrossRef]
- Sauvestre, F.; Mattuizzi, A.; Sentilhes, L.; Poingt, M.; Blanco, P.; Houssin, C.; Carles, D.; Pelluard, F.; Andre, G.; Lazaro, E. Chronic Intervillositis of Unknown Etiology: Development of a Grading and Scoring System That Is Strongly Associated with Poor Perinatal Outcomes. Am. J. Surg. Pathol. 2020, 44, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Evers, I.M. Placental Pathology in Women with Type 1 Diabetes and in a Control Group with Normal and Large-for-Gestational-Age Infants. Placenta 2003, 24, 819–825. [Google Scholar] [CrossRef]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef]
- GeurtsvanKessel, C.H.; Okba, N.M.A. An Evaluation of COVID-19 Serological Assays Informs Future Diagnostics and Exposure Assessment. Nat. Commun. 2020, 11, 3436. [Google Scholar] [CrossRef]
- Khong, T.Y.; Mooney, E.E.; Ariel, I.; Balmus, N.C.; Boyd, T.K.; Brundler, M.A.; Derricott, H.; Evans, M.J.; Faye-Petersen, O.M.; Gillan, J.E.; et al. Sampling and Definitions of Placental Lesions: Amsterdam Placental Workshop Group Consensus Statement. Arch. Pathol. Lab. Med. 2016, 140, 698–713. [Google Scholar] [CrossRef]
- Redline, R.W.; Ariel, I.; Baergen, R.N.; Desa, D.J.; Kraus, F.T.; Roberts, D.J.; Sander, C.M. Fetal Vascular Obstructive Lesions: Nosology and Reproducibility of Placental Reaction Patterns. Pediatr. Dev. Pathol. 2004, 7, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Bos, M.; Nikkels, P.; Cohen, D.; Schoones, J.; Bloemenkamp, K.; Bruijn, J.; Baelde, H.; van der Hoorn, M.; Turner, R. Towards standardized criteria for diagnosing chronic intervillositis of unknown etiology: A systematic review. Placenta 2018, 61, 80–88. [Google Scholar] [CrossRef]
- Redline, R.W.; Faye-Petersen, O.; Heller, D.; Qureshi, F.; Savell, V.; Vogler, C. Amniotic Infection Syndrome: Nosology and Reproducibility of Placental Reaction Patterns. Pediatr. Dev. Pathol. 2003, 6, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Bebell, L.M.; Siedner, M.J.; Ngonzi, J.; Le, M.H.; Adong, J.; Boatin, A.A.; Bassett, I.V.; Roberts, D.J. Brief Report: Chronic Placental Inflammation Among Women Living with HIV in Uganda. JAIDS J. Acquir. Immune Defic. Syndr. 2020, 85, 320–324. [Google Scholar] [CrossRef]
- Myerson, D.; Parkin, R.K.; Benirschke, K.; Tschetter, C.N.; Hyde, S.R. The Pathogenesis of Villitis of Unknown Etiology: Analysis with a New Conjoint Immunohistochemistry-in Situ Hybridization Procedure to Identify Specific Maternal and Fetal Cells. Pediatr. Dev. Pathol. 2006, 9, 257–265. [Google Scholar] [CrossRef]
- Tamiolakis, D.; Venizelos, I.; Lambropoulou, M.; Kotini, A.; Barbagadaki, S.; Nikolaidou, S.; Boglou, P.; Papadopoulos, N. Human Decidual Cells Activity in Women with Spontaneous Abortions of Probable CMV Aetiology During the First Trimester of Gestation. An Immunohistochemical Study with CMV-Associated Antigen. Acta Med. (Hradec Kralove) 2004, 47, 195–199. [Google Scholar] [CrossRef][Green Version]
- Kapur, P.; Rakheja, D.; Gomez, A.M.; Sheffield, J.; Sanchez, P.; Rogers, B.B. Characterization of Inflammation in Syphilitic Villitis and in Villitis of Unknown Etiology. Pediatr. Dev. Pathol. 2004, 7, 453–458. [Google Scholar] [CrossRef]
- Huynh, J.; Dawson, D.; Roberts, D.; Bentley-Lewis, R. A systematic review of placental pathology in maternal diabetes mellitus. Placenta 2015, 36, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Cribiù, F.M.; Erra, R.; Pugni, L.; Rubio-Perez, C.; Alonso, L.; Simonetti, S.; Croci, G.A.; Serna, G.; Ronchi, A.; Pietrasanta, C.; et al. Severe SARS-CoV-2 placenta infection can impact neonatal outcome in the absence of vertical transmission. J. Clin. Investig. 2021, 131, e145427. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 36) | Active COVID-19 (n = 17) | Resolved COVID-19 (n = 19) | p-Value | |
|---|---|---|---|---|
| Age, years, mean (±SD) | 33.5 (±5.1) | 34.4 (±4.5) | 32.8 (±5.7) | 0.38 |
| Primigravida, n = 35 (%) | 9 (25.0) | 6 (35.5) | 3 (15.8) | 0.21 |
| Pre-pregnancy BMI, kg/m2, n = 32, median (IQR) | 28.8 (10.4) | 31.6 (12.5) | 26.4 (9.5) | 0.12 |
| Geographic origin, n (%) | ||||
| Caucasian | 10 (27.8) | 4 (23.5) | 6 (31.6) | 0.59 |
| North African | 5 (13.9) | 2 (11.8) | 3 (15.8) | 0.73 |
| Turkish | 4 (11.1) | 2 (11.8) | 2 (10.5) | 0.91 |
| Other African | 3 (8.3) | 1 (5.9) | 2 (10.5) | 0.62 |
| Hispanic | 2 (5.6) | 1 (5.9) | 1 (5.3) | 0.94 |
| Asian | 2 (5.6 | 1 (5.9) | 1 (5.3) | 0.94 |
| Other | 6 (16.7) | 4 (23.5) | 2 (10.5) | 0.3 |
| Unknown | 4 (11.1) | 2 (11.8) | 2 (10.5) | 0.91 |
| Comorbidities, n = 34 (%) | ||||
| Autoimmune disease | 3 (8.3) | 1 (5.9) | 2 (10.5) | 0.88 |
| Pulmonary disease | 0 (0.0) | - | - | - |
| (Gestational) Diabetes | 6 (16.7) | 5 (29.4) | 1 (5.3) | 0.15 |
| Hypertensive disorders | 2 (5.6) | 0 (0.0) | 2 (10.5) | 0.39 |
| Periconceptional smoking, n = 25 (%) | 3 (8.3) | 2 (11.8) | 1 (5.3) | 0.68 |
| Singleton pregnancy, n (%) | 33 (91.7) | 16 (94.1) | 17 (89.5) | 0.62 |
| GA (in days) at diagnosis, median (IQR) | 237.5 (58) | 234 (50) | 241 (53) | 0.05 |
| Trimester at diagnosis, n (%) | ||||
| First trimester | 1 (2.8) | 0 (0) | 1 (5.3) | 0.34 |
| Second trimester | 4 (11.1) | 1 (5.9) | 3 (15.8) | 0.35 |
| Third trimester | 31 (86.1) | 16 (94.1) | 15 (78.9) | 0.19 |
| Admission during pregnancy, n (%) | 14 (38.9) | 9 (52.3) | 5 (26.3) | 0.1 |
| COVID-19 related | 8 (22.2) | 5 (29.4) | 3 (15.8)) | 0.33 |
| Respiratory failure | 7 (19.4) | 5 (29.4) | 2 (10.5) | |
| Vomiting | 1 (2.8) | 0 (0) | 1 (5.3) | |
| Non-COVID-19 related | 6 (16.7) | 4 (23.5) | 2 (10.5) | 0.3 |
| GA at delivery (days) median (IQR) | 269.5 (39) | 243 (48) | 271 (9) | 0.04 |
| Confirmed diagnosis to delivery (days) median (IQR) | 19.5 (33) | 4 (7) | 35 (52) | 0.00 |
| Preterm delivery, n (%) | 11 (30.6) | 9 (52.9) | 2 (10.5) | 0.01 |
| Spontaneous labour | 1 (2.8) | 0 (0) | 1 (5.3) | 0.03 |
| Labour by induction | 10 (27.8) | 9 (52.9) | 1 (5.3) | 0.03 |
| Mode of delivery, n (%) | ||||
| Vaginal spontaneous | 13 (36.1) | 4 (23.5) | 9 (47.4) | 0.14 |
| Vaginal instrumental | 1 (2.8) | 0 (0) | 1 (5.3) | 0.34 |
| Caesarean Section | 22 (61.2) | 13 (76.5) | 9 (47.4) | 0.07 |
| Type of caesarean section, n (%) | ||||
| Elective | 10 (27.8) | 3 (17.6) | 8 (42.1) | 0.19 |
| Emergency | 12 (32.3) | 10 (58.8) | 2 (10.5) | 0.002 |
| Indication for caesarean section, n (%) | ||||
| Elective | ||||
| Breech position | 3 (8.3) | 1 (5.9) | 2 (10.5) | 0.33 |
| Repeat caesarean section | 4 (11.1) | 2 (11.8) | 2 (10.5) | 0.68 |
| Other | 2 (5.6) | 0 (0) | 2 (10.5) | 0.08 |
| Unknown | 1 (2.8) | 0 (0) | 1 (5.3) | 0.22 |
| Emergency | ||||
| Foetal distress | 7 (19.4) | 5 (29.4) | 2 (10.5) | 0.42 |
| Maternal condition | 5 (13.9) | 5 (29.4) | 0 (0) | 0.03 |
| Total (n = 39) | Active COVID-19 (n = 18) | Resolved COVID-19 (n = 21) | p-Value | |
|---|---|---|---|---|
| Birthweight percentile for GA, median (IQR) | 48.0 (48) | 21.5 (69) | 51 (38) | 0.22 |
| SGA (BW < 10th percentile) | 9 (23.1) | 6 (33.3) | 3 (14.2) | 0.26 |
| Apgar <7 at 5 min, n (%) | 5 (12.8) | 5 (29.4) | 0 (0) | 0.03 |
| pH < 7.10, n (%) | 2 (5.1) | 2 (11.8) | 0 (0) | 0.17 |
| PCR nasopharynx SARS-CoV-2 positive (23/39) | 0 | - | - | - |
| PCR umbilical cord blood SARS-CoV-2 positive (12/39) | 0 | - | - | - |
| Ig neonatal SARS-CoV-2 positive (1/39) | 1 (2.6) | 0 (0.0) | 1 (5.3) | |
| Ig umbilical cord blood SARS-CoV-2 positive (14/39) | 6 (15.4) | 0 (0.0) | 6 (28.6) | 0.001 |
| Total (n = 39) | Active COVID-19 (n = 18) | Resolved COVID-19 (n = 21) | p-Value | Historical Healthy Term Controls, n = 583 [30,33] | |
|---|---|---|---|---|---|
| Normal maturation (%) | 29 (74.4) | 9 (50.0) | 20 (95.2) | 0.001 | 15–20% |
| Delayed maturation (%) | 10 (26.6) | 9 (50.0) | 1 (4.5) | ||
| Chorioamnionitis, n (%) | 10 (25.6) | 3 (16.7) | 7 (33.3) | 0.24 | 5–25% |
| Chronic membranitis, n (%) | 1 (2.6) | 1 (5.6) | 0 (0) | 0.27 | 8–19% (term) |
| Chronic deciduitis, n (%) | 6 (15.4) | 3 (16.7) | 3 (14.3) | 0.84 | 2–13% (term) |
| Chronic villitis, n (%) | 10 (25.6) | 5 (27.8) | 5 (23.8) | 0.78 | 2–25% |
| Resolved villitis, n (%) | 6 (15.4) | 2 (11.1) | 4 (19.0) | 0.49 | 5–15% |
| Chronic intervillositis, n (%) | 6 (15.4) | 3 (16.7) | 3 (14.3) | 0.84 | 0.4% |
| Positive SARS IHC, n (%) | 4 (10.3) | 3 (16.7) | 1 (4.8) | 0.22 | Not present |
| Perivillous fibrin | 9 (23.1) | 6 (33.3) | 3 (14.3) | 0.16 | 0.028% to 0.5% |
| Fibrinoid Necrosis | 4 (10.3) | 3 (16.7) | 1 (4.8) | 0.22 | Not present |
| Maternal CD20+ B-cell infiltrate | 5 (12.8) | 3 (16.7) | 2 (9.5) | 0.74 | Not present |
| PCR maternal side positive | 2 (5.1) | 2 (11.1) | 0 (0) | 0.12 | - |
| PCR foetal side positive | 2 (5.1) | 2 (11.1) | 0 (0) | 0.12 | - |
| Total (n = 36) | SARS-CoV-2 Immunohistochemical Positive Placenta (n = 4) | SARS-CoV2 Immunohistochemical Negative Placenta (n = 32) | p-Value | |
|---|---|---|---|---|
| Age, years, mean (±SD) | 33.5 (±5.1) | 31.5 (6.2) | 33.8 (5.0) | 0.40 |
| Pre-pregnancy BMI, kg/m2, median (IQR) | 28.8 (10.4) | 30.1 | 28.2 (10.1) | 1.00 |
| Missing (n = 4) | ||||
| Geographic origin, n (%) | ||||
| Caucasian | 10 (27.8) | 0 (0.0) | 10 (31.2) | 0.19 |
| Other | 26 (72.2) | 4 (100.0) | 22 (68.8) | 0.19 |
| Comorbidities (%) | ||||
| Missing (n = 2) | ||||
| Auto immune disease | 3 (8.3) | 0 (0.0) | 3 (9.8) | 0.17 |
| Pulmonary disease | 0 (0.0) | - | - | - |
| (Gestational) Diabetes | 6 (16.7) | 2 (50.0) | 4 (12.5) | 0.02 |
| Hypertensive disorders | 2 (5.6) | 0 (0.0) | 2 (6.3) | 0.18 |
| GA (in days) at diagnosis, median (IQR) | 237.5 (58) | 226.5 (34) | 243 (58) | 0.98 |
| Confirmed diagnosis to delivery (days) median (IQR) | 19.5 (33) | 2.5 (18) | 21 (51) | 0.06 |
| Admission during pregnancy, n (%) | 14 (38.9) | 2 (50.0) | 12 (37.5) | 0.63 |
| COVID-19 related | 8 (22.2) | 0 (0.0) | 8 (25.0) | 0.26 |
| Non-COVID-19 related | 6 (16.7) | 2 (50.0) | 4 (12.5) | 0.06 |
| Maternal PCR blood (n = 15) | 1 positive (2.8) | 1 (25.0) | 0 (0.0) | 0.000 |
| Maternal PCR fornix (n = 25) | 2 positive (5.6) | 2 (50.0) | 0 (0.0) | 0.000 |
| Signs of foetal distress, n (%) | 8 (22.2) | 3 (75.0) | 5 (15.6) | 0.007 |
| Total (n = 39) | SARS-CoV-2 Immunohistochemical Positive Placenta (n = 4) | SARS-CoV2 Immunohistochemical Negative Placenta (n = 35) | Historical Healthy Term Controls, n = 583 [30,33] | ||
|---|---|---|---|---|---|
| Perivillous fibrin, n (%) | 9 (23.1) | 4 (100.0) | 5 (14.3) | 0.000 | 0.028–0.5% |
| Fibrinoid Necrosis, n (%) | 4 (10.3) | 4 (100.0) | 0 (0.0) | 0.000 | Not present |
| Maternal CD20+ B-cell infiltrate, n (%) | 5 (12.8) | 4 (100.0) | 1 (2.9) | 0.000 | Not present |
| PCR maternal side positive (n = 30) | 2 positive (5.1) | 2 (50.0) | 0 (0.0) | 0.000 | |
| PCR foetal side positive (n = 29) | 2 positive (5.1) | 2 (50.0) | 0 (0.0) | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Husen, M.F.; van der Meeren, L.E.; Verdijk, R.M.; Fraaij, P.L.A.; van der Eijk, A.A.; Koopmans, M.P.G.; Freeman, L.; Bogers, H.; Trietsch, M.D.; Reiss, I.K.M.; et al. Unique Severe COVID-19 Placental Signature Independent of Severity of Clinical Maternal Symptoms. Viruses 2021, 13, 1670. https://doi.org/10.3390/v13081670
Husen MF, van der Meeren LE, Verdijk RM, Fraaij PLA, van der Eijk AA, Koopmans MPG, Freeman L, Bogers H, Trietsch MD, Reiss IKM, et al. Unique Severe COVID-19 Placental Signature Independent of Severity of Clinical Maternal Symptoms. Viruses. 2021; 13(8):1670. https://doi.org/10.3390/v13081670
Chicago/Turabian StyleHusen, Marjolein F., Lotte E. van der Meeren, Robert M. Verdijk, Pieter L. A. Fraaij, Annemiek A. van der Eijk, Marion P. G. Koopmans, Liv Freeman, Hein Bogers, Marjolijn D. Trietsch, Irwin K. M. Reiss, and et al. 2021. "Unique Severe COVID-19 Placental Signature Independent of Severity of Clinical Maternal Symptoms" Viruses 13, no. 8: 1670. https://doi.org/10.3390/v13081670
APA StyleHusen, M. F., van der Meeren, L. E., Verdijk, R. M., Fraaij, P. L. A., van der Eijk, A. A., Koopmans, M. P. G., Freeman, L., Bogers, H., Trietsch, M. D., Reiss, I. K. M., DeKoninck, P. L. J., & Schoenmakers, S. (2021). Unique Severe COVID-19 Placental Signature Independent of Severity of Clinical Maternal Symptoms. Viruses, 13(8), 1670. https://doi.org/10.3390/v13081670






