Efficacy of Unsupervised Self-Collected Mid-Turbinate FLOQSwabs for the Diagnosis of Coronavirus Disease 2019 (COVID-19)
Abstract
1. Introduction
2. Materials and Methods
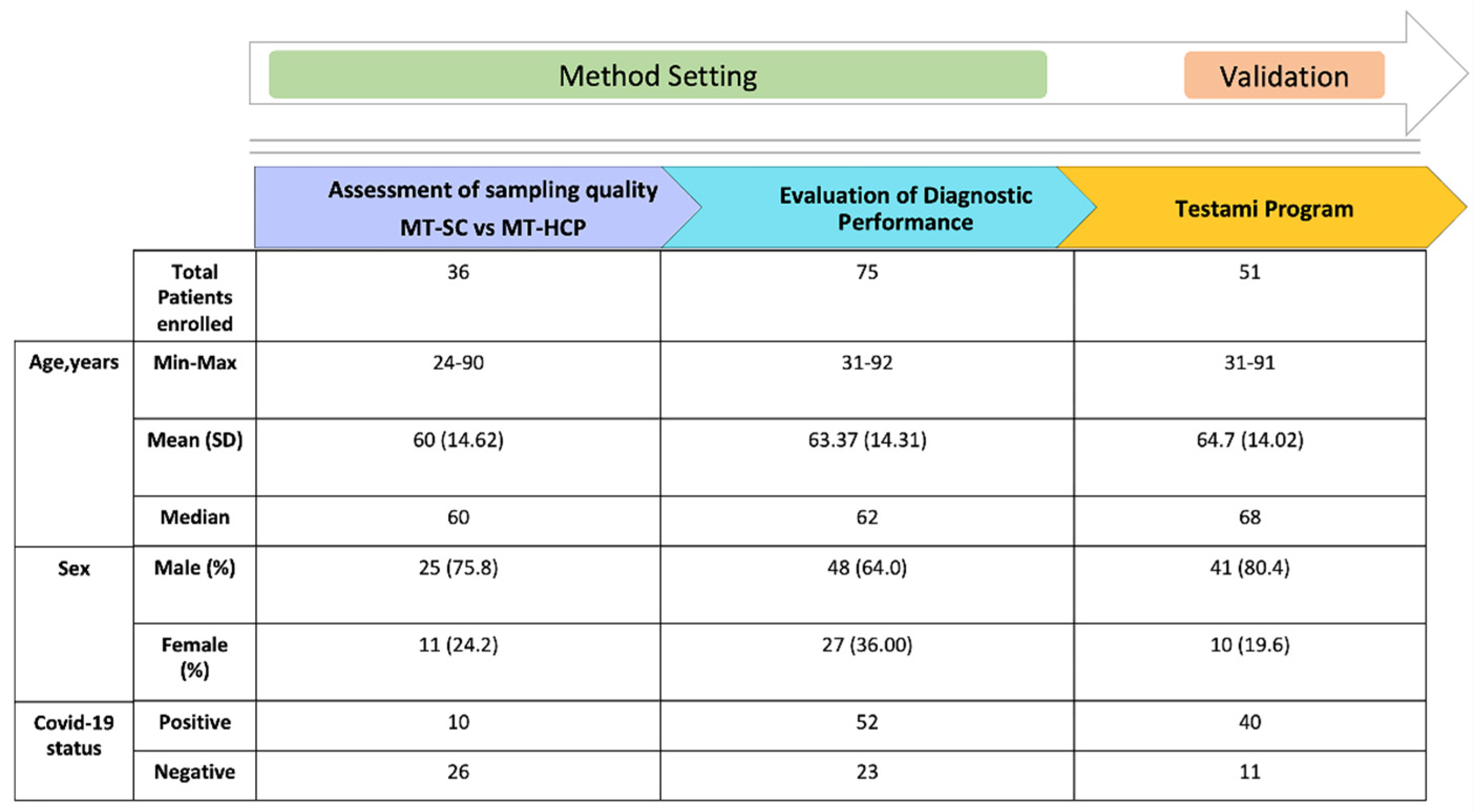
2.1. Subject and Speciemens
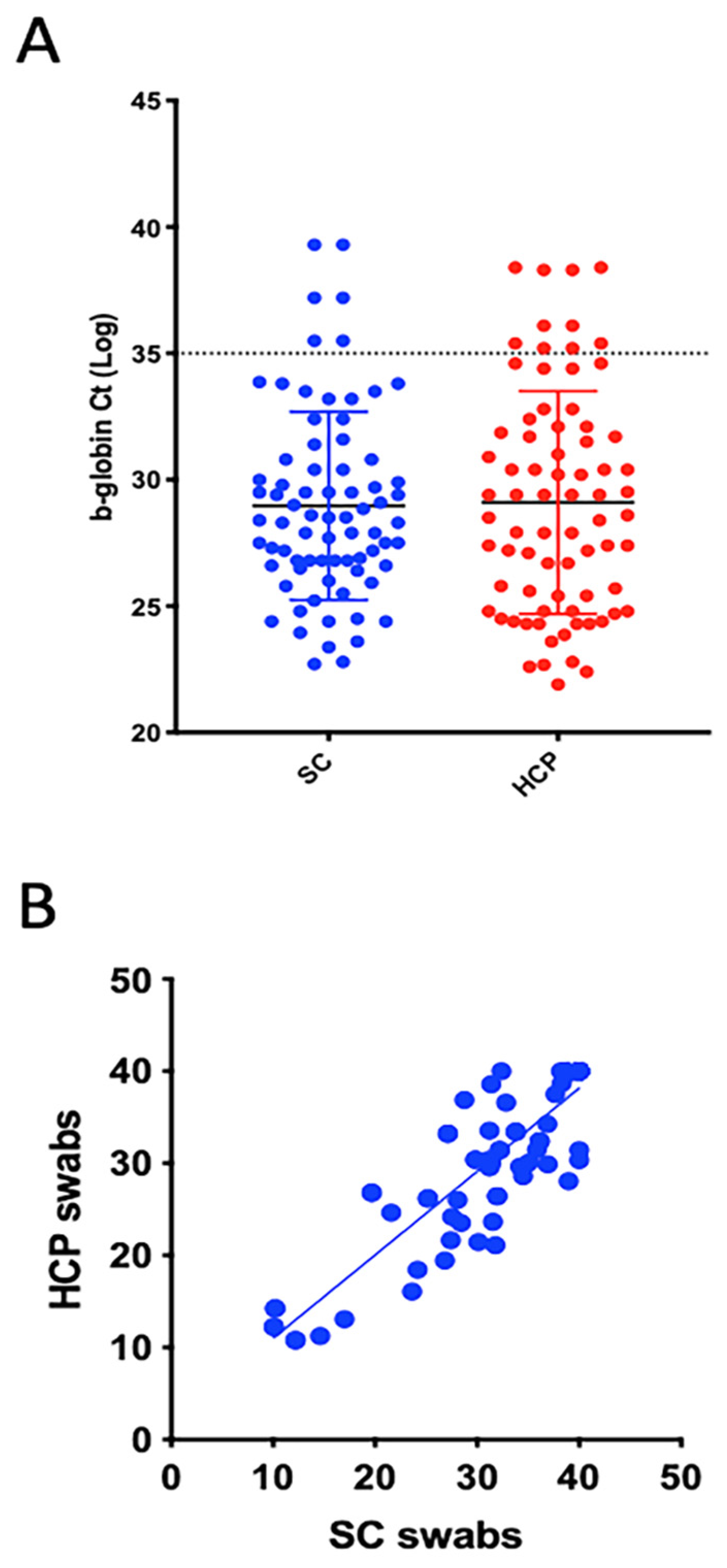
2.2. Specimen Collection
2.3. RNA Extraction and SARS-CoV-2 Detection
2.4. SARS-CoV-2 Virus Stability In Vitro and in Self-Collected MT Dry Swab
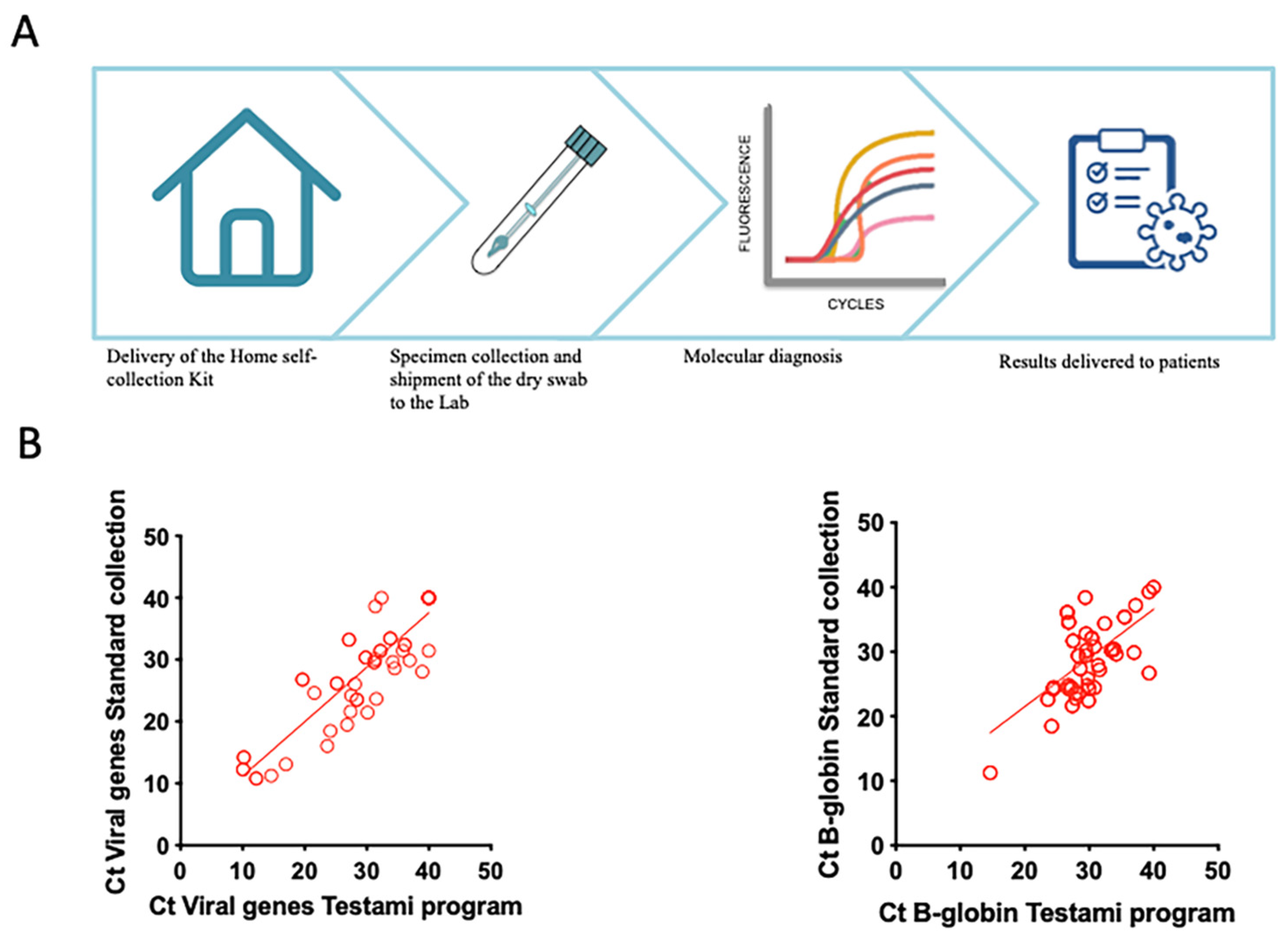
2.5. Evaluation of Diagnostic Performances of the “Testami Home Self-Collection Program”
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smieja, M.; Castriciano, S.; Carruthers, S.; So, G.; Chong, S.; Luinstra, K.; Mahony, J.B.; Petrich, A.; Chernesky, M.; Savarese, M.; et al. Development and Evaluation of a Flocked Nasal Midturbinate Swab for Self-Collection in Respiratory Virus Infection Diagnostic Testing. J. Clin. Microbiol. 2010, 48, 3340–3342. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. The species Severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Piralla, A.; Giardina, F.; Rovida, F.; Campanini, G.; Baldanti, F. Cellular DNA quantification in respiratory samples for the normalization of viral load: A real need? J. Clin. Virol. 2018, 107, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Faden, H. Comparison of Midturbinate Flocked-Swab Specimens with Nasopharyngeal Aspirates for Detection of Respiratory Viruses in Children by the Direct Fluorescent Antibody Technique. J. Clin. Microbiol. 2010, 48, 3742–3743. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Esposito, S.; Molteni, C.G.; Daleno, C.; Valzano, A.; Tagliabue, C.; Galeone, C.; Milani, G.; Fossali, E.; Marchisio, P.; Principi, N. Collection by trained pediatricians or parents of mid-turbinate nasal flocked swabs for the detection of influenza viruses in childhood. Virol. J. 2010, 7, 85. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, N.; Miller, R.M.; Finley, J.L.; Sztajnkrycer, M.D.; Nestler, D.M.; Boggust, A.J.; Jenkins, S.M.; Smith, T.F.; Wilson, J.W.; Cockerill, F.R.; et al. Effectiveness of Patient-Collected Swabs for Influenza Testing. Mayo Clin. Proc. 2012, 87, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Larios, O.E.; Coleman, B.L.; Drews, S.J.; Mazzulli, T.; Borgundvaag, B.; Green, K.; McGeer, A. STOP-Flu Study Group Self-Collected Mid-Turbinate Swabs for the Detection of Respiratory Viruses in Adults with Acute Respiratory Illnesses. PLoS ONE 2011, 6, e21335. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, Q.Y.; Li, Y.Y.; Wang, Y.F.; Yang, Z.F.; Zhong, N.S. Comparison among nasopharyngeal swab, nasal wash, and oropharyngeal swab for respiratory virus detection in adults with acute pharyngitis. BMC Infect. Dis. 2013, 13, 281. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.; Corden, S.; Sinha, J.; Jones, R. Dry cotton or flocked respiratory swabs as a simple collection technique for the molecular detection of respiratory viruses using real-time NASBA. J. Virol. Methods 2008, 153, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Davies, E.; Whitfield, T.; Machin, N.; Ahmad, S. The utility of beta-2-microglobulin testing as a human cellular control in COVID-19 testing. J. Clin. Virol. 2020, 129, 104449. [Google Scholar] [CrossRef] [PubMed]

| HCP Swabs | |||||
|---|---|---|---|---|---|
| Total | COVID-19 Positive | COVID-19 Negative | p-Value | ||
| SC Swabs | COVID-19 Positive | 50 | 49 | 1 | <0.05 |
| COVID-19 Negative | 25 | 3 | 22 | ||
| Total | 75 | 52 | 23 | ||
| Standard Diagnostic Program (eNat Swab) | |||||
|---|---|---|---|---|---|
| Total | COVID-19 Positive | COVID-19 Negative | p-Value | ||
| Testami program | COVID-19 Positive | 39 | 38 | 1 | <0.05 |
| COVID-19 Negative | 12 | 2 | 10 | ||
| Total | 51 | 40 | 11 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Andrea, E.L.; Cossu, A.M.; Scrima, M.; Messina, V.; Iuliano, P.; Di Perna, F.; Pizza, M.; Pizza, F.; Coppola, N.; Rinaldi, L.; et al. Efficacy of Unsupervised Self-Collected Mid-Turbinate FLOQSwabs for the Diagnosis of Coronavirus Disease 2019 (COVID-19). Viruses 2021, 13, 1663. https://doi.org/10.3390/v13081663
D’Andrea EL, Cossu AM, Scrima M, Messina V, Iuliano P, Di Perna F, Pizza M, Pizza F, Coppola N, Rinaldi L, et al. Efficacy of Unsupervised Self-Collected Mid-Turbinate FLOQSwabs for the Diagnosis of Coronavirus Disease 2019 (COVID-19). Viruses. 2021; 13(8):1663. https://doi.org/10.3390/v13081663
Chicago/Turabian StyleD’Andrea, Egildo Luca, Alessia Maria Cossu, Marianna Scrima, Vincenzo Messina, Pasquale Iuliano, Felice Di Perna, Marco Pizza, Fabio Pizza, Nicola Coppola, Luca Rinaldi, and et al. 2021. "Efficacy of Unsupervised Self-Collected Mid-Turbinate FLOQSwabs for the Diagnosis of Coronavirus Disease 2019 (COVID-19)" Viruses 13, no. 8: 1663. https://doi.org/10.3390/v13081663
APA StyleD’Andrea, E. L., Cossu, A. M., Scrima, M., Messina, V., Iuliano, P., Di Perna, F., Pizza, M., Pizza, F., Coppola, N., Rinaldi, L., Bellizzi, A. M., Pelosi, C., Cocca, C., Frieri, A., Lo Calzo, F., Capasso, G., Castriciano, S., Maggi, P., Fucci, A., & Caraglia, M. (2021). Efficacy of Unsupervised Self-Collected Mid-Turbinate FLOQSwabs for the Diagnosis of Coronavirus Disease 2019 (COVID-19). Viruses, 13(8), 1663. https://doi.org/10.3390/v13081663








