On the Use of Phylogeographic Inference to Infer the Dispersal History of Rabies Virus: A Review Study
Abstract
1. Introduction
2. (Lack of) Genomic Data, Geographic Scope and Methodological Expertise
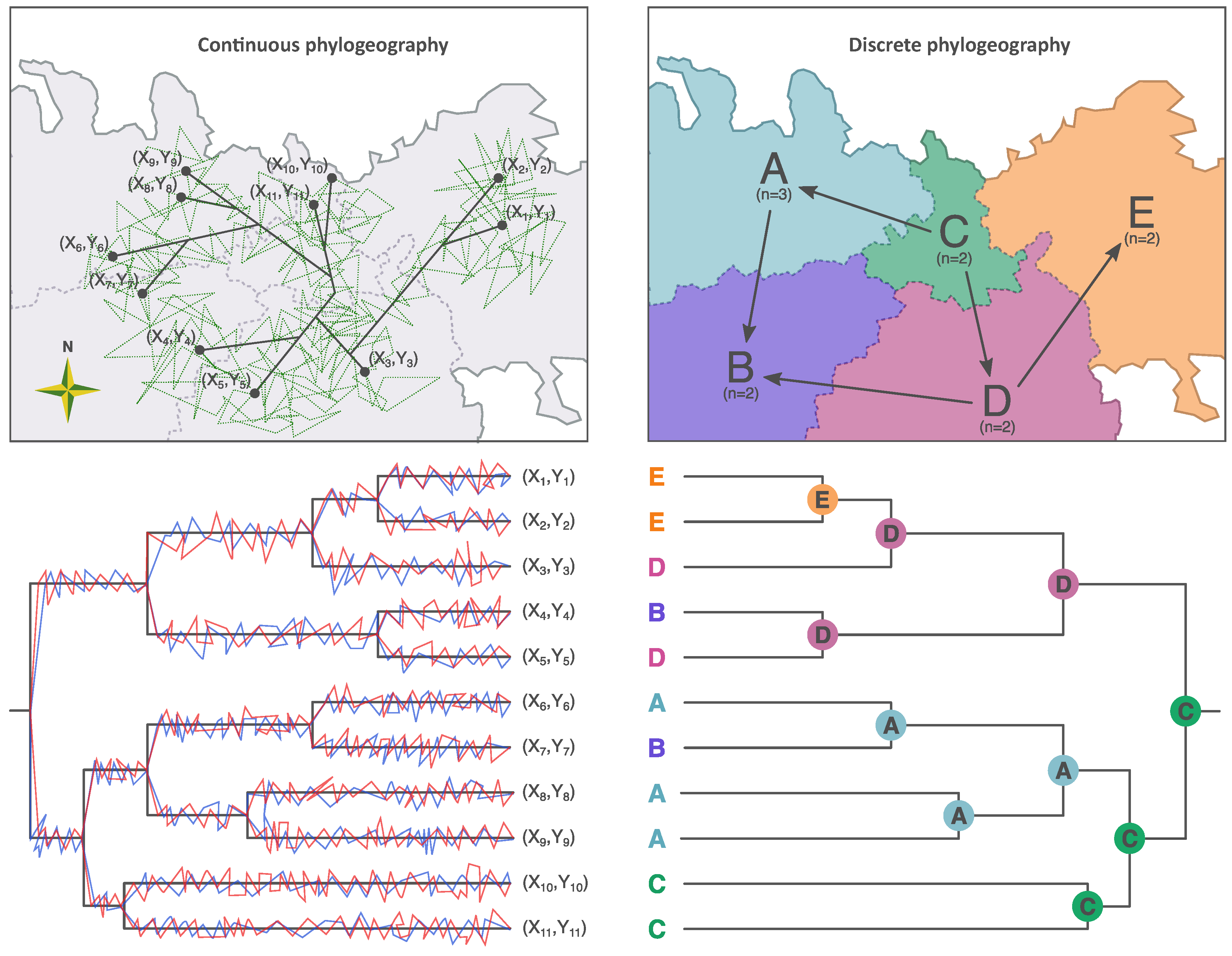
3. Discrete Phylogeographic Inference
3.1. Host Species
3.1.1. Dogs
3.1.2. Bats
3.1.3. Raccoons
3.1.4. Badgers
3.1.5. Livestock
4. Continuous Phylogeographic Inference
4.1. Host Species
4.1.1. Dogs
4.1.2. Bats
4.1.3. Skunks
4.1.4. Raccoons
4.1.5. Foxes
5. Meta-Analysis of Relevant Environmental Factors
6. Recommendations and Useful Resources
7. Novel Methodological Developments and Future Perspectives
7.1. Formal Assessment of Temporal Signal
7.2. Maximum-Likelihood Phylogeographic Inference
7.3. Mitigating Sampling Bias
7.4. Assessing (Hypothetical) Intervention Strategies
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| RABV | Rabies Virus |
| R&D | Research and Development |
| GDP | Gross domestic product |
| SCSK | south-central skunk RABV |
| VBRV | vampire bats RABV |
| MCMC | Markov chain Monte Carlo |
| ML | Maximum likelihood |
| BASTA | Bayesian structured coalescent approximation |
| FV | -Fleming-Viot |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
Appendix A
| Publication | Year | Tested Environmental Factors |
|---|---|---|
| Talbi et al. [43] | 2010 | geographical distances, human population size, road distances, a gravity model, spatial accessibility, population conductivity |
| Brunker et al. [6] | 2018 | dog density, dog presence, elevation, human-to-dog ratio, rivers, roads, slope, average vaccination coverage, number of vaccination campaigns, susceptible dog density |
| Lan et al. [53] | 2017 | ecological and geographical barriers |
| Publication | Year | Tested Environmental Factors |
|---|---|---|
| Streicker et al. [71] | 2016 | river valleys |
| Pepin et al. [67] | 2017 | neighbourhood, season, cardinal direction |
| Dellicour et al. [5] | 2017 | inaccessibility, barren vegetation, croplands, forests, grasslands, savannas, urban areas, elevation, human population density |
| Tian et al. [19] | 2018 | elevation, annual mean temperature, annual precipitation, key land cover variables, human population density, human footprint, major roads, and inaccessibility |
| Dellicour et al. [20] | 2019 | elevation, barren vegetation, shrublands, grasslands, croplands, major roads, inaccessibility, human population density |
References
- Brunker, K.; Mollentze, N. Rabies virus. Trends Microbiol. 2018, 26, 886–887. [Google Scholar] [CrossRef]
- Torres, C.; Lema, C.; Dohmen, F.G.; Beltran, F.; Novaro, L.; Russo, S.; Freire, M.; Velasco-Villa, A.; Mbayed, V.A.; Cisterna, D. Phylodynamics of vampire bat-transmitted rabies in A rgentina. Mol. Ecol. 2014, 23, 2340–2352. [Google Scholar] [CrossRef]
- Fenton, M.B.; Streicker, D.G.; Racey, P.A.; Tuttle, M.D.; Medellin, R.A.; Daley, M.J.; Recuenco, S.; Bakker, K.M. Knowledge gaps about rabies transmission from vampire bats to humans. Nat. Ecol. Evol. 2020, 4, 517–518. [Google Scholar] [CrossRef]
- Pybus, O.G.; Rambaut, A. Evolutionary analysis of the dynamics of viral infectious disease. Nat. Rev. Genet. 2009, 10, 540–550. [Google Scholar] [CrossRef]
- Dellicour, S.; Rose, R.; Faria, N.R.; Vieira, L.F.P.; Bourhy, H.; Gilbert, M.; Lemey, P.; Pybus, O.G. Using viral gene sequences to compare and explain the heterogeneous spatial dynamics of virus epidemics. Mol. Biol. Evol. 2017, 34, 2563–2571. [Google Scholar] [CrossRef]
- Brunker, K.; Lemey, P.; Marston, D.A.; Fooks, A.R.; Lugelo, A.; Ngeleja, C.; Hampson, K.; Biek, R. Landscape attributes governing local transmission of an endemic zoonosis: Rabies virus in domestic dogs. Mol. Ecol. 2018, 27, 773–788. [Google Scholar] [CrossRef]
- Fisher, C.R.; Streicker, D.G.; Schnell, M.J. The spread and evolution of rabies virus: Conquering new frontiers. Nat. Rev. Microbiol. 2018, 16, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Katz, I.; Mori, E.; Carnieli, P.; Vieira, L.; Batista, H.; Chaves, L.; Scheffer, K. Biotechnology advances: A perspective on the diagnosis and research of Rabies Virus. Biologicals 2013, 41, 217–223. [Google Scholar] [CrossRef]
- Quick, J.; Loman, N.J.; Duraffour, S.; Simpson, J.T.; Severi, E.; Cowley, L.; Bore, J.A.; Koundouno, R.; Dudas, G.; Mikhail, A.; et al. Real-time, portable genome sequencing for Ebola surveillance. Nature 2016, 530, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Biek, R.; Henderson, J.C.; Waller, L.A.; Rupprecht, C.E.; Real, L.A. A high-resolution genetic signature of demographic and spatial expansion in epizootic rabies virus. Proc. Natl. Acad. Sci. USA 2007, 104, 7993–7998. [Google Scholar] [CrossRef]
- Baele, G.; Suchard, M.A.; Rambaut, A.; Lemey, P. Emerging concepts of data integration in pathogen phylodynamics. Syst. Biol. 2017, 66, e47–e65. [Google Scholar] [CrossRef]
- Lemey, P.; Rambaut, A.; Drummond, A.J.; Suchard, M.A. Bayesian phylogeography finds its roots. PLoS Comput. Biol. 2009, 5, e1000520. [Google Scholar] [CrossRef] [PubMed]
- Lemey, P.; Rambaut, A.; Welch, J.J.; Suchard, M.A. Phylogeography takes a relaxed random walk in continuous space and time. Mol. Biol. Evol. 2010, 27, 1877–1885. [Google Scholar] [CrossRef]
- Suchard, M.A.; Lemey, P.; Baele, G.; Ayres, D.L.; Drummond, A.J.; Rambaut, A. Bayesian phylogenetic and phylodynamic data integration using BEAST 1.10. Virus Evol. 2018, 4, vey016. [Google Scholar] [CrossRef] [PubMed]
- Layan, M.; Dellicour, S.; Baele, G.; Cauchemez, S.; Bourhy, H. Mathematical modelling and phylodynamics for the study of dog rabies dynamics and control: A scoping review. PLoS Negl. Trop. Dis. 2021, 15, e0009449. [Google Scholar] [CrossRef] [PubMed]
- Brunker, K.; Jaswant, G.; Thumbi, S.; Lushasi, K.; Lugelo, A.; Czupryna, A.M.; Ade, F.; Wambura, G.; Chuchu, V.; Steenson, R.; et al. Rapid in-country sequencing of whole virus genomes to inform rabies elimination programmes. Wellcome Open Res. 2020, 5, 3. [Google Scholar] [CrossRef]
- Houlihan, C.F.; Frampton, D.; Ferns, R.B.; Raffle, J.; Grant, P.; Reidy, M.; Hail, L.; Thomson, K.; Mattes, F.; Kozlakidis, Z.; et al. Use of whole-genome sequencing in the investigation of a nosocomial influenza virus outbreak. J. Infect. Dis. 2018, 218, 1485–1489. [Google Scholar] [CrossRef]
- Maurier, F.; Beury, D.; Fléchon, L.; Varré, J.S.; Touzet, H.; Goffard, A.; Hot, D.; Caboche, S. A complete protocol for whole-genome sequencing of virus from clinical samples: Application to coronavirus OC43. Virology 2019, 531, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Feng, Y.; Vrancken, B.; Cazelles, B.; Tan, H.; Gill, M.S.; Yang, Q.; Li, Y.; Yang, W.; Zhang, Y.; et al. Transmission dynamics of re-emerging rabies in domestic dogs of rural China. PLoS Pathog. 2018, 14, e1007392. [Google Scholar] [CrossRef]
- Dellicour, S.; Troupin, C.; Jahanbakhsh, F.; Salama, A.; Massoudi, S.; Moghaddam, M.K.; Baele, G.; Lemey, P.; Gholami, A.; Bourhy, H. Using phylogeographic approaches to analyse the dispersal history, velocity and direction of viral lineages—Application to rabies virus spread in Iran. Mol. Ecol. 2019, 28, 4335–4350. [Google Scholar] [CrossRef]
- Duong, V.; Tarantola, A.; Ong, S.; Mey, C.; Choeung, R.; Ly, S.; Bourhy, H.; Dussart, P.; Buchy, P. Laboratory diagnostics in dog-mediated rabies: An overview of performance and a proposed strategy for various settings. Int. J. Infect. Dis. 2016, 46, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Kuzmina, N.A.; Lemey, P.; Kuzmin, I.V.; Mayes, B.C.; Ellison, J.A.; Orciari, L.A.; Hightower, D.; Taylor, S.T.; Rupprecht, C.E. The phylogeography and spatiotemporal spread of south-central skunk rabies virus. PLoS ONE 2013, 8, e82348. [Google Scholar] [CrossRef] [PubMed]
- Omodo, M.; Gouilh, M.A.; Mwiine, F.N.; Okurut, A.R.A.; Nantima, N.; Namatovu, A.; Nakanjako, M.F.; Isingoma, E.; Arinaitwe, E.; Esau, M.; et al. Rabies in Uganda: Rabies knowledge, attitude and practice and molecular characterization of circulating virus strains. BMC Infect. Dis. 2020, 20, 200. [Google Scholar] [CrossRef]
- Tao, X.Y.; Li, M.L.; Guo, Z.Y.; Yan, J.H.; Zhu, W.Y. Inner Mongolia: A Potential Portal for the Spread of Rabies to Western China. Vector-Borne Zoonotic Dis. 2019, 19, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Ahmed, K.; Karunanayake, D.; Wimalaratne, O.; Nanayakkara, S.; Perera, D.; Kobayashi, Y.; Nishizono, A. Molecular epidemiology of human rabies viruses in Sri Lanka. Infect. Genet. Evol. 2013, 18, 160–167. [Google Scholar] [CrossRef]
- Saito, M.; Oshitani, H.; Orbina, J.R.C.; Tohma, K.; de Guzman, A.S.; Kamigaki, T.; Demetria, C.S.; Manalo, D.L.; Noguchi, A.; Inoue, S.; et al. Genetic Diversity and Geographic Distribution of Genetically Distinct Rabies Viruses in the Philippines. PLoS Negl. Trop. Dis. 2013, 7, e2144. [Google Scholar] [CrossRef]
- Pant, G.R.; Lavenir, R.; Wong, F.Y.K.; Certoma, A.; Larrous, F.; Bhatta, D.R.; Bourhy, H.; Stevens, V.; Dacheux, L. Recent Emergence and Spread of an Arctic-Related Phylogenetic Lineage of Rabies Virus in Nepal. PLoS Negl. Trop. Dis. 2013, 7, e2560. [Google Scholar] [CrossRef]
- Cargnelutti, J.F.; de Quadros, J.M.; Martins, M.; Batista, H.B.C.R.; Weiblen, R.; Flores, E.F. Glycoprotein-G-gene-based molecular and phylogenetic analysis of rabies viruses associated with a large outbreak of bovine rabies in southern Brazil. Arch. Virol. 2017, 162, 3697–3704. [Google Scholar] [CrossRef]
- Sadeuh-Mba, S.A.; Momo, J.B.; Besong, L.; Loul, S.; Njouom, R. Molecular characterization and phylogenetic relatedness of dog-derived Rabies Viruses circulating in Cameroon between 2010 and 2016. PLoS Negl. Trop. Dis. 2017, 11, e0006041. [Google Scholar] [CrossRef]
- Tricou, V.; Bouscaillou, J.; Mebourou, E.K.; Koyanongo, F.D.; Nakouné, E.; Kazanji, M. Surveillance of Canine Rabies in the Central African Republic: Impact on Human Health and Molecular Epidemiology. PLoS Negl. Trop. Dis. 2016, 10, e0004433. [Google Scholar] [CrossRef]
- Zieger, U.; Marston, D.A.; Sharma, R.; Chikweto, A.; Tiwari, K.; Sayyid, M.; Louison, B.; Goharriz, H.; Voller, K.; Breed, A.C.; et al. The phylogeography of rabies in Grenada, West Indies, and implications for control. PLoS Negl. Trop. Dis. 2014, 8, e3251. [Google Scholar] [CrossRef] [PubMed]
- McElhinney, L.M.; Marston, D.A.; Freuling, C.M.; Cragg, W.; Stankov, S.; Lalosević, D.; Lalosević, V.; Müller, T.; Fooks, A.R. Molecular diversity and evolutionary history of rabies virus strains circulating in the Balkans. J. Gen. Virol. 2011, 92, 2171–2180. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Polupan, I.; Bezymennyi, M.; Gibaliuk, Y.; Drozhzhe, Z.; Rudoi, O.; Ukhovskyi, V.; Nedosekov, V.; Nardi, M.D. An Analysis of Rabies Incidence and Its Geographic Spread in the Buffer Area Among Orally Vaccinated Wildlife in Ukraine From 2012 to 2016. Front. Vet. Sci. 2019, 6, 290. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Zhou, H.; Zou, Y.; Yin, W.; Yu, H.; Si, Y.; Li, J.; Zhou, Y.; Zhou, X.; Magalhães, R.J.S. Geographical Analysis of the Distribution and Spread of Human Rabies in China from 2005 to 2011. PLoS ONE 2013, 8, e72352. [Google Scholar] [CrossRef]
- Bitek, A.O.; Osoro, E.; Munyua, P.M.; Nanyingi, M.; Muthiani, Y.; Kiambi, S.; Muturi, M.; Mwatondo, A.; Muriithi, R.; Cleaveland, S.; et al. A hundred years of rabies in Kenya and the strategy for eliminating dog-mediated rabies by 2030. AAS Open Res. 2019, 1, 23. [Google Scholar] [CrossRef]
- Kole, A.K.; Roy, R.; Kole, D.C. Human rabies in India: A problem needing more attention. Bull. World Health Organ. 2014, 92. [Google Scholar] [CrossRef]
- Ma, C.; Hao, X.; Deng, H.; Wu, R.; Liu, J.; Yang, Y.; Li, S.; Han, Y.; Wei, M.; Zhang, J.; et al. Re-emerging of rabies in Shaanxi province, China, from 2009 to 2015. J. Med Virol. 2017, 89, 1511–1519. [Google Scholar] [CrossRef]
- Hampson, K.; Coudeville, L.; Lembo, T.; Sambo, M.; Kieffer, A.; Attlan, M.; Barrat, J.; Blanton, J.D.; Briggs, D.J.; Cleaveland, S.; et al. Estimating the Global Burden of Endemic Canine Rabies. PLoS Negl. Trop. Dis. 2015, 9, e0003709. [Google Scholar] [CrossRef]
- Ferreira, M.A.; Suchard, M.A. Bayesian analysis of elapsed times in continuous-time Markov chains. Can. J. Stat. 2008, 36, 355–368. [Google Scholar] [CrossRef]
- Lemey, P.; Rambaut, A.; Bedford, T.; Faria, N.; Bielejec, F.; Baele, G.; Russell, C.A.; Smith, D.J.; Pybus, O.G.; Brockmann, D.; et al. Unifying viral genetics and human transportation data to predict the global transmission dynamics of human influenza H3N2. PLoS Path. 2014, 10, e1003932. [Google Scholar] [CrossRef]
- Brunker, K.; Marston, D.A.; Horton, D.L.; Cleaveland, S.; Fooks, A.R.; Kazwala, R.; Ngeleja, C.; Lembo, T.; Sambo, M.; Mtema, Z.J.; et al. Elucidating the phylodynamics of endemic rabies virus in eastern Africa using whole-genome sequencing. Virus Evol. 2015, 1, vev011. [Google Scholar] [CrossRef]
- Hayman, D.T.S.; Johnson, N.; Horton, D.L.; Hedge, J.; Wakeley, P.R.; Banyard, A.C.; Zhang, S.; Alhassan, A.; Fooks, A.R. Evolutionary History of Rabies in Ghana. PLoS Negl. Trop. Dis. 2011, 5, e1001. [Google Scholar] [CrossRef]
- Talbi, C.; Lemey, P.; Suchard, M.A.; Abdelatif, E.; Elharrak, M.; Jalal, N.; Faouzi, A.; Echevarría, J.E.; Morón, S.V.; Rambaut, A.; et al. Phylodynamics and human-mediated dispersal of a zoonotic virus. PLoS Pathog. 2010, 6, e1001166. [Google Scholar] [CrossRef]
- Mollentze, N.; Weyer, J.; Markotter, W.; le Roux, K.; Nel, L.H. Dog rabies in southern Africa: Regional surveillance and phylogeographical analyses are an important component of control and elimination strategies. Virus Genes 2013, 47, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Seetahal, J.F.R.; Velasco-Villa, A.; Allicock, O.M.; Adesiyun, A.A.; Bissessar, J.; Amour, K.; Phillip-Hosein, A.; Marston, D.A.; McElhinney, L.M.; Shi, M.; et al. Evolutionary History and Phylogeography of Rabies Viruses Associated with Outbreaks in Trinidad. PLoS Negl. Trop. Dis. 2013, 7, e2365. [Google Scholar] [CrossRef] [PubMed]
- Trewby, H.; Nadin-Davis, S.A.; Real, L.A.; Biek, R. Processes Underlying Rabies Virus Incursions across US–Canada Border as Revealed by Whole-Genome Phylogeography. Emerg. Infect. Dis. 2017, 23, 1454–1461. [Google Scholar] [CrossRef] [PubMed]
- Streicker, D.G.; González, S.L.F.; Luconi, G.; Barrientos, R.G.; Leon, B. Phylodynamics reveals extinction–recolonization dynamics underpin apparently endemic vampire bat rabies in Costa Rica. Proc. R. Soc. Biol. Sci. 2019, 286, 20191527. [Google Scholar] [CrossRef] [PubMed]
- Carnieli, P.; de Novaes Oliveira, R.; Macedo, C.I.; Castilho, J.G. Phylogeography of rabies virus isolated from dogs in Brazil between 1985 and 2006. Arch. Virol. 2011, 156, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Horton, D.L.; McElhinney, L.M.; Freuling, C.M.; Marston, D.A.; Banyard, A.C.; Goharrriz, H.; Wise, E.; Breed, A.C.; Saturday, G.; Kolodziejek, J.; et al. Complex Epidemiology of a Zoonotic Disease in a Culturally Diverse Region: Phylogeography of Rabies Virus in the Middle East. PLoS Negl. Trop. Dis. 2015, 9, e0003569. [Google Scholar] [CrossRef]
- Tohma, K.; Saito, M.; Kamigaki, T.; Tuason, L.T.; Demetria, C.S.; Orbina, J.R.C.; Manalo, D.L.; Miranda, M.E.; Noguchi, A.; Inoue, S.; et al. Phylogeographic analysis of rabies viruses in the Philippines. Infect. Genet. Evol. 2014, 23, 86–94. [Google Scholar] [CrossRef]
- Dibia, I.N.; Sumiarto, B.; Susetya, H.; Putra, A.A.G.; Scott-Orr, H.; Mahardika, G.N. Phylogeography of the current rabies viruses in Indonesia. J. Vet. Sci. 2015, 16, 459. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Chu, P.Y.; Chang, M.Y.; Hsiao, K.L.; Lin, J.H.; Liu, H.F. Spatial Temporal Dynamics and Molecular Evolution of Re-Emerging Rabies Virus in Taiwan. Int. J. Mol. Sci. 2016, 17, 392. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.C.; Wen, T.H.; Chang, C.C.; Liu, H.F.; Lee, P.F.; Huang, C.Y.; Chomel, B.B.; Chen, Y.M.A. Indigenous Wildlife Rabies in Taiwan: Ferret Badgers, a Long Term Terrestrial Reservoir. BioMed Res. Int. 2017, 2017, 5491640. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Li, H.; Tang, Q.; Rayner, S.; Han, N.; Guo, Z.; Liu, H.; Adams, J.; Fang, W.; Tao, X.; et al. The Spatial and Temporal Dynamics of Rabies in China. PLoS Negl. Trop. Dis. 2012, 6, e1640. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Tao, X.; Yin, C.; Han, N.; Yu, J.; Li, H.; Liu, H.; Fang, W.; Adams, J.; Wang, J.; et al. National Borders Effectively Halt the Spread of Rabies: The Current Rabies Epidemic in China Is Dislocated from Cases in Neighboring Countries. PLoS Negl. Trop. Dis. 2013, 7, e2039. [Google Scholar] [CrossRef]
- Zhang, Y.; Vrancken, B.; Feng, Y.; Dellicour, S.; Yang, Q.; Yang, W.; Zhang, Y.; Dong, L.; Pybus, O.G.; Zhang, H.; et al. Cross-border spread, lineage displacement and evolutionary rate estimation of rabies virus in Yunnan Province, China. Virol. J. 2017, 14, 102. [Google Scholar] [CrossRef]
- Wang, L.; Wu, X.; Bao, J.; Song, C.; Du, J. Phylodynamic and transmission pattern of rabies virus in China and its neighboring countries. Arch. Virol. 2019, 164, 2119–2129. [Google Scholar] [CrossRef]
- Yu, X.; Zhu, H.; Bo, Y.; Li, Y.; Zhang, J.; Jiang, L.; Chen, G.; Zhang, X.; Wen, Y. Molecular evolutionary analysis reveals Arctic-like rabies viruses evolved and dispersed independently in North and South Asia. J. Vet. Sci. 2021, 22, e5. [Google Scholar] [CrossRef]
- Bleck, T.P. CHAPTER 79—Rabies. In Tropical Infectious Diseases: Principles, Pathogens and Practice, 3rd ed.; Guerrant, R.L., Walker, D.H., Weller, P.F., Eds.; W.B. Saunders: Edinburgh, UK, 2011; pp. 525–531. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Suzuki, Y.; Itou, T.; Ito, F.H.; Sakai, T.; Gojobori, T. Evolutionary history of dog rabies in Brazil. J. Gen. Virol. 2011, 92, 85–90. [Google Scholar] [CrossRef]
- Carnieli, P., Jr.; de Oliveira Fahl, W.; Castilho, J.G.; de Novaes Oliveira, R.; Macedo, C.I.; Durymanova, E.; Jorge, R.S.; Morato, R.G.; Spíndola, R.O.; Machado, L.M.; et al. Characterization of Rabies virus isolated from canids and identification of the main wild canid host in Northeastern Brazil. Virus Res. 2008, 131, 33–46. [Google Scholar] [CrossRef]
- Amouri, I.K.; Kharmachi, H.; Djebbi, A.; Saadi, M.; Hogga, N.; Zakour, L.B.; Ghram, A. Molecular characterization of rabies virus isolated from dogs in Tunisia: Evidence of two phylogenetic variants. Virus Res. 2011, 158, 246–250. [Google Scholar] [CrossRef]
- Chang, S.S.; Tsai, H.J.; Chang, F.Y.; Lee, T.S.; Huang, K.C.; Fang, K.Y.; Wallace, R.; Inoue, S.; Fei, C.Y. Government Response to the Discovery of a Rabies Virus Reservoir Species on a Previously Designated Rabies-Free Island, Taiwan, 1999–2014. Zoonoses Public Health 2016, 63, 396–402. [Google Scholar] [CrossRef]
- Shih, T.H.; Chiang, J.T.; Wu, H.Y.; Inoue, S.; Tsai, C.T.; Kuo, S.C.; Yang, C.Y.; Fei, C.Y. Human exposure to ferret badger rabies in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 1347. [Google Scholar] [CrossRef]
- De Maio, N.; Wu, C.H.; O’Reilly, K.M.; Wilson, D. New routes to phylogeography: A Bayesian structured coalescent approximation. PLoS Genet. 2015, 11, e1005421. [Google Scholar] [CrossRef]
- Dellicour, S.; Rose, R.; Faria, N.R.; Lemey, P.; Pybus, O.G. SERAPHIM: Studying environmental rasters and phylogenetically informed movements. Bioinformatics 2016, 32, 3204–3206. [Google Scholar] [CrossRef] [PubMed]
- Pepin, K.M.; Davis, A.J.; Streicker, D.G.; Fischer, J.W.; VerCauteren, K.C.; Gilbert, A.T. Predicting spatial spread of rabies in skunk populations using surveillance data reported by the public. PLoS Negl. Trop. Dis. 2017, 11, e0005822. [Google Scholar] [CrossRef]
- Musial, T.; Duke-Sylvester, S.; Davis, R.; Biek, R.; Real, L.A. Persistent phylogeographic structure of an emerging virus on a homogeneous landscape. PeerJ Prepr. 2018, 6, e27260v1. [Google Scholar]
- Carnieli, P.; Batista, H.B.R.; de Novaes Oliveira, R.; Castilho, J.G.; Vieira, L.F.P. Phylogeographic dispersion and diversification of rabies virus lineages associated with dogs and crab-eating foxes (Cerdocyon thous) in Brazil. Arch. Virol. 2013, 158, 2307–2313. [Google Scholar] [CrossRef] [PubMed]
- Vieira, L.F.P.; Pereira, S.R.F.G.; Carnieli, P.; Tavares, L.C.B.; Kotait, I. Phylogeography of rabies virus isolated from herbivores and bats in the Espírito Santo State, Brazil. Virus Genes 2013, 46, 330–336. [Google Scholar] [CrossRef]
- Streicker, D.G.; Winternitz, J.C.; Satterfield, D.A.; Condori-Condori, R.E.; Broos, A.; Tello, C.; Recuenco, S.; Velasco-Villa, A.; Altizer, S.; Valderrama, W. Host-pathogen evolutionary signatures reveal dynamics and future invasions of vampire bat rabies. Proc. Natl. Acad. Sci. USA 2016, 113, 10926–10931. [Google Scholar] [CrossRef]
- Fusaro, A.; Monne, I.; Salomoni, A.; Angot, A.; Trolese, M.; Ferrè, N.; Mutinelli, F.; Holmes, E.C.; Capua, I.; Lemey, P.; et al. The introduction of fox rabies into Italy (2008–2011) was due to two viral genetic groups with distinct phylogeographic patterns. Infect. Genet. Evol. 2013, 17, 202–209. [Google Scholar] [CrossRef]
- Delpietro, H.; Marchevsky, N.; Simonetti, E. Relative population densities and predation of the common vampire bat (Desmodus rotundus) in natural and cattle-raising areas in north-east Argentina. Prev. Vet. Med. 1992, 14, 13–20. [Google Scholar] [CrossRef]
- Delpietro, H.; Russo, R.; Carter, G.; Lord, R.; Delpietro, G. Reproductive seasonality, sex ratio and philopatry in Argentina’s common vampire bats. R. Soc. Open Sci. 2017, 4, 160959. [Google Scholar] [CrossRef]
- Dellicour, S.; Rose, R.; Pybus, O.G. Explaining the geographic spread of emerging epidemics: A framework for comparing viral phylogenies and environmental landscape data. BMC Bioinform. 2016, 17, 82. [Google Scholar] [CrossRef] [PubMed]
- Nylinder, S.; Lemey, P.; De Bruyn, M.; Suchard, M.A.; Pfeil, B.E.; Walsh, N.; Anderberg, A.A. On the biogeography of Centipeda: A species-tree diffusion approach. Syst. Biol. 2014, 63, 178–191. [Google Scholar] [CrossRef] [PubMed]
- Dellicour, S.; Lemey, P.; Artois, J.; Lam, T.T.; Fusaro, A.; Monne, I.; Cattoli, G.; Kuznetsov, D.; Xenarios, I.; Dauphin, G.; et al. Incorporating heterogeneous sampling probabilities in continuous phylogeographic inference—Application to H5N1 spread in the Mekong region. Bioinformatics 2019, 36, 2098–2104. [Google Scholar] [CrossRef] [PubMed]
- Scotch, M.; Tahsin, T.; Weissenbacher, D.; O’Connor, K.; Magge, A.; Vaiente, M.; Suchard, M.A.; Gonzalez-Hernandez, G. Incorporating sampling uncertainty in the geospatial assignment of taxa for virus phylogeography. Virus Evol. 2019, 5, vey043. [Google Scholar] [CrossRef]
- Lemey, P.; Hong, S.L.; Hill, V.; Baele, G.; Poletto, C.; Colizza, V.; O’Toole, Á.; McCrone, J.T.; Andersen, K.G.; Worobey, M.; et al. Accommodating individual travel history and unsampled diversity in Bayesian phylogeographic inference of SARS-CoV-2. Nat. Commun. 2020, 11, 5110. [Google Scholar] [CrossRef]
- Kalkauskas, A.; Perron, U.; Sun, Y.; Goldman, N.; Baele, G.; Guindon, S.; De Maio, N. Sampling bias and model choice in continuous phylogeography: Getting lost on a random walk. PLoS Comput. Biol. 2021, 17, e1008561. [Google Scholar] [CrossRef]
- Ayres, D.L.; Cummings, M.P.; Baele, G.; Darling, A.E.; Lewis, P.O.; Swofford, D.L.; Huelsenbeck, J.P.; Lemey, P.; Rambaut, A.; Suchard, M.A. BEAGLE 3: Improved Performance, Scaling, and Usability for a High-Performance Computing Library for Statistical Phylogenetics. Syst. Biol. 2019, 68, 1052–1061. [Google Scholar] [CrossRef]
- Geoghegan, J.L.; Holmes, E.C. Predicting virus emergence amid evolutionary noise. Open Biol. 2017, 7, 170189. [Google Scholar] [CrossRef] [PubMed]
- Grubaugh, N.D.; Ladner, J.T.; Lemey, P.; Pybus, O.G.; Rambaut, A.; Holmes, E.C.; Andersen, K.G. Tracking virus outbreaks in the twenty-first century. Nat. Microbiol. 2019, 4, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C.; Rambaut, A.; Andersen, K.G. Pandemics: Spend on surveillance, not prediction. Nature 2018, 558, 180–182. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Lam, T.T.; Carvalho, L.M.; Pybus, O.G. Exploring the temporal structure of heterochronous sequences using TempEst (formerly Path-O-Gen). Virus Evol. 2016, 2, vew007. [Google Scholar] [CrossRef]
- Duchene, S.; Lemey, P.; Stadler, T.; Ho, S.Y.; Duchene, D.A.; Dhanasekaran, V.; Baele, G. Bayesian evaluation of temporal signal in measurably evolving populations. Mol. Biol. Evol. 2020, 37, 3363–3379. [Google Scholar] [CrossRef]
- Baele, G.; Lemey, P.; Bedford, T.; Rambaut, A.; Suchard, M.A.; Alekseyenko, A.V. Improving the accuracy of demographic and molecular clock model comparison while accommodating phylogenetic uncertainty. Mol. Biol. Evol. 2012, 29, 2157–2167. [Google Scholar] [CrossRef]
- Baele, G.; Lemey, P.; Suchard, M.A. Genealogical working distributions for Bayesian model testing with phylogenetic uncertainty. Syst. Biol. 2016, 65, 250–264. [Google Scholar] [CrossRef]
- Duchene, S.; Featherstone, L.; Haritopoulou-Sinanidou, M.; Rambaut, A.; Lemey, P.; Baele, G. Temporal signal and the phylodynamic threshold of SARS-CoV-2. Virus Evol. 2020, 6, veaa061. [Google Scholar] [CrossRef]
- Sagulenko, P.; Puller, V.; Neher, R. TreeTime: Maximum-likelihood phylodynamic analysis. Virus Evol. 2018, 4, vex042. [Google Scholar] [CrossRef]
- Ishikawa, S.A.; Zhukova, A.; Iwasaki, W.; Gascuel, O. A Fast Likelihood Method to Reconstruct and Visualize Ancestral Scenarios. Mol. Biol. Evol. 2019, 36, 2069–2085. [Google Scholar] [CrossRef]
- Dellicour, S.; Durkin, K.; Hong, S.L.; Vanmechelen, B.; Martí-Carreras, J.; Gill, M.S.; Meex, C.; Bontems, S.; André, E.; Gilbert, M.; et al. A phylodynamic workflow to rapidly gain insights into the dispersal history and dynamics of SARS-CoV-2 lineages. Mol. Biol. Evol. 2021, 38, 1608–1613. [Google Scholar] [CrossRef] [PubMed]
- Notohara, M. The coalescent and the genealogical process in geographically structured population. J. Math. Biol. 1990, 29, 59–75. [Google Scholar] [CrossRef] [PubMed]
- Müller, N.F.; Rasmussen, D.A.; Stadler, T. The structured coalescent and its approximations. Mol. Biol. Evol. 2017, 34, 2970–2981. [Google Scholar] [CrossRef]
- Müller, N.F.; Dudas, G.; Stadler, T. Inferring time-dependent migration and coalescence patterns from genetic sequence and predictor data in structured populations. Virus Evol. 2019, 5, vez030. [Google Scholar] [CrossRef]
- Baele, G.; Dellicour, S.; Suchard, M.A.; Lemey, P.; Vrancken, B. Recent advances in computational phylodynamics. Curr. Opin. Virol. 2018, 31, 24–32. [Google Scholar] [CrossRef]
- Gascuel, O.; Steel, M. A Darwinian uncertainty principle. Syst. Biol. 2019, 69, 521–529. [Google Scholar] [CrossRef]
- Dellicour, S.; Baele, G.; Dudas, G.; Faria, N.R.; Pybus, O.G.; Suchard, M.A.; Rambaut, A.; Lemey, P. Phylodynamic assessment of intervention strategies for the West African Ebola virus outbreak. Nat. Commun. 2018, 9, 2222. [Google Scholar] [CrossRef]
- Worobey, M.; Pekar, J.; Larsen, B.B.; Nelson, M.I.; Hill, V.; Joy, J.B.; Rambaut, A.; Suchard, M.A.; Wertheim, J.O.; Lemey, P. The emergence of SARS-CoV-2 in Europe and North America. Science 2020, 370, 564–570. [Google Scholar] [CrossRef]
- Rasigade, J.P.; Barray, A.; Shapiro, J.T.; Coquisart, C.; Vigouroux, Y.; Bal, A.; Destras, G.; Vanhems, P.; Lina, B.; Josset, L.; et al. A viral perspective on worldwide non-pharmaceutical interventions against COVID-19. medRxiv 2021. pre-print. [Google Scholar] [CrossRef]
- Paradis, E. Assessing temporal variations in diversification rates from phylogenies: Estimation and hypothesis testing. Proc. R. Soc. Lond. Ser. Biol. Sci. 1997, 264, 1141–1147. [Google Scholar] [CrossRef]
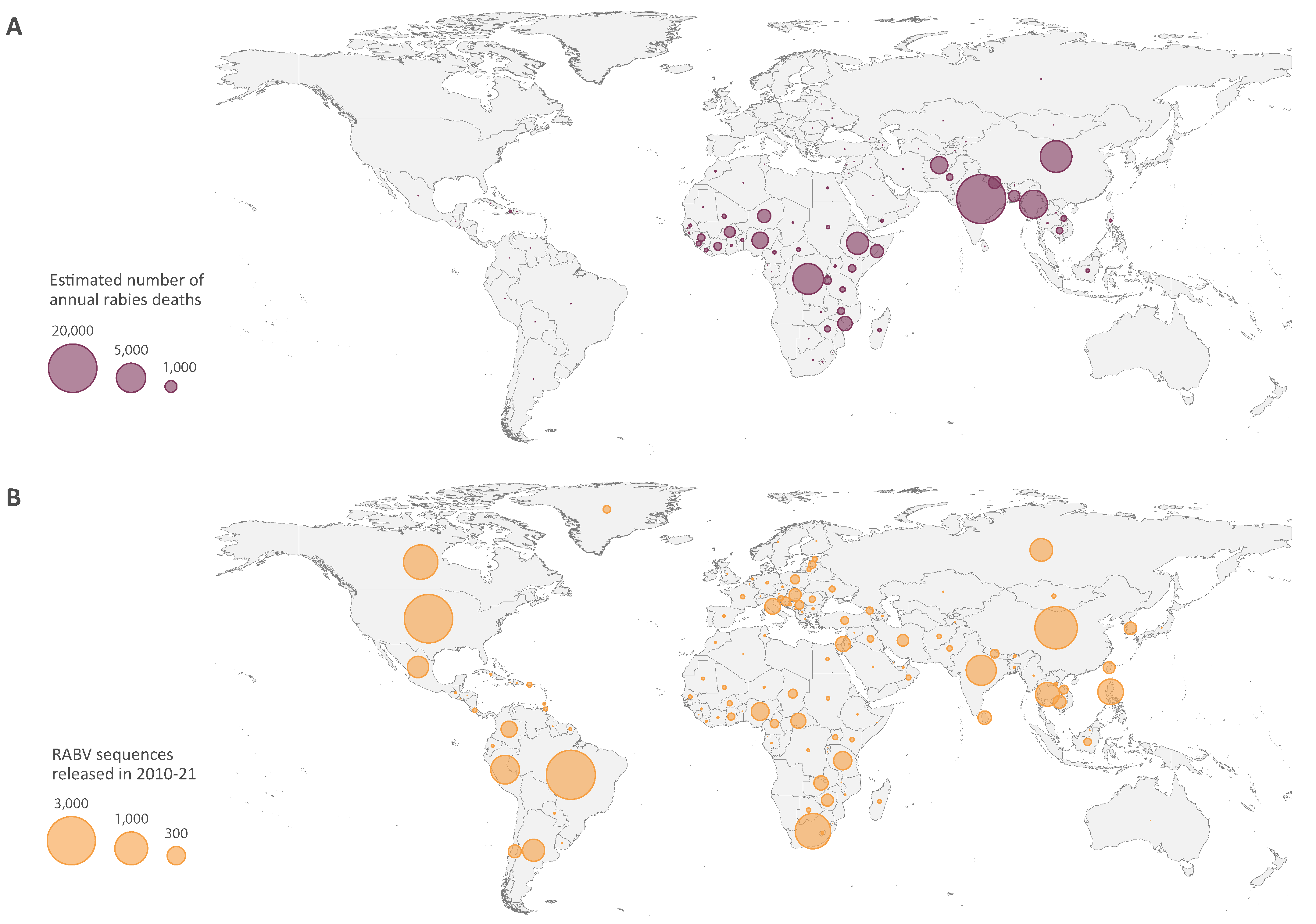
| Publication | Year | Sequences | Region | Species |
|---|---|---|---|---|
| Brunker et al. [41] | 2015 | 59 (whole) + 50 (partial) | Tanzania | dogs |
| Brunker et al. [6] | 2018 | 152 (whole) | Tanzania | dogs |
| Omodo et al. [23] | 2020 | 84 (partial) | Uganda | livestock, dogs, jackals and foxes |
| Hayman et al. [42] | 2021 | 139 + 88 (partial) | Ghana | dogs, cats |
| Talbi et al. [43] | 2010 | 287 (partial) | North Africa | dogs |
| Mollentze et al. [44] | 2013 | 636 (partial) | South Africa | dogs |
| Seetahal et al. [45] | 2013 | 183 (partial) | Trinidad | livestock (bovine, caprine, ovine, equine) |
| Trewby et al. [46] | 2017 | 289 (whole) | USA-Canada border | raccoon |
| Streicker et al. [47] | 2019 | 75 (partial) | Central America | vampire bats |
| Carnieli et al. [48] | 2011 | 71 (partial) | Brazil | dogs |
| Horton et al. [49] | 2015 | 139 (partial) | Middle East | domestic dogs, wildlife |
| Dellicour et al. [20] | 2019 | 109 (whole) | Iran | dogs, wolves, jackals, foxes |
| Tohma et al. [50] | 2014 | 233 (partial) | Philippines | dogs |
| Dibia et al. [51] | 2015 | 63 (partial) | Indonesia | dogs, cattle, goat, cat |
| Lin et al. [52] | 2016 | 220 (partial) | Taiwan | ferret badgers |
| Lan et al. [53] | 2017 | 156 (partial) | Taiwan | ferret badgers |
| Yu et al. [54] | 2012 | 110 + 90 (partial) | China | dogs, cats, deer, raccoon dogs, striped field mice, ferret badgers |
| Guo et al. [55] | 2013 | 232 (partial) | China | dogs |
| Ma et al. [37] | 2017 | 36 (partial) | Shaanxi (China) | dogs |
| Zhang et al. [56] | 2017 | 452 (partial) | Yunnan (China) | dogs, humans |
| Tian et al. [19] | 2018 | 1034 (partial) | Yunnan (China) | dogs |
| Wang et al. [57] | 2019 | 112 (partial) | China and neighbours | dogs |
| Yu et al. [58] | 2021 | 155 (partial) | North and South Asia | dogs, red fox, swift fox, raccoon dogs, cow, sheep camel |
| Publication | Year | Taxa (Whole Genomes) | Region | Species |
|---|---|---|---|---|
| Brunker et al. [6] | 2018 | 152 (whole) | Tanzania | dogs |
| Omodo et al. [23] | 2020 | 84 (partial) | Uganda | livestock, dogs, jackals and foxes |
| Kuzmina et al. [22] | 2013 | 241 (partial) | North America | skunks |
| Pepin et al. [67] | 2017 | 73 (partial) | Colorado (USA) | skunks |
| Musial et al. [68] | 2018 | 193 (partial) | Florida (US) | raccoons |
| Carnieli et al. [69] | 2013 | 53 (partial) | Brazil | dogs |
| Vieira et al. [70] | 2013 | 41 (partial) | Brazil | cattle, vampire bat |
| Torres et al. [2] | 2014 | 790 + 547 (partial) | Argentina | vampire bat |
| Streicker et al. [71] | 2016 | 264 (partial) | Peru | vampire bats |
| Streicker et al. [47] | 2019 | 75 + 40 (partial) | Costa Rica | vampire bats |
| Fusaro et al. [72] | 2013 | 160 (partial) | Italy and the Balkans | foxes |
| Tian et al. [19] | 2018 | 1034 (partial) | Yunnan (China) | dogs |
| Dellicour et al. [20] | 2019 | 109 (whole) | Iran | dogs, wolves, jackals, foxes |
| Dellicour et al. [5] (meta-analysis) | 2017 | [2,10,22,43,70] | North America, North Africa, Eastern Argentina, Eastern Brazil | skunk, raccoon, domestic dog, vampire bats |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nahata, K.D.; Bollen, N.; Gill, M.S.; Layan, M.; Bourhy, H.; Dellicour, S.; Baele, G. On the Use of Phylogeographic Inference to Infer the Dispersal History of Rabies Virus: A Review Study. Viruses 2021, 13, 1628. https://doi.org/10.3390/v13081628
Nahata KD, Bollen N, Gill MS, Layan M, Bourhy H, Dellicour S, Baele G. On the Use of Phylogeographic Inference to Infer the Dispersal History of Rabies Virus: A Review Study. Viruses. 2021; 13(8):1628. https://doi.org/10.3390/v13081628
Chicago/Turabian StyleNahata, Kanika D., Nena Bollen, Mandev S. Gill, Maylis Layan, Hervé Bourhy, Simon Dellicour, and Guy Baele. 2021. "On the Use of Phylogeographic Inference to Infer the Dispersal History of Rabies Virus: A Review Study" Viruses 13, no. 8: 1628. https://doi.org/10.3390/v13081628
APA StyleNahata, K. D., Bollen, N., Gill, M. S., Layan, M., Bourhy, H., Dellicour, S., & Baele, G. (2021). On the Use of Phylogeographic Inference to Infer the Dispersal History of Rabies Virus: A Review Study. Viruses, 13(8), 1628. https://doi.org/10.3390/v13081628






