Equine Parvovirus-Hepatitis Screening in Horses and Donkeys with Histopathologic Liver Abnormalities
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Histopathologic Evaluation
2.2. Detection of EqPV-H by Real-Time PCR
2.3. Quantification of Cellular Viral Load Using Digital PCR
3. Results
3.1. Clinical Records and Histopathologic Evaluation of Liver Samples
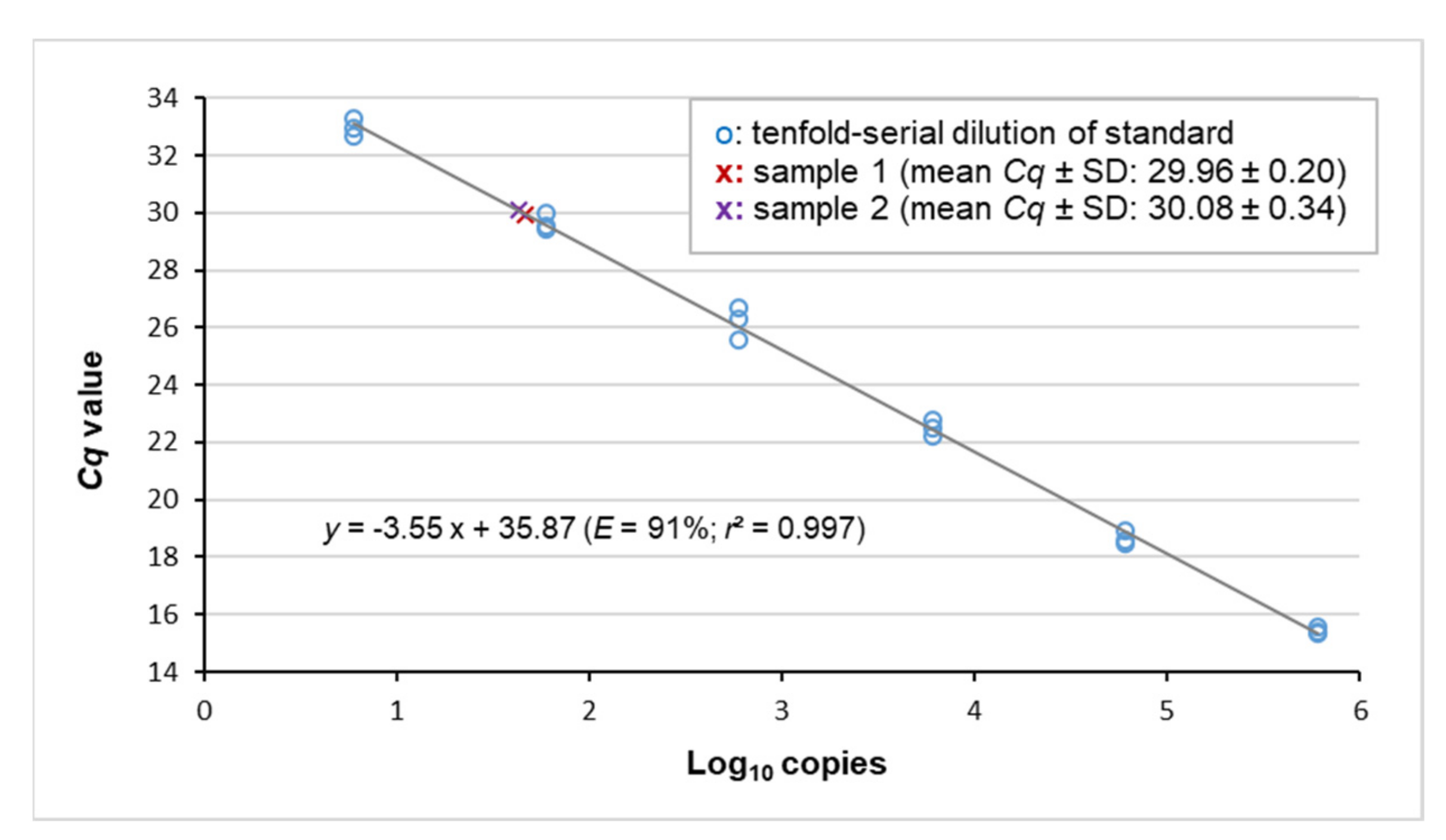
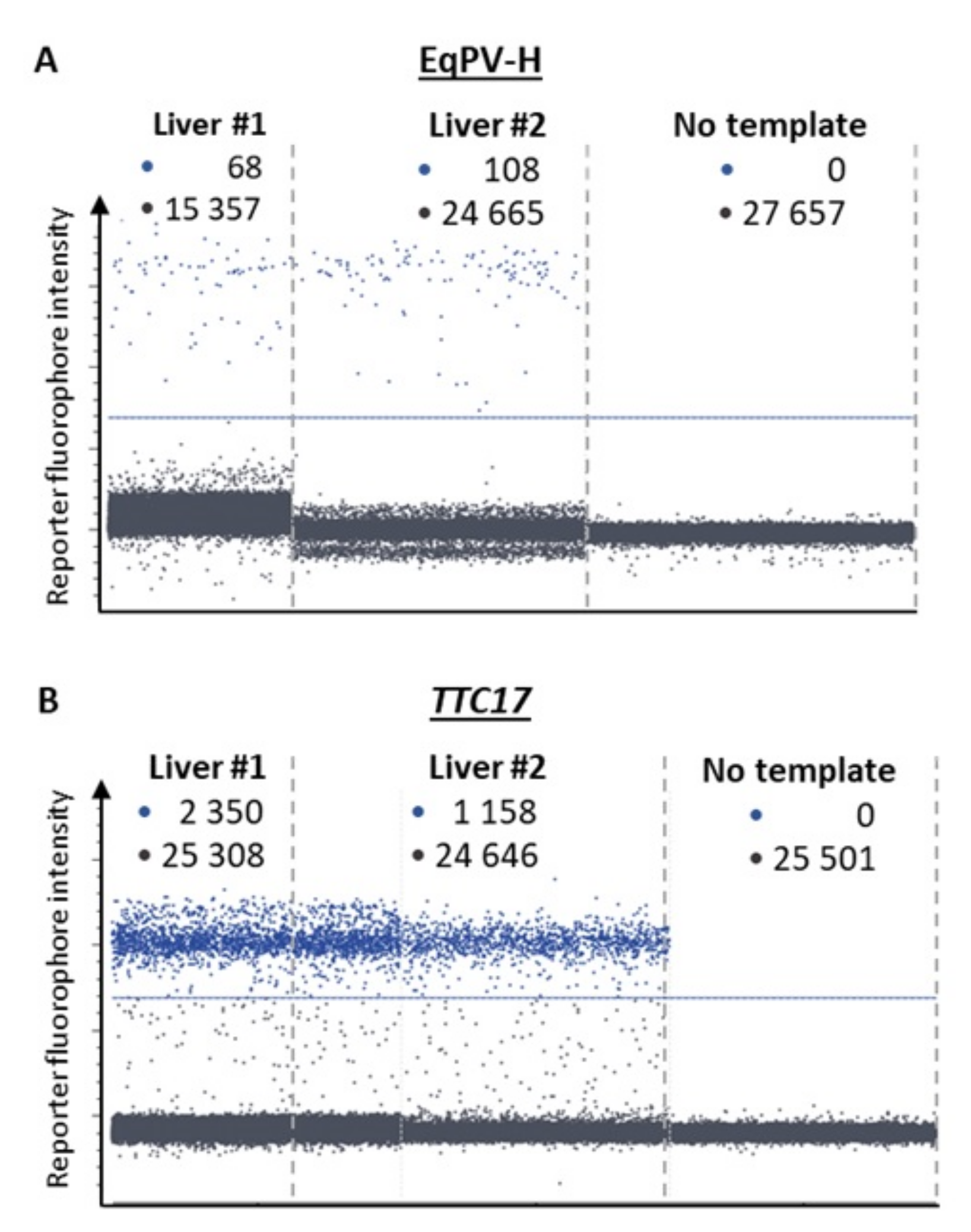
3.2. Virus Detection by Real-Time PCR and Copy Number Quantification by Digital PCR
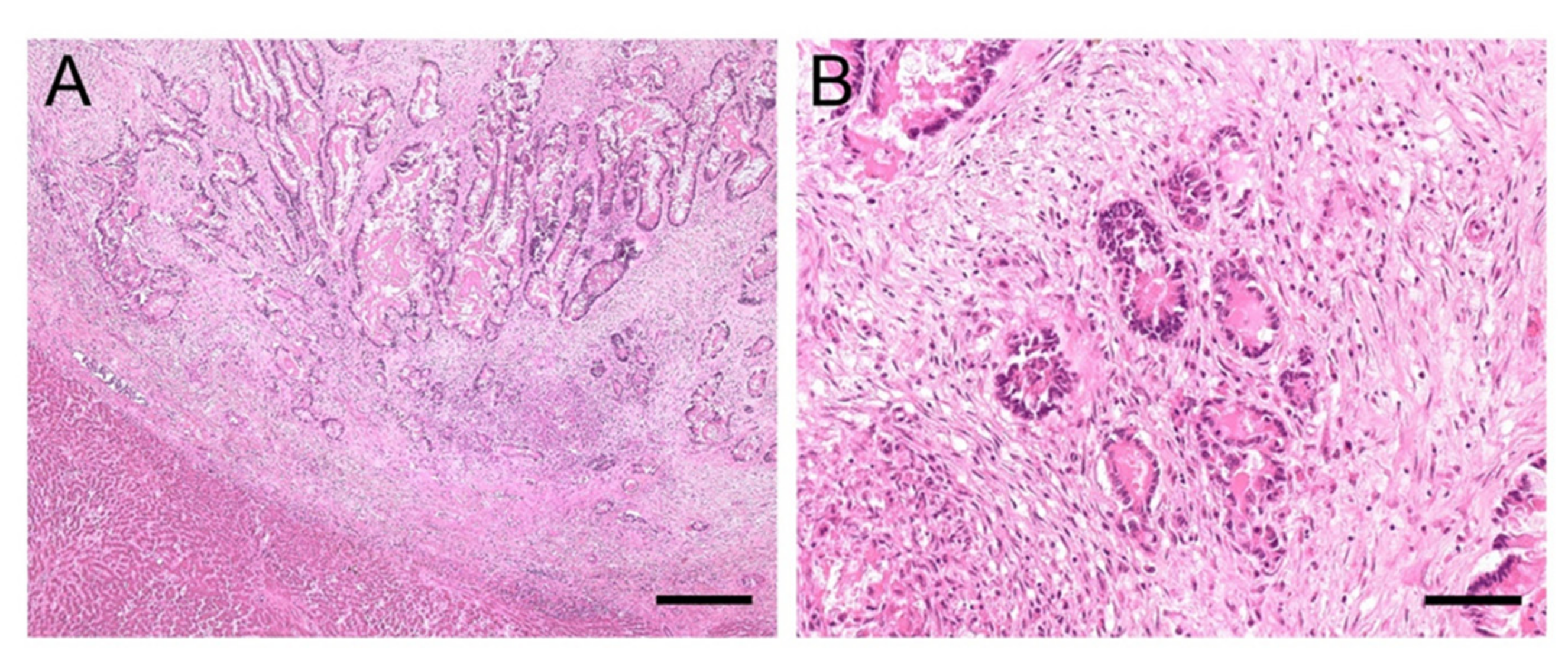
3.3. Clinical History and Pathological Findings of Horses with EqPV-H Positive Liver Tissue
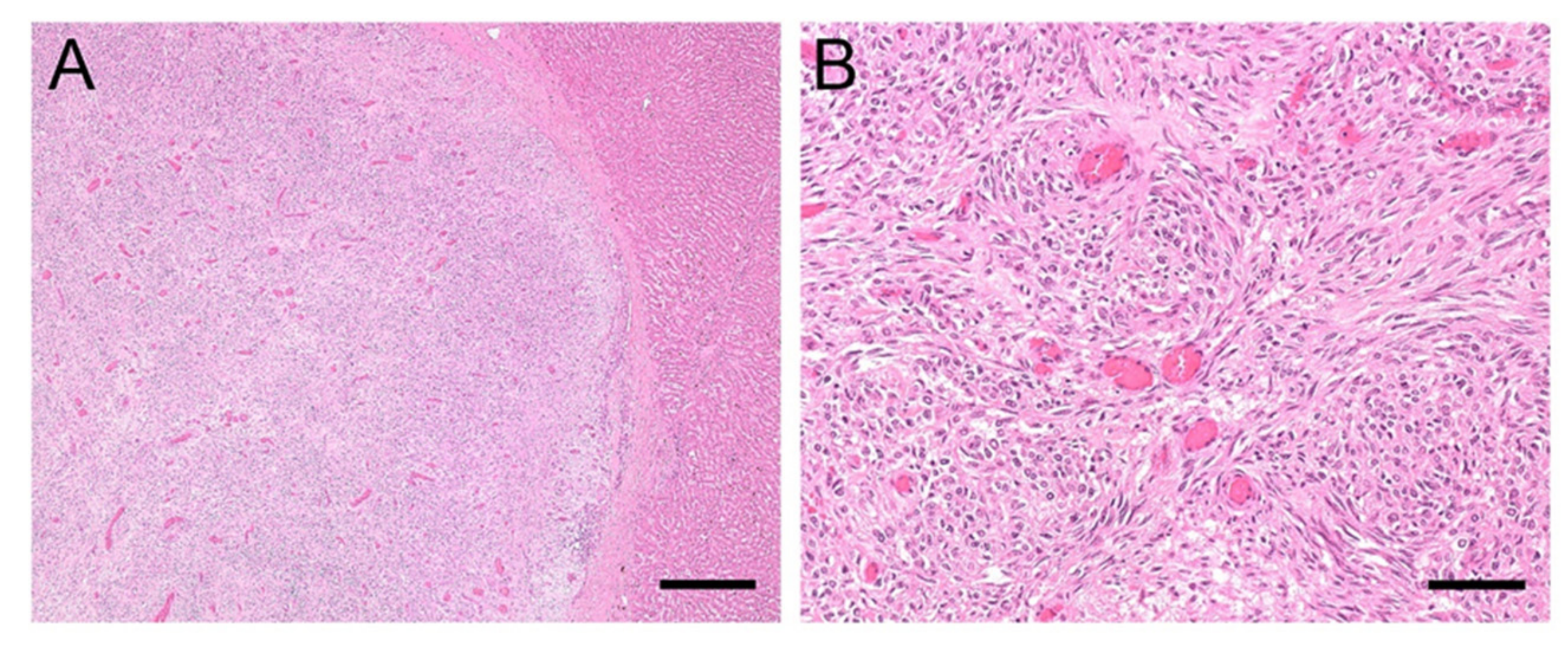
3.3.1. Case #1
3.3.2. Case #2
4. Discussion
5. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| S # | G 1 | G 2 | G 3 | G 4 | G 5 | G 6 | G 7 | Age | Sex | Breed | Clinical Diagnosis | Histology Liver | GG T< 30 U/L | AST < 550 U/L | BA < 20 µmol/L | GLDH < 13.00 U/L | AP < 250 U/L | BIL 0.7–3.1 mg/dL | NH3 < 40 µmol/L | ALB 2.4-4.5 g/dL | TG < 50.0 mg/dL | TP 5.5-7.5 g/dL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | x | 21 | M | Warmblood | Abdominal neoplasia | Adenocarcinoma | 13 | x | x | 3.07 | x | x | x | 1.41 | 28.0 | 6.89 | ||||||
| 2 | x | 19 | M | Warmblood | Liver neoplasia, cholelithiasis | Perivascular wall tumour | 132 | 619 | 6 | 20.08 | x | x | < LOD | 3.75 | x | 7.44 | ||||||
| 3 | x | 20 | M | Holsteiner | Colonic impaction, peritonitis, ventricular tachycardia | Adenocarcinoma biliary ducts | x | x | x | x | x | x | x | x | x | x | ||||||
| 4 | x | 20 | M | Haflinger | Renal failure, neoplasia suspected | Lymphoma | 27 | 556 | x | 8.06 | 171 | x | x | 2.90 | x | 8.09 | ||||||
| 5 | x | 16 | M | Warmblood | Colon displacement, malabsorption syndrome | Vascular hamartoma | 20 | x | x | 116.60 | 109 | x | x | 3.46 | x | 6.58 | ||||||
| 6 | x | 21 | F | Warmblood | Peritonitis | Carcinoma metastasis in liver | 18 | x | x | 70.99 | x | x | x | 3.14 | x | 7.56 | ||||||
| 7 | x | 25 | M | Warmblood | Hyperlipaemia, anaemia | Undifferentiated round cell neoplasia (myeloid leukosis suspected) | x | x | x | x | x | x | x | x | 709.0 | x | ||||||
| 8 | x | 3 | M | Thoroughbred | Liquidothorax (neoplasia suspected) | Multicentric lymphoma | x | x | x | x | x | x | x | x | x | x | ||||||
| 9 | x | 22 | F | Warmblood | Leukaemia | Lymphatic T-cell leukaemia | 30 | 245 | 5 | 1.45 | 651 | 0.90 | x | 2.69 | 134.0 | 5.23 | ||||||
| 10 | x | 22 | F | Haflinger | Haemothorax (neoplasia suspected) | Haemangiosarcoma metastasis in liver | x | x | x | x | x | x | x | x | x | x | ||||||
| 11 | x | 22 | M | Warmblood | Colon tympany, colon displacement | Adenocarcinoma metastasis in liver | x | x | x | x | x | x | x | x | x | x | ||||||
| 12 | x | 7 | F | Warmblood | Liquidothorax, ascites (neoplasia suspected) | Adenocarcinoma metastasis in liver | x | x | x | x | x | x | x | 2.64 | x | 6.07 | ||||||
| 13 | x | 25 | M | Fjord Horse | Spinal ataxia, kidney mass (neoplasia suspected) | Haemangiosarcoma metastasis in liver | x | x | x | x | x | x | x | x | x | 8.06 | ||||||
| 14 | x | 11 | F | Warmblood | Paraneoplastic syndrome | Lymphoma | 47 | x | 27 | 5.67 | x | x | x | 3.06 | x | 7.48 | ||||||
| 15 | x | 24 | F | Haflinger | Liver failure, immune mediated haemolytic anaemia and thrombocytopenia | Myeloid leukaemia | 741 | 889 | 162 | 107.43 | 1558 | 9.83 | 97 | 2.42 | 442.0 | 6.35 | ||||||
| 16 | x | 24 | M | Standardbred | Haemoperitoneum (lymphoma suspected) | Lymphoma | 31 | x | x | 2.09 | x | x | x | 2.54 | x | 6.90 | ||||||
| 17 | x | 26 | F | Icelandic Horse Mix | Hepatopathy, hepatoencephalopathy | Adenocarcinoma, liver infiltration | 332 | 449 | 33 | 77.03 | 1120 | x | 116 | 1.97 | 252.0 | 9.48 | ||||||
| 18 | x | 32 | F | Haflinger | Liquidothorax (neoplasia suspected) | Spindle cell carcinoma, liver metastasis | x | x | x | x | x | x | x | x | x | x | ||||||
| 19 | x | 25 | F | Warmblood | Haemoperitoneum | Lymphomaliver infiltration | x | x | x | x | x | x | x | 2.00 | x | x | ||||||
| 20 | x | 24 | F | Pony | Thoracic/ abdominal neoplasia | Haemangiosarcoma metastasis in liver | x | x | x | x | x | x | x | 2.27 | x | x | ||||||
| 21 | x | 21 | M | Warmblood | Ventricular tachycardia | Chronic eosinophilic granulomatous hepatitis | 20 | x | x | 9.01 | 213 | 2.26 | x | 2.18 | 466.0 | 6.82 | ||||||
| 22 | x | 6 | M | Welsh Pony | Ileus | Acute purulent cholangitis and necrotising hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 23 | x | 5 | M | Noriker | Retropharyngeal mass | Acute predominantly non purulent hepatitis | 17 | x | x | 1.29 | x | x | x | 2.94 | 31.0 | 7.58 | ||||||
| 24 | x | 18 | F | Haflinger | Inflammatory bowel disease, protein losing enteropathy | Chronic purulent hepatitis | 93 | x | x | 6.59 | x | x | x | 1.65 | 104.0 | 6.49 | ||||||
| 25 | x | 2 | M | Shetland Pony | Shock, tachycardia (gastric rupture suspected) | Acute purulent necrotising hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 26 | x | 1 | F | Morgan Horse | Hepatopathy | Acute purulent necrotising hepatitis | x | 5036 | x | 271.72 | x | x | 47 | 2.74 | 59.0 | 4.81 | ||||||
| 27 | x | 15 | F | Shetland Pony | Liver neoplasia, hepatoencephalopathy | Chronic purulent abscessing hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 28 | x | 1 | M | Warmblood | Septic peritonitis | Acute non purulent hepatitis, cholangitis | x | x | x | x | x | x | x | x | x | 9.10 | ||||||
| 29 | x | 1 m | M | Warmblood | Pneumonia | Acute purulent necrotising hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 30 | x | 10 | M | Standardbred | Morbus maculosus | Acute necrotising lymphoproliferative hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 31 | x | 13 | M | Shire Horse | Typhlocolitis, hepatopathy | Acute (partially) purulent hepatitis | 54 | x | 37 | 37.22 | x | 6.33 | 7 | 3.12 | x | 8.33 | ||||||
| 32 | x | 19 | F | Donkey | Laminitis, bronchitis, hepatopathy, hyperlipaemia | Chronic granulomatous hepatitis, interstitial fibrosis | 73 | 564 | 6 | 19.87 | x | x | 7 | 2.33 | 284.0 | 7.01 | ||||||
| 33 | x | 13 | M | Warmblood | Endocarditis, purulent meningitis | Acute (partially) purulent hepatitis | 21 | 275 | 4 | 1.17 | 196 | 2.96 | x | 2.90 | x | 7.49 | ||||||
| 34 | x | 23 | M | Icelandic Horse | Adhesions between jejunum and mesenterium, peritonitis, sepsis | Acute necrotising hepatitis | 42 | x | x | 67.18 | x | x | x | 2.18 | 646.0 | 4.40 | ||||||
| 35 | x | 8 | F | Shetland Pony | Ataxia, dysphagia | Acute necrotising hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 36 | x | 20 | M | Arabian | Neoplasia (equine multinodular pulmonary fibrosis suspected) | Acute non purulent hepatitis | 25 | x | x | 2.41 | x | x | x | 2.41 | 62.0 | 8.17 | ||||||
| 37 | x | 2 | F | Icelandic Horse | Intoxication | Acute purulent necrotising hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 38 | x | 16 | M | Warmblood | Gastric rupture | Acute purulent serositis | x | x | x | x | x | x | x | x | x | x | ||||||
| 39 | x | 9 | F | Shetland Pony | Right dorsal colitis | Acute non purulent hepatitis | 33 | x | x | 74.59 | x | x | x | 1.07 | 148.0 | 2.96 | ||||||
| 40 | x | 4 | F | Pony | Renal failure | Acute non purulent hepatitis | x | x | x | x | x | x | x | x | 72.0 | x | ||||||
| 41 | x | 22 | F | Welsh Pony | Choke, aspiration pneumonia | Acute non purulent hepatitis | x | x | x | x | x | x | x | 1.66 | 370.0 | x | ||||||
| 42 | x | 25 | M | Warmblood | Anaemia, thrombocytopenia, immune mediated vasculitis | Acute eosinophilic hepatitis | 20 | x | 4 | 18.35 | x | x | x | 2.55 | x | 8.63 | ||||||
| 43 | x | 20 | M | Icelandic Horse | Masses neck and sinuses | Chronic eosinophilic granulomatous hepatitis | x | x | x | x | x | x | x | x | x | 7.09 | ||||||
| 44 | x | 16 | M | Noriker | Gastric rupture, peritonitis | Acute purulent serositis | x | x | x | x | x | x | x | x | x | x | ||||||
| 45 | x | 13 | F | Warmblood | Hepatoencephalopathy | Cirrhosis | 148 | 4650 | x | 35.44 | 665 | x | x | 2.33 | 31.0 | 7.57 | ||||||
| 46 | x | 7 | F | Haflinger | Hepatoencephalopathy | Cirrhosis | 1309 | x | x | 83.71 | x | x | x | x | x | 9.25 | ||||||
| 47 | x | 7 | F | Warmblood | Left dorsal displacement, colonic impaction, hepatopathy | Cirrhosis | 139 | x | x | 532.30 | x | x | x | 3.59 | 48.0 | 8.11 | ||||||
| 48 | x | 30 | F | Pony | Laminitis, right dorsal colitis, hepatopathy, PPID | Biliary cirrhosis | 163 | x | 18 | 63.36 | x | x | 16 | 2.32 | 420.0 | 5.50 | ||||||
| 49 | x | 20 | F | Arabian | Hepatoencephalopathy | Cirrhosis | x | x | x | x | x | x | x | x | x | x | ||||||
| 50 | x | 17 | M | Warmblood | Cardiogenic shock, atrial fibrillation, ventricular tachycardia | Portal and central vein fibrosis, subacute liver congestion | x | x | x | x | x | x | x | x | x | x | ||||||
| 51 | x | 20 | F | Shetland Pony | Colon impaction, liver rupture | Liver haemorrhage | 86 | x | 15 | 76.07 | 877 | 1.63 | x | 3.03 | 1308.0 | 7.50 | ||||||
| 52 | x | 12 | F | Warmblood | Acute renal failure, septic pericarditis, right heart failure | Subacute liver congestion | 21 | x | x | 7.64 | x | x | x | 1.79 | x | 6.56 | ||||||
| 53 | x | 2 m | M | Haflinger | Cardiac cushion defect, atrial septum defect, hepatomegaly | Subacute liver congestion | x | x | x | x | x | x | x | x | x | x | ||||||
| 54 | x | 14 | M | Standardbred | Gastric neoplasia, multiple cardiac valve insufficiencies | Amyloidosis | 24 | x | x | 1.19 | 160 | 0.71 | x | 2.43 | 12.0 | 7.12 | ||||||
| 55 | x | 4 | F | Miniature Donkey | Hyperlipaemia | Hepatic lipidosis | 49 | x | x | 22.40 | x | x | < LOD | 1.80 | 1748.0 | x | ||||||
| 56 | x | 12 | F | Icelandic Horse | Enterocolitis, hepatoencephalopathy | Hepatic lipidosis | 96 | x | x | 95.30 | x | x | 152 | 2.14 | x | 5.38 | ||||||
| 57 | x | 10 | M | Shetland Pony | Shock, recumbency | Hepatic lipidosis | x | x | x | x | x | x | x | x | x | x | ||||||
| 58 | x | 12 | M | Pony | Enteral hyperammonaemia, encephalopathy | Hepatic lipidosis | 17 | x | x | 1.83 | x | x | 194 | 2.90 | x | 5.32 | ||||||
| 59 | x | 14 | M | Noriker | Weakness, recumbency | Severe siderosis | 11 | 1130 | x | 7.06 | x | x | x | 2.82 | x | 5.49 | ||||||
| 60 | x | 15 | F | Icelandic Horse | Hyperlipaemia | Hepatic lipidosis | 3319 | 3331 | 300 | 590.72 | x | 2.60 | 46 | 3.78 | 2410.0 | 7.46 | ||||||
| 61 | x | 12 | F | Icelandic Horse | Hyperlipaemia | Hepatic lipidosis | x | x | x | x | x | x | x | x | 4725.0 | x | ||||||
| 62 | x | 16 | F | Icelandic Horse | Quadriplegia | Hepatic lipidosis | x | x | x | x | x | x | x | x | x | x | ||||||
| 63 | x | 24 | M | Donkey | Paralytic ileus, renal failure, hyperlipaemia, encephalopathy | Hepatic lipidosis, chronic granulomatous hepatitis | x | x | x | x | x | x | x | x | x | x | ||||||
| 64 | x | 10 | F | Friesian | Paralytic ileus, endotoxaemia, acute renal failure, hyperammonaemia | Hepatic lipidosis | 17 | x | x | 10.15 | x | x | 217 | 1.67 | 1311.0 | 4.67 | ||||||
| 65 | x | 31 | M | Warmblood | Colon impaction, hypertriglyceridaemia | Liver sclerosis, hepatic lipidosis | x | x | x | x | x | x | x | 2.96 | 481.0 | 6.79 | ||||||
| 66 | x | 15 | M | Icelandic Horse | Hyperlipaemia | Hepatic lipidosis | 73 | x | 54 | 117.18 | x | 1.01 | 12 | 2.69 | 2588.0 | 6.69 | ||||||
| 67 | x | 14 | M | Donkey | Hyperlipaemia | Hepatic lipidosis | 251 | x | 10 | 11.86 | x | x | 7 | 3.28 | 7600.0 | 5.33 | ||||||
| 68 | o | o | x | 20 | M | Warmblood | Colitis, malabsorption syndrome | Amyloidosis, acute purulent hepatitis and pericholangitis | 42 | x | x | 2.83 | x | x | x | 0.78 | 331.0 | 4.53 | ||||
| 69 | o | o | x | 5 | M | Friesian | Ventricular tachycardia, myocarditis | Subacute liver congestion, centrolobular liver necrosis | 43 | 621 | x | 67.74 | x | x | x | 2.69 | 59.0 | 5.64 | ||||
| 70 | o | o | x | 7 | M | Friesian | Colitis, chronic kidney failure, ventricular tachycardia | Hepatic lipidosis and liver necrosis, acute non purulent hepatitis | 34 | x | x | 42.36 | x | x | x | 2.55 | x | 4.68 | ||||
| 71 | o | o | x | 2 | M | Quarter Horse | Pleural effusion, ascites (neoplasia suspected) | Subacute to chronic liver congestion, acute non purulent hepatitis | 12 | 364 | x | 3.21 | x | 1.90 | x | 1.67 | 37.0 | 5.89 | ||||
| 72 | o | o | x | 23 | F | Shetland Pony | Hyperlipaemia | Hepatic lipidosis, acute non purulent hepatitis | x | x | x | x | x | x | x | x | 801.0 | x | ||||
| 73 | o | o | x | 13 | F | Donkey | Hyperlipaemia | Hepatic lipidosis, acute purulent hepatitis | 97 | x | 14 | 27.49 | 476 | 0.40 | x | 2.80 | 1931.0 | 6.45 | ||||
| 74 | o | o | x | 17 | F | Haflinger | Protein losing enteropathy, inflammatory bowel disease | Cirrhosis, chronic purulent cholangitis and pericholangitis | 37 | 351 | 3 | 34.32 | x | x | x | 1.21 | x | 4.48 | ||||
| 75 | o | o | x | 20 | M | Thoroughbred | Delayed gastric emptying, hepatopathy (neoplasia suspected) | Squamous cell carcinoma, subacute to chronic liver congestion | 175 | 509 | 12 | 348.40 | 627 | 3.80 | x | 2.84 | 262.0 | 6.71 | ||||
| 76 | o | o | o | x | 31 | M | Warmblood | Paraneoplastic syndrome | Undifferentiated round cell neoplasia, subacute liver congestion, acute non purulent hepatitis | 32 | 351 | x | 17.54 | 244 | x | x | 2.10 | x | 7.35 | |||
| 77 | o | o | x | 24 | F | Warmblood | Ileus, colon displacement | Subacute liver congestion, focal acute non purulent hepatitis | x | x | x | x | x | x | x | x | x | x | ||||
| 78 | o | o | o | x | 13 | M | Pony | Ascites (neoplasia suspected) | Adenocarcinoma metastasis, acute non purulent hepatitis | x | x | x | x | x | x | x | x | x | 7.73 | |||
| 79 | o | o | x | 6 | F | Quarter Horse | Epiploic foramen entrapment, ileus, hyperlipaemia | Hepatic lipidosis, acute non purulent hepatitis | 9 | x | x | 18.27 | x | x | x | 2.15 | 548.0 | 3.78 | ||||
| 80 | o | o | o | x | 16 | F | Fjord Horse | Gastric distension, hyperammonaemia, hepatopathy | Carcinoma of biliary ducts, hepatic lipidosis, cirrhosis | 55 | x | 29 | 13.64 | x | x | 140 | 3.11 | 153.0 | 7.67 | |||
| 81 | o | o | x | 15 | F | Warmblood | Hepatopathy, hyperlipaemia, PPID | Cirrhosis, acute purulent hepatitis | 881 | 1265 | 135 | 217.11 | 2488 | 3.54 | 10 | 2.69 | 413.0 | 8.93 | ||||
| 82 | o | o | x | 12 | M | Warmblood | Multicentric lymphoma | Multicentric lymphoma - liver infiltration, chronic lymphoplasmocytic to eosinophilic hepatitis | 13 | x | x | 0.98 | x | x | x | 1.91 | x | 8.99 | ||||
| 83 | o | o | x | 4 | F | Warmblood | Equine grass sickness suspected | Acute (partially) purulent hepatitis and chronic pericholangitis | x | x | x | x | x | x | x | x | x | x | ||||
| 84 | o | o | x | 3 | F | Arabian | Aspiration pneumonia, liquidothorax | Hepatic lipidosis | x | x | x | x | x | x | x | x | x | x | ||||
| 85 | x | 8 | M | Warmblood | Encephalitis | Unremarkable | 8 | x | 60 | 1.93 | x | x | < LOD | 2.98 | 41.0 | 7.79 | ||||||
| 86 | x | 14 | M | Pura Raza Menorquina | Typhlocolitis, liquidothorax | Unremarkable | 21 | x | x | 6.45 | 157 | x | 103 | 2.00 | 149.0 | 5.23 | ||||||
| 87 | x | 4 | M | Warmblood | Chronic renal failure | Unremarkable | x | x | x | x | x | x | x | x | x | x | ||||||
| 88 | x | 2 | F | Warmblood | Mediastinal mass (lymphoma suspected) | Unremarkable | x | x | x | x | x | x | x | x | x | x | ||||||
| 89 | x | 12 | M | Icelandic Horse | Renal failure | Unremarkable | 15 | x | x | 44.40 | x | x | x | 2.55 | 255.0 | 5.49 | ||||||
| 90 | x | 6 | M | Arabian- Paint | Peritonitis | Unremarkable | x | x | x | x | x | x | x | x | x | x | ||||||
| 91 | x | 15 | M | Thoroughbred | Haemoperitoneum | Unremarkable | 4 | x | 1 | 5.07 | x | x | < LOD | 1.90 | x | 2.94 | ||||||
| 92 | x | 20 | F | Trakehner | Gastritis, colon impaction, PPID | Unremarkable | 26 | 230 | x | 11.21 | 334 | 5.63 | < LOD | 2.77 | 48.0 | 7.01 | ||||||
| T | 20 | 24 | 5 | 4 | 14 | 17 | 8 |
References
- Theiler, A. Acute Liver-Atrophy and Parenchymatous Hepatitis in Horses; Union of South Africa. Dept. of Agriculture. 5th and 6th Repts. of the Director of Veterinary Research, 1918; pp. 7–165. [Google Scholar]
- Thomsett, L.R. Acute Hepatic Failure in the Horse. Equine Veter. J. 1971, 3, 15–19. [Google Scholar] [CrossRef]
- Guglick, M.A.; MacAllister, C.G.; Ely, R.W.; Edwards, W.C. Hepatic disease associated with administration of tetanus antitoxin in eight horses. J. Am. Veter. Med. Assoc. 1995, 206, 1737–1740. [Google Scholar]
- Messer, N.T., 4th; Johnson, P.J. Idiopathic acute hepatic disease in horses: 12 cases (1982–1992). J. Am. Veter. Med. Assoc. 1994, 204, 1934–1937. [Google Scholar] [PubMed]
- Aleman, M.; Nieto, J.E.; Carr, E.A.; Carlson, G.P. Serum hepatitis associated with commercial plasma transfusion in horses. J. Veter. Intern. Med. 2005, 19, 120–122. [Google Scholar] [CrossRef]
- Chandriani, S.; Skewes-Cox, P.; Zhong, W.; Ganem, D.E.; Divers, T.J.; Van Blaricum, A.J.; Tennant, B.C.; Kistler, A.L. Identification of a previously undescribed divergent virus from the Flaviviridae family in an outbreak of equine serum hepatitis. Proc. Natl. Acad. Sci. USA 2013, 110, E1407–E1415. [Google Scholar] [CrossRef] [Green Version]
- Kopper, J.J.; Schott, H.C.; Divers, T.J.; Mullaney, T.; Huang, L.; Noland, E.; Smedley, R. Theiler’s disease associated with administration of tetanus antitoxin contaminated with nonprimate (equine) hepacivirus and equine parvovirus-hepatitis virus. Equine Veter. Educ. 2018, 32. [Google Scholar] [CrossRef]
- Divers, T.J.; Tennant, B.C.; Kumar, A.; McDonough, S.; Cullen, J.; Bhuva, N.; Jain, K.; Chauhan, L.S.; Scheel, T.; Lipkin, W.I.; et al. New Parvovirus Associated with Serum Hepatitis in Horses after Inoculation of Common Biological Product. Emerg. Infect. Dis. 2018, 24, 303–310. [Google Scholar] [CrossRef]
- Tomlinson, J.E.; Kapoor, A.; Kumar, A.; Tennant, B.C.; Laverack, M.A.; Beard, L.; Delph, K.; Davis, E.; Ii, H.S.; Lascola, K.; et al. Viral testing of 18 consecutive cases of equine serum hepatitis: A prospective study (2014–2018). J. Veter. Intern. Med. 2018, 33, 251–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomlinson, J.E.; Tennant, B.C.; Struzyna, A.; Mrad, D.; Browne, N.; Whelchel, D.; Johnson, P.J.; Jamieson, C.; Löhr, C.V.; Bildfell, R.; et al. Viral testing of 10 cases of Theiler’s disease and 37 in-contact horses in the absence of equine biologic product administration: A prospective study (2014–2018). J. Veter. Intern. Med. 2018, 33, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Ramsauer, A.S.; Badenhorst, M.; Cavalleri, J.V. Equine parvovirus hepatitis. Equine Veter. J. 2021. [Google Scholar] [CrossRef]
- Baird, J.; Tegtmeyer, B.; Arroyo, L.; Stang, A.; Brüggemann, Y.; Hazlett, M.; Steinmann, E. The association of Equine Parvovirus-Hepatitis (EqPV-H) with cases of non-biologic-associated Theiler’s disease on a farm in Ontario, Canada. Veter. Microbiol. 2020, 242, 108575. [Google Scholar] [CrossRef]
- Vengust, M.; Jager, M.C.; Zalig, V.; Cociancich, V.; Laverack, M.; Renshaw, R.W.; Dubovi, E.; Tomlinson, J.E.; Van de Walle, G.R.; Divers, T.J. First report of equine parvovirus-hepatitis-associated Theiler’s disease in Europe. Equine Veter. J. 2020, 52, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Sun, L.; Ou, J.; Xu, H.; Wu, L.; Li, S. Identification and genetic characterization of a novel parvovirus associated with serum hepatitis in horses in China. Emerg. Microbes Infect. 2018, 7, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badenhorst, M.; de Heus, P.; Auer, A.; Tegtmeyer, B.; Stang, A.; Dimmel, K.; Tichy, A.; Kubacki, J.; Bachofen, C.; Steinmann, E.; et al. Active equine parvovirus-hepatitis infection is most frequently detected in Austrian horses of advanced age. Equine Veter. J. 2021. [Google Scholar] [CrossRef]
- Altan, E.; Li, Y.; Jr, G.S.-S.; Sawaswong, V.; Barnum, S.; Pusterla, N.; Deng, X.; Delwart, E. Viruses in Horses with Neurologic and Respiratory Diseases. Viruses 2019, 11, 942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meister, T.L.; Tegtmeyer, B.; Brüggemann, Y.; Sieme, H.; Feige, K.; Todt, D.; Stang, A.; Cavalleri, J.-M.; Steinmann, E. Characterization of Equine Parvovirus in Thoroughbred Breeding Horses from Germany. Viruses 2019, 11, 965. [Google Scholar] [CrossRef] [Green Version]
- Lu, G.; Wu, L.; Ou, J.; Li, S. Equine Parvovirus-Hepatitis in China: Characterization of Its Genetic Diversity and Evidence for Natural Recombination Events Between the Chinese and American Strains. Front. Veter. Sci. 2020, 7. [Google Scholar] [CrossRef]
- Tomlinson, J.; Jager, M.; Struzyna, A.; Laverack, M.; Fortier, L.A.; Dubovi, E.; Foil, L.D.; Burbelo, P.D.; Divers, T.J.; Van De Walle, G.R. Tropism, pathology, and transmission of equine parvovirus-hepatitis. Emerg. Microbes Infect. 2020, 9, 651–663. [Google Scholar] [CrossRef] [Green Version]
- Zdobnov, E.M.; Tegenfeldt, F.; Kuznetsov, D.; Waterhouse, R.M.; Simão, F.A.; Ioannidis, P.; Seppey, M.; Loetscher, A.; Kriventseva, E.V. OrthoDB v9.1: Cataloging evolutionary and functional annotations for animal, fungal, plant, archaeal, bacterial and viral orthologs. Nucleic Acids Res. 2016, 45, D744–D749. [Google Scholar] [CrossRef]
- Lanini, S.; Ustianowski, A.; Pisapia, R.; Zumla, P.S.A.; Ippolito, G. Viral Hepatitis. Infect. Dis. Clin. N. Am. 2019, 33, 1045–1062. [Google Scholar] [CrossRef]
- Kwong, S.; Meyerson, C.; Zheng, W.; Kassardjian, A.; Stanzione, N.; Zhang, K.; Wang, H.L. Acute hepatitis and acute liver failure: Pathologic diagnosis and differential diagnosis. Semin. Diagn. Pathol. 2019, 36, 404–414. [Google Scholar] [CrossRef]
- Klastersky, J.; Aoun, M. Opportunistic infections in patients with cancer. Ann. Oncol. 2004, 15, iv329–iv335. [Google Scholar] [CrossRef] [PubMed]
- Allen, B.M.; Hiam, K.J.; Burnett, C.E.; Venida, A.; Debarge, R.; Tenvooren, I.; Marquez, D.M.; Cho, N.W.; Carmi, Y.; Spitzer, M.H. Systemic dysfunction and plasticity of the immune macroenvironment in cancer models. Nat. Med. 2020, 26, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Ringelhan, M.; McKeating, J.; Protzer, U. Viral hepatitis and liver cancer. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160274. [Google Scholar] [CrossRef] [Green Version]
- Corcioli, F.; Zakrzewska, K.; Rinieri, A.; Fanci, R.; Innocenti, M.; Civinini, R.; de Giorgi, V.; Di Lollo, S.; Azzi, A. Tissue persistence of parvovirus B19 genotypes in asymptomatic persons. J. Med. Virol. 2008, 80, 2005–2011. [Google Scholar] [CrossRef]
- Söderlund-Venermo, M.; Hokynar, K.; Nieminen, J.; Rautakorpi, H.; Hedman, K. Persistence of human parvovirus B19 in human tissues. Pathol. Biol. 2002, 50, 307–316. [Google Scholar] [CrossRef]
- Norja, P.; Hokynar, K.; Aaltonen, L.-M.; Chen, R.; Ranki, A.; Partio, E.K.; Kiviluoto, O.; Davidkin, I.; Leivo, T.; Eis-Hübinger, A.M.; et al. Bioportfolio: Lifelong persistence of variant and prototypic erythrovirus DNA genomes in human tissue. Proc. Natl. Acad. Sci. USA 2006, 103, 7450–7453. [Google Scholar] [CrossRef] [Green Version]
| Assay | GenBank Identity | 5′ to 3′ Sequence of Oligonucleotide | Amplicon Size (bp) |
|---|---|---|---|
| EqPV-H * | NC_040652.1, MG136722.1, MH500787.1 to MH500792.1, MN218583.1 to MN218592.1, MW256660.1 to MW256663.1 | F: AAG ATA TGC CGC CAT TTG AA | 77 |
| R: CTG AAA AGG CAT TCC GTC AG | |||
| P: FAM-CAG AGA AAT /ZEN/ CCT GAG CGG TGG CCT-IBFQ | |||
| PC: ATC TTC TAT AAA GAT ATG CCG CCA TTT GAA AAG GCC ACC GCT CAG GAT TTC TCT GAC TAT TAT GTT TCT GAC GGA ATG CCT TTT CAG ACT TTG TAT G | |||
| TTC17 | XM_023653901.1 | F: CTG GAC AAC AGC CAT GAC AAA | 147 |
| R: AAG TCT AAG GGC ATC TGA GTC CC | |||
| P: FAM-CAC AGG GTC /ZEN/ CTC CTC TGC TCC TGTC-IBFQ |
| Sample | TTC17 Copies/μL (1:20 Dilution) | EqPV-H Copies/μL | Cellular Viral Load | |
|---|---|---|---|---|
| Virus/Cells | GE/106 Cells | |||
| #1 | 151.5 ± 6.1 | 7.54 ± 1.81 | 1/201 | 5.0 × 103 |
| #2 | 78.4 ± 4.7 | 7.46 ± 1.42 | 1/105 | 9.5 × 103 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zehetner, V.; Cavalleri, J.-M.V.; Klang, A.; Hofer, M.; Preining, I.; Steinborn, R.; Ramsauer, A.S. Equine Parvovirus-Hepatitis Screening in Horses and Donkeys with Histopathologic Liver Abnormalities. Viruses 2021, 13, 1599. https://doi.org/10.3390/v13081599
Zehetner V, Cavalleri J-MV, Klang A, Hofer M, Preining I, Steinborn R, Ramsauer AS. Equine Parvovirus-Hepatitis Screening in Horses and Donkeys with Histopathologic Liver Abnormalities. Viruses. 2021; 13(8):1599. https://doi.org/10.3390/v13081599
Chicago/Turabian StyleZehetner, Verena, Jessika-M. V. Cavalleri, Andrea Klang, Martin Hofer, Irina Preining, Ralf Steinborn, and Anna S. Ramsauer. 2021. "Equine Parvovirus-Hepatitis Screening in Horses and Donkeys with Histopathologic Liver Abnormalities" Viruses 13, no. 8: 1599. https://doi.org/10.3390/v13081599
APA StyleZehetner, V., Cavalleri, J.-M. V., Klang, A., Hofer, M., Preining, I., Steinborn, R., & Ramsauer, A. S. (2021). Equine Parvovirus-Hepatitis Screening in Horses and Donkeys with Histopathologic Liver Abnormalities. Viruses, 13(8), 1599. https://doi.org/10.3390/v13081599






